ASTM F3018-17
(Guide)Standard Guide for Assessment of Hard-on-Hard Articulation Total Hip Replacement and Hip Resurfacing Arthroplasty Devices
Standard Guide for Assessment of Hard-on-Hard Articulation Total Hip Replacement and Hip Resurfacing Arthroplasty Devices
SIGNIFICANCE AND USE
4.1 This document provides guidance for a range of assessments and evaluations to aid in preclinical research and device development of hard-on-hard total hip replacement and hip resurfacing devices used for the repair of musculoskeletal disorders.
4.2 The user is encouraged to use appropriate ASTM International or ISO standards to conduct the physical, chemical, mechanical, biocompatibility, and preclinical tests on alloy fabricated forms, ceramic material samples, device components, or devices before assessment in an in vitro model.
4.3 Studies to support regulatory submissions should conform to appropriate regulatory requirements and guidelines for the development of medical devices.
4.4 Assessments with physical, chemical, mechanical, biocompatibility, and preclinical tests on hard-on-hard hip prosthesis components are not necessarily predictive of human results and therefore should be interpreted cautiously with respect to potential applicability to clinical conditions. Referenced metal-onmetal or ceramic-on-ceramic hip prosthesis publications can be found in the Bibliography section at the end of this guide for further review.
SCOPE
1.1 This guide covers materials and design recommendations and general test methods for the chemical, mechanical, and preclinical assessment of implantable devices with hard-on-hard articulations intended to replace a hip joint. The provided guidance is intended to encompass both Total Hip Replacement (THR) devices with stems that extend or fix within the intramedullary canal as well as Hip Resurfacing Arthroplasty (HRA) wherein only the hip articulating surfaces are replaced. There has been long term clinical experience with metal-on-metal articulating components manufactured from cobalt-28 % chromium-6 % molybdenum (Co28Cr6Mo) alloy (Specifications F75, F799, or F1537) or high purity alumina (ISO 6474-1) and ceramic-on-ceramic articulating components manufactured from high purity alumina (ISO 6474-1) or alumina matrix composite ceramics (ISO 6474-2). There has also been some limited clinical experience with metal (Co28Cr6Mo) on alumina matrix composite ceramic articulating components. This guide has been created based on the current understanding derived from those clinical histories. Device articulating components manufactured from other metallic alloys, ceramics or with coated or elementally modified articulating surfaces could also be evaluated with this guide. However, such materials that do not have a history of clinical use may present different risks.
1.2 This guide applies to the acetabular and femoral articulating components of hard-on-hard hip replacement devices. Acetabular components can be monoblock, or a modular component with a separate acetabular shell and acetabular liner. As stated above, articulating components have been made from Co28Cr6Mo for a metal-on-metal bearing; high purity alumina or alumina matrix composite ceramics for a ceramic-on-ceramic bearing; and Co28Cr6Mo and alumina matrix composite (ISO 6474-2) for a metal-on-ceramic bearing. Modular acetabular shells have to date been made from Ti-6Al-4V or Co28Cr6Mo. The shell is considered part of the acetabular component. Acetabular components may have external coating and/or porous structure intended for uncemented, press-fit or biological fixation; or, for use with bone cement.
1.3 This standard is a summary of available specifications, test methods, practices, and guides from published standards or the scientific literature. Their clinical relevance is unproven. Most of the methods do not have an established precision and bias; therefore, their repeatability and reproducibility has not been established. As the clinical relevance of these methods have not been established, consequently, most do not have performance requirements. This document does not require that all the listed methodologies are always necessary to evaluate these implant systems provided justifi...
General Information
- Status
- Published
- Publication Date
- 31-Aug-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Mar-2020
- Effective Date
- 15-May-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-Oct-2016
- Effective Date
- 15-Mar-2015
- Effective Date
- 15-Mar-2015
- Effective Date
- 15-Mar-2015
- Effective Date
- 01-Sep-2013
- Effective Date
- 01-Dec-2012
- Effective Date
- 01-Nov-2012
- Effective Date
- 15-May-2012
- Effective Date
- 15-Jan-2012
- Effective Date
- 15-Dec-2011
- Effective Date
- 15-Nov-2011
- Effective Date
- 01-Jun-2011
Overview
ASTM F3018-17 is the Standard Guide for Assessment of Hard-on-Hard Articulation Total Hip Replacement and Hip Resurfacing Arthroplasty Devices. Developed by ASTM International, this guide addresses the assessment and evaluation of implantable hip devices employing hard-on-hard bearing surfaces, including both total hip replacements (THR) and hip resurfacing arthroplasty (HRA) devices. Its primary focus is to aid preclinical research and device development for treating musculoskeletal disorders by providing materials and design recommendations, as well as general test methods for chemical, mechanical, and preclinical evaluation of hip prosthesis components.
This standard emphasizes the importance of using established ASTM and ISO methods to ensure comprehensive assessment of devices before clinical use, taking into account the complex interplay of physical, chemical, and biological factors influencing implant safety and performance.
Key Topics
- Materials and Design
- Covers both metal-on-metal and ceramic-on-ceramic articulations, including cobalt-chromium-molybdenum (Co28Cr6Mo) alloys and high-purity alumina ceramics.
- Addresses the assessment of acetabular and femoral components for both monoblock and modular configurations.
- Testing and Evaluation
- Suggests a suite of preclinical tests, including physical, chemical, mechanical, and biocompatibility evaluations.
- Encourages adherence to relevant ASTM and ISO standards for test procedures and reporting (e.g., wear, dimensional measurements, strength testing, and surface characterization).
- Regulatory Compliance and Clinical Relevance
- Recommends alignment with regulatory requirements for device submissions.
- Highlights that in vitro and preclinical assessments may not fully predict clinical outcomes, underlining the need for cautious interpretation of results.
- Analysis of Surface and Wear
- Discusses methods for evaluating surface roughness, sphericity, and wear behavior, including worst-case scenario testing.
- Stresses the significance of evaluating wear particles and changes in component surfaces using appropriate practices.
Applications
- Device Development and Preclinical Research
- Assists manufacturers and researchers in designing and evaluating hip prosthesis devices with hard-on-hard articulation.
- Facilitates the identification and mitigation of potential risks associated with new materials or innovative design features.
- Regulatory Submissions
- Provides guidance for compiling comprehensive test data to support regulatory filings for new hip replacement and resurfacing devices.
- Helps ensure that all required assessments, such as fatigue strength, wear testing, and modular interface evaluation, are performed per international standards.
- Quality Assurance and Comparative Studies
- Enables comparison between candidate devices and established successful predicates, ensuring materials and design choices meet high standards of safety and durability.
- Supports ongoing post-market surveillance efforts by providing a framework for consistent evaluation of new and modified products.
Related Standards
Materials and Components
- ASTM F75, F799, F1537 - Specifications for cobalt-chromium-molybdenum alloys for surgical implants
- ASTM F136 - Specification for wrought titanium-6 aluminum-4 vanadium alloy
- ISO 6474-1, ISO 6474-2 - Specifications for alumina ceramics and alumina matrix composite ceramics
Testing Methods
- ISO 14242 series - Wear testing for total hip-joint prostheses
- ASTM F2033 - Specification for total hip replacement joint prosthesis bearings
- ASTM F2345, ISO 7206-10 - Static and fatigue strength tests for ceramic heads
- ASTM F2582 - Impingement testing
- ISO 25178-6 - Surface texture measurement
Additional Guidance
- FDA Guidance Documents on test requirements for orthopedic implants
- ISO 7206 series - Mechanical testing of hip prosthesis components
- ASTM F1814, F1820 - Modular component evaluation and disassembly force testing
By employing ASTM F3018-17 in device assessment, manufacturers, researchers, and regulators can ensure high standards of safety, durability, and quality for hip replacement and resurfacing arthroplasty devices with hard-on-hard articulations. This guide serves as a key resource for harmonized and effective preclinical evaluation within the orthopedic medical device industry.
Buy Documents
ASTM F3018-17 - Standard Guide for Assessment of Hard-on-Hard Articulation Total Hip Replacement and Hip Resurfacing Arthroplasty Devices
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3018-17 is a guide published by ASTM International. Its full title is "Standard Guide for Assessment of Hard-on-Hard Articulation Total Hip Replacement and Hip Resurfacing Arthroplasty Devices". This standard covers: SIGNIFICANCE AND USE 4.1 This document provides guidance for a range of assessments and evaluations to aid in preclinical research and device development of hard-on-hard total hip replacement and hip resurfacing devices used for the repair of musculoskeletal disorders. 4.2 The user is encouraged to use appropriate ASTM International or ISO standards to conduct the physical, chemical, mechanical, biocompatibility, and preclinical tests on alloy fabricated forms, ceramic material samples, device components, or devices before assessment in an in vitro model. 4.3 Studies to support regulatory submissions should conform to appropriate regulatory requirements and guidelines for the development of medical devices. 4.4 Assessments with physical, chemical, mechanical, biocompatibility, and preclinical tests on hard-on-hard hip prosthesis components are not necessarily predictive of human results and therefore should be interpreted cautiously with respect to potential applicability to clinical conditions. Referenced metal-onmetal or ceramic-on-ceramic hip prosthesis publications can be found in the Bibliography section at the end of this guide for further review. SCOPE 1.1 This guide covers materials and design recommendations and general test methods for the chemical, mechanical, and preclinical assessment of implantable devices with hard-on-hard articulations intended to replace a hip joint. The provided guidance is intended to encompass both Total Hip Replacement (THR) devices with stems that extend or fix within the intramedullary canal as well as Hip Resurfacing Arthroplasty (HRA) wherein only the hip articulating surfaces are replaced. There has been long term clinical experience with metal-on-metal articulating components manufactured from cobalt-28 % chromium-6 % molybdenum (Co28Cr6Mo) alloy (Specifications F75, F799, or F1537) or high purity alumina (ISO 6474-1) and ceramic-on-ceramic articulating components manufactured from high purity alumina (ISO 6474-1) or alumina matrix composite ceramics (ISO 6474-2). There has also been some limited clinical experience with metal (Co28Cr6Mo) on alumina matrix composite ceramic articulating components. This guide has been created based on the current understanding derived from those clinical histories. Device articulating components manufactured from other metallic alloys, ceramics or with coated or elementally modified articulating surfaces could also be evaluated with this guide. However, such materials that do not have a history of clinical use may present different risks. 1.2 This guide applies to the acetabular and femoral articulating components of hard-on-hard hip replacement devices. Acetabular components can be monoblock, or a modular component with a separate acetabular shell and acetabular liner. As stated above, articulating components have been made from Co28Cr6Mo for a metal-on-metal bearing; high purity alumina or alumina matrix composite ceramics for a ceramic-on-ceramic bearing; and Co28Cr6Mo and alumina matrix composite (ISO 6474-2) for a metal-on-ceramic bearing. Modular acetabular shells have to date been made from Ti-6Al-4V or Co28Cr6Mo. The shell is considered part of the acetabular component. Acetabular components may have external coating and/or porous structure intended for uncemented, press-fit or biological fixation; or, for use with bone cement. 1.3 This standard is a summary of available specifications, test methods, practices, and guides from published standards or the scientific literature. Their clinical relevance is unproven. Most of the methods do not have an established precision and bias; therefore, their repeatability and reproducibility has not been established. As the clinical relevance of these methods have not been established, consequently, most do not have performance requirements. This document does not require that all the listed methodologies are always necessary to evaluate these implant systems provided justifi...
SIGNIFICANCE AND USE 4.1 This document provides guidance for a range of assessments and evaluations to aid in preclinical research and device development of hard-on-hard total hip replacement and hip resurfacing devices used for the repair of musculoskeletal disorders. 4.2 The user is encouraged to use appropriate ASTM International or ISO standards to conduct the physical, chemical, mechanical, biocompatibility, and preclinical tests on alloy fabricated forms, ceramic material samples, device components, or devices before assessment in an in vitro model. 4.3 Studies to support regulatory submissions should conform to appropriate regulatory requirements and guidelines for the development of medical devices. 4.4 Assessments with physical, chemical, mechanical, biocompatibility, and preclinical tests on hard-on-hard hip prosthesis components are not necessarily predictive of human results and therefore should be interpreted cautiously with respect to potential applicability to clinical conditions. Referenced metal-onmetal or ceramic-on-ceramic hip prosthesis publications can be found in the Bibliography section at the end of this guide for further review. SCOPE 1.1 This guide covers materials and design recommendations and general test methods for the chemical, mechanical, and preclinical assessment of implantable devices with hard-on-hard articulations intended to replace a hip joint. The provided guidance is intended to encompass both Total Hip Replacement (THR) devices with stems that extend or fix within the intramedullary canal as well as Hip Resurfacing Arthroplasty (HRA) wherein only the hip articulating surfaces are replaced. There has been long term clinical experience with metal-on-metal articulating components manufactured from cobalt-28 % chromium-6 % molybdenum (Co28Cr6Mo) alloy (Specifications F75, F799, or F1537) or high purity alumina (ISO 6474-1) and ceramic-on-ceramic articulating components manufactured from high purity alumina (ISO 6474-1) or alumina matrix composite ceramics (ISO 6474-2). There has also been some limited clinical experience with metal (Co28Cr6Mo) on alumina matrix composite ceramic articulating components. This guide has been created based on the current understanding derived from those clinical histories. Device articulating components manufactured from other metallic alloys, ceramics or with coated or elementally modified articulating surfaces could also be evaluated with this guide. However, such materials that do not have a history of clinical use may present different risks. 1.2 This guide applies to the acetabular and femoral articulating components of hard-on-hard hip replacement devices. Acetabular components can be monoblock, or a modular component with a separate acetabular shell and acetabular liner. As stated above, articulating components have been made from Co28Cr6Mo for a metal-on-metal bearing; high purity alumina or alumina matrix composite ceramics for a ceramic-on-ceramic bearing; and Co28Cr6Mo and alumina matrix composite (ISO 6474-2) for a metal-on-ceramic bearing. Modular acetabular shells have to date been made from Ti-6Al-4V or Co28Cr6Mo. The shell is considered part of the acetabular component. Acetabular components may have external coating and/or porous structure intended for uncemented, press-fit or biological fixation; or, for use with bone cement. 1.3 This standard is a summary of available specifications, test methods, practices, and guides from published standards or the scientific literature. Their clinical relevance is unproven. Most of the methods do not have an established precision and bias; therefore, their repeatability and reproducibility has not been established. As the clinical relevance of these methods have not been established, consequently, most do not have performance requirements. This document does not require that all the listed methodologies are always necessary to evaluate these implant systems provided justifi...
ASTM F3018-17 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3018-17 has the following relationships with other standards: It is inter standard links to ASTM F1537-20, ASTM F799-19, ASTM F561-19, ASTM F1877-16, ASTM F2091-15, ASTM F1854-15, ASTM F2068-15, ASTM F561-13, ASTM F136-12a, ASTM F136-12, ASTM F75-12, ASTM F2091-01(2012), ASTM F136-11, ASTM F1537-11, ASTM F799-11. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3018-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3018 − 17
Standard Guide for
Assessment of Hard-on-Hard Articulation Total Hip
Replacement and Hip Resurfacing Arthroplasty Devices
This standard is issued under the fixed designation F3018; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope acetabular component. Acetabular components may have ex-
ternal coating and/or porous structure intended for
1.1 This guide covers materials and design recommenda-
uncemented, press-fit or biological fixation; or, for use with
tions and general test methods for the chemical, mechanical,
bone cement.
and preclinical assessment of implantable devices with hard-
on-hard articulations intended to replace a hip joint. The 1.3 This standard is a summary of available specifications,
provided guidance is intended to encompass both Total Hip testmethods,practices,andguidesfrompublishedstandardsor
Replacement (THR) devices with stems that extend or fix the scientific literature. Their clinical relevance is unproven.
within the intramedullary canal as well as Hip Resurfacing Most of the methods do not have an established precision and
Arthroplasty (HRA) wherein only the hip articulating surfaces bias; therefore, their repeatability and reproducibility has not
arereplaced.Therehasbeenlongtermclinicalexperiencewith been established. As the clinical relevance of these methods
metal-on-metal articulating components manufactured from have not been established, consequently, most do not have
cobalt-28 % chromium-6 % molybdenum (Co28Cr6Mo) alloy performance requirements. This document does not require
(Specifications F75, F799,or F1537) or high purity alumina that all the listed methodologies are always necessary to
(ISO 6474-1) and ceramic-on-ceramic articulating components evaluate these implant systems provided justification for not
manufactured from high purity alumina (ISO 6474-1) or usingeachunusedmethodisprovided.Thisdocumentdoesnot
alumina matrix composite ceramics (ISO 6474-2). There has intend to prevent the use of new methodologies as they are
also been some limited clinical experience with metal developed.
(Co28Cr6Mo) on alumina matrix composite ceramic articulat-
1.4 This standard does not purport to address all of the
ing components. This guide has been created based on the
safety concerns, if any, associated with its use. It is the
current understanding derived from those clinical histories.
responsibility of the user of this standard to establish appro-
Device articulating components manufactured from other me-
priate safety, health and environmental practices and deter-
tallic alloys, ceramics or with coated or elementally modified
mine the applicability of regulatory limitations prior to use.
articulating surfaces could also be evaluated with this guide.
1.5 This international standard was developed in accor-
However, such materials that do not have a history of clinical
dance with internationally recognized principles on standard-
use may present different risks.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
1.2 This guide applies to the acetabular and femoral articu-
mendations issued by the World Trade Organization Technical
lating components of hard-on-hard hip replacement devices.
Barriers to Trade (TBT) Committee.
Acetabular components can be monoblock, or a modular
component with a separate acetabular shell and acetabular
2. Referenced Documents
liner.Asstatedabove,articulatingcomponentshavebeenmade
from Co28Cr6Mo for a metal-on-metal bearing; high purity
2.1 ASTM Standards:
alumina or alumina matrix composite ceramics for a ceramic-
F75 Specification for Cobalt-28 Chromium-6 Molybdenum
on-ceramic bearing; and Co28Cr6Mo and alumina matrix
Alloy Castings and Casting Alloy for Surgical Implants
composite (ISO 6474-2) for a metal-on-ceramic bearing.
(UNS R30075)
Modular acetabular shells have to date been made from
F136 Specification for Wrought Titanium-6Aluminum-
Ti-6Al-4V or Co28Cr6Mo. The shell is considered part of the
4Vanadium ELI (Extra Low Interstitial)Alloy for Surgical
Implant Applications (UNS R56401)
This test method is under the jurisdiction ofASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee For referenced ASTM standards, visit the ASTM website, www.astm.org, or
F04.22 on Arthroplasty. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved Sept. 1, 2017. Published September 2017. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F3018-17 the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3018 − 17
F561 Practice for Retrieval and Analysis of Medical prostheses -- Part 4: Determination of endurance proper-
Devices, and Associated Tissues and Fluids ties and performance of stemmed femoral components
F799 Specification for Cobalt-28Chromium-6Molybdenum ISO 7206-6 Implants for surgery -- Partial and total hip joint
Alloy Forgings for Surgical Implants (UNS R31537, prostheses -- Part 6: Endurance properties testing and
R31538, R31539) performance requirements of neck region of stemmed
F1537 Specification for Wrought Cobalt-28Chromium- femoral components
6Molybdenum Alloys for Surgical Implants (UNS ISO 7206-10 Implants for surgery -- Partial and total hip
R31537, UNS R31538, and UNS R31539) joint prostheses -- Part 10: Determination of resistance to
F1814 Guide for Evaluating Modular Hip and Knee Joint static load of modular femoral heads
Components ISO 7206-12 Implants for surgery -- Partial and total hip
F1820 Test Method for Determining the Forces for Disas- joint prostheses — Part 12: Deformation test method for
sembly of Modular Acetabular Devices acetabular shells
F1854 Test Method for Stereological Evaluation of Porous ISO 7206-13 Implants for surgery -- Partial and total hip
Coatings on Medical Implants joint prostheses -- Part 13: Determination of resistance to
F1877 Practice for Characterization of Particles torque of head fixation of stemmed femoral components
F2033 Specification for Total Hip Joint Prosthesis and Hip ISO 14242-1 Implants for surgery -- Wear of total hip-joint
Endoprosthesis Bearing Surfaces Made of Metallic, prostheses--Part1:Loadinganddisplacementparameters
Ceramic, and Polymeric Materials forwear-testingmachinesandcorrespondingenvironmen-
F2068 Specification for Femoral Prostheses—Metallic Im- tal conditions for test
plants ISO 14242-2 Implants for surgery -- Wear of total hip-joint
F2091 Specification for Acetabular Prostheses prostheses -- Part 2: Methods of Measurement
F2345 Test Methods for Determination of Static and Cyclic ISO 14242-3 Implants for surgery -- Wear of total hip-joint
Fatigue Strength of Ceramic Modular Femoral Heads prostheses--Part3:Loadinganddisplacementparameters
F2582 Test Method for Impingement ofAcetabular Prosthe- for orbital bearing type wear testing machines and corre-
ses sponding environmental conditions for test
F3047M Guide for High Demand Hip Simulator Wear Test- ISO 17853 Wear of implant materials -- Polymer and metal
ing of Hard-on-hard Articulations wear particles -- Isolation, characterization and quantifi-
cation
2.2 ISO Standards:
ISO21535 Non-activesurgicalimplants--Jointreplacement
ISO 1302 Geometrical Product Specifications (GPS) -- In-
implants -- Specific requirements for hip-joint replace-
dication of surface texture in technical product documen-
ment implants
tation
ISO 25178-6 Geometrical product specifications (GPS) --
ISO 4287 Geometrical Product Specifications (GPS) -- Sur-
Surface texture: areal -- Part 6: Classification of methods
face texture: Profile method -- Terms, definitions and
for measuring surface texture
surface texture parameters
2.3 Other Standards:
ISO 4288 Geometrical Product Specifications (GPS) -- Sur-
ASME Y14.36M Surface Texture Symbols
face texture: Profile method -- Rules and procedures for
US FDA 510(k) Information needed for Hydroxyapatite
the assessment of surface texture
CoatedOrthopedicImplantsMarch10,1995(revisedFeb.
ISO 5832-3 Implants for surgery -- Metallic materials – Part
20, 1997)
3: Wrought titanium 6-aluminium 4vanadium alloy
ISO 5832-4 Implants for surgery -- Metallic materials -- Part
3. Terminology
4: Cobalt-chromium-molybdenum casting alloy
3.1 Definitions of Terms Specific to This Standard:
ISO 5832-12 Implants for surgery -- Metallic materials --
3.1.1 alloy fabricated form, n—the raw material form of the
Part 12: Wrought cobalt-chromiummolybdenum alloy
metallic alloy and any processing techniques used to fabricate
ISO6474-1 Implantsforsurgery--Part1:Ceramicmaterials
the final form of the implant.
based on high purity alumina
ISO 6474-2 Implants for surgery -- Part 2: Composite 3.1.2 breakaway wear, n—a ‘higher’ unexpected wear rate
materials based on a high purity alumina matrix with
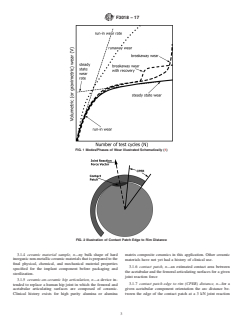
that follows a period of steady-state wear as illustrated in Fig.
zirconia reinforcement 1. (1)
ISO 7206-1 Implants for surgery -- Partial and total hip joint
3.1.3 breakaway wear with recovery, n—breakaway wear
prostheses -- Part 1: Classification and designation of
that returns to lower steady-state wear rates. The breakaway/
dimensions
recovery phenomenon can be a single event or multiple
ISO 7206-2 Implants for surgery -- Partial and total hip joint
‘episodic’ events during the steady-state conditions as illus-
prostheses -- Part 2: Articulating surfaces made of
trated in Fig. 1.
metallic, ceramic and plastics materials
ISO 7206-4 Implants for surgery -- Partial and total hip joint
Submission and Review of Sterility Information in Premarket Notification
(510(k)) Submissions for Devices Labeled as Sterile, issued January 21, 2016,
updated March 16, 2016.
3 5
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St., The boldface numbers in parentheses refer to a list of references at the end of
4th Floor, New York, NY 10036, http://www.ansi.org. this standard.
F3018 − 17
FIG. 1 Modes/Phases of Wear Illustrated Schematically (1)
FIG. 2 Illustration of Contact Patch Edge to Rim Distance
3.1.4 ceramic material sample, n—ny bulk shape of hard matrix composite ceramics in this application. Other ceramic
inorganicnon-metallicceramicmaterialsthatispreparedtothe materials have not yet had a history of clinical use.
final physical, chemical, and mechanical material properties
3.1.6 contact patch, n—an estimated contact area between
specified for the implant component before packaging and
the acetabular and the femoral articulating surfaces for a given
sterilization.
joint reaction force
3.1.5 ceramic-on-ceramic hip articulation, n—a device in-
3.1.7 contact patch edge to rim (CPER) distance, n—for a
tended to replace a human hip joint in which the femoral and
given acetabular component orientation the arc distance be-
acetabular articulating surfaces are composed of ceramic.
tween the edge of the contact patch ata3kN joint reaction
Clinical history exists for high purity alumina or alumina
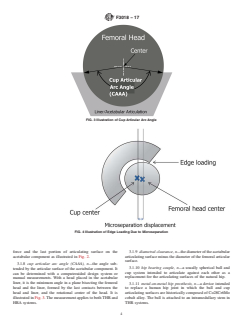
F3018 − 17
FIG. 3 Illustration of Cup Articular Arc Angle
FIG. 4 Illustration of Edge Loading Due to Microseparation
force and the last portion of articulating surface on the 3.1.9 diametral clearance, n—thediameteroftheacetabular
acetabular component as illustrated in Fig. 2. articulating surface minus the diameter of the femoral articular
surface.
3.1.8 cup articular arc angle (CAAA), n—the angle sub-
3.1.10 hip bearing couple, n—a usually spherical ball and
tended by the articular surface of the acetabular component. It
cup system intended to articulate against each other as a
can be determined with a computeraided design system or
replacement for the articulating surfaces of the natural hip.
manual measurements. With a head placed in the acetabular
liner, it is the minimum angle in a plane bisecting the femoral
3.1.11 metal-on-metal hip prosthesis, n—a device intended
head and the liner, formed by the last contacts between the
to replace a human hip joint in which the ball and cup
head and liner, and the rotational center of the head. It is articulating surfaces are historically composed of Co28Cr6Mo
illustrated in Fig. 3.The measurement applies to bothTHR and
cobalt alloy. The ball is attached to an intramedullary stem in
HRA systems. THR systems.
F3018 − 17
3.1.12 microseparation, n—a dynamic condition that can development of hard-on-hard total hip replacement and hip
occur in where the centers of rotation of the femoral head and resurfacing devices used for the repair of musculoskeletal
the acetabular cup are displaced during an activity. This can disorders.
lead to edge loading where the femoral head contacts the rim.
4.2 The user is encouraged to use appropriate ASTM
It is illustrated in Fig. 4. The phenomenon is relevant to both
International or ISO standards to conduct the physical,
THR and HRA systems.
chemical, mechanical, biocompatibility, and preclinical tests
3.1.13 modular acetabular device, n—a modular acetabular on alloy fabricated forms, ceramic material samples, device
system consisting of a minimum of two components, one of components,ordevicesbeforeassessmentinaninvitromodel.
which includes the bearing surface and the second component
4.3 Studies to support regulatory submissions should con-
is a modular acetabular shell intended to contain the bearing
form to appropriate regulatory requirements and guidelines for
liner and contact bone or bone cement.
the development of medical devices.
3.1.14 modular acetabular liner—portion of the modular
4.4 Assessments with physical, chemical, mechanical,
acetabular device with an internal hemispherical socket in-
biocompatibility, and preclinical tests on hard-on-hard hip
tended to articulate with the head of a femoral prosthesis. The
prosthesis components are not necessarily predictive of human
external geometry of this component interfaces with the
results and therefore should be interpreted cautiously with
acetabular shell through a locking mechanism which may be
respect to potential applicability to clinical conditions. Refer-
integral to the design of the liner and shell or may rely upon
enced metal-onmetal or ceramic-on-ceramic hip prosthesis
additionalcomponents(forexample,metalring,screws,andso
publications can be found in the Bibliography section at the
forth).
end of this guide for further review.
3.1.15 modular acetabular shell, n—the external, hollow
structure, usually metallic, that provides additional mechanical 5. Guidance for Device Description
support or reinforcement for an acetabular liner and whose
5.1 Specification F2068 provides appropriate descriptions
external features interface directly with the bones of the pelvic
and performance requirements for the femoral prostheses
socket (for example, through bone cement, intimate press-fit,
portion of the THR system.
coatings for attachment to bone cement or tissue, integral
5.2 Specification F2091 provides appropriate descriptions
screw threads, anchoring screws, pegs, and so forth). The
for the acetabular component of a THR and HRA systems.
acetabular shell may be solid or contain holes for fixation or
Additional features as described below are useful for defining
attachment of instrumentation.
the hard-on-hard articulation of a total hip replacement and hip
3.1.16 monoblock acetabular device, n—an acetabular sys-
resurfacing device.
tem manufactured as a single piece
5.2.1 Monoblock acetabular system
3.1.17 runaway wear, n—an initial high wear rate that
5.2.1.1 The location and size of features such as screw
shows no sign of achieving a lower steady-state wear rate as
holes, specific geometry intended for fixation, or an exterior
illustrated in Fig. 1.
surface coating intended for attachment to bone cement or
tissue, Note: There should be no interference of these features
3.1.18 run-in wear, n—wear rate that occurs when the
with the articulation of the system.
components are first implanted in vivo, or during the initial
5.2.2 Modular acetabular systems
phase of an in vitro hip simulator test. During this period, wear
5.2.2.1 Features intended to hold the modular system to-
rates are typically higher than during steady state as the
gether in clinical use.
articulating surfaces wear into conformity with each other and
5.2.2.2 Screw holes, specific geometry intended for fixation
any initially contacting surface asperities or form errors are
of the shell, or an exterior surface structure intended for
worn away as illustrated in Fig. 1. In hip simulator wear tests
attachment to bone cement or tissue. Note: there should be no
it is often considered to be about one million cycles. The
interferenceofthesefeatureswiththeattachmentoftheshellto
transition to steady-state wear can be estimated graphically
the liner or the articulation with the head.
from the plot of total wear vs. number of cycles.
5.2.2.3 Dimensional requirements of the modular interface
3.1.19 steady-state wear, n—an average wear rate that
of both the acetabular liner and the acetabular shell shall be
occurs after a transient run-in wear period as illustrated in Fig.
reported.
1. Typically, the steady-state wear rate is less than the run-in
5.2.2.4 The surface finish requirements of the contacting
wear rate. In hip simulator wear tests it is generally considered
modularsurfacesofboththeacetabularlinerandtheacetabular
to be estimated from values at 1 million cycles and above.
shell shall be reported before and after wear testing.
3.1.20 ZTA, n—Zirconia Toughened Alumina A ceramic
5.3 Materials
with an Alumina matrix than has Zirconia added to toughen
5.3.1 Hardonhardarticulationcomponentshavebeenmade
and strengthen the resultant composite ceramic.
frommaterialconformingtotherequirementsofSpecifications
F75, F799, F1537, ISO 5832-4, ISO 5832-12, ISO 6474-1, or
4. Significance and Use
ISO 6474-2. These specifications include, but are not limited
4.1 This document provides guidance for a range of assess- to, chemical, mechanical, inspection (including
ments and evaluations to aid in preclinical research and device microstructural), and supplier quality system requirements.
F3018 − 17
5.3.2 Modular acetabular shells have been manufactured 6. Device Evaluation
from one of the following materials: Specifications F75, F136,
6.1 Hip Simulator Testing for Hard-on-hard Systems:
F799, F1537, ISO 5832-3, ISO 5832-4, and ISO 5832-12.Any
6.1.1 Standard Wear Tests—Test specimen selection and
surfacemodificationorcoatingontheexterioroftheacetabular
justification.
component intended for contact with bone or bone cement
6.1.1.1 The test procedures shall be as described in ISO
should be tested for its intended purpose.
14242-1, ISO 14242-2, and ISO 14242-3 with the following
5.3.2.1 Test Method F1854 provides information on porous
specific provisions:
coating characterization (coating thickness, void content, and
6.1.1.2 The test shall include a minimum of 5 million cycles
mean intercept length).
of standard walking gait cycle as per ISO 14242-1 or ISO
5.3.2.2 Other important guidance includes the FDA Guid-
14242-3.
ance Documents for Testing Orthopedic Implants with Modi-
6.1.2 Test Specimen Selection and Justification:
fied Metallic Surfaces Opposing Bone or Bone Cement (2) and
6.1.2.1 Depending on the system design, the worst-case
Guidance for Industry on the Testing of Metallic Plasma
construct for ‘adverse conditions’ testing could possibly be
Sprayed Coatings on Orthopedic Implants to Support Recon-
different from the worstcase construct for ‘standard-pristine’
sideration of Postmarket Surveillance Requirements (3).
wear testing. It is recommended that the worst-case construct
5.3.2.3 The FDA Guidance Documents for “510(k) Infor-
for adverse conditions testing be investigated experimentally
mation needed for Hydroxyapatite Coated Orthopedic Im-
with shorter tests with fewer high demand conditions before
plants” provides guidance for characterizing hydroxyapatite
undertaking any five million cycle test. The Finite Element
coatings.
Method (FEM) may also be useful for some of these prelimi-
nary evaluations. The specific bearings used in the simulator
5.4 Dimensional and Physical Specifications
test should include the potentially worst-case (highest wear)
5.4.1 The diametral clearance requirements are described in
combinations taking into consideration the following design
Specification F2033.
parameters:
5.4.2 Articulating Surface Roughness
6.1.2.2 The diameter of the articulating surface; the mini-
5.4.2.1 Surface roughness of all bearing surfaces should be
mum thickness of the acetabular component; and in the case of
specified on drawings using suitable indications, such as
modular acetabular systems, the minimum thicknesses of the
ASME Y14.36M or ISO 1302.
liner and the shell.
5.4.2.2 Surface roughness of bearing surfaces should be
6.1.2.3 Small diametral clearance (highest contact area) and
measuredaccordingtotheproceduresdescribedina)ISO4287
largest diametral clearance (smallest contact area, potentially
and ISO 4288, or b) ISO 25178-6 for three-dimensional areal
highest contact stress) allowed by articulating surface manu-
measurements.
facturing tolerances are potential worst cases. Since the toler-
5.4.2.3 Specification F2033 and ISO 7206-2 describe sur-
ances are usually normal distributions, producing test samples
face finish requirements.
to the exact worst-case tolerances would be extremely difficult.
5.4.3 Articulating Surface Form Deviation (Sphericity)
Sufficient numbers of components shall be measured prior to
5.4.3.1 The surface deviation from the nominal print form
testingandpairedtoachievetheworstcasediametralclearance
of the bearing surfaces should be specified and measured. In
possible within the samples available.
the special case of an intentionally spherical bearing surface,
6.1.2.4 Some of these tests may require evaluation of all
this measurement is termed sphericity.
sizes. Other tests may require the justification of one or more
5.4.3.2 Specification F2033 and ISO 7206-2 describe sphe-
possible “worst case” sizes/combinations.
ricity form deviation requirements.
6.1.2.5 Thechoiceofsamplesforeachofthesetestsshallbe
5.4.4 Monoblock acetabular bearing minimum thickness,
justified.
variation of thickness by size, and approximate location shall
6.1.3 Adverse/high Demand Hip Simulator Testing for
be according to Specification F2091.
Hard-on-hard Systems:
5.4.5 Minimum thickness of the modular acetabular bearing
6.1.3.1 The Adverse/high demand wear testing should fol-
component, variation of minimum thickness by size and the
lowtherequirementsofGuideF3047M.Previousadverse/high
approximatelocationshallbeaccordingtoSpecificationF2091
demandtestsforhardonhardTHRarticulationshaveincluded:
or ISO 7206-1.
(1) A steep cup angle test, with cup inclinations up to 65°
5.4.6 Describe the cup articular arc angle (CAAA) of all
to horizontal (5, 6, 7).
liners according to .
(2) Microseparation tests (8, 9, 10, 11, 12, 13, 14).
5.4.7 One basis for comparison, the “worst-case” minimum (3) A combined steep cup angle plus microseparation (15,
contact patch edge to rim (CPER) distance shall be estimated 16, 10, 17).
for all articulating couples at an acetabular system inclination (4) High demand gait cycles such as ‘fast jogging’ with
of 65º, anteversion of 35º, and a vertical reaction force of 3kN. higher peak loads and faster test frequencies (18).
Selection of “worst-case” couples should take into consider- (5) Third body abrasive wear with bone cement, ceramic,
ation those couples with the smallest articular diameter, cup or titanium particles (19, 20, 21).
articulating arc angle (CAAA), and diametral clearance. The (6) Stop-Dwell-Start tests with dwell times and stop-dwell-
CPER distance shall also be estimated in any steep cup angle start cyclic rates representative of typical patient activities (22,
tests such as those described in 6.1.3.1.1 (4). 23).
F3018 − 17
6.1.4 Wear measurement shall be performed by one of the shell strength and fretting potential, acetabular shell size
following three methods: a) gravimetrically according to ISO should be evaluated.Aworst case for this test may or may not
14242-2 or b) dimensionally by measurement of surface be the same as the worst case required for wear testing. The
profiles or relative displacements (24), or c) optionally for Finite Element Method may be useful to help determine and
metal-on-metal bearings by measurement of the concentration justify “worst case”. Guide F1814 may provide guidance for
of cobalt, chromium and molybdenum (25, 26), in the test defining a test method and test parameters.
serum. If method (b) or (c) is used, an adequate correlation 6.2.3 Friction Torque Testing:
shall be demonstrated with method (a), gravimetric measure- 6.2.3.1 Published literature provide guidance on friction
ment. A correlation established for previous metalon-metal or torque testing of the bearing system (27, 28, 29). Friction
ceramic-on-ceramic testing is acceptable. In addition, if meth- torque testing should also include assessment of the starting
ods(b)and(c)areused,thengravimetricmeasurementsshould friction (stiction).
still be performed at the beginning (0 cycles) and end (five 6.2.3.2 The specific bearings used in the friction test should
million cycles) of the test to support the alternative wear include the potentially worst-case (highest friction) combina-
measurements. tions taking into consideration the design parameters listed in
6.1.5 Scanning electron microscopy (SEM) of the articular the hip simulator testing section above.
6.2.4 Disassociation of Modular Acetabular Devices
surfaces pre- and post-test may be useful to help describe
changes resulting from the tests. 6.2.4.1 The static disassociation of modular devices shall be
evaluated as described Test Method F1820.
6.1.6 Measurement of pre- and post-test dimensions and
(1) Liner push-out.
surface finishes are useful to help describe changes resulting
(2) Liner torque-out.
from the test.
(3) Liner lever-out.
6.1.7 Practices F561, F1877, and ISO 17853 provide gu
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...