ASTM E2160-23
(Test Method)Standard Test Method for Heat of Reaction of Thermally Reactive Materials by Differential Scanning Calorimetry
Standard Test Method for Heat of Reaction of Thermally Reactive Materials by Differential Scanning Calorimetry
SIGNIFICANCE AND USE
5.1 This test method is useful in determining the extrapolated onset temperature, the peak heat flow temperature and the heat of reaction of a material. Any onset temperature determined by this test method is not valid for use as the sole information used for determination of storage or processing conditions.
5.2 This test method is useful in determining the fraction of a reaction that has been completed in a sample prior to testing. This fraction of reaction that has been completed can be a measure of the degree of cure of a thermally reactive polymer or can be a measure of decomposition of a thermally reactive material upon aging.
5.3 The heat of reaction values may be used in Practice E1231 to determine hazard potential figures-of-merit Explosion Potential and Shock Sensitivity.
5.4 This test method may be used in research, process control, quality assurance, and specification acceptance.
SCOPE
1.1 This test method determines the exothermic heat of reaction of thermally reactive chemicals or chemical mixtures, using milligram specimen sizes, by differential scanning calorimetry. Such reactive materials may include thermally unstable or thermoset materials.
1.2 This test method also determines the extrapolated onset temperature and peak heat flow temperature for the exothermic reaction.
1.3 This test method may be performed on solids, liquids or slurries.
1.4 The applicable temperature range of this test method is 25 °C to 600 °C.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard is related to Test Method E537, but provides additional information.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Aug-2023
- Effective Date
- 01-May-2023
- Effective Date
- 01-Nov-2023
- Referred By
ASTM D5028-17 - Standard Test Method for Curing Properties of Pultrusion Resins by Thermal Analysis - Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
Overview
ASTM E2160-23: Standard Test Method for Heat of Reaction of Thermally Reactive Materials by Differential Scanning Calorimetry outlines a reliable testing protocol for measuring the exothermic heat of reaction in thermally reactive chemicals and mixtures. Using differential scanning calorimetry (DSC), this method is applicable to solids, liquids, and slurries, and covers specimen heating from 25°C to 600°C. The standard is widely implemented in research, process control, quality assurance, and specification acceptance, serving as a fundamental tool in thermal analysis of polymers and thermoset materials.
Key Topics
- Heat of Reaction Measurement: Utilizes DSC to accurately determine the total exothermic heat released by small samples (milligram scale) during reaction.
- Onset and Peak Temperatures: Establishes the extrapolated onset temperature (start of exotherm) and peak heat flow temperature (maximum rate).
- Degree of Cure/Fraction Reacted: Assesses the completion of reaction, useful for evaluating the curing status of polymers or assessing material decomposition upon aging.
- Hazard Assessment: The values generated can be used alongside ASTM E1231 to evaluate explosion potential and shock sensitivity, aiding in safety and hazard assessment.
- Sample Versatility: Applicable to thermally unstable materials, thermosets, and a variety of chemical sample types, enhancing its use in diverse industries.
- Measurement Precision: Includes guidance for calibration, appropriate container selection, and sample handling to maximize reproducibility and accuracy.
- Safety Practices: Stresses the importance of safety procedures due to the potential hazards of reactive materials, especially during sample preparation and heating.
Applications
ASTM E2160-23 is widely used across multiple sectors where understanding the thermal behavior of materials is critical:
- Research and Development: Assesses thermal reactivity in new chemical compounds, resins, and polymers.
- Process Control: Monitors cure progress of thermoset polymers to ensure quality during manufacturing.
- Quality Assurance: Validates batch consistency for reactive materials by verifying energy release and reaction characteristics.
- Hazard Analysis: Supports risk assessments by generating data on explosive or shock-sensitive substances.
- Material Aging Studies: Tracks changes in reactivity and decomposition products for materials subjected to thermal stress or aging.
Commonly, industries such as chemical manufacturing, aerospace, automotive, materials science, and safety engineering rely on this standard to ensure materials meet required thermal and safety specifications.
Related Standards
ASTM E2160-23 interfaces with several other standards and practices to provide a comprehensive approach to thermal analysis:
- ASTM E537 - Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry
- ASTM E473 - Terminology Relating to Thermal Analysis and Rheology
- ASTM E967 - Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
- ASTM E1142 - Terminology Relating to Thermophysical Properties
- ASTM E1231 - Practice for Calculation of Hazard Potential Figures of Merit for Thermally Unstable Materials
- ASTM E1860 - Test Method for Elapsed Time Calibration of Thermal Analyzers
- ASTM E3142 - Test Method for Thermal Lag of Thermal Analysis Apparatus
- NAS 1613 - Seal Element, Packing, Preformed, Ethylene Propylene Rubber (relevant for container evaluations)
Adherence to ASTM E2160-23 ensures the collection of consistent, accurate, and internationally recognized data on the heat of reaction and thermal properties of reactive materials, supporting safety, quality, and innovation across industries.
Buy Documents
ASTM E2160-23 - Standard Test Method for Heat of Reaction of Thermally Reactive Materials by Differential Scanning Calorimetry
REDLINE ASTM E2160-23 - Standard Test Method for Heat of Reaction of Thermally Reactive Materials by Differential Scanning Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E2160-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Heat of Reaction of Thermally Reactive Materials by Differential Scanning Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is useful in determining the extrapolated onset temperature, the peak heat flow temperature and the heat of reaction of a material. Any onset temperature determined by this test method is not valid for use as the sole information used for determination of storage or processing conditions. 5.2 This test method is useful in determining the fraction of a reaction that has been completed in a sample prior to testing. This fraction of reaction that has been completed can be a measure of the degree of cure of a thermally reactive polymer or can be a measure of decomposition of a thermally reactive material upon aging. 5.3 The heat of reaction values may be used in Practice E1231 to determine hazard potential figures-of-merit Explosion Potential and Shock Sensitivity. 5.4 This test method may be used in research, process control, quality assurance, and specification acceptance. SCOPE 1.1 This test method determines the exothermic heat of reaction of thermally reactive chemicals or chemical mixtures, using milligram specimen sizes, by differential scanning calorimetry. Such reactive materials may include thermally unstable or thermoset materials. 1.2 This test method also determines the extrapolated onset temperature and peak heat flow temperature for the exothermic reaction. 1.3 This test method may be performed on solids, liquids or slurries. 1.4 The applicable temperature range of this test method is 25 °C to 600 °C. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard is related to Test Method E537, but provides additional information. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is useful in determining the extrapolated onset temperature, the peak heat flow temperature and the heat of reaction of a material. Any onset temperature determined by this test method is not valid for use as the sole information used for determination of storage or processing conditions. 5.2 This test method is useful in determining the fraction of a reaction that has been completed in a sample prior to testing. This fraction of reaction that has been completed can be a measure of the degree of cure of a thermally reactive polymer or can be a measure of decomposition of a thermally reactive material upon aging. 5.3 The heat of reaction values may be used in Practice E1231 to determine hazard potential figures-of-merit Explosion Potential and Shock Sensitivity. 5.4 This test method may be used in research, process control, quality assurance, and specification acceptance. SCOPE 1.1 This test method determines the exothermic heat of reaction of thermally reactive chemicals or chemical mixtures, using milligram specimen sizes, by differential scanning calorimetry. Such reactive materials may include thermally unstable or thermoset materials. 1.2 This test method also determines the extrapolated onset temperature and peak heat flow temperature for the exothermic reaction. 1.3 This test method may be performed on solids, liquids or slurries. 1.4 The applicable temperature range of this test method is 25 °C to 600 °C. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard is related to Test Method E537, but provides additional information. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2160-23 is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2160-23 has the following relationships with other standards: It is inter standard links to ASTM E2160-04(2018), ASTM E1142-23b, ASTM E473-23b, ASTM E473-23a, ASTM E1142-23a, ASTM D8505/D8505M-23, ASTM D5028-17, ASTM D7957/D7957M-22. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2160-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2160 − 23
Standard Test Method for
Heat of Reaction of Thermally Reactive Materials by
Differential Scanning Calorimetry
This standard is issued under the fixed designation E2160; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
1.1 This test method determines the exothermic heat of 2.1 ASTM Standards:
reaction of thermally reactive chemicals or chemical mixtures, E473 Terminology Relating to Thermal Analysis and Rhe-
using milligram specimen sizes, by differential scanning calo- ology
rimetry. Such reactive materials may include thermally un- E537 Test Method for Thermal Stability of Chemicals by
stable or thermoset materials. Differential Scanning Calorimetry
E967 Test Method for Temperature Calibration of Differen-
1.2 This test method also determines the extrapolated onset
tial Scanning Calorimeters and Differential Thermal Ana-
temperature and peak heat flow temperature for the exothermic
lyzers
reaction.
E968 Practice for Heat Flow Calibration of Differential
1.3 This test method may be performed on solids, liquids or
Scanning Calorimeters (Withdrawn 2023)
slurries.
E1142 Terminology Relating to Thermophysical Properties
1.4 The applicable temperature range of this test method is E1231 Practice for Calculation of Hazard Potential Figures
of Merit for Thermally Unstable Materials
25 °C to 600 °C.
E1860 Test Method for Elapsed Time Calibration of Ther-
1.5 The values stated in SI units are to be regarded as
mal Analyzers
standard. No other units of measurement are included in this
E3142 Test Method for Thermal Lag of Thermal Analysis
standard.
Apparatus
1.6 This standard is related to Test Method E537, but
3. Terminology
provides additional information.
1.7 This standard does not purport to address all of the 3.1 Specific technical terms used in this standard are defined
safety concerns, if any, associated with its use. It is the in Terminologies E473 and E1142.
responsibility of the user of this standard to establish appro-
4. Summary of Test Method
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. 4.1 A small (milligram) quantity of the reactive material is
1.8 This international standard was developed in accor-
heated at 10 °C ⁄min through a temperature region where a
dance with internationally recognized principles on standard- chemical reaction takes place. The exothermic heat flow
ization established in the Decision on Principles for the
produced by the reaction is recorded as a function of tempera-
Development of International Standards, Guides and Recom- ture and time by a differential scanning calorimeter. Integration
mendations issued by the World Trade Organization Technical
of the exothermic heat flow over time yields the heat of
Barriers to Trade (TBT) Committee. reaction. If the heat flow is endothermic, then this test method
is not to be used.
1 2
This test method is under the jurisdiction of ASTM Committee E37 on Thermal For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Measurements and is the direct responsibility of Subcommittee E37.01 on Calo- contact ASTM Customer service at service@astm.org. For Annual Book of ASTM
rimetry and Mass Loss. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Nov. 1, 2023. Published November 2023. Originally the ASTM website.
approved in 2001. Last previous edition approved in 2018 as E2160 – 04 (2018). The last approved version of this historical standard is referenced on
DOI: 10.1520/E2160-23. www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2160 − 23
4.2 The test method can be used to determine the fraction of selected temperature limits (ambient temperature to 600 °C) at
a reaction that has occurred in a partially reacted sample. The a heating rate between 2 °C ⁄min and 20 °C ⁄min constant to
heat of reaction is determined for a specimen that is known to within 60.1 °C ⁄min.
be 100 % unreacted and is compared to the heat of reaction 6.1.1.6 A Data Collection Device, to provide a means of
determined for the partially reacted sample. Appropriate cal- acquiring, storing, and displaying measured or calculated
culation yields the fraction of the latter sample that was signals, or both. The minimum output signals required are heat
unreacted. flow, temperature, and time.
4.3 Subtracting the reaction fraction remaining from unity 6.2 Containers, (pans, crucibles, vials, etc. and lids) that are
inert to the specimen and reference materials and that are of
(1) yields the fraction reacted. The fraction reacted may be
expressed as percent. If the sample tested is a thermoset resin, suitable structural shape and integrity to contain the specimen
and reference in accordance with the specific requirements of
the percent reacted is often called the percent of cure.
this test method.
4.4 The extrapolated onset temperature and peak heat flow
6.3 Balance, with a capacity of 100 mg or greater to weigh
temperature are determined for the exothermic heat flow
specimens and containers, or both, readable to 61 μg.
thermal curve from 4.1.
7. Safety Precautions
5. Significance and Use
7.1 The use of this test method for materials of unknown
5.1 This test method is useful in determining the extrapo-
potential hazards requires that precautions be taken during the
lated onset temperature, the peak heat flow temperature and the
sample preparation and testing.
heat of reaction of a material. Any onset temperature deter-
7.2 Where particle size reduction by grinding is necessary,
mined by this test method is not valid for use as the sole
the user of this test method shall presume that the material is
information used for determination of storage or processing
hazardous.
conditions.
7.3 Toxic or corrosive effluents, or both, may be released
5.2 This test method is useful in determining the fraction of
when heating the test specimen and could be harmful to
a reaction that has been completed in a sample prior to testing.
personnel or the apparatus. Use of an exhaust system to remove
This fraction of reaction that has been completed can be a
such effluents is recommended.
measure of the degree of cure of a thermally reactive polymer
or can be a measure of decomposition of a thermally reactive
8. Calibration
material upon aging.
8.1 Perform any calibration procedures recommended by
5.3 The heat of reaction values may be used in Practice
the apparatus manufacturer as described in the Operations
E1231 to determine hazard potential figures-of-merit Explo-
Manual.
sion Potential and Shock Sensitivity.
8.2 Calibrate the temperature signal to within 62 °C using
5.4 This test method may be used in research, process
Test Method E967.
control, quality assurance, and specification acceptance.
8.3 Calibrate the heat flow signal to within 60.5 % using
Practice E968.
6. Apparatus
8.4 Calibrate the elapsed time signal, or ascertain its
6.1 Differential Scanning Calorimeter (DSC), capable of
accuracy, to within 60.5 % using Test Method E1860.
measuring and recording heat flow as a function of temperature
NOTE 2—Calibration or calibration verification of all signals is recom-
and time. Such a DSC is composed of:
mended at least annually.
6.1.1 Test Chamber, composed of:
6.1.1.1 Furnace(s), to provide uniform controlled heating of 9. Procedure
a specimen and reference to a constant temperature or at a
9.1 Into a tared sample container, weigh to within 61 μg,
constant rate within the temperature range of 25 °C to 600 °C.
1 mg to 2 mg of the test specimen. Record this mass as M in
6.1.1.2 Temperature Sensor, to provide an indication of the
mg. Close the sample. Weigh the sealed container to within
specimen or furnace temperature readable to within 60.5 °C.
61 μg and recorded this mass as N in mg.
6.1.1.3 Differential Sensor, to detect a heat flow difference
NOTE 3—Because of the reactive nature of the materials examined by
between the specimen and reference equivalent to 0.2 mW.
this test method, small specimen sizes shall be used unless the approxi-
6.1.1.4 Means of Sustaining a Test Chamber Environment,
mate reactivity of the test specimen is known. Other specimen sizes may
of inert (for example, nitrogen, helium or argon) or reactive
be used but shall be reported. Make sure that the specimen is homogenous
and represents the sample.
(for example, air) gas at a purge rate of 50 mL ⁄min 6
NOTE 4—Some substances may have non-reactive components mixed
5 mL ⁄min.
with the thermally reactive material. An example would be reinforcing
fibers mixed with a thermally-curing polymer. A specification of the
NOTE 1—Typically, at least 99 % pure nitrogen, helium or argon is
fraction of inert material in the mixture may accompany these materials.
employed when oxidation in air is a concern. Unless effects of moisture
The user should be aware that such specifications involve tolerances so
are to be studied, use of dry purge gas is recommended.
that the actual fraction of inert material may vary within these tolerances
6.1.1.5 Temperature Controller, capable of executing a spe-
from lot to lot. In such cases, the actual fraction of inert material must be
cific temperature program by operating the furnace(s) between taken into account.
E2160 − 23
NOTE 5—For highly reactive materials, the selection of sample con- baseline gives the heat of the reaction. Instrument software is most often
tainers can be particularly important. The material from which the used to integrate this area. Although such software packages display
container is constructed may catalyze the reaction or react with the sample thermal curves as in Fig. 1, they calculate the bound area on a basis of
material. Sealed containers may cause an autocatalytic effect or possibly time. If older instruments without these software packages are used, or if
a pressure effect. In open containers loss of material, and thereby loss of manual checks are performed on newer instruments, then the manual
heat, could be an issue. Excessive pressurization of a sample container can integration must be performed with the abscissa presented as a time
be avoided by using vented containers; however, vented or unsealed (seconds) coordinate.
containers may cause the measured heat of reaction to be much smaller NOTE 9—The amount of material should be chosen such that the
than the true value. See 12.4 for an example of such an effect.
maximum heat flow is less than 8 mW. This requirement reduces the
potential of obtaining adiabatic heating of the sample. Adiabatic heating of
9.2 Heat the test specimen at a controlled rate of 10 °C ⁄min
the sample results in “leaning” peaks, an example of which is shown in
6 0.1 °C ⁄min from ambient until the thermal curve returns to 4
Fig. 2 (adapted from Figure 11 of Jones (1996)). For highly energetic
baseline following the exothermic event. If the upper limit of
materials, it might be impossible to satisfy simultaneously the direction of
9.1 (using 1 mg to 2 mg of the test specimen) and the condition of this note
temperature for this test method, 600 °C, is reached before the
(maximum heat flow less
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2160 − 04 (Reapproved 2018) E2160 − 23
Standard Test Method for
Heat of Reaction of Thermally Reactive Materials by
Differential Scanning Calorimetry
This standard is issued under the fixed designation E2160; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method determines the exothermic heat of reaction of thermally reactive chemicals or chemical mixtures, using
milligram specimen sizes, by differential scanning calorimetry. Such reactive materials may include thermally unstable or
thermoset materials.
1.2 This test method also determines the extrapolated onset temperature and peak heat flow temperature for the exothermic
reaction.
1.3 This test method may be performed on solids, liquids or slurries.
1.4 The applicable temperature range of this test method is 25 to 600°C.25 °C to 600 °C.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 There is no ISO method equivalent to this standard.
1.6 This standard is related to Test Method E537 and to NAS 1613, , but provides additional information.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E473 Terminology Relating to Thermal Analysis and Rheology
E537 Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.01 on Calorimetry
and Mass Loss.
Current edition approved April 1, 2018Nov. 1, 2023. Published May 2018November 2023. Originally approved in 2001. Last previous edition approved in 20122018 as
E2160 – 04 (2012).(2018). DOI: 10.1520/E2160-04R18.10.1520/E2160-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2160 − 23
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E968 Practice for Heat Flow Calibration of Differential Scanning Calorimeters (Withdrawn 2023)
E1142 Terminology Relating to Thermophysical Properties
E1231 Practice for Calculation of Hazard Potential Figures of Merit for Thermally Unstable Materials
E1860 Test Method for Elapsed Time Calibration of Thermal Analyzers
E3142 Test Method for Thermal Lag of Thermal Analysis Apparatus
2.2 Other Standard:
NAS 1613 Seal Element, Packing, Preformed, Ethylene Propylene Rubber
3. Terminology
3.1 Specific technical terms used in this standard are defined in Terminologies E473 and E1142.
4. Summary of Test Method
4.1 A small (milligram) quantity of the reactive material is heated at 10°C/min10 °C ⁄min through a temperature region where a
chemical reaction takes place. The exothermic heat flow produced by the reaction is recorded as a function of temperature and time
by a differential scanning calorimeter. Integration of the exothermic heat flow over time yields the heat of reaction. If the heat flow
is endothermic, then this test method is not to be used.
4.2 The test method can be used to determine the fraction of a reaction that has occurred in a partially reacted sample. The heat
of reaction is determined for a specimen that is known to be 100 % unreacted and is compared to the heat of reaction determined
for the partially reacted sample. Appropriate calculation yields the fraction of the latter sample that was unreacted.
4.3 Subtracting the reaction fraction remaining from unity (1) yields the fraction reacted. The fraction reacted may be expressed
as percent. If the sample tested is a thermoset resin, the percent reacted is often called the percent of cure.
4.4 The extrapolated onset temperature and peak heat flow temperature are determined for the exothermic heat flow thermal curve
from 4.1.
5. Significance and Use
5.1 This test method is useful in determining the extrapolated onset temperature, the peak heat flow temperature and the heat of
reaction of a material. Any onset temperature determined by this test method is not valid for use as the sole information used for
determination of storage or processing conditions.
5.2 This test method is useful in determining the fraction of a reaction that has been completed in a sample prior to testing. This
fraction of reaction that has been completed can be a measure of the degree of cure of a thermally reactive polymer or can be a
measure of decomposition of a thermally reactive material upon aging.
5.3 The heat of reaction values may be used in Practice E1231 to determine hazard potential figures-of-merit Explosion Potential
and Shock Sensitivity.
5.4 This test method may be used in research, process control, quality assurance, and specification acceptance.
6. Apparatus
6.1 Differential Scanning Calorimeter (DSC),capable of measuring and recording heat flow as a function of temperature and time.
Such a DSC is composed of:
6.1.1 Test Chamber, composed of:
6.1.1.1 Furnace(s), to provide uniform controlled heating of a specimen and reference to a constant temperature or at a constant
rate within the temperature range of 25 to 600°C.25 °C to 600 °C.
The last approved version of this historical standard is referenced on www.astm.org.
E2160 − 23
6.1.1.2 Temperature Sensor, to provide an indication of the specimen or furnace temperature readable to within 60.5°C.60.5 °C.
6.1.1.3 Differential Sensor, to detect a heat flow difference between the specimen and reference equivalent to 0.2 mW.
6.1.1.4 Means of Sustaining a Test Chamber Environment, of inert (for example, nitrogen, helium or argon) or reactive (for
example, air) gas at a purge rate of 5050 mL ⁄min 6 5 5 mL mL/min.⁄min.
NOTE 1—Typically, at least 99 % pure nitrogen, helium or argon is employed when oxidation in air is a concern. Unless effects of moisture are to be
studied, use of dry purge gas is recommended.
6.1.1.5 Temperature Controller, capable of executing a specific temperature program by operating the furnace(s) between selected
temperature limits (ambient temperature to 600°C)600 °C) at a heating rate between 22 °C ⁄min and 20°C/min20 °C ⁄min constant
to within 60.1°C/min.60.1 °C ⁄min.
6.1.1.6 Recording A Data Collection Device, capable of recording and displaying any portion (including signal noise) of the
differential heat flow on the ordinate as a function of temperature or time on the abscissa.to provide a means of acquiring, storing,
and displaying measured or calculated signals, or both. The minimum output signals required are heat flow, temperature, and time.
6.2 Containers, (pans, crucibles, vials, etc. and lids) that are inert to the specimen and reference materials and that are of suitable
structural shape and integrity to contain the specimen and reference in accordance with the specific requirements of this test
method.
6.3 Balance, with a capacity of 100 mg or greater to weigh specimens and containers, or both, to a sensitivity of 61 μg.readable
to 61 μg.
7. Safety Precautions
7.1 The use of this test method for materials of unknown potential hazards requires that precautions be taken during the sample
preparation and testing.
7.2 Where particle size reduction by grinding is necessary, the user of this test method shall presume that the material is hazardous.
7.3 Toxic or corrosive effluents, or both, may be released when heating the test specimen and could be harmful to personnel or
the apparatus. Use of an exhaust system to remove such effluents is recommended.
8. Calibration
8.1 Perform any calibration procedures recommended by the apparatus manufacturer as described in the Operations Manual.
8.2 Calibrate the temperature signal to within 62°C62 °C using Test Method E967.
8.3 Calibrate the heat flow signal to within 60.5 % using Practice E968.
8.4 Calibrate the elapsed time signal, or ascertain its accuracy, to within 60.5 % using Test Method E1860.
NOTE 2—Calibration or calibration verification of all signals is recommended at least annually.
9. Procedure
9.1 Into a tared sample container, weigh to within 61μg, 161 μg, 1 mg to 2 mg of the test specimen. Record this mass as M in
mg. Close the sample. Weigh the sealed container to within 61 μg 61 μg and recorded this mass as N in mg.
NOTE 3—Because of the reactive nature of the materials examined by this test method, small specimen sizes shall be used unless the approximate reactivity
of the test specimen is known. Other specimen sizes may be used but shall be reported. Make sure that the specimen is homogenous and represents the
sample.
E2160 − 23
NOTE 4—Some substances may have non-reactive components mixed with the thermally reactive material. An example would be reinforcing fibers mixed
with a thermally-curing polymer. A specification of the fraction of inert material in the mixture may accompany these materials. The user should be aware
that such specifications involve tolerances so that the actual fraction of inert material may vary within these tolerances from lot to lot. In such cases, the
actual fraction of inert material must be taken into account.
NOTE 5—For highly reactive materials, the selection of sample containers can be particularly important. The material from which the container is
constructed may catalyze the reaction or react with the sample material. Sealed containers may cause an autocatalytic effect or possibly a pressure effect.
In open containers loss of material, and thereby loss of heat, could be an issue. Excessive pressurization of a sample container can be avoided by using
vented containers,containers; however, vented or unsealed containers may cause the measured heat of reaction to be much smaller than the true value.
See 12.4 for an example of such an effect.
9.2 Heat the test specimen at a controlled rate of 1010 °C ⁄min 6 0.1°C/min0.1 °C ⁄min from ambient until the thermal curve
returns to baseline following the exothermic event. If the upper limit of temperature for this test method, 600°C,600 °C, is reached
before the thermal curve returns to baseline, then this test method is not applicable.
NOTE 6—Other heating rates may be used but shall be reported.reported (see Appendix X1).
9.3 Cool the test specimen to ambient temperature upon completion of the experiment.
9.4 Reweigh the sample container. Compare this mass of the sealed sample container weight with N determined in 9.1. Report any
specimen weight loss observed.
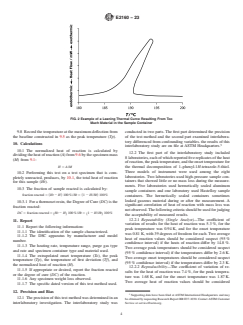
9.5 Construct a line connecting the baseline before the exothermic reaction to that after the reaction (see Fig. 1).
NOTE 7—For highly energetic reactions, a significant change may occur in the baseline prior to and following the exothermic reaction, due to a significant
change in the heat capacity of the reacted material in the sampl
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...