ASTM E1641-23
(Test Method)Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method
Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method
SIGNIFICANCE AND USE
5.1 Thermogravimetry provides a rapid method for determining the temperature-decomposition profile of a material.
5.2 This test method can be used for estimating lifetimes of materials, using Practice E1877 provided that a relationship has been established between the thermal endurance test results and actual lifetime tests.
SCOPE
1.1 This test method describes the determination of the kinetic parameters, Arrhenius activation energy, and pre-exponential factor by thermogravimetry, based on the assumption that the decomposition obeys first-order kinetics using the Ozawa/Flynn/Wall isoconversional method (1, 2).2
1.2 This test method is generally applicable to materials with well-defined decomposition profiles, namely, a smooth, continuous mass change with a single maximum rate.
1.3 This test method is normally applicable to decomposition occurring in the range from 400 K to 1300 K (nominally 100 °C to 1000 °C). The temperature range may be extended depending on the instrumentation used.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jul-2023
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Refers
ASTM E2040-19 - Standard Test Method for Mass Scale Calibration of Thermogravimetric Analyzers - Effective Date
- 01-Dec-2019
- Effective Date
- 15-Jan-2018
- Effective Date
- 01-May-2015
- Effective Date
- 01-Mar-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Apr-2014
- Refers
ASTM E2040-08(2014) - Standard Test Method for Mass Scale Calibration of Thermogravimetric Analyzers - Effective Date
- 15-Mar-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 15-Oct-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Nov-2011
Overview
ASTM E1641-23 is the Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method, developed by ASTM International. This standard provides laboratories and researchers with a structured approach for determining the kinetic parameters-specifically the Arrhenius activation energy and pre-exponential factor-of materials undergoing decomposition. Using thermogravimetric analysis (TGA) and the Ozawa/Flynn/Wall (OFW) isoconversional method, ASTM E1641-23 enables the analysis of materials exhibiting smooth, continuous mass loss under programmed heating rates. This method is especially significant for materials science and polymer research, as it offers a reliable means to understand decomposition mechanisms and estimate the thermal endurance or lifetime of materials.
Key Topics
- Thermogravimetric Analysis (TGA): A technique for tracking mass loss as a function of temperature, TGA is utilized to assess the decomposition profile of materials.
- Ozawa/Flynn/Wall Method: An isoconversional approach that assumes first-order decomposition kinetics, enabling calculation of activation energy and pre-exponential factor without detailed knowledge of reaction order.
- Kinetic Parameters: The standard focuses on quantifying the Arrhenius activation energy (E) and pre-exponential factor (A), critical values for modeling decomposition kinetics.
- Temperature Range: Typically, the method is applicable within 400 K to 1300 K (approximately 100°C to 1000°C), though the range can be extended if instrumentation allows.
- Sample Types: Applicable to materials with straightforward decomposition behavior, typically shown by a single, sharp weight loss event.
- Safety and Calibration: Emphasizes the need for proper calibration, sample handling, and consideration of safety, health, and environmental practices in accordance with international guidelines.
Applications
The ASTM E1641-23 standard test method has broad utility across various sectors:
- Material Lifetime Estimation: By determining kinetic parameters, this standard facilitates the estimation of thermal endurance and service lifetime for polymers, composites, and other materials.
- Quality Assurance: Manufacturers can benchmark material thermal stability and predict performance under high-temperature conditions.
- Research and Development: Scientists and engineers can employ this method to screen new formulations or understand degradation mechanisms efficiently.
- Comparative Analysis: Consistent methodology allows for reliable comparison of thermal stability between different batches or material types.
- Performance Testing: Frequently relied on in industries where thermal stability is crucial, such as aerospace, automotive, electronics, and advanced polymers.
Related Standards
For comprehensive thermal characterization and assurance of measurement accuracy, ASTM E1641-23 references the following related ASTM standards:
- ASTM E1877 - Practice for Calculating Thermal Endurance of Materials from Thermogravimetric Decomposition Data
- ASTM E1582 - Test Method for Temperature Calibration of Thermogravimetric Analyzers
- ASTM E2040 - Test Method for Mass Scale Calibration of Thermogravimetric Analyzers
- ASTM E691 - Practice for Conducting an Interlaboratory Study for Precision of Test Methods
- ASTM E29, E473, E1142 - Practices and terminologies for thermal analysis and statistical treatment
- ASTM E3007 - Practice for Selection and Use of Kinetic Reference Values
- ASTM E3142 - Test Method for Thermal Lag of Thermal Analysis Apparatus
Summary
ASTM E1641-23 is an essential standard for anyone involved in the evaluation of thermal stability and kinetic analysis via TGA. By providing a reproducible methodology for calculating decomposition kinetics using the Ozawa/Flynn/Wall approach, it supports accurate lifetime estimation, research innovation, and reliable material performance benchmarking in diverse high-temperature applications. For optimal implementation, integration with referenced ASTM standards and adherence to best practices in calibration and safety are strongly recommended.
Buy Documents
ASTM E1641-23 - Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method
REDLINE ASTM E1641-23 - Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E1641-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method". This standard covers: SIGNIFICANCE AND USE 5.1 Thermogravimetry provides a rapid method for determining the temperature-decomposition profile of a material. 5.2 This test method can be used for estimating lifetimes of materials, using Practice E1877 provided that a relationship has been established between the thermal endurance test results and actual lifetime tests. SCOPE 1.1 This test method describes the determination of the kinetic parameters, Arrhenius activation energy, and pre-exponential factor by thermogravimetry, based on the assumption that the decomposition obeys first-order kinetics using the Ozawa/Flynn/Wall isoconversional method (1, 2).2 1.2 This test method is generally applicable to materials with well-defined decomposition profiles, namely, a smooth, continuous mass change with a single maximum rate. 1.3 This test method is normally applicable to decomposition occurring in the range from 400 K to 1300 K (nominally 100 °C to 1000 °C). The temperature range may be extended depending on the instrumentation used. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Thermogravimetry provides a rapid method for determining the temperature-decomposition profile of a material. 5.2 This test method can be used for estimating lifetimes of materials, using Practice E1877 provided that a relationship has been established between the thermal endurance test results and actual lifetime tests. SCOPE 1.1 This test method describes the determination of the kinetic parameters, Arrhenius activation energy, and pre-exponential factor by thermogravimetry, based on the assumption that the decomposition obeys first-order kinetics using the Ozawa/Flynn/Wall isoconversional method (1, 2).2 1.2 This test method is generally applicable to materials with well-defined decomposition profiles, namely, a smooth, continuous mass change with a single maximum rate. 1.3 This test method is normally applicable to decomposition occurring in the range from 400 K to 1300 K (nominally 100 °C to 1000 °C). The temperature range may be extended depending on the instrumentation used. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1641-23 is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1641-23 has the following relationships with other standards: It is inter standard links to ASTM E473-23b, ASTM E1142-23b, ASTM E2040-19, ASTM E3142-18, ASTM E1142-15, ASTM E1877-15, ASTM E1142-14b, ASTM E473-14, ASTM E1142-14a, ASTM E2040-08(2014), ASTM E1142-14, ASTM E1877-13, ASTM E691-13, ASTM E1142-12, ASTM E691-11. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1641-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1641 − 23
Standard Test Method for
Decomposition Kinetics by Thermogravimetry Using the
Ozawa/Flynn/Wall Method
This standard is issued under the fixed designation E1641; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* E29 Practice for Using Significant Digits in Test Data to
Determine Conformance with Specifications
1.1 This test method describes the determination of the
E473 Terminology Relating to Thermal Analysis and Rhe-
kinetic parameters, Arrhenius activation energy, and pre-
ology
exponential factor by thermogravimetry, based on the assump-
E691 Practice for Conducting an Interlaboratory Study to
tion that the decomposition obeys first-order kinetics using the
2 Determine the Precision of a Test Method
Ozawa/Flynn/Wall isoconversional method (1, 2).
E1142 Terminology Relating to Thermophysical Properties
1.2 This test method is generally applicable to materials
E1582 Test Method for Temperature Calibration of Thermo-
with well-defined decomposition profiles, namely, a smooth,
gravimetric Analyzers
continuous mass change with a single maximum rate.
E1877 Practice for Calculating Thermal Endurance of Ma-
1.3 This test method is normally applicable to decomposi- terials from Thermogravimetric Decomposition Data
E1970 Practice for Statistical Treatment of Thermoanalytical
tion occurring in the range from 400 K to 1300 K (nominally
100 °C to 1000 °C). The temperature range may be extended Data
E2040 Test Method for Mass Scale Calibration of Thermo-
depending on the instrumentation used.
gravimetric Analyzers
1.4 The values stated in SI units are to be regarded as
E3007 Practice for Selection and Use of Kinetic Reference
standard. No other units of measurement are included in this
Values in the Study of Decomposition Reactions by
standard.
Thermogravimetry
1.5 This standard does not purport to address all of the
E3142 Test Method for Thermal Lag of Thermal Analysis
safety concerns, if any, associated with its use. It is the
Apparatus
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter- 3. Terminology
mine the applicability of regulatory limitations prior to use.
3.1 Definitions—Technical terms used in this test method
1.6 This international standard was developed in accor-
are defined in Terminologies E473 and E1142 and include
dance with internationally recognized principles on standard-
activation energy, Celsius, failure, failure criterion, and ther-
ization established in the Decision on Principles for the
mogravimetric analyzer.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical 4. Summary of Test Method
Barriers to Trade (TBT) Committee.
4.1 This test method is based upon the general rate equation
that takes the form of:
2. Referenced Documents
dα ⁄ dT 5 A 1 2 α exp 2 E ⁄ RT ⁄ β (1)
3 ~ ! @ #
2.1 ASTM Standards:
where:
α = fraction reacted (dimensionless),
This test method is under the jurisdiction of ASTM Committee E37 on Thermal
-1
A = pre-exponential factor (min ),
Measurements and is the direct responsibility of Subcommittee E37.01 on Calo-
rimetry and Mass Loss. β = heating rate (K/min),
Current edition approved Aug. 1, 2023. Published August 2023. Originally
E = activation energy (J/mol),
approved in 1994. Last previous edition approved in 2018 as E1641 – 18. DOI:
R = gas constant (= 8.316 J/(mol K)),
10.1520/E1641-23.
T = absolute temperature (K),
The boldface numbers in parentheses refer to the list of references at the end of
exp = Euler’s number exponential, and
this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or dα / dT = rate of change of α with T.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
4.2 Using the method of Ozawa, Flynn, and Wall (1, 2), Eq
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. 1 may be solved for activation energy:
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1641 − 23
E 5 2 R ⁄ b ∆log β ⁄ ∆ 1 ⁄ T (2) 4.6 This iterative process is continued until the value of
~ ! @ # ~ !
activation energy no longer changes with the next iteration.
where:
4.7 For first order reactions (n51), the value of the pre-
E = the derivative of the Doyle approximation (3) with
exponential factor (A) may be determined using Eq 3 (4).
values tabulated in Table 1, and
b = value from Table 1. a
A 5 2βR ⁄ E ln @1 2 α# 10 (3)
~ ! ~ !
4.3 Using a point of constant conversion from a series of
where:
decomposition curves obtained at different heat rates,
a = the Doyle approximation value from Table 1.
∆log β ⁄ ∆ 1 ⁄ T is obtained by linear regression.
@ # ~ !
4.8 This test method consists of heating a series of four or
4.4 Assuming an initial value of b50.457, a first approxima-
more test specimens, taken from the original sample, each at a
tion of activation energy (Eʹ) is obtained using Eq 2.
different heating rate between 1 K/min and 10 K/min, through
4.5 This approximate activation energy is then used to
their decomposition region. The specimen mass is recorded
determine a new value of bʹ using Table 1.
continuously as a function of temperature. The temperatures
for constant conversion are determined from the resultant mass
loss curves. The Arrhenius activation energy is then determined
from a plot of the logarithm of heating rate versus the
TABLE 1 Numerical Integration Constants
reciprocal of the absolute temperature at constant conversion
E/RT a b
level.
8 5.3699 0.5398
9 5.8980 0.5281
4.9 This activation energy may then be used to calculate
10 6.4167 0.5187
thermal endurance and an estimate of the lifetime of the
11 6.928 0.511
12 7.433 0.505
material at a certain temperature using Practice E1877.
13 7.933 0.500
14 8.427 0.494
5. Significance and Use
15 8.918 0.491
16 9.406 0.488
5.1 Thermogravimetry provides a rapid method for deter-
17 9.890 0.484
mining the temperature-decomposition profile of a material.
18 10.372 0.482
19 10.851 0.479
5.2 This test method can be used for estimating lifetimes of
20 11.3277 0.4770
21 11.803 0.475
materials, using Practice E1877 provided that a relationship
22 12.276 0.473
has been established between the thermal endurance test results
23 12.747 0.471
and actual lifetime tests.
24 13.217 0.470
25 13.686 0.469
26 14.153 0.467
6. Apparatus
27 14.619 0.466
28 15.084 0.465
6.1 The essential equipment required to provide the mini-
29 15.547 0.463
mum thermogravimetric analytical capability of this test
30 16.0104 0.4629
31 16.472 0.462
method includes:
32 16.933 0.461
6.1.1 A thermobalance, composed of (a) a furnace to
33 17.394 0.461
provide uniform controlled heating of a specimen at a constant
34 17.853 0.459
35 18.312 0.459
rate within the temperature range from ambient to 1300 K; (b)
36 18.770 0.458
a temperature sensor to provide an indication of the specimen/
37 19.228 0.458
furnace temperature to 60.1 K; (c) an electrobalance to
38 19.684 0.456
39 20.141 0.456
continuously measure the specimen mass with a minimum
40 20.5967 0.4558
capacity of 20 mg and a sensitivity of 650 μg; and (d) a means
41 21.052 0.455
of sustaining the specimen/container under atmospheric con-
42 21.507 0.455
43 21.961 0.454
trol of an inert or reactive purge gas of 99.99 % purity at a rate
44 22.415 0.454
of 20 mL/min to 50 mL ⁄min 6 5 mL ⁄min.
45 22.868 0.453
6.1.2 A temperature controller, capable of executing a
46 23.321 0.453
47 23.774 0.453
specific temperature program by operating the furnace between
48 24.226 0.452
selected temperature limits at a rate of temperature change
49 24.678 0.452
between 1 K/min and 10 K/min to within 60.1 K/min.
50 25.1295 0.4515
51 25.5806 0.4511
NOTE 1—The precision of results is strongly dependent upon the
52 26.0314 0.4508
53 26.4820 0.4506
precision of the heating rate; the greater the heating rate precision, the
54 26.9323 0.4503
greater the precision of results. The precision described here should be
55 27.3823 0.4500
considered to be the minimum suitable for this test.
56 27.8319 0.4498
57 28.2814 0.4495 6.1.3 A data collection device, to provide a means of
58 28.7305 0.4491
acquiring, storing, and displaying measured or calculated
59 29.1794 0.4489
signals, or both. The minimum output signals required for this
60 29.6281 0.4487
test method are mass, temperature, and time.
E1641 − 23
6.1.4 Containers (pans, crucibles, and so forth) which are 8.3 Certain materials require more sophisticated
inert to the specimen and that will remain dimensionally stable conditioning, such as maintaining the sample at a specified
over the temperature range from ambient to 1300 K. room temperature and relative humidity for an extended period
of time. Such conditioning may be conducted, but procedural
6.2 High-Purity (99.99 %) Nitrogen Supply, for purge gas.
details shall be included in the report.
NOTE 2—Other atmospheres may be used but shall be reported.
9. Calibration
6.3 Auxiliary apparatus considered necessary or useful in
conducting this test method include:
9.1 Prepare the thermogravimetric analyzer using any pro-
6.3.1 Cryogenic Mill to grind or mill test specimens to a fine cedures described in the manufacturer’s Operations manual.
powder at temperatures below –173 K (–100 °C).
9.2 Place the temperature sensor within 2 mm of the outside
of the specimen holder. Care must be taken to ensure that the
7. Precautions
specimen holder is not touched in any way by the sensor and
that it is not moved after temperature calibration.
7.1 It is essential that the samples be representative since
milligram quantities of specimen are to be used.
9.3 Maintain a constant flow rate of purge gas in the range
from 20 mL/min to 50 mL/min throughout the experiment.
7.2 The value of the calculated activation energy is inde-
pendent of reaction order in the early stages of decomposition.
NOTE 5—In the case of samples that may be sensitive to oxidative
This assumption does not hold for the later stages and shall be
degradation, it will be necessary to maintain inert gas purging for a time
sufficient to ensure that all residual oxygen is removed from the system
used with caution. An upper limit of 10 % decomposition is
prior to the start of the temperature program. It may be necessary to
suggested. It is strongly suggested that calculations be made at
evacuate the system prior to initiating inert gas purging for some
several different levels of decomposition, for example, 5 %,
instruments.
10 %, 15 %, and 20 %. Variations in the results among these
9.4 Calibrate the instrument furnace temperature in accor-
determinations could indicate the inapplicability of one of
dance with the calibration procedure in Test Method E1582
them. For instance, volatile, low-level impurities would affect
using the same heating rate, purge gas, and flow rate to be used
the results of the lowest conversion determination more than
for the specimens. The temperature calibration shall be per-
those at higher conversions. Consistent results for all conver-
formed both prior to every change in heating rate and at that
sions validate the method for the range of conversions exam-
heating rate (see Appendix X2).
ined.
9.5 Calibrate the mass signal using Test Method E2040.
7.3 Toxic or corrosive effluents, or both, may be released
NOTE 6—Quality initiatives call for calibration of all signals at least
during the heating process and may be harmful to the personnel
annually.
or apparatus.
10. Procedure
8. Sampling
10.1 Place 3 mg 6 1 mg of the specimen under test into a
8.1 Powdered or granular specimens that have a high
clean, tared instrument specimen holder.
surface-to-volume ratio, are preferred, although films, fibers,
NOTE 7—Other specimen quantity may be used but shall be indicated in
and fabrics may be used providing that care is taken to make all
the report.
of the specimens uniform in size and shape. Under circum-
NOTE 8—The specimen holder should be tared in the fully assembled
stances in which material parts are available, the specimens
system, with the purge gas flowing.
NOTE 9—Powdered or granular specimens should be distributed evenly
should be prepared by filing or rasping the part. All specimens
over the specimen holder so as to maximize the exposed surface. A
should be mixed thoroughly prior to sampling if possible, and
one-grain thick layer would be optimal.
they should be sampled by removing portions from various
10.2 Select an equilibrium temperature based upon the
parts of the container. These portions should in turn be
heating rate and known decomposition first-deviation-from-
combined and mixed well to ensure a representative specimen
baseline temperature of the specimen, where the equilibrium
for the determination.
temperature equals the decomposition temperature – (10 min ×
NOTE 3—Care should be exercised during sample preparation to avoid
heating rate). If the percentage mass loss is to be recorded,
contamination.
establish zero percent loss at this time.
NOTE 4—The specimen size and surface-to-volume ratio are known to
affect the results of this test. A narrow range of specimen sizes should be
NOTE 10—If zero percent mass loss is established at the time at which
used, as noted in 10.1. Uniformity in particle size can be achieved, without
the specimen is placed into the instrument, the specimen mass at the
the loss of volatiles, by using a cryogenic mill to grind the sample to a fine
equilibration temperature can be greater than 100 % due to buoyancy
powder. To prevent the condensation of moisture, the mill should be
effects. A blank should be run for accurate determination of the buoyancy
opened only after returning fully to ambient temperature, or the operation
effect throughout the temperature range of the experiment. The blank can
should be performed in a glove box filled with dry gas.
be a piece of platinum of approximately the same volume as the specimen.
The balance drift at any temperature can be determined in this manner.
8.2 In the absence of other information, the samples are
assumed to be analyzed as received except for the mechanical 10.3 Heat the specimen at a constant rate through the
treatment noted in 8.1. If some heat treatment, such as drying, decomposition profile until a constant mass is obtained or the
is applied to the sample prior to analysis, this treatment and any temperature is well beyond the useful temperature range of the
resulting mass loss must be noted in the report. material tested. Record the accompanying thermal curve, with
E1641 − 23
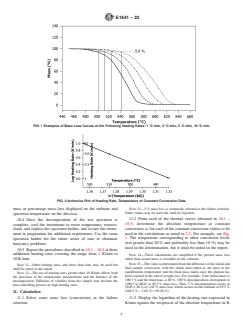
FIG. 1 Examples of Mass Loss Curves at the Following Heating Rates: 1 °C ⁄min, 2 °C ⁄min, 5 °C ⁄min, 10 °C ⁄min
FIG. 2 Arrhenius Plot of Heating Rate, Temperature of Constant Conversion Data
NOTE 13—5 % mass loss is commonly selected as the failure criterion.
mass or percentage mass loss displayed on the ordinate and
Other values may be used but shall be reported.
specimen temperature on the abscissa.
11.2 From each of the thermal curves obtained in 10.1 –
10.4 Once the decomposition
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1641 − 18 E1641 − 23
Standard Test Method for
Decomposition Kinetics by Thermogravimetry Using the
Ozawa/Flynn/Wall Method
This standard is issued under the fixed designation E1641; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method describes the determination of the kinetic parameters, Arrhenius activation energy, and pre-exponential factor
by thermogravimetry, based on the assumption that the decomposition obeys first-order kinetics using the Ozawa/Flynn/Wall
isoconversional method (1, 2).
1.2 This test method is generally applicable to materials with well-defined decomposition profiles, namely, a smooth, continuous
mass change with a single maximum rate.
1.3 This test method is normally applicable to decomposition occurring in the range from 400 K to 1300 K (nominally 100°C to
1000°C).100 °C to 1000 °C). The temperature range may be extended depending on the instrumentation used.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E473 Terminology Relating to Thermal Analysis and Rheology
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E1142 Terminology Relating to Thermophysical Properties
E1582 Test Method for Temperature Calibration of Thermogravimetric Analyzers
E1877 Practice for Calculating Thermal Endurance of Materials from Thermogravimetric Decomposition Data
E1970 Practice for Statistical Treatment of Thermoanalytical Data
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.01 on Calorimetry
and Mass Loss.
Current edition approved Nov. 1, 2018Aug. 1, 2023. Published November 2018August 2023. Originally approved in 1994. Last previous edition approved in 20162018
as E1641 – 16.E1641 – 18. DOI: 10.1520/E1641-18.10.1520/E1641-23.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1641 − 23
E2040 Test Method for Mass Scale Calibration of Thermogravimetric Analyzers
E3007 Practice for Selection and Use of Kinetic Reference Values in the Study of Decomposition Reactions by Thermogravi-
metry
E3142 Test Method for Thermal Lag of Thermal Analysis Apparatus
2.2 Other Standard:
ISO 11358-2 Plastics Thermogravimetry (TG) of Polymers Part 2: Determination of Kinetic Parameters
3. Terminology
3.1 Definitions—Technical terms used in this test method are defined in Terminologies E473 and E1142 and include activation
energy, Celsius, failure, failure criterion, and thermogravimetric analyzer.
4. Summary of Test Method
4.1 This test method is based upon the general rate equation that takes the form of:
dα ⁄ dT 5 A 1 2 α exp 2 E ⁄ RT ⁄ β (1)
~ ! @ #
where:
α = fraction reacted (dimensionless),
-1
A = pre-exponential factor (min ),
β = heating rate (K/min),
E = activation energy (J/mol),
R = gas constant (= 8.316 J/(mol K)),
T = absolute temperature (K),
exp = Euler’s number exponential, and
dα / dT = rate of change of α with T.
4.2 Using the method of Ozawa, Flynn, and Wall (1, 2),Eq 1 may be solved for activation energy:
E 52~R ⁄ b! ∆log@β# ⁄ ∆~1 ⁄ T! (2)
where:
E = the derivative of the Doyle approximation (3) with values tabulated in Table 1, and
b = value from Table 1.
4.3 Using a point of constant conversion from a series of decomposition curves obtained at different heat rates, ∆log@β# ⁄ ∆~1 ⁄ T! is
obtained by linear regression.
4.4 Assuming an initial value of b50.457, a first approximation of activation energy (Eʹ) is obtained using Eq 2.
4.5 This approximate activation energy is then used to determine a new value of bʹ using Table 1.
4.6 This iterative process is continued until the value of activation energy no longer changes with the next iteration.
4.7 For first order reactions (n51), the value of the pre-exponential factor (A) may be determined using Eq 3 (4).
a
A 5 2βR ⁄ E ln 1 2 α 10 (3)
~ ! ~ @ #!
where:
a = the Doyle approximation value from Table 1.
4.8 This test method consists of heating a series of four or more test specimens, taken from the original sample, each at a different
heating rate between 1 K/min and 10 K/min, through their decomposition region. The specimen mass is recorded continuously as
a function of temperature. The temperatures for constant conversion are determined from the resultant mass loss curves. The
Arrhenius activation energy is then determined from a plot of the logarithm of heating rate versus the reciprocal of the absolute
temperature at constant conversion level.
E1641 − 23
TABLE 1 Numerical Integration Constants
E/RT a b
8 5.3699 0.5398
9 5.8980 0.5281
10 6.4167 0.5187
11 6.928 0.511
12 7.433 0.505
13 7.933 0.500
14 8.427 0.494
15 8.918 0.491
16 9.406 0.488
17 9.890 0.484
18 10.372 0.482
19 10.851 0.479
20 11.3277 0.4770
21 11.803 0.475
22 12.276 0.473
23 12.747 0.471
24 13.217 0.470
25 13.686 0.469
26 14.153 0.467
27 14.619 0.466
28 15.084 0.465
29 15.547 0.463
30 16.0104 0.4629
31 16.472 0.462
32 16.933 0.461
33 17.394 0.461
34 17.853 0.459
35 18.312 0.459
36 18.770 0.458
37 19.228 0.458
38 19.684 0.456
39 20.141 0.456
40 20.5967 0.4558
41 21.052 0.455
42 21.507 0.455
43 21.961 0.454
44 22.415 0.454
45 22.868 0.453
46 23.321 0.453
47 23.774 0.453
48 24.226 0.452
49 24.678 0.452
50 25.1295 0.4515
51 25.5806 0.4511
52 26.0314 0.4508
53 26.4820 0.4506
54 26.9323 0.4503
55 27.3823 0.4500
56 27.8319 0.4498
57 28.2814 0.4495
58 28.7305 0.4491
59 29.1794 0.4489
60 29.6281 0.4487
4.9 This activation energy may then be used to calculate thermal endurance and an estimate of the lifetime of the material at a
certain temperature using Practice E1877.
5. Significance and Use
5.1 Thermogravimetry provides a rapid method for determining the temperature-decomposition profile of a material.
5.2 This test method can be used for estimating lifetimes of materials, using Practice E1877 provided that a relationship has been
established between the thermal endurance test results and actual lifetime tests.
6. Apparatus
6.1 The essential equipment required to provide the minimum thermogravimetric analytical capability of this test method includes:
E1641 − 23
6.1.1 A thermobalance, composed of (a) a furnace to provide uniform controlled heating of a specimen at a constant rate within
the temperature range from ambient to 1300 K; (b) a temperature sensor to provide an indication of the specimen/furnace
temperature to 60.1 K; (c) an electrobalance to continuously measure the specimen mass with a minimum capacity of 20 mg and
a sensitivity of 650 μg; and (d) a means of sustaining the specimen/container under atmospheric control of an inert or reactive
purge gas of 99.99 % purity at a rate of 20 mL/min to 50 6 550 mL ⁄min 6 5 mL ⁄min.
6.1.2 A temperature controller, capable of executing a specific temperature program by operating the furnace between selected
temperature limits at a rate of temperature change between 1 K/min and 10 K/min to within 60.1 K/min.
NOTE 1—The precision of results is strongly dependent upon the precision of the heating rate; the greater the heating rate precision, the greater the
precision of results. The precision described here should be considered to be the minimum suitable for this test.
6.1.3 A data collection device, to provide a means of acquiring, storing, and displaying measured or calculated signals, or both.
The minimum output signals required for this test method are mass, temperature, and time.
6.1.4 Containers (pans, crucibles, and so forth) which are inert to the specimen and that will remain dimensionally stable over
the temperature range from ambient to 1300 K.
6.2 High-Purity (99.99 %) Nitrogen Supply, for purge gas.
NOTE 2—Other atmospheres may be used but shall be reported.
6.3 Auxiliary apparatus considered necessary or useful in conducting this test method include:
6.3.1 Cryogenic Mill to grind or mill test specimens to a fine powder at temperatures below –173 K (–100°C).(–100 °C).
7. Precautions
7.1 It is essential that the samples be representative since milligram quantities of specimen are to be used.
7.2 The value of the calculated activation energy is independent of reaction order in the early stages of decomposition. This
assumption does not hold for the later stages and shall be used with caution. An upper limit of 10 % decomposition is suggested.
It is strongly suggested that calculations be made at several different levels of decomposition, for example, 5 %, 10 %, 15 %, and
20 %. Variations in the results among these determinations could indicate the inapplicability of one of them. For instance, volatile,
low-level impurities would affect the results of the lowest conversion determination more than those at higher conversions.
Consistent results for all conversions validate the method for the range of conversions examined.
7.3 Toxic or corrosive effluents, or both, may be released during the heating process and may be harmful to the personnel or
apparatus.
8. Sampling
8.1 Powdered or granular specimens that have a high surface-to-volume ratio, are preferred, although films, fibers, and fabrics may
be used providing that care is taken to make all of the specimens uniform in size and shape. Under circumstances in which material
parts are available, the specimens should be prepared by filing or rasping the part. All specimens should be mixed thoroughly prior
to sampling if possible, and they should be sampled by removing portions from various parts of the container. These portions
should in turn be combined and mixed well to ensure a representative specimen for the determination.
NOTE 3—Care should be exercised during sample preparation to avoid contamination.
NOTE 4—The specimen size and surface-to-volume ratio are known to affect the results of this test. A narrow range of specimen sizes should be used,
as noted in 10.1. Uniformity in particle size can be achieved, without the loss of volatiles, by using a cryogenic mill to grind the sample to a fine powder.
To prevent the condensation of moisture, the mill should be opened only after returning fully to ambient temperature, or the operation should be performed
in a glove box filled with dry gas.
E1641 − 23
8.2 In the absence of other information, the samples are assumed to be analyzed as received except for the mechanical treatment
noted in 8.1. If some heat treatment, such as drying, is applied to the sample prior to analysis, this treatment and any resulting mass
loss must be noted in the report.
8.3 Certain materials require more sophisticated conditioning, such as maintaining the sample at a specified room temperature and
relative humidity for an extended period of time. Such conditioning may be conducted, but procedural details shall be included
in the report.
9. Calibration
9.1 Prepare the thermogravimetric analyzer using any procedures described in the manufacturer’s Operations manual.
9.2 Place the temperature sensor within 2 mm of the outside of the specimen holder. Care must be taken to ensure that the
specimen holder is not touched in any way by the sensor and that it is not moved after temperature calibration.
9.3 Maintain a constant flow rate of purge gas in the range from 20 mL/min to 50 mL/min throughout the experiment.
NOTE 5—In the case of samples that may be sensitive to oxidative degradation, it will be necessary to maintain inert gas purging for a time sufficient to
ensure that all residual oxygen is removed from the system prior to the start of the temperature program. It may be necessary to evacuate the system prior
to initiating inert gas purging for some instruments.
9.4 Calibrate the instrument furnace temperature in accordance with the calibration procedure in Test Method E1582 using the
same heating rate, purge gas, and flow rate to be used for the specimens. The temperature calibration shall be performed both prior
to every change in heating rate and at that heating rate. rate (see Appendix X2).
9.5 Calibrate the mass signal using Test Method E2040.
NOTE 6—Quality initiatives call for calibration of all signals at least annually.
10. Procedure
10.1 Place 3 mg 6 1 mg of the specimen under test into a clean, tared instrument specimen holder.
NOTE 7—Other specimen quantity may be used but shall be indicated in the report.
NOTE 8—The specimen holder should be tared in the fully assembled system, with the purge gas flowing.
NOTE 9—Powdered or granular specimens should be distributed evenly over the specimen holder so as to maximize the exposed surface. A one-grain thick
layer would be optimal.
10.2 Select an equilibrium temperature based upon the heating rate and known decomposition first-deviation-from-baseline
temperature of the specimen, where the equilibrium temperature equals the decomposition temperature – (10 min × heating rate).
If the percentage mass loss is to be recorded, establish zero percent loss at this time.
NOTE 10—If zero percent mass loss is established at the time at which the specimen is placed into the instrument, the specimen mass at the equilibration
temperature can be greater than 100 % due to buoyancy effects. A blank should be run for accurate determination of the buoyancy effect throughout the
temperature range of the experiment. The blank can be a piece of platinum of approximately the same volume as the specimen. The balance drift at any
temperature can be determined in this manner.
10.3 Heat the specimen at a constant rate through the decomposition profile until a constant mass is obtained or the temperature
is well beyond the useful temperature range of the material tested. Record the accompanying thermal curve, with mass or
percentage mass loss displayed on the ordinate and specimen temperature on the abscissa.
10.4 Once the decomposition of the test specimen is complete, cool the instrument to room temperature, remove, clean, and
replace the specimen holder, and re-tare the instrument in preparation for additional experiments. Use the same specimen holder
for the entire series of runs to eliminate buoyancy problems.
E1641 − 23
FIG. 1 Examples of Mass Loss Curves at the Following Heating Rates: 1°C/min,1 °C 2°C/min,⁄min, 2 °C 5°C/min,⁄min, 5 °C 10°C/
min⁄min, 10 °C
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...