ASTM G3-89(1999)
(Practice)Standard Practice for Conventions Applicable to Electrochemical Measurements in Corrosion Testing
Standard Practice for Conventions Applicable to Electrochemical Measurements in Corrosion Testing
SCOPE
1.1 This practice is intended to provide conventions for reporting and displaying electrochemical corrosion data. Conventions for potential, current density, electrochemical impedance and admittance, as well as conventions for graphical presentation of such data are included.
1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
Relations
Standards Content (Sample)
NOTICE: This standard has either been superseded and replaced by a new version or withdrawn.
Contact ASTM International (www.astm.org) for the latest information
Designation:G3–89 (Reapproved 1999)
Standard Practice for
Conventions Applicable to Electrochemical Measurements
in Corrosion Testing
This standard is issued under the fixed designation G 3; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (e) indicates an editorial change since the last revision or reapproval.
1. Scope oxidizing condition at the electrode in question. The positive
direction has also been denoted as the noble direction because
1.1 This practice is intended to provide conventions for
the corrosion potentials of most noble metals, such as gold, are
reporting and displaying electrochemical corrosion data. Con-
more positive than the nonpassive base metals. On the other
ventions for potential, current density, electrochemical imped-
hand, the negative direction, often called the active direction, is
ance and admittance, as well as conventions for graphical
associated with reduction and consequently the corrosion
presentation of such data are included.
potentials of active metals, such as magnesium. This conven-
1.2 This standard does not purport to address all of the
tion was adopted unanimously by the 1953 International Union
safety concerns, if any, associated with its use. It is the
of Pure and Applied Chemistry as the standard for electrode
responsibility of the user of this standard to establish appro-
potential (1).
priate safety and health practices and determine the applica-
4.2 In the context of a specimen electrode of unknown
bility of regulatory limitations prior to use.
potential in an aqueous electrolyte, consider the circuit shown
2. Referenced Documents
in Fig. 1 with a reference electrode connected to the ground
terminal of an electrometer. If the electrometer reads on scale
2.1 ASTM Standards:
when the polarity switch is negative, the specimen electrode
IEEE/ASTM SI 10 Standard for Use of the International
potential is negative (relative to the reference electrode).
System of Units (SI) (the Modern Metric System)
Conversely, if the electrometer reads on scale when polarity is
3. Significance and Use
positive, the specimen potential is positive. On the other hand,
if the specimen electrode is connected to the ground terminal,
3.1 This practice provides guidance for reporting, display-
the potential will be positive if the meter is on scale when the
ing, and plotting electrochemical corrosion data and includes
polarity switch is negative, and vice versa.
recommendations on signs and conventions. Use of this prac-
tice will result in the reporting of electrochemical corrosion
NOTE 1—In cases where the polarity of a measuring instrument is in
data in a standard format, facilitating comparison between data
doubt, a simple verification test can be performed as follows: connect the
developed at different laboratories or at different times. The measuring instrument to a dry cell with the lead previously on the
reference electrode to the negative battery terminal and the lead previously
recommendations outlined in this standard may be utilized
on the specimen electrode to the positive battery terminal. Set the range
when recording and reporting corrosion data obtained from
switch to accommodate the dry cell voltage. The meter deflection will now
electrochemical tests such as potentiostatic and potentiody-
show the direction of positive potential.
namic polarization, polarization resistance, electrochemical
Also, the corrosion potential of magnesium or zinc should be negative
impedance and admittance measurements, galvanic corrosion,
ina1 N NaCl solution if measured against a saturated standard calomel
and open circuit potential measurements.
electrode (SCE).
4. Sign Convention for Electrode Potential
5. Sign Convention for Electrode Potential Temperature
Coefficients
4.1 The Stockholm sign invariant convention is recom-
mended for use in reporting the results of specimen potential
5.1 There are two types of temperature coefficients of
measurements in corrosion testing. In this convention, the electrode potential: isothermal temperature coefficients and the
positive direction of electrode potential implies an increasingly
thermal coefficients. The sign convention recommended for
both types of temperature coefficients is that the temperature
coefficient is positive when an increase in temperature pro-
duces an increase (that is, it becomes more positive) in the
This practice is under the jurisdiction of ASTM Committee G-1 on Corrosion
of Metals and is the direct responsibility of Subcommittee G01.11 on Electrochemi-
cal Measurements in Corrosion Testing.
Current edition approved Feb. 24, 1989. Published April 1989. Originally
e1 3
published as G 3 – 68. Last previous edition G3–74 (1981) . The boldface numbers in parentheses refer to the list of references at the end of
Annual Book of ASTM Standards, Vol 14.02 (excerpts in Vol 03.02). this practice.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959, United States.
G3–89 (1999)
where:
b = anodic Tafel slope,
a
b = cathodic Tafel slope, and
c
DE = the difference E − E , where E is the specimen
corr
potential.
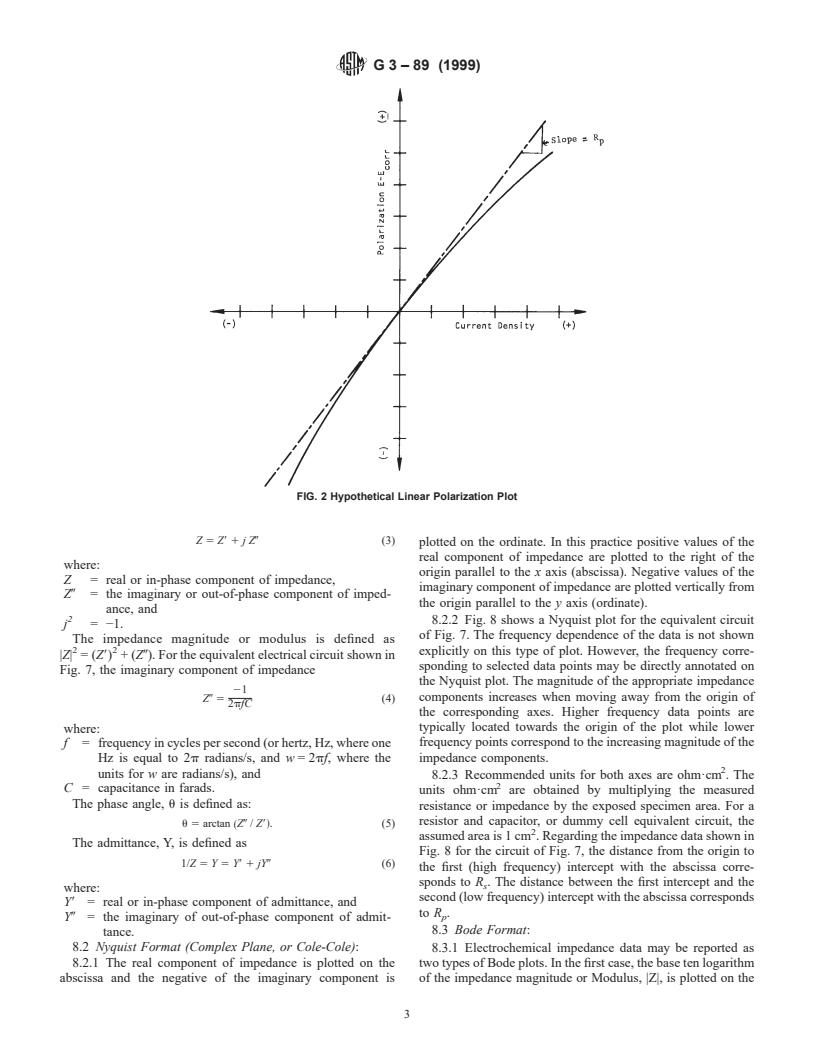
Fig. 2 is a plot of polarization, E − E , versus current density
corr
i (solid line) from which the polarization resistance R has been
p
determined as the slope of the curve at the corrosion potential
E .
corr
7.3 Potential Reference Points—In plots where electrode
potentials are displayed, some indication of the conversion of
the values displayed to both the standard hydrogen electrode
scale (SHE) and the saturated calomel electrode scale (SCE) is
recommended if they are known. For example, when electrode
potential is plotted as the ordinate, then the SCE scale could be
shown at the extreme left of the plot and the SHE scale shown
NOTE 1—The electrode potential of specimen is negative as shown.
FIG. 1 Schematic Diagram of an Apparatus to Measure Electrode
at the extreme right. An alternative, in cases where the
Potential of a Specimen
reference electrode was not either SCE or SHE, would be to
show on the potential axis the potentials of these electrodes
electrode potential. Likewise, the second temperature coeffi-
against the reference used. In cases where these points are not
cient is positive when an increase in temperature produces an
shown on the plot, an algebraic conversion could be indicated.
increase (that is, it becomes more positive) in the first tem-
For example, in the case of a silver-silver chloride reference
perature coefficient.
electrode (1 M KCl), the conversion could be shown in the title
box as:
6. Sign Convention for Current and Current Density
SCE 5 E 2 0.006 V (2)
6.1 The sign convention in which anodic currents and
SHE 5 E 1 0.235 V
current densities are considered positive and cathodic currents
and current densities are negative is recommended. When the
where E represents electrode potential measured against the
potential is plotted against the logarithm of the current density,
silver-silver chloride standard (1 M KCl).
only the absolute values of the current density can be plotted.
NOTE 2—A table of potentials for various common reference electrodes
In such plots, the values which are cathodic should be clearly
is presented in Appendix X2.
differentiated from the anodic values if both are present.
7.4 Units—The recommended unit of potential is the volt.
7. Conventions for Displaying Polarization Data
In cases where only small potential ranges are covered,
millivolts or microvolts may be used. The SI units for current
7.1 Sign Conventions—The standard mathematical practice
for plotting graphs is recommended for displaying electro- density are ampere per square metre or milliampere per square
centimetre (Practice E 380). Still in use are units expressed in
chemical corrosion data. In this practice, positive values are
plotted above the origin on the ordinate axis and to the right of amperes per square centimetre, and microamperes per square
centimetre.
the origin on the abscissa axis. In logarithmic plots, the
abscissa value increases from left to right and the ordinate 7.5 Sample Polarization Curves—Sample polarization plots
employing these recommended practices are shown in Figs.
value increases from bottom to top.
7.2 Current Density-Potential Plots—A uniform convention 2-6. Fig. 3 and Fig. 4 are hypothetical curves showing active
and active-passive anode behavior, respectively. Fig. 5 and Fig.
is recommended for plotting current density-potential data,
6 are actual polarization data for Type 430 stainless steel (UNS
namely, plot current density along the abscissa and potential
43000) (4) and two aluminum samples (5). Fig. 3 and Fig. 4 are
along the ordinate. In current density potential plots, the
exhibited to illustrate graphically the location of various points
current density may be plotted on linear or logarithmic axes. In
used in discussion of electrochemical methods of corrosion
general, logarithmic plots are better suited to incorporation of
testing. The purpose of Fig. 5 and Fig. 6 is to show how various
wide ranges of current density data and for demonstrating Tafel
relationships. Linear plots are recommended for studies in types of electrode behavior can be plotted in accordance with
the proposed conventions.
which the current density or potential range is small, or in cases
where the region in which the current density changes from
anodic to cathodic is important. Linear plots are also used for 8. Conventions for Displaying Electrochemical
the determination of the polarization resistance R , which is Impedance Data
p
defined as the slope of a potential-current density plot at the
8.1 Three graphical formats in common use for reporting
corrosion potential E . The relationship between the polar-
corr electrochemical impedance data are the Nyquist, Bode, and
ization resistance R and the corrosion current density i is as
p corr
Admittance formats. These formats are discussed for a simple
follows (2, 3):
electrode system modelled by an equivalent electrical circuit as
d~DE! b b shown in Fig. 7. In the convention utilized the impedance is
a c
5 R 5 (1)
F G
p
di 2.303~b 1 b !i defined as:
a c corr
DE 5 0
G3–89 (1999)
FIG. 2 Hypothetical Linear Polarization Plot
Z 5 Z8 1 jZ9 (3)
plotted on the ordinate. In this practice positive values of the
real component of impedance are plotted to the right of the
where:
origin parallel to the x axis (abscissa). Negative values of the
Z = real or in-phase component of impedance,
imaginary component of impedance are plotted vertically from
Z9 = the imaginary or out-of-phase component of imped-
the origin parallel to the y axis (ordinate).
ance, and
8.2.2 Fig. 8 shows a Nyquist plot for the equivalent circuit
j = −1.
of Fig. 7. The frequency dependence of the data is not shown
The impedance magnitude or modulus is defined as
2 2
explicitly on this type of plot. However, the frequency corre-
|Z| =(Z8) +(Z9). For the equivalent electrical circuit shown in
sponding to selected data points may be directly annotated on
Fig. 7, the imaginary component of impedance
the Nyquist plot. The magnitude of the appropriate impedance
components increases when moving away from the origin of
Z9 5 (4)
2pfC
the corresponding axes. Higher frequency data points are
typically located towards the origin of the plot while lower
where:
frequency points correspond to the increasing magnitude of the
f = frequency in cycles per second (or hertz, Hz, where one
Hz is equal to 2p radians/s, and w =2pf, where the impedance components.
units for w are radians/s), and
8.2.3 Recommended units for both axes are ohm·cm . The
C = capacitance in farads.
units ohm·cm are obtained by multiplying the measured
The phase angle, u is defined as:
resistance or impedance by the exposed specimen area. For a
resistor and capacitor, or dummy cell equivalent circuit, the
u5 arctan ~Z9 / Z8!. (5)
assumed area is 1 cm . Regarding the impedance data shown in
The admittance, Y, is defined as
Fig. 8 for the circuit of Fig. 7, the distance from the origin to
1/Z 5 Y 5 Y8 1 jY9 (6)
the first (high frequency) intercept with the abscissa corre-
sponds to R . The distance between the first intercept and the
s
where:
second (low frequency) intercept with the abscissa corresponds
Y8 = real or in-phase component of admittance, and
to R .
p
Y9 = the imaginary of out-of-phase component of admit-
8.3 Bode Format:
tance.
8.2 Nyquist Format (Complex Plane, or Cole-Cole): 8.3.1 Electrochemical impedance data may be reported as
8.2.1 The real component of impedance is plotted on the two types of Bode plots. In the first case, the base ten logarithm
abscissa and the negative of the imaginary component is of the impedance magnitude or Modulus, |Z|, is plotted on the
G3–89 (1999)
FIG. 3 Hypothetical Cathodic and Anodic Polarization Diagram
FIG. 4 Hypothetical Cathodic and Anodic Polarization Plots for a Passive Anode
ordinate and the base ten logarithm of the frequency is plotted origin itself is chosen at appropriate nonzero values of imped-
on the abscissa. In this practice increasing frequency values are ance magnitude and frequency.
plotted to the right of the origin parallel to the x axis (abscissa) 8.3.2 Fig. 9 shows a typical plot for the simple electrical
and increasing values of impedance magnitude are plotted circuit model of Fig. 7. The magnitude of the high frequency
vertically from the origin parallel to the y axis (ordinate). The impedance where the impedance magnitude is independent of
G3–89 (1999)
FIG. 5 Typical Potentiostatic Anodic Polarization Plot for Type 430 Stainless Steel in 1.0 N H SO
2 4
FIG. 6 Typical Polarization Plots for Aluminum Materials in 0.2 N NaCl Solution
frequency corresponds to R . The difference in magnitude (ordinate). In this format, a pure capacitive behavior is plotted
s
between the low frequency and the high frequency frequency- as a positive value of 90°. Fig. 10 shows a typical plot for the
independent regions of impedance magnitude corresponds to simple electrode model shown in Fig. 7.
R . These resistances are identical to those on the Nyquist 8.3.4 The units for the frequency on both plots are either
p
format plot shown in Fig. 8. hertz (cycles per second) or radians per second (radians per
8.3.3 In the second type of Bode plot, the negative of the second = 2p radians per cycle multiplied by the number of
phase angle, − u, is plotted on the ordinate and the base ten
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.