ASTM F2603-06(2020)
(Guide)Standard Guide for Interpreting Images of Polymeric Tissue Scaffolds
Standard Guide for Interpreting Images of Polymeric Tissue Scaffolds
SIGNIFICANCE AND USE
4.1 This document provides guidance for users who wish to obtain quantifiable data from images of tissue scaffolds manufactured from polymers that include both high water content gels and woven textiles.
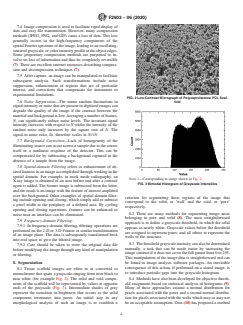
4.2 Information derived from tissue scaffold images can be used to optimize the structural characteristics of the matrix for a particular application, to develop better manufacturing procedures or to provide a measure of quality assurance and product traceability. Fig. 1 provides a summary of the key stages of image capture and analysis.
FIG. 1 Key Stages in Image Capture, Storage, and Analysis
4.3 There is a synergy between the analysis of pores in tissue scaffolds and that of particles that is reflected in standards cited and in the analysis described in Section 9. Guide E1919 provides a compendium of standards for particle analysis that includes measurement techniques, data analytical and sampling methodologies.
SCOPE
1.1 This guide covers the factors that need to be considered in obtaining and interpreting images of tissue scaffolds including technique selection, instrument resolution and image quality, quantification and sample preparation.
1.2 The information in this guide is intended to be applicable to porous polymer-based tissue scaffolds, including naturally derived materials such as collagen. However, some materials (both synthetic and natural) may require unique or varied sample preparation methods that are not specifically covered in this guide.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jul-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 01-Aug-2020
- Effective Date
- 01-Oct-2019
- Effective Date
- 15-Nov-2018
- Effective Date
- 01-May-2018
- Effective Date
- 01-Oct-2016
- Effective Date
- 15-Mar-2015
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 15-Jun-2009
- Effective Date
- 01-Jun-2009
- Effective Date
- 01-Dec-2007
Overview
ASTM F2603-06(2020), Standard Guide for Interpreting Images of Polymeric Tissue Scaffolds, is published by ASTM International. This guide provides comprehensive recommendations for interpreting and quantifying images of tissue scaffolds made from polymers, including high water content gels and woven textiles. The information extracted from these images plays a critical role in optimizing scaffold matrices for biomedical applications, improving manufacturing processes, and establishing quality assurance and product traceability in tissue engineering and regenerative medicine.
The guide outlines essential factors in imaging, such as technique selection, instrument resolution, image quality, quantification, and sample preparation. It also emphasizes the importance of using standardized methods and consistent units (SI units) in analysis, while noting the need for special preparation in certain natural or synthetic materials.
Key Topics
Imaging Methods: The guide discusses various imaging techniques suitable for polymeric tissue scaffolds, including optical microscopy, electron microscopy, magnetic resonance imaging (MRI), scanning acoustic microscopy (SAM), and X-ray micro-computed tomography (µ-CT). Technique selection depends on the scaffold material's properties, required contrast, and targeted pore size.
Resolution and Image Quality: Accurate interpretation relies on high-resolution images and appropriate dynamic range. The need for proper sampling frequency (per the Nyquist criterion) and the influence of compression and noise suppression on image quality are highlighted.
Sample Preparation: The guide addresses the potential for artifacts during preparation, especially for structures with high water content or delicate morphologies. Proper handling during processes like freezing, slicing, or fluid embedding is crucial to preserve scaffold structure.
Quantitative Analysis: Key scaffold parameters identified include overall porosity, pore size distribution, and pore interconnectivity/tortuosity. The guide details methods for segmenting images (thresholding, edge detection), binarizing for analysis, and measuring morphological features such as roundness, aspect ratio, elongation, and form factor.
Stereological Measurements: Methods for converting image data to meaningful 2-D and 3-D quantitative metrics are described, including area and volume fractions, and classification of pore types (open, closed, blind-ended).
Quality Control and Calibration: Emphasis is placed on equipment calibration, appropriate selection of reference materials, and ensuring statistically meaningful, representative data collection across multiple images and sample areas.
Applications
The practical value of ASTM F2603-06(2020) lies in its widespread application for:
- Biomaterials Research: Quantitative imaging informs the design and optimization of tissue scaffolds for applications such as bone, cartilage, and nerve regeneration.
- Medical Device Development: Image-derived data supports the development and improvement of polymeric scaffolds used in implants and tissue engineering products.
- Manufacturing Process Optimization: Imaging results help refine fabrication methods, supporting reproducibility and scalability in scaffold production.
- Quality Assurance and Product Traceability: Standardized imaging and quantification support regulatory compliance, product validation, and batch traceability.
- Academic and Industrial Research: Researchers use the guide to compare scaffold performance, evaluate new biomaterials, and ensure data quality in publications.
Related Standards
The guide references a number of related ASTM standards that support and complement its recommendations:
- ASTM F1854: Test Method for Stereological Evaluation of Porous Coatings on Medical Implants
- ASTM F1877: Practice for Characterization of Particles
- ASTM F2150: Guide for Characterization and Testing of Biomaterial Scaffolds Used in Regenerative Medicine and Tissue-Engineered Medical Products
- ASTM F2450: Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products
- ASTM E1919: Guide for Worldwide Published Standards Relating to Particle and Spray Characterization (withdrawn)
- ASTM E2245: Test Method for Residual Strain Measurements of Thin, Reflecting Films Using an Optical Interferometer
Summary
ASTM F2603-06(2020) provides users with a robust, standardized framework for obtaining reliable, quantifiable data from images of polymeric tissue scaffolds. Whether for quality control, research, or product development in the biomedical sector, its application ensures consistency, accuracy, and traceability across the tissue engineering workflow. This standard is indispensable for professionals seeking to harness imaging to advance scaffold technology and regenerative medicine.
Keywords: ASTM F2603, tissue scaffold imaging, polymeric scaffolds, image analysis, biomedical engineering, stereological analysis, scaffold characterization, regenerative medicine standards.
Buy Documents
ASTM F2603-06(2020) - Standard Guide for Interpreting Images of Polymeric Tissue Scaffolds
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2603-06(2020) is a guide published by ASTM International. Its full title is "Standard Guide for Interpreting Images of Polymeric Tissue Scaffolds". This standard covers: SIGNIFICANCE AND USE 4.1 This document provides guidance for users who wish to obtain quantifiable data from images of tissue scaffolds manufactured from polymers that include both high water content gels and woven textiles. 4.2 Information derived from tissue scaffold images can be used to optimize the structural characteristics of the matrix for a particular application, to develop better manufacturing procedures or to provide a measure of quality assurance and product traceability. Fig. 1 provides a summary of the key stages of image capture and analysis. FIG. 1 Key Stages in Image Capture, Storage, and Analysis 4.3 There is a synergy between the analysis of pores in tissue scaffolds and that of particles that is reflected in standards cited and in the analysis described in Section 9. Guide E1919 provides a compendium of standards for particle analysis that includes measurement techniques, data analytical and sampling methodologies. SCOPE 1.1 This guide covers the factors that need to be considered in obtaining and interpreting images of tissue scaffolds including technique selection, instrument resolution and image quality, quantification and sample preparation. 1.2 The information in this guide is intended to be applicable to porous polymer-based tissue scaffolds, including naturally derived materials such as collagen. However, some materials (both synthetic and natural) may require unique or varied sample preparation methods that are not specifically covered in this guide. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This document provides guidance for users who wish to obtain quantifiable data from images of tissue scaffolds manufactured from polymers that include both high water content gels and woven textiles. 4.2 Information derived from tissue scaffold images can be used to optimize the structural characteristics of the matrix for a particular application, to develop better manufacturing procedures or to provide a measure of quality assurance and product traceability. Fig. 1 provides a summary of the key stages of image capture and analysis. FIG. 1 Key Stages in Image Capture, Storage, and Analysis 4.3 There is a synergy between the analysis of pores in tissue scaffolds and that of particles that is reflected in standards cited and in the analysis described in Section 9. Guide E1919 provides a compendium of standards for particle analysis that includes measurement techniques, data analytical and sampling methodologies. SCOPE 1.1 This guide covers the factors that need to be considered in obtaining and interpreting images of tissue scaffolds including technique selection, instrument resolution and image quality, quantification and sample preparation. 1.2 The information in this guide is intended to be applicable to porous polymer-based tissue scaffolds, including naturally derived materials such as collagen. However, some materials (both synthetic and natural) may require unique or varied sample preparation methods that are not specifically covered in this guide. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2603-06(2020) is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics; 37.040.99 - Other standards related to photography. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2603-06(2020) has the following relationships with other standards: It is inter standard links to ASTM F2603-06(2012), ASTM F2150-19, ASTM F2450-18, ASTM E2245-11(2018), ASTM F1877-16, ASTM F1854-15, ASTM F2150-13, ASTM E2245-11, ASTM E2245-11e1, ASTM F1877-05(2010), ASTM F2450-10, ASTM E1919-09, ASTM F1854-09, ASTM F2450-09, ASTM F2150-07. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2603-06(2020) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2603 − 06 (Reapproved 2020)
Standard Guide for
Interpreting Images of Polymeric Tissue Scaffolds
This standard is issued under the fixed designation F2603; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F1854 Test Method for Stereological Evaluation of Porous
Coatings on Medical Implants
1.1 This guide covers the factors that need to be considered
F1877 Practice for Characterization of Particles
in obtaining and interpreting images of tissue scaffolds includ-
F2150 Guide for Characterization and Testing of Biomate-
ing technique selection, instrument resolution and image
rial Scaffolds Used in Regenerative Medicine and Tissue-
quality, quantification and sample preparation.
Engineered Medical Products
1.2 The information in this guide is intended to be appli-
F2450 Guide for Assessing Microstructure of Polymeric
cable to porous polymer-based tissue scaffolds, including
Scaffolds for Use in Tissue-Engineered Medical Products
naturally derived materials such as collagen. However, some
materials (both synthetic and natural) may require unique or
3. Terminology
varied sample preparation methods that are not specifically
3.1 Definitions:
covered in this guide.
3.1.1 aliasing, n—artifactual data that originates from an
1.3 The values stated in SI units are to be regarded as
insufficient sampling rate.
standard. No other units of measurement are included in this
3.1.2 biomaterial, n—a natural or synthetic material that is
standard.
suitable for introduction into living tissue especially as part of
1.4 This standard does not purport to address all of the
a medical device, such as an artificial heart valve or joint.
safety concerns, if any, associated with its use. It is the
3.1.3 blind (end) pore, n—a pore that is in contact with an
responsibility of the user of this standard to establish appro-
exposedinternalwallorsurfacethroughasingleorificesmaller
priate safety, health, and environmental practices and deter-
than the pore’s depth.
mine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accor- 3.1.4 closed cell, n—void within a solid, lacking any con-
nectivity with an external surface. Synonym: closed pore.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
3.1.5 feret diameter, n—the mean value of the distance
Development of International Standards, Guides and Recom-
between pairs of parallel tangents to the periphery of a pore
mendations issued by the World Trade Organization Technical
(adapted from Practice F1877).
Barriers to Trade (TBT) Committee.
3.1.6 hydrogel, n—a water-based open network of polymer
chains that are cross-linked either chemically or through
2. Referenced Documents
crystalline junctions or by specific ionic interactions.
2.1 ASTM Standards:
3.1.7 irregular, adj—an irregular pore that cannot be de-
E1919 GuideforWorldwidePublishedStandardsRelatingto
scribed as round or spherical. A set of reference figures that
Particle and Spray Characterization (Withdrawn 2014)
define the nomenclature are given in Appendix X2. (Adapted
E2245 Test Method for Residual Strain Measurements of
from Practice F1877).
Thin, Reflecting Films Using an Optical Interferometer
3.1.8 Nyquist criterion—a criterion that states that a signal
must be sampled at a rate greater than or equal to twice its
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
Surgical Materials and Devicesand is the direct responsibility of Subcommittee highest frequency component to avoid aliasing.
F04.42 on Biomaterials and Biomolecules for TEMPs.
3.1.9 permeability, n—a measure of fluid, particle, or gas
Current edition approved Aug. 1, 2020. Published August 2020. Originally
flow through an open pore structure.
approved in 2006. Last previous edition approved in 2012 as F2603 – 06 (2012).
DOI: 10.1520/F2603-06R20.
3.1.10 pixel, n—two-dimensional picture element.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
3.1.11 polymer, n—a long chain molecule composed of
Standards volume information, refer to the standard’s Document Summary page on
monomers.
the ASTM website.
3.1.11.1 Discussion—A polymer may be a natural or syn-
The last approved version of this historical standard is referenced on
www.astm.org. thetic material.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2603 − 06 (2020)
3.1.11.2 Discussion—Examples of polymers include colla-
gen and polycaprolactone.
3.1.12 pore, n—a liquid, fluid, or gas-filled externally con-
necting channel, void, or open space within an otherwise solid
or gelatinous material (for example, textile meshes composed
of many or single fibers (textile-based scaffolds), open cell
foams, (hydrogels). Synonyms: open pore, through pore.
3.1.13 porosity, n—property of a solid which contains an
inherent or induced network of channels and open spaces.
Porosity can be determined by measuring the ratio of pore
(void) volume to the apparent (total) volume of a porous
material and is commonly expressed as a percentage (Guide
F2150).
3.1.14 rectangular, adj—A pore that approximates a square
or rectangle in shape (derived from Practice F1877).
3.1.15 roundness (R), n—a measure of how closely an
object represents a circle (Practice F1877).
3.1.16 scaffold, n—a support, delivery vehicle, or matrix for
facilitating the migration, binding, or transport of cells or
bioactive molecules used to replace, repair, or regenerate
tissues. (Guide F2150).
FIG. 1 Key Stages in Image Capture, Storage, and Analysis
3.1.17 segmentation, n—a methodology for distinguishing
different regions (for example, pores and walls) within a tissue
analysis that includes measurement techniques, data analytical
scaffold image.
and sampling methodologies.
3.1.18 spherical pore, n—a pore with a generally spherical
5. Measurement Objectives
shape.
3.1.18.1 Discussion—A spherical pore appears round in a
5.1 Much of the research activity in tissue engineering is
photograph (Practice F1877).
focusedonthedevelopmentofsuitablematerialsandstructures
for optimal growth of a range of tissue types including
3.1.19 threshold, n—isolation of a range of grayscale values
cartilage, bone, and nerve. This requires a quantitative assess-
exhibited by one constituent within an image.
ment of the scaffold structure.
3.1.20 through pores, n—an inherent or induced network of
The key parameters that need to be determined are (1) the
voids or channels that permit flow of fluid from one side of the
overall level of porosity, (2) the pore size distribution, which
structure to the other.
can range from tens of nanometers to several hundred
3.1.21 tortuosity, n—a measure of the mean free path length
micrometres, and (3) the degree of interconnectivity and
of through pores relative to the sample thickness. Alternative
tortuosity of the pores.
definition: The squared ratio of the mean free path to the
6. Imaging Methods and Conditions
minimum possible path length.
6.1 There are many experimental ways of obtaining key
3.1.22 voxel, n—three-dimensional picture element.
scaffold physical parameters as described in Guide F2450.
When imaging and subsequent quantitative analysis is chosen
4. Significance and Use
as the method for determining these parameters, it is critical
4.1 This document provides guidance for users who wish to
that any image under consideration be a true representation of
obtain quantifiable data from images of tissue scaffolds manu-
the scaffold of interest. Some imaging methods require sample
factured from polymers that include both high water content
preparation.Somedonot.Whensamplepreparationisrequired
gels and woven textiles.
prior to imaging, care must be taken that the procedures do not
4.2 Information derived from tissue scaffold images can be
significantly alter the morphology of the scaffold. See Appen-
used to optimize the structural characteristics of the matrix for dix X1 for further information on sample preparation.
a particular application, to develop better manufacturing pro-
6.2 Images obtained using techniques such as light
cedures or to provide a measure of quality assurance and
microscopy, electron microscopy, and magnetic resonance
product traceability. Fig. 1 provides a summary of the key
imaging are two-dimensional (2-D) representations of a three-
stages of image capture and analysis.
dimensional (3-D) structure. These can be a planar or cross-
4.3 There is a synergy between the analysis of pores in sectional view with a relatively large depth of field or a series
tissue scaffolds and that of particles that is reflected in of physical or virtual 2-D slices, each with a small depth of
standards cited and in the analysis described in Section 9. field, that can be reassembled in a virtual environment to
Guide E1919 provides a compendium of standards for particle produce a 3-D mesostructure.
F2603 − 06 (2020)
6.3 There are limits to the extent an image (2-D or 3-D) can optical microscopy (1-3), optical coherence microscopy (4),
faithfully represent the physical artifacts that are influenced by MRI (5), and electron microscopies (6).
factors germane to the imaging method, such as spatial
6.6 The reconstruction of the mesostructure in 3-D from a
resolution and dynamic range, image contrast, and the signal-
series of 2-D images obtained from a sample that has been
to-noiseratio.Table1listssomeofthetechniquesavailablefor
physically sectioned requires considerably more effort than
producing images of porous structures, along with their con-
assembly of virtual sections produced by techniques that are
trast source, maximum demonstrated spatial resolution, and
able to focus on a plane within the sample. The virtual
typical dynamic range. Proper technique selection depends
approach is also less prone to sample distortion since it
both on the material properties of the scaffold (that is, optical
obviatestheneedforphysicalsectioningandregistrationerrors
methods cannot be used with opaque materials) the contrast
in the reassembly process. However, the techniques used to
available, and the target pore size range.
generate virtual 2-D images typically have limited penetration
6.4 The images generated by the techniques shown in Table depth.
1 cannot reproduce features smaller than the spatial resolution
6.7 Confocal microscopy (OCT), for example, has a pen-
of the method. Features that are faint, that is, those that do not
etration depth of approximately 100 µm, a value that depends
have significant contrast, or signal significantly above
on the wavelength of the light used and the amount of
background, will be resolved at length scales larger than the
scattering that occurs within the sample. Scanning acoustic
maximum resolution. Excessive contrast can also limit the
microscopy (SAM) can extend the penetration depth to ap-
penetration depth due to scattering effects. This is particularly
proximately 1 mm in polymer scaffolds albeit with a reduction
true of optical microscopies using differences in refractive
in image resolution.
index as the contrast mechanism. An appropriate level of
6.8 In general, using longer wavelength radiation to im-
contrast that can be established by experimentation is therefore
prove penetration of the radiation is accompanied by a reduc-
critical to high quality imaging.
tion in resolution.
6.5 Contrast can be enhanced by using exogenous agents,
such as florescence tags in optical microscopy and stains
7. Image Capture and Storage
containing heavy metal complexes in electron microscopies.
7.1 Image acquisition in this guide refers to the process of
Excessive contrast can be ameliorated in optical microscopies
capturing an image through digitization that is then stored for
by imbibing the structure with a fluid that has an index of
subsequent analysis. Care should be taken during this stage to
refraction similar to that of the solid making up the structure
avoid loss of fidelity by controllable factors that are not related
(this is termed “index-matching”). There are many excellent
to the methodology used to produce the image. These factors
resources describing factors influencing widefield and confocal
include the spatial sampling frequency of the detector system,
the dynamic range of analogue to digital (A/D) conversion,
segmenting (thresholding) operations (discussed in Section 9),
and both image compression and decompression.
7.2 Spatial sampling frequency and appropriate A/D con-
TABLE 1 Sources of Contrast and Techniques to Generate version are straightforward issues; the sampling frequency
Images of Tissue Scaffolds
should be at least twice the inverse spatial resolution, so as to
Generic method Contrast source Maximum resolution Physical slicing
fulfill the Nyquist criterion. Sampling at frequencies below this
(lateral/axial) required for 3D
will lead to the display of artifacts. Most image processing
imaging?
systems have anti-aliasing filters that remove frequencies
Widefield Optical Refractive index 1 µm/(10 µm) Y
greater than F /2 Hz, where F is the digital sampling rate. The
Microscopy Fluorescence
s s
Absorbance
A/D conversion should utilize a sufficient number of bits to
Confocal Optical Refractive index 0.5 µm/1 µm N
cover the dynamic range of the imaging / detector system.
Microscopy Fluorescence
Eight-bit conversion and recording is used for most common
Absorbance
Optical Coherence Refractive index 1 µm/1 µm N
imaging applications, resulting in images with 256 grayscale
Tomography (or
levels, where 0 corresponds to pure black and 255 to pure
Microscopy)
white respectively. If 8-bit conversion is used in a color (RGB)
Scanning Acoustic Acoustic 0.1 µm/0.1 µm N
Microscopy (SAM) impedance (depending on the
image there are 256 possible color combinations.
wavelength chosen)
Magnetic Nuclear spin 10 µm/10 µm N
NOTE 1—The gamut, or range of the grayscale reflects the image
Resonance
contrast.
Imaging (MRI)
X-ray Micro- Electron density 10 µm/10 µm N 7.3 It is important to record the minimum measurement
Computed
value (that is, the dimensions of a single pixel) when using
Tomography
digital capture or digitizing film-based images at all magnifi-
(µ-CT)
Transmission Electron density Approximately 0.2 Y
cations used in measurements (Test Method F1854).
Electron nm in plane
Microscopy (TEM)
Scanning Electron Electron density Approximately 10 nm N
Microscopy (SEM) The boldface numbers in parentheses refer to a list of references at the end of
this standard.
F2603 − 06 (2020)
7.4 Image compression is used to facilitate rapid display of
data and easy file transmission. However, many compression
methods (JPEG, PNG, and GIF) cause a loss of data. This loss
generally occurs in the high-frequency components of the
spatial Fourier spectrum of the image, leading to an oscillating,
smearedgrayscale,orcolorintensityprofileattheobjectedges.
Some proprietary compression methods are purported to in-
volve no loss of information and thus be completely reversible
(7). There are excellent internet resources describing compres-
sion and decompression techniques (7).
7.5 After capture, an image can be manipulated to facilitate
subsequent analysis. Such transformations include noise
suppression, enhancement of regions that are of particular
interest, and corrections that compensate for instrument or
experimental limitations.
FIG. 2 Low-Contrast Micrograph of Polycaprolactone PCL Scaf-
7.6 Noise Suppression—The minor random fluctuations in fold
signalintensityornoisethatarepresentindigitizedimagescan
degrade the quality of the image if the contrast between the
materialandbackgroundislow.Averaginganumberofframes,
N, can significantly reduce noise levels. The invariant signal
intensity increases with respect to N whilst the intensity of the
random noise only increases by the square root of N. The
signal-to-noise ratio, Sn, therefore scales as N/√N.
7.7 Background Correction—Lack of homogeneity of the
illuminatingsourcecanoccuracrossasampleduetothesource
itself or a nonlinear response of the detector. This can be
compensated for by subtracting a background captured in the
absence of a sample from the image.
7.8 Spatial-domain Filtering refers to enhancement of de-
siredfeaturesinanimageaccomplishedthroughworkinginthe
spatial domain. For example, in mask mode radiography, an
NOTE 1—Corresponding to image shown in Fig. 2.
X-ray image is obtained of an area before and after a contrast
FIG. 3 Bimodal Histogram of Grayscale Intensities
agent is added. The former image is subtracted from the latter,
and the result is an image with the feature of interest amplified
over the background. Other examples of spatial domain filter-
criterion for segmenting those regions of the image that
ing include opening and closing, which simply add or subtract
correspond to the solid, or ‘wall’ and the void, or ‘pore’
a pixel width to the periphery of a defined area. By cycling
respectively.
opening and closing operations, features can be enhanced or
8.2 There are many methods for segmenting image areas
noise near an interface can be eliminated.
belonging to pore and solid (8). The most straightforward
7.9 Frequency-domain Filtering:
approach is to define a grayscale threshold, in which the solid
7.9.1 In frequency-domain filtering, filtering operations are
appears as nearly white. Grayscale values below the threshold
performed on the 2-D or 3-D Fourier or similar transformation
are assigned to represent pores, and all others to represent the
of an image plane. The data is subsequently transformed back
wa
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...