ASTM E2469-16
(Test Method)Standard Test Method for Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion Chromatography
Standard Test Method for Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion Chromatography
SIGNIFICANCE AND USE
4.1 This test method provides for the quantitative determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG) and triethylene glycol (TEG) using ion chromatography with conductivity detection. The analysis time is less than 5 min with little or no sample preparation required. Conductivity detection is a universal detection mode and is linear over the range of the method. Acceptable levels of chloride in polyester-grade and low-conductivity-grade MEG vary with the manufacturer’s specifications but are normally in the low mg/kg range. Knowledge of the chloride content in polyester-grade and low-conductivity-grade MEG is required to establish whether the MEG product meets specification requirements.
4.2 Glycols have high viscosities and a dilution with high quality deionized water may be required depending on the capability of the autosampler, if used, to deliver the injection. All standards and samples, whether diluted or not should be treated in the same manner.
SCOPE
1.1 This test method covers the determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG) and triethylene glycol (TEG) in the range of 0.01 to 1.0 mg/kg by ion chromatography (IC).
1.2 Ethylene glycol can be analyzed directly by this test method without any sample preparation or diluted with high quality deionized water if an autosampler is used and dilution is necessary (that is, 50:50 or other suitable ratio).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 The exception is the additional information of (psi) in 9.3.3, 11.1.1, and 11.2.1.
1.4 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures and safety precautions.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and to determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2015
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.14 - Alcohols & Glycols
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 15-Oct-2008
- Effective Date
- 01-Nov-2007
- Effective Date
- 01-Nov-2007
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Dec-2000
Overview
ASTM E2469-16: Standard Test Method for Chloride in Mono-, Di-, and Tri-ethylene Glycol by Ion Chromatography provides a reliable and efficient procedure for the quantitative determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG), and triethylene glycol (TEG) using ion chromatography (IC) with conductivity detection. This method is essential for industries that require tight control of chloride content to meet stringent product specifications, particularly in polyester-grade and low-conductivity-grade glycol products.
Key highlights of the method include:
- A measurement range of 0.01 to 1.0 mg/kg chloride
- Analysis completed in less than five minutes
- Minimal sample preparation, with direct or diluted analysis based on the capabilities of the autosampler
- Applicability to a range of glycol grades vital for ensuring product quality and compliance
Key Topics
- Chloride Determination in Glycols: Quantitative assessment for MEG, DEG, and TEG is crucial for maintaining product integrity, especially for applications sensitive to inorganic ion contamination.
- Ion Chromatography with Conductivity Detection: Offers precise, linear response across the specified measurement range and supports high throughput with minimal manual intervention.
- Sample Preparation: Direct injection is possible for high-performance systems, while dilution with high-quality deionized water is recommended for viscous samples or when using an autosampler.
- Quality Assurance: Includes recommendations for calibration, use of certified reference materials, and routine verification of system performance.
- Interferences and Precautions: Addresses potential coeluting ions and the importance of contamination-free reagents, glassware, and apparatus to avoid false positives or negatives.
- Safety Considerations: Emphasizes adherence to applicable safety and health regulations as well as proper handling of glycols and reagents.
Applications
This standard test method has broad practical value in both industrial and research settings, including:
- Polyester Manufacturing: Ensures MEG is within stringent chloride content limits required for high-quality polyester fiber and resin production.
- Chemical Processing: Controls chloride impurities in DEG and TEG, which are used as solvents, plasticizers, and humectants in a variety of industrial processes.
- Purity Assessment for Electronic and Low-Conductivity Applications: Essential for verifying the suitability of glycols in applications where electrical conductivity and ionic contamination must be minimized.
- Quality Control and Regulatory Compliance: Supports manufacturers and laboratories in demonstrating robust analytical performance and adherence to product specifications.
Related Standards
ASTM E2469-16 aligns with and often references other important standards, including:
- ASTM D1193: Specification for Reagent Water, crucial for preparing dilution water and reagents free from chloride contamination.
- ASTM D6299: Practice for Applying Statistical Quality Assurance and Control Charting Techniques to Evaluate Analytical Measurement System Performance, referenced for ongoing performance verification.
- ASTM E180 (Withdrawn): Previously provided guidance on determining the precision of ASTM methods for analysis and testing of industrial and specialty chemicals.
- ASTM E300: Practice for Sampling Industrial Chemicals, relevant for obtaining representative samples for analysis.
By ensuring accurate chloride determination in ethylene glycols, ASTM E2469-16 helps maintain the high standards required across the chemicals, plastics, and electronics industries, where purity is paramount for both safety and product performance.
Keywords: ASTM E2469, chloride determination, monoethylene glycol, diethylene glycol, triethylene glycol, ion chromatography, conductivity detection, glycol purity, polyester-grade MEG, industrial standards, quality control, ASTM standards.
Buy Documents
ASTM E2469-16 - Standard Test Method for Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion Chromatography
REDLINE ASTM E2469-16 - Standard Test Method for Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E2469-16 is a standard published by ASTM International. Its full title is "Standard Test Method for Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 4.1 This test method provides for the quantitative determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG) and triethylene glycol (TEG) using ion chromatography with conductivity detection. The analysis time is less than 5 min with little or no sample preparation required. Conductivity detection is a universal detection mode and is linear over the range of the method. Acceptable levels of chloride in polyester-grade and low-conductivity-grade MEG vary with the manufacturer’s specifications but are normally in the low mg/kg range. Knowledge of the chloride content in polyester-grade and low-conductivity-grade MEG is required to establish whether the MEG product meets specification requirements. 4.2 Glycols have high viscosities and a dilution with high quality deionized water may be required depending on the capability of the autosampler, if used, to deliver the injection. All standards and samples, whether diluted or not should be treated in the same manner. SCOPE 1.1 This test method covers the determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG) and triethylene glycol (TEG) in the range of 0.01 to 1.0 mg/kg by ion chromatography (IC). 1.2 Ethylene glycol can be analyzed directly by this test method without any sample preparation or diluted with high quality deionized water if an autosampler is used and dilution is necessary (that is, 50:50 or other suitable ratio). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 The exception is the additional information of (psi) in 9.3.3, 11.1.1, and 11.2.1. 1.4 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures and safety precautions. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and to determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
SIGNIFICANCE AND USE 4.1 This test method provides for the quantitative determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG) and triethylene glycol (TEG) using ion chromatography with conductivity detection. The analysis time is less than 5 min with little or no sample preparation required. Conductivity detection is a universal detection mode and is linear over the range of the method. Acceptable levels of chloride in polyester-grade and low-conductivity-grade MEG vary with the manufacturer’s specifications but are normally in the low mg/kg range. Knowledge of the chloride content in polyester-grade and low-conductivity-grade MEG is required to establish whether the MEG product meets specification requirements. 4.2 Glycols have high viscosities and a dilution with high quality deionized water may be required depending on the capability of the autosampler, if used, to deliver the injection. All standards and samples, whether diluted or not should be treated in the same manner. SCOPE 1.1 This test method covers the determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene glycol (DEG) and triethylene glycol (TEG) in the range of 0.01 to 1.0 mg/kg by ion chromatography (IC). 1.2 Ethylene glycol can be analyzed directly by this test method without any sample preparation or diluted with high quality deionized water if an autosampler is used and dilution is necessary (that is, 50:50 or other suitable ratio). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 The exception is the additional information of (psi) in 9.3.3, 11.1.1, and 11.2.1. 1.4 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures and safety precautions. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and to determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
ASTM E2469-16 is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2469-16 has the following relationships with other standards: It is inter standard links to ASTM D6299-23a, ASTM D6299-17b, ASTM D6299-17a, ASTM D6299-17, ASTM D6299-13e1, ASTM D6299-10, ASTM D6299-10e2, ASTM D6299-09, ASTM D6299-08, ASTM D6299-07, ASTM D6299-07e1, ASTM D1193-06, ASTM D6299-02e1, ASTM D6299-02, ASTM D6299-00. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2469-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2469 − 16
Standard Test Method for
Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion
Chromatography
This standard is issued under the fixed designation E2469; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method covers the determination of inorganic
D1193 Specification for Reagent Water
chloride (chloride ion) in monoethylene glycol (MEG), dieth-
D6299 Practice for Applying Statistical Quality Assurance
ylene glycol (DEG) and triethylene glycol (TEG) in the range
and Control Charting Techniques to Evaluate Analytical
of 0.01 to 1.0 mg/kg by ion chromatography (IC).
Measurement System Performance
1.2 Ethylene glycol can be analyzed directly by this test
E180 Practice for Determining the Precision of ASTM
method without any sample preparation or diluted with high
Methods for Analysis and Testing of Industrial and Spe-
quality deionized water if an autosampler is used and dilution
cialty Chemicals (Withdrawn 2009)
is necessary (that is, 50:50 or other suitable ratio).
E300 Practice for Sampling Industrial Chemicals
1.3 The values stated in SI units are to be regarded as
3. Summary of Test Method
standard. No other units of measurement are included in this
3.1 An aliquot of the glycol sample is injected directly
standard.
(manually) or diluted (via autosampler) into an ion chromato-
1.3.1 The exception is the additional information of (psi) in
graph consisting of an injector with a fixed sample loop, two
9.3.3, 11.1.1, and 11.2.1.
anion exchange columns (guard and separator column), an
anion suppressor and a conductivity detector. Ions are sepa-
1.4 Review the current Safety Data Sheets (SDS) for de-
rated based on their affinity for the ion exchange sites of the
tailed information concerning toxicity, first-aid procedures and
resin with respect to the resin’s affinity for the eluent. The
safety precautions.
suppressor increases the sensitivity of the test method by both
1.5 This standard does not purport to address all of the
increasing the conductivity of the analytes and decreasing the
safety concerns, if any, associated with its use. It is the
conductivity of the eluent. The suppressor converts the eluent
responsibility of the user of this standard to establish appro-
and the analytes to the corresponding hydrogen form acids.
priate safety, health, and environmental practices and to
The chloride is detected by conductivity detection and identi-
determine the applicability of regulatory limitations prior to
fied by retention time. Quantitation is by peak area using an
use. For specific hazard statements, see Section 9.
external standard calibration curve. Instructions are provided
1.6 This international standard was developed in accor- for two equivalent IC systems.
dance with internationally recognized principles on standard-
4. Significance and Use
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
4.1 This test method provides for the quantitative determi-
mendations issued by the World Trade Organization Technical nation of inorganic chloride (chloride ion) in monoethylene
Barriers to Trade (TBT) Committee.
glycol (MEG), diethylene glycol (DEG) and triethylene glycol
(TEG) using ion chromatography with conductivity detection.
The analysis time is less than 5 min with little or no sample
preparation required. Conductivity detection is a universal
detection mode and is linear over the range of the method.
1 2
This test method is under the jurisdiction of ASTM Committee D16 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi- contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
bility of Subcommittee D16.14 on Alcohols & Glycols. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Jan. 1, 2016. Published March 2016. Originally the ASTM website.
approved in 2006. Last previous edition approved in 2008 as E2469 – 08a. DOI: The last approved version of this historical standard is referenced on
10.1520/E2469-16. www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2469 − 16
Acceptable levels of chloride in polyester-grade and low- analytical history of the product, the data should be confirmed
conductivity-grade MEG vary with the manufacturer’s speci- by an alternate test method.
fications but are normally in the low mg/kg range. Knowledge
of the chloride content in polyester-grade and low- 6. Apparatus
conductivity-grade MEG is required to establish whether the
6.1 Analytical Balance, capable of weighing 200 g to the
MEG product meets specification requirements.
nearest 0.0001 g. (See Note 1.)
4.2 Glycols have high viscosities and a dilution with high
6.2 Pipettes, capable of measuring from 100-µL to 10-mL.
quality deionized water may be required depending on the
(See Note 1.)
capability of the autosampler, if used, to deliver the injection.
NOTE1—Theaccuracyofbalancesandpipettesshouldbeconfirmedon
All standards and samples, whether diluted or not should be
a regular basis and documentation of the check should be kept.
treated in the same manner.
6.3 Ion Chromatograph, Configuration A (Bottled Eluent
System - Carbonate-Based) (see Note 2)—Analytical instru-
5. Interferences
ment with all the required accessories including an eluent
5.1 The identification of chloride is based on retention time.
pump, temperature-controlled low volume (< 2 µL) conductiv-
Interferences can be caused by ionic substances with retention
ity cell, conductivity detector, PEEK tubing, and a PEEK
times similar to that of chloride. If the eluent conditions are
injection valve with a fixed sample loop. An auxiliary regen-
changed or the column capacity changes, it is possible that
erant pump or pressurized 4-L reagent bottle is required for
other anions may coelute with chloride and cause an interfer-
external regenerant delivery. Autosampler (optional). The in-
ence.
strument must be suitable for analysis according to the oper-
ating conditions given in 11.1.
5.2 Any anion that elutes after chloride under the analysis
conditions used in the test method may cause an interference if
NOTE 2—The ion chromatograph (Configuration A) uses a carbonate
the run time of the method isn’t long enough to include that
based eluent system in which the eluent is prepared by the analyst from
anion.Whentheruntimeofamethodistooshort,alateeluting analytical grade reagents or commercially available concentrated carbon-
ate solutions. There is more variability in the retention time of chloride
anion from one analysis may be detected during the next
withthistypeofsystemasaresultofvariationsintheconcentrationofthe
analysis and cause an interference if it elutes at the same time
eluentpreparedbyanalysts.ThisistheoldestandmostcommonlyusedIC
as chloride. Carbonate, if present in a sample, may cause an
system.
interference in IC systems using hydroxide eluent (like Con-
6.3.1 AnionExchangeGuardColumn(forCarbonate-Based
figuration B) if the run time of the analysis is not long enough
Eluent), for protection of the analytical column from strongly
to include the carbonate peak. The elution time of carbonate
retained components and organics. Better separations are
under the analysis conditions used in this test method is
obtained with the additional plates of the guard column.
directly related to the amount of carbonate in the sample. The
6.3.2 Anion Exchange Separator Column (for Carbonate-
highertheconcentrationofcarbonateinasample,thefasterthe
Based Eluent), capable of producing separation of the chloride
carbonate elutes from the column.
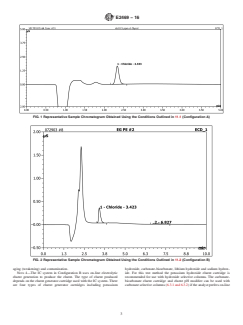
equivalent to or better than that shown in Fig. 1.
5.3 Method interferences can be caused by the contamina-
6.3.3 Anion Suppressor, an electrolytic self-regenerating
tion of glassware, eluent and reagents with chloride. Care must
membrane suppressor, micromembrane suppressor or equiva-
betakentoensureglasswareandapparatusarefreeofchloride.
lent suppressor capable of lowering the background conduc-
The use of latex gloves is recommended to prevent chloride
tance of the eluant to a level that allows the method detection
contamination while handling samples and reagents.
limit to be achieved.
6.3.4 Chromatography Data System, for data acquisition
5.4 In an IC system with an electrolytic membrane based
and data processing.
suppressor operated in the recycle mode, the eluent is recycled
back through the suppressor after it exits the conductivity cell 6.4 Ion Chromatograph, Configuration B (Eluent Genera-
to provide a source of water for electrolytic generation of
tion System - Hydroxide Eluent) (see Note 3)—Analytical
hydronium ions for the regenerant. Using this system instrumentwithalltherequiredaccessoriesincludinganeluent
configuration, there is an interference caused by the glycol
pump, temperature controlled low volume (< 2 µL) conductiv-
sample as it passes back through the suppressor. This interfer- ity cell, conductivity detector, PEEK tubing, PEEK injection
ence appears as a large broad peak that upsets the baseline
valve with a fixed sample loop and electrolytic eluent genera-
during the time chloride elutes from the column. Accurate
tion module.An auxiliary regenerant pump or pressurized 4-L
quantitation of chloride is very difficult if not impossible with
reagent bottle is required for external regenerant delivery.
this interference present.To avoid this interference, an external
Autosampler (optional). The instrument must be suitable for
supplyofreagentwaterisusedasthesourceofhydroniumions
analysis according to the operating conditions given in 11.2.
for the regenerant. In the external water mode, water flows
NOTE 3—The IC system (Configuration B) uses on-line electrolytic
countercurrent to the eluent through the suppressor. The water
eluent generation to produce a hydroxide eluent. The hydroxide eluent is
can be pressurized or pumped through the suppressor to
generated from reagent water using an eluent generator cartridge. The
achieve the required flow rate. concentration of hydroxide eluent is very reproducible, so the retention
time for chloride is less variable than with a carbonate eluent. This newer
5.5 No other direct interferences have been observed in the
technology for eluent generation eliminates the variability of eluent
use of this test method. If results are suspect based on the preparation by an analyst. It also eliminates the problems with eluent
E2469 − 16
FIG. 1 Representative Sample Chromatogram Obtained Using the Conditions Outlined in 11.1 (Configuration A)
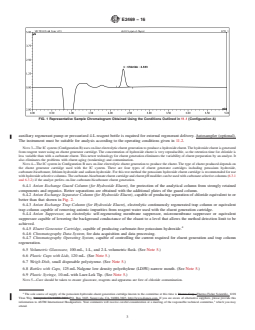
FIG. 2 Representative Sample Chromatogram Obtained Using the Conditions Outlined in 11.2 (Configuration B)
aging (weakening) and contamination. hydroxide, carbonate-bicarbonate, lithium hydroxide and sodium hydrox-
NOTE 4—The IC system in Configuration B uses on-line electrolytic ide. For this test method the potassium hydroxide eluent cartridge is
eluent generation to produce the eluent. The type of eluent produced recommended for use with hydroxide selective columns. The carbonate-
depends on the eluent generator cartridge used with the IC system. There bicarbonate eluent cartridge and eluent pH modifier can be used with
are four types of eluent generator cartridges including potassium carbonate selective columns (6.3.1 and 6.3.2) if the analyst prefers on-line
E2469 − 16
carbonate-bicarbonate eluent generation.
defined by Type II of Specification D1193. It is recommended
that all water be filtered through a 0.2-µm filter. For eluent
6.4.1 Anion Exchange Guard Column (for Hydroxide
preparation, it is recommended to degas the water by sparging
Eluent), for protection of the analytical column from strongly
with helium or vacuum degassing and sonication.
retained components and organics. Better separations are
obtained with the additional plates of the guard column.
7.3 Sodium Chloride, ACS reagent grade.
6.4.2 Anion Exchange Separator Column (for Hydroxide
7.4 Chloride Quality Assurance Check Standard, an
Eluent),capableofproducingseparationofchlorideequivalent
independent, certified 10 µg/mL chloride standard (not made
to or better than that shown in Fig. 2.
from sodium chloride in 7.3), may be purchased commercially.
6.4.3 Anion Exchange Trap Column (for Hydroxide Eluent),
7.5 Sodium Carbonate (Configuration A), ACS reagent
electrolyticcontinuouslyregeneratedtrapcolumnorequivalent
grade.
trap column capable of removing anionic impurities from
reagent water used with the eluent generation cartridge.
7.6 Sodium Bicarbonate (Configuration A), ACS reagent
6.4.4 Anion Suppressor, an electrolytic self-regenerating
grade.
membrane suppressor, micromembrane suppressor or equiva-
7.7 Sodium Carbonate Concentrate (Configuration A),
lent suppressor capable of lowering the background conduc-
0.5 M sodium carbonate, commercially available.
tance of the eluant to a level that allows the method detection
7.8 Sodium Bicarbonate Concentrate (Configuration A),
limit to be achieved.
0.5 M sodium bicarbonate, commercially available.
6.4.5 Eluent Generator Cartridge, capable of producing
carbonate-free potassium hydroxide.
7.9 Monoethylene Glycol (MEG) (High Purity), with low
6.4.6 Chromatography Data System, for data acquisition
concentrations of impurities.
and data processing.
7.10 Diethylene Glycol (DEG) (High Purity), with low
6.4.7 Chromatography Operating System, capable of con-
concentrations of impurities.
trolling the current required for eluent generation and trap
column regeneration. 7.11 Triethylene Glycol (TEG) (High Purity), with low
concentrations of impurities.
6.5 VolumetricGlassware,100-mL,1-L,and2-Lvolumetric
NOTE 6— Impurities in high-purity MEG, DEG or TEG used for
flask. (See Note 5.)
preparation of the chloride working standards or for sample dilution
shouldnotexceed0.01mg/kg.Thisinformationshouldbeprovidedbythe
6.6 Plastic Cups with Lids, 120 mL. (See Note 5.)
supplier or determined by the analyst before use.
6.7 Weigh Dish, small disposable polystyrene. (See Note 5.)
7.12 Sulfuric Acid (for use with anion micromembrane
6.8 Bottles with Caps, 125-mL Nalgene low density poly-
suppressor), concentrated sulfuric acid (95 to 98 %).
ethylene (LDPE) narrow mouth. (See Note 5.)
7.13 Anion Regenerant Concentrate (for use with anion
6.9 Plastic Syringe, 10-mL with Luer-Lok Tip. (See Note
micromembrane suppressor), 1 M sulfuric acid, commercially
5.)
available.
NOTE 5—Care should be taken to ensure glassware, reagents and
7.14 Isopropyl Alcohol (2-Propanol), ACS reagent grade.
apparatus are free of chloride contamination.
7.15 Methyl Alcohol (Methanol, alternate for Isopropyl
7. Reagents and Materials
Alcohol), ACS reagent grade.
7.1 Purity of Reagents—Reagent grade chemicals shall be
7.16 Monoethylene Glycol Quality Control Sample, polyes-
used in all tests. Unless otherwise indicated, it is intended that
ter grade MEG.
all reagents conform to the specifications of the Committee on
7.17 Diethylene Glycol Quality Control Sample.
Analytical Reagents of theAmerican Chemical Society (ACS)
where such specifications are available. Other grades may be
7.18 Triethylene Glycol Quality Control Sample.
used, provided it is first ascertained that the reagent is of
8. Reagent Solutions
sufficiently high purity to permit its use without lessening the
accuracy of the determination.
8.1 Chloride Stock Solution (1000 mg/kg Chloride in Wa-
ter):
7.2 High-Purity Water—Unless otherwise indicated, refer-
ences to water shall be understood to mean reagent water as
NOTE 7—As an alternative, a 1000 µg/mL chloride standard may be
purchased commercially.
NOTE 8—The density of the commercial chloride stock solution is
The sole source of supply of the potassium hydroxide eluent generation
assumed to be 1.0 g/mL at room temperature for wt/wt calculations in
cartridge known to the committee at this time is Thermo Fisher Scientific, 1228
mg/kg.
Titan Way, P.O. Box 3603, Sunnyvale, CA, 94088-3603, http://www.dionex.com. If
you are aware of alternative suppliers, please provide this information to ASTM
8.1.1 Place a 125-mL Nalgene LDPE narrow mouth bottle
International Headquarters. Your comments will receive careful consideration at a
1 on the balance and tare.
meeting of the responsible technical committee, which you may attend.
8.1.2 Weigh (and record to the nearest 0.0001 g) 0.1649 6
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
0.0010 g of sodium chloride (7.3) into the 125-mL bottle.
listed by the American Chemical Society, see Annual Standards for Laboratory
8.1.3 Diluteto100g(andrecordtothenearest0.001g)with
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
reagent water (7.2) and mix well. Prepare this calibration
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USP), Rockville,
MD. solution once a year.
E2469 − 16
TABLE 1 Chloride Working Standard
8.1.4 Calculate the concentration of chloride in the chloride
stock solution as follows: Chloride Weight of 10 mg/kg Final Weight of
Working Nominal Chloride Calibration Chloride Working
W 310 mg 3FW
NaCl Cl Standard # Concentration Solution (to add) Standard (MEG DEG or
C 5 (1)
CSS
TEG added)
W 31 kg 3FW
STD NaCl
mg/kg g (± 0.0001 g) g (± 0.001 g)
where:
1 0 0 100
2 0.01 0.1 100
C = the concentration (mg/kg) of chloride in the chlo-
CSS
3 0.02 0.2 100
ride stock solution,
4 0.05 0.5 100
W = weight (g) of sodium chloride added to the chlo-
5 0.1 1.0 100
NaCl
6 0.2 2.0 100
ride stock solution (8.1.2),
7 0.5 5.0 100
W = total weight (g) of the prepared chloride stock
STD
8 1.0 10.0 100
solution (8.1.3, nominally 100 g),
FW = formula weight of chloride (35.457 g/mol), and
Cl
FW = formula weight of sodium chloride (58.44 g/mol).
NaCl
8.2 Chloride Calibration Solution (10 mg/kg Chloride in
NOTE 11—Chloride working standards should be prepared a minimum
of every three months (monthly preparation is recommended). To avoid
Water):
contamination of the standard, always pour the working standard out of
NOTE9—Pipettesareusedtodispensetheestimatedamountofchloride
the bottle for use and never put anything (pipette and so forth) into the
stock solution (1000 mg/kg).
bottle. Minimize the number of times and length of time the working
standards are exposed to air. Cap securely after each use.
8.2.1 Place a 100-mL volumetric flask on the balance and
8.3.3 Determine the concentration (mg/kg) of chloride in
tare. Pipette (and record the weight to the nearest 0.0001 g)
each of the chloride working standards as follows:
1000 µL of 1000 mg/kg chloride stock solution (8.1) into the
100-mL volumetric flask.
C 3W
CS CS i
~ !
C 5 (3)
WS i
8.2.2 Dilute to 100 mL(and record the weight to the nearest ~ !
W
WS~i!
0.001 g) with reagent water (7.2) and mix well. Store the
where:
10 mg⁄kg chloride calibration solution in a 125-mL Nalgene
C = the concentration (mg/kg) of chloride in chloride
LDPE narrow mouth bottle. Prepare this calibration solution
WS(i)
once a year. working standard, i,
C = the concentration (mg/kg) of chloride in the chlo-
8.2.3 Determine the concentration (mg/kg) of chloride in CS
ride calibration solution (8.2),
the chloride calibration solution as follows:
W = weight (g) of chloride calibration solution added to
CS(i)
C 3W
CSS CSS
chloride working standard, i (8.3.1), and
C 5 (2)
CS
W
CS
W = final weight (g) of the prepared chloride working
WS(i)
standard, i (8.3.2, nominally 100 g).
where:
C = theconcentration(mg/kg)ofchlorideinthechloride
8.3.4 If utilizing an autosampler and dilution is required, it
CS
calibration solution,
is recommended to dilute standards with high purity deionized
C = theconcentration(mg/kg)ofchlorideinthechloride
water prior to calibration. Dilution of the standards should be
CSS
stock solution (8.1),
the same as dilution used for samples.Application of the same
W = weight (g) of chloride stock solution added to
CSS dilution parameter to the standards and samples will eliminate
chloride calibration solution (8.2.1), and
further calculations. Mix well to ensure a homogeneous solu-
W = final weight (g) of the prepared chloride calibration
CS
tion. Record the final weight of the chloride working standard
solution (8.2.2, nominally 100 g).
to the nearest 0.001 g.
8.3 Chloride Working Standards (0.01, 0.02, 0.05, 0.1, 0.2,
8.4 Anion Regenerant Solution (12.5 mM SulfuricAcid) (for
0.5, and 1.0 mg/kg in MEG, DEG, or TEG).
Use with Anion Micromembrane Suppressor):
NOTE 10—Pipettes are used to dispense the estimated amount of
NOTE 12—Consult instrument suppressor manufacturer for preparation
chloride calibration solution (10 mg/kg).
of the suppressor regenerate solution.
8.3.1 Prepare at least five chloride working standard solu-
8.4.1 Add approximately 1 L of reagent water to a 2-L
tions (8.3) covering the expected range for the glycol being volumetric flask. Slowly add 1.4 mL(or 2.6 g) of concentrated
analyzed. Choose the dilutions such that the resulting concen-
sulfuric acid (7.12) to the flask taking care to avoid splashing
trations are equally distributed over the range of interest. For and overheating. Bring to volume with reagent water and mix
each of the chloride working standards, place a 120-mLplastic
well. Prepare fresh regenerant solution at least once per month.
cup on the balance and tare. Using Table 1 for guidance, add 8.4.2 Alternative Preparation with Anion Regenerant
the appropriate amount of 10 mg/kg chloride calibration
Concentrate—Add 1 part of the anion regenerant concentrate
solution (8.2) and record the weight to the nearest 0.0001 g. (1 M sulfuric acid) (7.13) to 80 parts reagent water (for
8.3.2 Add the high purity MEG, DEG or TEG (7.9, 7.10 or example, add 25 mL of anion regenerant concentrate to a 2-L
7.11) to obtain the final weight of 100 g. Mix well to ensure a volumetric flask. Bring to volume with reagent water and mix
homogeneous solution. Record the final weight of the chloride well). Prepare fresh regenerant solution at least once per
working standard to the nearest 0.001 g. month.
E2469 − 16
8.5 Carbonate/Bicarbonate Eluent Solution (0.6 mM/ 9.3.1.3 Excessive force on the injection syringe (manual
0.4 mM) Prepared from Carbonate/Bicarbonate Salts (for use injection) can cause glycol to spray on the analyst. Wear
with Configuration A): gloves, chemical goggles, and lab coat or chemical-resistant
apron when injecting a sample. Use Luer-Lok syringes to load
NOTE 13—Consult with the anion analytical column manufacturer for
the sample into the injection port. Turn face away from the
recommended mobile phase concentration and preparation of the separa-
injection port while depressing the syringe plunger. Depress
tion (8.5 and 8.6).
syringe plunger slowly to avoid high back pressure as a result
8.5.1 Place a small plastic weigh dish on the balance and
of the viscosity of the glycol sample.
tare. Add 0.1250 6 0.0010 g of sodium carbonate (7.5)to
9.3.2 Electrical/Shock Hazard:
weigh dish.Transfer the sodium carbonate in the weigh dish to
9.3.2.1 Ion chromatographs (pump modules and conductiv-
a 2-L volumetric flask. Rinse the weigh dish into the volumet-
ity detectors) contain printed circuit boards and components
ric flask using a small amount of reagent water.
operating at dangerous voltages. The circuit boards and com-
8.5.2 Place a second small plastic weigh dish on the balance
ponents are covered with protective panels that are identified
and tare. Add 0.0625 6 0.0010 g of sodium bicarbonate (7.6)
by warning labels. Contact with these circuits boards and
totheweighdish.Transferthesodiumbicarbonateintheweigh
components can cause serious injury or painful electrical
dish to the 2-L volumetric flask used in 8.5.1. Rinse the weigh
shock. Turn off the system power and unplug the line cord
dish into the volumetric flask using a small amount of reagent
before removing the protective panels. After removing the
water.
protective panel in the pump or detector, wait 30 to 60 s before
8.5.3 Add reagent water to the 2-Lvolumetric flask to bring
touching any printed circuit boards to guarantee the discharge
ittovolumeandmixwell.Preparefresheluentsolutionatleast
of the capacitors.
once every two weeks.
9.3.2.2 To prevent damage to the pump circuitry and
8.6 Carbonate/Bicarbonate Eluent Solution (0.6 mM/ components,
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2469 − 08a E2469 − 16
Standard Test Method for
Chloride in Mono-, Di- and Tri-ethylene Glycol by Ion
Chromatography
This standard is issued under the fixed designation E2469; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of inorganic chloride (chloride ion) in monoethylene glycol (MEG), diethylene
glycol (DEG) and triethylene glycol (TEG) in the range of 0.01 to 1.0 mg/kg by ion chromatography (IC).
1.2 Ethylene glycol can be analyzed directly by this test method without any sample preparation or diluted with high quality
deionized water if an autosampler is used and dilution is necessary (that is, 50:50 or other suitable ratio).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 The exception is the additional information of (psi) in 9.3.3, 11.1.1, and 11.2.1.
1.4 Review the current Material Safety Data Sheets (MSDS)(SDS) for detailed information concerning toxicity, first-aid
procedures and safety precautions.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and to determine the applicability of regulatory
limitations prior to use. For specific hazard statements, see Section 9.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D6299 Practice for Applying Statistical Quality Assurance and Control Charting Techniques to Evaluate Analytical Measure-
ment System Performance
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
E300 Practice for Sampling Industrial Chemicals
3. Summary of Test Method
3.1 An aliquot of the glycol sample is injected directly (manually) or diluted (via autosampler) into an ion chromatograph
consisting of an injector with a fixed sample loop, two anion exchange columns (guard and separator column), an anion suppressor
and a conductivity detector. Ions are separated based on their affinity for the ion exchange sites of the resin with respect to the
resin’s affinity for the eluent. The suppressor increases the sensitivity of the test method by both increasing the conductivity of the
analytes and decreasing the conductivity of the eluent. The suppressor converts the eluent and the analytes to the corresponding
hydrogen form acids. The chloride is detected by conductivity detection and identified by retention time. Quantitation is by peak
area using an external standard calibration curve. Instructions are provided for two equivalent IC systems.
4. Significance and Use
4.1 This test method provides for the quantitative determination of inorganic chloride (chloride ion) in monoethylene glycol
(MEG), diethylene glycol (DEG) and triethylene glycol (TEG) using ion chromatography with conductivity detection. The analysis
time is less than 5 min with little or no sample preparation required. Conductivity detection is a universal detection mode and is
This test method is under the jurisdiction of ASTM Committee E15 on Industrial and Specialty Chemicals and is the direct responsibility of Subcommittee E15.02 on
Product Standards.
Current edition approved Dec. 15, 2008Jan. 1, 2016. Published January 2009March 2016. Originally approved in 2006. Last previous edition approved in 2008 as
E2469 – 08.E2469 – 08a. DOI: 10.1520/E2469-08A.10.1520/E2469-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2469 − 16
linear over the range of the method. Acceptable levels of chloride in polyester-grade and low-conductivity-grade MEG vary with
the manufacturer’s specifications but are normally in the low mg/kg range. Knowledge of the chloride content in polyester-grade
and low-conductivity-grade MEG is required to establish whether the MEG product meets specification requirements.
4.2 Glycols have high viscosities and a dilution with high quality deionized water may be required depending on the capability
of the autosampler, if used, to deliver the injection. All standards and samples, whether diluted or not should be treated in the same
manner.
5. Interferences
5.1 The identification of chloride is based on retention time. Interferences can be caused by ionic substances with retention times
similar to that of chloride. If the eluent conditions are changed or the column capacity changes, it is possible that other anions may
coelute with chloride and cause an interference.
5.2 Any anion that elutes after chloride under the analysis conditions used in the test method may cause an interference if the
run time of the method isn’t long enough to include that anion. When the run time of a method is too short, a late eluting anion
from one analysis may be detected during the next analysis and cause an interference if it elutes at the same time as chloride.
Carbonate, if present in a sample, may cause an interference in IC systems using hydroxide eluent (like Configuration B) if the
run time of the analysis is not long enough to include the carbonate peak. The elution time of carbonate under the analysis
conditions used in this test method is directly related to the amount of carbonate in the sample. The higher the concentration of
carbonate in a sample, the faster the carbonate elutes from the column.
5.3 Method interferences can be caused by the contamination of glassware, eluent and reagents with chloride. Care must be
taken to ensure glassware and apparatus are free of chloride. The use of latex gloves is recommended to prevent chloride
contamination while handling samples and reagents.
5.4 In an IC system with an electrolytic membrane based suppressor operated in the recycle mode, the eluent is recycled back
through the suppressor after it exits the conductivity cell to provide a source of water for electrolytic generation of hydronium ions
for the regenerant. Using this system configuration, there is an interference caused by the glycol sample as it passes back through
the suppressor. This interference appears as a large broad peak that upsets the baseline during the time chloride elutes from the
column. Accurate quantitation of chloride is very difficult if not impossible with this interference present. To avoid this
interference, an external supply of reagent water is used as the source of hydronium ions for the regenerant. In the external water
mode, water flows countercurrent to the eluent through the suppressor. The water can be pressurized or pumped through the
suppressor to achieve the required flow rate.
5.5 No other direct interferences have been observed in the use of this test method. If results are suspect based on the analytical
history of the product, the data should be confirmed by an alternate test method.
6. Apparatus
6.1 Analytical Balance, capable of weighing 200 g to the nearest 0.0001 g. (See Note 1.)
6.2 Pipettes, capable of measuring from 100-μL to 10-mL. (See Note 1.)
NOTE 1—The accuracy of balances and pipettes should be confirmed on a regular basis and documentation of the check should be kept.
6.3 Ion Chromatograph, Configuration A (Bottled Eluent System - Carbonate-Based) (see Note 2)—Analytical instrument with
all the required accessories including an eluent pump, temperature-controlled low volume (< 2 (< 2 μL) conductivity cell,
conductivity detector, PEEK tubing, and a PEEK injection valve with a fixed sample loop. An auxiliary regenerant pump or
pressurized 4-L reagent bottle is required for external regenerant delivery. Autosampler (optional). The instrument must be suitable
for analysis according to the operating conditions given in 11.1. Autosampler optional.
NOTE 2—The ion chromatographychromatograph (Configuration A) uses a carbonate based eluent system in which the eluent is prepared by the analyst
from analytical grade reagents or commercially available concentrated carbonate solutions. There is more variability in the retention time of chloride with
this type of system as a result of variations in the concentration of the eluent prepared by analysts. This is the oldest and most commonly used IC system.
6.3.1 Anion Exchange Guard Column (for Carbonate-Based Eluent), for protection of the analytical column from strongly
retained components and organics. Better separations are obtained with the additional plates of the guard column.
6.3.2 Anion Exchange Separator Column (for Carbonate-Based Eluent), capable of producing separation of the chloride
equivalent to or better than that shown in Fig. 1.
6.3.3 Anion Suppressor, an electrolytic self-regenerating membrane suppressor, micromembrane suppressor or equivalent
suppressor capable of lowering the background conductance of the eluant to a level that allows the method detection limit to be
achieved.
6.3.4 Chromatography Data System, for data acquisition and data processing.
6.4 Ion Chromatograph, Configuration B (Eluent Generation System - Hydroxide Eluent) (see Note 3)—Analytical instrument
with all the required accessories including an eluent pump, temperature controlled low volume (< 2 (< 2 μL) conductivity cell,
conductivity detector, PEEK tubing, PEEK injection valve with a fixed sample loop and electrolytic eluent generation module. An
E2469 − 16
FIG. 1 Representative Sample Chromatogram Obtained Using the Conditions Outlined in 11.1 (Configuration A)
auxiliary regenerant pump or pressurized 4-L reagent bottle is required for external regenerant delivery. Autosampler (optional).
The instrument must be suitable for analysis according to the operating conditions given in 11.2.
NOTE 3—The IC system (Configuration B) uses on-line electrolytic eluent generation to produce a hydroxide eluent. The hydroxide eluent is generated
from reagent water using an eluent generator cartridge. The concentration of hydroxide eluent is very reproducible, so the retention time for chloride is
less variable than with a carbonate eluent. This newer technology for eluent generation eliminates the variability of eluent preparation by an analyst. It
also eliminates the problems with eluent aging (weakening) and contamination.
NOTE 4—The IC system in Configuration B uses on-line electrolytic eluent generation to produce the eluent. The type of eluent produced depends on
the eluent generator cartridge used with the IC system. There are four types of eluent generator cartridges including potassium hydroxide,
carbonate-bicarbonate, lithium hydroxide and sodium hydroxide. For this test method the potassium hydroxide eluent cartridge is recommended for use
with hydroxide selective columns. The carbonate-bicarbonate eluent cartridge and eluent pH modifier can be used with carbonate selective columns (6.3.1
and 6.3.2) if the analyst prefers on-line carbonate-bicarbonate eluent generation.
6.4.1 Anion Exchange Guard Column (for Hydroxide Eluent), for protection of the analytical column from strongly retained
components and organics. Better separations are obtained with the additional plates of the guard column.
6.4.2 Anion Exchange Separator Column (for Hydroxide Eluent), capable of producing separation of chloride equivalent to or
better than that shown in Fig. 2.
6.4.3 Anion Exchange Trap Column (for Hydroxide Eluent), electrolytic continuously regenerated trap column or equivalent
trap column capable of removing anionic impurities from reagent water used with the eluent generation cartridge.
6.4.4 Anion Suppressor, an electrolytic self-regenerating membrane suppressor, micromembrane suppressor or equivalent
suppressor capable of lowering the background conductance of the eluant to a level that allows the method detection limit to be
achieved.
6.4.5 Eluent Generator Cartridge, capable of producing carbonate-free potassium hydroxide.
6.4.6 Chromatography Data System, for data acquisition and data processing.
6.4.7 Chromatography Operating System, capable of controlling the current required for eluent generation and trap column
regeneration.
6.5 Volumetric Glassware, 100-mL, 1-L, and 2-L volumetric flask. (See Note 5.)
6.6 Plastic Cups with Lids, 120 mL. (See Note 5.)
6.7 Weigh Dish, small disposable polystyrene. (See Note 5.)
6.8 Bottles with Caps, 125-mL Nalgene low density polyethylene (LDPE) narrow mouth. (See Note 5.)
6.9 Plastic Syringe, 10-mL with Luer-Lok Tip. (See Note 5.)
NOTE 5—Care should be taken to ensure glassware, reagents and apparatus are free of chloride contamination.
The sole source of supply of the potassium hydroxide eluent generation cartridge known to the committee at this time is Dionex Corp., Thermo Fisher Scientific, 1228
Titan Way, Sunnyvale CA 94088-3603. P.O. Box 3603, Sunnyvale, CA, 94088-3603, http://www.dionex.com. If you are aware of alternative suppliers, please provide this
information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee, which you may
attend.
E2469 − 16
FIG. 2 Representative Sample Chromatogram Obtained Using the Conditions Outlined in 11.2 (Configuration B)
7. Reagents and Materials
7.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society (ACS) where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
7.2 High-Purity Water—Unless otherwise indicated, references to water shall be understood to mean reagent water as defined
by Type II of Specification D1193. It is recommended that all water be filtered through a 0.2-μm filter. For eluent preparation, it
is recommended to degas the water by sparging with helium or vacuum degassing and sonication.
7.3 Sodium Chloride, ACS reagent grade.
7.4 Chloride Quality Assurance Check Standard, an independent, certified 10 μg/mL chloride standard (not made from sodium
chloride in 7.37.3),), may be purchased commercially.
7.5 Sodium Carbonate (Configuration A), ACS reagent grade.
7.6 Sodium Bicarbonate (Configuration A), ACS reagent grade.
7.7 Sodium Carbonate Concentrate (Configuration A), 0.5 0.5 M sodium carbonate, commercially available.
7.8 Sodium Bicarbonate Concentrate (Configuration A), 0.5 0.5 M sodium bicarbonate, commercially available.
7.9 Monoethylene Glycol (MEG) (High Purity), with low concentrations of impurities.
7.10 Diethylene Glycol (DEG) (High Purity), with low concentrations of impurities.
7.11 Triethylene Glycol (TEG) (High Purity), with low concentrations of impurities.
NOTE 6— Impurities in high-purity MEG, DEG or TEG used for preparation of the chloride working standards or for sample dilution should not exceed
0.01 mg/kg. This information should be provided by the supplier or determined by the analyst before use.
7.12 Sulfuric Acid (for use with anion micromembrane suppressor), concentrated sulfuric acid (95 to 98 %).
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For Suggestions on the testing of reagents not listed by
the American Chemical Society, see Annual Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),(USP), Rockville, MD.
E2469 − 16
7.13 Anion Regenerant Concentrate (for use with anion micromembrane suppressor), 1 M sulfuric acid, commercially
available.
7.14 Isopropyl Alcohol (2-Propanol), ACS reagent grade.
7.15 Methyl Alcohol (Methanol, alternate for Isopropyl Alcohol), ACS reagent grade.
7.16 Monoethylene Glycol Quality Control Sample, polyester grade MEG.
7.17 Diethylene Glycol Quality Control Sample.
7.18 Triethylene Glycol Quality Control Sample.
8. Reagent Solutions
8.1 Chloride Stock Solution (1000 mg/kg Chloride in Water):
NOTE 7—As an alternative, a 1000 μg/mL chloride standard may be purchased commercially.
NOTE 8—The density of the commercial chloride stock solution is assumed to be 1.0 g/mL at room temperature for wt/wt calculations in mg/kg.
8.1.1 Place a 125-mL Nalgene LDPE narrow mouth bottle on the balance and tare.
8.1.2 Weigh (and record to the nearest 0.0001 g) 0.1649 6 0.0010 g of sodium chloride (7.3) into the 125-mL bottle.
8.1.3 Dilute to 100 g (and record to the nearest 0.001 g) with reagent water (7.2) and mix well. Prepare this calibration solution
once a year.
8.1.4 Calculate the concentration of chloride in the chloride stock solution as follows:
W 310 mg 3FW
NaCl Cl
C 5 (1)
CSS
W 31 kg 3FW
STD NaCl
where:
where:
C = the concentration (mg/kg) of chloride in the chloride stock solution,
CSS
W = weight (g) of sodium chloride added to the chloride stock solution (8.1.2),
NaCl
W = total weight (g) of the prepared chloride stock solution (8.1.3, nominally 100 g),
STD
FW = formula weight of chloride (35.457 g/mol), and
Cl
FW = formula weight of sodium chloride (58.44 g/mol).
NaCl
8.2 Chloride Calibration Solution (10 mg/kg Chloride in Water):
NOTE 9—Pipettes are used to dispense the estimated amount of chloride stock solution (1000 mg/kg).
8.2.1 Place a 100-mL volumetric flask on the balance and tare. Pipette (and record the weight to the nearest 0.0001 g) 1000 μL
of 1000 mg/kg chloride stock solution (8.1) into the 100-mL volumetric flask.
8.2.2 Dilute to 100 mL (and record the weight to the nearest 0.001 g) with reagent water (7.2) and mix well. Store the 1010 mg ⁄
mg/kg kg chloride calibration solution in a 125-mL Nalgene LDPE narrow mouth bottle. Prepare this calibration solution once a
year.
8.2.3 Determine the concentration (mg/kg) of chloride in the chloride calibration solution as follows:
C 3W
CSS CSS
C 5 (2)
CS
W
CS
where:
where:
C = the concentration (mg/kg) of chloride in the chloride calibration solution,
CS
C = the concentration (mg/kg) of chloride in the chloride stock solution (8.1),
CSS
W = weight (g) of chloride stock solution added to chloride calibration solution (8.2.1), and
CSS
W = final weight (g) of the prepared chloride calibration solution (8.2.2, nominally 100 g).
CS
8.3 Chloride Working Standards (0.01, 0.02, 0.05, 0.1, 0.2, 0.5, and 1.0 mg/kg in MEG).MEG, DEG, or TEG).
NOTE 10—Pipettes are used to dispense the estimated amount of chloride calibration solution (10 mg/kg).
8.3.1 Prepare at least 3five chloride working standard solutions (8.3) covering the expected range for the glycol being analyzed.
Choose the dilutions such that the resulting concentrations are equally distributed over the range of interest. For each of the
chloride working standards, place a 120-mL plastic cup on the balance and tare. Using Table 1 for guidance, add the appropriate
amount of 10 mg/kg chloride calibration solution (8.2) and record the weight to the nearest 0.0001 g.
8.3.2 Add the high purity MEG, DEG or TEG (7.9, 7.10 or 7.11) to obtain the final weight of 100 g. Mix well to ensure a
homogeneous solution. Record the final weight of the chloride working standard to the nearest 0.001 g.
NOTE 11—Chloride working standards should be prepared a minimum of every three months (monthly preparation is recommended). To avoid
contamination of the standard, always pour the working standard out of the bottle for use and never put anything (pipette and so forth) into the
E2469 − 16
TABLE 1 Chloride Working Standard
Chloride Weight of 10 mg/kg Final Weight of
Working Nominal Chloride Calibration Chloride Working
Standard # Concentration Solution (to add) Standard (MEG DEG or
TEG added)
mg/kg g (± 0.0001 g) g (± 0.001 g)
1 0 0 100
2 0.01 0.1 100
3 0.02 0.2 100
4 0.05 0.5 100
5 0.1 1.0 100
6 0.2 2.0 100
7 0.5 5.0 100
8 1.0 10.0 100
bottle. Minimize the number of times and length of time the working standards are exposed to air. Cap securely after each use.
8.3.3 Determine the concentration (mg/kg) of chloride in each of the chloride working standards as follows:
C 3W
CS CS~i!
C 5 (3)
WS i
~ !
W
WS i
~ !
where:
where:
C = the concentration (mg/kg) of chloride in chloride working standard, i,
WS(i)
C = the concentration (mg/kg) of chloride in the chloride calibration solution (8.2),
CS
W = weight (g) of chloride calibration solution added to chloride working standard, i (8.3.1), and
CS(i)
W = final weight (g) of the prepared chloride working standard, i (8.3.2, nominally 100 g).
WS(i)
8.3.4 If utilizing an autosampler and dilution is required, it is recommended to dilute standards with high purity deionized water
prior to calibration. Dilution of the standards should be the same as dilution used for samples. Application of the same dilution
parameter to the standards and samples will eliminate further calculations. Mix well to ensure a homogeneous solution. Record
the final weight of the chloride working standard to the nearest 0.001 g.
8.4 Anion Regenerant Solution (12.5 mM Sulfuric Acid) (for Use with Anion Micromembrane Suppressor):
NOTE 12—Consult instrument suppressor manufacturer for preparation of the suppressor regenerate solution.
8.4.1 Add approximately 1 L of reagent water to a 2-L volumetric flask. Slowly add 1.4 mL (or 2.6 g) of concentrated sulfuric
acid (7.12) to the flask taking care to avoid splashing and overheating. Bring to volume with reagent water and mix well. Prepare
fresh regenerant solution at least once per month.
8.4.2 Alternative Preparation with Anion Regenerant Concentrate—Add 1 part of the anion regenerant concentrate (1 M
sulfuric acid) (7.13) to 80 parts reagent water (for example, add 25 mL of anion regenerant concentrate to a 2-L volumetric flask.
Bring to volume with reagent water and mix well). Prepare fresh regenerant solution at least once per month.
8.5 Carbonate/Bicarbonate Eluent Solution (0.6 mM/0.4 mM) mM/0.4 mM) Prepared from Carbonate/Bicarbonate Salts (for
use with Configuration A):
NOTE 13—Consult with the anion analytical column manufacturer for recommended mobile phase concentration and preparation of the separation (8.5
and 8.6).
8.5.1 Place a small plastic weigh dish on the balance and tare. Add 0.1250 6 0.0010 g of sodium carbonate (7.5) to weigh dish.
Transfer the sodium carbonate in the weigh dish to a 2-L volumetric flask. Rinse the weigh dish into the volumetric flask using
a small amount of reagent water.
8.5.2 Place a second small plastic weigh dish on the balance and tare. Add 0.0625 6 0.0010 g of sodium bicarbonate (7.6) to
the weigh dish. Transfer the sodium bicarbonate in the weigh dish to the 2-L volumetric flask used in 8.5.1. Rinse the weigh dish
into the volumetric flask using a small amount of reagent water.
8.5.3 Add reagent water to the 2-L volumetric flask to bring it to volume and mix well. Prepare fresh eluent solution at least
once every two weeks.
8.6 Carbonate/Bicarbonate Eluent Solution (0.6 mM/0.4 mM) mM/0.4 mM) Prepared from Carbonate/Bicarbonate Concen-
trates (for Use with Configuration A)—Add 2.4 mL of sodium carbonate concentrate (7.7) and 1.6 mL of sodium bicarbonate
concentrate (7.8) to a 2-L volumetric flask. Bring to volume with reagent water and mix well. Prepare fresh eluent solution at least
once every two weeks.
8.7 Potassium Hydroxide Solution (Configuration B)—5 mM 5 mM carbonate-free potassium hydroxide made electrolytically
on-line by the eluent generator module using reagent water and a potassium hydroxide cartridge which is part of the IC system
(6.4).
E2469 − 16
9. Hazards
9.1 Each analyst must be acquainted with the potential hazards of the equipment, reagents, products, solvents and procedures
before beginning laboratory work. Sources of information include: operation manuals, MSDS, literature, and other related data.
Safety information should be requested from the supplier. Disposal of waste materials, reagents, reactants, and solvents must be
in compliance with laws and regulations from all applicable governmental agencies.
9.2 MEG, DEG and TEG products are intended for industrial use only. Before handling or using these products, read the current
MSDS for each product (9.1).
9.3 The following hazards are associated with the application of this test method and the use of an ion chromatograph.
9.3.1 Chemical Hazard:
9.3.1.1 Warning—Concentrated sulfuric acid is corrosive and should be handled in a fume hood with gloves, chemical goggles,
and lab coat or chemical-resistant apron. When diluting concentrated sulfuric acid, always add acid to water.Warning—
Concentrated sulfuric acid is corrosive and should be handled in a fume hood with gloves, chemical goggles, and lab coat or
chemical-resistant apron. When diluting concentrated sulfuric acid, always add acid to water.
9.3.1.2 Warning—Methanol and isopropyl alcohol are flammable and toxic solvents that are used to prime the pump in an IC.
Be careful when handling a flammable solvent and work in a well-ventilated area away from sources of ignition.Warning—
Methanol and isopropyl alcohol are flammable and toxic solvents that are used to prime the pump in an IC. Be careful when
handling a flammable solvent and work in a well-ventilated area away from sources of ignition.
9.3.1.3 Excessive force on the injection syringe (manual injection) can cause glycol to spray on the analyst. Wear gloves,
chemical goggles, and lab coat or chemical-resistant apron when injecting a sample. Use Luer-Lok syringes to load the sample into
the injection port. Turn face away from the injection port while depressing the syringe plunger. Depress syringe plunger slowly
to avoid high back pressure as a result of the viscosity of the glycol sample.
9.3.2 Electrical/Shock Hazard:
9.3.2.1 Ion chromatographs (pump modules and conductivity detectors) contain printed circuit boards and components
operating at dangerous voltages. The circuit boards and components are covered with protective panels that are identified by
warning labels. Contact with these circuits boards and components can cause serious injury or painful electrical shock. Turn off
the system power and unplug the line cord bef
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...