ASTM E2037-24
(Test Method)Standard Test Method for Bromine Chloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
Standard Test Method for Bromine Chloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
SIGNIFICANCE AND USE
4.1 This test method was developed for the determination of bromine chloride in liquid chlorine. Bromide is a common contaminant in all salt sources that are used in the production of chlorine. This bromide content of the salt is converted into bromine chloride in the liquid chlorine product. This test method is sensitive enough to measure the levels of bromine chloride observed in normal production chlorine.
SCOPE
1.1 This test method uses high performance liquid chromatography (HPLC) to determine bromine chloride levels in liquid chlorine at the 10 μg/g (ppm) to 1400 μg/g (ppm) range.
1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 8.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 29-Feb-2024
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.13 - Chlorine
Relations
- Effective Date
- 01-Mar-2024
Overview
ASTM E2037-24: Standard Test Method for Bromine Chloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC) establishes procedures for the precise determination of bromine chloride content in liquid chlorine. Developed by ASTM International, this analytical standard addresses a critical quality control parameter in chlorine production, as bromide impurities from salt sources are converted to bromine chloride during manufacture. The method utilizes high performance liquid chromatography (HPLC), delivering reliable sensitivity across a wide range, from 10 μg/g (ppm) to 1400 μg/g (ppm) bromine chloride concentrations.
This test method is essential for producers and users of liquid chlorine, supporting regulatory compliance, product safety, and process optimization. The procedure aligns with international principles on standardization and meets key requirements for analytical precision, repeatability, and reporting.
Key Topics
Purpose and Significance
- Detects bromine chloride, a common contaminant in liquid chlorine derived from natural bromide in salt feedstocks.
- Enables consistent quality monitoring for manufacturers and end-users of liquid chlorine.
- Sensitive and selective, with minimal interference in the specified concentration range.
Test Method Highlights
- Employs HPLC equipped with UV detection and reverse phase C18 column for accurate analytical separation.
- Utilizes a clearly defined sample preparation, eluent composition, and calibration procedures using potassium bromide standards.
- Reports bromine chloride concentrations rounded in accordance with ASTM E29 for significant digits.
Safety and Handling
- Includes guidance for handling corrosive and toxic chlorine samples, requiring well-ventilated fume hoods, protective equipment, and adherence to current SDS, OSHA, and local regulations.
- Addresses specialized equipment needs, such as nonmetallic syringes and tubing to mitigate corrosion.
- Specifies safe procedures for sample collection, storage, and disposal, emphasizing environmental and personnel safety.
Quality Control
- Recommends implementation of laboratory quality assurance (QA) and statistical quality control (QC) protocols, referencing ASTM D6809 when no internal protocols exist.
- Suggests proficiency testing and interlaboratory comparison to validate test performance across different facilities.
Applications
Chlorine Production Quality Control
- Routine analysis to ensure specification compliance and minimize process upsets or product recalls due to bromine chloride contamination.
- Supplier and customer assurance, facilitating accurate commercial and regulatory reporting.
Regulatory and Safety Compliance
- Supports conformance with federal transport standards (such as 49 CFR 173) governing shipment and packaging of compressed and liquefied gases.
- Aids in meeting international and domestic health, safety, and environmental regulations concerning chlorine handling.
Process Development and Optimization
- Allows chemical manufacturers and process engineers to monitor impurity levels, optimize purification stages, and improve feedstock selection or brine purification techniques.
Related Standards
- ASTM E29 - Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
- ASTM D6809 - Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
- ASTM E806 - Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine (Superseded but referenced)
- Chlorine Institute Pamphlet No. 1 - Chlorine Basics (industry reference)
- 49 CFR 173.304, 173.314, 173.315 - Federal standards for transportation and packaging of compressed or liquefied chlorine
Keywords: ASTM E2037, bromine chloride analysis, liquid chlorine testing, HPLC, chlorine quality control, industrial chemical analysis, bromine impurities, standard test method, regulatory compliance, chemical process optimization.
Buy Documents
ASTM E2037-24 - Standard Test Method for Bromine Chloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
REDLINE ASTM E2037-24 - Standard Test Method for Bromine Chloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E2037-24 is a standard published by ASTM International. Its full title is "Standard Test Method for Bromine Chloride in Liquid Chlorine by High Performance Liquid Chromatography (HPLC)". This standard covers: SIGNIFICANCE AND USE 4.1 This test method was developed for the determination of bromine chloride in liquid chlorine. Bromide is a common contaminant in all salt sources that are used in the production of chlorine. This bromide content of the salt is converted into bromine chloride in the liquid chlorine product. This test method is sensitive enough to measure the levels of bromine chloride observed in normal production chlorine. SCOPE 1.1 This test method uses high performance liquid chromatography (HPLC) to determine bromine chloride levels in liquid chlorine at the 10 μg/g (ppm) to 1400 μg/g (ppm) range. 1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 8. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method was developed for the determination of bromine chloride in liquid chlorine. Bromide is a common contaminant in all salt sources that are used in the production of chlorine. This bromide content of the salt is converted into bromine chloride in the liquid chlorine product. This test method is sensitive enough to measure the levels of bromine chloride observed in normal production chlorine. SCOPE 1.1 This test method uses high performance liquid chromatography (HPLC) to determine bromine chloride levels in liquid chlorine at the 10 μg/g (ppm) to 1400 μg/g (ppm) range. 1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 8. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2037-24 is classified under the following ICS (International Classification for Standards) categories: 71.040.30 - Chemical reagents. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2037-24 has the following relationships with other standards: It is inter standard links to ASTM E2037-15. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2037-24 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2037 − 24
Standard Test Method for
Bromine Chloride in Liquid Chlorine by High Performance
Liquid Chromatography (HPLC)
This standard is issued under the fixed designation E2037; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* Replaced by D 2645 (Withdrawn 1967)
D6809 Guide for Quality Control and Quality Assurance
1.1 This test method uses high performance liquid chroma-
Procedures for Aromatic Hydrocarbons and Related Ma-
tography (HPLC) to determine bromine chloride levels in
terials
liquid chlorine at the 10 μg ⁄g (ppm) to 1400 μg/g (ppm) range.
E29 Practice for Using Significant Digits in Test Data to
1.2 Review the current safety data sheet (SDS) for detailed
Determine Conformance with Specifications
information concerning toxicity, first aid procedures, and safety
E180 Practice for Determining the Precision of ASTM
precautions.
Methods for Analysis and Testing of Industrial and Spe-
cialty Chemicals (Withdrawn 2009)
1.3 The values stated in SI units are to be regarded as
E806 Test Method for Carbon Tetrachloride and Chloroform
standard. No other units of measurement are included in this
in Liquid Chlorine by Direct Injection (Gas Chromato-
standard.
graphic Procedure)
1.4 The following applies for the purposes of determining 4
2.2 Federal Standards:
the conformance of the test results using this test method to
CFR 173 Title 49 Transportation; Shippers’ General Re-
applicable specifications, results shall be rounded off in accor-
quirements for Shipments and Packaging, including Sec-
dance with the rounding-off method of Practice E29.
tions:
1.5 This standard does not purport to address all of the
173.304 Charging of Cylinders with Liquefied Compressed
safety concerns, if any, associated with its use. It is the
Gas
responsibility of the user of this standard to establish appro-
173.314 Requirements for Compressed Gases in Tank Cars
priate safety, health, and environmental practices and deter-
173.315 Compressed Gases in Cargo Tanks and Portable
mine the applicability of regulatory limitations prior to use.
Tank Containers
Specific hazard statements are given in Section 8.
2.3 Other Document:
1.6 This international standard was developed in accor-
Chlorine Institute Pamphlet No. 1 Chlorine Basics
dance with internationally recognized principles on standard-
3. Summary of Test Method
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
3.1 Weighed samples of chlorine delivered into a cooled
mendations issued by the World Trade Organization Technical
graduated centrifuge tube. One mL of cooled HPLC eluent is
Barriers to Trade (TBT) Committee.
added before the chlorine is allowed to evaporate. After the
chlorine has evaporated the remaining eluent is analyzed
2. Referenced Documents
directly on the HPLC for bromine chloride concentration.
2.1 ASTM Standards:
4. Significance and Use
D180 Methods of Testing Tolerances for Cotton Yarns;
4.1 This test method was developed for the determination of
bromine chloride in liquid chlorine. Bromide is a common
contaminant in all salt sources that are used in the production
This test method is under the jurisdiction of ASTM Committee D16 on
of chlorine. This bromide content of the salt is converted into
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi-
bility of Subcommittee D16.13 on Chlorine.
Current edition approved March 1, 2024. Published March 2024. Originally
approved in 1999. Last previous edition approved in 2015 as E2037 – 15. DOI: The last approved version of this historical standard is referenced on
10.1520/E2037-24. www.astm.org.
2 4
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Available from U.S. Government Publishing Office, 732 N. Capitol St., NW,
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Washington, DC 20401-0001, http://www.gpo.gov.
Standards volume information, refer to the standard’s Document Summary page on Available from The Chlorine Institute, 1300 Wilson Blvd., Ste. 525, Arlington,
the ASTM website. VA 22209, http://www.chlorineinstitute.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2037 − 24
bromine chloride in the liquid chlorine product. This test
method is sensitive enough to measure the levels of bromine
chloride observed in normal production chlorine.
5. Interferences
5.1 This test method is selective for bromine chloride. At
this time there are no known interference in the materials used
FIG. 1 Sodium Acetate, reagent grade. CAS# 127-09-3.
in this test method.
5.2 Contact with any metal surfaces should be avoided due
to the corrosive nature of the sample.
6. Apparatus
6.1 A high performance liquid chromatograph (HPLC) com-
posed of the following:
6.1.1 HPLC Pump, capable of 1 mL/min flow,
6.1.2 HPLC UV Detector, capable of operating at 221 nm
with a 1 cm cell,
FIG. 2 Glacial Acetic Acid, reagent grade. CAS# 64-19-7.
6.1.3 HPLC Injection Valve, 20 μl loop, all nonmetal, and
6.1.4 HPLC Column, C18 reverse phase, 25 cm by 4.6 mm.
6.2 Plastic Syringes, 1 mL, 2.5 mL, 5 mL, 10 mL, 20 mL,
handling and to vent the test equipment when this product is
and 60 mL.
analyzed in the laboratory.
6.3 Nonmetallic Syringe Needles.
8.2 The analysis should be attempted only by persons who
6.4 Top Loader Balance, capable of 0.01 g resolution with a
are thoroughly familiar with the handling of chlorine, and even
1 kg capacity. an experienced person should not work alone. The operator
must be provided with adequate eye protection and a respirator.
6.5 TFE-Fluorocarbon Tubing, 1.59 mm outside diameter.
Splashes of liquid chlorine destroy clothing and if such
6.6 Stainless Steel Sample Cylinder, with a needle valve on
clothing is next to the skin, it will produce irritations and burns.
one end.
8.3 If liquid samples are to be taken in cylinders, do not
6.7 Graduated Centrifuge Tube, 15 mL.
allow the sample cylinder to become liquid full. Test Method
E806, 49 CFR 173.314, 173.315, and 173.304 advise that the
7. Reagents
weight of the chlorine in the cylinder should not be more than
7.1 Purity of Reagents—Unless otherwise indicated, it is
125 % of the weight of the water that the cylinder could
intended that all reagents shall conform to the specifications of
contain. See the Chlorine Institute Pamphlet No. 1 for addi-
the Committee on Analytical Reagents of the American Chemi-
tional technical information regarding liquid chlorine.
cal Society, where such specifications are available. Other
8.4 When sampling and working with chlorine out-of-doors,
grades may be used, provided it is first ascertained that the
people downwind from such an operation should be warned of
reagent is of sufficiently high purity to permit its use without
the possible release of chlorine vapors.
lessening the accuracy of the determination.
8.5 In the event chlorine is inhaled, first aid should be
7.2 Water, HPLC grade.
summoned immediately and oxygen administered without
7.3 Methanol, HPLC grade. CAS# 67-56-1.
delay.
7.4 Sodium Acetate, reagent grade. CAS# 127-09-3.
8.6 Store pressurized samples where involuntary release
7.5 Glacial Acetic Acid, reagent grade. CAS# 64-19-7.
would not cause excessive risk to people or property.
7.6 Dry Ice. CAS# 124-38-9, CO . 8.7 It is recommended that means be available for disposal
of excess chlorine in an environmentally safe and acceptable
7.7 Potassium Bromide, reagent grade. CAS# 7758-02-3.
manner. If chlorine cannot be disposed of in a chlorine
7.8 Chlorine, reagent grade. CAS# 7782-50-5, Cl .
consuming process, a chlorine absorption system should be
provided. When the analysis and sampling regimen requires an
8. Hazards
initial purging of chlorine from a container, the purged chlorine
8.1 Chlorine is a corrosive and toxic material. A well-
should be similarly handled. Purging to the atmosphere should
ventilated fume hood should be used to house all sample
be avoided.
8.8 Safety and Health Precautions—Exposure to all solvents
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
used in this test method should be avoided.
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by the American Chemical
8.9 Consult current OSHA regulations, suppliers’Safety
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
Data Sheets, and local regulations for all materials used in this
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. test method.
E2037 − 24
9. Typical Instrument Parameters 13.2 Withdraw 0.5 mL of a potassium bromide standard into
a 1 mL plastic syringe and then pull the plunger back to the
9.1 Adjust the chromatograph in accordance with the fol-
1 mL mark to fill the remainder of the syringe with air.
lowing parameters and allow the instrument to equilibrate until
a steady baseline is obtained: 13.3 Clamp a sample cylinder containing reagent chlorine
9.1.1 Column—C18 reverse phase ODS (C18) 25 cm by 4.6 vertically in a ring stand and attach a 152.40 mm piece of 1.59
mm, 10 μm, mm outside diameter TFE-fluorocarbon tubing to the top valve.
9.1.2 Eluent—60 % by volume methanol, 40 % by volume Take a second 1 mL plastic syringe and pull the plunger back
0.1 mol/L (M) acetate buffer, pH 4.5, helium sparged, to the 1 mL mark. Insert the 1.59 mm TFE-fluorocarbon tubing
9.1.3 Flow Rate—1 mL/min, into the syringe and gently purge chlorine through the syringe
9.1.4 Injection Volume—20 μl sample loop, and filling this second syringe with reagent chlorine gas. Remove
9.1.5 Detector Wavelength—UV at 221 nm. the TFE-fluorocarbon tubing from the syringe and attach a
nonmetal needle to the luer tip.
10. Preparation of Buffer Solution
13.4 Holding the first syringe pointed upward, use the
10.1 Sodium Acetate Buffer Stock Solution (1 mol/L (M))—
second syringe equipped with the nonmetal needle to bubble
Dissolve 136 g of sodium acetate (NaOOCCH ·3H O) and
3 2 the reagent chlorine gas through the potassium bromide stan-
60 g of glacial acetic acid (HOOCCH ) in water and dilute to
3 dard solution, converting the bromide ions to bromine chloride.
1 L.
One mL of reagent chlorine gas is required for the conversion
of each 500 μg/g (ppm) of bromide ion in the standard.
10.2 Sodium Acetate Buffer (0.1 mol/L (M))—Transfer 100
Although the amount of chlorine used to chlorinate the
mL of the stock buffer solution into a 1 L volumetric flask and
standards is extremely small when compared to that of the
dilute to volume with water.
sample, it is always a good idea to prepare the first standard in
11. Preparation of Eluent
any calibration curve without the addition of the potassium
bromide to serve as a reagent blank.
11.1 Add 600 mL of methanol to 400 mL of 0.1 mol/L (M)
sodium acetate buffer solution and mix well. Before use, purge
13.5 Using the entire 0.5 mL of standard flush the injection
the solution with helium for 20 min to remove dissolved
valve and fill the sample loop in the injection valve. Immedi-
oxygen.
ately inject this standard into the HPLC for analysis. The
bromine chloride peak will elute betwe
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2037 − 15 E2037 − 24
Standard Test Method for
Bromine Chloride in Liquid Chlorine by High Performance
Liquid Chromatography (HPLC)
This standard is issued under the fixed designation E2037; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method uses high performance liquid chromatography (HPLC) to determine bromine chloride levels in liquid chlorine
at the 1010 μg ⁄g (ppm) to 1400 μg/g (ppm) range.
1.2 Review the current safety data sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety
precautions.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 The following applies for the purposes of determining the conformance of the test results using this test method to applicable
specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. Specific hazard statements are given in Section 8.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D180 Methods of Testing Tolerances for Cotton Yarns; Replaced by D 2645 (Withdrawn 1967)
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
E806 Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic
Procedure)
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsibility of
Subcommittee D16.13 on Chlorine.
Current edition approved June 1, 2015March 1, 2024. Published June 2015March 2024. Originally approved in 1999. Last previous edition approved in 20072015 as
E2037– 07. DOI: 10.1520/E2037-15. – 15. DOI: 10.1520/E2037-24.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2037 − 24
2.2 Federal Standards:
CFR 173 Title 49 Transportation; Shippers’ General Requirements for Shipments and Packaging, including Sections:
173.304 Charging of Cylinders with Liquefied Compressed Gas
173.314 Requirements for Compressed Gases in Tank Cars
173.315 Compressed Gases in Cargo Tanks and Portable Tank Containers
2.3 Other Document:
Chlorine Institute Pamphlet No. 1 Chlorine Basics
3. Summary of Test Method
3.1 Weighed samples of chlorine delivered into a cooled graduated centrifuge tube. One mL of cooled HPLC eluent is added
before the chlorine is allowed to evaporate. After the chlorine has evaporated the remaining eluent is analyzed directly on the
HPLC for bromine chloride concentration.
4. Significance and Use
4.1 This test method was developed for the determination of bromine chloride in liquid chlorine. Bromide is a common
contaminant in all salt sources that are used in the production of chlorine. This bromide content of the salt is converted into bromine
chloride in the liquid chlorine product. This test method is sensitive enough to measure the levels of bromine chloride observed
in normal production chlorine.
5. Interferences
5.1 This test method is selective for bromine chloride. At this time there are no known interference in the materials used in this
test method.
5.2 Contact with any metal surfaces should be avoided due to the corrosive nature of the sample.
6. Apparatus
6.1 A high performance liquid chromatograph (HPLC) composed of the following:
6.1.1 HPLC Pump, capable of 1 mL/min flow,
6.1.2 HPLC UV Detector, capable of operating at 221 nm with a 1-cm1 cm cell,
6.1.3 HPLC Injection Valve, 20 μl loop, all nonmetal, and
6.1.4 HPLC Column, C18 reverse phase, 25 cm by 4.6 mm.
6.2 Plastic Syringes, 1, 2.5, 5, 10, 20,1 mL, 2.5 mL, 5 mL, 10 mL, 20 mL, and 60 mL.
6.3 Nonmetallic Syringe Needles.
6.4 Top Loader Balance, capable of 0.01 g resolution with a 1-kg1 kg capacity.
6.5 TFE-Fluorocarbon Tubing, 1.59 mm outside diameter.
6.6 Stainless Steel Sample Cylinder, with a needle valve on one end.
6.7 Graduated Centrifuge Tube, 15 mL.
Available from U.S. Government Publishing Office, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://www.gpo.gov.
Available from The Chlorine Institute, 1300 Wilson Blvd., Ste. 525, Arlington, VA 22209, http://www.chlorineinstitute.org.
E2037 − 24
7. Reagents
7.1 Purity of Reagents—Unless otherwise indicated, it is intended that all reagents shall conform to the specifications of the
Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
7.2 Water, HPLC grade.
7.3 Methanol, HPLC grade. CAS# 67-56-1.
7.4 Sodium Acetate, reagent grade. CAS# 127-09-3.
FIG. 1 Sodium Acetate, reagent grade. CAS# 127-09-3.
7.5 Glacial Acetic Acid, reagent grade.grade. CAS# 64-19-7.
7.6 Dry Ice. CAS# 124-38-9, CO .
7.7 Potassium Bromide, reagent grade. CAS# 7758-02-3.
7.8 Chlorine, reagent grade. CAS# 7782-50-5, Cl .
FIG. 2 Glacial Acetic Acid, reagent grade. CAS# 64-19-7.
8. Hazards
8.1 Chlorine is a corrosive and toxic material. A well-ventilated fume hood should be used to house all sample handling and to
vent the test equipment when this product is analyzed in the laboratory.
8.2 The analysis should be attempted only by persons who are thoroughly familiar with the handling of chlorine, and even an
experienced person should not work alone. The operator must be provided with adequate eye protection and a respirator. Splashes
of liquid chlorine destroy clothing and if such clothing is next to the skin, it will produce irritations and burns.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
E2037 − 24
8.3 If liquid samples are to be taken in cylinders, do not allow the sample cylinder to become liquid full. Test Method E806, 49
CFR 173.314, 173.315, and 173.304 advise that the weight of the chlorine in the cylinder should not be more than 125 % of the
weight of the water that the cylinder could contain. See the Chlorine Institute Pamphlet No. 1 for additional technical information
regarding liquid chlorine.
8.4 When sampling and working with chlorine out-of-doors, people downwind from such an operation should be warned of the
possible release of chlorine vapors.
8.5 In the event chlorine is inhaled, first aid should be summoned immediately and oxygen administered without delay.
8.6 Store pressurized samples where involuntary release would not cause excessive risk to people or property.
8.7 It is recommended that means be available for disposal of excess chlorine in an environmentally safe and acceptable manner.
If chlorine cannot be disposed of in a chlorine consuming process, a chlorine absorption system should be provided. When the
analysis and sampling regimen requires an initial purging of chlorine from a container, the purged chlorine should be similarly
handled. Purging to the atmosphere should be avoided.
8.8 Safety and Health Precautions—Exposure to all solvents used in this test method should be avoided.
8.9 Consult current OSHA regulations, suppliers’Safety Data Sheets, and local regulations for all materials used in this test
method.
9. Typical Instrument Parameters
9.1 Adjust the chromatograph in accordance with the following parameters and allow the instrument to equilibrate until a steady
baseline is obtained:
9.1.1 Column—C18 reverse phase ODS (C18) 25 cm by 4.6 mm, 10 μm,
9.1.2 Eluent—60 % by volume methanol, 40 % by volume 0.1 mol/L (M) acetate buffer, pH 4.5, helium sparged,
9.1.3 Flow Rate—1 mL/min,
9.1.4 Injection Volume—20-μl20 μl sample loop, and
9.1.5 Detector Wavelength—UV at 221 nm.
10. Preparation of Buffer Solution
10.1 Sodium Acetate Buffer Stock Solution (1 mol/L (M))—Dissolve 136 g of sodium acetate (NaOOCCH ·3H O) and 60 g 60 g
3 2
of glacial acetic acid (HOOCCH ) in water and dilute to 1 L.
10.2 Sodium Acetate Buffer (0.1 mol/L (M))—Transfer 100 mL of the stock buffer solution into a 1-L1 L volumetric flask and dilute
to volume with water.
11. Preparation of Eluent
11.1 Add 600 mL of methanol to 400 mL of 0.1 mol/L (M) sodium acetate buffer solution and mix well. Before use, purge the
solution with helium for 20 min to remove dissolved oxygen.
12. Preparation of the Sample Cylinder
12.1 Clamp the sample cylinder in a vertical position with the needle valve in the downward position. Insert the 6.35 mm end of
the reducing tube fitting into the needle valve and set the 6.35 mm nut and ferrule of the fitting. Insert the 1.59 mm fluoropolymer
E2037 − 24
tubing in the reducing tube fitting and tighten the 1.59 mm nut and ferrule. See Fig. 13. It may be helpful to cut a 6.35 mm circle
of fluoropolymer frit material and place it into the reducing fitting prior to assembly to prevent plugging of the 1.59 mm tubing.
Cut the length of the 1.59 mm tubing so that only 38.10 mm protrudes out of the fitting.
13. Standardization of the HPLC
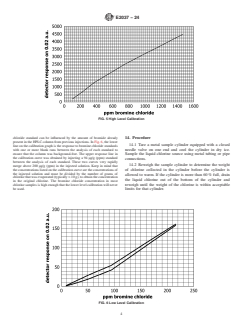
13.1 Prepare a 1000 μg/g (ppm) stock solution of potassium bromide in water. Make a series of standards of potassium bromide
by serial dilution covering the range between 11 μg ⁄g (ppm) and 650 μg/g (ppm) potassium bromide. Calculate the bromine
chloride concentration of each standard from the potassium bromide concentration by multiplying by the ratio of the molecular
weights:
BrCl/KBr 5 115.4/119 5 0.97 (1)
~ ! ~ !
13.2 Withdraw 0.5 mL of a potassium bromide standard into a 1-mL1 mL plastic syringe and then pull the plunger back to the
1 mL 1 mL mark to fill the remainder of the syringe with air.
13.3 Clamp a sample cylinder containing reagent chlorine vertically in a ring stand and attach a 152.40 mm p
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...