ASTM D3895-19
(Test Method)Standard Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry
Standard Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry
SIGNIFICANCE AND USE
5.1 The OIT is a qualitative assessment of the level (or degree) of stabilization of the material tested. This test has the potential to be used as a quality control measure to monitor the stabilization level in formulated resin as received from a supplier, prior to extrusion.
Note 2: The OIT measurement is an accelerated thermal-aging test and as such can be misleading. Caution should be exercised in data interpretation since oxidation reaction kinetics are a function of temperature and the inherent properties of the additives contained in the sample. For example, OIT results are often used to select optimum resin formulations. Volatile antioxidants may generate poor OIT results even though they may perform adequately at the intended use temperature of the finished product.
Note 3: There is no accepted sampling procedure, nor have any definitive relationships been established for comparing OIT values on field samples to those on unused products, hence the use of such values for determining life expectancy is uncertain and subjective.
SCOPE
1.1 This test method outlines a procedure for the determination of oxidative-induction time (OIT) of polymeric materials by differential scanning calorimetry (DSC). It is applicable to polyolefin resins that are in a fully stabilized/compounded form.
1.2 The values stated in SI units are to be regarded as the standard.
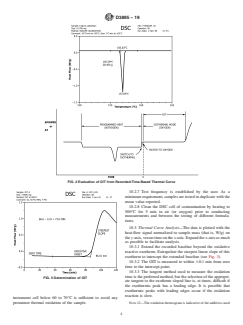
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards information is given in Section 8.
Note 1: This standard and ISO 11357–6 2013 address the same subject matter, but differ in technical content.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2019
- Technical Committee
- D20 - Plastics
- Drafting Committee
- D20.30 - Thermal Properties
Relations
- Effective Date
- 01-Oct-2023
- Refers
ASTM E2935-17 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2017
- Refers
ASTM E2935-16 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 15-Nov-2016
- Refers
ASTM E2935-15 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2015
- Refers
ASTM E2935-14 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2014
- Effective Date
- 15-Aug-2014
- Refers
ASTM E2935-13 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Aug-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-Apr-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Aug-2009
- Effective Date
- 01-Oct-2008
Overview
ASTM D3895-19 defines the standard test method for determining the Oxidative-Induction Time (OIT) of polyolefins using differential scanning calorimetry (DSC). OIT is a relative indicator of the oxidation resistance of polyolefinic materials, such as polyethylene and polypropylene. This test method is crucial for assessing the degree of stabilization of polyolefin resins and compounds, helping manufacturers understand how materials will perform under thermal stress and in processing environments.
Employing this ASTM standard ensures consistent evaluations across the plastics and polyolefin industries, facilitating quality control and material selection in a variety of manufacturing sectors.
Key Topics
Oxidative-Induction Time (OIT):
OIT measures the time interval before a rapid exothermic oxidation reaction occurs in a polyolefin sample held at a specified temperature under an oxygen atmosphere. It serves as a qualitative assessment of stabilization level and the oxidation resistance of the polymer.Differential Scanning Calorimetry (DSC):
DSC is used to measure the heat flow in a polymer sample during controlled heating conditions. After preconditioning in nitrogen, the sample is exposed to oxygen at a set temperature, and the DSC records the onset of oxidation.Application to Polyolefins:
The method applies specifically to fully stabilized/compounded polyolefin resins, such as:- HDPE (high-density polyethylene)
- LDPE (low-density polyethylene)
- LLDPE (linear low-density polyethylene)
- Polypropylene
Quality Control Implications:
The OIT test serves as a potential quality control tool for monitoring the effectiveness of antioxidant stabilization in supplied resins prior to further processing, such as extrusion or molding.Limitations and Data Interpretation:
- OIT is an accelerated aging test; oxidation reaction kinetics depend on temperature and additive properties.
- OIT results may not directly reflect real-life product performance, especially when volatile antioxidants are involved.
- There are no universally accepted sampling procedures or direct correlations between OIT values of field samples and unused products.
Applications
Manufacturing Quality Assurance:
OIT testing is widely used by manufacturers of plastics, geomembranes, films, and cable insulation to verify the sufficient incorporation of stabilizers in polyolefin resins before processing.Supplier Evaluation:
Resin buyers and converters use OIT results to compare batches, select suppliers, and ensure material consistency over time.Material Formulation and Research:
Polymer scientists employ OIT to optimize antioxidant packages and resin formulations aimed at improving thermal and oxidative stability.Industry Sectors:
- Packaging
- Wire and cable insulation and sheathing
- Geomembranes and barrier films
- Automotive and construction plastics
Related Standards
ISO 11357-6
(Plastics-Differential scanning calorimetry (DSC)-Part 6: Determination of oxidation induction time (isothermal OIT) and oxidation induction temperature (dynamic OIT)):
This international standard uses a similar approach for determining OIT, but technical details may differ from ASTM D3895.ASTM D4703
Practice for Compression Molding Thermoplastic Materials into Test Specimens, Plaques, or SheetsASTM E473
Terminology Relating to Thermal Analysis and RheologyASTM E691
Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test MethodASTM E967
Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
Summary
ASTM D3895-19 is an essential standard for evaluating the oxidative induction time of polyolefins, supporting robust quality control and informed material selection. By standardizing OIT measurements via DSC, this method helps stakeholders in the plastics industry maintain product consistency and optimize the long-term performance of polyolefin-based products. For full compliance and accurate data interpretation, users should refer directly to the latest official ASTM D3895 standard.
Buy Documents
ASTM D3895-19 - Standard Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry
REDLINE ASTM D3895-19 - Standard Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

Smithers Quality Assessments
US management systems and product certification.

DIN CERTCO
DIN Group product certification.
Sponsored listings
Frequently Asked Questions
ASTM D3895-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 The OIT is a qualitative assessment of the level (or degree) of stabilization of the material tested. This test has the potential to be used as a quality control measure to monitor the stabilization level in formulated resin as received from a supplier, prior to extrusion. Note 2: The OIT measurement is an accelerated thermal-aging test and as such can be misleading. Caution should be exercised in data interpretation since oxidation reaction kinetics are a function of temperature and the inherent properties of the additives contained in the sample. For example, OIT results are often used to select optimum resin formulations. Volatile antioxidants may generate poor OIT results even though they may perform adequately at the intended use temperature of the finished product. Note 3: There is no accepted sampling procedure, nor have any definitive relationships been established for comparing OIT values on field samples to those on unused products, hence the use of such values for determining life expectancy is uncertain and subjective. SCOPE 1.1 This test method outlines a procedure for the determination of oxidative-induction time (OIT) of polymeric materials by differential scanning calorimetry (DSC). It is applicable to polyolefin resins that are in a fully stabilized/compounded form. 1.2 The values stated in SI units are to be regarded as the standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards information is given in Section 8. Note 1: This standard and ISO 11357–6 2013 address the same subject matter, but differ in technical content. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The OIT is a qualitative assessment of the level (or degree) of stabilization of the material tested. This test has the potential to be used as a quality control measure to monitor the stabilization level in formulated resin as received from a supplier, prior to extrusion. Note 2: The OIT measurement is an accelerated thermal-aging test and as such can be misleading. Caution should be exercised in data interpretation since oxidation reaction kinetics are a function of temperature and the inherent properties of the additives contained in the sample. For example, OIT results are often used to select optimum resin formulations. Volatile antioxidants may generate poor OIT results even though they may perform adequately at the intended use temperature of the finished product. Note 3: There is no accepted sampling procedure, nor have any definitive relationships been established for comparing OIT values on field samples to those on unused products, hence the use of such values for determining life expectancy is uncertain and subjective. SCOPE 1.1 This test method outlines a procedure for the determination of oxidative-induction time (OIT) of polymeric materials by differential scanning calorimetry (DSC). It is applicable to polyolefin resins that are in a fully stabilized/compounded form. 1.2 The values stated in SI units are to be regarded as the standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards information is given in Section 8. Note 1: This standard and ISO 11357–6 2013 address the same subject matter, but differ in technical content. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D3895-19 is classified under the following ICS (International Classification for Standards) categories: 83.080.20 - Thermoplastic materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3895-19 has the following relationships with other standards: It is inter standard links to ASTM E473-23b, ASTM E2935-17, ASTM E2935-16, ASTM E2935-15, ASTM E2935-14, ASTM E473-14, ASTM E2935-13, ASTM E691-13, ASTM E691-11, ASTM E473-11a, ASTM E473-11, ASTM D4703-10, ASTM E473-10, ASTM E473-09, ASTM E691-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3895-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D3895 − 19
Standard Test Method for
Oxidative-Induction Time of Polyolefins by Differential
Scanning Calorimetry
This standard is issued under the fixed designation D3895; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* tial Scanning Calorimeters and Differential Thermal Ana-
lyzers
1.1 This test method outlines a procedure for the determi-
E2935 Practice for Evaluating Equivalence of Two Testing
nation of oxidative-induction time (OIT) of polymeric materi-
Processes
als by differential scanning calorimetry (DSC). It is applicable
to polyolefin resins that are in a fully stabilized/compounded
3. Terminology
form.
3.1 Definitions—Definitions of terms applying to thermal
1.2 The values stated in SI units are to be regarded as the
analysis appear in Terminology E473.
standard.
3.2 Definitions of Terms Specific to This Standard:
1.3 This standard does not purport to address all of the
3.2.1 oxidative induction time—a relative measure of a
safety concerns, if any, associated with its use. It is the
material’s resistance to oxidative decomposition; it is deter-
responsibility of the user of this standard to establish appro-
mined by the thermoanalytical measurement of the time
priate safety, health, and environmental practices and deter-
interval to onset of exothermic oxidation of a material at a
mine the applicability of regulatory limitations prior to use.
specified temperature in an oxygen atmosphere.
Specific hazards information is given in Section 8.
3.2.2 Abbreviations:
NOTE 1—This standard and ISO 11357–6 2013 address the same subject
3.2.3 HDPE—high-density polyethylene.
matter, but differ in technical content.
3.2.4 LDPE—low-density polyethylene.
1.4 This international standard was developed in accor-
3.2.5 LLDPE—linear low-density polyethylene.
dance with internationally recognized principles on standard-
3.2.6 OIT—oxidative induction time.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
4. Summary of Test Method
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
4.1 The sample to be tested and the reference material are
heated at a constant rate in an inert gaseous environment
2. Referenced Documents
(nitrogen). When the specified temperature has been reached,
the atmosphere is changed to oxygen maintained at the same
2.1 ASTM Standards:
flow rate. The specimen is then held at constant temperature
D4703 Practice for Compression Molding Thermoplastic
until the oxidative reaction is displayed on the thermal curve.
Materials into Test Specimens, Plaques, or Sheets
The OIT is determined from the data recorded during the
E473 Terminology Relating to Thermal Analysis and Rhe-
isothermal period. The time interval from when the oxygen
ology
flow is first initiated to the oxidative reaction is referred to as
E691 Practice for Conducting an Interlaboratory Study to
the induction period.
Determine the Precision of a Test Method
4.1.1 The end of the induction period is signaled by an
E967 Test Method for Temperature Calibration of Differen-
abrupt increase in the specimen’s evolved heat or temperature
and will be recorded as an exothermic event by a differential
scanning calorimeter (DSC).
This test method is under the jurisdiction of ASTM Committee D20 on Plastics
and is the direct responsibility of Subcommittee D20.30 on Thermal Properties.
4.2 The type of containment system used depends on the
Current edition approved May 1, 2019. Published June 2019. Originally
intended application use of the material being tested. Polyole-
approved in 1980. Last previous edition approved in 2014 as D3895 – 14. DOI:
10.1520/D3895-19.
fins used in the wire and cable industry typically require copper
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
or aluminum pans, whereas polyolefins used in geomembrane
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
and vapor-barrier film applications exclusively use aluminum
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. pans.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3895 − 19
conditioning (degreasing) are given in Annex A2 – Annex A4.
4.3 Unless otherwise specified, the analysis temperature
NOTE 7—The material composition of the specimen holder can influ-
used in this test is set at 200.0°C. For samples that have
ence the OIT test result significantly (that is, including any associated
relatively low or high stabilization levels, it is permissible to
catalytic effects).
adjust the temperature (typically between 180 and 220°C) to
6.9 Compression-Molding Device with Heated Platens.
yield a thermal curve that can be interpreted and analyzed
easily. 6.10 Spacer Plates, Shim Stock, Caul Plates, etc.
6.11 Polyethylene Terephthalate Film (PET) or Polytet-
5. Significance and Use
rafluoroethylene (PTFE) Coated Cloth, for sample-plaque
5.1 The OIT is a qualitative assessment of the level (or
preparation.
degree) of stabilization of the material tested. This test has the
6.12 Thickness Gauge.
potential to be used as a quality control measure to monitor the
6.13 Laboratory Gas Burner, for copper-pan oxidation.
stabilization level in formulated resin as received from a
supplier, prior to extrusion.
6.14 Boiling Flask, with condenser and heating mantle.
NOTE 2—The OIT measurement is an accelerated thermal-aging test
6.15 Forced-Air Oven.
and as such can be misleading. Caution should be exercised in data
interpretation since oxidation reaction kinetics are a function of tempera-
7. Reagents and Materials
ture and the inherent properties of the additives contained in the sample.
7.1 All chemical reagents used in this procedure shall be
For example, OIT results are often used to select optimum resin
formulations. Volatile antioxidants may generate poor OIT results even analytical grade unless otherwise specified.
though they may perform adequately at the intended use temperature of
7.2 Oxygen—Ultra-high-purity grade (extra dry).
the finished product.
NOTE 3—There is no accepted sampling procedure, nor have any
7.3 Nitrogen—Ultra-high-purity grade (extra dry).
definitive relationships been established for comparing OIT values on field
7.4 Aluminum Pan Degreasing Solvent.
samples to those on unused products, hence the use of such values for
determining life expectancy is uncertain and subjective.
7.5 Indium (99.999 % purity).
6. Apparatus
7.6 Tin (99.999 % purity).
6.1 Differential Scanning Calorimeter—As a minimum
8. Hazards
requirement, the thermal analysis equipment shall be capable
8.1 Oxygen is a strong oxidizer that accelerates combustion
of measuring heat flow of at least 10-mW full scale. The
vigorously. Keep oil and grease away from equipment using or
instrument recorder shall be capable of displaying heat flow or
containing oxygen.
temperature differential on the Y-axis and time on the X-axis.
The time base must be accurate to 61 % and be readable to 0.1 8.2 The use of pressurized gas requires safe and proper
handling.
min.
NOTE 4—The OIT test is a function of a particular compound’s
9. Sampling
stabilizer system and should not be used as a basis of comparison between
9.1 The following sample preparation procedures are rec-
formulations that might contain, different resins, stabilizers, or additive
ommended: the test sample is compression molded into sheet
packages, or all of these.
format (thickness of 250 6 15 μm) prior to analysis to yield
6.2 Gas-Selector Switch and Regulators, for high-purity
consistent sample morphology and weight. Specimen disks cut
nitrogen and oxygen.The distance between the gas-switching
from the sheet using 6.4-mm diameter bore-hole cutter will
point and the instrument cell shall be such that the time
have a weight of approximately 5 to 10 mg, depending on
required to transition to an oxygen environment is less than one
sample density.
minute. At a flow rate of 50 mL/min, this equates to a
maximum switching volume of less than 50 mL.
NOTE 8—If the sample requires homogenization prior to analysis, the
melt compounding procedure given in Appendix X1 is recommended.
6.3 Analytical Balance, 0.1-mg sensitivity.
Poor sample uniformity will affect test precision adversely.
6.4 Bore-Hole Cutter, 6.4-mm diameter.
9.1.1 Obtain the required mass of the sample and place the
6.5 Specimen-Encapsulating Press. material in the center of the appropriately sized spacer between
two sheets of Polyethylene Terephthalate or PTFE coated cloth
6.6 Forceps, Scalpel, and Cutting Board.
and two caul plates.
6.7 Electronic Mass Flow Controller, Rotometer (Cali-
9.1.2 Place the assembly into the compression-molding
brated) or Soap-Film Flowmeter, for gas-flow calibration.
device. The preheat and pressing temperature is 160°C for
polyethylene and 190°C for polypropylene.
6.8 Specimen or Sample Pans—Degreased aluminum or
9.1.3 Heat the sample with appropriate pressure and time
oxidized-copper pans (6.0 to 7.0-mm diameter, 1.5-mm
settings to obtain a plaque with uniform thickness.
height). Use the appropriate pan type for the material being
9.1.4 Remove the plaque assembly and place it between two
tested.
thick steel plates (heat sink) and cool the plaque to ambient
NOTE 5—Aluminum lids are required for temperature calibration.
temperature. Alternatively, quench the plaque in ice water.
NOTE 6—Both types of pans are commercially available. Alternatively,
9.1.5 Determine the average thickness of the sheet to ensure
the copper pans can be fabricated manually. Details on copper pan
preparation and oxidation as well as instructions for aluminum pan that it is within the allowable tolerances.
D3895 − 19
FIG. 1 Indium and Tin Melting Thermal Curves
9.1.6 Use the bore-hole cutter to punch out a disk from the extended baseline and the extended tangent to the first slope of
plaque and record the specimen weight. the endotherm (that is, the onset). See Fig. 1.
9.1.7 Place the specimen disk into the appropriate pan type.
NOTE 11—An inadequate melting thermal curve is occasionally ob-
Use an identical empty pan as the reference. (Do not crimp or
tained due to poor surface contact of the calibrant material to the pan
seal the pans.)
surface. If this occurs, repeat the calibration step. (After one melting/
crystallization cycle the calibrant material should coat the bottom of the
NOTE 9—If controlled cooling is not necessary, the option to prepare the
pan evenly.)
test sample using Practice D4703, Annex 1 (Procedure C), is acceptable.
10.2 Instrument Operation:
10. Procedure
10.2.1 Load the specimen and reference pans into the cell.
10.1 Instrumental Calibration—This procedure uses a two-
10.2.2 Allow 5 min for a nitrogen prepurge prior to begin-
point calibration step. Indium and tin are used as the calibrants
ning the heating cycle to eliminate any residual oxygen.
since their respective melting points encompass the specified
Commence programmed heating of the specimen (under nitro-
analysis temperature range (180 to 220°C). Calibrate the
gen flow of 50 6 5 mL ⁄min) from ambient temperature to
instrument in accordance with the manufacturer’s instructions
200°C (set point) at a rate of 20°C/min.
using the following procedure. Calibrate the instrument at least
10.2.3 When the set temperature has been reached, discon-
once per month or before use if longer than one month.
tinue programmed heating and equilibrate the sample for 5 min
10.1.1 Place 5 6 0.5 mg of indium/tin into an aluminum
at the set temperature. Turn on the recorder. If the instrument
sample pan. Place an aluminum cover over the pan, and seal
being used does not have an isothermal temperature-control-
using the encapsulating press. Prepare an empty sealed pan to
mode feature, follow the alternate procedure outlined in Annex
be used as the reference. Place the specimen and reference pans
A1 or alternatively ensure accurate temperature control by
into their respective locations in the instrument cell.
monitoring and adjusting continually, as required.
10.1.2 Turn on the nitrogen-gas flow at a rate of 50 mL/min
10.2.4 Once the equilibrium time has expired, change the
(with an absolute pressure of 140 kPa).
gas to oxygen at a flow rate of 50 6 mL ⁄min. (Record this
10.1.3 Use the following melting profiles:
event.) This c
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D3895 − 14 D3895 − 19
Standard Test Method for
Oxidative-Induction Time of Polyolefins by Differential
Scanning Calorimetry
This standard is issued under the fixed designation D3895; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method outlines a procedure for the determination of oxidative-induction time (OIT) of polymeric materials by
differential scanning calorimetry (DSC). It is applicable to polyolefin resins that are in a fully stabilized/compounded form.
1.2 The values stated in SI units are to be regarded as the standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. Specific hazards information is given in Section 8.
NOTE 1—This standard and ISO 11357–6 2013 address the same subject matter, but differ in technical content.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D4703 Practice for Compression Molding Thermoplastic Materials into Test Specimens, Plaques, or Sheets
E473 Terminology Relating to Thermal Analysis and Rheology
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E2935 Practice for Conducting Equivalence Testing in Laboratory Applications
3. Terminology
3.1 Definitions—Definitions of terms applying to thermal analysis appear in Terminology E473.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 oxidative induction time—a relative measure of a material’s resistance to oxidative decomposition; it is determined by the
thermoanalytical measurement of the time interval to onset of exothermic oxidation of a material at a specified temperature in an
oxygen atmosphere.
3.2.2 Abbreviations:
3.2.3 HDPE—high-density polyethylene.
3.2.4 LDPE—low-density polyethylene.
3.2.5 LLDPE—linear low-density polyethylene.
3.2.6 OIT—oxidative induction time.
4. Summary of Test Method
4.1 The sample to be tested and the reference material are heated at a constant rate in an inert gaseous environment (nitrogen).
When the specified temperature has been reached, the atmosphere is changed to oxygen maintained at the same flow rate. The
specimen is then held at constant temperature until the oxidative reaction is displayed on the thermal curve. The OIT is determined
This test method is under the jurisdiction of ASTM Committee D20 on Plastics and is the direct responsibility of Subcommittee D20.30 on Thermal Properties.
Current edition approved Dec. 1, 2014May 1, 2019. Published January 2015June 2019. Originally approved in 1980. Last previous edition approved in 20072014 as
D3895 – 07.D3895 – 14. DOI: 10.1520/D3895-14.10.1520/D3895-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3895 − 19
from the data recorded during the isothermal period. The time interval from when the oxygen flow is first initiated to the oxidative
reaction is referred to as the induction period.
4.1.1 The end of the induction period is signaled by an abrupt increase in the specimen’s evolved heat or temperature and will
be recorded as an exothermic event by a differential scanning calorimeter (DSC).
4.2 The type of containment system used depends on the intended application use of the material being tested. Polyolefins used
in the wire and cable industry typically require copper or aluminum pans, whereas polyolefins used in geomembrane and
vapor-barrier film applications exclusively use aluminum pans.
4.3 Unless otherwise specified, the analysis temperature used in this test has been is set arbitrarily at 200.0°C. For samples that
have relatively low or high stabilization levels, it is possiblepermissible to adjust the temperature (typically between 180 and
220°C) to yield a thermal curve that can be interpreted and analyzed easily.
5. Significance and Use
5.1 The OIT is a qualitative assessment of the level (or degree) of stabilization of the material tested. This test has the potential
to be used as a quality control measure to monitor the stabilization level in formulated resin as received from a supplier, prior to
extrusion.
NOTE 2—The OIT measurement is an accelerated thermal-aging test and as such can be misleading. Caution should be exercised in data interpretation
since oxidation reaction kinetics are a function of temperature and the inherent properties of the additives contained in the sample. For example, OIT
results are often used to select optimum resin formulations. Volatile antioxidants may generate poor OIT results even though they may perform adequately
at the intended use temperature of the finished product.
NOTE 3—There is no accepted sampling procedure, nor have any definitive relationships been established for comparing OIT values on field samples
to those on unused products, hence the use of such values for determining life expectancy is uncertain and subjective.
6. Apparatus
6.1 Differential Scanning Calorimeter—As a minimum requirement, the thermal analysis equipment shall be capable of
measuring heat flow of at least 10-mW full scale. The instrument recorder shall be capable of displaying heat flow or temperature
differential on the Y-axis and time on the X-axis. The time base must be accurate to 61 % and be readable to 0.1 min.
NOTE 4—The OIT test is a function of a particular compound’s stabilizer system and should not be used as a basis of comparison between formulations
that might contain, different resins, stabilizers, or additive packages, or all of these.
6.2 Gas-Selector Switch and Regulators, for high-purity nitrogen and oxygen.The distance between the gas-switching point and
the instrument cell shall be such that the time required to transition to an oxygen environment is less than one minute. At a flow
rate of 50 mL/min, this equates to a maximum switching volume of less than 50 mL.
6.3 Analytical Balance, 0.1-mg sensitivity.
6.4 Bore-Hole Cutter, 6.4-mm diameter.
6.5 Specimen-Encapsulating Press.
6.6 Forceps, Scalpel, and Cutting Board.
6.7 Electronic Mass Flow Controller, Rotometer (Calibrated) or Soap-Film Flowmeter, for gas-flow calibration.
6.8 Specimen Holders—or Sample Pans—Degreased aluminum or oxidized-copper pans (6.0 to 7.0-mm diameter, 1.5-mm
height). Use the appropriate pan type for the material being tested.
NOTE 5—Aluminum lids are required for temperature calibration.
NOTE 6—Both types of pans are commercially available. Alternatively, the copper pans can be fabricated manually. Details on copper pan preparation
and oxidation as well as instructions for aluminum pan conditioning (degreasing) are given in Annex A2 – Annex A4.
NOTE 7—The material composition of the specimen holder can influence the OIT test result significantly (that is, including any associated catalytic
effects).
6.9 Compression-Molding Device with Heated Platens.
6.10 Spacer Plates, Shim Stock, Caul Plates, etc.
6.11 Polyethylene Terephthalate Film (PET) or Polytetrafluoroethylene (PTFE) Coated Cloth, for sample-plaque preparation.
6.12 Thickness Gauge.
6.13 Laboratory Gas Burner, for copper-pan oxidation.
6.14 Boiling Flask, with condenser and heating mantle.
6.15 Forced-Air Oven.
7. Reagents and Materials
7.1 All chemical reagents used in this procedure shall be analytical grade unless otherwise specified.
7.2 Oxygen—Ultra-high-purity grade (extra dry).
D3895 − 19
7.3 Nitrogen—Ultra-high-purity grade (extra dry).
7.4 Aluminum Pan Degreasing Solvent.
7.5 Indium (99.999 % purity).
7.6 Tin (99.999 % purity).
8. Hazards
8.1 Oxygen is a strong oxidizer that accelerates combustion vigorously. Keep oil and grease away from equipment using or
containing oxygen.
8.2 The use of pressurized gas requires safe and proper handling.
9. Sampling
9.1 The following sample preparation procedures are recommended: the test sample is compression molded into sheet format
(thickness of 250 6 15 μm) prior to analysis to yield consistent sample morphology and weight. Specimen disks (6.4-mm
diameter) cut from the sheet using 6.4-mm diameter bore-hole cutter will have a weight of approximately 5 to 10 mg, depending
on sample density.
NOTE 8—If the sample requires homogenization prior to analysis, the melt compounding procedure given in Appendix X1 is recommended. Poor
sample uniformity will affect test precision adversely.
9.1.1 Obtain the required mass of the sample and place the material in the center of the appropriately sized spacer between two
sheets of Polyethylene Terephthalate or PTFE coated cloth and two caul plates.
9.1.2 Place the assembly into the compression-molding device. The preheat and pressing temperature is 160°C for polyethylene
and 190°C for polypropylene.
9.1.3 Heat the sample with appropriate pressure and time settings to obtain a plaque with uniform thickness.
9.1.4 Remove the plaque assembly and place it between two thick steel plates (heat sink) and cool the plaque to ambient
temperature. Alternatively, quench the plaque in ice water.
9.1.5 Determine the average thickness of the sheet to ensure that it is within the allowable tolerances.
9.1.6 Use the bore-hole cutter to punch out a disk from the plaque and record the specimen weight.
9.1.7 Place the specimen disk into the appropriate pan type. Use an identical empty pan as the reference. (Do not crimp or seal
the pans.)
NOTE 9—If controlled cooling is not necessary, the option to prepare the test sample using Practice D4703, Annex 1 (Procedure C), is acceptable.
10. Procedure
10.1 Instrumental Calibration—This procedure uses a two-point calibration step. Indium and tin are used as the calibrants since
their respective melting points encompass the specified analysis temperature range (180 to 220°C). Calibrate the instrument in
accordance with the manufacturer’s instructions using the following procedure. Calibrate the instrument at least once per month
or before use if longer than one month.
10.1.1 Place 5 6 0.5 mg of indium/tin into an aluminum sample pan. Place an aluminum cover over the pan, and seal using the
encapsulating press. Prepare an empty sealed pan to be used as the reference. Place the specimen and reference pans into their
respective locations in the instrument cell.
10.1.2 Turn on the nitrogen-gas flow at a rate of 50 mL/min (with an absolute pressure of 140 kPa).
10.1.3 Use the following melting profiles:
Indium: = ambient to 145°C at 10°C/min, 145 to 165°C at 1°C/min
Tin: = ambient to 220°C at 10°C/min, 220 to 240°C at 1°C/min
NOTE 10—The specified heating rates are for calibration use only.
10.1.4 Adjust the temperature-calibration software (or potentiometer) to set the melting point at 156.63 and 231.97°C (see
Practice E967) for indium and tin, respectively. The melting point of the calibrant is defined as the intercept of the extended
baseline and the extended tangent to the first slope of the endotherm (that is, the onset). See Fig. 1.
NOTE 11—An inadequate melting thermal curve is occasionally obtained due to poor surface contact of the calibrant material to the pan surface. If this
occurs, repeat the calibration step. (After one melting/crystallization cycle the calibrant material should coat the bottom of the pan evenly.)
10.2 Instrument Operation:
10.2.1 Load the specimen and reference pans into the cell.
D3895 − 19
FIG. 1 Indium and Tin Melting Thermal Curves
10.2.2 Allow 5 min for a nitrogen prepurge prior to beginning the heating cycle to eliminate any residual oxygen. Commence
programmed heating of the specimen (under nitrogen flow of 50 6 5 mL ⁄min) from ambient temperature to 200°C (set point) at
a rate of 20°C/min.
10.2.3 When the set temperature has been reached, discontinue programmed heating and equilibrate the sample for 5 min at the
set temperature. Turn on the recorder. If the instrument being used does not have an isothermal temperature-control-mode feature,
follow the alternate procedure outlined in Annex A1 or alternatively ensure accurate temperature control by monitoring and
adjusting continually, as required.
10.2.4 Once the equilibrium time has expired, change the gas to oxygen at a flow rate of 50 6 mL ⁄min. (Record this event.)
This changeover point to oxygen flow is considered the zero time of the experiment.
10.2.5 Continue isothermal operation until the maximum exotherm has been reached to allow a complete examination of the
entire exotherm. (see Fig. 2
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...