ASTM E3174-22
(Practice)Standard Practice for Determination of Kinetic Reaction Model Using Differential Scanning Calorimetry
Standard Practice for Determination of Kinetic Reaction Model Using Differential Scanning Calorimetry
SIGNIFICANCE AND USE
5.1 Information concerning the reaction model aids in the selection of the appropriate method (and test method) for evaluation of kinetic parameters. nth order reaction may be treated by isoconversion methods such as Test Methods E698 and E2890. Autocatalytic reactions are treated by Test Methods E2070.
5.2 This practice may be used in research, forensic analysis, trouble shooting, product evaluation, and hazard potential evaluation.
SCOPE
1.1 This practice describes a procedure for determining the “model” of an exothermic reaction using differential scanning calorimetry. The procedure is typically performed on 1 mg to 3 mg specimen sizes over the temperature range from ambient to 600 °C.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2022
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Apr-2018
- Effective Date
- 15-Jan-2018
- Effective Date
- 01-May-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 15-Sep-2013
- Effective Date
- 01-Dec-2012
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Sep-2012
Overview
ASTM E3174-22: Standard Practice for Determination of Kinetic Reaction Model Using Differential Scanning Calorimetry provides a recognized methodology for determining the kinetic reaction model of exothermic chemical reactions by means of differential scanning calorimetry (DSC). This standard practice applies to tests typically conducted on small sample sizes (1 mg to 3 mg) within a temperature range from ambient up to 600 °C. The approach is essential for identifying whether the reaction under study follows an nth order or autocatalytic kinetic model, information that is fundamental for further kinetic parameter evaluation, method selection, and safety assessments.
Key Topics
- Kinetic Reaction Models: Differentiates between nth order and autocatalytic reactions, which are critical in understanding reaction mechanisms and rates.
- Differential Scanning Calorimetry (DSC): Describes the use of DSC instrumentation for precise thermal measurements, aiding in the interpretation and classification of reaction behavior.
- Specimen Handling: Outlines preparation protocols for representative small-scale samples, emphasizing the minimization of thermal gradients and avoidance of hazards.
- Calibration and Accuracy: Stresses the importance of calibrating temperature and heat flow using referenced ASTM methods to ensure reliable and standardized results.
- Safety Considerations: Highlights necessary precautions due to the potential hazards of testing thermally unstable or volatile materials.
Applications
ASTM E3174-22 is valuable across various industries where kinetic modeling of exothermic reactions is required. Key applications include:
- Research and Development: Utilized by laboratories and researchers to investigate reaction mechanisms and optimize chemical processes.
- Forensic Analysis: Supports the identification and investigation of unknown substances based on their thermal decompositional behavior.
- Troubleshooting: Assists chemical engineers and quality assurance professionals in diagnosing issues within production systems by revealing thermal instabilities.
- Product Evaluation: Enables material scientists and manufacturers to evaluate product formulations for stability and reaction hazards.
- Hazard Potential Assessment: Provides safety professionals with kinetic model data vital to risk assessment and safe handling guidelines for chemicals, especially those prone to exothermic runaway reactions.
Related Standards
Several ASTM standards are frequently referenced alongside E3174-22 to ensure comprehensive analysis and calibration when determining reaction kinetics via DSC:
- ASTM E698: Test Method for Kinetic Parameters for Thermally Unstable Materials Using DSC (Flynn/Wall/Ozawa Method)
- ASTM E2890: Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by DSC Using the Kissinger and Farjas Methods
- ASTM E2070: Test Methods for Kinetic Parameters by DSC Using Isothermal Methods - especially for autocatalytic reactions
- ASTM E537: Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry - often used for initial scouting runs
- ASTM E967 and E968: Methods for temperature and heat flow calibration, ensuring accuracy in DSC measurements
- ASTM E3142: Guide for measuring thermal lag of analysis apparatus
- ASTM E473 and E1142: Terminology for consistency in reporting and documentation
Practical Value
ASTM E3174-22 delivers a robust, standardized framework for identifying reaction models, contributing to improved process safety, product reliability, and regulatory compliance. By following this practice, organizations can decisively select appropriate kinetic evaluation methods and mitigate risks linked to thermally unstable or exothermic materials. This standard is an essential resource for professionals in chemical engineering, materials science, quality control, and industrial safety seeking to leverage DSC data for kinetic analysis.
Keywords: ASTM E3174-22, kinetic reaction model, differential scanning calorimetry, DSC, nth order reaction, autocatalytic reaction, reaction kinetics, thermal analysis, kinetic parameter evaluation, chemical safety.
Buy Documents
ASTM E3174-22 - Standard Practice for Determination of Kinetic Reaction Model Using Differential Scanning Calorimetry
REDLINE ASTM E3174-22 - Standard Practice for Determination of Kinetic Reaction Model Using Differential Scanning Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E3174-22 is a standard published by ASTM International. Its full title is "Standard Practice for Determination of Kinetic Reaction Model Using Differential Scanning Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 Information concerning the reaction model aids in the selection of the appropriate method (and test method) for evaluation of kinetic parameters. nth order reaction may be treated by isoconversion methods such as Test Methods E698 and E2890. Autocatalytic reactions are treated by Test Methods E2070. 5.2 This practice may be used in research, forensic analysis, trouble shooting, product evaluation, and hazard potential evaluation. SCOPE 1.1 This practice describes a procedure for determining the “model” of an exothermic reaction using differential scanning calorimetry. The procedure is typically performed on 1 mg to 3 mg specimen sizes over the temperature range from ambient to 600 °C. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Information concerning the reaction model aids in the selection of the appropriate method (and test method) for evaluation of kinetic parameters. nth order reaction may be treated by isoconversion methods such as Test Methods E698 and E2890. Autocatalytic reactions are treated by Test Methods E2070. 5.2 This practice may be used in research, forensic analysis, trouble shooting, product evaluation, and hazard potential evaluation. SCOPE 1.1 This practice describes a procedure for determining the “model” of an exothermic reaction using differential scanning calorimetry. The procedure is typically performed on 1 mg to 3 mg specimen sizes over the temperature range from ambient to 600 °C. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E3174-22 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E3174-22 has the following relationships with other standards: It is inter standard links to ASTM E473-23b, ASTM E2070-23, ASTM E1142-23b, ASTM E537-20, ASTM E2070-13(2018), ASTM E3142-18, ASTM E1142-15, ASTM E1142-14b, ASTM E473-14, ASTM E1142-14a, ASTM E1142-14, ASTM E2070-13, ASTM E537-12, ASTM E1142-12, ASTM E2890-12e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E3174-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E3174 − 22
Standard Practice for
Determination of Kinetic Reaction Model Using Differential
Scanning Calorimetry
This standard is issued under the fixed designation E3174; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* E968 Practice for Heat Flow Calibration of Differential
Scanning Calorimeters
1.1 This practice describes a procedure for determining the
E1142 Terminology Relating to Thermophysical Properties
“model” of an exothermic reaction using differential scanning
E2070 Test Methods for Kinetic Parameters by Differential
calorimetry. The procedure is typically performed on 1 mg to
Scanning Calorimetry Using Isothermal Methods
3 mg specimen sizes over the temperature range from ambient
E2890 Test Method for Determination of Kinetic Parameters
to 600 °C.
and Reaction Order for Thermally Unstable Materials by
1.2 The values stated in SI units are to be regarded as
Differential Scanning Calorimetry Using the Kissinger
standard. No other units of measurement are included in this
and Farjas Methods
standard.
E3142 Test Method for Thermal Lag of Thermal Analysis
1.3 This standard does not purport to address all of the
Apparatus
safety concerns, if any, associated with its use. It is the
3. Terminology
responsibility of the user of this standard to establish appro-
3.1 Definitions—Technical terms used in this standard are
priate safety, health, and environmental practices and deter-
provided in Terminologies E473 and E1142 including:
mine the applicability of regulatory limitations prior to use.
calorimeter, Celsius, derivative, differential scanning
1.4 This international standard was developed in accor-
calorimeter, extrapolated onset, Kelvin, reaction, reaction
dance with internationally recognized principles on standard-
order, and temperature.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
3.2 Definitions of Terms Specific to This Standard:
mendations issued by the World Trade Organization Technical
3.2.1 autocatalytic, n—a chemical reaction where one or
Barriers to Trade (TBT) Committee.
more reaction products are a catalyst for the same reaction.
3.2.1.1 Discussion—An example of an autocatalytic model
2. Referenced Documents
is:
2.1 ASTM Standards:
m n
dα⁄dt 5α k 1 2 α (1)
~ !
E473 Terminology Relating to Thermal Analysis and Rhe-
ology
where:
E537 Test Method for Thermal Stability of Chemicals by
α = the fraction reacted,
Differential Scanning Calorimetry
t = time,
E698 Test Method for Kinetic Parameters for Thermally
k = the rate constant, and
Unstable Materials Using Differential Scanning Calorim-
m and n = the reaction orders.
etry and the Flynn/Wall/Ozawa Method
3.2.2 autocatalytic model, n—a kinetic model used to de-
E967 Test Method for Temperature Calibration of Differen-
scribe an autocatalytic reaction.
tial Scanning Calorimeters and Differential Thermal Ana-
3.2.3 exotherm, n—in thermal analysis, the thermal record
lyzers
of a transition where heat is evolved by the specimen.
This practice is under the jurisdiction of ASTM Committee E37 on Thermal 3.2.4 model, kinetic, n—a mathematical construct used to
Measurements and is the direct responsibility of Subcommittee E37.01 on Calo-
describe the rates of a chemical reaction.
rimetry and Mass Loss.
3.2.5 nth order, n—a kinetic model in which the rate of
CurrenteditionapprovedJune1,2022.PublishedJuly2022.Originallyapproved
in 2019. Last previous edition approved in 2021 as E3174 – 21. DOI: 10.1520/
reaction (dα⁄dt) is proportional to the power of the current
E3174-22.
concentration of the reactant(s).
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
3.2.5.1 Discussion—An example of an nth order model is:
Standards volume information, refer to the standard’s Document Summary page on
n
the ASTM website. dα⁄dt 5 k 1 2 α (2)
~ !
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3174 − 22
where: signals, or both. The minimum output signals required for
differential scanning calorimetry are heat flow, temperature,
α = the fraction reacted,
and time.
t = time,
k = the rate constant, and 6.1.5 Containers (pans, crucibles, vials, etc.) and lids that
n = the reaction order. areinerttothespecimenandreferencematerialsandthatareof
suitable structure, shape and integrity to contain and seal the
3.2.6 rate constant (k), n—a coefficient of proportionality
nominal 5-mg test specimen and reference.
relating the rate of a chemical reaction at a given temperature
to a function of the reactant’s concentration.
6.2 A balance, with a capacity of at least 100 mg, to weigh
specimens and/or containers to within 610 µg.
4. Summary of Practice
6.3 A cooling system, to cool the chamber and the test
4.1 A scouting experiment is conducted at a constant heat-
specimen to ambient temperature at an initial rate of 50 °C/min
ing rate to determine the overall exothermic profile of the
or greater.
chemical reaction. From this thermal curve, the temperature of
maximumrateandtheextrapolatedonset(test)temperatureare 7. Hazards
determined.
7.1 This practice is applicable to thermally unstable
4.2 A fresh test specimen is heated at a slow rate through a materials, the potential hazards for which are unknown. This
small fraction of its reaction exotherm. The test specimen is requires that caution be taken during sample preparation and
testing. The user should avoid test specimens greater than 10
then rapidly cooled to a temperature where the reaction rate
goes to “zero” thereby “quenching” the reaction. The test mgandbeawarethatsamplesizereductiontechniques(suchas
grinding) that may cause localized heating.
specimen is again heated at a slow rate through the whole of
thereactionexotherm.Theshiftornon-shiftofthetemperature
7.2 Toxic or corrosive effluents, or both, may be released
of maximum rate indicates the reaction model.
when heating the specimen that could be harmful to the user or
the apparatus. Use of an exhaust system is recommended to
5. Significance and Use
remove such effluents.
5.1 Information concerning the reaction model aids in the
8. Specimen Preparation
selection of the appropriate method (and test method) for
evaluation of kinetic parameters. nth order reaction may be
8.1 Specimens shall be representative of the sample being
treated by isoconversion methods such as Test Methods E698
studied.
andE2890.AutocatalyticreactionsaretreatedbyTestMethods
8.2 Specimen size shall be kept small to minimize tempera-
E2070.
ture gradient within it. In general, a sample mass resulting in a
5.2 This practice may be used in research, forensic analysis,
maximum heat generation rate of less than 8 mW is satisfac-
trouble shooting, product evaluation, and hazard potential
tory.
evaluation.
NOTE 2—A typical test specimen is 1 mg to 3 mg.
6. Apparatus
8.3 Specimens shall be placed in the containers so that good
thermal contact is achieved between the specimen and con-
6.1 A Differential Scanning Calorimeter (DSC) consisting
tainer and sealed so that evolved gases are retained in the
of:
vicinity of the specimen.
6.1.1 A test chamber consisting of:
6.1.1.1 A furnace or furnaces to provide a uniform con-
8.4 Fig. 1 and Fig. 2 illustrate one acceptable specimen and
trolled heating of a test specimen and reference at a constant
container configuration.
rate of 5 °C/min (61 %) from ambient temperature to 600 °C.
8.5 The specimen container shall be inert (nonreactive) to
6.1.1.2 Atemperature sensor to provide an indication of the
the specimen or its reaction products.
specimen temperature readable to 60.1 °C.
8.6 The reference container is normally an empty container
6.1.1.3 A differential sensor to provide an indication of the
and lid.
difference in heat flow between the specimen and reference
material to within 610 µW.
8.7 Specimens that have appreciable volatility over the
6.1.2 A means of sustaining a test chamber environment of
temperature range of interest may require sealing in hermetic
an inert purge gas at a rate of 10 mL/min to 50 mL/min.
containers or a high-pressure chamber, or both, to prevent
vaporization interference and mass loss of unreacted material.
NOTE 1—Typically 99+ % pure nitrogen, argon, or helium are em-
ployed.
9. Calibration
6.1.3 A temperature controller, capable of executing a
9.1 Perform any
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E3174 − 21 E3174 − 22
Standard Practice for
Determination of Kinetic Reaction Model Using Differential
Scanning Calorimetry
This standard is issued under the fixed designation E3174; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This practice describes a procedure for determining the “model” of an exothermic reaction using differential scanning
calorimetry. The procedure is typically performed on 1 mg to 3 mg specimen sizes over the temperature range from ambient to
600 °C.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E473 Terminology Relating to Thermal Analysis and Rheology
E537 Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry
E698 Test Method for Kinetic Parameters for Thermally Unstable Materials Using Differential Scanning Calorimetry and the
Flynn/Wall/Ozawa Method
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E968 Practice for Heat Flow Calibration of Differential Scanning Calorimeters
E1142 Terminology Relating to Thermophysical Properties
E2070 Test Methods for Kinetic Parameters by Differential Scanning Calorimetry Using Isothermal Methods
E2890 Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by
Differential Scanning Calorimetry Using the Kissinger and Farjas Methods
E3142 Test Method for Thermal Lag of Thermal Analysis Apparatus
3. Terminology
3.1 Definitions—Technical terms used in this standard are provided in Terminologies E473 and E1142 including: calorimeter,
Celsius, derivative, differential scanning calorimeter, extrapolated onset, Kelvin, reaction, reaction order, and temperature.
This practice is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.01 on Calorimetry and
Mass Loss.
Current edition approved May 1, 2021June 1, 2022. Published July 2021July 2022. Originally approved in 2019. Last previous edition approved in 20202021 as E3174
– 20.21. DOI: 10.1520/E3174-21.10.1520/E3174-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3174 − 22
3.2 Definitions of Terms Specific to This Standard:
3.2.1 autocatalytic, n—a chemical reaction where one or more reaction products are a catalyst for the same reaction.
3.2.1.1 Discussion—
An example of an autocatalytic model is:
m n
α⁄dt 5 α k 1 2 α (1)
~ !
m n
dα⁄dt 5 α k~1 2 α! (1)
where:
α = the fraction reacted,
t = time,
k = the rate constant, and
m and n = the reaction orders.
3.2.2 autocatalytic model, n—a kinetic model used to describe an autocatalytic reaction.
3.2.3 exotherm, n—in thermal analysis, the thermal record of a transition where heat is evolved by the specimen.
3.2.4 model, kinetic, n—a mathematical construct used to describe the rates of a chemical reaction.
3.2.5 nth order, n—a kinetic model in which the rate of reaction (dα⁄dt) is proportional to the power of the current concentration
of the reactant(s).
3.2.5.1 Discussion—
An example of an nth order model is:
n
dα⁄dt 5 k 1 2 α (2)
~ !
where
n
dα⁄dt 5 k~1 2 α! (2)
where:
α = the fraction reacted,
t = time,
k = the rate constant, and
n = the reaction order.
α is the fraction reacted, t is time, k is the rate constant, and n is the reaction order.
3.2.6 rate constant (k), n—a coefficient of proportionality relating the rate of a chemical reaction at a given temperature to a
function of the reactant’s concentration.
4. Summary of Practice
4.1 A scouting experiment is conducted at a constant heating rate to determine the overall exothermic profile of the chemical
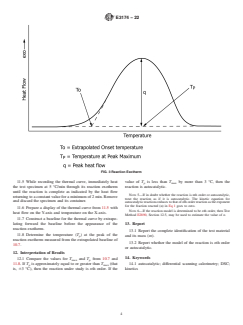
reaction. From this thermal curve, the temperature of maximum rate and the extrapolated onset (test) temperature are determined.
4.2 A fresh test specimen is heated at a slow rate through a small fraction of its reaction exotherm. The test specimen is then
rapidly cooled to a temperature where the reaction rate goes to “zero” thereby “quenching” the reaction. The test specimen is again
heated at a slow rate through the whole of the reaction exotherm. The shift or non-shift of the temperature of maximum rate
indicates the reaction model.
5. Significance and Use
5.1 Information concerning the reaction model aids in the selection of the appropriate method (and test method) for evaluation of
E3174 − 22
kinetic parameters. nth order reaction may be treated by isoconversion methods such as Test Methods E698 and E2890.
Autocatalytic reactions are treated by Test Methods E2070.
5.2 This practice may be used in research, forensic analysis, trouble shooting, product evaluation, and hazard potential evaluation.
6. Apparatus
6.1 A Differential Scanning Calorimeter (DSC) consisting of:
6.1.1 A test chamber consisting of:
6.1.1.1 A furnace or furnaces to provide a uniform controlled heating of a test specimen and reference at a constant rate of 5
°C/min (61 %) from ambient temperature to 600 °C.
6.1.1.2 A temperature sensor to provide an indication of the specimen temperature readable to 60.1 °C.
6.1.1.3 A differential sensor to provide an indication of the difference in heat flow between the specimen and reference material
to within 610 μW.
6.1.2 A means of sustaining a test chamber environment of an inert purge gas at a rate of 10 mL/min to 50 mL/min.
NOTE 1—Typically 99+ % pure nitrogen, argon, or helium are employed.
6.1.3 A temperature controller, capable of executing a specific temperature program by operating the furnace(s) between selected
temperature limits at a rate of 5 °C/min constant to 60.1 °C/min or at an isothermal temperature constant to 60.1 °C.
6.1.4 A data collection device, to provide a means of acquiring, storing, and displaying measured or calculated signals, or both.
The minimum output signals required for differential scanning calorimetry are heat flow, temperature, and time.
6.1.5 Containers (pans, crucibles, vials, etc.) and lids that are inert to the specimen and reference materials and that are of suitable
structure, shape and integrity to contain and seal the nominal 5-mg test specimen and reference.
6.2 A balance, with a capacity of at least 100 mg, to weigh specimens and/or containers to within 610 μg.
6.3 A cooling system, to cool the chamber and the test specimen to ambient temperature at an initial rate of 50 °C/min or greater.
7. Hazards
7.1 This practice is applicable to thermally unstable materials, the potential hazards for which are unknown. This requires that
caution be taken during sample preparation and testing. The user should avoid test specimens greater than 10 mg and be aware
that sample size reduction techniques (such as grinding) that may cause localized heating.
7.2 Toxic or corrosive effluents, or both, may be released when heating the specimen that could be harmful to the user or the
apparatus. Use of an exhaust system is recommended to remove such effluents.
8. Specimen Preparation
8.1 Specimens shall be representative of the sample being studied.
8.2 Specimen size shall be kept small to minimize temperature gradient within it. In general, a sample mass resulting in a
maximum heat generation rate of less than 8 mW is satisfactory.
NOTE 2—A typical test specimen is 1 mg to 3 mg.
8.3 Specimens shall be placed in the containers so that good thermal contact is achieved between the specimen and container and
sealed so that evolved gases are retained in the vicinity of the specimen.
E3174 − 22
8.4 Fig. 1 and Fig. 2 illustrate one acceptable specimen and container configuration.
8.5 The s
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...