ASTM C1753/C1753M-21a
(Practice)Standard Practice for Evaluating Early Hydration of Hydraulic Cementitious Mixtures Using Thermal Measurements
Standard Practice for Evaluating Early Hydration of Hydraulic Cementitious Mixtures Using Thermal Measurements
SIGNIFICANCE AND USE
5.1 This practice provides a means of assessing the relative early hydration performance of various test mixtures compared with control mixtures that are prepared in a similar manner.
5.2 Thermal profiles are used to evaluate the hydration behavior of hydraulic cementitious mixtures after the addition of water. They may provide indications concerning setting characteristics, compatibility of different materials, sulfate balance, relative heat of hydration, and early strength development. They can be used to evaluate the effects of compositions, proportions, and time of addition of materials as well as the initial mixture and test temperatures. Thermal profile testing is an effective tool for identifying performance sensitivities or trends, and may help to reduce the number of concrete test mixtures required to develop and qualify mixtures, especially those to be subject to variable ambient environments. It may be used by concrete producers, materials suppliers, and other practitioners to support mixture development, selection of material types or sources, optimization of proportions, or troubleshooting of field problems.
5.3 This practice can be used to understand concrete problems related to slump loss, setting, and early strength, but results may not predict field concrete performance. Performance verification with concrete is needed to quantify the trends identified using thermal testing.
5.4 This practice can be used to evaluate the effects of chemical admixtures on the thermal profiles of cementitious mixtures. This can be especially useful in selecting dosages appropriate for different ambient conditions.
5.5 Thermal measurement testing as described in this practice may have similar significance and use as isothermal calorimetry described by Practice C1679 or some types of near-adiabatic calorimetry. The selection of which practice or methods to use may depend on specific applications and circumstances. The thermal profiles obtained by this practic...
SCOPE
1.1 This practice describes the apparatus and procedure for evaluating relative differences in early hydration of hydraulic cementitious mixtures such as paste, mortar, or concrete, including those containing chemical admixtures, various supplementary cementitious materials (SCMs), and other finely divided materials, by measuring the temperature history of a specimen.
1.2 Calorimetry is the measurement of heat lost or gained during a chemical reaction such as cement hydration; calorimetric measurements as a function of time can be used to describe and evaluate hydration and related early-age property development. Calorimetry may be performed under isothermal conditions (as described in Practice C1679) or under adiabatic or semi-adiabatic conditions. This practice cannot be described as calorimetry because no attempt is made to measure or compute the heat evolved from test specimens due to hydration, but it can in many cases be used for similar evaluations. Variables that should be considered in the application of this practice are discussed in the Appendix.
1.3 Units—The values stated in either SI units or inch-pound units shall be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system must be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. Some values have only SI units because the inch-pound equivalents are not used in practice.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
Note 1: Warning: Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.2
1.5 This...
General Information
- Status
- Published
- Publication Date
- 30-Nov-2021
- Technical Committee
- C09 - Concrete and Concrete Aggregates
Relations
- Effective Date
- 01-Apr-2024
- Refers
ASTM C39/C39M-23 - Standard Test Method for Compressive Strength of Cylindrical Concrete Specimens - Effective Date
- 01-Dec-2023
- Effective Date
- 01-Aug-2020
- Effective Date
- 15-Jul-2020
- Effective Date
- 01-Apr-2020
- Effective Date
- 15-Dec-2019
- Effective Date
- 15-Nov-2019
- Effective Date
- 01-Jun-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Jul-2018
- Effective Date
- 01-May-2018
- Effective Date
- 01-Jan-2018
- Refers
ASTM C39/C39M-17b - Standard Test Method for Compressive Strength of Cylindrical Concrete Specimens - Effective Date
- 01-Aug-2017
- Effective Date
- 15-Mar-2017
Overview
ASTM C1753/C1753M-21a - Standard Practice for Evaluating Early Hydration of Hydraulic Cementitious Mixtures Using Thermal Measurements establishes protocols for assessing the relative early hydration performance of hydraulic cementitious mixtures, such as paste, mortar, or concrete. This practice utilizes thermal measurements-tracking the temperature history of test specimens-to provide insights into hydration behavior, setting characteristics, sulfate balance, heat evolution, and early strength development.
By comparing thermal profiles of different mixtures under controlled conditions, this standard offers a practical, efficient, and cost-effective tool for concrete producers, materials suppliers, and researchers involved in mixture development, selection of materials and proportions, and troubleshooting.
Key Topics
- Early Hydration Assessment:
Measures relative differences in early hydration between test mixtures and control mixtures. - Thermal Profile Analysis:
Monitors temperature changes after mixing water with cementitious materials to evaluate reactions leading to setting and strength development. - Influence of Variables:
Assesses the impact of chemical admixtures, supplementary cementitious materials (SCMs), composition, proportions, and temperatures on hydration kinetics. - Sulfate Balance Evaluation:
Identifies mixture compositions close to sulfate imbalance threshold to avoid abnormal hydration behaviors. - Performance Trends:
Detects trends such as retarding or accelerating effects, compatibility issues, and abnormal behaviors early on. - Reduction of Test Mixtures:
Streamlines the mixture qualification process by minimizing the number of required trial batches, especially for variable field conditions.
Applications
This ASTM standard is widely applicable in the fields of concrete technology, construction, and materials science for:
- Mixture Development and Optimization:
Enables formulating high-performance cementitious mixtures tailored to specific project requirements or environmental conditions. - Material Selection:
Assists in evaluating compatibility and performance of different cements, SCMs, and chemical admixtures. - Quality Control & Troubleshooting:
Provides a rapid assessment method for identifying batch-to-batch variability, potential setting problems, and early strength development issues. - Laboratory and Field Simulation:
Supports laboratory-based simulation of anticipated field conditions to evaluate thermal behavior and hydration kinetics. - Sulfate Balance Studies:
Assists practitioners in adjusting mixtures or dosages to mitigate sulfate imbalance-related problems. - Educational and Research Use:
A valuable tool for researchers and educators when studying hydration reactions and early-age properties of cement-based materials.
Users of This Standard Include:
- Concrete producers and suppliers
- Cement manufacturers
- Materials laboratories
- Construction quality control teams
- Researchers in concrete technology
Related Standards
For a comprehensive approach to evaluating hydraulic cementitious mixtures, consider referencing these related ASTM standards:
- ASTM C1679 - Practice for Measuring Hydration Kinetics via Isothermal Calorimetry
(for quantitative heat evolution measurements) - ASTM C403/C403M - Test Method for Time of Setting by Penetration Resistance
- ASTM C305 - Practice for Mechanical Mixing of Hydraulic Cement Pastes and Mortars
- ASTM C494/C494M - Specification for Chemical Admixtures for Concrete
- ASTM C39/C39M - Test Method for Compressive Strength of Cylindrical Concrete Specimens
- ASTM C192/C192M - Practice for Making and Curing Concrete Test Specimens in the Laboratory
- ASTM C125 & C219 - Terminology Standards for Concrete and Hydraulic Cements
Practical Value
Thermal measurement testing, as outlined in ASTM C1753/C1753M-21a, offers a practical, economical, and informative approach for evaluating early hydration in cementitious mixtures. While it does not replace performance verification with concrete or provide absolute heat evolution values, it serves as an expedient screening method. This makes it particularly valuable for supporting mixture development, material optimization, and quality control in modern concrete production and construction.
Keywords: Early hydration, hydraulic cementitious mixtures, thermal measurements, cement hydration, setting characteristics, sulfate balance, concrete testing, ASTM standards, quality control, admixture evaluation.
Buy Documents
ASTM C1753/C1753M-21a - Standard Practice for Evaluating Early Hydration of Hydraulic Cementitious Mixtures Using Thermal Measurements
REDLINE ASTM C1753/C1753M-21a - Standard Practice for Evaluating Early Hydration of Hydraulic Cementitious Mixtures Using Thermal Measurements
Get Certified
Connect with accredited certification bodies for this standard

ICC Evaluation Service
Building products evaluation and certification.

QAI Laboratories
Building and construction product testing and certification.

Aboma Certification B.V.
Specialized in construction, metal, and transport sectors.
Sponsored listings
Frequently Asked Questions
ASTM C1753/C1753M-21a is a standard published by ASTM International. Its full title is "Standard Practice for Evaluating Early Hydration of Hydraulic Cementitious Mixtures Using Thermal Measurements". This standard covers: SIGNIFICANCE AND USE 5.1 This practice provides a means of assessing the relative early hydration performance of various test mixtures compared with control mixtures that are prepared in a similar manner. 5.2 Thermal profiles are used to evaluate the hydration behavior of hydraulic cementitious mixtures after the addition of water. They may provide indications concerning setting characteristics, compatibility of different materials, sulfate balance, relative heat of hydration, and early strength development. They can be used to evaluate the effects of compositions, proportions, and time of addition of materials as well as the initial mixture and test temperatures. Thermal profile testing is an effective tool for identifying performance sensitivities or trends, and may help to reduce the number of concrete test mixtures required to develop and qualify mixtures, especially those to be subject to variable ambient environments. It may be used by concrete producers, materials suppliers, and other practitioners to support mixture development, selection of material types or sources, optimization of proportions, or troubleshooting of field problems. 5.3 This practice can be used to understand concrete problems related to slump loss, setting, and early strength, but results may not predict field concrete performance. Performance verification with concrete is needed to quantify the trends identified using thermal testing. 5.4 This practice can be used to evaluate the effects of chemical admixtures on the thermal profiles of cementitious mixtures. This can be especially useful in selecting dosages appropriate for different ambient conditions. 5.5 Thermal measurement testing as described in this practice may have similar significance and use as isothermal calorimetry described by Practice C1679 or some types of near-adiabatic calorimetry. The selection of which practice or methods to use may depend on specific applications and circumstances. The thermal profiles obtained by this practic... SCOPE 1.1 This practice describes the apparatus and procedure for evaluating relative differences in early hydration of hydraulic cementitious mixtures such as paste, mortar, or concrete, including those containing chemical admixtures, various supplementary cementitious materials (SCMs), and other finely divided materials, by measuring the temperature history of a specimen. 1.2 Calorimetry is the measurement of heat lost or gained during a chemical reaction such as cement hydration; calorimetric measurements as a function of time can be used to describe and evaluate hydration and related early-age property development. Calorimetry may be performed under isothermal conditions (as described in Practice C1679) or under adiabatic or semi-adiabatic conditions. This practice cannot be described as calorimetry because no attempt is made to measure or compute the heat evolved from test specimens due to hydration, but it can in many cases be used for similar evaluations. Variables that should be considered in the application of this practice are discussed in the Appendix. 1.3 Units—The values stated in either SI units or inch-pound units shall be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system must be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. Some values have only SI units because the inch-pound equivalents are not used in practice. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Note 1: Warning: Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.2 1.5 This...
SIGNIFICANCE AND USE 5.1 This practice provides a means of assessing the relative early hydration performance of various test mixtures compared with control mixtures that are prepared in a similar manner. 5.2 Thermal profiles are used to evaluate the hydration behavior of hydraulic cementitious mixtures after the addition of water. They may provide indications concerning setting characteristics, compatibility of different materials, sulfate balance, relative heat of hydration, and early strength development. They can be used to evaluate the effects of compositions, proportions, and time of addition of materials as well as the initial mixture and test temperatures. Thermal profile testing is an effective tool for identifying performance sensitivities or trends, and may help to reduce the number of concrete test mixtures required to develop and qualify mixtures, especially those to be subject to variable ambient environments. It may be used by concrete producers, materials suppliers, and other practitioners to support mixture development, selection of material types or sources, optimization of proportions, or troubleshooting of field problems. 5.3 This practice can be used to understand concrete problems related to slump loss, setting, and early strength, but results may not predict field concrete performance. Performance verification with concrete is needed to quantify the trends identified using thermal testing. 5.4 This practice can be used to evaluate the effects of chemical admixtures on the thermal profiles of cementitious mixtures. This can be especially useful in selecting dosages appropriate for different ambient conditions. 5.5 Thermal measurement testing as described in this practice may have similar significance and use as isothermal calorimetry described by Practice C1679 or some types of near-adiabatic calorimetry. The selection of which practice or methods to use may depend on specific applications and circumstances. The thermal profiles obtained by this practic... SCOPE 1.1 This practice describes the apparatus and procedure for evaluating relative differences in early hydration of hydraulic cementitious mixtures such as paste, mortar, or concrete, including those containing chemical admixtures, various supplementary cementitious materials (SCMs), and other finely divided materials, by measuring the temperature history of a specimen. 1.2 Calorimetry is the measurement of heat lost or gained during a chemical reaction such as cement hydration; calorimetric measurements as a function of time can be used to describe and evaluate hydration and related early-age property development. Calorimetry may be performed under isothermal conditions (as described in Practice C1679) or under adiabatic or semi-adiabatic conditions. This practice cannot be described as calorimetry because no attempt is made to measure or compute the heat evolved from test specimens due to hydration, but it can in many cases be used for similar evaluations. Variables that should be considered in the application of this practice are discussed in the Appendix. 1.3 Units—The values stated in either SI units or inch-pound units shall be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system must be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. Some values have only SI units because the inch-pound equivalents are not used in practice. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Note 1: Warning: Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.2 1.5 This...
ASTM C1753/C1753M-21a is classified under the following ICS (International Classification for Standards) categories: 91.100.10 - Cement. Gypsum. Lime. Mortar. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1753/C1753M-21a has the following relationships with other standards: It is inter standard links to ASTM C219-24, ASTM C39/C39M-23, ASTM C219-20a, ASTM C305-20, ASTM C219-20, ASTM C125-19a, ASTM C219-19a, ASTM C219-19, ASTM C125-19, ASTM C125-18b, ASTM C125-18a, ASTM C1005-18, ASTM C125-18, ASTM C39/C39M-17b, ASTM C1005-17. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1753/C1753M-21a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:C1753/C1753M −21a
Standard Practice for
Evaluating Early Hydration of Hydraulic Cementitious
Mixtures Using Thermal Measurements
This standard is issued under the fixed designation C1753/C1753M; the number immediately following the designation indicates the
year of original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last
reapproval. A superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
NOTE 1—Warning: Fresh hydraulic cementitious mixtures are caustic
1. Scope*
and may cause chemical burns to skin and tissue upon prolonged
1.1 This practice describes the apparatus and procedure for
exposure.
evaluating relative differences in early hydration of hydraulic
1.5 This international standard was developed in accor-
cementitious mixtures such as paste, mortar, or concrete,
dance with internationally recognized principles on standard-
including those containing chemical admixtures, various
ization established in the Decision on Principles for the
supplementarycementitiousmaterials(SCMs),andotherfinely
Development of International Standards, Guides and Recom-
divided materials, by measuring the temperature history of a
mendations issued by the World Trade Organization Technical
specimen.
Barriers to Trade (TBT) Committee.
1.2 Calorimetry is the measurement of heat lost or gained
2. Referenced Documents
during a chemical reaction such as cement hydration; calori-
metric measurements as a function of time can be used to
2.1 ASTM Standards:
describe and evaluate hydration and related early-age property
C39/C39MTest Method for Compressive Strength of Cylin-
development. Calorimetry may be performed under isothermal
drical Concrete Specimens
conditions (as described in Practice C1679) or under adiabatic
C125Terminology Relating to Concrete and Concrete Ag-
orsemi-adiabaticconditions.Thispracticecannotbedescribed
gregates
as calorimetry because no attempt is made to measure or
C172/C172MPractice for Sampling Freshly Mixed Con-
compute the heat evolved from test specimens due to
crete
hydration, but it can in many cases be used for similar
C192/C192MPracticeforMakingandCuringConcreteTest
evaluations. Variables that should be considered in the appli-
Specimens in the Laboratory
cation of this practice are discussed in the Appendix.
C219Terminology Relating to Hydraulic and Other Inor-
ganic Cements
1.3 Units—The values stated in either SI units or inch-
C305Practice for Mechanical Mixing of Hydraulic Cement
pound units shall be regarded separately as standard. The
Pastes and Mortars of Plastic Consistency
values stated in each system may not be exact equivalents;
C403/C403MTest Method for Time of Setting of Concrete
therefore,eachsystemmustbeusedindependentlyoftheother.
Mixtures by Penetration Resistance
Combining values from the two systems may result in non-
C494/C494MSpecification for Chemical Admixtures for
conformancewiththestandard.SomevalueshaveonlySIunits
Concrete
because the inch-pound equivalents are not used in practice.
C1005Specification for Reference Masses and Devices for
1.4 This standard does not purport to address all of the
DeterminingMassandVolumeforUseinPhysicalTesting
safety concerns, if any, associated with its use. It is the
of Hydraulic Cements
responsibility of the user of this standard to establish appro-
C1679Practice for Measuring Hydration Kinetics of Hy-
priate safety, health, and environmental practices and deter-
draulic Cementitious Mixtures Using Isothermal Calorim-
mine the applicability of regulatory limitations prior to use.
etry
1 2
This practice is under the jurisdiction of ASTM Committee C09 on Concrete Section on Safety Precautions, Manual of Aggregate and Concrete Testing,
andConcreteAggregatesandisthedirectresponsibilityofSubcommitteeC09.48on Annual Book of ASTM Standards, Vol. 04.02.
Performance of Cementitious Materials and Admixture Combinations. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2021. Published January 2022. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2015. Last previous edition approved in 2021 as C1753/C1753M–21. Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/C1753_C1753M-21A. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1753/C1753M−21a
3. Terminology mixture proportions, materials properties, initial mixture
temperature, and test temperature.
3.1 Definitions—For definitions of terms used in this
practice, refer to Terminology C125, Terminology C219, and
3.2.7 sulfate imbalance threshold, n—the condition of a
Practice C1679. cementitious mixture in terms of mixture proportions, materi-
als properties, initial mixture temperature, and test
3.2 Definitions of Terms Specific to This Standard:
temperature,forwhichasmallchangeinanyofthesevariables
3.2.1 adiabatic, adj—occurring without exchange of heat
can result in abnormal hydration behavior due to depletion of
with the environment.
calcium sulfate in solution.
3.2.2 exotherm, n—heat evolution during hydration as evi-
3.2.8 test specimen, n—a hydraulic cementitious mixture
denced by an increase in measured specimen temperature
being evaluated for its thermal response.
shown in the thermal profile.
3.2.9 test temperature, n—the temperature of the air or
3.2.3 inert specimen, n—specimen placed within the same
insulation, if any, surrounding the test specimen containers at
thermal environment as the test specimen(s), made of a
the start of temperature measurement, normally intended to
nonreactive material of similar heat capacity and the same
remain constant.
mass as the reacting test specimen(s).
3.2.3.1 Discussion—The difference between the tempera-
3.2.10 thermal profile, n—the temperature of a hydrating
ture of the hydrating test specimen(s) and the inert specimen
mixture (before or after subtraction of the reference
represents the change in specimen temperature due to hydra-
temperature), plotted as a function of hydration time, that
tion. Interpretation can often be improved by comparing
provides an indication of the rate of hydration over time.
temperature histories after subtracting the temperature of the
3.2.10.1 Discussion—An example thermal profile is shown
corresponding inert specimen (reference temperature), which
in Fig. 1. On the vertical axis T refers to the temperature of
test
tends to account for the effects of changing environment
the test specimen andT refers to the temperature of the inert
ref
temperature during the measurement period.
(reference) specimen. The shape of the thermal profile is
3.2.4 main peak response, n—the initial temperature rise
affected not only by mixture hydration but also by the
and subsequent temperature drop in the measured thermal
specimen type and mass, mixture proportions, specimen initial
profile that starts at the end of the dormant period and, for a
temperature, specimen container size and shape, insulation (if
mixture with normal sulfate balance, lasts for several hours.
any)providedaroundthespecimencontainer,andthetempera-
ture of the surrounding environment. Additional guidance is
3.2.5 reference temperature, n—the temperature of the inert
provided in the Appendix.
specimen in a test series at the time corresponding to a
particular temperature of the test specimen.
3.2.11 time of setting marker, n—the point marked on the
3.2.6 sulfate demand, n—thelevelofsolublecalciumsulfate thermal profile indicating the hydration time when a selected
fraction of the main peak amplitude is attained and that is used
in a hydrating cementitious mixture required to maintain
normal hydration behavior for a specific combination of as a relative indicator of time of setting.
NOTE 1—(A) initial exotherm from dissolution of cement and initial hydration, principally of calcium aluminates; (B) dormant period temperature
reduction associated with very low heat evolution indicating slow and well-controlled hydration; (C) main peak response associated primarily with
hydration reactions contributing to setting and early strength development, with maximum temperature at (D). The maximum temperature (D) and the
rates of temperature rise and fall that shape the main peak response (C) are affected not only by hydration but by the related cooling response of the
specimen.
FIG. 1Example Thermal Profile of a Portland Cement Paste Mixture (Inert Specimen Temperature Subtracted from Test Specimen Tem-
perature)
C1753/C1753M−21a
4. Summary of Practice may have similar shapes to isothermal hydration profiles as
obtained by Practice C1679, but thermal profiles from this
4.1 Athermalmeasurementtestsystemconsistsoftempera-
practice do not provide quantitative measurement of heat of
ture measuring devices, data collection equipment, and speci-
hydration, are affected by various details of the test conditions
mencontainersofsimilarvolume,shape,andmaterial,capable
and mixtures (see 3.2.10 and theAppendix), and are subject to
ofsimilarlyisolatingeachtestspecimenandaninertspecimen.
greater variability. Equipment used for this practice is less
The specific insulation values for specimen containers and the
expensive than isothermal or near-adiabatic calorimeters and
test temperature are selected based on the intended test
maybemoreeasilyadaptedforuseinthefieldorwherealarge
objectives. Related guidance is provided in the Appendix.
number of different specimens and mixtures must be evaluated
4.2 Mixtures composed of cementitious materials, water,
in a short time period. Identification of the sulfate depletion
and optionally chemical admixtures, or aggregate, or both, are
point of a mixture (as described in Practice C1679)isnot
prepared and introduced into specimen containers for collec-
generally possible using thermal measurement testing.
tion of temperature data.
5.6 To evaluate the potential for abnormal hydration, it is
4.3 Thermal profiles are plotted using a common time scale
importantthatthetesttemperaturesandtheinitialtemperatures
that begins at the time of initial mixing of water with
of the mixture be selected to represent the range of expected
cementitious materials, which is the start of hydration time.
initial concrete field temperatures.
The measured thermal profiles allow qualitative comparison of
5.7 This practice is not intended to provide results that can
early hydration kinetics, such as retarding or accelerating
be compared across laboratories using different equipment nor
trends, as influenced by different combinations of materials,
to provide quantitative measurements or corrected approxima-
and abnormal hydration behaviors that can interfere with
tions of actual hydration heat. It should not be cited in project
setting and strength development.
specifications or otherwise used for the purpose of acceptance
or rejection of concrete. It is intended to serve as a simple and
5. Significance and Use
expedient tool for comparison of the relative early-age hydra-
5.1 This practice provides a means of assessing the relative
tion performance of different specific combinations of materi-
earlyhydrationperformanceofvarioustestmixturescompared
als that are prepared and stored under the same conditions.
with control mixtures that are prepared in a similar manner.
5.2 Thermal profiles are used to evaluate the hydration 6. Apparatus
behavior of hydraulic cementitious mixtures after the addition
6.1 Devices for Preparing Specimens:
of water. They may provide indications concerning setting
6.1.1 Weights and Weighing Devices,usedforpreparationof
characteristics, compatibility of different materials, sulfate
laboratory test mixtures up to 5 kg [11 lb] total mass shall
balance, relative heat of hydration, and early strength devel-
conform to the requirements of Specification C1005. For
opment. They can be used to evaluate the effects of
preparingtestmixturesofgreatertotalmassincludingconcrete
compositions, proportions, and time of addition of materials as
batches in the laboratory, weighing devices shall conform to
well as the initial mixture and test temperatures. Thermal
the requirements of Practice C192/C192M.
profile testing is an effective tool for identifying performance
6.1.2 Graduated Cylinders, shall conform to the require-
sensitivities or trends, and may help to reduce the number of
ments of Specification C1005. The permissible variation for
concrete test mixtures required to develop and qualify
graduated cylinders of less than 100-mL capacity shall be 6
mixtures, especially those to be subject to variable ambient
1.0% of the indicated capacity.
environments. It may be used by concrete producers, materials
6.1.3 Graduated Syringes, if used, shall be of suitable
suppliers, and other practitioners to support mixture
capacities to contain the desired volume of liquid admixture
development, selection of material types or sources, optimiza-
and shall be accurate to 6 3% of the required volume.
tion of proportions, or troubleshooting of field problems.
6.1.4 Mixing Apparatus, capable of producing a uniform
5.3 This practice can be used to understand concrete prob-
mixture.
lems related to slump loss, setting, and early strength, but
6.2 Thermal Measurement Test Equipment and Data Acqui-
results may not predict field concrete performance. Perfor-
sition System—Actual design of the equipment, whether com-
mance verification with concrete is needed to quantify the
mercial or custom-built, may vary, but it shall meet the
trends identified using thermal testing.
following requirements for the selected type, shape, and mass
5.4 This practice can be used to evaluate the effects of
of the specimen, insulation (if any) surrounding the specimen
chemical admixtures on the thermal profiles of cementitious
container, initial mixture temperature, and test temperature.
mixtures. This can be especially useful in selecting dosages
6.2.1 Temperature sensors shall be thermistors or thermo-
appropriate for different ambient conditions.
couples with measurement accuracy of 6 1.0 °C [2 °F].
5.5 Thermal measurement testing as described in this prac- 6.2.2 Thesignal-to-noiseratioshallbeatleast5.0.Signalis
tice may have similar significance and use as isothermal defined as the difference between the highest and the lowest
calorimetry described by Practice C1679 or some types of temperatures measured from the dormant period through the
near-adiabatic calorimetry. The selection of which practice or main peak response (Fig. 1) for a test specimen in the test
methods to use may depend on specific applications and series without admixture or SCMs (Fig. 2). Noise is defined as
circumstances. The thermal profiles obtained by this practice the difference between the highest and the lowest temperatures
C1753/C1753M−21a
FIG. 2Examples of Signal and Noise Determination for Verification of Signal-to-Noise Ratio
measured during the time period in which the signal is ture in the ambient space around the hydrating test specimens
established(Fig.2)foraninertspecimenhavingamasssimilar without the use of forced air circulation. Portable concrete
to that of the test specimens. The inert specimen shall remain cylinder curing boxes that employ circulating temperature-
in the same environment as the test specimens to indicate both conditioned water below a specimen shelf have been success-
the effects of changes in ambient temperature as well as any fully used. Other types of chambers that use forced air in the
thermal influences of adjacent test specimens (see also 6.2.5). regulationofinternaltemperatureshouldnotbeused,however,
asairmovementaroundthespecimencontainerscanadversely
NOTE 2—Exceeding the minimum signal-to-noise ratio is more impor-
influence test results.
tant than specific requirements for insulation value of the specimen
6.2.4 The data acquisition equipment shall be capable of
containerorenvironment(seeAppendixforguidance).Selectedspecimen
containers and insulation configurations (if any) may vary with mixture
performingcontinuousloggingofthetemperatureswithatime
type, specimen mass, and initial mixture and test temperatures. A
interval between recorded measurements not greater than 60 s.
satisfactory inert specimen may be obtained using quantities of sand and
6.2.5 Specimen Containers of volume and insulating value
water having masses within 6 10 % of the combined solids and water
as needed to meet the requirements of 6.2.1 for the test
contents of the test specimens. Thermal influences from other test
specimens may be reduced by providing adequate spaces between
mixtures and conditions that can be sealed while providing
specimens in the test environment, depending on the insulating values of
access for the temperature sensors of the thermal measurement
the specimen containers. The intent of a minimum signal-to-noise ratio
system, if required (see Note 4). For systems without continu-
requirement is to assure a well-defined thermal profile that is minimally
ous insulation between specimen containers, provide a clear
influenced by ambient temperature changes and the presence of other test
specimens.The maximum main peak temperature should be similar to the distance of at least 70 mm [3 in.] between individual specimen
maximumtemperaturethatwouldbeexpectedforin-placeconcreteinthe
containers.
application of interest. Because the type, shape, and mass of the test
6.2.6 The location of temperature sensors relative to speci-
specimen, insulation around the specimen container, and initial mixture
men containers shall be similar for all test specimens and for
and test temperatures all influence main peak response levels, it is
the inert specimen.
important to balance these factors to meet the requirements of 8.2.2
without causing unrealistic main peak response temperatures (see Note 4
and the Appendix for guidance).
7. Materials
6.2.3 The air space or insulation, or both, surrounding the
7.1 Mixture Materials:
specimen containers,whetherthetestspecimenisstoredunder
7.1.1 Mixture materials, including cementitious materials
ambientconditionsorinsideaconditionedchamberintendedto
and admixtures, shall be obtained from the concrete producer,
replicate field conditions of interest, shall be controlled to
or otherwise obtained to be representative of those specific to
ensure that the measured temperature of the inert specimen
the purpose of the test.
(referencetemperature)doesnotvaryfromthetesttemperature
7.2 Calcium Sulfate:
by more than 3 °C [5 °F] during testing, unless deliberate
change of ambient conditions during the period of temperature 7.2.1 Use reagent grade calcium sulfate dihydrate or hemi-
measurement is part of the test program. hydrate prepared from reagent grade calcium sulfate dihydrate
6.2.3.1 A conditioned chamber may be a laboratory test or calcium sulfate anhydrite to verify whether a mixture is in
chamber, portable curing chamber, or other temperature- sulfate balance. See the Appendix for examples of sulfate
controlled container capable of maintaining the test tempera- addition for evaluation of sulfate balance.
C1753/C1753M−21a
7.2.2 It is permissible to use a source-specific calcium 8.3.1 Any effective mixing procedure is allowed; various
sulfate for performing a test series that is related to a specific suitable mixing methods are described in the Appendix. De-
cement production source. pending on the method used, the order of the introduction of
materials to the mixing bowl or container may differ. Dispense
liquid admixtures into mixing water to form a solution before
8. Procedure
introduction into the cementitious materials. The solution
8.1 Temperature Conditions:
containing admixtures may consist of all of the mix water or
8.1.1 Specimen Preparation Temperature—Maintain the
some portion, if admixture addition is to be delayed. Liquid
temperature of the air in the vicinity of all equipment and
admixturesmaybeintroduceddirectlytomixingwaterusinga
materials used in specimen preparation at the test temperature
graduated syringe or obtained from a stock solution at appro-
to within 6 3.0 °C [5 °F].
priate dilution. Inspect stock solutions for separation and
8.1.2 Materials and Initial Mixture Temperatures—
remix, if necessary. Record the time of initial mixing (when
Precondition all materials as necessary to achieve an initial
wetting of cementitious materials first occurs), to the nearest
mixturetemperatureof23.0°C 62.0°C[73.5°F 63.5°F]or
minute.
other specific initial mixture temperature according to test
8.3.2 Because mixing intensity is a variable that may
objectives.
influence the interaction of materials used to prepare test
specimens, in many cases different mixing procedures (speeds
NOTE 3—Depending on test objectives, a test temperature representa-
or durations) may be needed, depending on the goal of the
tive of typical or extreme field conditions may be selected. For other
evaluations, a test temperature equal to the laboratory temperature is
testing. Unless mixing intensity is a defined variable in a
typically used. Regardless of test temperature, the initial mixture and
testing program, mixtures prepared using different mixing
specimen temperatures should usually be controlled to be as close to the
procedure shall not be compared.
test temperature as possible so that measured changes in specimen
temperature over time result essentially only from hydration influences,
8.4 Mortar:
and so that the initial (calcium aluminate) hydration and dormant periods
8.4.1 If mortar is to be tested, it can be prepared indepen-
arecapturedinthethermalprofile.Iftheinitialmixturetemperaturediffers
dently or obtained from fresh concrete by wet sieving in
fromthetesttemperature,itbecomesdifficulttousethethermalprofilefor
accordance with Practice C172/C172M.
a relative indication of time of setting.
8.5 Transferring Mixture to Specimen Container and Test
8.1.3 Thermal Measurement System and Ambient
Environment:
Temperature—The temperature of the thermal measurement
8.5.1 Place the appropriate mass of the batch contents into
system and the surrounding ambient environment shall be
the specimen container, using a suitable clean spatula, spoon,
within 6 2.0 °C [3.5 °F] of the test temperature before
or scoop; pouring is permitted if the batch is sufficiently fluid
beginning a test. Allow sufficient time for the temperature
(see Note 5). If necessary, consolidate the specimen by
measurement system to stabilize to the ambient temperature.
rodding, tamping, or tapping. Cover and seal the specimen
8.2 Test Specimens:
container, providing access for temperature sensors (such as
8.2.1 The number of specimens and number of test batches
thermocouples) that must be inserted into the test specimen.
depend on the purpose of the test program (see the Appendix
NOTE5—Itmaybeusefultomeasureslump,flow,minislump orother
for examples of test programs).
properties for comparing consistency. Specimen type and consistency
8.2.2 The volume and mass (see Note 4 and theAppendix)
govern which method(s) could be used.
of the test specimen depend on the thermal measurement
8.5.2 Immediately place the specimen container in the test
equipment, insulating value of the specimen container and any
environment and begin recording specimen temperature.
surrounding insulation, test temperature, the type of mixture
8.6 Thermal Measurements:
(paste, mortar, or concrete), and the test objectives. Masses of
8.6.1 Ensurethattemperaturesensorsareincontactwiththe
specimens that will be compared with each other shall not
specimen or container as required for the equipment used.
differ by more than 5% of the average.
Record, to the nearest minute, the time at the start of mixing
NOTE 4—Typical specimen mass is 300g to 1000 g [0.7 to 2.2 lb] for
(time of initial contact of water with cementitious materials)
pasteand1500gto4000g[3.3lbto8.8lb]formortarorconcrete,though
and the time at which temperature measurements are initiated
acceptable temperature measurements have been reported with mortar
or when the specimen temperature is first measured using
specimens of as little as 750 g [1.7 lb]. Corresponding container volumes
are approximately 150mL to 600 mL [10in. to 35 in. ] for paste and continuously logged data (see Note 6).
650mL to 1650 mL [40in. to 100 in. ] for mortar or concrete. The
8.6.2 The time delay between the start of mixing and initial
selection of specimen mass and the use of insulation around specimen
measurement of specimen temperature may vary according to
containers must be balanced; specimens with greater mass require less
testseriesandspecimendetailsbuttheextentofthisdelayshall
insulation. Thermal testing with concrete or mortar specimens is usually
be controlled to within 6 15 seconds for all specimens being
preferred when time of setting trends are being evaluated, but testing with
paste specimens of similar proportions may be equally useful and may be
compared.
more convenient. Thermal profiles for paste specimens with the same
NOTE 6—The time delay between the start of mixing and initial
proportions as the paste fractions of concrete mixtures being evaluated,
without the aggregates, have been shown to consistently produce indica-
tions of longer times of setting than those for concrete or mortar
Kantro, D.L. (1980). “Influence of water-reducing admixtures on properties of
specimens, but trends are similar.
cement paste—a miniature slump test,” Cement, Concrete, and Aggregates, 2, pp.
8.3 Mixing: 95-102.
C1753/C1753M−21a
measurement of specimen temperature should be as short as possible.
10.1.1 Type of equipment used including descriptions of
specimen containers, layout and spacing of individual speci-
8.6.3 For typical test durations of less than 48 hours,
men containers, type and locations of temperature sensors, and
measure the specimen temperatures at intervals of no greater
any insulation used.
than 60 seconds until at least two hours after the maximum
10.1.2 Signal-to-noise ratio as determined from test data for
temperature of the main peak response has been reached (Fig.
the specimens and test equipment and conditions, noting
1). Alternatively, greater intervals are permitted to simplify
compliance with 6.2.1.
data management for extremely gradual rates of specimen
10.1.3 Sourceandidentityofallmaterialstested,methodof
temperaturechangeand/ortestdurationsinexcessof48hours.
conditioningthemtotesttemperature,andtemperaturepriorto
9. Evaluation of Test Results
mixing.
9.1 Test results are evaluated typically by comparing differ-
10.1.3.1 If calcium sulfate was added, describe the specific
ences in thermal profiles from different test mixtures. See
type of calcium sulfate used and its source, as well as the
examples in the Appendix.
timing of addition. If calcium sulfate hemihydrate was used,
evidence shall be supplied of its hydration form before testing.
9.2 Plotspecimentemperatureasafunctionoftime,usinga
10.1.4 Mixture proportions, including the concentrations of
common time scale relative to time at the start of mixing (t =
any stock solutions used.
0 at the time of first wetting of cementitious materials, to the
10.1.5 Mixing method and duration, including sequences
nearest minute) for all mixtures to be compared. Optionally,
and timing of mixing and scraping down, volume of mixing
plot specimen temperature after subtraction of the temperature
bowl or container used for mixing, and speed of mixer.
of the inert specimen at the corresponding elapsed time for
10.1.6 Addition sequence for all materials, and method of
each data point to isolate temperature changes due to hydra-
addition of admixture(s).
tion.Itispermissibletoplotsegmentsofthethermalprofilefor
10.1.7 Method or description of any consolidation effort
specialevaluation(seeNote7).Smoothingoftemperaturedata
used.
is permissible if errant data points were logged that can be
10.1.8 Anyunusualbehavior,suchasearlystiffeningduring
reasonably attributed to spurious data or any type of malfunc-
specimen preparation.
tion of measurement equipment.
NOTE7—Itmaybeusefultoseparatelyplottemperaturedataduringthe 10.1.9 Mass of the test specimens placed in the specimen
first 30 to 60 minutes of hydration, or other time period showing rapid
containers.
temperaturechange,inordertoexpandthetimescaleforbetterdisplayof
10.1.10 Test temperatures, initial mixture temperatures at
rapid temperature changes due to calcium aluminate hydration. The
the conclusion of mixing, date, time at the start of mixing and
usefulness of such early data may depend on timing of initial temperature
elapsed time to the first recorded specimen temperature, and
measurements, insulation properties of specimen container and
environment, specimen mass, and other equipment configuration details.
duration of thermal measurements for each test mixture.
10.1.11 If thermal indication of relative time of setting is
9.3 Indications of relative time of setting for different
used, the fraction or percentage of main peak response tem-
mixtures, if called for, shall be evaluated using the same
perature rise used in evaluation.
fractionofthemainpeakresponsetemperaturerise(maximum
10.1.12 Theresultsandtestmethodusedtomeasurefluidity
temperatureminusminimumdormantperiodtemperature).See
or consistency of specimen, if applicable.
Note 8 and examples in the Appendix.
NOTE 8—A fixed fraction or percentage of the main peak response 10.1.13 Plotsofthermalprofilesforalltestmixturesandthe
temperature rise is used as a temperature indication of relative time of
temperature history of the inert specimen from the start of
setting when comparing different mixtures. For the given conditions, a
testing. See Appendix for examples.
fraction that approximates the times of initial or final setting of concrete,
10.1.14 Explanation of any periods of missing or flawed
as defined by Test Method C403/C403M, may be selected to permit
temperature data affecting individual thermal profiles, includ-
correlation with penetration resistance data. It is, however, often conve-
nient to select a percentage that can be used easily in visual evaluation of
ing any non-uniformity of the elapsed time from the start of
thermal profiles, such as 50%. In such cases, the thermal indication of
mixing to first recorded specimen temperature.
timesofsettingmaydiffersignificantlyfromtimesofsettingbasedonTest
10.1.15 Statementthatthetestwascarriedoutinaccordance
Method C403/C403M, but can still be useful in evaluation of the relative
with this practice and notes of any deviations from intended
effects of different mixture variables on setting. See the Appendix for
examplesofuseofthermalprofilesforevaluationofsettingtrendsandthe test conditions.
influences of variables related to materials, proportions, and temperature
conditions.
11. Keywords
10. Report
11.1 cement – admixture interactions; hydration; setting;
10.1 Report the following information: sulfate balance; thermal measurement testing; thermal profiles
C1753/C1753M−21a
APPENDIX
(Nonmandatory Information)
X1. TYPICAL APPLICATIONS
X1.1 Introduction dosages that are both lower and higher than the envisioned
dosage, in order to establish the mixture sensitivity to those
X1.1.1 Thermal measurement testing can be used to study
materials.Thedosagesequencingprotocolforconcretebatches
setting characteristics, relative early-age hydration efficiency,
may also be a variable of interest, as delayed addition of
and the potential for abnormal behavior in paste, mortar, or
chemical admixtures, seconds or minutes after initial introduc-
concrete mixtures. As such it can be used as part of concrete
tion of mix water and mixing effort, can be useful in avoiding
quality control, for the evaluation of candidate materials
sulfate-balance issues.
sources or materials variability, and to investigate the influ-
ences of different component materials, proportions, and con-
X1.2.4 Variation of both the initial mixture temperature and
crete temperatures. test temperature (usually simulating field temperatures of
interest) are important to include in the experiment, because
X1.1.2 Several examples of experimental evaluations are
time of setting can vary unpredictably and sulfate-balance
shown in this appendix. Each example represents a specific set
effects can change unexpectedly with temperature changes.
ofmaterials,andtheresultscannotbeextrapolatedtoothersets
While the effects of these variations can be evaluated using
of materials.
fieldtestingatactualambienttemperatures,itisoftenusefulto
simulatefieldtemperaturesinlaboratoryexperiments.Depend-
X1.2 Experiment Design and Planning
ing on the laboratory equipment and the number and distance
X1.2.1 While uses of thermal testing may include routine
between test specimens during testing, precise control of test
concrete or mortar mixtures for quality control or benchmark-
temperatures is often a challenge due to the collective contri-
ing of setting trends, many applications may be designed to
butionofhydrationheatfromthetestspecimens.Insuchcases,
answer questions about the influences of alternative materials
the number of test specimens contained in a temperature-
sources, material variability, proportions, initial concrete
controlled cabinet or vessel may need to be limited in order to
temperature, and test temperatures. Thermal measurement
meet the reference temperature requirement of 6.2.3. The
testing should generally be planned to include a number of
plotting of specimen temperature after the subtraction of
similar but distinct mixtures featuring specific variables, the
reference temperature (see 9.2) usually helps to minimize the
performance effects of which are to be compared.
unwanted influences of changes in ambient temperature during
X1.2.1.1 Measurements of temperature according to this
testing.
practice are typically subject to more variability than the
X1.2.5 Cementitious mixtures of all types, including
temperature or heat measurements of more sophisticated calo-
concrete, mortar, soil stabilization mixtures, grout, and paste,
rimetry methods. Standardization of equipment is not usually
can be used in thermal measurement testing. Depending on the
warranted, and control of the test temperature may be
configuration of the available test apparatus, size and shape of
approximate, affecting results to some extent. For these
specimen containers, and the insulation that will surround
reasons, replicate test mixtures should include several incre-
hydratingspecimens,ifany,selectionoftheappropriatetypeof
ments of the variables of interest so that performance trends
mixturemayinfluencetheapplicabilityofdataproduced.Peak
can be identified and inherent test variability evaluated and
hydrationtemperaturesduringtestingwilltypicallybereduced
considered. Comparisons of thermal profiles obtained in dif-
as the proportions of aggregate in the mixture increases.
ferent test series, at different locations, or using different
Likewise, as the volume and mass of the test specimen
equipment are not usually appropriate.
increases, the peak hydration temperature increases, other
X1.2.2 The objectives of thermal measurement experiments
factors being equal. The most useful data are generally
may include evaluation of the effects of different cements,
produced by balancing these factors so that peak temperatures
supplementary cementitious materials (SCMs), chemical
achieved during testing result in adequate signal-to-noise ratio
admixtures, dosage rates, and addition sequences. Other pa-
(see 6.2.2, Note 2, and Fig. 2) without exceeding the expected
rameters such as mix water source, presence of finely divided
peak temperatures of field concrete in place. Artificially high
particles, materials variability, mixing method, initial mixture
peak temperatures during testing will often result in unrealistic
temperature, and test temperature can be studied as well.
thermal profiles, because different chemical compounds in the
Experimentsmaybeintendedtoevaluatethesulfatebalanceof
hydrating mixture respond to temperature differently with
amixture,thatis,whetherthesolublecalciumsulfate(contrib-
respect to the rate of hydration.
uted typically by the portland or blended cement) in a mixture
X1.2.5.1 In general, concrete or other mixtures with high
isadequateforthematerials,proportions,andprojecttempera-
aggregatecontentrequirelarger(moremassive)testspecimens
tures of interest.
surrounded by insulation. The quality of data for concrete
X1.2.3 Chemical admixtures and SCMs may be selected mixtures can often be improved by testing only the mortar
and dosed based on submitted or envisioned concrete mixtures fraction, obtained in accordance with Practice C172/C172M,
or supplier recommendations. It is recommended to include especially when specimens are smaller (less massive) than
C1753/C1753M−21a
ideal. Likewise, laboratory testing of paste-only specimens is X1.2.7 A mixture plan should be prepared before each test
often done using smaller specimens, without insulation around series, for efficiency of batching and mixing procedures during
specimen containers. test execution and to serve as a record of the mixture materials
X1.2.5.2 Thereshouldbeuniformityofmixtureconsistency and proportions. An example mixture plan is shown in Table
for the test specimens in a test series. Consolidate test X1.1. Test results from this mixture series are presented and
specimens, if needed, to remove excessive entrapped air. Mix discussed in X1.5.4.1.
water should be proportioned to result in a uniform mixture
X1.3 Mixing Methods
without excessive segregation.
X1.3.1 Actual project concrete for thermal measurement
X1.2.6 Experiments intended to evaluate mixture sulfate
testing can be sampled in the field according to Practice
balance should include a range of key variables (usually
C172/C172M. Concrete mixed in the laboratory according to
including all possible combinations of admixtures, SCMs and
Practice C192/C192M can also be used. In either case, data
extremes of field temperatures) sufficient to demonstrate the
variability can often be reduced and signal-to-noise increased
relative contribution of each variable to sulfate balance issues.
by testing mortar sieved from the concrete in accordance with
This will typically require including overdoses of admixtures
Practice C172/C172M, although sieving of mortar from the
and SCMs and initial and test temperatures higher than those
concrete will increase the elapsed hydration time prior to first
anticipated in the field. Sulfate balance-related abnormal be-
measurement of temperatures.
havior may occur with only a slight incremental change in a
X1.3.2 Various laboratory mixing methods for mortar and
critical variable.As such, even normal variability of a compo-
paste have been used successfully, including methods pre-
nent material should be anticipated as a possible source of
sented in Practice C305 and other methods as indicated in
performance issues when a mixture is near its sulfate imbal-
PracticeC1679.Modificationsoftheseandothermethodshave
ance threshold.
been found advantageous, as well. Some method variations
X1.2.6.1 Theevaluationofsulfatebalanceforagivensetof
include shorter mixing times (often 60 seconds or less) that
materials and proportions can also be approached using incre-
facilitateearlierdatacollectionduringinitialhydration.Mixing
mental sulfate contents (see X1.5.6), through the addition of
devices and hardware may include kitchen mixers, paint
calcium sulfate in replicate mixtures or the use of multiple
stirrers, and simple mechanical agitation. For short duration
cementsamplesfromthesamesourcethatvaryinSO content,
mixing, cementitious materials are often introduced to the
to determine if normal main peak response (or sulfate “bal-
mixing bowl in a dry state, and mix water with dispersed
ance” as defined in Practice C1679) can be restored (see 5.2
chemical admixtures is added subsequently as mixing begins.
and 5.6). Additions of reagent grade calcium sulfate may not
Hand-held kitchen mixers can also be used in such a way that
necessarily result in the same performance as the equivalent
separate scraping of the sides of the mixing bowl is unneces-
increments of calcium sulfate introduced during the cement
sary. Specific protocols can be adjusted to include delayed
grinding process, but performance trends will be similar. Fig.
admixture additions to simulate what might occur at the
X1.1 shows an example of the influences of incremental
concrete plant, as an experiment variable.
cementsulfatecontentinpastemadewith25%ClassCflyash
and water reducing admixture. The abnormally-shaped, dual X1.3.3 Regardless of the specific mixing protocol used in a
peak thermal profiles, reduced peaks, and delayed setting test series, it is important that the method produce homoge-
evident in the mixtures with lower sulfate levels can be neous mixtures, that the method is repeatable, resulting in
confirmed as effects of sulfate imbalance, because a single, identical mixing for each mixture to be compared, and that
higher peak and normal setting was restored as sulfate was timing of each stage of mixing be controlled to reproduce the
increased. mixing process as accurately as possible for each test mixture.
FIG. X1.1The Effects of Incremental Cement SO Content on Main Peak Response for a Mixture with 25% Class C Fly Ash Replace-
ment and Water Reducing Admixture at 35 °C [95 °F] Initial Mixture and Test Temperatures, w/cm = 0.40
C1753/C1753M−21a
TABLE X1.1 Example Mixture Proportions for a Laboratory Paste Test Series
NOTE 1—1 mL/100 kg = 0.0154 oz/100 lb.
Admixture by product
Cement SCM Water
time
(rate, mL/100 kg) - dose, mL
Temp,
Channel Mixture description @data
°C
Type &
start
Type & source mass (g) mass (g) w/cm mass (g) A/B/D A/F A/F/MR
source
A1 23 No admix - 23 °C Type II - project 500 0.40 200
A2 23 Type II - project 500 0.40 200 (195) -
ABD @ 195 - 23 °C
0.98
A3 23 Type II - project 500 0.40 200 (390) -
ABD @ 390 - 23 °C
1.95
A4 23 Type II - project 500 0.40 200 (195) -
AF @ 195 - 23 °C
0.98
A5 23 Type II - project 500 0.40 200 (390) -
AF @ 390 - 23 °C
1.95
A6 23 not used
A7 23 not used
A8 23 23 °C reference - sand + water
B1 13 No admix - 13 °C Type II - project 500 0.40 200 (195) -
0.98
B2 13 ABD @ 195 - 13 °C Type II - project 500 0.40 200 (195) -
0.98
B3 13 AF @ 195 - 13 °C Type II - project 500 0.40 200 (390) -
1.95
B4 13 AF @ 390 - 13 °C Type II - project 500 0.40 200 (195) -
0.98
B5 13 AMR @ 195 - 13 °C Type II - project 500 0.40 200 (390) -
1.95
B6 13 AMR @ 390 - 13 °C Type II - project 500 0.40 200
B7 13 not used
B8 13 13 °C reference - sand + water
X1.3.4 Itispossiblethatmixingintensitymayaffectresults, and 50% fraction time of setting markers, which occur at
especially when the shear applied by mixing differs signifi- approximately 4.9 and 6.4 hours of hydration time, respec-
cantly from that in concrete mixing. Thus test comparisons tively.Aminimum temperature during the dormant period (see
using different mixing methods for the same mixture propor- Fig. X1.2) may not be clearly evident in some thermal profiles
tions may be warranted to verify the suitability of the mixing or may be missing from data (often the case for tests of actual
protocol selected. project concrete), and in such cases the reliability of relative
times of setting based on fraction times determined from the
X1.4 Evaluation of Results
thermal profiles is questionable.
X1.4.2.1 The hydration times at which a given fraction of
X1.4.1 Results of thermal measurement testing can be
the main peak response temperature rise occur can be com-
evaluated effectively only if comparative graphs include a
puted from a data record using a spreadsheet or can be
control mixture and other mixtures of significance. Evaluation
estimated from the graphs (50% fraction times are convenient
of results from a single test series may require multiple
for scaling). The hydration times corresponding to a selected
comparison graphs, each featuring a certain category of mix-
fraction may differ somewhat from actual times of initial or
ture variation (such as admixture dosag
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C1753/C1753M − 21 C1753/C1753M − 21a
Standard Practice for
Evaluating Early Hydration of Hydraulic Cementitious
Mixtures Using Thermal Measurements
This standard is issued under the fixed designation C1753/C1753M; the number immediately following the designation indicates the
year of original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last
reapproval. A superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This practice describes the apparatus and procedure for evaluating relative differences in early hydration of hydraulic
cementitious mixtures such as paste, mortar, or concrete, including those containing chemical admixtures, various supplementary
cementitious materials (SCMs), and other finely divided materials, by measuring the temperature history of a specimen.
1.2 Calorimetry is the measurement of heat lost or gained during a chemical reaction such as cement hydration; calorimetric
measurements as a function of time can be used to describe and evaluate hydration and related early-age property development.
Calorimetry may be performed under isothermal conditions (as described in Practice C1679) or under adiabatic or semi-adiabatic
conditions. This practice cannot be described as calorimetry because no attempt is made to measure or compute the heat evolved
from test specimens due to hydration, but it can in many cases be used for similar evaluations. Variables that should be considered
in the application of this practice are discussed in the Appendix.
1.3 Units—The values stated in either SI units or inch-pound units shall be regarded separately as standard. The values stated in
each system may not be exact equivalents; therefore, each system must be used independently of the other. Combining values from
the two systems may result in non-conformance with the standard. Some values have only SI units because the inch-pound
equivalents are not used in practice.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
NOTE 1—Warning: Fresh hydraulic cementitious mixtures are caustic and may cause chemical burns to skin and tissue upon prolonged exposure.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C39/C39M Test Method for Compressive Strength of Cylindrical Concrete Specimens
This practice is under the jurisdiction of ASTM Committee C09 on Concrete and Concrete Aggregates and is the direct responsibility of Subcommittee C09.48 on
Performance of Cementitious Materials and Admixture Combinations.
Current edition approved Sept. 1, 2021Dec. 1, 2021. Published October 2021January 2022. Originally approved in 2015. Last previous edition approved in 20152021 as
ɛ1
C1753/C1753M–15–21. . DOI: 10.1520/C1753_C1753M-21.10.1520/C1753_C1753M-21A.
Section on Safety Precautions, Manual of Aggregate and Concrete Testing, Annual Book of ASTM Standards, Vol. 04.02.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1753/C1753M − 21a
C125 Terminology Relating to Concrete and Concrete Aggregates
C172/C172M Practice for Sampling Freshly Mixed Concrete
C192/C192M Practice for Making and Curing Concrete Test Specimens in the Laboratory
C219 Terminology Relating to Hydraulic and Other Inorganic Cements
C305 Practice for Mechanical Mixing of Hydraulic Cement Pastes and Mortars of Plastic Consistency
C403/C403M Test Method for Time of Setting of Concrete Mixtures by Penetration Resistance
C494/C494M Specification for Chemical Admixtures for Concrete
C1005 Specification for Reference Masses and Devices for Determining Mass and Volume for Use in Physical Testing of
Hydraulic Cements
C1679 Practice for Measuring Hydration Kinetics of Hydraulic Cementitious Mixtures Using Isothermal Calorimetry
3. Terminology
3.1 Definitions—For definitions of terms used in this practice, refer to Terminology C125, Terminology C219, and Practice C1679.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 adiabatic, adj—occurring without exchange of heat with the environment.
3.2.2 exotherm, n—heat evolution during hydration as evidenced by an increase in measured specimen temperature shown in the
thermal profile.
3.2.3 inert specimen, n—specimen placed within the same thermal environment as the test specimen(s), made of a nonreactive
material of similar heat capacity and the same mass as the reacting test specimen(s).
3.2.3.1 Discussion—
The difference between the temperature of the hydrating test specimen(s) and the inert specimen represents the change in specimen
temperature due to hydration. Interpretation can often be improved by comparing temperature histories after subtracting the
temperature of the corresponding inert specimen (reference temperature), which tends to account for the effects of changing
environment temperature during the measurement period.
3.2.4 main peak response, n—the initial temperature rise and subsequent temperature drop in the measured thermal profile that
starts at the end of the dormant period and, for a mixture with normal sulfate balance, lasts for several hours.
3.2.5 reference temperature, n—the temperature of the inert specimen in a test series at the time corresponding to a particular
temperature of the test specimen.
3.2.6 sulfate demand, n—the level of soluble calcium sulfate in a hydrating cementitious mixture required to maintain normal
hydration behavior for a specific combination of mixture proportions, materials properties, initial mixture temperature, and test
temperature.
3.2.7 sulfate imbalance threshold, n—the condition of a cementitious mixture in terms of mixture proportions, materials
properties, initial mixture temperature, and test temperature, for which a small change in any of these variables can result in
abnormal hydration behavior due to depletion of calcium sulfate in solution.
3.2.8 test specimen, n—a hydraulic cementitious mixture being evaluated for its thermal response.
3.2.9 test temperature, n—the temperature of the air or insulation, if any, surrounding the test specimen containers at the start of
temperature measurement, normally intended to remain constant.
3.2.10 thermal profile, n—the temperature of a hydrating mixture (before or after subtraction of the reference temperature), plotted
as a function of hydration time, that provides an indication of the rate of hydration over time.
3.2.10.1 Discussion—
An example thermal profile is shown in Fig. 1. On the vertical axis T refers to the temperature of the test specimen and T refers
test ref
to the temperature of the inert (reference) specimen. The shape of the thermal profile is affected not only by mixture hydration but
also by the specimen type and mass, mixture proportions, specimen initial temperature, specimen container size and shape,
insulation (if any) provided around the specimen container, and the temperature of the surrounding environment. Additional
guidance is provided in the Appendix.
C1753/C1753M − 21a
NOTE 1—(A) initial exotherm from dissolution of cement and initial hydration, principally of calcium aluminates; (B) dormant period temperature
reduction associated with very low heat evolution indicating slow and well-controlled hydration; (C) main peak response associated primarily with
hydration reactions contributing to setting and early strength development, with maximum temperature at (D). The maximum temperature (D) and the
rates of temperature rise and fall that shape the main peak response (C) are affected not only by hydration but by the related cooling response of the
specimen.
FIG. 1 Example Thermal Profile of a Portland Cement Paste Mixture (Inert Specimen Temperature Subtracted from Test Specimen Tem-
perature)
3.2.11 time of setting marker, n—the point marked on the thermal profile indicating the hydration time when a selected fraction
of the main peak amplitude is attained and that is used as a relative indicator of time of setting.
4. Summary of Practice
4.1 A thermal measurement test system consists of temperature measuring devices, data collection equipment, and specimen
containers of similar volume, shape, and material, capable of similarly isolating each test specimen and an inert specimen. The
specific insulation values for specimen containers and the test temperature are selected based on the intended test objectives.
Related guidance is provided in the Appendix.
4.2 Mixtures composed of cementitious materials, water, and optionally chemical admixtures, or aggregate, or both, are prepared
and introduced into specimen containers for collection of temperature data.
4.3 Thermal profiles are plotted using a common time scale that begins at the time of initial mixing of water with cementitious
materials, which is the start of hydration time. The measured thermal profiles allow qualitative comparison of early hydration
kinetics, such as retarding or accelerating trends, as influenced by different combinations of materials, and abnormal hydration
behaviors that can interfere with setting and strength development.
5. Significance and Use
5.1 This practice provides a means of assessing the relative early hydration performance of various test mixtures compared with
control mixtures that are prepared in a similar manner.
5.2 Thermal profiles are used to evaluate the hydration behavior of hydraulic cementitious mixtures after the addition of water.
They may provide indications concerning setting characteristics, compatibility of different materials, sulfate balance, relative heat
of hydration, and early strength development. They can be used to evaluate the effects of compositions, proportions, and time of
addition of materials as well as the initial mixture and test temperatures. Thermal profile testing is an effective tool for identifying
performance sensitivities or trends, and may help to reduce the number of concrete test mixtures required to develop and qualify
mixtures, especially those to be subject to variable ambient environments. It may be used by concrete producers, materials
suppliers, and other practitioners to support mixture development, selection of material types or sources, optimization of
proportions, or troubleshooting of field problems.
C1753/C1753M − 21a
5.3 This practice can be used to understand concrete problems related to slump loss, setting, and early strength, but results may
not predict field concrete performance. Performance verification with concrete is needed to quantify the trends identified using
thermal testing.
5.4 This practice can be used to evaluate the effects of chemical admixtures on the thermal profiles of cementitious mixtures. This
can be especially useful in selecting dosages appropriate for different ambient conditions.
5.5 Thermal measurement testing as described in this practice may have similar significance and use as isothermal calorimetry
described by Practice C1679 or some types of near-adiabatic calorimetry. The selection of which practice or methods to use may
depend on specific applications and circumstances. The thermal profiles obtained by this practice may have similar shapes to
isothermal hydration profiles as obtained by Practice C1679, but thermal profiles from this practice do not provide quantitative
measurement of heat of hydration, are affected by various details of the test conditions and mixtures (see 3.2.10 and the Appendix),
and are subject to greater variability. Equipment used for this practice is less expensive than isothermal or near-adiabatic
calorimeters and may be more easily adapted for use in the field or where a large number of different specimens and mixtures must
be evaluated in a short time period. Identification of the sulfate depletion point of a mixture (as described in Practice C1679) is
not generally possible using thermal measurement testing.
5.6 To evaluate the potential for abnormal hydration, it is important that the test temperatures and the initial temperatures of the
mixture be selected to represent the range of expected initial concrete field temperatures.
5.7 This practice is not intended to provide results that can be compared across laboratories using different equipment nor to
provide quantitative measurements or corrected approximations of actual hydration heat. It should not be cited in project
specifications or otherwise used for the purpose of acceptance or rejection of concrete. It is intended to serve as a simple and
expedient tool for comparison of the relative early-age hydration performance of different specific combinations of materials that
are prepared and stored under the same conditions.
6. Apparatus
6.1 Devices for Preparing Specimens:
6.1.1 Weights and Weighing Devices, used for preparation of laboratory test mixtures up to 5 kg [11 lb] total mass shall conform
to the requirements of Specification C1005. For preparing test mixtures of greater total mass including concrete batches in the
laboratory, weighing devices shall conform to the requirements of Practice C192/C192M.
6.1.2 Graduated Cylinders, shall conform to the requirements of Specification C1005. The permissible variation for graduated
cylinders of less than 100-mL capacity shall be 6 1.0% of the indicated capacity.
6.1.3 Graduated Syringes, if used, shall be of suitable capacities to contain the desired volume of liquid admixture and shall be
accurate to 6 3% of the required volume.
6.1.4 Mixing Apparatus, capable of producing a uniform mixture.
6.2 Thermal Measurement Test Equipment and Data Acquisition System—Actual design of the equipment, whether commercial
or custom-built, may vary, but it shall meet the following requirements for the selected type, shape, and mass of the specimen,
insulation (if any) surrounding the specimen container, initial mixture temperature, and test temperature.
6.2.1 Temperature sensors shall be thermistors or thermocouples with measurement accuracy of 6 1.0 °C [2 °F].
6.2.2 The signal-to-noise ratio shall be at least 5.0. Signal is defined as the difference between the highest and the lowest
temperatures measured from the dormant period through the main peak response (Fig. 1) for a test specimen in the test series
without admixture or SCMs (Fig. 2). Noise is defined as the difference between the highest and the lowest temperatures measured
during the time period in which the signal is established (Fig. 2) for an inert specimen having a mass similar to that of the test
specimens. The inert specimen shall remain in the same environment as the test specimens to indicate both the effects of changes
in ambient temperature as well as any thermal influences of adjacent test specimens (see also 6.2.5).
C1753/C1753M − 21a
FIG. 2 Examples of Signal and Noise Determination for Verification of Signal-to-Noise Ratio
NOTE 2—Exceeding the minimum signal-to-noise ratio is more important than specific requirements for insulation value of the specimen container or
environment (see Appendix for guidance). Selected specimen containers and insulation configurations (if any) may vary with mixture type, specimen
mass, and initial mixture and test temperatures. A satisfactory inert specimen may be obtained using quantities of sand and water having masses within
6 10 % of the combined solids and water contents of the test specimens. Thermal influences from other test specimens may be reduced by providing
adequate spaces between specimens in the test environment, depending on the insulating values of the specimen containers. The intent of a minimum
signal-to-noise ratio requirement is to assure a well-defined thermal profile that is minimally influenced by ambient temperature changes and the presence
of other test specimens. The maximum main peak temperature should be similar to the maximum temperature that would be expected for in-place concrete
in the application of interest. Because the type, shape, and mass of the test specimen, insulation around the specimen container, and initial mixture and
test temperatures all influence main peak response levels, it is important to balance these factors to meet the requirements of 8.2.2 without causing
unrealistic main peak response temperatures (see Note 4 and the Appendix for guidance).
6.2.3 The air space or insulation, or both, surrounding the specimen containers, whether the test specimen is stored under ambient
conditions or inside a conditioned chamber intended to replicate field conditions of interest, shall be controlled to ensure that the
measured temperature of the inert specimen (reference temperature) does not vary from the test temperature by more than 3 °C
[5 °F] during testing, unless deliberate change of ambient conditions during the period of temperature measurement is part of the
test program.
6.2.3.1 A conditioned chamber may be a laboratory test chamber, portable curing chamber, or other temperature-controlled
container capable of maintaining the test temperature in the ambient space around the hydrating test specimens without the use
of forced air circulation. Portable concrete cylinder curing boxes that employ circulating temperature-conditioned water below a
specimen shelf have been successfully used. Other types of chambers that use forced air in the regulation of internal temperature
should not be used, however, as air movement around the specimen containers can adversely influence test results.
6.2.4 The data acquisition equipment shall be capable of performing continuous logging of the temperatures with a time interval
between recorded measurements not greater than 60 s.
6.2.5 Specimen Containers of volume and insulating value as needed to meet the requirements of 6.2.1 for the test mixtures and
conditions that can be sealed while providing access for the temperature sensors of the thermal measurement system, if required
(see Note 4). For systems without continuous insulation between specimen containers, provide a clear distance of at least 70 mm
[3 in.] between individual specimen containers.
6.2.6 The location of temperature sensors relative to specimen containers shall be similar for all test specimens and for the inert
specimen.
7. Materials
7.1 Mixture Materials:
C1753/C1753M − 21a
7.1.1 Mixture materials, including cementitious materials and admixtures, shall be obtained from the concrete producer, or
otherwise obtained to be representative of those specific to the purpose of the test.
7.2 Calcium Sulfate:
7.2.1 Use reagent grade calcium sulfate dihydrate or hemihydrate prepared from reagent grade calcium sulfate dihydrate or
calcium sulfate anhydrite to verify whether a mixture is in sulfate balance. See the Appendix for examples of sulfate addition for
evaluation of sulfate balance.
7.2.2 It is permissible to use a source-specific calcium sulfate for performing a test series that is related to a specific cement
production source.
8. Procedure
8.1 Temperature Conditions:
8.1.1 Specimen Preparation Temperature—Maintain the temperature of the air in the vicinity of all equipment and materials used
in specimen preparation at the test temperature to within 6 3.0 °C [5 °F].
8.1.2 Materials and Initial Mixture Temperatures—Precondition all materials as necessary to achieve an initial mixture
temperature of 23.0 °C 6 2.0 °C [73.5 °F 6 3.5 °F] or other specific initial mixture temperature according to test objectives.
NOTE 3—Depending on test objectives, a test temperature representative of typical or extreme field conditions may be selected. For other evaluations,
a test temperature equal to the laboratory temperature is typically used. Regardless of test temperature, the initial mixture and specimen temperatures
should usually be controlled to be as close to the test temperature as possible so that measured changes in specimen temperature over time result
essentially only from hydration influences, and so that the initial (calcium aluminate) hydration and dormant periods are captured in the thermal profile.
If the initial mixture temperature differs from the test temperature, it becomes difficult to use the thermal profile for a relative indication of time of setting.
8.1.3 Thermal Measurement System and Ambient Temperature—The temperature of the thermal measurement system and the
surrounding ambient environment shall be within 6 2.0 °C [3.5 °F] of the test temperature before beginning a test. Allow sufficient
time for the temperature measurement system to stabilize to the ambient temperature.
8.2 Test Specimens:
8.2.1 The number of specimens and number of test batches depend on the purpose of the test program (see the Appendix for
examples of test programs).
8.2.2 The volume and mass (see Note 4 and the Appendix) of the test specimen depend on the thermal measurement equipment,
insulating value of the specimen container and any surrounding insulation, test temperature, the type of mixture (paste, mortar, or
concrete), and the test objectives. Masses of specimens that will be compared with each other shall not differ by more than 5%
of the average.
NOTE 4—Typical specimen mass is 300 g to 1000 g [0.7 to 2.2 lb] for paste and 1500 g to 4000 g [3.3 lb to 8.8 lb] for mortar or concrete, though acceptable
temperature measurements have been reported with mortar specimens of as little as 750 g [1.7 lb]. Corresponding container volumes are approximately
3 3
150 mL to 600 mL [10 in. to 35 in. ] for paste and 650 mL to 1650 mL [40 in. to 100 in. ] for mortar or concrete. The selection of specimen mass and
the use of insulation around specimen containers must be balanced; specimens with greater mass require less insulation. Thermal testing with concrete
or mortar specimens is usually preferred when time of setting trends are being evaluated, but testing with paste specimens of similar proportions may
be equally useful and may be more convenient. Thermal profiles for paste specimens with the same proportions as the paste fractions of concrete mixtures
being evaluated, without the aggregates, have been shown to consistently produce indications of longer times of setting than those for concrete or mortar
specimens, but trends are similar.
8.3 Mixing:
8.3.1 Any effective mixing procedure is allowed; various suitable mixing methods are described in the Appendix. Depending on
the method used, the order of the introduction of materials to the mixing bowl or container may differ. Dispense liquid admixtures
into mixing water to form a solution before introduction into the cementitious materials. The solution containing admixtures may
consist of all of the mix water or some portion, if admixture addition is to be delayed. Liquid admixtures may be introduced directly
C1753/C1753M − 21a
to mixing water using a graduated syringe or obtained from a stock solution at appropriate dilution. Inspect stock solutions for
separation and remix, if necessary. Record the time of initial mixing (when wetting of cementitious materials first occurs), to the
nearest minute.
8.3.2 Because mixing intensity is a variable that may influence the interaction of materials used to prepare test specimens, in many
cases different mixing procedures (speeds or durations) may be needed, depending on the goal of the testing. Unless mixing
intensity is a defined variable in a testing program, mixtures prepared using different mixing procedure shall not be compared.
8.4 Mortar:
8.4.1 If mortar is to be tested, it can be prepared independently or obtained from fresh concrete by wet sieving in accordance with
Practice C172/C172M.
8.5 Transferring Mixture to Specimen Container and Test Environment:
8.5.1 Place the appropriate mass of the batch contents into the specimen container, using a suitable clean spatula, spoon, or scoop;
pouring is permitted if the batch is sufficiently fluid (see Note 5). If necessary, consolidate the specimen by rodding, tamping, or
tapping. Cover and seal the specimen container, providing access for temperature sensors (such as thermocouples) that must be
inserted into the test specimen.
NOTE 5—It may be useful to measure slump, flow, mini slump or other properties for comparing consistency. Specimen type and consistency govern
which method(s) could be used.
8.5.2 Immediately place the specimen container in the test environment and begin recording specimen temperature.
8.6 Thermal Measurements:
8.6.1 Ensure that temperature sensors are in contact with the specimen or container as required for the equipment used. Record,
to the nearest minute, the time at the start of mixing (time of initial contact of water with cementitious materials) and the time at
which temperature measurements are initiated or when the specimen temperature is first measured using continuously logged data
(see Note 6).
8.6.2 The time delay between the start of mixing and initial measurement of specimen temperature may vary according to test
series and specimen details but the extent of this delay shall be controlled to within 6 15 seconds for all specimens being
compared.
NOTE 6—The time delay between the start of mixing and initial measurement of specimen temperature should be as short as possible.
8.6.3 For typical test durations of less than 48 hours, measure the specimen temperatures at intervals of no greater than 60 seconds
until at least two hours after the maximum temperature of the main peak response has been reached (Fig. 1). Alternatively, greater
intervals are permitted to simplify data management for extremely gradual rates of specimen temperature change and/or test
durations in excess of 48 hours.
9. Evaluation of Test Results
9.1 Test results are evaluated typically by comparing differences in thermal profiles from different test mixtures. See examples in
the Appendix.
9.2 Plot specimen temperature as a function of time, using a common time scale relative to time at the start of mixing (t = 0 at
the time of first wetting of cementitious materials, to the nearest minute) for all mixtures to be compared. Optionally, plot specimen
temperature after subtraction of the temperature of the inert specimen at the corresponding elapsed time for each data point to
isolate temperature changes due to hydration. It is permissible to plot segments of the thermal profile for special evaluation (see
Note 7). Smoothing of temperature data is permissible if errant data points were logged that can be reasonably attributed to
spurious data or any type of malfunction of measurement equipment.
Kantro, D.L. (1980). “Influence of water-reducing admixtures on properties of cement paste—a miniature slump test,” Cement, Concrete, and Aggregates, 2, pp. 95-102.
C1753/C1753M − 21a
NOTE 7—It may be useful to separately plot temperature data during the first 30 to 60 minutes of hydration, or other time period showing rapid temperature
change, in order to expand the time scale for better display of rapid temperature changes due to calcium aluminate hydration. The usefulness of such early
data may depend on timing of initial temperature measurements, insulation properties of specimen container and environment, specimen mass, and other
equipment configuration details.
9.3 Indications of relative time of setting for different mixtures, if called for, shall be evaluated using the same fraction of the main
peak response temperature rise (maximum temperature minus minimum dormant period temperature). See Note 8 and examples
in the Appendix.
NOTE 8—A fixed fraction or percentage of the main peak response temperature rise is used as a temperature indication of relative time of setting when
comparing different mixtures. For the given conditions, a fraction that approximates the times of initial or final setting of concrete, as defined by Test
Method C403/C403M, may be selected to permit correlation with penetration resistance data. It is, however, often convenient to select a percentage that
can be used easily in visual evaluation of thermal profiles, such as 50 %. In such cases, the thermal indication of times of setting may differ significantly
from times of setting based on Test Method C403/C403M, but can still be useful in evaluation of the relative effects of different mixture variables on
setting. See the Appendix for examples of use of thermal profiles for evaluation of setting trends and the influences of variables related to materials,
proportions, and temperature conditions.
10. Report
10.1 Report the following information:
10.1.1 Type of equipment used including descriptions of specimen containers, layout and spacing of individual specimen
containers, type and locations of temperature sensors, and any insulation used.
10.1.2 Signal-to-noise ratio as determined from test data for the specimens and test equipment and conditions, noting compliance
with 6.2.1.
10.1.3 Source and identity of all materials tested, method of conditioning them to test temperature, and temperature prior to
mixing.
10.1.3.1 If calcium sulfate was added, describe the specific type of calcium sulfate used and its source, as well as the timing of
addition. If calcium sulfate hemihydrate was used, evidence shall be supplied of its hydration form before testing.
10.1.4 Mixture proportions, including the concentrations of any stock solutions used.
10.1.5 Mixing method and duration, including sequences and timing of mixing and scraping down, volume of mixing bowl or
container used for mixing, and speed of mixer.
10.1.6 Addition sequence for all materials, and method of addition of admixture(s).
10.1.7 Method or description of any consolidation effort used.
10.1.8 Any unusual behavior, such as early stiffening during specimen preparation.
10.1.9 Mass of the test specimens placed in the specimen containers.
10.1.10 Test temperatures, initial mixture temperatures at the conclusion of mixing, date, time at the start of mixing and elapsed
time to the first recorded specimen temperature, and duration of thermal measurements for each test mixture.
10.1.11 If thermal indication of relative time of setting is used, the fraction or percentage of main peak response temperature rise
used in evaluation.
10.1.12 The results and test method used to measure fluidity or consistency of specimen, if applicable.
10.1.13 Plots of thermal profiles for all test mixtures and the temperature history of the inert specimen from the start of testing.
See Appendix for examples.
10.1.14 Explanation of any periods of missing or flawed temperature data affecting individual thermal profiles, including any
non-uniformity of the elapsed time from the start of mixing to first recorded specimen temperature.
C1753/C1753M − 21a
10.1.15 Statement that the test was carried out in accordance with this practice and notes of any deviations from intended test
conditions.
11. Keywords
11.1 cement – admixture interactions; hydration; setting; sulfate balance; thermal measurement testing; thermal profiles
APPENDIX
(Nonmandatory Information)
X1. TYPICAL APPLICATIONS
X1.1 Introduction
X1.1.1 Thermal measurement testing can be used to study setting characteristics, relative early-age hydration efficiency, and the
potential for abnormal behavior in paste, mortar, or concrete mixtures. As such it can be used as part of concrete quality control,
for the evaluation of candidate materials sources or materials variability, and to investigate the influences of different component
materials, proportions, and concrete temperatures.
X1.1.2 Several examples of experimental evaluations are shown in this appendix. Each example represents a specific set of
materials, and the results cannot be extrapolated to other sets of materials.
X1.2 Experiment Design and Planning
X1.2.1 While uses of thermal testing may include routine concrete or mortar mixtures for quality control or benchmarking of
setting trends, many applications may be designed to answer questions about the influences of alternative materials sources,
material variability, proportions, initial concrete temperature, and test temperatures. Thermal measurement testing should generally
be planned to include a number of similar but distinct mixtures featuring specific variables, the performance effects of which are
to be compared.
X1.2.1.1 Measurements of temperature according to this practice are typically subject to more variability than the temperature or
heat measurements of more sophisticated calorimetry methods. Standardization of equipment is not usually warranted, and control
of the test temperature may be approximate, affecting results to some extent. For these reasons, replicate test mixtures should
include several increments of the variables of interest so that performance trends can be identified and inherent test variability
evaluated and considered. Comparisons of thermal profiles obtained in different test series, at different locations, or using different
equipment are not usually appropriate.
X1.2.2 The objectives of thermal measurement experiments may include evaluation of the effects of different cements,
supplementary cementitious materials (SCMs), chemical admixtures, dosage rates, and addition sequences. Other parameters such
as mix water source, presence of finely divided particles, materials variability, mixing method, initial mixture temperature, and test
temperature can be studied as well. Experiments may be intended to evaluate the sulfate balance of a mixture, that is, whether the
soluble calcium sulfate (contributed typically by the portland or blended cement) in a mixture is adequate for the materials,
proportions, and project temperatures of interest.
X1.2.3 Chemical admixtures and SCMs may be selected and dosed based on submitted or envisioned concrete mixtures or supplier
C1753/C1753M − 21a
recommendations. It is recommended to include dosages that are both lower and higher than the envisioned dosage, in order to
establish the mixture sensitivity to those materials. The dosage sequencing protocol for concrete batches may also be a variable
of interest, as delayed addition of chemical admixtures, seconds or minutes after initial introduction of mix water and mixing effort,
can be useful in avoiding sulfate-balance issues.
X1.2.4 Variation of both the initial mixture temperature and test temperature (usually simulating field temperatures of interest) are
important to include in the experiment, because time of setting can vary unpredictably and sulfate-balance effects can change
unexpectedly with temperature changes. While the effects of these variations can be evaluated using field testing at actual ambient
temperatures, it is often useful to simulate field temperatures in laboratory experiments. Depending on the laboratory equipment
and the number and distance between test specimens during testing, precise control of test temperatures is often a challenge due
to the collective contribution of hydration heat from the test specimens. In such cases, the number of test specimens contained in
a temperature-controlled cabinet or vessel may need to be limited in order to meet the reference temperature requirement of 6.2.3.
The plotting of specimen temperature after the subtraction of reference temperature (see 9.2) usually helps to minimize the
unwanted influences of changes in ambient temperature during testing.
X1.2.5 Cementitious mixtures of all types, including concrete, mortar, soil stabilization mixtures, grout, and paste, can be used
in thermal measurement testing. Depending on the configuration of the available test apparatus, size and shape of specimen
containers, and the insulation that will surround hydrating specimens, if any, selection of the appropriate type of mixture may
influence the applicability of data produced. Peak hydration temperatures during testing will typically be reduced as the proportions
of aggregate in the mixture increases. Likewise, as the volume and mass of the test specimen increases, the peak hydration
temperature increases, other factors being equal. The most useful data are generally produced by balancing these factors so that
peak temperatures achieved during testing result in adequate signal-to-noise ratio (see 6.2.2, Note 2, and Fig. 2) without exceeding
the expected peak temperatures of field concrete in place. Artificially high peak temperatures during testing will often result in
unrealistic thermal profiles, because different chemical compounds in the hydrating mixture respond to temperature differently with
respect to the rate of hydration.
X1.2.5.1 In general, concrete or other mixtures with high aggregate content require larger (more massive) test specimens
surrounded by insulation. The quality of data for concrete mixtures can often be improved by testing only the mortar fraction,
obtained in accordance with Practice C172/C172M, especially when specimens are smaller (less massive) than ideal. Likewise,
laboratory testing of paste-only specimens is often done using smaller specimens, without insulation around specimen containers.
X1.2.5.2 There should be uniformity of mixture consistency for the test specimens in a test series. Consolidate test specimens, if
needed, to remove excessive entrapped air. Mix water should be proportioned to result in a uniform mixture without excessive
segregation.
X1.2.6 Experiments intended to evaluate mixture sulfate balance should include a range of key variables (usually including all
possible combinations of admixtures, SCMs and extremes of field temperatures) sufficient to demonstrate the relative contribution
of each variable to sulfate balance issues. This will typically require including overdoses of admixtures and SCMs and initial and
test temperatures higher than those anticipated in the field. Sulfate balance-related abnormal behavior may occur with only a slight
incremental change in a critical variable. As such, even normal variability of a component material should be anticipated as a
possible source of performance issues when a mixture is near its sulfate imbalance threshold.
X1.2.6.1 The evaluation of sulfate balance for a given set of materials and proportions can also be approached using incremental
sulfate contents (see X1.5.6), through the addition of calcium sulfate in replicate mixtures or the use of multiple cement samples
from the same source that vary in SO content, to determine if normal main peak response (or sulfate “balance” as defined in
Practice C1679) can be restored (see 5.2 and 5.6). Additions of reagent grade calcium sulfate may not necessarily result in the same
performance as the equivalent increments of calcium sulfate introduced during the cement grinding process, but performance
trends will be similar. Fig. X1.1 shows an example of the influences of incremental cement sulfate content in paste made with 25 %
Class C fly ash and water reducing admixture. The abnormally-shaped, dual peak thermal profiles, reduced peaks, and delayed
setting evident in the mixtures with lower sulfate levels can be confirmed as effects of sulfate imbalance, because a single, higher
peak and normal setting was restored as sulfate was increased.
C1753/C1753M − 21a
FIG. X1.1 The Effects of Incremental Cement SO Content on Main Peak Response for a Mixture with 25 % Class C Fly Ash Replace-
ment and Water Reducing Admixture at 35 °C [95 °F] Initial Mixture and Test Temperatures, w/cm = 0.40
X1.2.7 A mixture plan should be prepared before each test series, for efficiency of batching and mixing procedures during test
execution and to serve as a record of the mixture materials and proportions. An example mixture plan is shown in Table X1.1. Test
results from this mixture series are presented and discussed in X1.5.4.1.
X1.3 Mixing Methods
X1.3.1 Actual project concrete for thermal measurement testing can be sampled in the field according to Practice C172/C172M.
Concrete mixed in the laboratory according to Practice C192/C192M can also be used. In either case, data variability can often
TABLE X1.1 Example Mixture Proportions for a Laboratory Paste Test Series
NOTE 1—1 mL/100 kg = 0.0154 oz/100 lb.
Admixture by product
Cement SCM Water
time
(rate, mL/100 kg) - dose, mL
Temp,
Channel Mixture description @data
°C
Type &
start
Type & source mass (g) mass (g) w/cm mass (g) A/B/D A/F A/F/MR
source
A1 23 No admix - 23 °C Type II - project 500 0.40 200
A2 23 Type II - project 500 0.40 200 (195) -
ABD @ 195 - 23 °C
0.98
A3 23 Type II - project 500 0.40 200 (390) -
ABD @ 390 - 23 °C
1.95
A4 23 Type II - project 500 0.40 200 (195) -
AF @ 195 - 23 °C
0.98
A5 23 Type II - project 500 0.40 200 (390) -
AF @ 390 - 23 °C
1.95
A6 23 not used
A7 23 not used
A8 23 23 °C reference - sand + water
B1 13 No admix - 13 °C Type II - project 500 0.40 200 (195) -
0.98
B2 13 ABD @ 195 - 13 °C Type II - project 500 0.40 200 (195) -
0.98
B3 13 AF @ 195 - 13 °C Type II - project 500 0.40 200 (390) -
1.95
B4 13 AF @ 390 - 13 °C Type II - project 500 0.40 200 (195) -
0.98
B5 13 AMR @ 195 - 13 °C Type II - project 500 0.40 200 (390) -
1.95
B6 13 AMR @ 390 - 13 °C Type II - project 500 0.40 200
B7 13 not used
B8 13 13 °C reference - sand + water
C1753/C1753M − 21a
be reduced and signal-to-noise increased by testing mortar sieved from the concrete in accordance with Practice C172/C172M,
although sieving of mortar from the concrete will increase the elapsed hydration time prior to first measurement of temperatures.
X1.3.2 Various laboratory mixing methods for mortar and paste have been used successfully, including methods presented in
Practice C305 and other methods as indicated in Practice C1679. Modifications of these and other methods have been found
advantageous, as well. Some method variations include shorter mixing times (often 60 seconds or less) that facilitate earlier data
collection during initial hydration. Mixing devices and hardware may include kitchen mixers, paint stirrers, and simple mechanical
agitation. For short duration mixing, cementitious materials are often introduced to the mixing bowl in a dry state, and mix water
with dispersed chemical admixtures is added subsequently as mixing begins. Hand-held kitchen mixers can also be used in such
a way that separate scraping of the sides of the mixing bowl is unnecessary. Specific protocols can be adjusted to include delayed
admixture additions to simulate what might occur at the concrete plant, as an experiment variable.
X1.3.3 Regardless of the specific mixing protocol used in a test series, it is important that the method produce homogeneous
mixtures, that the method is repeatable, resulting in identical mixing for each mixture to be compared, and that timing of each stage
of mixing be controlled to reproduce the mixing process as accurately as possible for each test mixture.
X1.3.4 It is possible that mixing intensity may affect results, especially when the shear applied by mixing differs significantly from
that in concrete mixing. Thus test comparisons using different mixing methods for the same mixture proportions may be warranted
to verify the suitability of the mixing protocol selected.
X1.4 Evaluation of Results
X1.4.1 Results of thermal measurement testing can be evaluated effectively only if comparative graphs include a control mixture
and other mixtures of significance. Evaluation of results from
...

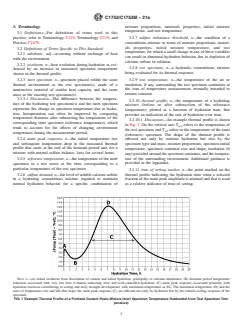

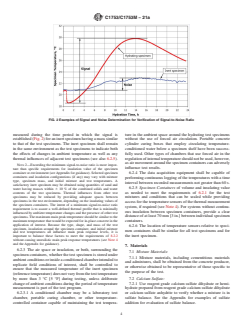


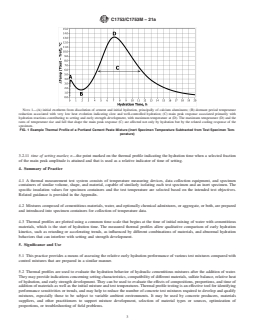

Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...