ASTM F3395/F3395M-19
(Specification)Standard Specification for Neurosurgical Head Holder Devices
Standard Specification for Neurosurgical Head Holder Devices
SCOPE
1.1 This specification covers standards a manufacturer shall meet in the performance testing of neurosurgical head holder devices (skull clamps).
1.2 This specification covers neurosurgical head holder devices (skull clamps) made of metal (nonradiolucent) as well as neurosurgical head holder devices (skull clamps) made of plastic material (radiolucent).
1.3 This specification represents the best currently available test procedures and is a minimum safety and performance standard.
1.4 This specification covers only those neurosurgical head holders (skull clamps) intended for use on humans for neurosurgical and spinal clinical applications. This specification assumes the user is well trained in the procedures and use of these devices including selection of the correct device type and accessories.
1.5 This specification describes those devices commonly known as skull clamps and accessories, such as skull pins, attachments, and various adaptors.
1.6 This specification only describes head holder devices that provide rigid skeletal fixation of the skull by means of three skull pins that penetrate the outer surface or outer layers of the patient’s head during neurosurgical or spinal procedures (compare with Ref (1)).2 Two pins are typically located in a 2-pin rocker, whereas the force delivery component is equipped with a single pin.
1.7 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. The values given in parentheses are for information only.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
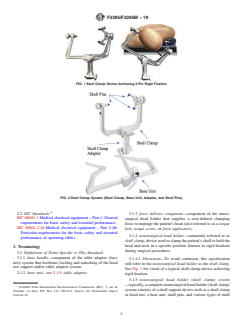
FIG. 1 Skull Clamp Device Achieving 3-Pin Rigid Fixation
FIG. 2 Skull Clamp System (Skull Clamp, Base Unit, Adaptor, and Skull Pins)
1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2019
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 01-Apr-2023
- Effective Date
- 15-Sep-2019
- Effective Date
- 15-Sep-2019
- Effective Date
- 15-Sep-2019
- Effective Date
- 01-Sep-2017
- Effective Date
- 15-May-2014
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-Oct-2011
- Effective Date
- 15-Apr-2011
- Effective Date
- 01-Mar-2011
- Effective Date
- 15-May-2010
- Effective Date
- 01-Apr-2010
- Effective Date
- 15-Nov-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Aug-2008
Overview
ASTM F3395/F3395M-19: Standard Specification for Neurosurgical Head Holder Devices provides a comprehensive framework for the performance testing, safety, and mechanical requirements of neurosurgical head holder devices, commonly known as skull clamps. Developed by ASTM International, this standard ensures manufacturers meet essential quality benchmarks when producing devices intended for rigid skeletal fixation of the skull during neurosurgical and spinal procedures. The specification addresses both metal (nonradiolucent) and plastic (radiolucent) head holders used in human clinical applications and establishes minimum requirements for safety, reliability, and performance.
Key Topics
Performance Testing and Safety
Manufacturers must meet precise mechanical and safety requirements, including load resistance, torque stability, and efficacy of fixation. The standard prescribes test methods for static load, creep (load loss), torque resistance, skull pin shear, and system integrity.Materials and Construction
Devices may use a variety of materials including stainless steel, polyoxymethylene, aluminum, and other rigid, non-creeping materials. All mechanical fixation components must be corrosion resistant and maintain functional integrity over their service life.Device Configuration
The specification covers neurosurgical head holders employing a three-pin design for rigid cranial fixation. Accessories such as skull pins, adaptors, and base units are also included under the requirements.MRI Compatibility
For devices intended for use in magnetic resonance environments, ASTM F3395/F3395M-19 mandates testing for MR safety, including requirements on materials (minimal or non-magnetic, non-conductive), displacement force, torque, RF-induced heating, and imaging artifact per referenced ASTM and IEC methods.Quality Assurance and Maintenance
Manufacturers are responsible for providing maintenance recommendations and ensuring the device performs safely and effectively across its lifecycle, including reprocessing suitability.
Applications
Neurosurgical Procedures
Rigid fixation of the patient’s head is critical during delicate neurosurgical operations. ASTM F3395/F3395M-19-standardized head holders minimize movement, thus reducing the risk of intraoperative injury and supporting surgical precision.Spinal Surgeries
Spinal procedures often require immobilization of the head and neck. Skull clamps conforming to this ASTM standard provide the required stability during complex procedures.Intraoperative Imaging
Devices built under this standard can be selected for use with MRI when appropriately tested and labeled, enabling real-time imaging and navigation during surgery.Medical Device Manufacturing and Procurement
Hospitals and surgical centers rely on this standard to evaluate and purchase neurosurgical head holder devices, ensuring compliance with international safety and performance benchmarks.
Related Standards
- ASTM D638 – Test Method for Tensile Properties of Plastics
- ASTM D695 – Test Method for Compressive Properties of Rigid Plastics
- ASTM D792 – Test Methods for Density and Specific Gravity of Plastics
- ASTM F2052, F2119, F2182, F2213, F2503 – Test methods for evaluation and marking of medical devices in MR environments
- IEC 60601-1 – General requirements for safety of medical electrical equipment
- IEC 60601-2-46 – Particular requirements for safety of operating tables
These referenced standards complement ASTM F3395/F3395M-19, supplying recognized procedures for performance testing and device labeling in clinical and MRI settings.
Keywords: ASTM neurosurgical head holder standard, skull clamp, medical device testing, MR compatibility, surgical fixation device, ASTM F3395, cranial fixation, patient safety, surgery instrumentation standards
Buy Documents
ASTM F3395/F3395M-19 - Standard Specification for Neurosurgical Head Holder Devices
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3395/F3395M-19 is a technical specification published by ASTM International. Its full title is "Standard Specification for Neurosurgical Head Holder Devices". This standard covers: SCOPE 1.1 This specification covers standards a manufacturer shall meet in the performance testing of neurosurgical head holder devices (skull clamps). 1.2 This specification covers neurosurgical head holder devices (skull clamps) made of metal (nonradiolucent) as well as neurosurgical head holder devices (skull clamps) made of plastic material (radiolucent). 1.3 This specification represents the best currently available test procedures and is a minimum safety and performance standard. 1.4 This specification covers only those neurosurgical head holders (skull clamps) intended for use on humans for neurosurgical and spinal clinical applications. This specification assumes the user is well trained in the procedures and use of these devices including selection of the correct device type and accessories. 1.5 This specification describes those devices commonly known as skull clamps and accessories, such as skull pins, attachments, and various adaptors. 1.6 This specification only describes head holder devices that provide rigid skeletal fixation of the skull by means of three skull pins that penetrate the outer surface or outer layers of the patient’s head during neurosurgical or spinal procedures (compare with Ref (1)).2 Two pins are typically located in a 2-pin rocker, whereas the force delivery component is equipped with a single pin. 1.7 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. The values given in parentheses are for information only. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. FIG. 1 Skull Clamp Device Achieving 3-Pin Rigid Fixation FIG. 2 Skull Clamp System (Skull Clamp, Base Unit, Adaptor, and Skull Pins) 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SCOPE 1.1 This specification covers standards a manufacturer shall meet in the performance testing of neurosurgical head holder devices (skull clamps). 1.2 This specification covers neurosurgical head holder devices (skull clamps) made of metal (nonradiolucent) as well as neurosurgical head holder devices (skull clamps) made of plastic material (radiolucent). 1.3 This specification represents the best currently available test procedures and is a minimum safety and performance standard. 1.4 This specification covers only those neurosurgical head holders (skull clamps) intended for use on humans for neurosurgical and spinal clinical applications. This specification assumes the user is well trained in the procedures and use of these devices including selection of the correct device type and accessories. 1.5 This specification describes those devices commonly known as skull clamps and accessories, such as skull pins, attachments, and various adaptors. 1.6 This specification only describes head holder devices that provide rigid skeletal fixation of the skull by means of three skull pins that penetrate the outer surface or outer layers of the patient’s head during neurosurgical or spinal procedures (compare with Ref (1)).2 Two pins are typically located in a 2-pin rocker, whereas the force delivery component is equipped with a single pin. 1.7 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system are not necessarily exact equivalents; therefore, to ensure conformance with the standard, each system shall be used independently of the other, and values from the two systems shall not be combined. The values given in parentheses are for information only. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. FIG. 1 Skull Clamp Device Achieving 3-Pin Rigid Fixation FIG. 2 Skull Clamp System (Skull Clamp, Base Unit, Adaptor, and Skull Pins) 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3395/F3395M-19 is classified under the following ICS (International Classification for Standards) categories: 11.040.30 - Surgical instruments and materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3395/F3395M-19 has the following relationships with other standards: It is inter standard links to ASTM F2503-23e1, ASTM F2182-19, ASTM F2182-19e2, ASTM F2182-19e1, ASTM F2213-17, ASTM F2052-14, ASTM F2503-13, ASTM F2213-06(2011), ASTM F2182-11a, ASTM F2182-11, ASTM D638-10, ASTM D695-10, ASTM F2182-09, ASTM F2503-08, ASTM D695-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3395/F3395M-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3395/F3395M −19
Standard Specification for
Neurosurgical Head Holder Devices
ThisstandardisissuedunderthefixeddesignationF3395/F3395M;thenumberimmediatelyfollowingthedesignationindicatestheyear
of original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.
A superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope used independently of the other, and values from the two
systems shall not be combined. The values given in parenthe-
1.1 This specification covers standards a manufacturer shall
ses are for information only.
meet in the performance testing of neurosurgical head holder
1.8 This standard does not purport to address all of the
devices (skull clamps).
safety concerns, if any, associated with its use. It is the
1.2 This specification covers neurosurgical head holder
responsibility of the user of this standard to establish appro-
devices (skull clamps) made of metal (nonradiolucent) as well
priate safety, health, and environmental practices and deter-
as neurosurgical head holder devices (skull clamps) made of
mine the applicability of regulatory limitations prior to use.
plastic material (radiolucent).
1.9 This international standard was developed in accor-
1.3 Thisspecificationrepresentsthebestcurrentlyavailable
dance with internationally recognized principles on standard-
test procedures and is a minimum safety and performance
ization established in the Decision on Principles for the
standard.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.4 This specification covers only those neurosurgical head
Barriers to Trade (TBT) Committee.
holders (skull clamps) intended for use on humans for neuro-
surgical and spinal clinical applications. This specification
2. Referenced Documents
assumes the user is well trained in the procedures and use of
thesedevicesincludingselectionofthecorrectdevicetypeand 2.1 ASTM Standards:
accessories. D638Test Method for Tensile Properties of Plastics
D695Test Method for Compressive Properties of Rigid
1.5 This specification describes those devices commonly
Plastics
known as skull clamps and accessories, such as skull pins,
D792Test Methods for Density and Specific Gravity (Rela-
attachments, and various adaptors.
tive Density) of Plastics by Displacement
1.6 This specification only describes head holder devices
F2052Test Method for Measurement of Magnetically In-
that provide rigid skeletal fixation of the skull by means of
duced Displacement Force on Medical Devices in the
three skull pins that penetrate the outer surface or outer layers
Magnetic Resonance Environment
of the patient’s head during neurosurgical or spinal procedures
F2119Test Method for Evaluation of MR Image Artifacts
(compare with Ref (1)). Two pins are typically located in a
from Passive Implants
2-pin rocker, whereas the force delivery component is
F2182Test Method for Measurement of Radio Frequency
equipped with a single pin.
Induced Heating On or Near Passive Implants During
1.7 The values stated in either SI units or inch-pound units Magnetic Resonance Imaging
are to be regarded separately as standard. The values stated in F2213Test Method for Measurement of Magnetically In-
each system are not necessarily exact equivalents; therefore, to duced Torque on Medical Devices in the Magnetic Reso-
ensure conformance with the standard, each system shall be nance Environment
F2503Practice for Marking Medical Devices and Other
Items for Safety in the Magnetic Resonance Environment
ThistestmethodisunderthejurisdictionofASTMCommitteeF04onMedical
andSurgicalMaterialsandDevicesandisthedirectresponsibilityofSubcommittee
F04.25 on Spinal Devices.
Current edition approved Sept. 1, 2019. Published October 2019. DOI: 10.1520/ For referenced ASTM standards, visit the ASTM website, www.astm.org, or
F3395_F3395M-19. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to a list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3395/F3395M − 19
FIG. 1 Skull Clamp Device Achieving 3-Pin Rigid Fixation
FIG. 2 Skull Clamp System (Skull Clamp, Base Unit, Adaptor, and Skull Pins)
2.2 IEC Standards: 3.1.3 force delivery component—component of the neuro-
IEC 60601-1Medical electrical equipment – Part 1: General
surgical head holder that supplies a user-defined clamping
requirements for basic safety and essential performance
force to impinge the patient’s head (also referred to as a torque
IEC 60601-2-46Medical electrical equipment – Part 2-46:
bolt, torque screw,or force applicator).
Particular requirements for the basic safety and essential
3.1.4 neurosurgical head holder—commonly referred to as
performance of operating tables
skull clamp,deviceusedtoclampthepatient’sskulltoholdthe
head and neck in a specific position (known as rigid fixation)
3. Terminology
during surgical procedures.
3.1 Definitions of Terms Specific to This Standard:
3.1.1 base handle—component of the table adaptor (base
3.1.4.1 Discussion—To avoid confusion, this specification
unit) system that facilitates locking and unlocking of the head
will refer to the neurosurgical head holder as the skull clamp.
rest support and/or table adaptor system.
See Fig. 1 for visual of a typical skull clamp device achieving
3.1.2 base unit—see 3.1.9, table adaptor.
rigid fixation.
3.1.5 neurosurgical head holder (skull clamp) system
—typically,acompleteneurosurgicalheadholder(skullclamp)
Available from International Electrotechnical Commission (IEC), 3, rue de
system consists of a skull support device such as a skull clamp
Varembé, 1st floor, P.O. Box 131, CH-1211, Geneva 20, Switzerland, https://
www.iec.ch. or head rest, a base unit, skull pins, and various types of skull
F3395/F3395M − 19
clamp adaptors. See Fig. 2 for a visual (with high level 6. Conformance
terminology) of a typical skull clamp system.
6.1 Presently, this specification is voluntary and not re-
3.1.6 skull clamp—see 3.1.4, neurosurgical head holder.
quired by law. A manufacturer may label a product as con-
forming to this specification only if the product meets all the
3.1.7 skull clamp adaptor—accessory of the neurosurgical
head holder (skull clamp) system that allows connection of requirements of this specification.
various components to the table adaptor.
7. Classification
3.1.8 skull pin—rigid pin point devices used in conjunction
with skull clamping devices which, when properly applied,
7.1 Intraoperative Neurosurgical Head Holder (Skull
impinge the skull. Skull pins are intended to be sterile at the
Clamp) System—Typically, a complete neurosurgical head
time of use.
holder (skull clamp) system consists of a skull support device
3.1.9 table adaptor (base assembly)—commonly referred to such as a skull clamp or head rest, a base unit, skull pins, and
various types of system adaptors. The uniqueness of these
as base unit, device used to provide attachment from the
systems is their ability to provide surgeons with numerous
operating room table to a skull clamp or other neurosurgical
device. degrees of freedom and patient position options.
3.1.9.1 Discussion—To avoid confusion, this specification
8. Magnetic Resonance Imaging (MRI) Compatibility
will refer to the table adaptor as the base unit.
Requirements
3.1.10 torque screw—(sometimes referred to as torque bolt
or force applicator) supplies a user-defined clamping force to 8.1 Forneurosurgicalheadholders(skullclamps)tobeused
with intraoperative MRI imaging only:
impinge the patients head.
8.1.1 These neurosurgical head holder device (skull clamp)
3.1.10.1 Discussion—To avoid confusion, this specification
MRI compatibility requirements are intended to protect the
will refer to the torque screw as the force delivery component
patient and users from harm in the MR environment.
(see 3.1.3, force delivery component.
8.1.2 ManufacturersshallberesponsiblefortestingtheMRI
4. Recommended Auxiliary Materials safety and efficacy of the device.
8.1.3 Test Methods
4.1 Definitions of Auxiliary Materials for this Specification:
8.1.3.1 For neurosurgical head holders (skull clamps) de-
4.1.1 Analogue Cortical Bone—Material that mimics a hu-
signed for use in the MR environment, the manufacturer shall
man cortical skull bone and features the following properties:
–3
ensurethatitsconstructionusesaminimumamountofmetallic
4.1.1.1 Density, 1.7 60.2gcm (Test Methods D792).
and/or conductive components.
4.1.1.2 Compression Modulus, 15.0 6 3.0 GPa (Test
Method D695).
8.1.3.2 Where metallic components are present and cannot
4.1.1.3 Transverse Tension Modulus, 10.4 6 3.0 GPa (Test be avoided, the materials used shall be non-magnetic and
Method D638).
non-conductive to the greatest extent possible.
4.1.1.4 These properties are in line with values of a human
8.1.3.3 Perform testing and analyses referenced in Practice
cortical skull bone given in the literature (2), (3).
F2503 to evaluate the safety of the device in the MR environ-
4.1.2 Rigid Non-Creeping Material—Materials with a
ment. Label the device MR Safe, MR Unsafe, or MR Condi-
Young’s modulus (E) > 2.5 GPa. Effective materials include
tional as defined in Practice F2503.
the following:
8.1.3.4 Theworst-caseconfigurationshallbedeterminedfor
4.1.2.1 Stainless Steel, E ≈ 203.0 GPa.
each evaluation, and the tests shall be conducted on the
4.1.2.2 Polyoxymethylene (POM), E ≈ 2.6 to 3.1 GPa.
identified worst-case configurations.
4.1.2.3 Aluminum 6061-T6, E ≈ 68.9 GPa.
8.1.3.5 Evaluate the magnetically induced displacement
4.1.2.4 Garolite (G-10 Fiberglass Epoxy Laminate), E ≈
produced by the device. Perform testing and analyses outlined
18.6 GPa).
in Test Method F2052.
4.1.2.5 Phenolic Laminate, E ≈ 9.65 GPa.
8.1.3.6 Evaluate the magnetically induced torque produced
4.1.2.6 Oak Wood, E ≈ 8.6 to 11.0 GPa.
by the device. Perform testing and analyses outlined in Test
4.1.2.7 Solid Polyurethane Foam, E ≈ 10 to 16 GPa.
Method F2213.
5. Recommended Tolerances 8.1.3.7 Assessment of radio frequency (RF)-induced heat-
ing for these devices is complex. While Test Method F2182 is
5.1 Force Tolerance—The recommended tolerances for the
limited to devices entirely implanted inside the body, RF-
force values listed throughout this specification are 68.9 N
induced heating can be evaluated experimentally and/or com-
(62.0 lbf) unless otherwise specified.
putationally using a method similar to that described in Test
5.2 Mass Tolerance—The recommended tolerances for the
Method F2182, with modifications for head holder devices.
mass values listed throughout this specification are 61.0 kg
The components that are in contact with the patient need to be
(62.2 lb) unless otherwise specified.
monitored, and particular attention needs to be paid to deter-
5.3 Time Tolerance—The recommended tolerances for the mining the worst-case configuration (4). In addition to the
time values listed throughout this specification are 61.0 s possibility of antenna resonant effects, these devices may
unless otherwise specified. create conductive loops that may result in heating.
F3395/F3395M − 19
8.1.3.8 Evaluate the MR image artifact produced by the 25.0lb).Witha2×safetyfactor,theskullclampshallbetested
device. Perform testing and analyses outlined in Test Method toholdaminimumverticalshearforceofatleast222.4N(50.0
F2119. lbf).
9.4.4 The load requirements may vary due to its intended
8.1.3.9 Testing shall encompass the entirety of the cranial
use. IEC 60601-1 may be consulted for load distributions for
stabilization system exposed to the MR environment including
normal patients. IEC 60601-2-46 may also be consulted for
accessories and ancillary devices.
weight distributions of larger patients.
9.4.5 The test method is defined in Section 14.
9. Mechanical Testing
9.5 Force Delivery Accuracy Verification
9.1 Static Load Test
9.5.1 The skull clamp force delivery component is used to
9.1.1 To ensure mechanical integrity of the skull clamp
apply a user-defined force to the patient’s head.
underload,theskullclampistestedtowithstandstaticloading.
9.5.2 Theforcedeliverycomponentistypicallyanassembly
9.1.2 To ensure that the device is free from deformation
of various components. As such, the totality of the finished
under load, the minimum applied force shall be equal to the
assembly shall be verified to ensure delivery of the prescribed
maximum force able to be exerted by the device’s force
force (e.g., not only a compression spring).
delivery component for a period of 24 h. A typical surgical
9.5.3 The test method is defined in Section 15.
procedure involving these devices could range from 8 to 12 h,
with 24 h representing a worst-case duration to account for 9.6 Skull Pin Shear Test
extensive surgical procedures. Because mechanical failure of 9.6.1 The skull pin must be capable of resisting shear force
the device could lead to a sudden loss of functionality and/or arising out of the weight of the patient’s head, and surgical
patient support, the system shall be tested at this maximum activities without breaking, and without skull pin locomotion
force (simulating the maximum clamping force of the force perpendicular to the pinned skull bone surface. Potential
delivery component) for 24 h. vibrations of the patient’s head occurring during surgery are
not considered in this skull pin test, as vibrations may occur
9.1.3 The test method is defined in Section 11.
although the mechanical integrity of the skull pin is given.
9.2 Creep Test (Load Loss)
9.6.2 Ahumanheadweighsapproximately3.6to5.4kg(8.0
9.2.1 The skull clamp must be able to achieve and maintain
to11.9lb).Consideringadditionalloadsappliedbythesurgeon
a user-defined force for a specified period of time during
andinstrumentstheskullpinsmustsustainloadsashighas9.1
utilization.
to 11.3 kg (20.0 to 25.0 lb). During surgical application, this
9.2.2 Extensive surgical procedures for which this device
load is distributed between all utilized skull pins.The theoreti-
will be used may be up to 12 h in duration. To account for
calmaximumloadaskullpinisexposedtooccursonthesingle
extensive surgical procedures, this test method represents a
pin side in a 3-pin rigid cranial fixation system. Including a 2×
worst-casedurationof24h.Itisimportantthattheskullclamp
safety factor, this load reaches 9.1 to 11.3 kg (20.0 to 25.0 lb).
maintains its support throughout clinical use.
Therefore, a skull pin shall be tested to withstand a perpen-
9.2.3 The test method is defined in Section 12.
dicular tensile force of 111.0 N (25.0 lbf).
9.6.3 The test method is defined in Section 16.
9.3 Torque Load Resistance Test
9.3.1 Askull clamp is designed to allow proper positioning
10. Performance Requirements
and locking by the surgeon to achieve rigid fixation. This
locking is critical as the patient’s head must be restricted from
10.1 The purpose of these requirements is to ensure that the
unintentional movement.
skull clamp devices meet the minimum performance require-
9.3.2 The torque load test is intended to ensure the skull ments as originally designed. The skull clamp device require-
clamp can withstand rotational movement when in use without ments should not vary from procedure to procedure provided
impacting the locking mechanism. they are used and maintained according to the manufacturer’s
9.3.3 The test method is defined in Section 13. recommendation.
10.2 Performance Requirement
9.4 System Test
10.2.1 All mechanical fixation components shall be manu-
9.4.1 The skull clamp system, which consists of the skull
factured out of corrosion resistant materials.
clampandthetableadaptor,isintendedtoensurerigidityofthe
10.2.2 All components shall be manufactured from materi-
system. The test verifies that the system is capable of resisting
alscapableofprovidingfunctionalintegrityovertheusefullife
forces imposed by the patient and the surgeon when in use and
of the device.
in the locked position.
10.2.3 The manufacturer shall be responsible for maintain-
9.4.2 The table adaptor system is an assembly of various
ing adequate mechanical test data or equivalent clinical data
components (table adaptor, skull clamp adapter). As such, the
regarding the suitability of design, useful life, and diagnostic
totalityofthefinishedassemblyincludingtheskullclampshall
imaging compatibility of the system.
be verified to ensure its capability to resist movement when
10.2.4 The manufacturer shall be responsible for supplying
locked and in use.
materials that are suitable to be reprocessed by the manufac-
9.4.3 Taking into account the average weight of a human
turer’s recommended reprocessing techniques.
head and corresponding applied loads detailed in 9.4.1, the
device could see static loads as high as 9.1 to 11.3 kg (20.0 to 10.3 Maintenance Requirement
F3395/F3395M − 19
10.3.1 Themanufacturershallberesponsibleforidentifying 11.1.4.2 Fasten the weight application linkage to the clamp
appropriate maintenance requirements and intervals necessary extension so that the force will be applied along the axis of the
to ensure device performance throughout its useful life. force delivery component.
11.1.4.3 Gradually increase the weight applied to the link-
10.4 If required, CT / MRI compatibility shall be tested.
ageuntiltheclampsupportsaforceequivalenttothemaximum
force able to b
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...