ASTM F3574-22
(Test Method)Standard Test Methods for Sacroiliac Joint Fusion Devices
Standard Test Methods for Sacroiliac Joint Fusion Devices
SIGNIFICANCE AND USE
4.1 The function of the SIJ fusion device assembly is to stabilize the SIJ to facilitate arthrodesis of the motion segment. This test method outlines materials and methods for the characterization and evaluation of the mechanical performance of different SIJ fusion device assemblies so that comparisons can be made between different designs.
4.2 These test methods are designed to quantify the static and dynamic characteristics of different designs of SIJ fusion device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of SIJ fusion device assemblies to specific force modalities.
4.3 The forces applied to the SIJ fusion device assemblies during the tests described herein may differ from the complex loading seen in vivo and, therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare mechanical performance of different SIJ fusion device assemblies.
4.4 Since the environment may affect the dynamic performance of SIJ fusion device assemblies, dynamic testing in a saline environment may be considered for implants with wearing surfaces or with movable components or for implants with components that are temperature dependent. Fatigue tests should first be conducted in air (at ambient temperature) for comparison purposes since the environmental effects could be significant. If a simulated in vivo environment is necessary, the investigator should consider testing in a saline environmental bath at 37 °C (for example, 0.9 g NaCl per 100 mL water). A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37 °C could also be used with adequate justification.
4.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution as described in 4.4. Devices that require physiologic solution for testing should be tested in the same type of soluti...
SCOPE
1.1 These test methods cover the materials and methods for the static and dynamic testing of sacroiliac joint (SIJ) fusion device assemblies, SIJ implants designed to promote arthrodesis at the sacroiliac joint.
1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic SIJ fusion device assemblies. These test methods allow for comparison of SIJ fusion device assemblies intended to be implanted with a trajectory in line with the joint space (in-line implant) or for comparison of SIJ fusion devices intended for implantation across the joint space (transverse implant). These test methods are intended enable the user to compare SIJ fusion device assemblies mechanically and do not purport to provide performance standards for SIJ fusion device assemblies.
1.3 These tests describe static and dynamic tests by specifying force types and specific methods of applying these forces. These tests are designed to allow for the comparative evaluation of SIJ device assemblies.
1.4 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the stiffness and strength of the SIJ fusion device assemblies.
1.5 Some SIJ fusion device assemblies may not be testable in all test configurations.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurements are included in this standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recogniz...
General Information
- Status
- Published
- Publication Date
- 31-May-2022
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 15-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Feb-2018
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
Overview
ASTM F3574-22 is an international standard developed by ASTM International, specifying test methods for sacroiliac joint (SIJ) fusion devices. These devices are intended to stabilize the sacroiliac joint for the purpose of facilitating arthrodesis-fusion of the motion segment between the sacrum and ilium. This standard provides comprehensive procedures for mechanical performance testing, enabling manufacturers and researchers to evaluate and compare SIJ fusion device assemblies under controlled laboratory conditions.
The standard addresses both static and dynamic testing of SIJ fusion implants. Testing is conducted in vitro to assess static and fatigue characteristics, with emphasis on comparability across various design configurations. While the applied forces and conditions may not completely replicate in vivo environments, the results offer reliable metrics for device evaluation and regulatory submissions.
Key Topics
- Mechanical Characterization: Outlines methodologies for evaluating mechanical properties such as yield force/moment, ultimate force/moment, stiffness, and displacement. Both in-line and transverse implant trajectories are addressed.
- Static and Dynamic Testing: Defines test protocols for both static (single loading event) and dynamic (cyclic loading/fatigue) testing, including criteria for failure, runouts, and data reporting.
- Environmental Considerations: Recognizes the significance of testing in ambient conditions (air at room temperature) and in simulated physiologic environments (e.g., saline at 37°C) for devices sensitive to factors like temperature or lubrication.
- Test Configurations: Specifies the use of test blocks or potting mediums to simulate the joint space geometry and secure the device during testing. Provides guidance for both screw-based and other mechanical fixation designs.
- Comparative Analysis: Enables side-by-side comparison of mechanical performance for past, present, and future nonbiologic SIJ fusion devices, supporting device innovation and benchmarking.
- Reporting Requirements: Establishes detailed guidelines for documentation, including test setup, specimen information, loading protocols, failure modes, and statistical analysis.
Applications
ASTM F3574-22 is widely used in the development, validation, and regulatory approval of SIJ fusion devices. Its practical value includes:
- Product Development: Assisting medical device manufacturers in assessing mechanical strength, durability, and performance of new and existing SIJ implants.
- Research and Innovation: Facilitating academic and clinical research by providing standardized methods for evaluating novel device designs or materials for SIJ fusion.
- Regulatory Compliance: Supporting pre-clinical testing submissions for regulatory bodies by providing recognized, reproducible test results.
- Quality Control: Enabling quality assurance teams to consistently monitor the mechanical attributes of finished SIJ devices.
The standard is applicable to various implant designs, including both in-line (within the joint space) and transverse implants (across the joint space), and accommodates variations in surgical approach and fixation methodology.
Related Standards
ASTM F3574-22 references and is often used in conjunction with several other ASTM standards relevant to medical and spinal devices, such as:
- ASTM E4 - Practices for Force Calibration and Verification of Testing Machines
- ASTM E6 - Terminology Relating to Methods of Mechanical Testing
- ASTM E691 - Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM F543 - Specification and Test Methods for Metallic Medical Bone Screws
- ASTM F2077 - Test Methods for Intervertebral Body Fusion Devices
- ASTM F2193 - Specifications and Test Methods for Components Used in the Surgical Fixation of the Spinal Skeletal System
These related standards support comprehensive testing and validation of orthopaedic and spinal implant devices, ensuring consistent terminology, methodology, and data integrity across the industry.
Keywords: ASTM F3574, sacroiliac joint fusion device, SIJ implant testing, mechanical performance, static and dynamic testing, orthopedic device standard, regulatory compliance, SI joint arthrodesis, fatigue testing
Buy Documents
ASTM F3574-22 - Standard Test Methods for Sacroiliac Joint Fusion Devices
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3574-22 is a standard published by ASTM International. Its full title is "Standard Test Methods for Sacroiliac Joint Fusion Devices". This standard covers: SIGNIFICANCE AND USE 4.1 The function of the SIJ fusion device assembly is to stabilize the SIJ to facilitate arthrodesis of the motion segment. This test method outlines materials and methods for the characterization and evaluation of the mechanical performance of different SIJ fusion device assemblies so that comparisons can be made between different designs. 4.2 These test methods are designed to quantify the static and dynamic characteristics of different designs of SIJ fusion device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of SIJ fusion device assemblies to specific force modalities. 4.3 The forces applied to the SIJ fusion device assemblies during the tests described herein may differ from the complex loading seen in vivo and, therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare mechanical performance of different SIJ fusion device assemblies. 4.4 Since the environment may affect the dynamic performance of SIJ fusion device assemblies, dynamic testing in a saline environment may be considered for implants with wearing surfaces or with movable components or for implants with components that are temperature dependent. Fatigue tests should first be conducted in air (at ambient temperature) for comparison purposes since the environmental effects could be significant. If a simulated in vivo environment is necessary, the investigator should consider testing in a saline environmental bath at 37 °C (for example, 0.9 g NaCl per 100 mL water). A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37 °C could also be used with adequate justification. 4.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution as described in 4.4. Devices that require physiologic solution for testing should be tested in the same type of soluti... SCOPE 1.1 These test methods cover the materials and methods for the static and dynamic testing of sacroiliac joint (SIJ) fusion device assemblies, SIJ implants designed to promote arthrodesis at the sacroiliac joint. 1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic SIJ fusion device assemblies. These test methods allow for comparison of SIJ fusion device assemblies intended to be implanted with a trajectory in line with the joint space (in-line implant) or for comparison of SIJ fusion devices intended for implantation across the joint space (transverse implant). These test methods are intended enable the user to compare SIJ fusion device assemblies mechanically and do not purport to provide performance standards for SIJ fusion device assemblies. 1.3 These tests describe static and dynamic tests by specifying force types and specific methods of applying these forces. These tests are designed to allow for the comparative evaluation of SIJ device assemblies. 1.4 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the stiffness and strength of the SIJ fusion device assemblies. 1.5 Some SIJ fusion device assemblies may not be testable in all test configurations. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurements are included in this standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recogniz...
SIGNIFICANCE AND USE 4.1 The function of the SIJ fusion device assembly is to stabilize the SIJ to facilitate arthrodesis of the motion segment. This test method outlines materials and methods for the characterization and evaluation of the mechanical performance of different SIJ fusion device assemblies so that comparisons can be made between different designs. 4.2 These test methods are designed to quantify the static and dynamic characteristics of different designs of SIJ fusion device assemblies. These tests are conducted in vitro to allow for analysis and comparison of the mechanical performance of SIJ fusion device assemblies to specific force modalities. 4.3 The forces applied to the SIJ fusion device assemblies during the tests described herein may differ from the complex loading seen in vivo and, therefore, the results from these tests may not directly predict in vivo performance. The results, however, can be used to compare mechanical performance of different SIJ fusion device assemblies. 4.4 Since the environment may affect the dynamic performance of SIJ fusion device assemblies, dynamic testing in a saline environment may be considered for implants with wearing surfaces or with movable components or for implants with components that are temperature dependent. Fatigue tests should first be conducted in air (at ambient temperature) for comparison purposes since the environmental effects could be significant. If a simulated in vivo environment is necessary, the investigator should consider testing in a saline environmental bath at 37 °C (for example, 0.9 g NaCl per 100 mL water). A simulated body fluid, a saline drip or mist, distilled water, or other type of lubrication at 37 °C could also be used with adequate justification. 4.5 If the devices are known to be temperature and environment dependent, testing should be conducted in physiologic solution as described in 4.4. Devices that require physiologic solution for testing should be tested in the same type of soluti... SCOPE 1.1 These test methods cover the materials and methods for the static and dynamic testing of sacroiliac joint (SIJ) fusion device assemblies, SIJ implants designed to promote arthrodesis at the sacroiliac joint. 1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future nonbiologic SIJ fusion device assemblies. These test methods allow for comparison of SIJ fusion device assemblies intended to be implanted with a trajectory in line with the joint space (in-line implant) or for comparison of SIJ fusion devices intended for implantation across the joint space (transverse implant). These test methods are intended enable the user to compare SIJ fusion device assemblies mechanically and do not purport to provide performance standards for SIJ fusion device assemblies. 1.3 These tests describe static and dynamic tests by specifying force types and specific methods of applying these forces. These tests are designed to allow for the comparative evaluation of SIJ device assemblies. 1.4 Guidelines are established for measuring displacements, determining the yield force or moment, and evaluating the stiffness and strength of the SIJ fusion device assemblies. 1.5 Some SIJ fusion device assemblies may not be testable in all test configurations. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurements are included in this standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recogniz...
ASTM F3574-22 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3574-22 has the following relationships with other standards: It is inter standard links to ASTM E1823-24a, ASTM E1823-24, ASTM F2193-20, ASTM E1823-20, ASTM E2309/E2309M-20, ASTM F2193-18a, ASTM F2193-18, ASTM F2077-17, ASTM F1582-98(2016), ASTM F2077-14, ASTM F2193-14, ASTM E4-14, ASTM F543-13, ASTM E691-13, ASTM F543-13e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3574-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3574 − 22
Standard Test Methods for
Sacroiliac Joint Fusion Devices
This standard is issued under the fixed designation F3574; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
1.1 These test methods cover the materials and methods for
mendations issued by the World Trade Organization Technical
the static and dynamic testing of sacroiliac joint (SIJ) fusion
Barriers to Trade (TBT) Committee.
device assemblies, SIJ implants designed to promote arthrod-
esis at the sacroiliac joint.
2. Referenced Documents
1.2 These test methods are intended to provide a basis for
2.1 ASTM Standards:
the mechanical comparison among past, present, and future
E4Practices for Force Calibration and Verification of Test-
nonbiologic SIJ fusion device assemblies. These test methods
ing Machines
allow for comparison of SIJ fusion device assemblies intended
E6Terminology Relating to Methods of Mechanical Testing
to be implanted with a trajectory in line with the joint space
E691Practice for Conducting an Interlaboratory Study to
(in-line implant) or for comparison of SIJ fusion devices
Determine the Precision of a Test Method
intended for implantation across the joint space (transverse
E1823TerminologyRelatingtoFatigueandFractureTesting
implant). These test methods are intended enable the user to
E2309/E2309MPractices for Verification of Displacement
compareSIJfusiondeviceassembliesmechanicallyanddonot
Measuring Systems and Devices Used in MaterialTesting
purporttoprovideperformancestandardsforSIJfusiondevice
Machines
assemblies.
F543Specification and Test Methods for Metallic Medical
1.3 These tests describe static and dynamic tests by speci-
Bone Screws
fying force types and specific methods of applying these
F1582Terminology Relating to Spinal Implants
forces. These tests are designed to allow for the comparative
F1839SpecificationforRigidPolyurethaneFoamforUseas
evaluation of SIJ device assemblies.
a Standard Material for Testing Orthopaedic Devices and
Instruments
1.4 Guidelinesareestablishedformeasuringdisplacements,
F2077TestMethodsForIntervertebralBodyFusionDevices
determining the yield force or moment, and evaluating the
F2193Specifications and Test Methods for Components
stiffness and strength of the SIJ fusion device assemblies.
Used in the Surgical Fixation of the Spinal Skeletal
1.5 Some SIJ fusion device assemblies may not be testable
System
in all test configurations.
1.6 The values stated in SI units are to be regarded as 3. Terminology
standard. No other units of measurements are included in this
3.1 For definitions of terms, refer to Terminologies E6,
standard, with the exception of angular measurements, which
E1823, and F1582, and the Terminology section in Specifica-
may be reported in terms of either degrees or radians.
tions F543 and F2193.
1.7 This standard does not purport to address all of the
3.2 Definitions of Terms Specific to This Standard:
safety concerns, if any, associated with its use. It is the
3.2.1 axial pullout strength, n—the maximum tensile force
responsibility of the user of this standard to establish appro-
perAnnexA2requiredtofailorremoveatransversesacroiliac
priate safety, health, and environmental practices and deter-
joint fusion implant from a material into which the device has
mine the applicability of regulatory limitations prior to use.
been inserted.
1.8 This international standard was developed in accor-
3.2.2 bending fatigue runout moment (N-m), n—valueofthe
dance with internationally recognized principles on standard-
maximum moment under dynamic cantilever bending per
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Subcommittee F04.25 on Spinal Devices. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved June 1, 2022. Published June 2022. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
F3574-22. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3574 − 22
Annex A2 that can be applied to a transverse sacroiliac joint 3.2.8 coordinate system/axes (transverse implants),
fusionimplantwhereallthetestedspecimenshaveexperienced n—threeorthogonalaxesforatransverseSIJfusionimplantare
2500000loadingcycleswithoutafailureataspecificR-ratio. defined in terms of the implant design and the joint space (Fig.
5).TheoriginofthetransverseSIJcoordinatesystemislocated
3.2.3 bending moment arm, L (mm), n—distance in mm
at the geometric center of the device assembly. The X-axis
between the point where a transverse sacroiliac joint fusion
corresponds to the long axis of the implant in the direction of
implant test specimen is gripped (typically the axis of the
trajectory. The Y passes in the superior-inferior direction
longitudinal element) and the line of action for the applied
through a plane parallel to the plane tangential to the joint
force in cantilever bending per Annex A2 prior to any
space.TheZ-axisistheresultantaxisdependentontheimplant
deformation of the assembly.
trajectory. Torsional force is defined to be the component of
3.2.4 bending stiffness, S (N/mm), n—slope of the initial
moment about the X-axis.
linear elastic portion of the load versus total displacement
3.2.9 core diameter, n—the smallest diameter of the
curve (slope of Line Om in Fig. A1.3) for static cantilever
threadedportionofathreadedtransversesacroiliacjointfusion
bending of a transverse sacroiliac joint fusion implant.
implant measured at the thread root. This is also known as the
3.2.5 bending ultimate moment (N-m), n—maximum bend-
minor diameter.
ing moment in static cantilever bending that can be applied to
atransversesacroiliacjointfusionimplanttestsample;Point E
3.2.10 crack, n—anexternallyvisiblephysicaldiscontinuity
in Fig. A1.3. in the form of a narrow opening that arises from mechanical
forces.
3.2.6 bending yield moment (N-m), n—bending moment in
static cantilever bending necessary to produce a 0.2% offset
3.2.11 fatigue life, n—the number of cycles, N, that the SIJ
displacement in the transverse sacroiliac joint fusion implant.
fusion device assembly can sustain at a particular force or
If the specimen fractures before the test reaches the 0.2%
moment before mechanical or functional failure.
offsetdisplacementpoint,thebendingmomentshallbedefined
3.2.12 force point, n—the point through which the resultant
as the bending moment at fracture.
force on the SIJ device passes.
3.2.7 coordinate system/axes (in-line implants), n—three
3.2.13 functional failure, n—permanent deformation that
orthogonal axes for an in-line SIJ fusion implant are defined in
renders the SIJ fusion device assembly ineffective or unable to
termsofthejointspaceandtheimplantdesign(Figs.1-4).The
resist force and/or maintain attachment adequately.
origin of the in-line SIJ coordinate system is located at the
geometric center of the device assembly. The X-axis corre-
3.2.14 gauge length, n—the distance between the holding
sponds to the trajectory of the implant. The Y-axis passes
device (for example, a split collet) and the underside of the
tangentially through the joint space. The Z-axis passes normal
headforatransversesacroiliacjointfusionimplantintorsional
to the joint space. The XY plane is to bisect the joint space
testing.
between iliac (lateral) and sacral (medial) surfaces. Force
3.2.15 grip length, n—thenumberofthreadsheldfastinthe
components parallel to the XY plane are shear components of
split collet or holding mechanism during torsional testing of a
loading. Torsional force is defined to be the component of
transverse sacroiliac joint fusion implant.
moment about the Z-axis.
3.2.16 ideal insertion location, n—theimplantlocationwith
respect to the simulated ilium (lateral) and sacrum (medial)
articulating surfaces (bone cement) dictated by the type,
design, and manufacturer’s surgical installation instructions.
3.2.17 in-line implant, n—adeviceintendedtobeimplanted
with a trajectory primarily within the sacroiliac joint space
(Figs. 1-4); this kind of device may have integrated fixation
(that is, screws, blades) into the sacrum and ilium.
3.2.18 insertion depth, n—the length of a transverse sacro-
iliac joint fusion implant that is inserted into the test block for
axial pullout testing.
3.2.19 intended method of application, n—SIJfusiondevice
assemblies may contain different types of stabilizing anchors
such as threads, spikes, and knurled surfaces. Each type of
anchor has an intended method of application or attachment to
the sacral and iliac bones.
3.2.20 intended SIJ location, n—the anatomic region of the
sacroiliacjointintendedfortheSIJfusiondeviceassembly.SIJ
fusion device assemblies may be designed and developed for
specific anatomical regions such as primarily within the joint
FIG. 1 Orthogonal Coordinate System for Testing of an In-Line
SIJ Fusion Implant space or across the joint space. Also, there exist different
F3574 − 22
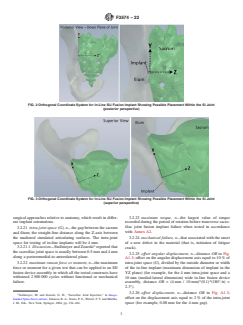
FIG. 2 Orthogonal Coordinate System for In-Line SIJ Fusion Implant Showing Possible Placement Within the SI Joint
(posterior perspective)
FIG. 3 Orthogonal Coordinate System for In-Line SIJ Fusion Implant Showing Possible Placement Within the SI Joint
(superior perspective)
surgical approaches relative to anatomy, which result in differ- 3.2.23 maximum torque, n—the largest value of torque
ent implant orientations. recorded during the period of rotation before transverse sacro-
iliac joint fusion implant failure when tested in accordance
3.2.21 intra-joint space (G), n—thegapbetweenthesacrum
with Annex A2.
and ilium; the straight-line distance along the Z-axis between
the unaltered simulated articulating surfaces. The intra-joint 3.2.24 mechanical failure, n—that associated with the onset
space for testing of in-line implants will be 4mm.
of a new defect in the material (that is, initiation of fatigue
3.2.21.1 Discussion—Stallmeyer and Zoarski reported that crack).
the sacroiliac joint space is usually between 0.5mm and 4mm
3.2.25 offset angular displacement, n—distance OB in Fig.
along a posteromedial-to-anterolateral plane.
A1.3; offset on the angular displacement axis equal to 10% of
3.2.22 maximum runout force or moment, n—the maximum
intra-joint space (G), divided by the outside diameter or width
force or moment for a given test that can be applied to an SIJ of the in-line implant (maximum dimension of implant in the
fusion device assembly in which all the tested constructs have
YZ plane) (for example, for the 4mm intra-joint space and a
withstood 2500000 cycles without functional or mechanical 10mm (medial-lateral dimension) wide in-line fusion device
failure.
assembly, distance OB = (4mm / 10mm)*(0.1)*(180°⁄π)=
2.3°).
3.2.26 offset displacement, n—distance OB in Fig. A1.3;
Stallmeyer, M. and Zoarski, G. H., “Sacroiliac Joint Injection,” in Image-
offset on the displacement axis equal to 2% of the intra-joint
Guided Spine Interventions, Johnson, B.A., Staats, P. S., Wetzel, F. T. and Matthis,
J. M., Eds., New York, Springer, 2004, pp. 234–244. space (for example, 0.08mm for the 4mm gap).
F3574 − 22
FIG. 4 Orthogonal Coordinate System for In-Line SIJ Fusion Implant Showing Possible Placement Within the SI Joint
(lateral perspective)
NOTE 1—Sacroiliac joint fusion screws are typically implanted in pairs or in sets of three.
FIG. 5 Orthogonal Coordinate System for Transverse SIJ Fusion Implant Showing Possible Placement Within the SI Joint
(the example shown has a lateral approach, but the surgical approach for transverse implants may be lateral or posterior)
3.2.27 permanent deformation, n—the remaining displace- 3.2.29 sacroiliac joint (SIJ) fusion device, n—a structure
ment(mmordegreesorradians)relativetotheinitialunloaded (biologicorsynthetic)thatisplacedinoracrossthejointspace
condition of the SIJ fusion device assembly after the applied to provide support for eventual arthrodesis of the joint.
force has been removed.
3.2.30 stiffness (N/mm or N*mm/degree (radian)), n—slope
3.2.28 pilot hole, n—the hole drilled into the bone (or test of Line Om in Fig.A1.3 or slope of Line OA in Fig.A2.5; the
block) into which a transverse sacroiliac joint fusion implant slope of the initial linear portion of the force-displacement
tip is inserted. The pilot hole is normally slightly larger than curve or the slope of the initial linear portion of the moment-
implant’s core diameter. angular displacement curve.
F3574 − 22
3.2.31 test block, n—thecomponentofthetestapparatusfor 4.2 These test methods are designed to quantify the static
mounting the SIJ fusion device assembly for the intended test and dynamic characteristics of different designs of SIJ fusion
configuration. device assemblies. These tests are conducted in vitro to allow
for analysis and comparison of the mechanical performance of
3.2.32 torsional yield strength (N-m), n—the point at which
SIJ fusion device assemblies to specific force modalities.
the transverse sacroiliac joint fusion implant reaches its pro-
4.3 The forces applied to the SIJ fusion device assemblies
portionallimitwhentestedinaccordancewithAnnexA2.This
during the tests described herein may differ from the complex
willbedeterminedbytheoffsetmethod.A2°offsetvalueshall
loading seen in vivo and, therefore, the results from these tests
be used.
may not directly predict in vivo performance. The results,
3.2.33 transverse implant, n—a device intended to be im-
however, can be used to compare mechanical performance of
planted with a trajectory across the sacroiliac joint by being
different SIJ fusion device assemblies.
implanted first into the iliac or sacral bone, subsequently
4.4 Since the environment may affect the dynamic perfor-
through the joint space, and finally into the opposite (sacral or
mance of SIJ fusion device assemblies, dynamic testing in a
iliac bone) (Fig. 5); this type of device may be implanted from
saline environment may be considered for implants with
a lateral approach or a posterior approach, but will be inserted
wearing surfaces or with movable components or for implants
through one of the articulating bones and extend into the other
with components that are temperature dependent. Fatigue tests
articulating bone. A screw design is common for transverse
should first be conducted in air (at ambient temperature) for
implants. Note: sacroiliac joint fusion screws are typically
comparison purposes since the environmental effects could be
implanted in pairs or in sets of three.
significant.Ifasimulatedinvivoenvironmentisnecessary,the
3.2.34 ultimate displacement (mm or degrees or radians),
investigator should consider testing in a saline environmental
n—displacement OF in Fig.A1.3; the displacement associated
bath at 37°C (for example, 0.9g NaCl per 100mL water). A
with the ultimate force or ultimate moment.
simulated body fluid, a saline drip or mist, distilled water, or
other type of lubrication at 37°C could also be used with
3.2.35 ultimate force or moment (N or N*mm), n—point Ein
adequate justification.
Fig. A1.3; the maximum applied force, F, transmitted by the
test frame actuator or the applied moment, M, that can be
4.5 If the devices are known to be temperature and envi-
applied during testing of a SIJ fusion device assembly. ronmentdependent,testingshouldbeconductedinphysiologic
solution as described in 4.4. Devices that require physiologic
3.2.36 yield displacement, n—distance OA in Fig.A1.3; the
solution for testing should be tested in the same type of
displacement (mm) or angular displacement (deg) when an SIJ
solution for comparison purposes.
fusion device assembly has a permanent deformation equal to
4.6 The location within the simulated joint space and
the offset displacement or the offset angular displacement.
position of the SIJ fusion device assembly with respect to the
3.2.37 yield force or moment (N or N*mm), n—point C in
loading axis will be dependent upon the design, the manufac-
Fig. A1.3 or Point b in Fig. A2.5; the applied force, F,
turer’s recommendation, or the surgeon’s preferred method for
transmitted by actuator, or the applied moment, M, required to
implant placement.
produce a permanent deformation equal to the offset displace-
4.7 It is well known that failure of materials is dependent
ment or to the offset angular displacement.
upon stress, test frequency, surface treatments, and environ-
mental factors. Therefore, when determining the effect of
4. Significance and Use
changing one of these parameters (for example, frequency,
4.1 The function of the SIJ fusion device assembly is to
material, or environment), all others must be kept constant to
stabilizetheSIJtofacilitatearthrodesisofthemotionsegment.
facilitate interpretation of results.
This test method outlines materials and methods for the
5. Keywords
characterizationandevaluationofthemechanicalperformance
of different SIJ fusion device assemblies so that comparisons 5.1 dynamic test methods; sacroiliac joint (SIJ) fusion
can be made between different designs. device; SIJ implants; static test methods
F3574 − 22
ANNEXES
(Mandatory Information)
A1. TEST METHODS FOR IN-LINE SACROILIAC FUSION IMPLANTS
A1.1 Summary of Test Methods Note: testing data for devices assembled in test blocks might
not be comparable to testing data for devices assembled in a
A1.1.1 These test methods are proposed for the mechanical
potting medium.
testing of SIJ fusion device assemblies intended as posterior
trajectory for implantation within the joint space (in-line
A1.1.3 Static and fatigue testing of the SIJ fusion device
implants). The test method for SIJ fusion device assemblies assemblies will simulate a motion segment via a gap between
intended to cross the joint space (transverse implants) is
the sacral and iliac sides of the implant.
describedinAnnexA2.Thesetestsaredesignedtocharacterize
A1.1.4 The test fixtures holding the test blocks and implant
thestructuralintegrityofthedeviceandarenotintendedtotest
(orpottedspecimen)shallbemanufacturedfromstainlesssteel
the bone-implant interface.
or other suitably rigid material.
A1.1.2 The implant is to be positioned in the test setup
A1.1.5 Static and dynamic tests will evaluate the SIJ fusion
within test blocks with device-matched pockets such that the
device assembly. The user of this test method must decide
sacral and iliac sides of the implant are rigidly held and such
which series of tests are applicable to the to the SIJ fusion
that there is an intra-joint space of 4mm to simulate the joint
device assembly in question. The user of this test method may
space (Figs. A1.1 and A1.2) which shall be held within test
choose to use all or a section of the tests described in this test
fixtures attached to the test frame table top and actuator.
method for testing a particular SIJ fusion device assembly.
Material selection for test blocks should be according to the
test block recommendations per Test Methods F2077.
A1.2 Significance and Use
A1.1.2.1 If stability cannot be maintained between the
A1.2.1 The function of the SIJ fusion device assembly is to
device and the test blocks during testing, the use of a potting
support the SIJ joint space to facilitate arthrodesis of the
medium in place of test blocks should be considered. Polym-
motion segment. These test methods outline materials and
ethyl methylcrylate (PMMA) is recommended as a potting
methods for the characterization and evaluation of the me-
medium; alternative potting materials such as metal-filled
chanical performance of different SIJ fusion device assemblies
epoxies may be considered if a stronger potting material is
so that comparisons in shear strength or torsional strength can
necessary. The potting medium should have a modulus of
be made between different designs.
elasticity between 2500MPa and 6000MPa. The potting
medium should have a minimum thickness of 5mm between A1.2.2 These test methods are designed to quantify the
the implant and the test fixtures on all sides of the implant. static and dynamic characteristics of different designs of SIJ
FIG. A1.1 Test Configuration for Shear
F3574 − 22
FIG. A1.2 Test Configuration for Torsion
FIG. A1.3 Typical Force-Displacement Curve
fusion device assemblies implanted within the joint space. simulated body fluid, a saline drip or mist, distilled water, or
These tests are conducted in vitro to allow for analysis and other type of lubrication at 37°C could also be used with
comparison of the mechanical performance of SIJ fusion
adequate justification.
device assemblies to specific force modalities.
A1.2.4.1 If the devices are known to be temperature and
A1.2.3 The forces applied to the in-line SIJ fusion device environment dependent, testing should be conducted in physi-
assemblies may differ from the complex loading seen in vivo ologic solution as described in A1.2.4. Devices that require
and, therefore, the results from these tests may not directly physiologic solution for testing should be tested in the same
predict in vivo performance.The results, however, can be used type of solution for comparison purposes.
to compare mechanical performance of different SIJ fusion
A1.2.5 The location within the simulated joint space and
device assemblies.
position of the SIJ fusion device assembly with respect to the
A1.2.4 Since the environment may affect the dynamic
loading axis will be dependent upon the design, the manufac-
performance of SIJ fusion device assemblies, dynamic testing
turer’s recommendation, or the surgeon’s preferred method for
in a saline environment may be considered for implants with
implant placement.
wearing surfaces or with movable components or for implants
A1.2.6 It is well known that failure of materials is depen-
with components that are temperature dependent. Fatigue tests
dent upon stress, test frequency, surface treatments, and envi-
should first be conducted in air (at ambient temperature) for
ronmental factors. Therefore, when determining the effect of
comparison purposes since the environmental effects could be
changing one of these parameters (for example, frequency,
significant.Ifasimulatedinvivoenvironmentisnecessary,the
investigator should consider testing in a saline environmental material, or environment), all others must be kept constant to
bath at 37°C (for example, 0.9g NaCl per 100mL water). A facilitate interpretation of results.
F3574 − 22
A1.3 Apparatus shall not be greater than 450N.Aregression analysis shall be
conducted on the force or moment versus number of cycles to
A1.3.1 Test machines will conform to the requirements of
failure data.
Practices E4.
A1.3.2 Shear Testing Apparatus—The shear testing appara- A1.5 Procedure for Static Tests
tuswillbeconfiguredsuchthatashearforcewillbeappliedto
A1.5.1 The SIJ fusion device is to be assembled in the test
the geometric center of the SIJ fusion device at 90° from the
setup with an intra-joint space of 4mm.
Z-axis in the YZ plane. The actuator of the testing machine
A1.5.2 The force, F, and moment, M , are to be applied as
z
may be connected to the upper test fixture by a universal joint,
describedinA1.3ofthismethodinpositioncontrolatarateno
rigid joint, or other suitable adapting joint. The bottom test
greater than 25 mm/min or 60°/min (radians/minute) until
fixture will be rigidly attached to the test frame table top. The
functional or mechanical failure of the SIJ fusion device
resultant tensile force, F, is a purely shear force. The user
assemblyisobtained.Thestatictorsionloadshallbeappliedin
shouldmonitorlateraltranslation(intheZ-direction)ofthetest
a clockwise direction.
fixtures during testing and make appropriate adjustments if
necessary.
A1.5.3 Aphysiologic compressive preload of 100N for SIJ
fusion device assemblies is required for the static torsion
NOTE A1.1—Pel et al., using a validated biomechanical musculoskel-
4,5,6
test. Other loads may be used with adequate justification.
etal pelvis model, showed how the SIJ reaction force is primarily shear at
approximately 81° from the direction normal to the joint space.The shear
NOTE A1.2—Pel et al., using a validated biomechanical musculoskel-
orientation has been set to 90° for ease of testing and repeatability.
etal model, showed that the SIJ reaction force is primarily shear at 81°
from the direction normal to the joint space, and suprasacral weight is
A1.3.3 Torsion Testing Apparatus—The torsion testing ap-
approximately 60% of the body weight per Snijders et al. At a body
paratus will be configured such that a moment will be applied
weight of 100 kg without considering the musculo-ligamentous
throughthegeometriccenteroftheSIJdeviceabouttheZ-axis.
interactions, the compressive component of the joint reaction force would
The torsion test apparatus (Fig. A1.2) will consist of the
be 92N (100 kg*9.81 N/kg*60%*sin(90°-81°)). With a biomechanical
implant held within test blocks (or fixed within a potting
musculoskeletal pelvis model, Grüneweller et al. estimated the medial-
lateral force across the sacroiliac joint to be up to approximately 100N
medium) which will be held in rigid test fixtures on the test
during normal gait.
frame so that the implant’s Z-axis is in line with the actuator.
Thebottomtestfixturewillbeheldrigidtothetestframetable A1.5.4 Theforce-displacementcurveshallberecorded.The
top, and the upper test fixture will be attached rigidly to the yield displacement (mm or degrees or radians), stiffness
actuator. (N/mm or N*mm/degree (radian)), yield force or moment (N
or N*mm), ultimate displacement (mm or degrees or radians),
A1.4 Sampling
and ultimate force or moment (N or N*mm) are to be
A1.4.1 All components in the SIJ fusion device assembly established. The user may reference Practices E2309/E2309M
for assistance in static test yield determination.
shall be previously unused parts only; no implants shall be
retested.
A1.6 Procedures for Dynamic Tests
A1.4.2 The test assemblies (that is, the SIJ fusion device
A1.6.1 TheSIJfusiondeviceassemblyistobeassembledin
assembled within test blocks) shall be labeled and shall be
the test setup with an intra-joint space of 4mm.
maintained according to good laboratory practice.
A1.4.2.1 Fordevicesusingapottingmediuminplaceoftest A1.6.2 Force, F, and moment, M , are to be selected as
z
blocks due to instability in dynamic testing, the test assembly describedinA1.4.Force, F,andmoment, M ,aretobeapplied
z
cannot be disassembled after potting. as described in A1.3 in load or moment control. A semi-log
fatigue graph of maximum applied force, F, or moment, M ,
z
A1.4.3 All static tests should have a minimum of six test
versus the number of cycles to failure shall be plotted. The
specimens.
endurance limit is to be determined.Acompressive preload of
A1.4.4 The user of these test methods should select the
100N for SIJ fusion device assemblies is required for the
necessary fatigue loads to establish the endurance limit of the
dynamictorsiontest.Theendofatestisdefinedasafunctional
SIJ fusion device assembly and develop a curve of cyclical
failure of the construct or attainment of 2500000 cycles
shear or torsional moment versus the number of cycles to
without functional failure. However, any mechanical failure
failure.Thiscurveshallbecomprisedofatleastsixdatapoints.
(for example, crack initiation or propagation) should be noted
Initial fatigue loading conditions may be determined primarily
at the 2500000 cycle point.
by the user’s experience. The precision of the endurance limit
shallbeestablishedbyensuringthatthelowestloadthatresults
in a failed construct is not greater than 1.5× the highest runout
Snijders,C.J.,Vleeming,A.,andStoeckart,R.,“TransferofLumbosacralLoad
load. For example, if the highest established runout load is
to Iliac Bones and Legs Part 1: Biomechanics of Self-Bracing of the Sacroiliac
300N, then the lowest load that results in a failed construct
Joints and Its Significance forTreatment and Exercise,” Clinical Biomechanics,Vol
8, No. 6, 1993, pp. 285–294.
Grüneweller, N., Raschke, M. J., Zderic, I., Widmer, D., Wahnert, D.,
Pel,J.J.M.,Spoor,C.W.,Pool-Goudzwaard,A.L.,HoekvanDijke,G.A.,and Guerguiev, B., Richards, R. G., Fuchs, T., and Windolf, M., “Biomechanical
Snijders,C.J.,“BiomechanicalAnalysisofReducingSacroiliacJointShearLoadby Comparison of Augmented Versus Non-Augment Sacroiliac Screws in a Novel
Optimization of Pelvic Muscle and Ligament Forces,” Annals of Biomedical Hemi-PelvisTestModel,” Journal of Orthopedic Research,Vol35,No.7,2017,pp.
Engineering, Vol 36, No. 3, 2008, pp. 415–424. 1485–1493.
F3574 − 22
A1.6.3 During dynamic tests, observations of any mechani- A1.7.3.1 Test Machine/Channel: List the manufacturer and
cal failures (for example, cracks) shall be documented with a model number of load frame. List type of control channel used
complete description of the mechanical failure, number of for testing (for example, force, torque), maximum capacity,
cycles at the initial observation, and subsequent changes, if date of most recent calibration, and calibrated range.
any, in mechanical behavior of the construct. It is recom-
A1.7.3.2 Test Fixtures: Provide descriptions of all fixtures
mendedthatimplantsshallbeexaminedformechanicalfailure
and connections including degrees of freedom. The following
atapproximatelyhalfwaythroughthedynamictests.However,
is a list of fixtures and connections that shall be described,
it is also recommended that the implant not be removed from
followed by what is called for in this standard.Any deviations
the test apparatus for examination of mechanical failure since
from what is recommended in this standard must be fully
test endpoints are determined by functional failure, and re-
described and justified.
moval and insertion of the implant in the test apparatus may
A1.7.3.3 Describe the connector between the test machine
affect implant performance (for example, failure to reinsert the
actuator and the upper fixture.
implant in the same position could either increase or decrease
A1.7.3.4 Describe the connection between the inferior fix-
the chances of crack propagation and potentially alter assess-
ture and the test machine base, which must be a rigid
ment of functional failure of the device). If a crack or other
attachment to the test machine base allowing zero degrees of
mechanical failure is found, the crack location and cycle count
freedom.
along with the size and description at which it was discovered
A1.7.3.5 Describe initial fixture alignment with the test
shall be recorded.At the engineering judgment of the user, the
specimen.Forsheartesting,theresultantforcemustbeapplied
test may be continued following the observation of a mechani-
through the geometric center of the test specimen. For torsion
cal failure to evaluate the ability of the implant to function
testing, the Z-axis of the test specimen must be co-linear with
under the applied forces. If a mechanical failure is detected
the axis of the test machine’s actuator and load cell.
following a 2500000 cycle runout, the failure shall be
A1.7.3.6 Test Blocks: Provide a description and an engi-
recorded (that is, location, size, and description) at the last
neering drawing of the test blocks. List the material of
cycle count at inspection without any detectable cracks. For
construction (for example, polyacetal for fatigue testing or
example, if an implant reached runout and a crack was
stainless steel for static testing). Describe the test block pocket
discovered on the implant upon removal, this crack shall be
geometry, including how closely it matches the geometry.
adequately described and noted and assigned the previous
Describe any fixation aids use in addition to the test block
examination cycle count before 2500000 cycles were at-
pocket (for example, epoxy) and provide a justification for
tained. Functionally, however, this implant would still be
their use.
considered a runout.
A1.7.3.6.1 Potting: Should a potting medium be used rather
A1.6.4 An R-value of 10 shall be used for the shear tests,
than test blocks, provide a description of the potting design or
and an R-value of –1 shall be used for the torsional testing.
molding for the potting blocks. List the material of the potting
medium (for example, PMMA (polymethyl methacrylate)).
A1.6.5 The frequency of the dynamic test shall be deter-
Describe any fixation aids used in addition to the potting and
mined by the user of this test method and recorded. Frequen-
provide a justification for their use.
ciesover10Hzmayresultinheatingandmeltingoftestblocks
A1.7.3.7 Test Environment: List the test medium,
(or potting medium). Since this phenomenon is device and
temperature, and any other environmental parameters that
environment specific, the user of these test methods is left to
might affect the results.
discern an appropriate cyclic frequency.
A1.7.3.8 TestSetupPhotographs:Provideanoverallpicture
A1.7 Report
of every test setup showing entire extent of fixturing. Provide
A1.7.1 The test report shall contain the following items. a close-up picture of the test specimen in fixtures (test medium
(for example, saline) may be omitted from the photo for image
Note that additional items may be necessary depending on the
specific device being tested. clarity, but this should be noted).
A1.7.4 Procedure:
A1.7.2 Test Samples—Thereportshallspecifythefollowing
regarding the SIJ fusion device(s) tested: manufacturer, trade
A1.7.4.1 Sampling: State the number of samples tested.
name,partnumber(s)andlotnumber(s),devicematerial(s)and
A1.7.4.2 Loading: For static tests, state the loading rate in
nominal dimensions (that is, width/diameter (medial-lateral
mm/min or degrees/min and the direction of loading. For
dimension), length (anterior-posterior dimension), height/
dynamic tests, state the cyclic loading frequency and the
diameter (cephalad-caudal dimension)). Additional features
) applied and
R-value. For torsion tests, state the axial load (F
z
such as integrated fixation screws or expansion features shall
the F actuator setting through testing (for example, load
z
be described. If more than one configuration is possible,
control or position control).
describe the configuration in which the device was tested (for
A1.7.4.3 Data Acquisition: State the data acquisition rate,
example, fully expanded). All other information necessary to
and for dynamic tests state if the acquisition time scale was
reproduce the assembly shall also be included. Provide repre-
linear or logarithmic.
sentative photographs of a test specimen showing, at a
A1.7.4.4 Parameters Calculated: State which parameters
minimum, three orthogonal views.
were calculated. Specifically report if alternative methods for
A1.7.3 Apparatus: calculation of yield load or torque and yield displacement or
F3574 − 22
angle were used and provide a justification and the expected A1.7.5.3 Semi-LogLoadversusNumberofCyclesPlot:For
impact on the calculated value. dynamic tests, provide a plot of load versus number of cycles.
Perform a regression analysis of the force or moment versus
A1.7.4.5 Deviations: Provide a detailed description of all
number of cycles. Runouts may be plotted, but should not be
procedure deviations, along with a description of why the
included in regression analysis.
deviation is acceptable and the effect the deviation is expected
A1.7.5.4 Failure Modes: Provide a complete description of
to have on the results of the test.
all failures (functional and mechanical), modes of failure, and
A1.7.5 Results: deformation of the SIJ fusion assembly or test apparatus.
Fatigue failures should be described completely with the
A1.7.5.1 ResultsTable: For static tests, report the following
following information: failure or crack initiation site, propaga-
intabularformatforeachindividualspecimen,aswellasmean
tion zone, and ultimate failure zone.Any wear or loosening of
and standard deviation: stiffness (N/mm or N*m/degree), yield
the assembly shall be described. Provide high-resolution pho-
loadortorque(NorN*m),yielddisplacementorangle(mmor
tographsforeveryspecimenshowingfailuremodesandfatigue
degrees), ultimate load or torque (N or N*m), and ultimate
runouts. Ensure use of appropriate magnification(s) to depict
displacement (mm or degrees). For dynamic tests, report the
the failure mode.
following for each individual specimen: the maximum load
applied (N or N*m), the minimum load applied (N or N*m),
A1.8 Precision and Bias
number of cycles achieved, and failure mode (or runout with
A1.8.1 Precision—The repeatability and reproducibility of
no visible failure).
this test method will be provided when resources are available
A1.7.5.2 Load versus Displacement Plots: For every static
to conduct an interlaboratory study.
test, provide a load or torque versus displacement plot that
A1.8.2 Bias—No statement can be made as to the bias of
includes the following: stiffness line, offset line, yield load/
these test methods since no acceptable reference values are
displacement marker, and ultimate load/displacement marker.
available, nor can they be obtained because of the destructive
Provide an explanation of any discontinuities or unexpected
nature of the tests.
behaviorsthatoccurpriortothecalculatedyieldloadortorque.
A1.9 Keywords
Optionally, in addition to individual plots, you may also
provide a single load versus displacement plot with plots for
A1.9.1 dynamic test methods; sacroiliac joint (SIJ) fusion
every specimen overlaid. device; SIJ implants; static test methods
A2. TEST METHODS FOR TRANSVERSE SACROILIAC FUSION IMPLANTS
A2.1 Summary of Test Methods sothatcomparisonsintorsionalstrength,bendingstrength,and
pullout resistance can be made between different designs.
A2.1.1 These test methods are proposed for the mechanical
A2.2.2 These test methods are designed to quantify the
testing of SIJ fusion device assemblies intended for implanta-
static and dynamic characteristics of different designs of SIJ
tion across the joint space (transverse implants) such as
fusion device assemblies implanted across the joint space.
sacroiliac screws. The test method for SIJ fusion device
These tests are conducted in vitro to allow for analysis and
assemblies intended to be implanted with a trajectory into the
comparison of the mechanical performance of SIJ fusion
joint space (in-line implants) is described in AnnexA1. These
device assemblies to specific force modalities.
tests are designed to characterize the structural integrity of the
A2.2.3 The forces applied to the in-line SIJ fusion device
device.
assemblies may differ from the complex loading seen in vivo
A2.1.2 Static and dynamic tests will evaluate the SIJ fusion
and, therefore, the results from these tests may not directly
device assembly. The user of these test methods must decide
predict in vivo performance.The results, however, can be used
which series of tests are applicable to the to the SIJ fusion
to compare mechanical performance of different SIJ fusion
device assembly in question. The user of these test methods
device assemblies.
maychoosetouseallorasectionofthetestsdescribedinthese
A2.2.4 Since the environment may affect the dynamic
testmethodsfortestingaparticularSIJfusiondeviceassembly.
performance of SIJ fusion device assemblies, dynamic testing
in a saline environment may be considered. Fatigue tests
A2.2 Significance and Use
should first be conducted in air (at ambient temperature) for
A2.2.1 The function of the SIJ fusion device assembly is to
comparison purposes since the environmental effects could be
support the SIJ joint space to facilitate arthrodesis of the
significant. If a simulated in vivo environment is desired, the
motion segment. These test methods outline materials and
investigator should consider testing in a saline environmental
methods for the characterization and evaluation of the me-
bath at 37°C (for example, 0.9g NaCl per 100mL water). A
chanical performance of different SIJ fusion device assemblies simulated body fluid, a saline drip or mist, distilled water, or
F3574 − 22
other type of lubrication at 37°C could also be used with A2.3.3 Pullout Testing Apparatus—The pullout testing ap-
adequate justification. paratus that is to be used for applying the required force to
A2.2.4.1 If the devices are known to be temperature and disengage the specimen from the test block shall be calibrated
environment dependent, testing should be conducted in physi- for the range of force levels and displacements used in the
ologic solution as described in A2.2.4. Devices that require determination of implant pullout resistance. A suitable testing
physiologic solution for testing should be tested in the same fixture to measure pullout resistance is shown in Fig. A2.2.
type of solution for comparison purposes. This fixture shall incorporate a Grade 20 lb/ft polyurethane
foam test block and clamp.
A2.2.5 The location within the simulated joint space and
position of the SIJ fusion device assembly with respect to the
NOTE A2.1—Zheng et al. measured the densities of the S1 body and
3 3
sacralalatobeapproximately23.8lb⁄ft and18.5lb⁄ft ;perSpecification
loading axis will be dependent upon the design, the manufac-
F1839, 20lb⁄ft polyurethane foam will be used.
turer’s recommendation, or the surgeon’s preferred method for
implant placement. A2.3.3.1 In addition to these requirements, the test block
clampshouldbesufficientlyrigidsuchthatdeflectionunderthe
A2.2.6 It is well known that failure of materials is depen-
required loading conditions is negligible.The test block clamp
dent upon stress, test frequency, surface treatments, and envi-
should have a minimum grip span of five times the major
ronmental factors. Therefore, when determining the effect of
diameter of the implant with the implant centered between the
changing one of these parameters (for example, frequency,
grips. The grip span should be consistent throughout testing.
material, or environment), all others must be kept constant to
The top and bottom test block surfaces shall be flat, smooth,
facilitate interpretation of results.
and parallel (within 60.4mm) as required to ensure that the
test block will be supported in the fixture with the top surface
A2.3 Apparatus
at an angle of 90° to the centerline of the test specimen. The
A2.3.1 Test machines will conform to the requirements of
edges of the test block shall be of such contour or squareness
Practices E4.
as required to ensure that the test block clamp shall hold the
A2.3.2 Cantilever Bending Testing Apparatus—Thecantile-
test block free of relative motion without deformation of the
verbendingtestingapparatusthatistobeusedforapplyingthe
requiredcantileverbendtothespecimenshallbecalibratedfor
the range of force levels and displacements used in the 7
Zheng,Y., Lu, W., Zhu, Q., Qin, L., Zhong, S., and Leong, J. C., “Variation in
determination. A suitable testing fixture for measuring the
BoneMineralDensityoftheSacruminYoungAdultsandItsSignificanceforSacral
Fixation,” Spine, Vol 25, No. 3, 2000, pp. 353–357.
bending strength (yield moment) and dynamic runout moment
to 2500000 cycles is illustrated in Fig.A2.1.Any test fixture
implemented shall be sufficiently rigid so that its deformation
under maximum load is less than 1% of the test specimen’s
deformation.Thedistaltipofthedeviceshouldberigidlyfixed
to the test frame table top or anchoring fixture with clearance
for bending deformation of the implant’s proximal end. The
applied bending force is transmitted from the test frame
actuator to the proximal end of the implant via rigid point
loader.
FIG. A2.1 Test Apparatus for Determination of Static and
Dynamic Cantilever Bending Strength FIG. A2.2 Test Apparatus
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...