ASTM F2028-17
(Test Method)Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation
Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation
SIGNIFICANCE AND USE
5.1 This test method is intended to investigate the resistance of a glenoid component to loosening. Glenoid loosening is the most common clinical complication in total shoulder arthroplasty (see X1.1). The method assumes that loosening occurs because of edge loading, often called the rocking-horse phenomenon.
5.2 This test method can be used both to detect potential problems and to compare design features. Factors affecting loosening performance include articular geometry, flange geometry, materials, fixation design, bone quality, and surgical technique.
SCOPE
1.1 These test methods measure how much a prosthetic anatomic glenoid component rocks or pivots following cyclic displacement of the humeral head to opposing glenoid rims (for example, superior-inferior or anterior-posterior). Motion is quantified by the tensile displacement opposite each loaded rim after dynamic rocking. Similarly, these test methods measure how much a prosthetic reverse glenoid component rocks or pivots following cyclic articulation with a mating humeral liner. Motion is quantified by the magnitude of displacement measured before and after cyclic loading.
1.2 The same setup can be used to test the locking mechanisms of modular glenoid components, for example, disassociation of both anatomic and reverse shoulder components.
1.3 These test methods cover shoulder replacement designs with monolithic or modular glenoid components for cemented fixation as well as reverse glenoid components for uncemented fixation.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Replaces
ASTM F2028-14 - Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation - Effective Date
- 01-Dec-2017
- Effective Date
- 15-Dec-2018
- Effective Date
- 01-Dec-2017
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-Jan-2012
- Effective Date
- 15-Dec-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 15-Nov-2008
- Effective Date
- 15-Nov-2008
- Effective Date
- 15-Nov-2008
Overview
ASTM F2028-17: Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation is a critical international standard developed by ASTM International. It specifies test methods designed to assess the resistance of shoulder prosthesis glenoid components to loosening or module disassociation under dynamic, cyclic loading conditions. Glenoid loosening is acknowledged as the most common complication in total shoulder arthroplasty, and this standard is essential for evaluating implant designs, materials, and fixation techniques.
Key Topics
Dynamic Resistance Testing
The standard addresses how to simulate real-world loading of glenoid components by subjecting them to repeated stress in multiple directions, replicating the forces experienced following total shoulder replacement.Edge Loading and the Rocking-Horse Phenomenon
The main mechanism of loosening is assumed to be edge loading, often referred to as the “rocking-horse phenomenon.”Test Methods for Multiple Implant Types:
- Anatomic glenoid components (monolithic and modular, typically cemented)
- Reverse glenoid components (primarily uncemented, utilizing baseplates and glenospheres)
- Locking mechanisms of modular glenoid components
Quantification of Movement
Motion is measured by comparing displacements before and after cyclic loading, both at loaded rim positions and opposite, to assess tensile displacement and potential loosening.Influencing Factors Analysed
The protocol allows for comparison based on variables such as:- Articular and flange geometry
- Material selection
- Fixation methods
- Bone quality of the substitute material
- Surgical implantation technique
Applications
ASTM F2028-17 is widely used in the research and development context for shoulder arthroplasty device manufacturers, biomechanical testing labs, regulatory submissions, and academic research. Its principal applications include:
Preclinical Testing of Glenoid Implants
Manufacturers employ this standard to verify the stability and performance of new designs, cemented or uncemented fixation systems, and to optimize implant geometry and materials.Comparative Design Evaluation
The ability to compare different implant designs and materials under controlled, repeatable loading helps inform product development and regulatory approval processes.Assessing Fixation Integrity
The test establishes a reproducible method for quantifying the ability of a glenoid component to resist loosening due to cyclic load, as well as to assess the locking strength of modular mechanisms.Regulatory and Clinical Relevance
Results generated according to ASTM F2028-17 provide critical evidence for demonstrating compliance with international safety and efficacy requirements for orthopedic implants.
Related Standards
ASTM E4 – Practices for Force Verification of Testing Machines
Ensures mechanical or hydraulic testing equipment meets the necessary force specifications.ASTM F1378 – Specification for Shoulder Prostheses
Provides minimum requirements for shoulder prosthesis products and their components.ASTM F1839 – Specification for Rigid Polyurethane Foam
Details requirements for bone substitute materials used in the dynamic test procedures outlined in F2028-17.
Practical Value
Applying ASTM F2028-17 helps ensure high-quality, reliable, and safe shoulder arthroplasty components by providing a standard approach for dynamic evaluation of glenoid loosening or disassociation. It supports innovation in implant design, improves patient outcomes, and meets the expectations of regulatory agencies worldwide.
Keywords: arthroplasty, glenoid loosening, disassociation, total shoulder replacement, shoulder prosthesis, implant testing, ASTM F2028-17, cyclic loading, orthopedic implant standards.
Buy Documents
ASTM F2028-17 - Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation
REDLINE ASTM F2028-17 - Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2028-17 is a standard published by ASTM International. Its full title is "Standard Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is intended to investigate the resistance of a glenoid component to loosening. Glenoid loosening is the most common clinical complication in total shoulder arthroplasty (see X1.1). The method assumes that loosening occurs because of edge loading, often called the rocking-horse phenomenon. 5.2 This test method can be used both to detect potential problems and to compare design features. Factors affecting loosening performance include articular geometry, flange geometry, materials, fixation design, bone quality, and surgical technique. SCOPE 1.1 These test methods measure how much a prosthetic anatomic glenoid component rocks or pivots following cyclic displacement of the humeral head to opposing glenoid rims (for example, superior-inferior or anterior-posterior). Motion is quantified by the tensile displacement opposite each loaded rim after dynamic rocking. Similarly, these test methods measure how much a prosthetic reverse glenoid component rocks or pivots following cyclic articulation with a mating humeral liner. Motion is quantified by the magnitude of displacement measured before and after cyclic loading. 1.2 The same setup can be used to test the locking mechanisms of modular glenoid components, for example, disassociation of both anatomic and reverse shoulder components. 1.3 These test methods cover shoulder replacement designs with monolithic or modular glenoid components for cemented fixation as well as reverse glenoid components for uncemented fixation. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is intended to investigate the resistance of a glenoid component to loosening. Glenoid loosening is the most common clinical complication in total shoulder arthroplasty (see X1.1). The method assumes that loosening occurs because of edge loading, often called the rocking-horse phenomenon. 5.2 This test method can be used both to detect potential problems and to compare design features. Factors affecting loosening performance include articular geometry, flange geometry, materials, fixation design, bone quality, and surgical technique. SCOPE 1.1 These test methods measure how much a prosthetic anatomic glenoid component rocks or pivots following cyclic displacement of the humeral head to opposing glenoid rims (for example, superior-inferior or anterior-posterior). Motion is quantified by the tensile displacement opposite each loaded rim after dynamic rocking. Similarly, these test methods measure how much a prosthetic reverse glenoid component rocks or pivots following cyclic articulation with a mating humeral liner. Motion is quantified by the magnitude of displacement measured before and after cyclic loading. 1.2 The same setup can be used to test the locking mechanisms of modular glenoid components, for example, disassociation of both anatomic and reverse shoulder components. 1.3 These test methods cover shoulder replacement designs with monolithic or modular glenoid components for cemented fixation as well as reverse glenoid components for uncemented fixation. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2028-17 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2028-17 has the following relationships with other standards: It is inter standard links to ASTM F2028-14, ASTM F1378-18, ASTM F1378-17, ASTM E4-14, ASTM F1839-08(2012)e1, ASTM F1839-08(2012), ASTM F1378-12, ASTM F1378-05(2010), ASTM E4-10, ASTM E4-09a, ASTM E4-09, ASTM E4-08, ASTM F1839-08e2, ASTM F1839-08, ASTM F1839-08e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2028-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2028 − 17
Standard Test Methods for
Dynamic Evaluation of Glenoid Loosening or
Disassociation
This standard is issued under the fixed designation F2028; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 These test methods measure how much a prosthetic
E4 Practices for Force Verification of Testing Machines
anatomic glenoid component rocks or pivots following cyclic
F1378 Specification for Shoulder Prostheses
displacementofthehumeralheadtoopposingglenoidrims(for
F1839 Specification for Rigid Polyurethane Foam for Use as
example, superior-inferior or anterior-posterior). Motion is
a Standard Material for Testing Orthopaedic Devices and
quantifiedbythetensiledisplacementoppositeeachloadedrim
Instruments
after dynamic rocking. Similarly, these test methods measure
how much a prosthetic reverse glenoid component rocks or
3. Terminology
pivots following cyclic articulation with a mating humeral
3.1 Anatomic Total Shoulder Replacement (TSR) Definitions
liner. Motion is quantified by the magnitude of displacement
3.1.1 anatomic total shoulder arthroplasty system,
measured before and after cyclic loading.
n—shoulder implant system that has a concave glenoid com-
1.2 The same setup can be used to test the locking mecha- ponent and a convex humeral component design.
nisms of modular glenoid components, for example, disasso-
3.1.2 anatomic glenoid component, n—the concave pros-
ciation of both anatomic and reverse shoulder components.
thetic portion that replaces, in part or in total, the glenoid fossa
of the scapula and articulates with the natural humeral head or
1.3 These test methods cover shoulder replacement designs
a prosthetic replacement.
with monolithic or modular glenoid components for cemented
3.1.3 glenoid backing, n—the metallic or composite mate-
fixation as well as reverse glenoid components for uncemented
rial prosthetic portion of a multi-piece anatomic glenoid
fixation.
component that attaches to the scapula.
1.4 The values stated in SI units are to be regarded as
3.1.4 glenoid liner, n—the polymeric prosthetic portion of a
standard. No other units of measurement are included in this
multiple-piece anatomic glenoid component that articulates
standard.
with the humeral head.
1.5 This standard does not purport to address all of the
3.2 Reverse TSR Definitions
safety concerns, if any, associated with its use. It is the
3.2.1 reverse total shoulder arthroplasty system,
responsibility of the user of this standard to establish appro-
n—shoulder implant system that has a convex glenoid compo-
priate safety and health practices and determine the applica-
nent and a concave humeral component design.
bility of regulatory limitations prior to use.
3.2.2 reverse glenoid component, n—the convex prosthetic
1.6 This international standard was developed in accor-
portion that replaces the glenoid fossa of the scapula and
dance with internationally recognized principles on standard-
articulates with a concave prosthetic replacement of the hu-
ization established in the Decision on Principles for the
meral head in reverse total shoulder arthroplasty applications.
Development of International Standards, Guides and Recom-
The reverse glenoid may consist of one or more components
mendations issued by the World Trade Organization Technical
from one or more materials; most commonly, the reverse
Barriers to Trade (TBT) Committee.
glenoid is composed of a metal glenosphere that is modularly
connected to a metal glenoid baseplate which is fixed to the
glenoid fossa.
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of
Subcommittee F04.22 on Arthroplasty. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2017. Published January 2018. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2000. Last previous edition approved in 2014 as F2028 – 14. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F2028-17. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2028 − 17
3.2.3 glenoid baseplate, n—the nonarticular portion of the the back (medial) surface. For a reverse shoulder it is defined
reverse glenoid component that modularly connects to the as the plane created by the face of the glenoid baseplate (see
glenosphere and is commonly fixed to the glenoid fossa of the Fig. 2).
scapula using bone screws without the use of cement.
3.3.4.1 Discussion—Although the glenoid fossa is not truly
3.2.4 glenosphere, n—the convex prosthetic articular por-
a planar structure, the terms plane of the glenoid and glenoid
tion of the reverse glenoid component that articulates with the
planehavebothbeenusedinthescientificliteraturetodescribe
concave prosthetic replacement of the proximal humerus or
the anatomic orientation of the glenoid.
humeral head (for example, the humeral liner).
3.3.5 humeral head, n—the bearing member that articulates
with the glenoid.
3.2.5 glenosphere thickness, n—the height of the truncated
section of the sphere which composes the glenosphere. Note
3.3.6 humeral component, n—the prosthetic portion that
thatthedifferencebetweentheglenospherearticularradiusand
replaces, in part or in total, the proximal humerus or humeral
thickness defines the medial/lateral position of the glenoid
head and articulates with the natural glenoid fossa or a
center of rotation (see Fig. 1).The glenosphere thickness could
prosthetic replacement.
also be affected by the geometric relationship between the
3.3.7 humeral stem, n—segment intended for insertion
glenosphere and the glenoid baseplate.
within the humeral medullary canal.
3.2.6 humeral liner, n—theconcaveprostheticportionofthe
3.4 Definitions of Terms Specific to This Standard:
reversehumeralcomponentthatreplacestheproximalhumerus
3.4.1 anterior/posterior (AP), n—the widest dimension of
or humeral head and articulates with the convex prosthetic
the glenoid component that is perpendicular to the SI axis (see
replacement of the glenoid (for example, the glenosphere).
Fig. 3 and Fig. 4).
3.3 Definitions Common to Anatomic and Reverse TSRs
3.4.2 axial force; axial translation, n—the force and
3.3.1 collar, n—flange at the junction of the humeral neck
displacement, respectively, perpendicular to the glenoid plane.
and stem.
The axial force simulates the net compressive external and
3.3.2 keel, (or pegs), n—single or multiple projections that
active and passive soft tissue forces (see Fig. 2).
provide resistance to translation or rotation of the glenoid
3.4.3 edge displacement, n—the translation, perpendicular
component, or both, by mating with cavities created in the
to the glenoid plane, of a specific point on the outside edge of
glenoid fossa.
the glenoid, when subjected to loading (see Fig. 5, Fig. 6 and
3.3.3 neck—segment connecting the head and the stem. Fig. 7).
3.3.4 glenoid plane, n—in symmetrical anatomic glenoids, 3.4.4 runout, n—apredeterminednumberofcyclesatwhich
the glenoid plane is defined by joining the two articular edges; the testing on a particular specimen will be stopped, and no
in planar and asymmetric anatomic glenoids, it is defined by further testing on that specimen will be performed.
FIG. 1 Glenosphere Thickness
F2028 − 17
FIG. 2 Glenoid Plane and Force Directions
FIG. 3 Anatomic Glenoid Axes and Origin
FIG. 4 Reverse Glenoid Baseplate Axes
3.4.5 shear force; shear translation, n—the force and ANATOMIC SHOULDER GLENOID LOOSENING
displacement, respectively, parallel to the glenoid plane, TEST METHOD
applied, for example, in the superior/inferior or anterior/
posterior direction. The shear force simulates the net external
4. Summary of Test Method
shear and active and passive soft tissue forces (see Fig. 2).
4.1 The prosthetic glenoid component is fixed with bone
3.4.6 subluxation force, n—thepeakshearforcerequiredfor
cement into a bone substitute using the normal surgical
subluxation (for example, the peak resistive force at the
technique.
glenoidarticularrimopposingmovementofthehumeralhead).
4.2 The subluxation translation is determined experimen-
3.4.7 subluxation translation, n—the distance from the gle-
tally on additional components. This is accomplished using a
noid origin (see Fig. 3), parallel to the glenoid plane, to the
biaxial apparatus (see Fig. 5) by applying an axial load
point at which the subluxation load occurs.
perpendicular to the glenoid, then translating the humeral head
3.4.8 superior/inferior (SI), n—the longest dimension of the parallel to the glenoid plane until encountering a peak shear
glenoid component (see Fig. 3 and Fig. 4). load.This is performed in both directions, corresponding to the
F2028 − 17
FIG. 5 Biaxial Testing Apparatus for Anatomic Shoulders
FIG. 6 Displacement Test Configuration
direction of intended rocking (for example, superior-inferior, subluxation load would be acceptable, but is not practical
anterior-posterior, or an alternative angle).
because of the large displacements, quick speeds, and deform-
able polyethylene).
4.3 The edge displacements of the glenoid are measured
before cycling: a given axial load is first applied perpendicular
4.4 The humeral head is cycled to 90 % of the subluxation
to the glenoid, then the edge displacements are measured with
distance for a fixed number of cycles.
the humeral head in three positions: at the glenoid origin, and
positioned to 90 % of the subluxation translation (see X1.2), in 4.5 Theedgedisplacements(4.3)areeitherrepeatedfollow-
both directions, as defined in 4.2. (Cycling to 90 % of the ing the cycling or measured continuously during the cycling.
F2028 − 17
FIG. 7 Alternative Displacement Test Configuration
5. Significance and Use 7. Sampling and Test Specimens
5.1 This test method is intended to investigate the resistance 7.1 Aminimum of three samples shall be tested.Additional
of a glenoid component to loosening. Glenoid loosening is the
samples may be used to reflect test variability. At least two
most common clinical complication in total shoulder arthro- additional components should be used to determine the sub-
plasty (see X1.1). The method assumes that loosening occurs
luxation translation. The test may be conducted along the
because of edge loading, often called the rocking-horse phe- superior-inferior axis, the anterior-posterior axis, or another
nomenon.
axis of interest to the user.
5.2 This test method can be used both to detect potential
7.2 All glenoid components shall be in the final manufac-
problems and to compare design features. Factors affecting
tured condition. All plastic components shall be sterilized
loosening performance include articular geometry, flange
according to the manufacturer-recommended specifications for
geometry, materials, fixation design, bone quality, and surgical
clinical use.
technique.
7.3 The humeral head shall include the identical radius or
radii and material as the actual implant. Other features of the
6. Apparatus and Equipment
humeral component such as the shaft may be omitted. The
6.1 Thetestapparatusshallbeconstructedsuchthatanaxial
same head may be used for all tests unless the surface becomes
force is applied perpendicular to the glenoid plane and a shear
damaged.
force is applied parallel to the glenoid plane (see Fig. 2). Fig.
7.4 Glenoid and humeral components are used in total
5 shows the axial force to be horizontal and the shear force to
shoulder arthroplasty and should conform to the criteria
be vertical; however, this arrangement may be reversed.
specified in Specification F1378.
6.2 A bone substitute representing the strength or glenoid
cancellous bone (see X1.5) shall be used. If a polyurethane
8. Procedure
foam is used, it shall conform to Specification F1839.
8.1 Thefollowingstepsarecommontoboththesubluxation
6.3 The glenoid and humeral head shall be enclosed in a
(4.2) and rocking (4.3 – 4.5) tests:
chamber with water heated to 37 6 2°C, at least for the
8.1.1 Secure the glenoid component in a bone substitute
dynamic portion of the test (see X1.6).Abuffer may be added,
with bone cement using the normal surgical procedure and
if the tester deems this necessary.
instrumentation. Do not perform tests until the cement has
6.4 A means to measure the axial load, shear load, shear
cured properly.
translation, and glenoid edge displacements is required. A
8.1.2 Position the path of the humeral head on the glenoid
means to measure the axial translation is desirable.
within 60.5 mm (sideways) of the desired path, for example,
6.5 The tests shall be performed on either mechanical or by using a dye to locate the contact point of the humeral head;
hydraulic load frames with adequate load capacity and shall a dye is unnecessary for congruent prostheses. Locate the
meet the criteria of Practices E4. center of the path (for the subluxation test, this need not be
F2028 − 17
exact; for the rocking test, the peak loads at each rim during 8.6 Repeat the glenoid edge displacement measurements
cycling should be within 610 % of each other for symmetrical (8.3) if measurements were not taken continuously.
designs).
8.7 Testing may be continued to a higher number of cycles
8.1.3 Perform the static measurements (subluxation and
if desired.
edge displacements) either in air at room temperature or in
water at 37°C. The cyclic testing shall be performed in 37°C
9. Report
water (see 6.3, X1.3, and X1.6).
9.1 The test report shall include the following:
8.1.4 Apply a given axial load to the glenoid, for example,
9.1.1 All details relevant to the particular implants tested
750 6 7.5 N (see X1.4).
including type, size, and lot number as well as the glenoid
8.2 Determinethesubluxationtranslationexperimentallyon radius, humeral head radius or radii, and the prosthesis
separate components (see X1.2): material.
9.1.2 Theaxisanddirectionoftesting(forexample,central-
8.2.1 After applying the axial load, displace the humeral
head at a constant rate to a given displacement, ensuring that a superior-inferior).
9.1.3 Subluxation Test—The subluxation load and transla-
peak load is achieved in both directions. A rate of 50 mm/min
is recommended to avoid polyethylene creep. tion for each specimen, as well as the axial load and displace-
ment rate. A chart plotting the load versus displacement with
8.2.2 Yielding is expected at the recommended load and
does not constitute a failure. The test shall be terminated if the the 90 and 100 % subluxation loads clearly marked should be
included.
insert of a modular glenoid disassociates.
9.1.4 Rocking Test—The axial load, cyclic displacement,
8.2.3 Record the axial load, subluxation load, and sublux-
maximum number of cycles, testing frequency, and cause of
ation translation.
test termination. Testing parameters that differ from those
8.3 Measure the edge displacements before rocking:
recommended shall be justified.
8.3.1 Create a foundation for measurements at both ends of
9.1.5 Displacement Test—The edge displacements before
the glenoid at a similar distance from the back surface of the
andfollowingcycling,highlightingthetensiledisplacementon
glenoid for all prostheses. One possibility is to insert 2-mm-
theunloadedsidefollowingrocking(forexample,thedisplace-
diameter screws into the outside edge at each end of the
ment opposite the loaded side minus the value with the head at
glenoid prosthesis, parallel to the articular surface (to avoid
the glenoid origin).
exiting either into the articular surface or into the bone
9.1.6 If the amplitude of the axial translation decreases
substitute).Flattenthescrewheadparalleltotheglenoidplane.
suddenly during the test (indicating a tilt of the glenoid and the
Alternative methods are acceptable (see X1.8).
probable onset of loosening), the number of cycles at which
8.3.2 Rest a displacement measuring device, for example, a
this occurred should be recorded.
linear variable differential transformer (LVDT), differential
variable reluctance transducer (DVRT), or dial gauge, on each
10. Precision and Bias
foundation to measure the displacements perpendicular to the
10.1 Precision—The precision of this test method was
glenoid plane (see X1.8). Continuous measurement is
established by an interlaboratory comparison among four
desirable, but measurement at the beginning and end of the
laboratories, with each laboratory testing three specimens. The
rocking is sufficient.
specimens tested were commercially available UHMWPE
8.3.3 Condition the prosthesis/bone substitute system, for
glenoid components and cobalt chrome humeral heads. The
example, for ten cycles at 0.25 Hz.
population mean micromotion before and after testing was 368
8.3.4 Measure the edge displacements with the humeral
6 330 µm and 496 6 275 µm, respectively. Each laboratory
head located at the glenoid origin (see Fig. 3 and Fig. 4).
utilized different methods for measuring the edge
8.3.5 Translate the humeral head parallel to the glenoid
displacements, and one laboratory performed the test using a
plane to 90 % of the subluxation translation determined previ-
lubricantatthecontactsurfaceinsteadofperformingthetestin
ously (8.2) in one direction. Measure both edge displacements.
solution (see X1.8).
8.3.6 Translate the humeral head to 90 % of the subluxation
10.1.1 Repeatability—For replicate results obtained by the
translation in the opposite direction and measure both edge
same laboratory on nominally identical test specimens, the
displacements.
repeatability standard deviation (s ) was 72.3 µm before testing
r
8.3.7 Repeat the three readings at least once to ensure
and 268.0 µm after testing. All laboratories were within the
repeatability.
critical k values for the before and after testing conditions.
8.4 Cyclically translate the humeral head to 90 % of the 10.1.2 Reproducibility—For replicate results obtained by
subluxation translation to cause a rocking motion of the
the same laboratory on nominally identical test specimens, the
glenoid at a given frequency (for example, 2 Hz as a result of reproducibility standard deviation (s ) was 335.9 µm before
R
the large translations, or up to a maximum of 6 Hz) to a
testingand359.4µmaftertesting.Onelaboratoryexceededthe
maximumnumberofcycles(forexample,100 000)(seeX1.7). critical h value for the before testing condition (h=1.50 versus
Maintain the axial load and specified displacement.
h =1.49).Alllaboratorieswerewithinthecritical hvaluesfor
crit
the after testing condition.
8.5 Terminate the test when either the maximum number of
cycles has been reached or a modular glenoid insert disasso- 10.2 The above round robin data represent initial efforts at
ciates. establishing a precision and bias statement for this test method
F2028 − 17
and have been published before documentation of full lab 15. Sampling and Test Specimens
participation was completed (4 out of 6). Additionally, some
15.1 A minimum of three samples shall be tested. Addi-
labs experienced difficulty with measurement of micromotion
tional samples may be used to reflect test variability. The test
resulting in test method variances. Further testing is warranted
maybeconductedalongthesuperior-inferioraxis,theanterior-
and a revised precision and bias statement incorporating
posterior axis, or another axis of interest to the user.The initial
participation by additional labs with reduced methodology
shear displacement or load should be set just below the
variances is intended for future publication.
subluxation displacement or load. Each test will result either in
afailureor,ifnodisassociationoccurswithinthesetnumberof
11. Keywords
cycles, a runout. The load should be progressively stepped
11.1 arthroplasty; glenoid; loosening; subluxation; total
down until at least one runout occurs.
shoulder replacement
15.2 All glenoid components shall be in the final manufac-
tured condition. All plastic components shall be sterilized
MODULAR DISASSOCIATION TEST METHOD according to the manufacturer-recommended specifications for
clinical use.
12. Summary of Test Method
15.3 The humeral head shall include the identical radius or
12.1 The prosthetic glenoid component is fixed into a bone
radii and material as the actual implant. Other features of the
substitute with bone cement using the normal surgical tech-
humeral component such as the shaft may be omitted. The
nique.
same head may be used for all tests unless the surface becomes
damaged.
12.2 The subluxation translation is determined experimen-
tally on the intended test samples or additional components.
15.4 Glenoid and humeral components are used in total
This is accomplished using a biaxial apparatus (see Fig. 5), by
shoulder arthroplasty and should conform to the criteria in
first applying an axial load perpendicular to the glenoid, then
Specification F1378.
translating the humeral head parallel to the glenoid plane until
encountering a peak shear load. This is performed in both
16. Procedure
directions, corresponding to the direction of intended rocking
16.1 The following steps are common to both the sublux-
(for example, superior-inferior, anterior-posterior, or an alter-
ation (12.2) and rocking (12.3) tests:
native angle).
16.1.1 Secure the glenoid component in a bone substitute
12.3 The humeral head is cycled to 90 % of the subluxation
with bone cement using the normal surgical procedure and
distance for a fixed number of cycles (see X1.2). (Cycling to
instrumentation. Do not perform tests until the cement has
90 % of the subluxation load would be acceptable, but is not
cured properly.
practical because of the large displacements, quick speeds, and
16.1.2 Position the path of the humeral head on the glenoid
deformable polyethylene).
within 60.5 mm (sideways) of the desired path, for example,
by using a dye to locate the contact point of the humeral head.
13. Significance and Use
A dye is unnecessary for congruent prostheses. Locate the
13.1 This test method is intended to investigate the locking
center of the path (for the subluxation test, this need not be
mechanismofamodularglenoid.Disassociationoftheinsertis
exact; for the rocking test, the peak loads at each run during
the greatest issue in modular glenoid components. This test
cycling should be within 610 % of each other for symmetrical
method can be used either to detect potential problems or to
designs).
compare design features.
16.1.3 Perform the measurements in 37°C water (see 14.2,
X1.3 and X1.6).
14. Apparatus and Equipment
16.1.4 Apply a given axial load to the glenoid, for example,
14.1 The test apparatus shall be constructed such that an
750 6 7.5 N (see X1.4).
axial force is applied perpendicular to the glenoid plane and a
16.2 Determine the subluxation translation experimentally
shear force is applied parallel to the glenoid plane (see Fig. 2).
on the intended test specimens or separate components (see
Fig.5showstheaxialforcetobehorizontalandtheshearforce
X1.2):
to be vertical; however, this arrangement may be reversed.
16.2.1 After applying the axial load, displace the humeral
14.2 The glenoid and humeral head shall be enclosed in a
head at a constant rate to a given displacement, ensuring that a
chamber with water heated to 37 6 2°C, at least for the
peak load is achieved in both directions. A rate of 50 mm/min
dynamic portion of the test (see X1.6).Abuffer may be added,
is recommended to avoid polyethylene creep.
if the tester deems this necessary.
16.2.2 Yielding is expected at the recommended load and
14.3 Ameanstomeasuretheaxialloadandsheartranslation
does not constitute a failure. The test shall be terminated if the
is required.
modular insert disassociates.
14.4 The tests shall be performed on either mechanical or 16.2.3 Record the axial load and subluxation translation.
hydraulic load frames with adequate load capacity and shall The subluxation load is not required for the rocking test, but
meet the criteria of Practices E4. may be of interest to characterize the prosthesis.
F2028 − 17
16.3 Cyclically translate the humeral head to 90 % of the 19. Keywords
subluxation translation to cause a rocking motion of the
19.1 arthroplasty; disassociation; glenoid; subluxation; total
glenoid at a given frequency (for example, 2 Hz as a result of
shoulder replacement
the large translations, or up to a maximum of 6 Hz) to a
maximum number of cycles (for example, 100 000 or higher,
REVERSE SHOULDER GLENOID LOOSENING/
see X1.7). Maintain the axial load and specified displacement.
DISASSOCIATION TEST METHOD
16.4 Terminate the test when either the maximum number
of cycles has been reached or the glenoid insert disassociates.
20. Summary of Test Method
The load should be set high enough to produce a failure, then
reduced to produce at least one runout. 20.1 The prosthetic reverse glenoid baseplate is fixed with
bone screws into a bone substitute using the normal surgical
16.5 Testing may be continued to a higher number of cycles
technique.
if desired.
20.2 The initial glenoid baseplate fixation to the bone
17. Report
substitute is measured before cyclic loading. Fixation can be
17.1 The test report shall include the following:
measured directly from the glenoid baseplate or with the
17.1.1 All details relevant to the particular implants tested
glenosphere assembled. Fixation is measured as an axial
including type, size, and lot number as well as the glenoid
compressive load is applied approximately through the center
radius, humeral head radius or radii, and the prosthesis
of rotation, perpendicular to the glenoid plane as a shear load
materials.
is applied parallel to the glenoid plane. The induced displace-
17.1.2 The axis and direction of testing (for example,
ment of the glenoid baseplate or glenoid baseplate/glenosphere
central-superior-inferior).
assembly in the directions of the shear and axial compressive
17.1.3 Subluxation Test—The subluxation load and transla-
loads should be measured. If the glenoid baseplate is noncir-
tion for each specimen, as well as the axial load and displace-
cular in shape (see Fig. 4), then the shear force should be
ment rate. A chart plotting the load versus displacement with
applied (and the associated displacements measured) along the
the 90 and 100 % subluxation loads clearly marked should be
device’s major and minor axes, typically in the superior/
included.
inferior and anterior/posterior directions (see X2.10).
17.1.4 Rocking Test—The axial load, cyclic displacement,
20.3 The glenosphere is connected to the glenoid baseplate
maximum number of cycles, testing frequency, and cause of
(if not already assembled), mated with the reverse humeral
test termination. Testing parameters that differ from those
component, and the assembly is secured to a biaxial apparatus.
recommended shall be justified.
20.4 Using the biaxial apparatus, the reverse glenoid com-
18. Precision and Bias
ponent is rotated about the humeral liner for a fixed number of
18.1 The precision and bias of this test method has not been cycles as an axial compressive load is applied through the
established. Test results that could be used to establish preci- humeral liner into the glenoid component (see Fig. 8 and
sion and bias are solicited. X2.6).
FIG. 8 Biaxial Testing Apparatus for Cyclic Test of Reverse Shoulders
F2028 − 17
20.5 The glenoid fixation is measured after cyclic loading 22. Apparatus and Equipment
according to the method described in 20.2.
22.1 The biaxial test apparatus shall be constructed such
20.6 As this cyclic test loads the reverse shoulder assembly
that an axial compressive load is applied approximately
in a physiologically relevant manner, the cyclic test is also
through the center of rotation as the glenoid component is
applicable to evaluate the resistance of a modular reverse
rotated about the humeral liner in the cyclic test (see Fig. 8).
shoulder design to disassociation or dislocation.
Fig.9depictstheaxialcompressiveloadbeingappliedthrough
thehumerallinerastheglenoidcomponentiscyclicallyrotated
21. Significance and Use
in the superior/inferior direction (see X2.6 and X2.7).
21.1 This test method is intended to investigate the resis-
22.2 Thetestapparatusshouldalsopermittheapplicationof
tance of a reverse shoulder glenoid baseplate to loosening,
a shear load approximately parallel to the glenoid plane as an
disassociation of modular components, and/or dislocation.
axial compressive load is applied perpendicular to the glenoid
Glenoid loosening is a common clinical complication of
planeinthedisplacementtest.Fig.6depictstheshearandaxial
reverse total shoulder arthroplasty. The method assumes that
compressive loads applied directly to the glenoid baseplate.
loosening occurs because of the cyclic loading of conforming
The point of application of loading should be chosen to
articular curvatures and not due to edge loading of noncon-
minimize the creation of a moment on the baseplate. Fig. 7
forming articular curvatures common to anatomic total shoul-
depicts an alternative method in which the shear and axial
der arthroplasty (see X2.2, X2.3, X2.6).
compressive loads are applied through the glenosphere/glenoid
21.2 This test method can be used to detect potential
baseplate assembly (see X2.10).
problems and compare design features. Factors affecting loos-
22.3 A bone substitute representing the strength of glenoid
ening performance include the type of screw (for example,
cancellous bone shall be used. If a polyurethane foam is used,
compression versus locking), screw length and diameter, screw
it shall conform to Specification F1839 (see X2.5).
angulation, screw positioning or configuration, glenoid base-
22.4 The cyclic loading of the reverse components can be
plate contact area, glenoid baseplate backside geometry (for
performed in air at room temperature; however, the post-cyclic
example, flat or curved), glenoid baseplate fixation post geom-
displacement measurements should be made only after the
etry (for example, cylinder, taper, or screw), the amount of the
tested components have cooled to room temperature following
glenoid baseplate pressfit, glenosphere thickness, glenosphere
cyclic loading (for example, 18 to 25°C). A fan or air jet may
diameter, glenoid component center of rotation (for example,
beusedtocoolthetestcomponentsduringcyclicloadingorthe
medialized, lateralized, or inferiorly shifted), articular
test may be performed in a lubricated environment, if the tester
geometry, materials, surface roughness, bone quality, and
deems this necessary.
surgical technique (see X2.4).
21.3 This test method is intended to investigate short-term 22.5 Ameanstomeasuretheaxialcompressiveloadandthe
fixation only and does not evaluate the contribution of biologi- angle of rotation during cyclic loading is required during the
cal fixation. cyclic test. A means to measure the axial compressive load,
FIG. 9 Cyclic Test Configuration
F2028 − 17
shear load, and the glenoid baseplate displacement (or 24.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2028 − 14 F2028 − 17
Standard Test Methods for
Dynamic Evaluation of Glenoid Loosening or
Disassociation
This standard is issued under the fixed designation F2028; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods measure how much a prosthetic anatomic glenoid component rocks or pivots following cyclic
displacement of the humeral head to opposing glenoid rims (for example, superior-inferior or anterior-posterior). Motion is
quantified by the tensile displacement opposite each loaded rim after dynamic rocking. Similarly, these test methods measure how
much a prosthetic reverse glenoid component rocks or pivots following cyclic articulation with a mating humeral liner. Motion is
quantified by the magnitude of displacement measured before and after cyclic loading.
1.2 The same setup can be used to test the locking mechanisms of modular glenoid components, for example, disassociation
of both anatomic and reverse shoulder components.
1.3 These test methods cover shoulder replacement designs with monolithic or modular glenoid components for cemented
fixation as well as reverse glenoid components for uncemented fixation.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Verification of Testing Machines
F1378 Specification for Shoulder Prostheses
F1839 Specification for Rigid Polyurethane Foam for Use as a Standard Material for Testing Orthopaedic Devices and
Instruments
3. Terminology
3.1 Anatomic Total Shoulder Replacement (TSR) Definitions
3.1.1 anatomic total shoulder arthroplasty system, n—shoulder implant system that has a concave glenoid component and a
convex humeral component design.
3.1.2 anatomic glenoid component, n—the concave prosthetic portion that replaces, in part or in total, the glenoid fossa of the
scapula and articulates with the natural humeral head or a prosthetic replacement.
3.1.3 glenoid backing, n—the metallic or composite material prosthetic portion of a multi-piece anatomic glenoid component
that attaches to the scapula.
3.1.4 glenoid liner, n—the polymeric prosthetic portion of a multiple-piece anatomic glenoid component that articulates with the
humeral head.
These test methods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and are the direct responsibility of Subcommittee
F04.22 on Arthroplasty.
Current edition approved March 1, 2014Dec. 1, 2017. Published July 2014January 2018. Originally approved in 2000. Last previous edition approved in 20122014 as
ε1
F2028 – 08F2028 – 14.(2012) . DOI: 10.1520/F2028-14.10.1520/F2028-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2028 − 17
3.2 Definitions:Reverse TSR Definitions
3.2.1 anatomicreverse total shoulder arthroplasty, arthroplasty system, n—shoulder implant system that has a concaveconvex
glenoid component and a convexconcave humeral component design.
3.1.1.1 anatomic glenoid, n—the concave prosthetic portion that replaces the glenoid fossa of the scapula and articulates with
a convex prosthetic replacement of the humeral head in anatomic total shoulder arthroplasty applications. It may consist of one
or more components from one or more materials, for example, either all-polyethylene or a metal baseplate with a polymeric insert.
3.1.1.2 humeral head, n—the convex prosthetic portion that replaces the proximal humerus or humeral head and articulates with
the natural glenoid fossa or an anatomic prosthetic replacement.
3.2.2 reverse total shoulder arthroplasty, glenoid component, n—shoulder implants that have a convex glenoid component and
a concave humeral component design.the convex prosthetic portion that replaces the glenoid fossa of the scapula and articulates
with a concave prosthetic replacement of the humeral head in reverse total shoulder arthroplasty applications. The reverse glenoid
may consist of one or more components from one or more materials; most commonly, the reverse glenoid is composed of a metal
glenosphere that is modularly connected to a metal glenoid baseplate which is fixed to the glenoid fossa.
3.1.2.1 glenoid baseplate, n—the nonarticular portion of the reverse glenoid component that modularly connects to the
glenosphere and is usually fixed to the glenoid fossa of the scapula using bone screws without the use of cement.
3.1.2.2 glenosphere, n—the convex prosthetic articular portion of the reverse glenoid component that articulates with the
concave prosthetic replacement of the proximal humerus or humeral head (for example, the humeral liner).
3.1.2.3 glenosphere thickness, n—the height of the truncated section of the sphere which composes the glenosphere. Note that
the difference between the glenosphere articular radius and thickness defines the medial/lateral position of the glenoid center of
rotation (see Fig. 1). The glenosphere thickness could also be affected by the geometric relation between the glenosphere and the
glenoid baseplate.
3.1.2.4 humeral liner, n—the concave prosthetic portion of the reverse humeral component that replaces the proximal humerus
or humeral head and articulates with the convex prosthetic replacement of the glenoid (for example, the glenosphere).
3.1.2.5 reverse glenoid, n—the convex prosthetic portion that replaces the glenoid fossa of the scapula and articulates with a
concave prosthetic replacement of the humeral head in reverse total shoulder arthroplasty applications. The reverse glenoid may
consist of one or more components from one or more materials; most commonly, the reverse glenoid is composed of a metal
glenosphere that is modularly connected to a metal glenoid baseplate which is fixed to the glenoid fossa.
3.1.3 anterior/posterior (AP), n—the AP axis is the widest dimension of the glenoid component (see Fig. 2 and Fig. 3).
3.2.3 axial load; axial translation, glenoid baseplate, n—the force and displacement, respectively, perpendicularnonarticular
portion of the reverse glenoid component that modularly connects to the glenosphere and is commonly fixed to the glenoid plane.
The axial load simulates the net compressive external and active and passive soft tissue forces (see fossa of the scapula using bone
screws without the use of cement.Fig. 4).
3.2.4 edge displacements, glenosphere, n—the translation, perpendicular to the glenoid plane, of a specific point on the outside
edge of the glenoid, when subjected to loading (seeconvex prosthetic articular portion of the reverse glenoid component that
articulates with the concave prosthetic replacement of the proximal humerus or humeral head (for Fig. 5,example, Fig. 6 andthe
humeral Fig. 7).liner).
3.1.6 glenoid plane (see X1.9),n—in symmetrical anatomic glenoids, the glenoid plane is defined by joining the two articular
edges; in planar and asymmetric anatomic glenoids, it is defined by the back (medial) surface. For a reverse shoulder it is defined
as the plane created by the face of the glenoid baseplate (see Fig. 4).
3.1.7 runout, n—a predetermined number of cycles at which the testing on a particular specimen will be stopped, and no further
testing on that specimen will be performed.
3.2.5 shear load; shear translation, glenosphere thickness, n—the force and displacement, respectively, parallel to the glenoid
plane, applied, for example, in the superior/inferior or anterior/posterior direction. The shear load simulates the net external shear
and active and passive soft tissue forcesheight of the truncated section of the sphere which composes the glenosphere. Note that
the difference between the glenosphere articular radius and thickness defines the medial/lateral position of the glenoid center of
rotation (see Fig. 41). The glenosphere thickness could also be affected by the geometric relationship between the glenosphere and
the glenoid baseplate.
3.1.9 subluxation load, n—the peak shear load required for subluxation (for example, the peak resistive force at the glenoid
articular rim opposing movement of the humeral head).
3.2.6 subluxation translation,humeral liner, n—the distance from the glenoid origin (seeconcave prosthetic portion of the
reverse humeral component Fig. 2), parallel to the glenoid plane, to the point at which the subluxation load occurs.that replaces
the proximal humerus or humeral head and articulates with the convex prosthetic replacement of the glenoid (for example, the
glenosphere).
3.1.11 superior/inferior (SI), n—the SI axis is the longest dimension of the glenoid component (see Fig. 2 and Fig. 3).
F2028 − 17
FIG. 1 Glenosphere Thickness
3.3 Definitions Common to Anatomic and Reverse TSRs
3.3.1 collar, n—flange at the junction of the humeral neck and stem.
3.3.2 keel, (or pegs), n—single or multiple projections that provide resistance to translation or rotation of the glenoid
component, or both, by mating with cavities created in the glenoid fossa.
3.3.3 neck—segment connecting the head and the stem.
3.3.4 glenoid plane, n—in symmetrical anatomic glenoids, the glenoid plane is defined by joining the two articular edges; in
planar and asymmetric anatomic glenoids, it is defined by the back (medial) surface. For a reverse shoulder it is defined as the plane
created by the face of the glenoid baseplate (see Fig. 2).
3.3.4.1 Discussion—
Although the glenoid fossa is not truly a planar structure, the terms plane of the glenoid and glenoid plane have both been used
in the scientific literature to describe the anatomic orientation of the glenoid.
FIG. 2 Anatomic Glenoid AxesPlane and OriginForce Directions
F2028 − 17
3.3.5 humeral head, n—the bearing member that articulates with the glenoid.
3.3.6 humeral component, n—the prosthetic portion that replaces, in part or in total, the proximal humerus or humeral head and
articulates with the natural glenoid fossa or a prosthetic replacement.
3.3.7 humeral stem, n—segment intended for insertion within the humeral medullary canal.
3.4 Definitions of Terms Specific to This Standard:
3.4.1 anterior/posterior (AP), n—the widest dimension of the glenoid component that is perpendicular to the SI axis (see Fig.
3 and Fig. 4).
3.4.2 axial force; axial translation, n—the force and displacement, respectively, perpendicular to the glenoid plane. The axial
force simulates the net compressive external and active and passive soft tissue forces (see Fig. 2).
3.4.3 edge displacement, n—the translation, perpendicular to the glenoid plane, of a specific point on the outside edge of the
glenoid, when subjected to loading (see Fig. 5, Fig. 6 and Fig. 7).
3.4.4 runout, n—a predetermined number of cycles at which the testing on a particular specimen will be stopped, and no further
testing on that specimen will be performed.
3.4.5 shear force; shear translation, n—the force and displacement, respectively, parallel to the glenoid plane, applied, for
example, in the superior/inferior or anterior/posterior direction. The shear force simulates the net external shear and active and
passive soft tissue forces (see Fig. 2).
3.4.6 subluxation force, n—the peak shear force required for subluxation (for example, the peak resistive force at the glenoid
articular rim opposing movement of the humeral head).
3.4.7 subluxation translation, n—the distance from the glenoid origin (see Fig. 3), parallel to the glenoid plane, to the point at
which the subluxation load occurs.
3.4.8 superior/inferior (SI), n—the longest dimension of the glenoid component (see Fig. 3 and Fig. 4).
ANATOMIC SHOULDER GLENOID LOOSENING TEST METHOD
4. Summary of Test Method
4.1 The prosthetic glenoid component is fixed with bone cement into a bone substitute using the normal surgical technique.
4.2 The subluxation translation is determined experimentally on additional components. This is accomplished using a biaxial
apparatus (see Fig. 5) by applying an axial load perpendicular to the glenoid, then translating the humeral head parallel to the
glenoid plane until encountering a peak shear load. This is performed in both directions, corresponding to the direction of intended
rocking (for example, superior-inferior, anterior-posterior, or an alternative angle).
4.3 The edge displacements of the glenoid are measured before cycling: a given axial load is first applied perpendicular to the
glenoid, then the edge displacements are measured with the humeral head in three positions: at the glenoid origin, and positioned
to 90 % of the subluxation translation (see X1.2), in both directions, as defined in 4.2. (Cycling to 90 % of the subluxation load
would be acceptable, but is not practical because of the large displacements, quick speeds, and deformable polyethylene).
4.4 The humeral head is cycled to 90 % of the subluxation distance for a fixed number of cycles.
4.5 The edge displacements (4.3) are either repeated following the cycling or measured continuously during the cycling.
5. Significance and Use
5.1 This test method is intended to investigate the resistance of a glenoid component to loosening. Glenoid loosening is the most
common clinical complication in total shoulder arthroplasty (see X1.1). The method assumes that loosening occurs because of edge
loading, often called the rocking-horse phenomenon.
FIG. 3 Reverse Glenoid Baseplate AxesAnatomic Glenoid Axes and Origin
F2028 − 17
FIG. 4 Glenoid Plane and Load DirectionsReverse Glenoid Baseplate Axes
FIG. 5 Biaxial Testing Apparatus for Anatomic Shoulders
FIG. 6 Displacement Test Configuration
5.2 This test method can be used both to detect potential problems and to compare design features. Factors affecting loosening
performance include articular geometry, flange geometry, materials, fixation design, bone quality, and surgical technique.
F2028 − 17
FIG. 7 Alternative Displacement Test Configuration
6. Apparatus and Equipment
6.1 The test apparatus shall be constructed such that an axial loadforce is applied perpendicular to the glenoid plane and a shear
loadforce is applied parallel to the glenoid plane (see Fig. 42). Fig. 5 shows the axial loadforce to be horizontal and the shear
loadforce to be vertical; however, this arrangement may be reversed.
6.2 A bone substitute representing the strength or glenoid cancellous bone (see X1.5) shall be used. If a polyurethane foam is
used, it shall conform to Specification F1839.
6.3 The glenoid and humeral head shall be enclosed in a chamber with water heated to 37 6 2°C, at least for the dynamic
portion of the test (see X1.6). A buffer may be added, if the tester deems this necessary.
6.4 A means to measure the axial load, shear load, shear translation, and glenoid edge displacements is required. A means to
measure the axial translation is desirable.
6.5 The tests shall be performed on either mechanical or hydraulic load frames with adequate load capacity and shall meet the
criteria of Practices E4.
7. Sampling and Test Specimens
7.1 A minimum of three samples shall be tested. Additional samples may be used to reflect test variability. At least two
additional components should be used to determine the subluxation translation. The test may be conducted along the
superior-inferior axis, the anterior-posterior axis, or another axis of interest to the user.
7.2 All glenoid components shall be in the final manufactured condition. All plastic components shall be sterilized according
to the manufacturer-recommended specifications for clinical use.
7.3 The humeral head shall include the identical radius or radii and material as the actual implant. Other features of the humeral
component such as the shaft may be omitted. The same head may be used for all tests unless the surface becomes damaged.
7.4 Glenoid and humeral components are used in total shoulder arthroplasty and should conform to the criteria specified in
Specification F1378.
8. Procedure
8.1 The following steps are common to both the subluxation (4.2) and rocking (4.3 – 4.5) tests:
8.1.1 Secure the glenoid component in a bone substitute with bone cement using the normal surgical procedure and
instrumentation. Do not perform tests until the cement has cured properly.
8.1.2 Position the path of the humeral head on the glenoid within 60.5 mm (sideways) of the desired path, for example, by using
a dye to locate the contact point of the humeral head; a dye is unnecessary for congruent prostheses. Locate the center of the path
F2028 − 17
(for the subluxation test, this need not be exact; for the rocking test, the peak loads at each rim during cycling should be within
610 % of each other for symmetrical designs).
8.1.3 Perform the static measurements (subluxation and edge displacements) either in air at room temperature or in water at
37°C. The cyclic testing shall be performed in 37°C water (see 6.3, X1.3, and X1.6).
8.1.4 Apply a given axial load to the glenoid, for example, 750 6 7.5 N (see X1.4).
8.2 Determine the subluxation translation experimentally on separate components (see X1.2):
8.2.1 After applying the axial load, displace the humeral head at a constant rate to a given displacement, ensuring that a peak
load is achieved in both directions. A rate of 50 mm/min is recommended to avoid polyethylene creep.
8.2.2 Yielding is expected at the recommended load and does not constitute a failure. The test shall be terminated if the insert
of a modular glenoid disassociates.
8.2.3 Record the axial load, subluxation load, and subluxation translation.
8.3 Measure the edge displacements before rocking:
8.3.1 Create a foundation for measurements at both ends of the glenoid at a similar distance from the back surface of the glenoid
for all prostheses. One possibility is to insert 2-mm-diameter screws into the outside edge at each end of the glenoid prosthesis,
parallel to the articular surface (to avoid exiting either into the articular surface or into the bone substitute). Flatten the screw head
parallel to the glenoid plane. Alternative methods are acceptable (see X1.8).
8.3.2 Rest a displacement measuring device, for example, a linear variable differential transformer (LVDT), differential variable
reluctance transducer (DVRT), or dial gauge, on each foundation to measure the displacements perpendicular to the glenoid plane
(see X1.8). Continuous measurement is desirable, but measurement at the beginning and end of the rocking is sufficient.
8.3.3 Condition the prosthesis/bone substitute system, for example, for ten cycles at 0.25 Hz.
8.3.4 Measure the edge displacements with the humeral head located at the glenoid origin (see Fig. 23 and Fig. 34).
8.3.5 Translate the humeral head parallel to the glenoid plane to 90 % of the subluxation translation determined previously (8.2)
in one direction. Measure both edge displacements.
8.3.6 Translate the humeral head to 90 % of the subluxation translation in the opposite direction and measure both edge
displacements.
8.3.7 Repeat the three readings at least once to ensure repeatability.
8.4 Cyclically translate the humeral head to 90 % of the subluxation translation to cause a rocking motion of the glenoid at a
given frequency (for example, 2 Hz as a result of the large translations, or up to a maximum of 6 Hz) to a maximum number of
cycles (for example, 100 000) (see X1.7). Maintain the axial load and specified displacement.
8.5 Terminate the test when either the maximum number of cycles has been reached or a modular glenoid insert disassociates.
8.6 Repeat the glenoid edge displacement measurements (8.3) if measurements were not taken continuously.
8.7 Testing may be continued to a higher number of cycles if desired.
9. Report
9.1 The test report shall include the following:
9.1.1 All details relevant to the particular implants tested including type, size, and lot number as well as the glenoid radius,
humeral head radius or radii, and the prosthesis material.
9.1.2 The axis and direction of testing (for example, central-superior-inferior).
9.1.3 Subluxation Test—The subluxation load and translation for each specimen, as well as the axial load and displacement rate.
A chart plotting the load versus displacement with the 90 and 100 % subluxation loads clearly marked should be included.
9.1.4 Rocking Test—The axial load, cyclic displacement, maximum number of cycles, testing frequency, and cause of test
termination. Testing parameters that differ from those recommended shall be justified.
9.1.5 Displacement Test—The edge displacements before and following cycling, highlighting the tensile displacement on the
unloaded side following rocking (for example, the displacement opposite the loaded side minus the value with the head at the
glenoid origin).
9.1.6 If the amplitude of the axial translation decreases suddenly during the test (indicating a tilt of the glenoid and the probable
onset of loosening), the number of cycles at which this occurred should be recorded.
10. Precision and Bias
10.1 Precision—The precision of this test method was established by an interlaboratory comparison among four laboratories,
with each laboratory testing three specimens. The specimens tested were commercially available UHMWPE glenoid components
and cobalt chrome humeral heads. The population mean micromotion before and after testing was 368 6 330 μm and 496 6 275
μm, respectively. Each laboratory utilized different methods for measuring the edge displacements, and one laboratory performed
the test using a lubricant at the contact surface instead of performing the test in solution (see X1.8).
10.1.1 Repeatability—For replicate results obtained by the same laboratory on nominally identical test specimens, the
repeatability standard deviation (s ) was 72.3 μm before testing and 268.0 μm after testing. All laboratories were within the critical
r
k values for the before and after testing conditions.
F2028 − 17
10.1.2 Reproducibility—For replicate results obtained by the same laboratory on nominally identical test specimens, the
reproducibility standard deviation (s ) was 335.9 μm before testing and 359.4 μm after testing. One laboratory exceeded the critical
R
h value for the before testing condition (h=1.50 versus h =1.49). All laboratories were within the critical h values for the after
crit
testing condition.
10.2 The above round robin data represent initial efforts at establishing a precision and bias statement for this test method and
have been published before documentation of full lab participation was completed (4 out of 6). Additionally, some labs experienced
difficulty with measurement of micromotion resulting in test method variances. Further testing is warranted and a revised precision
and bias statement incorporating participation by additional labs with reduced methodology variances is intended for future
publication.
11. Keywords
11.1 arthroplasty; glenoid; loosening; subluxation; total shoulder replacement
MODULAR DISASSOCIATION TEST METHOD
12. Summary of Test Method
12.1 The prosthetic glenoid component is fixed into a bone substitute with bone cement using the normal surgical technique.
12.2 The subluxation translation is determined experimentally on the intended test samples or additional components. This is
accomplished using a biaxial apparatus (see Fig. 5), by first applying an axial load perpendicular to the glenoid, then translating
the humeral head parallel to the glenoid plane until encountering a peak shear load. This is performed in both directions,
corresponding to the direction of intended rocking (for example, superior-inferior, anterior-posterior, or an alternative angle).
12.3 The humeral head is cycled to 90 % of the subluxation distance for a fixed number of cycles (see X1.2). (Cycling to 90 %
of the subluxation load would be acceptable, but is not practical because of the large displacements, quick speeds, and deformable
polyethylene).
13. Significance and Use
13.1 This test method is intended to investigate the locking mechanism of a modular glenoid. Disassociation of the insert is the
greatest issue in modular glenoid components. This test method can be used either to detect potential problems or to compare
design features.
14. Apparatus and Equipment
14.1 The test apparatus shall be constructed such that an axial loadforce is applied perpendicular to the glenoid plane and a shear
loadforce is applied parallel to the glenoid plane (see Fig. 42). Fig. 5 shows the axial loadforce to be horizontal and the shear
loadforce to be vertical; however, this arrangement may be reversed.
14.2 The glenoid and humeral head shall be enclosed in a chamber with water heated to 37 6 2°C, at least for the dynamic
portion of the test (see X1.6). A buffer may be added, if the tester deems this necessary.
14.3 A means to measure the axial load and shear translation is required.
14.4 The tests shall be performed on either mechanical or hydraulic load frames with adequate load capacity and shall meet the
criteria of Practices E4.
15. Sampling and Test Specimens
15.1 A minimum of three samples shall be tested. Additional samples may be used to reflect test variability. The test may be
conducted along the superior-inferior axis, the anterior-posterior axis, or another axis of interest to the user. The initial shear
displacement or load should be set just below the subluxation displacement or load. Each test will result either in a failure or, if
no disassociation occurs within the set number of cycles, a runout. The load should be progressively stepped down until at least
one runout occurs.
15.2 All glenoid components shall be in the final manufactured condition. All plastic components shall be sterilized according
to the manufacturer-recommended specifications for clinical use.
15.3 The humeral head shall include the identical radius or radii and material as the actual implant. Other features of the humeral
component such as the shaft may be omitted. The same head may be used for all tests unless the surface becomes damaged.
15.4 Glenoid and humeral components are used in total shoulder arthroplasty and should conform to the criteria in Specification
F1378.
16. Procedure
16.1 The following steps are common to both the subluxation (12.2) and rocking (12.3) tests:
F2028 − 17
16.1.1 Secure the glenoid component in a bone substitute with bone cement using the normal surgical procedure and
instrumentation. Do not perform tests until the cement has cured properly.
16.1.2 Position the path of the humeral head on the glenoid within 60.5 mm (sideways) of the desired path, for example, by
using a dye to locate the contact point of the humeral head. A dye is unnecessary for congruent prostheses. Locate the center of
the path (for the subluxation test, this need not be exact; for the rocking test, the peak loads at each run during cycling should be
within 610 % of each other for symmetrical designs).
16.1.3 Perform the measurements in 37°C water (see 14.2, X1.3 and X1.6).
16.1.4 Apply a given axial load to the glenoid, for example, 750 6 7.5 N (see X1.4).
16.2 Determine the subluxation translation experimentally on the intended test specimens or separate components (see X1.2):
16.2.1 After applying the axial load, displace the humeral head at a constant rate to a given displacement, ensuring that a peak
load is achieved in both directions. A rate of 50 mm/min is recommended to avoid polyethylene creep.
16.2.2 Yielding is expected at the recommended load and does not constitute a failure. The test shall be terminated if the
modular insert disassociates.
16.2.3 Record the axial load and subluxation translation. The subluxation load is not required for the rocking test, but may be
of interest to characterize the prosthesis.
16.3 Cyclically translate the humeral head to 90 % of the subluxation translation to cause a rocking motion of the glenoid at
a given frequency (for example, 2 Hz as a result of the large translations, or up to a maximum of 6 Hz) to a maximum number
of cycles (for example, 100 000 or higher, see X1.7). Maintain the axial load and specified displacement.
16.4 Terminate the test when either the maximum number of cycles has been reached or the glenoid insert disassociates. The
load should be set high enough to produce a failure, then reduced to produce at least one runout.
16.5 Testing may be continued to a higher number of cycles if desired.
17. Report
17.1 The test report shall include the following:
17.1.1 All details relevant to the particular implants tested including type, size, and lot number as well as the glenoid radius,
humeral head radius or radii, and the prosthesis materials.
17.1.2 The axis and direction of testing (for example, central-superior-inferior).
17.1.3 Subluxation Test—The subluxation load and translation for each specimen, as well as the axial load and displacement
rate. A chart plotting the load versus displacement with the 90 and 100 % subluxation loads clearly marked should be included.
17.1.4 Rocking Test—The axial load, cyclic displacement, maximum number of cycles, testing frequency, and cause of test
termination. Testing parameters that differ from those recommended shall be justified.
18. Precision and Bias
18.1 The precision and bias of this test method has not been established. Test results that could be used to establish precision
and bias are solicited.
19. Keywords
19.1 arthroplasty; disassociation; glenoid; subluxation; total shoulder replacement
REVERSE SHOULDER GLENOID LOOSENING/DISASSOCIATION TEST METHOD
20. Summary of Test Method
20.1 The prosthetic reverse glenoid baseplate is fixed with bone screws into a bone substitute using the normal surgical
technique.
20.2 The initial glenoid baseplate fixation to the bone substitute is measured before cyclic loading. Fixation can be measured
directly from the glenoid baseplate or with the glenosphere assembled. Fixation is measured as an axial compressive load is applied
approximately through the center of rotation, perpendicular to the glenoid plane as a shear load is applied parallel to the glenoid
plane. The induced displacement of the glenoid baseplate or glenoid baseplate/glenosphere assembly in the directions of the shear
and axial compressive loads should be measured. If the glenoid baseplate is noncircular in shape (see Fig. 34), then the shear
loadforce should be applied (and the associated displacements measured) along the device’s major and minor axes, typically in the
superior/inferior and anterior/posterior directions (see X2.10).
20.3 The glenosphere is connected to the glenoid baseplate (if not already assembled), mated with the reverse humeral
component, and the assembly is secured to a biaxial apparatus.
20.4 Using the biaxial apparatus, the reverse glenoid component is rotated about the humeral liner for a fixed number of cycles
as an axial compressive load is applied through the humeral liner into the glenoid component (see Fig. 8 and X2.6).
F2028 − 17
FIG. 8 Biaxial Testing Apparatus for Cyclic Test of Reverse Shoulders
20.5 The glenoid fixation is measured after cyclic loading according to the method described in 20.2.
20.6 As this cyclic test loads the reverse shoulder assembly in a physiologically relevant manner, the cyclic test is also
applicable to evaluate the resistance of a modular reverse shoulder design to disassociation or dislocation.
21. Significance and Use
21.1 This test method is intended to investigate the resistance of a reverse shoulder glenoid baseplate to loosening,
disassociation of modular components, and/or dislocation. Glenoid loosening is a common clinical complication of reverse total
shoulder arthroplasty. The method assumes that loosening occurs because of the cyclic loading of conforming articular curvatures
and not due to edge loading of nonconforming articular curvatures common to anatomic total shoulder arthroplasty (see X2.2,
X2.3, X2.6).
21.2 This test method can be used to detect potential problems and compare design features. Factors affecting loosening
performance include the type of screw (for example, compression versus locking), screw length and diameter, screw angulation,
screw positioning or configuration, glenoid baseplate contact area, glenoid baseplate backside geometry (for example, flat or
curved), glenoid baseplate fixation post geometry (for example, cylinder, taper, or screw), the amount of the glenoid baseplate
pressfit, glenosphere thickness, glenosphere diameter, glenoid component center of rotation (for example, medialized, lateralized,
or inferiorly shifted), articular geometry, materials, surface roughness, bone quality, and surgical technique (see X2.4).
21.3 This test method is intended to investigate short-term fixation only and does not evaluate the contribution of biological
fixation.
22. Apparatus and Equipment
22.1 The biaxial test apparatus shall be constructed such that an axial compressive load is applied approximately through the
center of rotation as the glenoid component is rotated about the humeral liner in the cyclic test (see Fig. 8). Fig. 9 depicts the axial
compressive load being applied through the humeral liner as the glenoid component is cyclically rotated in the superior/inferior
direction (see X2.6 and X2.7).
22.2 The test apparatus should also permit the application of a shear load approximately parallel to the glenoid plane as an axial
compressive load is applied perpendicular to the glenoid plane in the displacement test. Fig. 6 depicts the shear and axial
compressive loads applied directly to the glenoid baseplate. The point of application of loading should be chosen to minimize the
creation of a moment on the baseplate. Fig. 7 depicts an alternative method in which the shear and axial compressive loads are
applied through the glenosphere/glenoid baseplate assembly (see X2.10).
22.3 A bone substitute representing the strength of glenoid cancellous bone shall be used. If a polyurethane foam is used, it shall
conform to Specification F1839 (see X2.5).
F2028 − 17
FIG. 9 Cyclic Test Configuration
22.4 The cyclic loading of the reverse components can be performed in air at room temperature; however, the post-cyclic
displacement measurements should be made only after the tested components have cooled to room temperature following cyclic
loading (for example, 18 to 25°C). A fan or air jet may be used to cool the test components during cyclic loading or the test may
be performed in a lubricated environment, if the tester deems this necessary.
22.5 A means to measure the axial compressive load and the angle of rotation during cyclic loading is required during the cyclic
test. A means to measure the axial compressive load, shear load, and the glenoid baseplate displacement (or glenosphere/glenoid
baseplate assembly displacement) in the directions of both the shear and axial compressive loads is required during the
displacement test (see Fig. 5, Fig. 6, Fig. 7, and X2.4).
22.6 The tests shall be performed on either electromechanical or servohydraulic load frames with adequate load capacity and
shall meet the criteria of Practices E4. A loading frequency of 0.5 Hz is recommended; however, a faster or slower rate of rotation
may be used if desired though it should not exceed 1.0 Hz (see X2.9).
23. Sampling and Test Specimens
23.1 A minimum of three samples shall be tested. Additional samples may be used to reflect test variability. The reverse glenoid
component should be cyclically rotated about the humeral liner along the superior-inferior glenoid axis as this simulates humeral
abduction, the primary motion generated by the deltoid. The displacement test should be conducted such that displacement is
measured in the directions of both the applied shear and axial compressive loads; however, this shear load may be applied along
the superior-inferior axis, the anterior-posterior axis, or another axis of interest to the user. If the glenoid baseplate is noncircular
in shape (see Fig. 34), then the shear loadforce should be applied (and the associated displacements measured) along the
devicesdevice’s major and minor axes (see X2.10).
23.2 All reverse glenoid components shall be in the final manufactured condition. The same glenosphere may be used for all
tests unless the surface or locking mechanism becomes damaged.
23.3 The humeral liner shall include the identical radius or radii and material as the actual implant. If the test is also being used
to assess the resistance of the humeral component design to disassociation, the mating locking mechanisms shall also be in the final
manufactured condition. Other features of the humeral component such as the shaft/stem may be omitted. A new humeral liner
should be used in each test.
23.4 All plastic components shall be sterilized according to the manufacturer-recommended specifications for clinical use.
23.5 Glenoid and humeral components used in reverse total shoulder arthroplasty should conform to the criteria specified in
Specification F1378.
24. Procedure
24.1 Method to measure the initial glenoid baseplate (or glenosphere/glenoid baseplate assembly) displacement (see Fig. 6 and
Fig. 7).
F2028 − 17
24.1.1 Secure the glenoid baseplate to the bone substitute with bone screws using the normal surgical technique and
instrumentation.
24.1.2 Position the glenoid component in the testing apparatus so that a shear load can be applied parallel to the glenoid plane
as an axial compressive load is applied perpendicular to the glenoid plane, approximately through the center of rotation (see Fig.
6 and Fig. 7).
24.1.3 Apply a shear load of 350N 6 15N as an axial compressive load of 430N 6 15N is applied perpendicular to the glenoid
plane, the rate of the shear and compressive loads should not exceed 200N/sec (see Fig. 6, Fig. 7, and X2.10). A smaller magnitude
compressive load may be applied if desired.
24.1.4 Record the applied shear and axial compressive loads.
24.1.5 Use a dial indicator or similar device to measure the initial (pre-cyclic loading) displacement of the glenoid component
in the directions of both the applied shear and axial compressive loads. Static measurements can be performed at room temperature.
A measurement accuracy of at least 5 μm is required.
24.1.6 Record the glenoid baseplate (or glenosphere/glenoid baseplate assembly) displacement in the directions of both the
applied shear and axial compressive loads at the peak shear/compression loads.
24.1.7 Unload the specimen and repeat sections 24.1.2 through 24.1.6 with the same specimen for a total of at least three
displacement measurement sets.
24.2 Method to cyclically load the reverse glenoid components (see Fig. 8 and Fig. 9).
24.2.1 Secure the glenosphere to the glenoid baseplate and position the components in the biaxial testing apparatus.
24.2.2 Secure the humeral liner component in the biaxial testing apparatus and align the center of the humeral liner with the
reverse glenoid component.
24.2.3 Apply an axial compressive load of 750N 6 15N to the back of the humeral liner through the center of rotation. A larger
magnitude compressive load may be applied if desired (see Fig. 8 and X2.8).
24.2.4 Rotate the glenoid component about the humeral liner along the superior-inferior axis for 10,000 cycles at a rate of 0.5
Hz; the glenoid component should be rotated at least 45° (see Fig. 9, X2.7, and X2.9). The angular position of the glenoid
component may be biased at any position along the superior/inferior axis, if the tester deems this necessary.
24.2.5 Testing may be conducted in air at room temperature; a fan or air jet may be used to cool the test components during
cyclic loading or the test may be performed in a lubricated environment, if the tester deems this necessary.
24.2.6 Record the axial load and the magnitude of glenoid component rotation. The test shall be terminated if the construct
dislocates, disassociates, or fails in any way.
24.2.7 Testing may be continued to a higher number of cycles if desired.
24.3 Method to measure the post-cyclic glenoid baseplate (or glenosphere/glenoid baseplate assembly) displacement (see Fig.
6 and Fig. 7).
24.3.1 Dislocate the glenoid component from the humeral liner.
24.3.2 Position the glenoid component in the testing apparatus so that a shear load can be applied parallel to the glenoid plane
as an axial compressive load is applied perpendicular to the glenoid plane approximately through the center of rotation (see Fig.
6 and Fig. 7).
24.3.3 Apply a shear load of 350N 6 15N as an axial compressive load of 430N 6 15N is applied perpendicular to the glenoid
plane, the rate of the shear and compressive loads should not exceed 200N/sec (see Fig. 6, Fig. 7, and X2.10). A smaller magnitude
compressive load may be applied if desired.
24.3.4 Record the applied shear and axial compressive loads.
24.3.5 Use a dial indicator or similar device to measure the post-cyclic loading displacement of the glenoid component in the
directions of both the applied shear and axial compressive loads. Static measurements can be performed at room temperature.
24.3.6 Record the glenoid baseplate (or glenosphere/glenoid baseplate assembly) displacement in the directions of both the
applied shear and axial compressive loads at the peak shear/compression loads.
24.3.7 Unload the specimen and repeat sections 24.3.2 through 24.3.6 with the same specimen for a total of at least three
displacement measurement sets.
25. Report
25.1 The test report shall include the following:
25.1.1 All details relevant to the particular implants tested including type, size (for example, glenosphere radius and glenosphere
thickness), and lot number as well as the number/location of screws used, the orientation of the glenoid components, and the
density of polyurethane bone substitute used.
25.1.2 The axis and direction of cyclic testing, for example, superior-inferior.
25.1.3 The axis and direction of displacement testing, for example, superior-inferior, anterior-posterior, or another axis of
interest to the user.
25.1.4 Environmental temperature during testing.
25.1.5 Use of lubricant or cooling of the bearing surface during testing.
F2028 − 17
25.1.6 Cyclic Test—The axial load, amount of glenoid component rotation, maximum number of cycles, testing frequency, and
cause of test termination. Testing parameters that differ from those recommended shall be justified (see X2.6).
25.1.7 Displacement Test—The glenoid component displacements before and after cycling and the magnitude of axial
compressive and shear loads associated with each displacement measurement (see X2.10).
26. Precision and Bias
26.1 The precision and bias of this test method has not been established. Test results that could be used to establish precision
and bias are solicited.
27. Keywords
27.1 arthroplasty; glenoid loosening; reverse total shoulder replacement; sublu
...



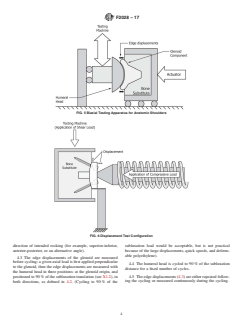


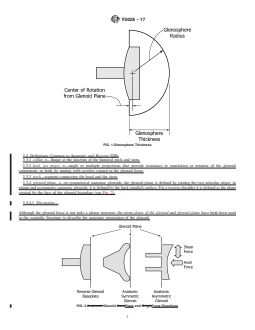

Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...