ASTM F2129-19a
(Test Method)Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices
Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices
SIGNIFICANCE AND USE
5.1 Corrosion of implantable medical devices can have deleterious effects on the device performance or may result in the release of corrosion products with harmful biological consequences; therefore it is important to determine the general corrosion behavior as well as the susceptibility of the devices to localized corrosion.
5.2 The forming and finishing steps used to create an implantable device may have significant effects on the corrosion resistance of the material out of which the device is fabricated. During the selection process of a material for use as an implantable device, testing the corrosion resistance of the material is an essential step; however, it does not necessarily provide critical data regarding device performance.
5.3 To accommodate the wide variety of device shapes and sizes encountered, a variety of holding devices can be used.
5.4 Note that the method is intentionally designed to reach conditions that are sufficiently severe to cause breakdown and deterioration of the medical devices and that these conditions may not necessarily be encountered in vivo. The results of this corrosion test conducted in artificial physiological electrolytes can provide useful data for comparison of different device materials, designs, or manufacturing processes. However, note that this test method does not take into account the effects of cells, proteins, and so forth, on the corrosion behavior in vivo.
SCOPE
1.1 This test method assesses the corrosion susceptibility of small, metallic, implant medical devices, or components thereof, using cyclic (forward and reverse) potentiodynamic polarization. Examples of device types that may be evaluated by this test method include, but are not limited to, vascular stents, ureteral stents (Specification F1828), filters, support segments of endovascular grafts, cardiac occluders, aneurysm or ligation clips, staples, and so forth.
1.2 This test method is used to assess a device in its final form and finish, as it would be implanted. These small devices should be tested in their entirety. The upper limit on device size is dictated by the electrical current delivery capability of the test apparatus (see Section 6). It is assumed that test methods, such as Reference Test Method G5 and Test Method G61 have been used for material screening.
1.3 Because of the variety of configurations and sizes of implants, this test method provides a variety of specimen holder configurations.
1.4 This test method is intended for use on implantable devices made from metals with a relatively high resistance to corrosion.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jan-2019
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 15-Jan-2019
- Effective Date
- 01-May-2019
- Effective Date
- 01-May-2018
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-May-2016
- Effective Date
- 15-Dec-2014
- Effective Date
- 01-Nov-2014
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
Overview
ASTM F2129-19a is a key international standard published by ASTM International that outlines the procedures for evaluating the corrosion susceptibility of small metallic implant devices through cyclic potentiodynamic polarization measurements. Corrosion is a critical concern for implantable medical devices, as it can compromise device performance and release harmful corrosion products in vivo. This test method provides a reliable, comparative means to assess the general and localized corrosion behavior of finished, small-scale medical implants using controlled laboratory conditions.
Key Topics
- Corrosion Susceptibility Assessment: The standard describes how to employ forward and reverse cyclic potentiodynamic polarization to evaluate the corrosion resistance of small medical implants in their final, clinical form and finish.
- Device Types: Applicable to a wide range of small metallic implant devices such as vascular stents, ureteral stents, cardiac occluders, endovascular graft support segments, aneurysm clips, and surgical staples.
- Testing Environment: Testing is conducted in artificial physiological electrolytes, such as phosphate-buffered saline, reflecting conditions relevant to human body environments.
- Critical Corrosion Potentials:
- Rest Potential (Eₑ): Stabilization period to assess natural surface properties.
- Breakdown Potential (E_b): Indicates onset of pitting corrosion.
- Protection Potential (E_p): Measures ability of the device surface to repassivate.
- Test Conditions: Devices are evaluated in their entirety where possible, and test equipment must accommodate various shapes and sizes, using different custom holders.
- Limitations: The test simulates aggressive environments to provoke corrosion, which may not directly correspond to true physiological conditions. Effects from biological elements such as cells or proteins are not considered.
Applications
The test method specified in ASTM F2129-19a is widely used throughout the medical device industry to:
- Evaluate Material and Process Selection: Manufacturers can compare corrosion resistance among different materials, device designs, or finishing processes to inform product development and ensure regulatory compliance.
- Finalize Device Readiness: The method helps verify that the surface and metallurgical condition of the finished device-post-fabrication and all processing steps-are suitable before clinical use.
- Quality Control and Risk Management: Provides essential data for risk assessments and quality assurance, documenting corrosion performance for regulatory submissions.
- Research and Development: Aids in the innovation of new biomaterials or device designs by supplying comparative corrosion data under standardized laboratory conditions.
- Comparative Analysis: Enables benchmarking of device corrosion resistance against established reference devices with known clinical outcomes.
- Simulated Use Testing: Encourages preconditioning to simulate real-world deployment and anatomical placement scenarios, which is essential for devices subjected to handling or deformation during insertion.
Related Standards
Several referenced documents are integral to the application of ASTM F2129-19a, including:
- ASTM G5 – Reference Test Method for Making Potentiodynamic Anodic Polarization Measurements
- ASTM G61 – Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility
- ASTM F1828 – Specification for Ureteral Stents
- ASTM D1193 – Specification for Reagent Water
- ASTM E177 & E691 – Practices for Precision and Bias in ASTM Test Methods
- ASTM G215 – Guide for Electrode Potential Measurement
Keywords: corrosion susceptibility, cyclic potentiodynamic polarization, medical implant devices, ASTM F2129, bio-compatibility, medical device testing, pitting potential, protection potential, biomedical materials, implant performance, metallic implants.
ASTM F2129-19a remains an essential resource for ensuring that small metallic implantable medical devices meet stringent corrosion resistance requirements and support safe long-term use in clinical settings.
Buy Documents
ASTM F2129-19a - Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices
REDLINE ASTM F2129-19a - Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2129-19a is a standard published by ASTM International. Its full title is "Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Susceptibility of Small Implant Devices". This standard covers: SIGNIFICANCE AND USE 5.1 Corrosion of implantable medical devices can have deleterious effects on the device performance or may result in the release of corrosion products with harmful biological consequences; therefore it is important to determine the general corrosion behavior as well as the susceptibility of the devices to localized corrosion. 5.2 The forming and finishing steps used to create an implantable device may have significant effects on the corrosion resistance of the material out of which the device is fabricated. During the selection process of a material for use as an implantable device, testing the corrosion resistance of the material is an essential step; however, it does not necessarily provide critical data regarding device performance. 5.3 To accommodate the wide variety of device shapes and sizes encountered, a variety of holding devices can be used. 5.4 Note that the method is intentionally designed to reach conditions that are sufficiently severe to cause breakdown and deterioration of the medical devices and that these conditions may not necessarily be encountered in vivo. The results of this corrosion test conducted in artificial physiological electrolytes can provide useful data for comparison of different device materials, designs, or manufacturing processes. However, note that this test method does not take into account the effects of cells, proteins, and so forth, on the corrosion behavior in vivo. SCOPE 1.1 This test method assesses the corrosion susceptibility of small, metallic, implant medical devices, or components thereof, using cyclic (forward and reverse) potentiodynamic polarization. Examples of device types that may be evaluated by this test method include, but are not limited to, vascular stents, ureteral stents (Specification F1828), filters, support segments of endovascular grafts, cardiac occluders, aneurysm or ligation clips, staples, and so forth. 1.2 This test method is used to assess a device in its final form and finish, as it would be implanted. These small devices should be tested in their entirety. The upper limit on device size is dictated by the electrical current delivery capability of the test apparatus (see Section 6). It is assumed that test methods, such as Reference Test Method G5 and Test Method G61 have been used for material screening. 1.3 Because of the variety of configurations and sizes of implants, this test method provides a variety of specimen holder configurations. 1.4 This test method is intended for use on implantable devices made from metals with a relatively high resistance to corrosion. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Corrosion of implantable medical devices can have deleterious effects on the device performance or may result in the release of corrosion products with harmful biological consequences; therefore it is important to determine the general corrosion behavior as well as the susceptibility of the devices to localized corrosion. 5.2 The forming and finishing steps used to create an implantable device may have significant effects on the corrosion resistance of the material out of which the device is fabricated. During the selection process of a material for use as an implantable device, testing the corrosion resistance of the material is an essential step; however, it does not necessarily provide critical data regarding device performance. 5.3 To accommodate the wide variety of device shapes and sizes encountered, a variety of holding devices can be used. 5.4 Note that the method is intentionally designed to reach conditions that are sufficiently severe to cause breakdown and deterioration of the medical devices and that these conditions may not necessarily be encountered in vivo. The results of this corrosion test conducted in artificial physiological electrolytes can provide useful data for comparison of different device materials, designs, or manufacturing processes. However, note that this test method does not take into account the effects of cells, proteins, and so forth, on the corrosion behavior in vivo. SCOPE 1.1 This test method assesses the corrosion susceptibility of small, metallic, implant medical devices, or components thereof, using cyclic (forward and reverse) potentiodynamic polarization. Examples of device types that may be evaluated by this test method include, but are not limited to, vascular stents, ureteral stents (Specification F1828), filters, support segments of endovascular grafts, cardiac occluders, aneurysm or ligation clips, staples, and so forth. 1.2 This test method is used to assess a device in its final form and finish, as it would be implanted. These small devices should be tested in their entirety. The upper limit on device size is dictated by the electrical current delivery capability of the test apparatus (see Section 6). It is assumed that test methods, such as Reference Test Method G5 and Test Method G61 have been used for material screening. 1.3 Because of the variety of configurations and sizes of implants, this test method provides a variety of specimen holder configurations. 1.4 This test method is intended for use on implantable devices made from metals with a relatively high resistance to corrosion. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2129-19a is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2129-19a has the following relationships with other standards: It is inter standard links to ASTM F2129-19, ASTM G3-14(2019), ASTM G61-86(2018), ASTM G215-17, ASTM G215-16, ASTM G3-14, ASTM G5-14, ASTM F1828-97(2014), ASTM G61-86(2014), ASTM E177-14, ASTM G3-13, ASTM E177-13, ASTM E691-13, ASTM G5-13e1, ASTM G5-13. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2129-19a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2129 − 19a
Standard Test Method for
Conducting Cyclic Potentiodynamic Polarization
Measurements to Determine the Corrosion Susceptibility of
Small Implant Devices
This standard is issued under the fixed designation F2129; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 This test method assesses the corrosion susceptibility of
Barriers to Trade (TBT) Committee.
small, metallic, implant medical devices, or components
thereof, using cyclic (forward and reverse) potentiodynamic
2. Referenced Documents
polarization. Examples of device types that may be evaluated
by this test method include, but are not limited to, vascular 2.1 ASTM Standards:
D1193 Specification for Reagent Water
stents, ureteral stents (Specification F1828), filters, support
E177 Practice for Use of the Terms Precision and Bias in
segments of endovascular grafts, cardiac occluders, aneurysm
ASTM Test Methods
or ligation clips, staples, and so forth.
E691 Practice for Conducting an Interlaboratory Study to
1.2 This test method is used to assess a device in its final
Determine the Precision of a Test Method
form and finish, as it would be implanted. These small devices
F1828 Specification for Ureteral Stents
shouldbetestedintheirentirety.Theupperlimitondevicesize
G3 Practice for Conventions Applicable to Electrochemical
is dictated by the electrical current delivery capability of the
Measurements in Corrosion Testing
test apparatus (see Section 6). It is assumed that test methods,
G5 Reference Test Method for Making Potentiodynamic
such as Reference Test Method G5 and Test Method G61 have
Anodic Polarization Measurements
been used for material screening.
G15 Terminology Relating to Corrosion and Corrosion Test-
1.3 Because of the variety of configurations and sizes of
ing (Withdrawn 2010)
implants, this test method provides a variety of specimen
G61 Test Method for Conducting Cyclic Potentiodynamic
holder configurations.
Polarization Measurements for Localized Corrosion Sus-
ceptibility of Iron-, Nickel-, or Cobalt-Based Alloys
1.4 This test method is intended for use on implantable
G215 Guide for Electrode Potential Measurement
devices made from metals with a relatively high resistance to
corrosion.
3. Terminology
1.5 The values stated in SI units are to be regarded as
3.1 Definitions:
standard. No other units of measurement are included in this
3.1.1 potentiodynamic cyclic polarization (forward and re-
standard.
verse polarization), n—a technique in which the potential of
1.6 This standard does not purport to address all of the
the test specimen is controlled and the corrosion current
safety concerns, if any, associated with its use. It is the
measured by a potentiostat. The potential is scanned in the
responsibility of the user of this standard to establish appro-
positive or noble (forward) direction as defined in Practice G3.
priate safety, health, and environmental practices and deter-
The potential scan is continued until a predetermined potential
mine the applicability of regulatory limitations prior to use.
or current density is reached.Typically, the scan is run until the
1.7 This international standard was developed in accor-
transpassive region is reached, and the specimen no longer
dance with internationally recognized principles on standard-
demonstratespassivity,asdefinedinPracticeG3.Thepotential
ization established in the Decision on Principles for the
1 2
This test method is under the jurisdiction ofASTM Committee F04 on Medical For referenced ASTM standards, visit the ASTM website, www.astm.org, or
and Surgical Materials and Devices and is the direct responsibility of Subcommittee contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
F04.15 on Material Test Methods. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Jan. 15, 2019. Published March 2019. Originally the ASTM website.
approved in 2001. Last previous edition approved in 2019 as F2129–19. DOI: The last approved version of this historical standard is referenced on
10.1520/F2129–19A. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2129 − 19a
scan direction is then reversed until the specimen repassivates 5. Significance and Use
or the potential reaches a preset value.
5.1 Corrosion of implantable medical devices can have
3.1.2 potentiostat, n—an instrument for automatically main-
deleterious effects on the device performance or may result in
taining an electrode in an electrolyte at a constant potential or
the release of corrosion products with harmful biological
controlled potentials with respect to a suitable reference
consequences;thereforeitisimportanttodeterminethegeneral
electrode (see Terminology G15).
corrosion behavior as well as the susceptibility of the devices
3.1.3 scan rate, n—the rate at which the controlling voltage to localized corrosion.
is changed.
5.2 The forming and finishing steps used to create an
3.2 Symbols: implantable device may have significant effects on the corro-
sion resistance of the material out of which the device is
3.2.1 E = Breakdown or Critical Pitting Potential—the
b
least noble potential at which pitting or crevice corrosion or fabricated. During the selection process of a material for use as
an implantable device, testing the corrosion resistance of the
bothwillinitiateandpropagateasdefinedinTerminologyG15.
An increase in the resistance to pitting corrosion is associated material is an essential step; however, it does not necessarily
with an increase in E . provide critical data regarding device performance.
b
3.2.2 E = Final Potential—a preset potential at which the
5.3 To accommodate the wide variety of device shapes and
f
scan is stopped.
sizes encountered, a variety of holding devices can be used.
3.2.3 E = Initial Potential—the potential at which the
i
5.4 Note that the method is intentionally designed to reach
potentiostat begins the controlled potentiodynamic scan.
conditions that are sufficiently severe to cause breakdown and
3.2.4 E = Protection Potential—the potential at which the
deterioration of the medical devices and that these conditions
p
reverse scan intersects the forward scan at a value that is less
may not necessarily be encountered in vivo. The results of this
noble than E . E cannot be determined if there is no
corrosion test conducted in artificial physiological electrolytes
b p
breakdown. Whereas, pitting will occur on a pit-free surface
can provide useful data for comparison of different device
above E , it will occur only in the range of potentials between
materials, designs, or manufacturing processes. However, note
b
E and E if the surface is already pitted. The severity of
that this test method does not take into account the effects of
p b
crevice corrosion susceptibility increases with increasing hys-
cells, proteins, and so forth, on the corrosion behavior in vivo.
teresis of the polarization curve, the difference between E and
b
E .
p 6. Apparatus
3.2.5 E = Rest Potential—the potential of the working
r
6.1 Potentiostat, calibrated in accordance with Reference
electrode relative to the reference electrode measured under
Test Method G5.
virtual open-circuit conditions (working electrode is not polar-
6.2 Working Electrode, to be used as the test specimen, as
ized).
describedinSection9.Itsconfigurationandholderwilldepend
3.2.6 E = Vertex Potential—a preset potential, at which the
v
on the type of specimen being tested, as described in Section 7.
scan direction is reversed.
In all cases, the metallurgical and surface condition of a
3.2.7 E = Zero Current Potential—the potential at which
zc
specimen simulating a device must be in the same condition as
the current reaches a minimum during the forward scan.
the device.
3.2.8 i = Threshold Current Density (mA/cm )—a preset
t
6.3 Reference Electrode—A standard reference electrode
current density, at which the scan direction is reversed.
should be used in the test. Examples of standard electrodes are
Typically, the scan is reversed when a current density two
provided in Guide G215, along with a table showing conver-
decades higher than the current density at the breakdown
sions between electrodes. The reference electrode used in the
potential (E ) is reached.
b
test shall be identified along with the conversion used, if
necessary. Individual electrochemical potentials such as E and
4. Summary of Test Method
b
E shall be reported relative to the saturated calomel electrode
r
4.1 The device is placed in an appropriate deaerated simu-
(SCE). (For example, the saturated Ag/AgCl electrode is 45
lated physiological solution, and the rest potential (E)is
r
mV electronegative to SCE. A reading of 0 mV vs. Sat’d.
recorded for1hor, alternatively, until the rest potential
Ag/AgCl would be equivalent to a reading of –45 mVvs. SCE.
stabilizes to a rate of change less than 3 mV/min. The
Therefore, to convert potentials measured using a Sat’d.
potentiodynamic scan is then started at E and scanned in the
r
Ag/AgCl electrode to the SCE scale, 45 mV should be
positiveornoble(forward)direction.Thescanisreversedafter
subtracted.) When plotting curves, potentials may be plotted
either the vertex potential (E ) is reached or the current density
v
using raw data, that is, to the scale of the reference electrode
hasreachedavalueapproximatelytwodecadesgreaterthanthe
used during the test and this shall be clearly shown on the axis
current density measured at the breakdown potential. The
label (for example, “Potential vs. SCE (mV)” or “Potential vs.
reverse scan is stopped after the current has become less than
Sat’d. Ag/AgCl (mV)”).
that in the forward direction or the potential reaches E . The
r
data is plotted with the current density in mA/cm on the x axis 6.4 Salt Bridge, such as a Luggin probe, shall be used
(logarithmic axis) versus the potential in mV on the y axis between the working and reference electrode, such as the type
(linear axis). shown in Reference Test Method G5.
F2129 − 19a
6.5 Auxiliary Electrodes: some components in bile solutions are not available in reagent
6.5.1 Two platinum auxiliary electrodes may be prepared grade). Such reagents shall conform to the specifications of the
from high-purity rod stock. The surfaces may be platinized, as Committee on Analytical Reagents of the American Chemical
per Reference Test Method G5. Society.
6.5.2 Alternatively,high-puritygraphiteauxiliaryelectrodes 8.1.1 The water shall be distilled or deionized conforming
may be used in accordance with Reference Test Method G5. to the purity requirements of Specification D1193, Type IV
Care should be taken to ensure that they do not get contami- reagent water.
nated during a test. 8.1.2 Unless otherwise specified, phosphate buffered saline
6.5.3 The auxiliary electrode surface area should be at least (PBS) should be used as the standard test solution.Arepresen-
four times greater than the sample surface area. Use of tative PBS formulation is given in Appendix X2, along with
wire-mesh platinum might be more cost-effective than plati- the formulations of two simulated bile solutions for testing
num cylinders when testing larger specimens or whole devices. implantable medical devices intended for use in the biliary
system, the formulations of two artificial urine solutions for
6.6 Suitable Polarization Cell, with a sufficient volume to
testing implantable indwelling materials intended for use in the
allow the solution to cover the sample and the counter
urinary tract, and the compositions of two other commonly
electrode, and to prevent changes in pH during testing.
used physiological solutions.
Furthermore, the cell needs to be appropriately sealed to avoid
8.1.3 ThepHoftheelectrolyteshouldbeadjustedasneeded
oxygen access and include a secondary bubbler for the release
based on the nature of the solution (for example, for PBS,
of exhaust gas without the back diffusion of oxygen. The test
adjust the pH to a value of 7.4 6 0.2 by the addition of
cell must be able to hold a minimum of 500 ml.
NaH PO (acid) or Na HPO (base)). When the electrolyte is
2 4 2 4
6.7 Water Bath, or other heating appliance capable of
deaerated, its pH may change significantly if it is not suffi-
maintaining the test solution temperature at 37 6 1 °C (see
ciently buffered. Several pH controlling methods are provided
X1.5).
in Appendix X2.
6.8 Purge Gas Delivery System, capable of delivering nitro- 8.1.4 Nitrogen gas with a minimum purity of 99.99 %
gen gas at 150 cm /min.
should be used for purging the test solution of oxygen.
7. Specimen Holders
9. Test Specimen
7.1 There are a variety of holders that may be used in this
9.1 Unless otherwise justified, all samples selected for
test method. Each is designed for a specific type or class of
testing should be taken from finished, clinical-quality product.
device.
Cosmetic rejects or other nonclinical samples may be used if
7.2 Short wire or coil specimens: the cause for rejection does not affect the corrosion behavior of
7.2.1 Specimens can be held suspended from a clamping
the device. Sterilization may be omitted if it can be demon-
device. For example, the threaded end of a Reference Test strated that prior sterilization has no effect on the corrosion
Method G5 holder can be used to hold two stainless steel nuts.
behavior of the device.
The wire test specimen is clamped between these nuts and bent 9.1.1 Test specimens used for design parameter studies can
so as to enter the test solution.
be prepared as detailed in Reference Test Method G5 for
7.2.2 The surface area of the test specimen shall be calcu- working electrodes, with the requirement that the metallurgical
lated based on the length of wire or coil immersed in the test
and surface conditions of the specimens are the same as the
solution. intended implantable medical device.
7.2.3 This type of holder exposes the specimen to the
9.2 Preconditioning can impact the corrosion behavior of
air-liquid interface, which is subject to localized crevice
implants; therefore, prior to testing, devices should be sub-
corrosion. Test specimens should be examined carefully after
jected to simulated use or other preconditioning that is appro-
testing to ensure that there is no localized corrosion at or just
priate to their application.Ajustification should be provided if
below the interface. If specimens show evidence of localized
simulated use or other preconditioning is omitted. Additional
corrosion at the air-liquid interface, then the portion of the
information on simulated use is found in Appendix X4.
specimen passing across this interface shall be sealed with an
impervious coating.
10. Procedure
7.2.4 Alternatively, one may choose to coat the portion of
10.1 Prepare the specimen such that the portion exposed to
the specimen out of the solution and the connection to the
the test solution is in the same metallurgical and surface
specimen holder with a suitable coating. The surfaces out of
condition as the implantable form of the medical device being
solution will tend to have test solution condensed on them and
studied.
this may lead to undesirable results.
7.3 One method for holding stents or cylindrical devices is
Reagent Chemicals, American Chemical Society Specifications, American
shown in Appendix X3.
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
listed by the American Chemical Society, see Analar Standards for Laboratory
8. Reagents
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
8.1 Reagent grade chemicals shall be used for this test
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
method when they are commercially available (for example, MD.
F2129 − 19a
10.1.1 Where reasonable, calculate the approximate surface 10.7 At the end of the E recording period, start the
r
area exposed to solution; where not reasonable, for example, potentiodynamic scan in the positive or noble (forward)
with a porous surface or covered device, utilize a calculated
direction, as defined in Practice G3. The scanning program
estimate of surface area exposed to solution and include the
should be set with the following parameters:
reasoning behind that estimate. The exposed surface area is
10.7.1 Starting or initial potential (E)at E .
i r
used to calculate the current density (current per surface area)
10.7.2 Ascan rate of either 0.167 mV/s or 1 mV/s should be
generated by the specimen during the test.
used. Note that the scan rate may affect the breakdown
10.2 Prepareenoughtestsolutiontoimmersethedeviceand
potential of the device and the shape of the passive region of
auxiliary electrodes and so avoid any appreciable change in the
the polarization curve. Comparisons should not be made
solution corrosivity during the test through exhaustion of the
between test results using different scan rates, even if all other
corrosive constituents or by accumulation of corrosion prod-
experimental parameters are held constant.
ucts that may affect further corrosion. At a minimum, transfer
10.7.3 Acurrent density threshold two decades greater than
500 mLof electrolyte to a clean polarization cell. Measure and
the current density recorded at breakdown can be used to
record the pH of the solution before and after each test.
reverse the voltage scan.
10.3 Place the auxiliary electrodes, salt bridge probe,
10.7.3.1 Alternatively, a minimum reversing or vertex po-
thermometer, and gas purge diffuser in the test chamber and
tential (E ) of 800 mV (SCE) may be used to control the
v
bring the temperature of the test solution to 37 6 1 °C.
potentiostat (see X1.6).
10.4 Purge the solution for a minimum of 30 min with
10.7.4 The final potential (E) is set to E . The reverse scan
3 f r
nitrogen gas at a flow rate of 150 cm /min.
may be manually stopped at potentials above E in cases in
r
10.5 Gently immerse the test specimen in the test solution
which a protection potential (E ) is observed as a drop in
p
and connect it to a potentiostat. Continue the nitrogen purge
current density below that of the passive current density or
throughout the test.
when no hysteresis loop is formed once the scan is reversed
(E ), indicating repassivation (Fig. 1a), no protection potential
10.6 Record E for1hor, alternatively, until the rest
r v
potential stabilizes to a rate of change less than 3 mV/min. (Fig. 1b), or oxygen evolution (Fig. 1c).
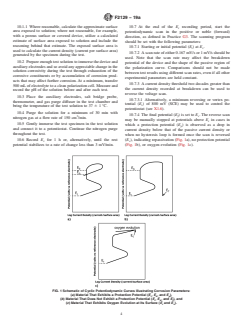
FIG. 1 Schematic of Cyclic Potentiodynamic Curves Illustrating Corrosion Parameters:
(a) Material That Exhibits a Protection Potential (E , E , and E ),
r b p
(b) Material That Does Not Exhibit a Protection Potential (E , E , and E ), and
r b f
(c) Material That Exhibits Oxygen Evolution at Its Surface (E and E ).
r v
F2129 − 19a
TABLE 1 Precision of Rest Potential E (mV)
r
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Standard Deviation Standard Deviation Limit Limit
316 SS –7 33 64 93 178
455 SS –30 38 67 105 187
Nitinol A –519 35 49 98 137
Nitinol B –482 21 49 60 138
TABLE 2 Precision of Breakdown Potential E (mV)
b
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Standard Deviation Standard Deviation Limit Limit
316 SS 679 161 190 451 531
455 SS 269 36 40 100 113
Nitinol A 160 82 108 230 302
Nitinol B 180 54 94 152 263
TABLE 3 Precision of Repassivation Potential E (mV)
p
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Standard Deviation Standard Deviation Limit Limit
316 SS . . . . .
455 SS . . . . .
Nitinol A –171 57 108 160 302
Nitinol B –126 38 58 107 162
10.8 Ifcontrolspecimensareused,theyshallbetestedusing 11.5 A copy of the cyclic polarization curve (plotted using
the same method as the investigated devices. the raw data collected in the experiment) should be provided in
the report. The reference electrode scale used for the curve
11. Report
shall be clearly indicated in the axis label (for example,
11.1 The report should contain a detailed description of the “Potential (mV vs. SCE)” or “Potential (mV vs. sat’d Ag/
AgCl)”).
test specimen, including metallurgical and surface condition-
ing.
11.6 A generic description of the appearance of any corro-
11.1.1 When specimens are not finished devices, for
sion observed on the specimen should be described. Photo-
example, surrogates, the sample preparation should be de-
graphic documentation may be appropriate.
scribed in detail.
12. Precision and Bias
11.2 A description of the test conditions should also be
12.1 An interlaboratory study was conducted in accordance
reported, including the following:
with Practice E691 in twelve laboratories with four different
11.2.1 The volume of the test cell;
materials. Each laboratory tested eig
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2129 − 19 F2129 − 19a
Standard Test Method for
Conducting Cyclic Potentiodynamic Polarization
Measurements to Determine the Corrosion Susceptibility of
Small Implant Devices
This standard is issued under the fixed designation F2129; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method assesses the corrosion susceptibility of small, metallic, implant medical devices, or components thereof,
using cyclic (forward and reverse) potentiodynamic polarization. Examples of device types that may be evaluated by this test
method include, but are not limited to, vascular stents, ureteral stents (Specification F1828), filters, support segments of
endovascular grafts, cardiac occluders, aneurysm or ligation clips, staples, and so forth.
1.2 This test method is used to assess a device in its final form and finish, as it would be implanted. These small devices should
be tested in their entirety. The upper limit on device size is dictated by the electrical current delivery capability of the test apparatus
(see Section 6). It is assumed that test methods, such as Reference Test Method G5 and Test Method G61 have been used for
material screening.
1.3 Because of the variety of configurations and sizes of implants, this test method provides a variety of specimen holder
configurations.
1.4 This test method is intended for use on implantable devices made from metals with a relatively high resistance to corrosion.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
F1828 Specification for Ureteral Stents
G3 Practice for Conventions Applicable to Electrochemical Measurements in Corrosion Testing
G5 Reference Test Method for Making Potentiodynamic Anodic Polarization Measurements
G15 Terminology Relating to Corrosion and Corrosion Testing (Withdrawn 2010)
G61 Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility of
Iron-, Nickel-, or Cobalt-Based Alloys
G215 Guide for Electrode Potential Measurement
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.15 on Material Test Methods.
Current edition approved Jan. 1, 2019Jan. 15, 2019. Published March 2019. Originally approved in 2001. Last previous edition approved in 20172019 as F2129–17b.–19.
DOI: 10.1520/F2129–19.10.1520/F2129–19A.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2129 − 19a
3. Terminology
3.1 Definitions:
3.1.1 potentiodynamic cyclic polarization (forward and reverse polarization), n—a technique in which the potential of the test
specimen is controlled and the corrosion current measured by a potentiostat. The potential is scanned in the positive or noble
(forward) direction as defined in Practice G3. The potential scan is continued until a predetermined potential or current density is
reached. Typically, the scan is run until the transpassive region is reached, and the specimen no longer demonstrates passivity, as
defined in Practice G3. The potential scan direction is then reversed until the specimen repassivates or the potential reaches a preset
value.
3.1.2 potentiostat, n—an instrument for automatically maintaining an electrode in an electrolyte at a constant potential or
controlled potentials with respect to a suitable reference electrode (see Terminology G15).
3.1.3 scan rate, n—the rate at which the controlling voltage is changed.
3.2 Symbols:
3.2.1 E = Breakdown or Critical Pitting Potential—the least noble potential at which pitting or crevice corrosion or both will
b
initiate and propagate as defined in Terminology G15. An increase in the resistance to pitting corrosion is associated with an
increase in E .
b
3.2.2 E = Final Potential—a preset potential at which the scan is stopped.
f
3.2.3 E = Initial Potential—the potential at which the potentiostat begins the controlled potentiodynamic scan.
i
3.2.4 E = Protection Potential—the potential at which the reverse scan intersects the forward scan at a value that is less noble
p
than E .E cannot be determined if there is no breakdown. Whereas, pitting will occur on a pit-free surface above E , it will occur
b p b
only in the range of potentials between E and E if the surface is already pitted. The severity of crevice corrosion susceptibility
p b
increases with increasing hysteresis of the polarization curve, the difference between E and E .
b p
3.2.5 E = Rest Potential—the potential of the working electrode relative to the reference electrode measured under virtual
r
open-circuit conditions (working electrode is not polarized).
3.2.6 E = Vertex Potential—a preset potential, at which the scan direction is reversed.
v
3.2.7 E = Zero Current Potential—the potential at which the current reaches a minimum during the forward scan.
zc
3.2.8 i = Threshold Current Density (mA/cm )—a preset current density, at which the scan direction is reversed. Typically, the
t
scan is reversed when a current density two decades higher than the current density at the breakdown potential (E ) is reached.
b
4. Summary of Test Method
4.1 The device is placed in an appropriate deaerated simulated physiological solution, and the rest potential (E ) is recorded for
r
1 h or, alternatively, until the rest potential stabilizes to a rate of change less than 3 mV/min. The potentiodynamic scan is then
started at E and scanned in the positive or noble (forward) direction. The scan is reversed after either the vertex potential (E ) is
r v
reached or the current density has reached a value approximately two decades greater than the current density measured at the
breakdown potential. The reverse scan is stopped after the current has become less than that in the forward direction or the potential
reaches E . The data is plotted with the current density in mA/cm on the x axis (logarithmic axis) versus the potential in mV on
r
the y axis (linear axis).
5. Significance and Use
5.1 Corrosion of implantable medical devices can have deleterious effects on the device performance or may result in the release
of corrosion products with harmful biological consequences; therefore it is important to determine the general corrosion behavior
as well as the susceptibility of the devices to localized corrosion.
5.2 The forming and finishing steps used to create an implantable device may have significant effects on the corrosion resistance
of the material out of which the device is fabricated. During the selection process of a material for use as an implantable device,
testing the corrosion resistance of the material is an essential step; however, it does not necessarily provide critical data regarding
device performance.
5.3 To accommodate the wide variety of device shapes and sizes encountered, a variety of holding devices can be used.
5.4 Note that the method is intentionally designed to reach conditions that are sufficiently severe to cause breakdown and
deterioration of the medical devices and that these conditions may not necessarily be encountered in vivo. The results of this
corrosion test conducted in artificial physiological electrolytes can provide useful data for comparison of different device materials,
designs, or manufacturing processes. However, note that this test method does not take into account the effects of cells, proteins,
and so forth, on the corrosion behavior in vivo.
6. Apparatus
6.1 Potentiostat, calibrated in accordance with Reference Test Method G5.
F2129 − 19a
6.2 Working Electrode, to be used as the test specimen, as described in Section 9. Its configuration and holder will depend on
the type of specimen being tested, as described in Section 7. In all cases, the metallurgical and surface condition of a specimen
simulating a device must be in the same condition as the device.
6.3 Reference Electrode—A standard reference electrode should be used in the test. Examples of standard electrodes are
provided in Guide G215, along with a table showing conversions between electrodes. The reference electrode used in the test shall
be identified along with the conversion used, if necessary. Individual electrochemical potentials such as E and E shall be reported
b r
relative to the saturated calomel electrode (SCE), as described in Reference Test Method(SCE). (For example, the saturated
Ag/AgCl electrode G5, shall be used as a reference electrode.is 45 mV electronegative to SCE. A reading of 0 mV vs. Sat’d.
Ag/AgCl would be equivalent to a reading of –45 mV vs. SCE. Therefore, to convert potentials measured using a Sat’d. Ag/AgCl
electrode to the SCE scale, 45 mV should be subtracted.) When plotting curves, potentials may be plotted using raw data, that is,
to the scale of the reference electrode used during the test and this shall be clearly shown on the axis label (for example, “Potential
vs. SCE (mV)” or “Potential vs. Sat’d. Ag/AgCl (mV)”).
6.4 Salt Bridge, such as a Luggin probe, shall be used between the working and reference electrode, such as the type shown
in Reference Test Method G5.
F2129 − 19a
6.5 Auxiliary Electrodes:
6.5.1 Two platinum auxiliary electrodes may be prepared from high-purity rod stock. The surfaces may be platinized, as per
Reference Test Method G5.
6.5.2 Alternatively, high-purity graphite auxiliary electrodes may be used in accordance with Reference Test Method G5. Care
should be taken to ensure that they do not get contaminated during a test.
6.5.3 The auxiliary electrode surface area should be at least four times greater than the sample surface area. Use of wire-mesh
platinum might be more cost-effective than platinum cylinders when testing larger specimens or whole devices.
F2129 − 19a
6.6 Suitable Polarization Cell, with a sufficient volume to allow the solution to cover the sample and the counter electrode, and
to prevent changes in pH during testing. Furthermore, the cell needs to be appropriately sealed to avoid oxygen access and include
a secondary bubbler for the release of exhaust gas without the back diffusion of oxygen. The test cell must be able to hold a
minimum of 500 ml.
6.7 Water Bath, or other heating appliance capable of maintaining the test solution temperature at 37 6 1 °C (see X1.5).
6.8 Purge Gas Delivery System, capable of delivering nitrogen gas at 150 cm /min.
7. Specimen Holders
7.1 There are a variety of holders that may be used in this test method. Each is designed for a specific type or class of device.
7.2 Short wire or coil specimens:
7.2.1 Specimens can be held suspended from a clamping device. For example, the threaded end of a Reference Test Method G5
holder can be used to hold two stainless steel nuts. The wire test specimen is clamped between these nuts and bent so as to enter
the test solution.
7.2.2 The surface area of the test specimen shall be calculated based on the length of wire or coil immersed in the test solution.
7.2.3 This type of holder exposes the specimen to the air-liquid interface, which is subject to localized crevice corrosion. Test
specimens should be examined carefully after testing to ensure that there is no localized corrosion at or just below the interface.
If specimens show evidence of localized corrosion at the air-liquid interface, then the portion of the specimen passing across this
interface shall be sealed with an impervious coating.
7.2.4 Alternatively, one may choose to coat the portion of the specimen out of the solution and the connection to the specimen
holder with a suitable coating. The surfaces out of solution will tend to have test solution condensed on them and this may lead
to undesirable results.
7.3 One method for holding stents or cylindrical devices is shown in Appendix X3.
8. Reagents
8.1 Reagent grade chemicals shall be used for this test method when they are commercially available (for example, some
components in bile solutions are not available in reagent grade). Such reagents shall conform to the specifications of the Committee
on Analytical Reagents of the American Chemical Society.
8.1.1 The water shall be distilled or deionized conforming to the purity requirements of Specification D1193, Type IV reagent
water.
8.1.2 Unless otherwise specified, phosphate buffered saline (PBS) should be used as the standard test solution. A representative
PBS formulation is given in Appendix X2, along with the formulations of two simulated bile solutions for testing implantable
medical devices intended for use in the biliary system, the formulations of two artificial urine solutions for testing implantable
indwelling materials intended for use in the urinary tract, and the compositions of two other commonly used physiological
solutions.
8.1.3 The pH of the electrolyte should be adjusted as needed based on the nature of the solution (for example, for PBS, adjust
the pH to a value of 7.4 6 0.2 by the addition of NaH PO (acid) or Na HPO (base)). When the electrolyte is deaerated, its pH
2 4 2 4
may change significantly if it is not sufficiently buffered. Several pH controlling methods are provided in Appendix X2.
8.1.4 Nitrogen gas with a minimum purity of 99.99 % should be used for purging the test solution of oxygen.
9. Test Specimen
9.1 Unless otherwise justified, all samples selected for testing should be taken from finished, clinical-quality product. Cosmetic
rejects or other nonclinical samples may be used if the cause for rejection does not affect the corrosion behavior of the device.
Sterilization may be omitted if it can be demonstrated that prior sterilization has no effect on the corrosion behavior of the device.
9.1.1 Test specimens used for design parameter studies can be prepared as detailed in Reference Test Method G5 for working
electrodes, with the requirement that the metallurgical and surface conditions of the specimens are the same as the intended
implantable medical device.
9.2 Preconditioning can impact the corrosion behavior of implants; therefore, prior to testing, devices should be subjected to
simulated use or other preconditioning that is appropriate to their application. A justification should be provided if simulated use
or other preconditioning is omitted. Additional information on simulated use is found in Appendix X4.
10. Procedure
10.1 Prepare the specimen such that the portion exposed to the test solution is in the same metallurgical and surface condition
as the implantable form of the medical device being studied.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
F2129 − 19a
10.1.1 Calculate the total surface area of the specimen exposed to the solution in order to determineWhere reasonable, calculate
the approximate surface area exposed to solution; where not reasonable, for example, with a porous surface or covered device,
utilize a calculated estimate of surface area exposed to solution and include the reasoning behind that estimate. The exposed surface
area is used to calculate the current density (current per surface area) generated by the specimen during the test.
10.2 Prepare enough test solution to immerse the device and auxiliary electrodes and so avoid any appreciable change in the
solution corrosivity during the test through exhaustion of the corrosive constituents or by accumulation of corrosion products that
may affect further corrosion. At a minimum, transfer 500 mL of electrolyte to a clean polarization cell. Measure and record the
pH of the solution before and after each test.
10.3 Place the auxiliary electrodes, salt bridge probe, thermometer, and gas purge diffuser in the test chamber and bring the
temperature of the test solution to 37 6 1 °C.
10.4 Purge the solution for a minimum of 30 min with nitrogen gas at a flow rate of 150 cm /min.
10.5 Gently immerse the test specimen in the test solution and connect it to a potentiostat. Continue the nitrogen purge
throughout the test.
10.6 Record E for 1 h or, alternatively, until the rest potential stabilizes to a rate of change less than 3 mV/min.
r
10.7 At the end of the E recording period, start the potentiodynamic scan in the positive or noble (forward) direction, as defined
r
in Practice G3. The scanning program should be set with the following parameters:
10.7.1 Starting or initial potential (E ) at E .
i r
10.7.2 A scan rate of either 0.167 mV/s or 1 mV/s should be used. Note that the scan rate may affect the breakdown potential
of the device and the shape of the passive region of the polarization curve. Comparisons should not be made between test results
using different scan rates, even if all other experimental parameters are held constant.
10.7.3 A current density threshold two decades greater than the current density recorded at breakdown can be used to reverse
the voltage scan.
10.7.3.1 Alternatively, a minimum reversing or vertex potential (E ) of 800 mV (SCE) may be used to control the potentiostat
v
(see X1.6).
FIG. 1 Schematic of Cyclic Potentiodynamic Curves Illustrating Corrosion Parameters:
(a) Material That Exhibits a Protection Potential (E ,E , and E ),
r b p
(b) Material That Does Not Exhibit a Protection Potential (E ,E , and E ), and
r b f
(c) Material That Exhibits Oxygen Evolution at Its Surface (E and E ).
r v
F2129 − 19a
10.7.4 The final potential (E ) is set to E . The reverse scan may be manually stopped at potentials above E in cases in which
f r r
a protection potential (E ) is observed as a drop in current density below that of the passive current density or when no hysteresis
p
loop is formed once the scan is reversed (E ), indicating repassivation (Fig. 1a), no protection potential (Fig. 1b), or oxygen
v
evolution (Fig. 1c).
10.8 If control specimens are used, they shall be tested using the same method as the investigated devices.
11. Report
11.1 The report should contain a detailed description of the test specimen, including metallurgical and surface conditioning.
11.1.1 When specimens are not finished devices, for example, surrogates, the sample preparation should be described in detail.
11.2 A description of the test conditions should also be reported, including the following:
11.2.1 The volume of the test cell;
11.2.2 The approximate volume of solution used;
11.2.3 If performed, a description of the preconditioning/simulated use, together with the rationale for the choice of the
preconditioning;
11.2.4 A description of the method used to determine the total estimated exposed surface area of the specimen.
TABLE 1 Precision of Rest Potential E (mV)
r
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Standard Deviation Standard Deviation Limit Limit
316 SS –7 33 64 93 178
455 SS –30 38 67 105 187
Nitinol A –519 35 49 98 137
Nitinol B –482 21 49 60 138
F2129 − 19a
TABLE 2 Precision of Breakdown Potential E (mV)
b
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Standard Deviation Standard Deviation Limit Limit
316 SS 679 161 190 451 531
455 SS 269 36 40 100 113
Nitinol A 160 82 108 230 302
Nitinol B 180 54 94 152 263
TABLE 3 Precision of Repassivation Potential E (mV)
p
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Standard Deviation Standard Deviation Limit Limit
316 SS . . . . .
455 SS . . . . .
Nitinol A –171 57 108 160 302
Nitinol B –126 38 58 107 162
TABLE 4 Precision of Breakdown Potential minus Rest Potential:E — E (mV)
b r
Repeatability Reproducibility 95 % Repeatability 95 % Reproducibility
Material Grand Mean
Stan
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...