ASTM G215-17
(Guide)Standard Guide for Electrode Potential Measurement
Standard Guide for Electrode Potential Measurement
SIGNIFICANCE AND USE
5.1 Electrode potential is the reversible work that is required to transfer a unit of positive charge between the surface in question and a reference electrode through the electrolyte that is in contact with both electrodes. The sign of the electrode potential is determined by the Gibbs Stockholm Convention described in Practice G3.
5.2 The electrode potential of a surface is related to the Gibbs free energy of the oxidation/reduction reactions occurring at the surface in question compared to the Gibbs free energy of the reactions occurring on the reference electrode surface.4
5.3 Electrode potentials are used together with potential-pH (Pourbaix) diagrams to determine the corrosion products that would be in equilibrium with the environment and the electrode surface.5
5.4 Electrode potentials are used in the estimation of corrosion rates by several methods. One example is by means of Tafel line extrapolation, see Practices G3 and G102. Polarization resistance measurements are also determined using electrode potential measurements, see Test Method G59 and Guide G96.
5.5 Corrosion potential measurements are used to determine whether metal surfaces are passive in the environment in question, see Test Method C876.
5.6 Corrosion potential measurements are used in the evaluation of alloys to determine their resistance or susceptibility to various forms of localized corrosion, see Test Methods F746, F2129, G61, and G150.
5.7 Corrosion potentials are used to determine the metallurgical condition of some aluminum alloys, see Test Method G69. Similar measurements have been used with hot dipped galvanized steel to determine their ability to cathodically polarize steel. See Appendix X2.
5.8 Corrosion potentials are used to evaluate aluminum and magnesium alloys as sacrificial anodes for underground and immersion cathodic protection application, see Test Method G97 and NACE TM0190–2012.
5.9 Corrosion potentials are used to evaluate the galvanic performanc...
SCOPE
1.1 This guide provides guidance on the measurement of electrode potentials in laboratory and field studies both for corrosion potentials and polarized potentials.
1.2 The values stated in SI units are to be regarded as standard. Any other units of measurements included in this standard are present because of their wide usage and acceptance.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2016
- Technical Committee
- G01 - Corrosion of Metals
- Drafting Committee
- G01.11 - Electrochemical Measurements in Corrosion Testing
Relations
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2019
- Effective Date
- 01-May-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-May-2018
- Effective Date
- 01-May-2018
- Effective Date
- 01-May-2018
- Effective Date
- 01-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Mar-2015
- Effective Date
- 15-Dec-2014
- Effective Date
- 01-Nov-2014
- Effective Date
- 01-May-2014
Overview
ASTM G215-17: Standard Guide for Electrode Potential Measurement is a key international reference that provides comprehensive guidance for the measurement of electrode potentials in laboratory and field settings. Developed by ASTM International, this standard is essential to the accurate assessment of corrosion behavior and electrochemical characteristics of metals and alloys. The guide addresses both corrosion potentials (open circuit) and polarized potentials, ensuring precise evaluation under various environmental and operational conditions.
Measuring electrode potential is crucial for understanding the reversible work required to transfer positive charge between a test surface and a reference electrode within a shared electrolyte. The standard emphasizes the importance of proper techniques, equipment, and reference electrode selection for reliable electrochemical measurements.
Key Topics
- Electrode Potential Fundamentals
Explains the reversible work associated with charge transfer and its determination according to international conventions (referencing the Gibbs Stockholm Convention and related ASTM practices). - Reference Electrode Selection
Outlines the use of standard (e.g., Standard Hydrogen Electrode, Saturated Calomel Electrode, Silver/Silver Chloride Electrode, Copper/Copper Sulfate Electrode) and nonstandard reference electrodes, including practical selection criteria for diverse environments. - Potential Measurement Procedures
Describes best practices for setting up electrodes, utilizing high impedance voltmeters or electrometers to measure potential differences without affecting the electrodes, and handling both laboratory and field applications. - Sources of Error and Corrections
Details typical sources of measurement error-such as liquid junction potentials, contamination, and environmental effects-and provides guidance on minimizing or correcting these errors. - Maintenance and Calibration
Suggests procedures for storage, maintenance, and periodic verification of reference electrodes to ensure data reliability. - Reporting Requirements
Recommends essential data reporting, including reference electrode type, measurement conditions, and error correction methods associated with electrode potential measurements.
Applications
ASTM G215-17 finds broad practical value across several industries and research fields, notably:
- Corrosion Rate Estimation
Electrode potentials aid in quantifying corrosion rates (using techniques such as Tafel extrapolation and polarization resistance, as referenced in ASTM G3, G59, and G102). - Corrosion Product Prediction
By integrating potential measurements with Pourbaix diagrams, users can predict stable corrosion products under specific environmental conditions. - Material Susceptibility Assessment
Evaluates alloy resistance to localized corrosion forms (pitting, crevice corrosion) and helps in material selection and design. - Cathodic Protection Verification
Assesses the effectiveness of cathodic protection systems for infrastructure such as pipelines, storage tanks, and immersed metal structures. - Hydrogen Embrittlement Testing
Supports the determination of hydrogen overvoltage and related embrittlement phenomena in metals. - Field and Laboratory Use
From in-situ monitoring of reinforced concrete and galvanized steel to qualification of sacrificial anodes, the guide is structured for flexible application.
Related Standards
For comprehensive electrochemical measurement and corrosion evaluation, ASTM G215-17 should be used in conjunction with related ASTM and NACE standards, including:
- ASTM G3: Conventions for Electrochemical Measurements in Corrosion Testing
- ASTM G59: Potentiodynamic Polarization Resistance Measurements
- ASTM G61, G69, G102, G150: Various methods for corrosion susceptibility and rate determination
- ASTM C876: Corrosion Potentials of Reinforcing Steel in Concrete
- ASTM F746, F2129, F3044: Corrosion analysis for medical implants
- NACE TM0190, TM0497, TM0108, TM0113: Standards for cathodic protection and reference electrode evaluation
Keywords: electrode potential measurement, reference electrode, corrosion potential, standard guide, polarization, ASTM G215, calomel electrode, silver/silver chloride electrode, copper sulfate electrode, cathodic protection, corrosion testing, electrochemical measurements
ASTM G215-17 delivers essential procedures and best practices for reliable electrode potential measurements, supporting accurate corrosion assessment, material performance analysis, and improved infrastructure durability.
Buy Documents
ASTM G215-17 - Standard Guide for Electrode Potential Measurement
REDLINE ASTM G215-17 - Standard Guide for Electrode Potential Measurement
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

National Aerospace and Defense Contractors Accreditation Program (NADCAP)
Global cooperative program for special process quality in aerospace.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM G215-17 is a guide published by ASTM International. Its full title is "Standard Guide for Electrode Potential Measurement". This standard covers: SIGNIFICANCE AND USE 5.1 Electrode potential is the reversible work that is required to transfer a unit of positive charge between the surface in question and a reference electrode through the electrolyte that is in contact with both electrodes. The sign of the electrode potential is determined by the Gibbs Stockholm Convention described in Practice G3. 5.2 The electrode potential of a surface is related to the Gibbs free energy of the oxidation/reduction reactions occurring at the surface in question compared to the Gibbs free energy of the reactions occurring on the reference electrode surface.4 5.3 Electrode potentials are used together with potential-pH (Pourbaix) diagrams to determine the corrosion products that would be in equilibrium with the environment and the electrode surface.5 5.4 Electrode potentials are used in the estimation of corrosion rates by several methods. One example is by means of Tafel line extrapolation, see Practices G3 and G102. Polarization resistance measurements are also determined using electrode potential measurements, see Test Method G59 and Guide G96. 5.5 Corrosion potential measurements are used to determine whether metal surfaces are passive in the environment in question, see Test Method C876. 5.6 Corrosion potential measurements are used in the evaluation of alloys to determine their resistance or susceptibility to various forms of localized corrosion, see Test Methods F746, F2129, G61, and G150. 5.7 Corrosion potentials are used to determine the metallurgical condition of some aluminum alloys, see Test Method G69. Similar measurements have been used with hot dipped galvanized steel to determine their ability to cathodically polarize steel. See Appendix X2. 5.8 Corrosion potentials are used to evaluate aluminum and magnesium alloys as sacrificial anodes for underground and immersion cathodic protection application, see Test Method G97 and NACE TM0190–2012. 5.9 Corrosion potentials are used to evaluate the galvanic performanc... SCOPE 1.1 This guide provides guidance on the measurement of electrode potentials in laboratory and field studies both for corrosion potentials and polarized potentials. 1.2 The values stated in SI units are to be regarded as standard. Any other units of measurements included in this standard are present because of their wide usage and acceptance. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 Electrode potential is the reversible work that is required to transfer a unit of positive charge between the surface in question and a reference electrode through the electrolyte that is in contact with both electrodes. The sign of the electrode potential is determined by the Gibbs Stockholm Convention described in Practice G3. 5.2 The electrode potential of a surface is related to the Gibbs free energy of the oxidation/reduction reactions occurring at the surface in question compared to the Gibbs free energy of the reactions occurring on the reference electrode surface.4 5.3 Electrode potentials are used together with potential-pH (Pourbaix) diagrams to determine the corrosion products that would be in equilibrium with the environment and the electrode surface.5 5.4 Electrode potentials are used in the estimation of corrosion rates by several methods. One example is by means of Tafel line extrapolation, see Practices G3 and G102. Polarization resistance measurements are also determined using electrode potential measurements, see Test Method G59 and Guide G96. 5.5 Corrosion potential measurements are used to determine whether metal surfaces are passive in the environment in question, see Test Method C876. 5.6 Corrosion potential measurements are used in the evaluation of alloys to determine their resistance or susceptibility to various forms of localized corrosion, see Test Methods F746, F2129, G61, and G150. 5.7 Corrosion potentials are used to determine the metallurgical condition of some aluminum alloys, see Test Method G69. Similar measurements have been used with hot dipped galvanized steel to determine their ability to cathodically polarize steel. See Appendix X2. 5.8 Corrosion potentials are used to evaluate aluminum and magnesium alloys as sacrificial anodes for underground and immersion cathodic protection application, see Test Method G97 and NACE TM0190–2012. 5.9 Corrosion potentials are used to evaluate the galvanic performanc... SCOPE 1.1 This guide provides guidance on the measurement of electrode potentials in laboratory and field studies both for corrosion potentials and polarized potentials. 1.2 The values stated in SI units are to be regarded as standard. Any other units of measurements included in this standard are present because of their wide usage and acceptance. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM G215-17 is classified under the following ICS (International Classification for Standards) categories: 25.220.99 - Other treatments and coatings; 77.060 - Corrosion of metals. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM G215-17 has the following relationships with other standards: It is inter standard links to ASTM G215-16, ASTM G69-20, ASTM G71-81(2019), ASTM G3-14(2019), ASTM F2129-19, ASTM G150-18, ASTM G96-90(2018), ASTM G61-86(2018), ASTM F2129-17b, ASTM F2129-17a, ASTM F2129-17, ASTM F2129-15, ASTM G3-14, ASTM G5-14, ASTM G71-81(2014). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM G215-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: G215 − 17
Standard Guide for
Electrode Potential Measurement
This standard is issued under the fixed designation G215; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope ceptibility of Iron-, Nickel-, or Cobalt-Based Alloys
G69Test Method for Measurement of Corrosion Potentials
1.1 This guide provides guidance on the measurement of
of Aluminum Alloys
electrode potentials in laboratory and field studies both for
G71Guide for Conducting and Evaluating Galvanic Corro-
corrosion potentials and polarized potentials.
sion Tests in Electrolytes
1.2 The values stated in SI units are to be regarded as
G82Guide for Development and Use of a Galvanic Series
standard. Any other units of measurements included in this
for Predicting Galvanic Corrosion Performance
standard are present because of their wide usage and accep-
G96Guide for Online Monitoring of Corrosion in Plant
tance.
Equipment (Electrical and Electrochemical Methods)
1.3 This standard does not purport to address all of the G97Test Method for Laboratory Evaluation of Magnesium
safety concerns, if any, associated with its use. It is the
SacrificialAnodeTest Specimens for UndergroundAppli-
responsibility of the user of this standard to establish appro- cations
priate safety and health practices and determine the applica-
G102Practice for Calculation of Corrosion Rates and Re-
bility of regulatory limitations prior to use. lated Information from Electrochemical Measurements
G106Practice for Verification of Algorithm and Equipment
2. Referenced Documents
for Electrochemical Impedance Measurements
G150Test Method for Electrochemical Critical PittingTem-
2.1 ASTM Standards:
perature Testing of Stainless Steels
C876Test Method for Corrosion Potentials of Uncoated
G193Terminology and Acronyms Relating to Corrosion
Reinforcing Steel in Concrete
2.2 NACE Standards:
F746Test Method for Pitting or Crevice Corrosion of
TM0497–2012Measurement Techniques Related to Criteria
Metallic Surgical Implant Materials
for Cathodic Protection on Underground or Submerged
F2129Test Method for Conducting Cyclic Potentiodynamic
Metallic Piping Systems
Polarization Measurements to Determine the Corrosion
TM0101–2012Measurement Techniques Related to Criteria
Susceptibility of Small Implant Devices
for Cathodic Protection of Underground Storage Tank
F3044Test Method for Test Method for Evaluating the
Systems
Potential for Galvanic Corrosion for Medical Implants
TM0108–2012Testing of Catalyzed Titanium Anodes for
G3Practice for Conventions Applicable to Electrochemical
Use in Soils or Natural Waters
Measurements in Corrosion Testing
TM0109–2009Aboveground Survey Techniques for the
G5Reference Test Method for Making Potentiodynamic
Evaluation of Underground Pipeline Coating Condition
Anodic Polarization Measurements
TM0190–2012Impressed Current Laboratory Testing of
G59TestMethodforConductingPotentiodynamicPolariza-
Aluminum Alloy Anodes
tion Resistance Measurements
TM0211–2011Durability Test for Copper/Copper Sulfate
G61Test Method for Conducting Cyclic Potentiodynamic
Permanent Reference Electrodes for Direct Burial Appli-
Polarization Measurements for Localized Corrosion Sus-
cations
TM0113–2013Evaluating theAccuracy of Field Grade Ref-
erence Electrode
This guide is under the jurisdiction ofASTM Committee G01 on Corrosion of
Metals and is the direct responsibility of Subcommittee G01.11 on Electrochemical
3. Terminology
Measurements in Corrosion Testing.
Current edition approved Jan. 1, 2017. Published January 2017. Originally
3.1 Definitions—The terminology used herein shall be in
approved in 2016. Last previous edition approved in 2016 as G215–16. DOI:
accordance with Terminology G193.
10.1520/G0215-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on AvailablefromNACEInternational(NACE),15835ParkTenPl.,Houston,TX
the ASTM website. 77084, http://www.nace.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G215 − 17
4. Summary of Practice systems and to confirm the performance of these systems in
soils,concrete,andnaturalwaters,seeNACETM0497,NACE
4.1 Electrode potential measurements are made by electri-
TM0108, and NACE TM0109.
cally connecting a high impedance voltmeter or electrometer
between the specimen electrode and a suitable reference 5.11 Electrodepotentialmeasurementsarenecessaryforthe
half-cell electrode. See Practice G3. determination of hydrogen overvoltage values in testing for
hydrogen embrittlement and related issues with hydrogen
5. Significance and Use
cracking. See Appendix X3.
5.1 Electrodepotentialisthereversibleworkthatisrequired
6. Potential Measurement
to transfer a unit of positive charge between the surface in
6.1 Electrodepotentialsaremeasuredbyplacingareference
question and a reference electrode through the electrolyte that
is in contact with both electrodes. The sign of the electrode electrode in the corrosive electrolyte and electrically connect-
ing a high impedance potential measuring instrument, such as
potential is determined by the Gibbs Stockholm Convention
described in Practice G3. an electrometer, potentiometer, or high impedance voltmeter,
betweenthereferenceelectrodeandtheobjectwiththesurface
5.2 The electrode potential of a surface is related to the
inquestion.Themeasuringinstrumentmustbeabletomeasure
Gibbs free energy of the oxidation/reduction reactions occur-
thepotentialdifferencewithoutaffectingeitherelectrodetoany
ring at the surface in question compared to the Gibbs free
significant degree. In general, devices with input impedances
energy of the reactions occurring on the reference electrode
4 greater than 10 ohms have been found to be acceptable in
surface.
most corrosion related measurements. In cases where the
5.3 Electrode potentials are used together with potential-pH
specimen is polarized by an external power source, it may be
(Pourbaix) diagrams to determine the corrosion products that
desirable to connect the potential measuring instrument di-
would be in equilibrium with the environment and the elec-
rectly to the specimen rather than using the conductor carrying
trode surface.
the polarizing current to the specimen.
5.4 Electrode potentials are used in the estimation of corro-
NOTE 1—When using a potential measuring instrument such as a high
sion rates by several methods. One example is by means of
impedance voltmeter, the reference electrode should be connected to the
Tafel line extrapolation, see Practices G3 and G102. Polariza- negativeorground(black)terminalinordertohavetheinstrumentrecord
the proper sign of the reading in accordance with Practice G3. However,
tion resistance measurements are also determined using elec-
for instruments that read only positive potentials, it may be necessary to
trodepotentialmeasurements,seeTestMethodG59andGuide
reverse these connections to obtain the reading.
G96.
6.2 Two types of reference electrodes have been used in
5.5 Corrosionpotentialmeasurementsareusedtodetermine
corrosion testing: standard reference electrodes and nonstan-
whether metal surfaces are passive in the environment in
dard reference electrodes.
question, see Test Method C876.
6.2.1 Standard reference electrodes are widely used and
5.6 Corrosionpotentialmeasurementsareusedintheevalu-
they provide a known half-cell potential value versus the
ation of alloys to determine their resistance or susceptibility to standard hydrogen electrode, SHE, half-cell. These electrodes
various forms of localized corrosion, see Test Methods F746,
are stable, and in most cases commercially available. It is
F2129, G61, and G150. possible also to construct them using known techniques.
6.2.2 Nonstandard reference electrodes are used in cases
5.7 Corrosion potentials are used to determine the metallur-
where it is not necessary to know the actual value of the
gical condition of some aluminum alloys, see Test Method
potential with reference to a chemical reaction, but it is
G69. Similar measurements have been used with hot dipped
important to know how the potential has changed as a surface
galvanized steel to determine their ability to cathodically
is polarized or when environmental changes occur. These
polarize steel. See Appendix X2.
nonstandard reference electrodes should be stable with time,
5.8 Corrosion potentials are used to evaluate aluminum and
and they should not be significantly affected by the measuring
magnesium alloys as sacrificial anodes for underground and
process. Guide G96 provides information on nonstandard
immersion cathodic protection application, see Test Method
reference electrodes used in polarization resistance measure-
G97 and NACE TM0190–2012.
ments. In some cases the nonstandard reference electrode is
5.9 Corrosion potentials are used to evaluate the galvanic
identical with the test electrode. In these cases a drift in the
performance of alloy pairs for use in seawater and other potential with time is acceptable as long as both the test and
conductive electrolytes, see Test Method F3044, Guide G71, reference electrodes experience the same drift.
and Guide G82. 6.2.3 In some cases nonstandard reference electrodes are
used because the environmental conditions are not suitable for
5.10 Electrode potential measurements are used to establish
standard reference electrodes. Pure zinc and zinc alloy (UNS
cathodic protection levels to troubleshoot cathodic protection
Z12001, and Z12002, or Z14002) reference electrodes have
been used in seawater and similar aqueous solutions although
4 nd
Moore, Walter J. Physical Chemistry,2 Edition, Prentice Hall, Englewood
Cliffs, NJ, 1955.
5 6
Pourbaix, Marcel, Atlas of Electrochemical Equilibria in Aqueous Solutions, Ives, David J. G. and Janz, George, J., Reference Electrodes Theory and
NACE International, Houston, TX, 1974. Practice, Academic Press, New York, NY, 1961.
G215 − 17
theyhavebeenobservedtohavesignificantpotentialdriftwith gas at a pressure of 101.3 kPa (1 atm) and 25°C. Although
exposure. The potentials of these electrodes are determined by theseelectrodeshavebeenusedextensivelyinelectrochemical
the corrosion potential of metal in the seawater. For pure zinc, studies to determine the thermodynamic properties of ions,
the potential versus SHE is approximately -0.78 V, while for they are almost never used in corrosion studies. However, this
the zinc alloys, the potential is approximately -0.8 V. In some
electrode is the reference point for all other standard reference
cases the corrosion potential of the zinc electrode has been electrodes.
measuredagainstastandardelectrodeinaknownenvironment
7.1.2 Saturated Calomel Electrode—This electrode, desig-
before and after usage to obtain a measure of the drift that
nated SCE, has been the most widely used standard reference
occurred.
electrode for corrosion studies.The reason for its popularity is
that it has been used in commercial electrometric pH meters,
7. Standard Reference Electrodes
and consequently it has been easily available and is very
7.1 Standard reference electrodes are based on having the
reproducible. The SCE is based on the following reactions:
primary electrochemical reaction occurring on the electrode
2Hg 5Hg 12e (3)
surface at equilibrium. This implies that both the forward and
11 2
Hg 12Cl 5Hg Cl (4)
reverse reactions are occurring at the same rate. In the general 2 2 2
The compound, Hg Cl , mercurous chloride, is also known
2 2
case, the electrochemical reaction can be expressed as shown
as calomel, and that is the reason for the electrode’s designa-
in Eq 1:
tion. The mercury/mercurous chloride mixture is immersed
n1
Me 5Me 1ne (1) in a saturated potassium chloride solution so that the mercu-
Where Me represents a metal with a valence of n, and e rep- rous ion concentration is determined by its solubility at that
resents an electron. The potential of this reaction is shown in chloride level. This electrode has been designated a second
Eq 2: kind electrode. See Table 1 for information on the potential
of this electrode. Although these standard reference elec-
0 21 n1
E 5E 10.0592 T 1 273.2 n298.2 log Me (2)
~ !~ ! @ #
trodes have been widely used for many laboratory corrosion
tests including Test Methods G5, G59, and others, their use
where:
may be restricted because of bans on mercury and its com-
E = the electrode potential of the half-cell V,
pounds.
E = the electrode potential of the reaction at unit
NOTE 3—The term “saturated” when used to describe standard refer-
activity, V,
n+
ence electrodes refers to the metal ion concentration, not the anion.
[Me ] = activity of the Me ion,
n = thenumberofelectronstransferredinthereaction,
7.1.3 Saturated Silver/Silver Chloride Electrode—There are
and
four silver/silver chloride electrodes, saturated with respect to
T = electrode temperature, °C.
the silver ion concentration, that have been used as standard
reference electrodes. All of these electrodes are based on
NOTE 2—The activity of an ion is equal to the concentration of the ion
multiplied by its activity coefficient. reactions (5) and (6) below:
7.1.1 Standard Hydrogen Electrode—The standard hydro-
Ag 5Ag 1e (5)
gen electrode, SHE, is a first kind standard reference elec-
1 2
Ag 1Cl 5AgCl (6)
trode. This electrode is composed of a platinized platinum
Because silver chloride is slightly soluble, the silver ion con-
electrode immersed in an acid solution with a hydrogen ion
centration is based on the chloride concentration. The silver/
activityof1(approximately1N)andincontactwithhydrogen silver chloride combination is immersed in KCl solutions of
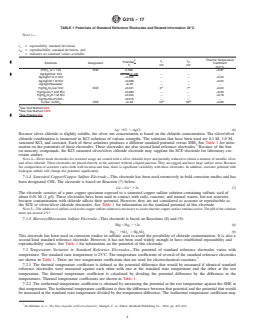
TABLE 1 Potentials of Standard Reference Electrodes and Related Information 25°C
NOTE 1—
s = repeatability standard deviation,
r
s = reproducibility standard deviation, and
R
– = indicates no standard values available.
Thermal Temperature
C
Potential s s
r R
Electrode Designation Coefficient
V
mV mV
mV/°C
(Pt)H (a = 1.0) SHE 0.000 +0.87
Ag/AgCl/sat. KCl +0.196 – – –
Ag/AgCl/1.0 m KCl +0.235 – – +0.25
Ag/AgCl/0.1 M KCl +0.288 – – +0.22
Ag/AgCl/Seawater +0.25 – – –
A A
Hg/Hg Cl /sat. KCl SCE +0.241 3 7 +0.22
2 2
Hg/Hg Cl /1.0 M KCl +0.280 – – +0.59
2 2
Hg/Hg Cl /0.1 M KCl +0.334 – – +0.79
2 2
Hg/Hg SO /H SO +0.616 – – –
2 4 2 4
B B
Cu/sat. CuSO CSE +0.30 10 30 +0.90
A
See Test Method G69.
B
See Test Method C876.
C
See Practice G3.
G215 − 17
various strengths. The solutions that have been used are 0.1
7.2.2 The isothermal temperature coefficient is obtained by
M, 1.0 M, saturated KCl, and seawater. Each of these solu-
measuringthepotentialatthetesttemperatureagainsttheSHE
tions produces a different standard potential versus SHE. See
at that temperature. The isothermal temperature coefficient is
Table 1 for information on the potentials of these electrodes.
5 thenthedifferencebetweenthatpotentialandthepotentialthat
These electrodes are also second kind reference electrodes.
wouldbemeasuredatthestandardstatetemperaturedividedby
Because of the ban on mercury compounds, the KCl satu-
the temperature difference. The isothermal temperature coeffi-
rated silver/silver chloride electrode may supplant the SCE
electrode for laboratory corrosion studies. cient may be calculated by subtracting 0.87 mV from the
NOTE 4—Silver mesh electrodes for seawater usage are coated with a
thermal temperature coefficient. Isothermal temperature coef-
silver chloride layer and partially reduced to obtain a mixture of metallic
ficients can be used in predicting the thermodynamic stability
silver and silver chloride. These electrodes are placed directly in the
of corrosion products at temperatures different from 25°C.
seawaterwithoutaliquidjunction.Theyareruggedandhavelargesurface
7.2.3 In many cases involving laboratory testing, a salt
areas. Because the composition of seawater varies both with location and
time, there is significant variability with these electrodes. In addition, bridgeisusedtoconnectthestandardreferenceelectrodetothe
seawater polluted with hydrogen sulfide will change the potential signifi-
test environment. In these cases the standard reference elec-
cantly.
trode may be at a different temperature than the test tempera-
7.1.4 Saturated Copper/Copper Sulfate Electrode—This
ture.Inthesecases,thetemperaturecorrectionshouldbemade
electrode has been used extensively in field corrosion studies
based on the temperature of the standard reference electrode.
and has been designated CSE. The electrode is based on
See Appendix X1 for a sample calculation.
Reaction (7) below:
8. Standard Reference Electrodes—Errors, Issues, and
Cu 5Cu 12e (7)
Corrections
The electrode consists of a pure copper specimen exposed to
a saturated copper sulfate solution containing sulfuric acid of
8.1 Liquid Junction Potentials—Because standard reference
about 0.01 M (1 g/l). These electrodes have been used in
electrodes have a specific environment that is required to
contact with soils, concrete, and natural waters, but not
achieve their standard potential value, it is necessary to have a
seawater, because contamination with chloride affects their
liquid junction between the standard reference electrode solu-
potential. However, they are not considered as accurate or
tion and the test environment solution. This liquid junction
reproducible as the SCE or silver/silver chloride electrodes.
See Table 1 for information on the standard potential of th
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: G215 − 16 G215 − 17
Standard Guide for
Electrode Potential Measurement
This standard is issued under the fixed designation G215; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide provides guidance on the measurement of electrode potentials in laboratory and field studies both for corrosion
potentials and polarized potentials.
1.2 The values stated in SI units are to be regarded as standard. Any other units of measurements included in this standard are
present because of their wide usage and acceptance.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
C876 Test Method for Corrosion Potentials of Uncoated Reinforcing Steel in Concrete
F746 Test Method for Pitting or Crevice Corrosion of Metallic Surgical Implant Materials
F2129 Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements to Determine the Corrosion Suscepti-
bility of Small Implant Devices
F3044 Test Method for Test Method for Evaluating the Potential for Galvanic Corrosion for Medical Implants
G3 Practice for Conventions Applicable to Electrochemical Measurements in Corrosion Testing
G5 Reference Test Method for Making Potentiodynamic Anodic Polarization Measurements
G59 Test Method for Conducting Potentiodynamic Polarization Resistance Measurements
G61 Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility of
Iron-, Nickel-, or Cobalt-Based Alloys
G69 Test Method for Measurement of Corrosion Potentials of Aluminum Alloys
G71 Guide for Conducting and Evaluating Galvanic Corrosion Tests in Electrolytes
G82 Guide for Development and Use of a Galvanic Series for Predicting Galvanic Corrosion Performance
G96 Guide for Online Monitoring of Corrosion in Plant Equipment (Electrical and Electrochemical Methods)
G97 Test Method for Laboratory Evaluation of Magnesium Sacrificial Anode Test Specimens for Underground Applications
G102 Practice for Calculation of Corrosion Rates and Related Information from Electrochemical Measurements
G106 Practice for Verification of Algorithm and Equipment for Electrochemical Impedance Measurements
G150 Test Method for Electrochemical Critical Pitting Temperature Testing of Stainless Steels
G193 Terminology and Acronyms Relating to Corrosion
2.2 NACE Standards:
TM0497–2012 Measurement Techniques Related to Criteria for Cathodic Protection on Underground or Submerged Metallic
Piping Systems
TM0101–2012 Measurement Techniques Related to Criteria for Cathodic Protection of Underground Storage Tank Systems
TM0108–2012 Testing of Catalyzed Titanium Anodes for Use in Soils or Natural Waters
TM0109–2009 Aboveground Survey Techniques for the Evaluation of Underground Pipeline Coating Condition
TM0190–2012 Impressed Current Laboratory Testing of Aluminum Alloy Anodes
This guide is under the jurisdiction of ASTM Committee G01 on Corrosion of Metals and is the direct responsibility of Subcommittee G01.11 on Electrochemical
Measurements in Corrosion Testing.
Current edition approved May 1, 2016Jan. 1, 2017. Published May 2016January 2017. Originally approved in 2016. Last previous edition approved in 2016 as G215 – 16.
DOI: 10.1520/G0215-16.10.1520/G0215-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from NACE International (NACE), 15835 Park Ten Pl., Houston, TX 77084, http://www.nace.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G215 − 17
TM0211–2011 Durability Test for Copper/Copper Sulfate Permanent Reference Electrodes for Direct Burial Applications
TM0113–2013 Evaluating the Accuracy of Field Grade Reference Electrode
3. Terminology
3.1 Definitions—The terminology used herein shall be in accordance with Terminology G193.
4. Summary of Practice
4.1 Electrode potential measurements are made by electrically connecting a high impedance voltmeter or electrometer between
the specimen electrode and a suitable reference half-cell electrode. See Practice G3.
5. Significance and Use
5.1 Electrode potential is the reversible work that is required to transfer a unit of positive charge between the surface in question
and a reference electrode through the electrolyte that is in contact with both electrodes. The sign of the electrode potential is
determined by the Gibbs Stockholm Convention described in Practice G3.
5.2 The electrode potential of a surface is related to the Gibbs free energy of the oxidation/reduction reactions occurring at the
surface in question compared to the Gibbs free energy of the reactions occurring on the reference electrode surface.
5.3 Electrode potentials are used together with potential-pH (Pourbaix) diagrams to determine the corrosion products that would
be in equilibrium with the environment and the electrode surface.
5.4 Electrode potentials are used in the estimation of corrosion rates by several methods. One example is by means of Tafel line
extrapolation, see Practices G3 and G102. Polarization resistance measurements are also determined using electrode potential
measurements, see Test Method G59 and Guide G96.
5.5 Corrosion potential measurements are used to determine whether metal surfaces are passive in the environment in question,
see Test Method C876.
5.6 Corrosion potential measurements are used in the evaluation of alloys to determine their resistance or susceptibility to
various forms of localized corrosion, see Test Methods F746, F2129, G61, and G150.
5.7 Corrosion potentials are used to determine the metallurgical condition of some aluminum alloys, see Test Method G69.
Similar measurements have been used with hot dipped galvanized steel to determine their ability to cathodically polarize steel. See
Appendix X2.
5.8 Corrosion potentials are used to evaluate aluminum and magnesium alloys as sacrificial anodes for underground and
immersion cathodic protection application, see Test Method G97 and NACE TM0190–2012.
5.9 Corrosion potentials are used to evaluate the galvanic performance of alloy pairs for use in seawater and other conductive
electrolytes, see Test Method F3044, Guide G71, and Guide G82.
5.10 Electrode potential measurements are used to establish cathodic protection levels to troubleshoot cathodic protection
systems and to confirm the performance of these systems in soils, concrete, and natural waters, see NACE TM0497, NACE
TM0108, and NACE TM0109.
5.11 Electrode potential measurements are necessary for the determination of hydrogen overvoltage values in testing for
hydrogen embrittlement and related issues with hydrogen cracking. See Appendix X3.
6. Potential Measurement
6.1 Electrode potentials are measured by placing a reference electrode in the corrosive electrolyte and electrically connecting
a high impedance potential measuring instrument, such as an electrometer, potentiometer, or high impedance voltmeter, between
the reference electrode and the object with the surface in question. The measuring instrument must be able to measure the potential
difference without affecting either electrode to any significant degree. In general, devices with input impedances greater than 10
ohms have been found to be acceptable in most corrosion related measurements. In cases where the specimen is polarized by an
external power source, it may be desirable to connect the potential measuring instrument directly to the specimen rather than using
the conductor carrying the polarizing current to the specimen.
NOTE 1—When using a potential measuring instrument such as a high impedance voltmeter, the reference electrode should be connected to the negative
or ground (black) terminal in order to have the instrument record the proper sign of the reading in accordance with Practice G3. However, for instruments
that read only positive potentials, it may be necessary to reverse these connections to obtain the reading.
6.2 Two types of reference electrodes have been used in corrosion testing: standard reference electrodes and nonstandard
reference electrodes.
nd
Moore, Walter J. Physical Chemistry, 2 Edition, Prentice Hall, Englewood Cliffs, NJ, 1955.
Pourbaix, Marcel, Atlas of Electrochemical Equilibria in Aqueous Solutions, NACE International, Houston, TX, 1974.
G215 − 17
6.2.1 Standard reference electrodes are widely used and they provide a known half-cell potential value versus the standard
hydrogen electrode, SHE, half-cell. These electrodes are stable, and in most cases commercially available. It is possible also to
construct them using known techniques.
6.2.2 Nonstandard reference electrodes are used in cases where it is not necessary to know the actual value of the potential with
reference to a chemical reaction, but it is important to know how the potential has changed as a surface is polarized or when
environmental changes occur. These nonstandard reference electrodes should be stable with time, and they should not be
significantly affected by the measuring process. Guide G96 provides information on nonstandard reference electrodes used in
polarization resistance measurements. In some cases the nonstandard reference electrode is identical with the test electrode. In
these cases a drift in the potential with time is acceptable as long as both the test and reference electrodes experience the same
drift.
6.2.3 In some cases nonstandard reference electrodes are used because the environmental conditions are not suitable for
standard reference electrodes. Pure zinc and zinc alloy (UNS Z12001, and Z12002, or Z14002) reference electrodes have been used
in seawater and similar aqueous solutions although they have been observed to have significant potential drift with exposure. The
potentials of these electrodes are determined by the corrosion potential of metal in the seawater. For pure zinc, the potential versus
SHE is approximately -0.78 V, while for the zinc alloys, the potential is approximately -0.8 V. In some cases the corrosion potential
of the zinc electrode has been measured against a standard electrode in a known environment before and after usage to obtain a
measure of the drift that occurred.
7. Standard Reference Electrodes
7.1 Standard reference electrodes are based on having the primary electrochemical reaction occurring on the electrode surface
at equilibrium. This implies that both the forward and reverse reactions are occurring at the same rate. In the general case, the
electrochemical reaction can be expressed as shown in Eq 1:
n1
Me 5 Me 1ne (1)
Where Me represents a metal with a valence of n, and e represents an electron. The potential of this reaction is shown in Eq
2:
0 21 n1
E5 E 10.0592 T 1 273.2 n298.2 log@ Me # (2)
~ !~ !
where:
E = the electrode potential of the half-cell V,
E = the electrode potential of the reaction at unit activity, V,
n+
[Me ] = activity of the Me ion,
n = the number of electrons transferred in the reaction, and
T = electrode temperature, °C.
NOTE 2—The activity of an ion is equal to the concentration of the ion multiplied by its activity coefficient.
7.1.1 Standard Hydrogen Electrode—The standard hydrogen electrode, SHE, is a first kind standard reference electrode. This
electrode is composed of a platinized platinum electrode immersed in an acid solution with a hydrogen ion activity of 1
(approximately 1 N) and in contact with hydrogen gas at a pressure of 101.3 kPa (1 atm) and 25°C. Although these electrodes have
been used extensively in electrochemical studies to determine the thermodynamic properties of ions, they are almost never used
in corrosion studies. However, this electrode is the reference point for all other standard reference electrodes.
7.1.2 Saturated Calomel Electrode—This electrode, designated SCE, has been the most widely used standard reference
electrode for corrosion studies. The reason for its popularity is that it has been used in commercial electrometric pH meters, and
consequently it has been easily available and is very reproducible. The SCE is based on the following reactions:
2Hg 5 Hg 12e (3)
11 2
Hg 12Cl 5 Hg Cl (4)
2 2 2
The compound, Hg Cl , mercurous chloride, is also known as calomel, and that is the reason for the electrode’s designation.
2 2
The mercury/mercurous chloride mixture is immersed in a saturated potassium chloride solution so that the mercurous ion
concentration is determined by its solubility at that chloride level. This electrode has been designated a second kind elec-
trode. See Table 1 for information on the potential of this electrode. Although these standard reference electrodes have been
widely used for many laboratory corrosion tests including Test Methods G5, G59, and others, their use may be restricted be-
cause of bans on mercury and its compounds.
NOTE 3—The term “saturated” when used to describe standard reference electrodes refers to the metal ion concentration, not the anion.
7.1.3 Saturated Silver/Silver Chloride Electrode—There are four silver/silver chloride electrodes, saturated with respect to the
silver ion concentration, that have been used as standard reference electrodes. All of these electrodes are based on reactions (5)
and (6) below:
Ag 5 Ag 1e (5)
Ives, David J. G. and Janz, George, J., Reference Electrodes Theory and Practice, Academic Press, New York, NY, 1961.
G215 − 17
TABLE 1 Potentials of Standard Reference Electrodes and Related Information 25°C
NOTE 1—
s = repeatability standard deviation,
r
s = reproducibility standard deviation, and
R
– = indicates no standard values available.
Thermal Temperature
C
s s
Potential
r R
Electrode Designation Coefficient
V
mV mV
mV/°C
(Pt)H (a = 1.0) SHE 0.000 +0.87
Ag/AgCl/sat. KCl +0.194+0.196 – – –
Ag/AgCl/1.0 m KCl +0.235 – – +0.25
Ag/AgCl/0.1 M KCl +0.288 – – +0.22
Ag/AgCl/Seawater +0.25 – – –
A A
Hg/Hg Cl /sat. KCl SCE +0.241 3 7 +0.22
2 2
Hg/Hg Cl /1.0 M KCl +0.280 – – +0.59
2 2
Hg/Hg Cl /0.1 M KCl +0.334 – – +0.79
2 2
Hg/Hg SO /H SO +0.616 – – –
2 4 2 4
B B
Cu/sat. CuSO CSE +0.30 10 30 +0.90
A
See Test Method G69.
B
See Test Method C876.
C
See Practice G3.
1 2
Ag 1Cl 5 AgCl (6)
Because silver chloride is slightly soluble, the silver ion concentration is based on the chloride concentration. The silver/silver
chloride combination is immersed in KCl solutions of various strengths. The solutions that have been used are 0.1 M, 1.0 M,
saturated KCl, and seawater. Each of these solutions produces a different standard potential versus SHE. See Table 1 for infor-
mation on the potentials of these electrodes. These electrodes are also second kind reference electrodes. Because of the ban
on mercury compounds, the KCl saturated silver/silver chloride electrode may supplant the SCE electrode for laboratory cor-
rosion studies.
NOTE 4—Silver mesh electrodes for seawater usage are coated with a silver chloride layer and partially reduced to obtain a mixture of metallic silver
and silver chloride. These electrodes are placed directly in the seawater without a liquid junction. They are rugged and have large surface areas. Because
the composition of seawater varies both with location and time, there is significant variability with these electrodes. In addition, seawater polluted with
hydrogen sulfide will change the potential significantly.
7.1.4 Saturated Copper/Copper Sulfate Electrode—This electrode has been used extensively in field corrosion studies and has
been designated CSE. The electrode is based on Reaction (7) below:
Cu 5 Cu 12e (7)
The electrode consists of a pure copper specimen exposed to a saturated copper sulfate solution containing sulfuric acid of
about 0.01 M (1 g/l). These electrodes have been used in contact with soils, concrete, and natural waters, but not seawater,
because contamination with chloride affects their potential. However, they are not considered as accurate or reproducible as
the SCE or silver/silver chloride electrodes. See Table 1 for information on the standard potential of this electrode.
NOTE 5—The addition of sulfuric acid to the copper sulfate solution is necessary to assure that the copper surface remains active. The pH of the solution
must not exceed 2.9.
7.1.5 Mercury/Mercurous Sulfate Electrode—This electrode is based on Reactions (8) and (9):
2Hg 5 Hg 12e (8)
11 2
Hg 1SO 5 Hg SO (9)
2 4 2 4
This electrode has been used in corrosion studies in sulfuric acid to avoid the possibility of chloride contamination. It is also a
second kind standard reference electrode. However, it has not been used widely enough to have established repeatability and
reproducibility values. See Table 1 for information on the potential of this electrode.
7.2 Temperature Variation in Standard Reference Electrodes—The potential of standard reference electrodes varies with
temperature. The standard state temperature is 25°C. The temperature coefficients of several of the standard reference electrodes
are shown in Table 1. There are two temperature coefficients that are used for electrochemical reactions.
7.2.1 The thermal temperature coefficient is defined as the potential difference that would be measured if identical standard
reference electrodes were measured against each other with one at the standard state temperature and the other at the test
temperature. The thermal temperature coefficient is calculated by dividing the potential difference by the differ
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...