ASTM F2475-20
(Guide)Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials
Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials
SIGNIFICANCE AND USE
5.1 The compatibility of packaging materials with a medical device is a requirement of many regulatory bodies. Since most medical devices are used or implanted in, around, or on the human body, the benefits of these devices must outweigh the risks. Therefore, the packaging materials that come in contact with the medical device must also be evaluated and determined to be safe for use with the human body in that they have no negative impact on the physical, chemical, or biological properties of the device. This evaluation may include both a study of relevant experience with, and actual testing of, packaging materials. Such an evaluation may result in the conclusion that no testing is needed if the material has a demonstrable history of safe use in the specific role that is the same as that of the package under design.
5.2 The medical device manufacturer determines the need for appropriate testing, with consideration of the device/package interactions, if any. When screening information is needed regarding the biocompatibility of the packaging, cytotoxicity testing from the supplier is typically performed.
SCOPE
1.1 This guide provides information to determine the appropriate testing for biocompatibility of medical device packaging materials that have the potential to contact the patient directly or indirectly.
1.2 This guide does not apply to secondary or tertiary packaging materials.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2019
- Technical Committee
- F02 - Primary Barrier Packaging
- Drafting Committee
- F02.15 - Chemical/Safety Properties
Relations
- Replaces
ASTM F2475-11 - Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials - Effective Date
- 01-Jan-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Oct-2018
- Effective Date
- 15-Aug-2018
- Effective Date
- 01-Jun-2017
- Effective Date
- 01-Aug-2013
- Effective Date
- 15-Apr-2013
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-Aug-2008
- Effective Date
- 01-Sep-2007
- Effective Date
- 01-May-2007
- Effective Date
- 01-Dec-2006
- Effective Date
- 10-Oct-2002
- Effective Date
- 10-Oct-1998
- Referred By
ASTM F2559/F2559M-21 - Standard Guide for Writing a Specification for Sterilizable Peel Pouches - Effective Date
- 01-Jan-2020
Overview
ASTM F2475-20: Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials provides a systematic approach for evaluating whether packaging materials used with medical devices are biocompatible. As medical devices often come into direct or indirect contact with the human body, it is essential that their packaging does not adversely impact their safety or performance. This standard helps manufacturers determine what level of testing or justification is needed to ensure that packaging materials are safe and suitable for use with medical devices.
This guide is particularly important for regulatory compliance, as many global regulatory bodies require evidence that all components in contact with medical devices, including packaging, are biocompatible. The standard applies specifically to materials in sterile barrier systems that may contact the medical device itself, either directly or indirectly, but excludes secondary and tertiary packaging.
Key Topics
- Biocompatibility of Packaging Materials: Evaluates if materials used in medical device packaging interact chemically, physically, or biologically with the device in ways that might affect patient safety.
- Risk Assessment: Encourages a risk-based approach, considering historical use, material properties, and extent of contact between packaging and device to determine if testing is required or can be justified based on prior safe use.
- Testing Recommendations: Advises on appropriate biocompatibility tests, such as cytotoxicity, irritation, sensitization, and chemical analysis (when necessary), following guidance from standards like ISO 10993-1 and FDA guidelines.
- Documentation and Traceability: Stresses the importance of documenting material details, manufacturing processes, and any secondary treatments or additives, which can influence biocompatibility.
- Material and Device Interactions: Accounts for the degree and nature of contact, highlighting that testing needs may vary depending on whether the device is solid, semi-solid, or liquid, and the likelihood for material migration or chemical interaction.
Applications
- Medical Device Sterile Barrier Systems: Ensures that primary packaging used for sterilized medical devices does not compromise the sterility or performance of the product.
- Regulatory Submissions: Provides a framework for manufacturers to demonstrate compliance with FDA, EU MDR, and other regulatory requirements regarding packaging safety and biocompatibility.
- Risk Management in Manufacturing: Supports manufacturers in implementing risk-based controls, helping to avoid unnecessary testing when safe-use history exists, or to direct robust evaluation when packaging is novel or higher-risk.
- Supplier Collaboration: Guides procurement and quality assurance teams in communicating requirements to material and packaging suppliers, such as requesting cytotoxicity or chemical characterization data.
Related Standards
ASTM F2475-20 references several other important standards and guidance documents to align with global best practices in biocompatibility and packaging evaluation:
- ANSI/AAMI/ISO 11607: Packaging for terminally sterilized medical devices - specifies requirements for materials, sterile barrier systems, and packaging systems.
- ISO 10993-1: Biological evaluation of medical devices, focusing on test selection within a risk management process.
- USP <1031>: Biocompatibility of materials used in drug containers, medical devices, and implants.
- FDA Guidance (2016): Use of International Standard ISO 10993-1 for evaluating the biocompatibility of medical devices.
Practical Value
Adopting ASTM F2475-20 provides medical device manufacturers with:
- A clear pathway for evaluating and justifying the biocompatibility of packaging materials.
- Improved compliance with regulatory authorities, speeding up product approvals.
- Reduced risk of adverse patient outcomes associated with packaging-device interactions.
- Enhanced documentation and traceability for quality assurance and audit readiness.
By following ASTM F2475-20, stakeholders in medical device manufacturing and packaging can demonstrate due diligence, optimize testing strategies, and reinforce patient safety through effective biocompatibility evaluation.
Buy Documents
ASTM F2475-20 - Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials
REDLINE ASTM F2475-20 - Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2475-20 is a guide published by ASTM International. Its full title is "Standard Guide for Biocompatibility Evaluation of Medical Device Packaging Materials". This standard covers: SIGNIFICANCE AND USE 5.1 The compatibility of packaging materials with a medical device is a requirement of many regulatory bodies. Since most medical devices are used or implanted in, around, or on the human body, the benefits of these devices must outweigh the risks. Therefore, the packaging materials that come in contact with the medical device must also be evaluated and determined to be safe for use with the human body in that they have no negative impact on the physical, chemical, or biological properties of the device. This evaluation may include both a study of relevant experience with, and actual testing of, packaging materials. Such an evaluation may result in the conclusion that no testing is needed if the material has a demonstrable history of safe use in the specific role that is the same as that of the package under design. 5.2 The medical device manufacturer determines the need for appropriate testing, with consideration of the device/package interactions, if any. When screening information is needed regarding the biocompatibility of the packaging, cytotoxicity testing from the supplier is typically performed. SCOPE 1.1 This guide provides information to determine the appropriate testing for biocompatibility of medical device packaging materials that have the potential to contact the patient directly or indirectly. 1.2 This guide does not apply to secondary or tertiary packaging materials. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The compatibility of packaging materials with a medical device is a requirement of many regulatory bodies. Since most medical devices are used or implanted in, around, or on the human body, the benefits of these devices must outweigh the risks. Therefore, the packaging materials that come in contact with the medical device must also be evaluated and determined to be safe for use with the human body in that they have no negative impact on the physical, chemical, or biological properties of the device. This evaluation may include both a study of relevant experience with, and actual testing of, packaging materials. Such an evaluation may result in the conclusion that no testing is needed if the material has a demonstrable history of safe use in the specific role that is the same as that of the package under design. 5.2 The medical device manufacturer determines the need for appropriate testing, with consideration of the device/package interactions, if any. When screening information is needed regarding the biocompatibility of the packaging, cytotoxicity testing from the supplier is typically performed. SCOPE 1.1 This guide provides information to determine the appropriate testing for biocompatibility of medical device packaging materials that have the potential to contact the patient directly or indirectly. 1.2 This guide does not apply to secondary or tertiary packaging materials. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2475-20 is classified under the following ICS (International Classification for Standards) categories: 11.120.99 - Other standards related to pharmaceutics; 55.020 - Packaging and distribution of goods in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2475-20 has the following relationships with other standards: It is inter standard links to ASTM F2475-11, ASTM F17-20, ASTM F17-18a, ASTM F17-18, ASTM F17-17, ASTM F17-13a, ASTM F17-13, ASTM F17-12, ASTM F17-08, ASTM F17-07a, ASTM F17-07, ASTM F17-06, ASTM F17-02, ASTM F17-98, ASTM F2559/F2559M-21. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2475-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2475 − 20
Standard Guide for
Biocompatibility Evaluation of Medical Device Packaging
Materials
This standard is issued under the fixed designation F2475; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope for materials, sterile barrier systems, and packaging sys-
tems
1.1 This guide provides information to determine the appro-
FDA Center for Devices and Radiological Health: 2016
priate testing for biocompatibility of medical device packaging
Biocompatibility Guidance: Use of International Standard
materials that have the potential to contact the patient directly
ISO 10993-1, "Biological evaluation of medical devices –
or indirectly.
Part 1: Evaluation and testing within a risk management
1.2 This guide does not apply to secondary or tertiary
process" (June 16, 2016)
packaging materials.
3. Terminology
1.3 This standard does not purport to address all of the
3.1 Definitions—Forterminologyrelatedtobarriermaterials
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro- for medical packaging see Terminology F17.
priate safety, health, and environmental practices and deter-
3.2 Definitions of Terms Specific to This Standard:
mine the applicability of regulatory limitations prior to use.
3.2.1 extent of contact—degree to which the packaged
1.4 This international standard was developed in accor-
device will contact the patient (refer to ISO 10993-1). When
dance with internationally recognized principles on standard-
referringtothepackaging,extentofcontactreferstothedegree
ization established in the Decision on Principles for the
to which the packaging will interact with the device. Degree of
Development of International Standards, Guides and Recom-
packaging contact (interaction) is related to the physical-
mendations issued by the World Trade Organization Technical
chemical nature of the packaging materials and the device, the
Barriers to Trade (TBT) Committee.
intended use of the device (which relates to the categorization
of contact with the body), and the extent to which the
2. Referenced Documents
packaging may negatively impact the contained device.
2.1 ASTM Standards: 3.2.2 packaging biocompatibility—inherent ability of a ma-
terial to protect against a chemical change in the packaged
F17 Terminology Relating to Primary Barrier Packaging
medical device that could result in an adverse response to the
2.2 Other Standards:
host in its intended application.
ISO 10993-1:2018 Biological evaluation of medical devices
3.2.3 packaging biocompatibility testing—series of chemi-
– Part 1: Evaluation and testing within a risk management
cal and biological tests that a material is subjected to in order
process
to determine the ability of the packaging material to protect
ANSI/AAMI/ISO 11607-1:2006(R2010) Packaging for ter-
against a chemical change in the packaged medical device that
minally sterilized medical devices – Part 1: Requirements
could result in an adverse response to the host in its intended
application.
3.2.4 sterile barrier system—minimum package that pre-
This guide is under the jurisdiction of ASTM Committee F02 on Primary
Barrier Packaging and is the direct responsibility of Subcommittee F02.15 on vents ingress of microorganisms and allows aseptic presenta-
Chemical/Safety Properties.
tion of the product at the point of use.
Current edition approved Jan. 1, 2020. Published February 2020. Originally
approved in 2005. Last previous edition approved in 2011 as F2475 – 11. DOI:
4. Summary of Practice
10.1520/F2475-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
4.1 Materials used in packaging are to be evaluated per
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
definedguidelines,suchasAAMI/ANSI/ISO11607.Theeffect
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website.
Available from International Organization for Standardization (ISO), ISO
Central Secretariat, BIBC II, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
Geneva, Switzerland, http://www.iso.org. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2475 − 20
of packaging on the patient should be first and foremost contact with the medical device. Consult the referenced ISO
addressed through the biocompatibility evaluation of the de- and FDAguidances for selection of appropriate biocompatibil-
vice itself as this represents the clinical exposure. Additional ity evaluations. Because medical device packages are consid-
biocompatibility testing for packaging materials may be re- ered to have indirect patient contact, it is unlikely that the
quired based on the conditions and extent of packaging packaging biocompatibility assessments will exceed the re-
material contact with the medical device, the subsequent quirements for those medical devices intended for direct or
degreetowhichthemedicaldevicewillcontactthepatient,and indirect patient contact. However, due to the diversity of
the intended use of the device. While the scope of ISO medical devices and the packaging materials used for them, it
10993-1, and CDRH’s 2016 Biocompatibility Guidance refer- isrecognizedthatallevaluationsidentifiedforacertaincontact
encedin2.2donotdirectlyapplytomedicaldevicepackaging, category may not always result in the need for testing for some
use of these documents will address the intent ofAAMI/ANSI/ packaging material while other materials may require addi-
ISO 11607. tional testing to support a medical device regulatory applica-
tion.
4.2 The reader is advised to consult these standards and
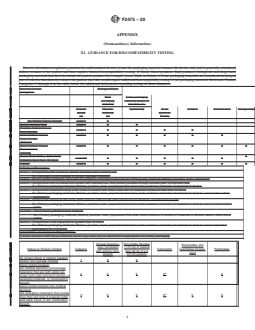
6.1.1 If it is determined that testing is needed, variations
guidances in determining which evaluations apply for a given
from standard testing plans may be justified to either reduce or
packaging application, and whether testing would be needed to
expand tests to be done based on (1) known potential contact
address those evaluation
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2475 − 11 F2475 − 20
Standard Guide for
Biocompatibility Evaluation of Medical Device Packaging
Materials
This standard is issued under the fixed designation F2475; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide provides information to determine the appropriate testing for biocompatibility of materials (or packaging
materials) in sterile barrier systems used to contain a medical device.medical device packaging materials that have the potential
to contact the patient directly or indirectly.
1.2 This guide does not apply to secondary or tertiary packaging materials.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and to determine the
applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
F17 Terminology Relating to Primary Barrier Packaging
2.2 Other Standards:
ANSI/AAMI/ISO 11607 Packaging for Terminally Sterilized Medical Devices
ISO 10993-1:2009 10993-1:2018 Biological Evaluationevaluation of Medical Devicesmedical devices – Part 1: Evaluation and
Testingtesting within a risk management process
USP <1031>ANSI/AAMI/ISO 11607-1:2006(R2010) The Biocompatibility of Materials Used in Drug Containers, Medical
Devices, and ImplantsPackaging for terminally sterilized medical devices – Part 1: Requirements for materials, sterile barrier
systems, and packaging systems
FDA – Center for Devices and Radiological Health: Required Biocompatability Training and Toxicology Profiles for Evaluation
of Medical Devices (#G95-1)2016 Biocompatibility Guidance: Use of International Standard ISO 10993-1, "Biological
evaluation of medical devices – Part 1: Evaluation and testing within a risk management process" (June 16, 2016)
3. Terminology
3.1 Definitions—For terminology related to barrier materials for medical packaging see Terminology F17.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 biocompatibility—the inherent ability of a material to remain biologically inert with the host in its intended application.
3.2.2 biocompatibility testing—the series of chemical and biological tests that a material is subjected to in order to determine
the ability of the material to remain biologically inert with the host in its intended application.
This guide is under the jurisdiction of ASTM Committee F02 on FlexiblePrimary Barrier Packaging and is the direct responsibility of Subcommittee F02.15 on
Chemical/Safety Properties.
Current edition approved April 1, 2011Jan. 1, 2020. Published April 2011February 2020. Originally approved in 2005. Last previous edition approved in 20052011 as
F2475 – 05.F2475 – 11. DOI: 10.1520/F2475-11.10.1520/F2475-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from International Organization for Standardization (ISO), ISO Central Secretariat, BIBC II, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
Switzerland, http://www.iso.org.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2475 − 20
3.2.1 extent of contact—the degree to which the packaged device will contact the patient (refer to ISO 10993-1 for levels of
contact of the device with the human body). 10993-1). When referring to the packaging, extent of contact refers to the degree to
which the packaging will interact with the device. Degree of packaging contact (interaction) is related to the physical-chemical
nature of the packaging materials and the device, the intended use of the device (levels (which relates to the categorization of
contact with the body), and the extent to which the packaging may negatively impact the contained device.
3.2.2 packaging biocompatibility—inherent ability of a material to protect against a chemical change in the packaged medical
device that could result in an adverse response to the host in its intended application.
3.2.3 packaging biocompatibility testing—series of chemical and biological tests that a material is subjected to in order to
determine the ability of the packaging material to protect against a chemical change in the packaged medical device that could
result in an adverse response to the host in its intended application.
3.2.4 sterile barrier system—minimum package that prevents ingress of microorganisms and allows aseptic presentation of the
product at the point of use.
4. Summary of Practice
4.1 Materials used in packaging are to be evaluated per defined guidelines, such as AAMI/ANSI/ISO 11607. The effect of
packaging on the patient should be first and foremost addressed through the biocompatibility evaluation of the device itself as this
represents the clinical exposure. Additional biocompatibility testing for packaging materials may be required based on the
conditions and extent of packaging material contact with the contained medical device, the subsequent degree to which the
packagedmedical device (product) will contact the patient, and the intended use of the device. When selecting the appropriate tests
for biological evaluation of medical devices, the chemical characteristicsWhile the scope of ISO 10993-1, and CDRH’s 2016
Biocompatibility Guidance referenced in 2.2 of the device materials, as well as the nature, degree, frequency and duration of the
device’s exposure to the body must be considered. Similar testing may be considered for medical packaging, when there is not a
history of safe use of packaging materials for their intended sue or there may be a question as to whether the packaging may
negatively impact the contained device. Guidelines for biocompatibility verification of medical device packaging are based on
FDA guidance (Memorandum #G-95), ANSI/AAMI/ISO 10993-1 and USP <1031> The Biocompatibility of Materials Used in
Drug Containers, Medical Devices, and Implants. While the scope of these standards does do not directly apply to medical device
packaging, use of them these documents will address the intent of ISOAAMI/ANSI/ISO 11607.
The reader is advised to consult these standards in determining which tests apply for a given packaging application. All medical
device packages are considered to have indirect patient contact, at a minimum. Therefore, the tests selected will not typically
require more extensive testing than that required for medical devices intended for indirect patient contact.
However, test selection should also be based on the extent of contact between the package and the device, and the probability
that the package may negatively impact the properties of the contained medical device. For example, a device that is a solid
structure is less likely to interact with its packaging than a device composed of a semi-solid or liquid material.
4.2 The reader is advised to consult these standards and guidances in determining which evaluations apply for a given packaging
application, and whether testing would be needed to address those evaluations. If chemical information is needed to support that
there is no interaction between the device and the packaging, the need for analytical chemistry information, and the type of physical
or chemical testing selected (some options can include Fourier-transform infrared spectroscopy (FTIR), Scanning Electron
Microscopy (SEM) and extractable or leachable analyses) should also be based on the extent of contact between the package and
the device or the package and device materials, to assess whether the package may negatively impact the properties of the
contained medical device. For example, a device that is a solid structure may be less likely to interact with its packaging than a
device composed of a semi-solid or liquid material. The chemical stability of the packaging materials over the shelf life of the
product, and the potential for interaction between the packaging and the device should be considered and justified by an
appropriately qualified individual using experimental data or preexisting information.
5. Significance and Use
5.1 The compatibility of packaging materials with a medical device is a requirement of many regulatory bodies. Since most
medical devices are used or implanted in, around, or on the human body, the benefits of these devices must do no harm.outweigh
the risks. Therefore, the packaging materials that come in contact with the medical device must also be evaluated and determined
to be safe for use with the human body in that they have no negative impact on the physical, chemical, or biological properties
of the device. . This evaluation may include both a study of relevant experience with, and actual testing of, packaging materials.
Such an evaluation may result in the conclusion that no testing is needed if the material has a demonstrable history of safe use in
the specific role that is the same as that of the package under design.
The medical device manufacturer determines the need for appropriate testing, with consideration of the device/package
i
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...