ASTM D4127-18a
(Terminology)Standard Terminology Used with Ion-Selective Electrodes
Standard Terminology Used with Ion-Selective Electrodes
SCOPE
1.1 This terminology covers those terms recommended by the International Union of Pure and Applied Chemistry (IUPAC),2 and is intended to provide guidance in the use of ion-selective electrodes for analytical measurement of species in water, wastewater, and brines.
1.2 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2018
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Feb-2006
- Effective Date
- 01-Mar-2004
- Effective Date
- 01-Mar-2004
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Mar-2003
- Effective Date
- 10-Jul-2002
- Effective Date
- 10-Jul-2002
- Effective Date
- 10-Feb-2002
- Effective Date
- 10-Feb-2002
Overview
ASTM D4127-18a: Standard Terminology Used with Ion-Selective Electrodes is an essential international standard issued by ASTM International. This document establishes a consistent and authoritative set of terms commonly used in the field of ion-selective electrode (ISE) measurements. These terms are based on recommendations from the International Union of Pure and Applied Chemistry (IUPAC) and are intended to provide guidance for the accurate analytical measurement of ionic species in water, wastewater, and brines.
The standard is developed in alignment with globally recognized principles, supporting harmonized communication in laboratory analysis, water quality monitoring, and regulatory reporting. By defining critical concepts and terminology, ASTM D4127-18a helps professionals properly interpret results and ensures comparability across laboratories and industries.
Key Topics
Core terminology defined in ASTM D4127-18a includes:
- Ion-selective electrodes (ISEs): Sensors that measure the activity of specific ions in solution, critical for chemical analysis.
- Calibration and measurement: Terms like calibration curve, activity standard, and concentration standard clarify best practices for electrode use and data interpretation.
- Electrode characteristics: Definitions cover pH electrode, combination electrode, reference electrode (including single-junction, double-junction, and types like calomel, silver/silver chloride), response time, slope, and electrode life.
- Analytical errors and interferences: Descriptions of acid error, alkaline error, interfering substances, and limitations related to selectivity constants.
- Measurement techniques: Concepts such as direct measurement, standard addition/subtraction, titration methods, and the use of selective ion meters are detailed.
- Physical and chemical parameters: Terms such as activity, activity coefficient, ionic strength, junction potential, and membrane types are precisely explained for technical accuracy.
This terminology ensures consistency and clarity when reporting or interpreting results involving ISE technology across various applications.
Applications
ASTM D4127-18a finds broad application in:
- Water and wastewater analysis: Laboratories use ISEs for the accurate measurement of ionic species such as pH, sodium, chloride, fluoride, calcium, and other ions within water quality testing, environmental monitoring, and process control.
- Analysis of brines: The standard is particularly relevant for brine solutions, including industrial and environmental samples, where high ionic strength demands specialized terminology and understanding.
- Industrial process control: Chemical and manufacturing industries rely on ion-selective electrodes to monitor electrolytes and maintain compliance with environmental standards.
- Educational and research settings: Universities and research institutions reference these standardized definitions to ensure teaching and experimentation are consistent with international practice.
- Regulatory and reporting compliance: The terminology supports uniform interpretation in reports submitted for regulatory approval or quality assurance under global trade and environmental agreements.
By using the standard vocabulary, professionals can minimize ambiguity and ensure that data from different sources are directly comparable and reliable.
Related Standards
To enhance understanding and effective application, the following related ASTM and international standards can be referenced:
- ASTM D1129: Terminology Relating to Water – provides foundational water analysis terminology referenced within D4127-18a.
- IUPAC Recommendations for Nomenclature of Ion-Selective Electrodes: Offers additional international guidance aligning with the terminology in this standard.
- ISO standards related to water quality and analytical laboratory practice may complement ASTM D4127-18a for broader context.
For more detailed information or the most current version of ASTM D4127-18a, visit the ASTM International website.
Keywords: ion-selective electrodes, analytical measurement, ISE, water analysis, wastewater, brines, standard terminology, ASTM D4127, laboratory standards, electrode calibration, selectivity, reference electrode, pH measurement.
Buy Documents
ASTM D4127-18a - Standard Terminology Used with Ion-Selective Electrodes
REDLINE ASTM D4127-18a - Standard Terminology Used with Ion-Selective Electrodes
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D4127-18a is a standard published by ASTM International. Its full title is "Standard Terminology Used with Ion-Selective Electrodes". This standard covers: SCOPE 1.1 This terminology covers those terms recommended by the International Union of Pure and Applied Chemistry (IUPAC),2 and is intended to provide guidance in the use of ion-selective electrodes for analytical measurement of species in water, wastewater, and brines. 1.2 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SCOPE 1.1 This terminology covers those terms recommended by the International Union of Pure and Applied Chemistry (IUPAC),2 and is intended to provide guidance in the use of ion-selective electrodes for analytical measurement of species in water, wastewater, and brines. 1.2 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4127-18a is classified under the following ICS (International Classification for Standards) categories: 01.040.71 - Chemical technology (Vocabularies); 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4127-18a has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D1129-10, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D1129-06, ASTM D1129-04, ASTM D1129-04e1, ASTM D1129-03a, ASTM D1129-03, ASTM D1129-02a, ASTM D1129-01, ASTM D1129-02, ASTM D1129-99a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4127-18a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4127 − 18a
Standard Terminology Used with
Ion-Selective Electrodes
This standard is issued under the fixed designation D4127; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
concentration. Ionic activity, not concentration, determines both the
1. Scope*
rate and the extent of chemical reactions.
1.1 This terminology covers those terms recommended by
activity coefficient, n—afactor, γ,thatrelatesactivity, A,tothe
the International Union of Pure and Applied Chemistry
(IUPAC), and is intended to provide guidance in the use of concentration, C of a species in solution:
ion-selective electrodes for analytical measurement of species
A 5 γC
in water, wastewater, and brines.
DISCUSSION—The activity coefficient is dependent on the ionic
strength of the solution. Ions of similar size and charge have similar
1.2 This international standard was developed in accor-
activity coefficients.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
alkaline error, n—in alkaline solutions, where hydrogen ion
Development of International Standards, Guides and Recom-
activity becomes very small, some glass electrodes respond
mendations issued by the World Trade Organization Technical
to other cations, such as sodium.
Barriers to Trade (TBT) Committee. DISCUSSION—AnegativeerrorinthepHreadingresults.Bychanging
thecompositionoftheglass,theaffinityoftheglassforsodiumioncan
2. Referenced Documents be reduced. Such electrodes are known as lithium glass, high-pH, or
full-range electrodes.
2.1 ASTM Standards:
D1129Terminology Relating to Water analate, n—the sample being analyzed; used in the terms
“analate addition” and “analate subtraction.”
3. Terminology
DISCUSSION—This term differs from the term “analyte,” which
describes the chemical species of interest in an analytical test.
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to
asymmetry potential, n—the potential across a glass pH
Terminology D1129.
electrode membrane when the inside and outside of the
membrane are in contact with solutions of identical pH.
3.2 Definitions of Terms Relevant to Ion-Selective Electrode
DISCUSSION—This term has also been used to define the observed
Technology:
potential differences between identical electrode pairs placed in iden-
acid error, n—in very acid solutions, the activity of water is
tical solutions.
reduced (less than unity) causing a non-Nernstian response
in glass electrodes. calibration curve, n—a plot of the potential (emf or E) of a
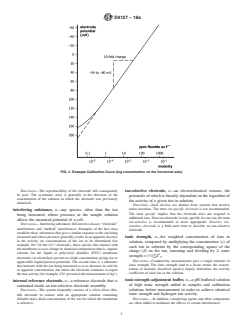
DISCUSSION—A positive error in the pH reading results.
given ion-selective electrode cell assembly (ion-selective
electrode combined with an identified reference electrode)
activity, n—the thermodynamically effective concentration of
versus the logarithm of the ionic activity (or concentration)
a free ion in solution.
of a given species.
DISCUSSION—In dilute solutions, ionic activity, and concentration are
DISCUSSION—For uniformity, it is recommended that the potential be
practically identical, but in solutions of high ionic strength, or in the
plotted on the ordinate (vertical axis) with the more positive potentials
presence of complexing agents, activity may differ significantly from
at the top of the graph and that pa (−log activity of the species
A
measured, A)orpc (−log concentration of species measured, A)be
A
1 plotted on the abscissa (horizontal axis) with increasing activity to the
This terminology is under the jurisdiction ofASTM Committee D19 on Water
right. See Fig. 1 and Fig. 2. Region I of Fig. 1 represents the segment
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
in Water. of the curve where the potential no longer changes in response to
Current edition approved May 1, 2018. Published June 2018. Originally
changes of the measured species activity and the electrode no longer
approved in 1982. Last previous edition approved in 2018 as D4127–18. DOI:
demonstrates Nernstian response.
10.1520/D4127-18a.
IUPAC
Recommendations for Nomenclature of Ion-Selective Electrodes, IUPAC Com-
mission on Analytical Nomenclature, Pergamon Press, Oxford, 1976.
activity standard, n—a standardizing solution whose value is
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
reported in terms of ionic activity.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
DISCUSSION— If the electrode is calibrated using activity standards,
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. the activity of the free, unbound ion in the sample is determined.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4127 − 18a
electrolyte, n—a substance that ionizes in aqueous solution;
also, a solution containing ions.
DISCUSSION—Weakelectrolytesareonlyslightlydissociatedintoions
in solution (acetic acid), and strong electrolytes are highly dissociated
(HCl, NaCl).
equitransference, n—equal diffusion rates of the positively
and negatively charged ions of an electrolyte across a liquid
junction.
equitransferent filling solution, n—a reference electrode
filling solution in which the diffusion rates of negatively and
positively charged ions are equal.
FIG. 1 Example Response Curve (log activity on the horizontal
axis)
filling solution, n—the solution inside a sensing or reference
electrode that is replenished periodically.
DISCUSSION— Solutions that are permanently sealed within the
concentration standard, n—a standardizing solution whose
electrode (like the buffer inside a pH electrode) are usually called
value is reported in terms of total concentration of the ion of
internal reference solutions to differentiate them from filling solutions.
interest.
DISCUSSION—If the electrode is calibrated using pure-concentration
internal filling solution of sensing electrode, n—in liquid
standards and measurements made on untreated samples, results must
membrane electrodes, an aqueous internal filling solution
be corrected for the sample ionic strength and the presence of
contacts the internal reference element and the membrane,
complexingagents.Morecommonly,areagentisaddedtoallstandards
which is saturated with ion exchanger.
and samples before measurement in order to fix the ionic strength, thus
avoiding the need for correction.
DISCUSSION—The filling solution normally contains a fixed level of
chloride and of the ion for which the electrode was designed; the
combination electrode, n—an electrochemical apparatus that
concentration of this ion determines the zero potential point of the
incorporates an ion-selective electrode and a reference
electrode. In addition, the filling solution is saturated with silver
electrode in a single assembly thereby avoiding the need for chloride to prevent the silver chloride of the internal reference element
from dissolving.
a separate reference electrode.
concentration, n—the actual amount of a substance in a given reference electrode filling solution, n—a concentrated salt
solution contacting the internal reference element and the
volume of solution.
DISCUSSION—When measuring ionic concentrations by electrode, a
sample solution.
distinction is made between the concentration of the free, unbound ion,
DISCUSSION—The composition of the filling solution is chosen to
and total concentration that includes ions bound to complexing agents.
maximize stability of the potentials developed at the internal reference
element/filling solution interface and the filling solution/sample junc-
dissociation constant, n—a number indicating the extent to
tion. In general, filling solutions for AgCl internal construction refer-
which a substance dissociates in solution.
−
ence electrodes should: (1) contain Cl and be saturated withAgCl to
DISCUSSION—For a simple two-species complex AB, the constant is
prevent the reference element from dissolving; (2) be at least ten times
given by the product of the molar concentrations of A and of B divided
higherintotalionicstrengththanthesample;(3)beequitransferent;(4)
by the molar concentrations of the undissociated species AB. For
not contain the ion being measured or an ion that interferes with the
example, with hydrofluoric acid:
measurement.
1 2 24
~@H #@F #!/~@HF#! 5 K 5 6.7 310 at25°C
flowthrough electrodes, n—ion-selective and reference elec-
The smaller the value of K, the less the complex is dissociated. K
trodes designed for anaerobic measurements.
varies with temperature, ionic strength, and the nature of the solvent.
DISCUSSION—The two electrodes are connected by plastic tubing to a
syringe or peristaltic pump, and the sample is pumped through the
drift, n—this is the slow nonrandom change with time in the
electrodes at a constant rate. Ion-selective electrodes can be made in a
potential (emf) of an ion-selective electrode cell assembly
flow through configuration for the measurement of very small samples
maintained in a solution of constant composition and tem-
(0.2 to 0.3 mL) or samples that must be measured anaerobically.
perature.
Gran’s plots, n—a method of plotting apparent concentration
electrode life, n—thelengthoftimethatanelectrodefunctions
(as derived from the electrode potential) versus the volume
usefully.
of reagent added to the sample.
DISCUSSION—Life-time of solid-state and glass electrodes is limited
DISCUSSION—Gran’s plots are especially useful for plotting titrations
by mechanical failure of the electrode body or chemical attack on the
that would give poor end-point breaks if plotted conventionally. These
sensing membrane, and can range from a few days, if the electrode is
plots can also be used to determine concentration by known addition
used continuously in hot or abrasive flowing streams, to several years
with greater precision than can be obtained by a single addition
under normal laboratory conditions. The life-time of liquid membrane
measurement.
electrodesislimitedbylossofionexchangerwithuse,andisgenerally
1 to 6 months.
hysteresis (electrode memory), n—hysteresis is said to have
electrode pair, n—a sensing electrode and a reference elec- occurred if, after the concentration has been changed and
trode; the reference electrode may be separate or combined restored to its original value, there is a different potential
in one body with the sensing electrode. observed.
D4127 − 18a
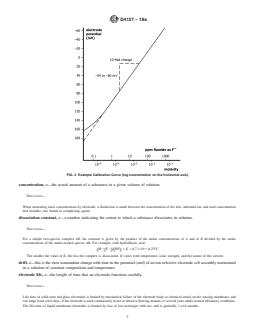
FIG. 2 Example Calibration Curve (log concentration on the horizontal axis)
DISCUSSION—The reproducibility of the electrode will consequently
ion-selective electrode, n—an electrochemical sensors, the
be poor. The systematic error is generally in the direction of the
potentials of which is linearly dependent on the logarithm of
concentration of the solution in which the electrode was previously
the activity of a given ion in solution.
immersed.
DISCUSSION—Such devices are distinct from systems that involve
redox reactions. The term ion-specific electrode is not recommended.
interfering substance, n—any species, other than the ion
The term specific implies that the electrode does not respond to
being measured, whose presence in the sample solution
additionalions.Sincenoelectrodeistrulyspecificforoneion,theterm
affects the measured potential of a cell.
ion-selective is recommended as more appropriate. Selective ion-
DISCUSSION—Interferingsubstancesfallintotwoclasses:“electrode”
sensitive electrode is a little-used term to describe an ion-selective
interferences and “method” interferences. Examples of the first class
electrode.
would be those substances that give a similar response to the ion being
measured and whose presence generally results in an apparent increase
ionic strength, n—the weighted concentration of ions in
in the activity (or concentration) of the ion to be determined (for
solution, computed by multiplying the concentration (c)of
+ ++
example, Na for the Ca electrode), those species that interact with
each ion in solution by the corresponding square of the
themembranesoastochangeitschemicalcomposition(thatis,organic
charge (Z) on the ion, summing and dividing by 2: ionic
solvents for the liquid or poly(vinyl chloride) (PVC) membrane
strength=( ⁄2)∑Z .
C
electrodes) or electrolytes present at a high concentration giving rise to
DISCUSSION—Conductivity measurements give a rough estimate of
appreciable liquid-junction potentials. The second class is a substance
ionic strength. The ionic strength (and to a lesser extent, the concen-
that interacts with the ion being measured so as to decrease its activity
tration of nonionic dissolved species) largely determines the activity
or apparent concentration, but where the electrode continues to report
− +
coefficient of each ion in the solution.
thetrueactivity(forexample,CN presentinthemeasurementofAg ).
ionic-strength adjustment buffer, n—a pH buffered solution
internal reference electrode, n—a reference electrode that is
of high ionic strength added to samples and calibration
contained inside an ion-selective electrode assembly.
solutions before measurement in order to achieve identical
DISCUSSION—The system frequently consists of a silver-silver chlo-
ride electrode in contact with an appropriate solution containing ionic strength and hydrogen ion activity.
chloride and a fixed concentration of the ion for which the membrane DISCUSSION— In addition, complexing agents and other components
is selective. are often added to minimize the effects of certain interferences.
D4127 − 18a
isopotential point, n—for a cell containing an ion-selective Nernst factor, n—in the Nernst equation, the term 2.3RT/zF,
electrode and a reference electrode there is often a particular which is equal (at T=25°C) to 59.16 mV when z=1 and
activity of the ion concerned for which the potential of the 29.58 mV when z=2, and which includes the sign of the
cell is independent of temperature. charge on the ion in the term z.
DISCUSSION—That activity, and the corresponding potential, define DISCUSSION—The Nernst factor varies with temperature. This factor
the isopotential point. The identity of the reference electrode, and the is often referred to as the electrode slope.
composition of the filling solution of the measuring electrode, must be
Nernstian response, n—electrodebehaviorwhichconformsto
specified.
the Nernst equation. For example, when a plot of the
junction potential, n—for the total observed potential devel-
potentialofsuchanelectrodeinconjunctionwithareference
oped between the sensing and reference electrodes, the
electrodeversusthelogarithmoftheionicactivityofagiven
portion that is formed at the liquid/liquid junction between
measured species (a ) is linear with a slope of 2.303×10
A
F
the reference electrode filling solution and the sample
RT/z mV/decade (59.16/z mV per unit of pa at 25°C).
A A A
solution.
pH electrode, n—an ion-selective electrode, made of glass,
DISCUSSION— For accuracy, the junction potential should be as low
that responds to hydrogen ion activity.
and as constant as possible despite variations in the composition of the
DISCUSSION—Wide-range pH electrodes function over the activity
sample solution. Reference electrode filling solutions should be judi-
+ −14 +
range 1 M H (pH 0) to 10 M H (pH 14). pH electrodes may be
ciously chosen to minimize liquid junction potential.
subject to “acid error” in strongly acidic solutions and are also subject
mean ionic activity coefficient, n—for a salt that is composed
to “alkaline error” caused by response to sodium or other cations in
of two monovalent ions, the geometric mean of the indi- basic solutions.
vidual ionic activity coefficients.
DISCUSSION—An ISFET (Ion-Selective Field Effect Transistor) pH
DISCUSSION— (The geometric mean is obtained, in this case, by
electrode uses a pH sensitive metal oxide layer over the gate of a
multiplying the two individual ionic activity coefficients and then
transistor that is compactly packaged with corrosion-resistant materials
taking the square root.) This mean coefficient is important because,
anddoesnotcontainglass,makingitparticularlyapplicableinfoodand
unlike individual ionic activity coefficients, it can be measured by a
life sciences industries.
variety of techniques, such as freezing point depression and vapor
DISCUSSION—An ISFET pH electrode may have negligible alkaline
pressure, as well as by paired sensing electrodes.
error. An ISFET pH electrode is usually combined with a reference
membrane, n—a thin space of material covering a structure of
electrode and a temperature sensor into a single probe.
separating solutions and permitting selection transport of a
pH/mV meter, n—an instrument that measures and displays
chemical species between the two solutions.
the voltage developed between a sensing electrode and a
liquid ion-exchange electrode membrane, n—a porous plas- reference electrode placed in a solution, and can convert the
voltage developed by a pH electrode to a corresponding pH
ticdisk,permeabletotheionexchanger,andimpermeableto
water, that allows the ion exchanger to contact the sample value.
solution and separates the internal filling solution from the
analog pH/mV meter, n—aninstrumentthatdisplaysvoltages,
sample.
pH units, or other concentration units by means of a needle
pointer and scale.
gas electrode membrane, n—a plastic film, permeable to
gases but impermeable to water, separating the electrode
digital pH/mV meter, n—aninstrumentwithdigitaldisplayof
from the simple solution.
millivolts or pH units.
DISCUSSION— Less operator error is associated with digital instru-
crystal membrane or solid-state electrode membrane, n—a
ments since there is no need for interpolation and no risk of confusing
thin single or mixed crystal that is an ionic conductor and
scales.
that separates the internal reference element or inter
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4127 − 18 D4127 − 18a
Standard Terminology Used with
Ion-Selective Electrodes
This standard is issued under the fixed designation D4127; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This terminology covers those terms recommended by the International Union of Pure and Applied Chemistry (IUPAC),
and is intended to provide guidance in the use of ion-selective electrodes for analytical measurement of species in water,
wastewater, and brines.
1.2 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Relevant to Ion-Selective Electrode Technology:
acid error, n—in very acid solutions, the activity of water is reduced (less than unity) causing a non-Nernstian response in glass
electrodes.
DISCUSSION—
A positive error in the pH reading results.
activity, n—the thermodynamically effective concentration of a free ion in solution.
DISCUSSION—
In dilute solutions, ionic activity, and concentration are practically identical, but in solutions of high ionic strength, or in the presence of complexing
agents, activity may differ significantly from concentration. Ionic activity, not concentration, determines both the rate and the extent of chemical
reactions.
activity coefficient, n—a factor, γ, that relates activity, A, to the concentration, C of a species in solution:
A 5 γC
DISCUSSION—
The activity coefficient is dependent on the ionic strength of the solution. Ions of similar size and charge have similar activity coefficients.
This terminology is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved Feb. 1, 2018May 1, 2018. Published May 2018June 2018. Originally approved in 1982. Last previous edition approved in 20122018 as
D4127 – 12.D4127 – 18. DOI: 10.1520/D4127-18.10.1520/D4127-18a.
Recommendations for Nomenclature of Ion-Selective Electrodes, IUPAC Commission on Analytical Nomenclature, Pergamon Press, Oxford, 1976.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4127 − 18a
alkaline error, n—in alkaline solutions, where hydrogen ion activity becomes very small, some glass electrodes respond to other
cations, such as sodium.
DISCUSSION—
A negative error in the pH reading results. By changing the composition of the glass, the affinity of the glass for sodium ion can be reduced. Such
electrodes are known as lithium glass, high-pH, or full-range electrodes.
analate, n—the sample being analyzed; used in the terms “analate addition” and “analate subtraction.”
DISCUSSION—
This term differs from the term “analyte,” which describes the chemical species of interest in an analytical test.
asymmetry potential, n—the potential across a glass pH electrode membrane when the inside and outside of the membrane are
in contact with solutions of identical pH.
DISCUSSION—
This term has also been used to define the observed potential differences between identical electrode pairs placed in identical solutions.
calibration curve, n—a plot of the potential (emf or E) of a given ion-selective electrode cell assembly (ion-selective electrode
combined with an identified reference electrode) versus the logarithm of the ionic activity (or concentration) of a given species.
DISCUSSION—
For uniformity, it is recommended that the potential be plotted on the ordinate (vertical axis) with the more positive potentials at the top of the graph
and that pa (−log activity of the species measured, A) or pc (−log concentration of species measured, A) be plotted on the abscissa (horizontal axis)
A A
with increasing activity to the right. See Fig. 1 and Fig. 2. Region I of Fig. 1 represents the segment of the curve where the potential no longer changes
in response to changes of the measured species activity and the electrode no longer demonstrates Nernstian response.
IUPAC
activity standard, n—a standardizing solution whose value is reported in terms of ionic activity.
DISCUSSION—
If the electrode is calibrated using activity standards, the activity of the free, unbound ion in the sample is determined.
concentration standard, n—a standardizing solution whose value is reported in terms of total concentration of the ion of interest.
DISCUSSION—
If the electrode is calibrated using pure-concentration standards and measurements made on untreated samples, results must be corrected for the sample
ionic strength and the presence of complexing agents. More commonly, a reagent is added to all standards and samples before measurement in order
to fix the ionic strength, thus avoiding the need for correction.
combination electrode, n—an electrochemical apparatus that incorporates an ion-selective electrode and a reference electrode in
a single assembly thereby avoiding the need for a separate reference electrode.
FIG. 1 Example Response Curve (log activity on the horizontal axis)
D4127 − 18a
FIG. 2 Example Calibration Curve (log concentration on the horizontal axis)
concentration, n—the actual amount of a substance in a given volume of solution.
DISCUSSION—
When measuring ionic concentrations by electrode, a distinction is made between the concentration of the free, unbound ion, and total concentration
that includes ions bound to complexing agents.
dissociation constant, n—a number indicating the extent to which a substance dissociates in solution.
DISCUSSION—
For a simple two-species complex AB, the constant is given by the product of the molar concentrations of A and of B divided by the molar
concentrations of the undissociated species AB. For example, with hydrofluoric acid:
1 2 24
~@H # @F #!/~@HF#!5 K 5 6.7 310 at 25°C
The smaller the value of K, the less the complex is dissociated. K varies with temperature, ionic strength, and the nature of the solvent.
drift, n—this is the slow nonrandom change with time in the potential (emf) of an ion-selective electrode cell assembly maintained
in a solution of constant composition and temperature.
electrode life, n—the length of time that an electrode functions usefully.
DISCUSSION—
Life-time of solid-state and glass electrodes is limited by mechanical failure of the electrode body or chemical attack on the sensing membrane, and
can range from a few days, if the electrode is used continuously in hot or abrasive flowing streams, to several years under normal laboratory conditions.
The life-time of liquid membrane electrodes is limited by loss of ion exchanger with use, and is generally 1 to 6 months.
D4127 − 18a
electrode pair, n—a sensing electrode and a reference electrode; the reference electrode may be separate or combined in one body
with the sensing electrode.
electrolyte, n—a substance that ionizes in aqueous solution; also, a solution containing ions.
DISCUSSION—
Weak electrolytes are only slightly dissociated into ions in solution (acetic acid), and strong electrolytes are highly dissociated (HCl, NaCl).
equitransference, n—equal diffusion rates of the positively and negatively charged ions of an electrolyte across a liquid junction.
equitransferent filling solution, n—a reference electrode filling solution in which the diffusion rates of negatively and positively
charged ions are equal.
filling solution, n—the solution inside a sensing or reference electrode that is replenished periodically.
DISCUSSION—
Solutions that are permanently sealed within the electrode (like the buffer inside a pH electrode) are usually called internal reference solutions to
differentiate them from filling solutions.
internal filling solution of sensing electrode, n—in liquid membrane electrodes, an aqueous internal filling solution contacts the
internal reference element and the membrane, which is saturated with ion exchanger.
DISCUSSION—
The filling solution normally contains a fixed level of chloride and of the ion for which the electrode was designed; the concentration of this ion
determines the zero potential point of the electrode. In addition, the filling solution is saturated with silver chloride to prevent the silver chloride of
the internal reference element from dissolving.
reference electrode filling solution, n—a concentrated salt solution contacting the internal reference element and the sample
solution.
DISCUSSION—
The composition of the filling solution is chosen to maximize stability of the potentials developed at the internal reference element/filling solution
interface and the filling solution/sample junction. In general, filling solutions for AgCl internal construction reference electrodes should: (1) contain
−
Cl and be saturated with AgCl to prevent the reference element from dissolving; (2) be at least ten times higher in total ionic strength than the sample;
(3) be equitransferent; (4) not contain the ion being measured or an ion that interferes with the measurement.
flowthrough electrodes, n—ion-selective and reference electrodes designed for anaerobic measurements.
DISCUSSION—
The two electrodes are connected by plastic tubing to a syringe or peristaltic pump, and the sample is pumped through the electrodes at a constant rate.
Ion-selective electrodes can be made in a flow through configuration for the measurement of very small samples (0.2 to 0.3 mL) or samples that must
be measured anaerobically.
Gran’s plots, n—a method of plotting apparent concentration (as derived from the electrode potential) versus the volume of
reagent added to the sample.
DISCUSSION—
Gran’s plots are especially useful for plotting titrations that would give poor end-point breaks if plotted conventionally. These plots can also be used
to determine concentration by known addition with greater precision than can be obtained by a single addition measurement.
hysteresis (electrode memory), n—hysteresis is said to have occurred if, after the concentration has been changed and restored
to its original value, there is a different potential observed.
DISCUSSION—
The reproducibility of the electrode will consequently be poor. The systematic error is generally in the direction of the concentration of the solution
in which the electrode was previously immersed.
D4127 − 18a
interfering substance, n—any species, other than the ion being measured, whose presence in the sample solution affects the
measured potential of a cell.
DISCUSSION—
Interfering substances fall into two classes: “electrode” interferences and “method” interferences. Examples of the first class would be those substances
that give a similar response to the ion being measured and whose presence generally results in an apparent increase in the activity (or concentration)
+ ++
of the ion to be determined (for example, Na for the Ca electrode), those species that interact with the membrane so as to change its chemical
composition (that is, organic solvents for the liquid or poly(vinyl chloride) (PVC) membrane electrodes) or electrolytes present at a high concentration
giving rise to appreciable liquid-junction potentials. The second class is a substance that interacts with the ion being measured so as to decrease its
− +
activity or apparent concentration, but where the electrode continues to report the true activity (for example, CN present in the measurement of Ag ).
internal reference electrode, n—a reference electrode that is contained inside an ion-selective electrode assembly.
DISCUSSION—
The system frequently consists of a silver-silver chloride electrode in contact with an appropriate solution containing chloride and a fixed concentration
of the ion for which the membrane is selective.
ion-selective electrode, n—an electrochemical sensors, the potentials of which is linearly dependent on the logarithm of the
activity of a given ion in solution.
DISCUSSION—
Such devices are distinct from systems that involve redox reactions. The term ion-specific electrode is not recommended. The term specific implies
that the electrode does not respond to additional ions. Since no electrode is truly specific for one ion, the term ion-selective is recommended as more
appropriate. Selective ion-sensitive electrode is a little-used term to describe an ion-selective electrode.
ionic strength, n—the weighted concentration of ions in solution, computed by multiplying the concentration (c) of each ion in
solution by the corresponding square of the charge (Z) on the ion, summing and dividing by 2: ionic strength = ( ⁄2)∑Z .
C
DISCUSSION—
Conductivity measurements give a rough estimate of ionic strength. The ionic strength (and to a lesser extent, the concentration of nonionic dissolved
species) largely determines the activity coefficient of each ion in the solution.
ionic-strength adjustment buffer, n—a pH buffered solution of high ionic strength added to samples and calibration solutions
before measurement in order to achieve identical ionic strength and hydrogen ion activity.
DISCUSSION—
In addition, complexing agents and other components are often added to minimize the effects of certain interferences.
isopotential point, n—for a cell containing an ion-selective electrode and a reference electrode there is often a particular activity
of the ion concerned for which the potential of the cell is independent of temperature.
DISCUSSION—
That activity, and the corresponding potential, define the isopotential point. The identity of the reference electrode, and the composition of the filling
solution of the measuring electrode, must be specified.
junction potential, n—for the total observed potential developed between the sensing and reference electrodes, the portion that
is formed at the liquid/liquid junction between the reference electrode filling solution and the sample solution.
DISCUSSION—
For accuracy, the junction potential should be as low and as constant as possible despite variations in the composition of the sample solution. Reference
electrode filling solutions should be judiciously chosen to minimize liquid junction potential.
mean ionic activity coefficient, n—for a salt that is composed of two monovalent ions, the geometric mean of the individual ionic
activity coefficients.
D4127 − 18a
DISCUSSION—
(The geometric mean is obtained, in this case, by multiplying the two individual ionic activity coefficients and then taking the square root.) This mean
coefficient is important because, unlike individual ionic activity coefficients, it can be measured by a variety of techniques, such as freezing point
depression and vapor pressure, as well as by paired sensing electrodes.
membrane, n—a thin space of material covering a structure of separating solutions and permitting selection transport of a chemical
species between the two solutions.
liquid ion-exchange electrode membrane, n—a porous plastic disk, permeable to the ion exchanger, and impermeable to water,
that allows the ion exchanger to contact the sample solution and separates the internal filling solution from the sample.
gas electrode membrane, n—a plastic film, permeable to gases but impermeable to water, separating the electrode from the simple
solution.
crystal membrane or solid-state electrode membrane, n—a thin single or mixed crystal that is an ionic conductor and that
separates the internal reference element or internal reference solution from the sample solution.
Nernst equation, n—a mathematical description of electrode behavior:
E 5 E 12.3 RT/zFlogA
x
where:
E = total potential (in volts) developed between the sensing and reference electrodes
E = potential (in volts) dependent on the choice of reference electrodes, V,
x
RT/zF = Nernst factor,
R = gas constants, 8.3143 V C/K·mol,
F = Faraday constant, 96 485 C/mol,
z = charge on the ion, including sign,
T = absolute temperature in Kelvin, and
A = activity of the ion to which the electrode is responding.
Nernst factor, n—in the Nernst equation, the term 2.3RT/zF, which is equal (at T = 25°C) to 59.16 mV when z = 1 and 29.58 mV
when z = 2, and which includes the sign of the charge on the ion in the term z.
DISCUSSION—
The Nernst factor varies with temperature. This factor is often referred to as the electrode slope.
Nernstian response, n—electrode behavior which conforms to the Nernst equation. For example, when a plot of the potential of
such an electrode in conjunction with a reference electrode versus the logarithm of the ionic activity of a given measured species
3 F
(a ) is linear with a slope of 2.303 × 10 RT/z mV/decade (59.16/z mV per unit of pa at 25°C).
A A A A
pH electrode, n—an ion-selective electrode, made of glass, that responds to hydrogen ion activity.
DISCUSSION—
+ −14 +
Wide-range pH electrodes function over the activity range 1 M H (pH 0) to 10 M H (pH 14). pH electrodes may be subject to “acid error” in
strongly acidic solutions and are also subject to “alkaline error” caused by response to sodium or other cations in basic solutions.
DISCUSSION—
An ISFET (Ion-Selective Field Effect Transistor) pH electrode uses a pH sensitive metal oxide layer over the gate of a transistor that is compactly
packaged with corrosion-resistant materials and does not contain glass, making it particularly applicable in food and life sciences industries.
DISCUSSION—
An ISFET pH electrode may have negligible alkaline error. An ISFET pH electrode is usually combined with a reference electrode and a temperature
sensor into a single probe.
pH/mV meter, n—an instrument that measur
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...