ASTM E2009-23
(Test Method)Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry
Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry
SIGNIFICANCE AND USE
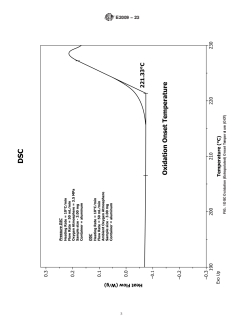
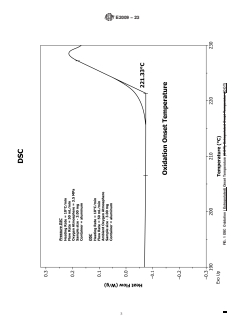
5.1 Oxidation onset temperature is a relative measure of the degree of oxidative stability of the material evaluated at a given heating rate and oxidative environment (e.g., oxygen); the higher the OOT value the more stable the material. The OOT is described in Fig. 1. The OOT values can be used for comparative purposes and are not an absolute measurement, like the oxidation induction time (OIT) at a constant temperature (see Test Method E1858). The presence or effectiveness of antioxidants may be determined by these test methods.
FIG. 1 DSC Oxidation (Extrapolated) Onset Temperature (OOT)
5.2 Typical uses of these test methods include the oxidative stability of edible oils and fats (oxidative rancidity), lubricants, greases, and polyolefins.
SCOPE
1.1 These test methods describe the determination of the oxidative properties of hydrocarbons by differential scanning calorimetry or pressure differential scanning calorimetry under linear heating rate conditions and are applicable to hydrocarbons, which oxidize exothermically in their analyzed form.
1.2 Test Method A—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of oxygen.
1.3 Test Method B—A pressure DSC (PDSC) is used at high pressure (e.g., 3.5 MPa (500 psig) of oxygen).
1.4 Test Method C—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of air.
1.5 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2022
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-Oct-2023
- Effective Date
- 15-Jan-2018
- Effective Date
- 01-Oct-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-Apr-2012
- Refers
ASTM D3350-12e1 - Standard Specification for Polyethylene Plastics Pipe and Fittings Materials - Effective Date
- 01-Apr-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-Apr-2011
- Effective Date
- 15-Nov-2010
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-May-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Jan-2010
Overview
ASTM E2009-23: Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry establishes standardized methods for determining the oxidative properties of hydrocarbons. Using tools such as Differential Scanning Calorimetry (DSC) or Pressure Differential Scanning Calorimetry (PDSC), these methods measure the Oxidation Onset Temperature (OOT), which serves as a relative indicator of a material’s oxidative stability under defined heating conditions. This standard is widely used in industries handling hydrocarbons, including edible oils, lubricants, greases, and polyolefins, to evaluate material performance and quality.
Key Topics
- Oxidation Onset Temperature (OOT): A comparative measure of the oxidative stability of hydrocarbons. A higher OOT indicates greater stability under the test conditions.
- Test Environments:
- Method A: Uses DSC at ambient pressure in an oxygen atmosphere.
- Method B: Uses PDSC at elevated pressures (e.g., 3.5 MPa of oxygen).
- Method C: Uses DSC at ambient pressure in air.
- Relevance of OOT: The OOT values are not absolute measures (in contrast to Oxidation Induction Time, OIT), but serve effectively for comparative and quality control purposes.
- Effect of Antioxidants: The method can be used to assess the presence or effectiveness of antioxidants in tested materials.
- Calibration and Accuracy: Regular calibration and proper setup of DSC/PDSC equipment are critical for precise measurements. The sensor must be in close thermal contact with the specimen, and calibration materials such as indium and tin are employed.
- Safety Considerations: Handling of oxygen and other reactive gases requires adherence to safety practices to minimize hazards.
Applications
ASTM E2009-23 test methods have several practical applications across industries working with hydrocarbons:
- Quality Assurance: Routine determination of OOT values assists manufacturers in monitoring the oxidative stability of products such as edible oils, lubricants, greases, and polymer-based materials like polyolefins.
- Research and Development: Comparative OOT measurements are valuable during product development, especially for materials where oxidation resistance is a key property.
- Antioxidant Evaluation: By measuring OOT, the impact of antioxidant additives can be determined, guiding formulation and development of more stable products.
- Process Control: Monitoring changes in OOT over time or after certain treatments can help detect issues in manufacturing or storage conditions.
- Comparative Analysis: Enables the benchmarking of new materials or formulations against established standards, supporting informed material selection and assurance.
Related Standards
When implementing or referencing ASTM E2009-23, the following related ASTM standards offer complementary methods and terminology:
- ASTM D3350: Specification for Polyethylene Plastics Pipe and Fittings Materials
- ASTM D3895: Test Method for Oxidative-Induction Time of Polyolefins by DSC
- ASTM D4565: Test Methods for Physical and Environmental Performance Properties of Insulations and Jackets for Telecommunications Wire and Cable
- ASTM D5483: Test Method for Oxidation Induction Time of Lubricating Greases by PDSC
- ASTM E473: Terminology Relating to Thermal Analysis and Rheology
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM E967: Test Method for Temperature Calibration of DSC and Differential Thermal Analyzers
- ASTM E1858: Test Methods for Determining Oxidation Induction Time of Hydrocarbons by DSC
- ASTM E3142: Test Method for Thermal Lag of Thermal Analysis Apparatus
These referenced documents reinforce standardization, comparability, and precision in oxidation stability and hydrocarbon testing.
Keywords: ASTM E2009-23, oxidation onset temperature, differential scanning calorimetry, hydrocarbons, oxidative stability, pressure differential scanning calorimetry, quality control, lubricant testing, edible oil stability, polymer oxidative stability.
Buy Documents
ASTM E2009-23 - Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry
REDLINE ASTM E2009-23 - Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E2009-23 is a standard published by ASTM International. Its full title is "Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 Oxidation onset temperature is a relative measure of the degree of oxidative stability of the material evaluated at a given heating rate and oxidative environment (e.g., oxygen); the higher the OOT value the more stable the material. The OOT is described in Fig. 1. The OOT values can be used for comparative purposes and are not an absolute measurement, like the oxidation induction time (OIT) at a constant temperature (see Test Method E1858). The presence or effectiveness of antioxidants may be determined by these test methods. FIG. 1 DSC Oxidation (Extrapolated) Onset Temperature (OOT) 5.2 Typical uses of these test methods include the oxidative stability of edible oils and fats (oxidative rancidity), lubricants, greases, and polyolefins. SCOPE 1.1 These test methods describe the determination of the oxidative properties of hydrocarbons by differential scanning calorimetry or pressure differential scanning calorimetry under linear heating rate conditions and are applicable to hydrocarbons, which oxidize exothermically in their analyzed form. 1.2 Test Method A—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of oxygen. 1.3 Test Method B—A pressure DSC (PDSC) is used at high pressure (e.g., 3.5 MPa (500 psig) of oxygen). 1.4 Test Method C—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of air. 1.5 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Oxidation onset temperature is a relative measure of the degree of oxidative stability of the material evaluated at a given heating rate and oxidative environment (e.g., oxygen); the higher the OOT value the more stable the material. The OOT is described in Fig. 1. The OOT values can be used for comparative purposes and are not an absolute measurement, like the oxidation induction time (OIT) at a constant temperature (see Test Method E1858). The presence or effectiveness of antioxidants may be determined by these test methods. FIG. 1 DSC Oxidation (Extrapolated) Onset Temperature (OOT) 5.2 Typical uses of these test methods include the oxidative stability of edible oils and fats (oxidative rancidity), lubricants, greases, and polyolefins. SCOPE 1.1 These test methods describe the determination of the oxidative properties of hydrocarbons by differential scanning calorimetry or pressure differential scanning calorimetry under linear heating rate conditions and are applicable to hydrocarbons, which oxidize exothermically in their analyzed form. 1.2 Test Method A—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of oxygen. 1.3 Test Method B—A pressure DSC (PDSC) is used at high pressure (e.g., 3.5 MPa (500 psig) of oxygen). 1.4 Test Method C—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of air. 1.5 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2009-23 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2009-23 has the following relationships with other standards: It is inter standard links to ASTM E473-23b, ASTM E3142-18, ASTM D5483-05(2015), ASTM E473-14, ASTM E691-13, ASTM D3350-12, ASTM D3350-12e1, ASTM E691-11, ASTM E473-11a, ASTM E473-11, ASTM D3350-10a, ASTM D5483-05(2010), ASTM D4565-10, ASTM E473-10, ASTM D3350-10. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2009-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2009 − 23
Standard Test Methods for
Oxidation Onset Temperature of Hydrocarbons by
Differential Scanning Calorimetry
This standard is issued under the fixed designation E2009; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D3350 Specification for Polyethylene Plastics Pipe and Fit-
tings Materials
1.1 These test methods describe the determination of the
D3895 Test Method for Oxidative-Induction Time of Poly-
oxidative properties of hydrocarbons by differential scanning
olefins by Differential Scanning Calorimetry
calorimetry or pressure differential scanning calorimetry under
D4565 Test Methods for Physical and Environmental Per-
linear heating rate conditions and are applicable to
formance Properties of Insulations and Jackets for Tele-
hydrocarbons, which oxidize exothermically in their analyzed
communications Wire and Cable
form.
D5483 Test Method for Oxidation Induction Time of Lubri-
1.2 Test Method A—A differential scanning calorimeter
catingGreasesbyPressureDifferentialScanningCalorim-
(DSC) is used at ambient pressure of one atmosphere of
etry
oxygen.
E473 Terminology Relating to Thermal Analysis and Rhe-
ology
1.3 Test Method B—Apressure DSC (PDSC) is used at high
pressure (e.g., 3.5 MPa (500 psig) of oxygen). E691 Practice for Conducting an Interlaboratory Study to
Determine the Precision of a Test Method
1.4 Test Method C—A differential scanning calorimeter
E967 Test Method for Temperature Calibration of Differen-
(DSC) is used at ambient pressure of one atmosphere of air.
tial Scanning Calorimeters and Differential Thermal Ana-
1.5 Units—The values stated in SI units are to be regarded
lyzers
as standard. No other units of measurement are included in this
E1858 Test Methods for Determining Oxidation Induction
standard.
Time of Hydrocarbons by Differential Scanning Calorim-
1.6 This standard does not purport to address all of the etry
safety concerns, if any, associated with its use. It is the E3142 Test Method for Thermal Lag of Thermal Analysis
responsibility of the user of this standard to establish appro- Apparatus
priate safety, health, and environmental practices and deter-
3. Terminology
mine the applicability of regulatory limitations prior to use.
3.1 Definitions:
1.7 This international standard was developed in accor-
3.1.1 For definitions of terms used in these test methods,
dance with internationally recognized principles on standard-
refer to Terminology E473.
ization established in the Decision on Principles for the
3.2 Definitions of Terms Specific to This Standard:
Development of International Standards, Guides and Recom-
3.2.1 oxidation (extrapolated) onset temperature (OOT)—a
mendations issued by the World Trade Organization Technical
relative measure of oxidative stability at the cited heating rate
Barriers to Trade (TBT) Committee.
is determined from data recorded during a DSC scanning
2. Referenced Documents temperature test. The temperature at which the onset to the
observed oxidation is taken as the OOT.
2.1 ASTM Standards:
4. Summary of Methods
1 4.1 In thermal analysis, a physical property of a material is
These test methods are under the jurisdiction of ASTM Committee E37 on
ThermalMeasurementsandarethedirectresponsibilityofSubcommitteeE37.01on measured either as a function of time at a specified constant
Calorimetry and Mass Loss.
temperature, or more frequently, as a function of temperature
Current edition approved Jan. 1, 2023. Published February 2023. Originally
under conditions of a fixed rate of temperature change. The
ε1
approved in 1999. Last previous edition approved in 2014 as E2009 – 08(2014) .
measured property is the dependent variable, and the measured
DOI: 10.1520/E2009-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
temperature is the independent variable.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
4.2 The test specimen in an aluminum container and an
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. empty reference aluminum container or pan are heated at a
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2009 − 23
specified constant heating rate in an oxygen (or air) environ- 6.2 Temperature sensors are subject to degraded perfor-
ment. Heat flow out of the specimen is monitored as a function mance with age and exposure to the DSC test chamber
of temperature until the oxidative reaction is manifested by atmosphere. Therefore, it is imperative that the apparatus is
heat evolution on the thermal curve. The oxidation (extrapo- temperature calibrated regularly. At a minimum, annual cali-
lated) onset temperature (OOT), a relative measure of oxida- bration is recommended for all instrument signals.
tive stability at the cited heating rate, is determined from data
recorded during the scanning temperature test. The OOT
7. Apparatus
measurementisinitiateduponreachingtheexothermicreaction
7.1 Differential Scanning Calorimeter (DSC) or Pressure
and measuring the extrapolated onset temperature.
Differential Scanning Calorimeter (PDSC)—Multiple genera-
4.3 For some particularly stable materials, the OOT may be
tions of DSCs from numerous commercial suppliers, as well as
quite high (> 300 °C) at the specified heating rate of the
in-house custom apparatus, utilizing a variety of sensor con-
experiment. Under these circumstances, the OOT may be
figurations may be available to the user. While all such
reduced by increasing the pressure of oxygen purge gas.
apparatus capabilities may not be equivalent, for purposes of
Conversely,reducingthepartialpressureofoxygen(suchasby
this test method, any DSC that meets the following criteria
the use of air) may retard reactions that proceed too rapidly,
should be able to generate acceptable results.
with a corresponding increase of the OOT. By admixing
7.1.1 A DSC test chamber composed of: a furnace to
oxygen gas with a suitable diluent, for example, nitrogen, the
provide uniform controlled heating of a specimen and a
OOT will be increased (see Specification D3350 and Test
referencetoaconstantheatingrateofatleast10 °C⁄minwithin
Methods D3895, D4565, and D5483).
the applicable temperature range for these test methods; a
temperature sensor to provide an indication of the specimen
NOTE 1—For some systems, the use of copper pans to catalyze
temperature readable to 60.1 °C; a differential sensor to detect
oxidation will reduce the oxidation onset temperature. The results,
however, will not necessarily correlate with non-catalyzed tests.
heat flow (power) difference between the specimen and the
reference to 0.1 mW; and the instrument should have the
5. Significance and Use
capability of measuring heat flow of at least 6 mW, with
5.1 Oxidation onset temperature is a relative measure of the
provision for less sensitive ranges.
degreeofoxidativestabilityofthematerialevaluatedatagiven
NOTE 2—In certain cases when the sample under study is of high
volatility (e.g., low molecular weight hydrocarbons), the use of pressures
heating rate and oxidative environment (e.g., oxygen); the
≥ 0.1 MPa (1 atmosphere) is needed. The operator is cautioned to verify
higher the OOT value the more stable the material. The OOT
(with apparatus designer) the maximum oxygen pressure at which the
is described in Fig. 1. The OOT values can be used for
apparatus may be safely operated. A PDSC is used in Method B.
comparative purposes and are not an absolute measurement,
7.2 A data collection device to provide a means of
like the oxidation induction time (OIT) at a constant tempera-
acquiring, storing, and displaying measured or calculated
ture (seeTest Method E1858).The presence or effectiveness of
signals,orboth.TheminimumoutputsignalsrequiredforDSC
antioxidants may be determined by these test methods.
are heat flow, temperature, and time.
5.2 Typical uses of these test methods include the oxidative
7.3 A high-pressure gas regulator or similar device to adjust
stability of edible oils and fats (oxidative rancidity), lubricants,
the applied pressure in the test chamber to less than 65%,
greases, and polyolefins.
including any temperature dependence on the transducer, is
6. Interferences used in Method B. (Warning—Use metal free of organic
matter or fluoropolymer tubing with oxygen rather than the
6.1 This test method involves the continuous monitoring of
commonly used rubber or polyvinyl chloride plastic tubing.
the specimen temperature within the test chamber’s enclosed
There have been hazardous situations with prolonged use of
environment of a flowing, static, or self-generated gaseous
certain polymer tubing with oxygen.)
atmosphere (or vacuum) during execution of the stipulated
procedure. In DSC apparatus, the sensor employed to measure
NOTE 3—Gas delivery tubing should be kept as short as possible to
the specimen temperature is not in direct contact with the
minimize dead volume. The link between the test chamber and pressure
transducer should allow fast pressure equilibration to ensure accurate
specimen but is in fixed close thermal contact assumed to be
recording of the pressure above the specimen during testing.
representative of the specimen, such that the measured tem-
perature is that of the sensor itself and the actual specimen 7.4 Specimen containers are aluminum sample pans and
temperature will lag behind this measured temperature during should be inert to the specimen and reference material as well
heating or cooling (seeTest Method E3142).The magnitude of as the oxidizing gas. The specimen containers should be of
this temperature offset depends upon a number of systematic suitable structural shape and integrity to contain the specimen
and random factors including, but not limited to, type and size and reference in accordance with the specific requirements of
of sensor, rate of temperature change, size and thermal con- these test methods, including a pressure system consisting of a
ductance of the specimen, specimen container, and thermal pressure vessel or similar means of sealing the test chamber at
contact between the specimen and the specimen container any applied pressure within the pressure limits required for
during the measurement. To obtain the correct specimen these test methods. The specimen containers should be clean,
temperature,theDSCapparatusmustbetemperaturecalibrated dry, and flat. A typical cylindrical specimen container has the
at equivalent experimental conditions so that the recorded following dimensions: height, (1.5 to 2.5) mm and outer
temperature correctly indicates the specimen temperature. diameter, (5.0 to 7.0) mm.
E2009 − 23
FIG. 1 DSC Oxidation (Extrapolated) Onset Temperature (OOT)
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: E2009 − 08 (Reapproved 2014) E2009 − 23
Standard Test Methods for
Oxidation Onset Temperature of Hydrocarbons by
Differential Scanning Calorimetry
This standard is issued under the fixed designation E2009; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Warning notes were editorially updated throughout in March 2014.
1. Scope
1.1 These test methods describe the determination of the oxidative properties of hydrocarbons by differential scanning calorimetry
or pressure differential scanning calorimetry under linear heating rate conditions and are applicable to hydrocarbons, which oxidize
exothermically in their analyzed form.
1.2 Test Method A—A differential scanning calorimeter (DSC) is used at ambient pressure,pressure of one atmosphere of oxygen.
1.3 Test Method B—A pressure DSC (PDSC) is used at high pressure, for example, pressure (e.g., 3.5 MPa (500 psig) oxygen.of
oxygen).
1.4 Test Method C—A differential scanning calorimeter (DSC) is used at ambient pressure of one atmosphere of air.
1.5 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this
standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D3350 Specification for Polyethylene Plastics Pipe and Fittings Materials
D3895 Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry
D4565 Test Methods for Physical and Environmental Performance Properties of Insulations and Jackets for Telecommunications
Wire and Cable
These test methods are under the jurisdiction of ASTM Committee E37 on Thermal Measurements and are the direct responsibility of Subcommittee E37.01 on
Calorimetry and Mass Loss.
Current edition approved March 15, 2014Jan. 1, 2023. Published April 2014February 2023. Originally approved in 1999. Last previous edition approved in 20082014 as
ε1
E2009 – 08.E2009 – 08(2014) . DOI: 10.1520/E2009-08R14E01.10.1520/E2009-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2009 − 23
D5483 Test Method for Oxidation Induction Time of Lubricating Greases by Pressure Differential Scanning Calorimetry
E473 Terminology Relating to Thermal Analysis and Rheology
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E1858 Test Methods for Determining Oxidation Induction Time of Hydrocarbons by Differential Scanning Calorimetry
E3142 Test Method for Thermal Lag of Thermal Analysis Apparatus
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in these test methods, refer to Terminology E473.
3.2 Definitions of Terms Specific to This Standard:—For definitions of terms used in these test methods, refer to Terminology
E473.
3.2.1 oxidation (extrapolated) onset temperature (OOT)—a relative measure of oxidative stability at the cited heating rate is
determined from data recorded during a DSC scanning temperature test. The temperature at which the onset to the observed
oxidation is taken as the OOT.
4. Summary of Methods
4.1 In thermal analysis, a physical property of a material is measured either as a function of time at a specified constant
temperature, or more frequently, as a function of temperature under conditions of a fixed rate of temperature change. The measured
property is the dependent variable, and the measured temperature is the independent variable.
4.2 The test specimen in an aluminum container and an empty reference aluminum container or pan are heated at a specified
constant heating rate in an oxygen (or air) environment. Heat flow out of the specimen is monitored as a function of temperature
until the oxidative reaction is manifested by heat evolution on the thermal curve. The oxidation (extrapolated) onset temperature
(OOT), a relative measure of oxidative stability at the cited heating rate, is determined from data recorded during the scanning
temperature test. The OOT measurement is initiated upon reaching the exothermic reaction and measuring the extrapolated onset
temperature.
4.3 For some particularly stable materials, the OOT may be quite high (>300°C)(> 300 °C) at the specified heating rate of the
experiment. Under these circumstances, the OOT may be reduced by increasing the pressure of oxygen purge gas. Conversely,
reducing the partial pressure of oxygen (such as by the use of air) may retard reactions that proceed too rapidly, with a
corresponding increase of the OOT. By admixing oxygen gas with a suitable diluent, for example, nitrogen, the OOT will be
increased (see Specification D3350 and Test Methods D3895, D4565, and D5483).
NOTE 1—For some systems, the use of copper pans to catalyze oxidation will reduce the oxidation onset temperature. The results, however, will not
necessarily correlate with non-catalyzed tests.
5. Significance and Use
5.1 Oxidation onset temperature is a relative measure of the degree of oxidative stability of the material evaluated at a given
heating rate and oxidative environment, for example, oxygen; environment (e.g., oxygen); the higher the OOT value the more
stable the material. The OOT is described in Fig. 1. The OOT values can be used for comparative purposes and are not an absolute
measurement, like the oxidation induction time (OIT) at a constant temperature (see Test Method E1858). The presence or
effectiveness of antioxidants may be determined by these test methods.
5.2 Typical uses of these test methods include the oxidative stability of edible oils and fats (oxidative rancidity), lubricants,
greases, and polyolefins.
6. Interferences
6.1 This test method involves the continuous monitoring of the specimen temperature within the test chamber’s enclosed
environment of a flowing, static, or self-generated gaseous atmosphere (or vacuum) during execution of the stipulated procedure.
In DSC apparatus, the sensor employed to measure the specimen temperature is not in direct contact with the specimen but is in
fixed close thermal contact assumed to be representative of the specimen, such that the measured temperature is that of the sensor
E2009 − 23
FIG. 1 DSC Oxidation (Extrapolated) Onset Temperature (OOT), Extrapolated Onset Temperature(OOT)
E2009 − 23
itself and the actual specimen temperature will lag behind this measured temperature during heating or cooling (see Test Method
E3142). The magnitude of this temperature offset depends upon a number of systematic and random factors including, but not
limited to, type and size of sensor, rate of temperature change, size and thermal conductance of the specimen, specimen container,
and thermal contact between the specimen and the specimen container during the measurement. To obtain the correct specimen
temperature, the DSC apparatus must be temperature calibrated at equivalent experimental conditions so that the recorded
temperature correctly indicates the specimen temperature.
6.2 Temperature sensors are subject to degraded performance with age and exposure to the DSC test chamber atmosphere.
Therefore, it is imperative that the apparatus is temperature calibrated regularly. At a minimum, annual calibration is recommended
for all instrument signals.
7. Apparatus
7.1 Differential Scanning Calorimeter (DSC) or Pressure Differential Scanning Calorimeter (PDSC)—The essential instrumen-
tation required to provide the minimum differential scanning calorimetric capability for these test methods includes: a DSC
chamber composed of a furnace to provide uniform controlled heating of a specimen and a reference to a constant heating rate of
at least 10°C/min within the applicable temperature range for these test methods; a temperature sensor to provide an indication of
the specimen temperature to 60.1°C; a differential sensor to detect heat flow (power) difference between the specimen and the
reference to 0.1 mW; and the instrument should have the capability of measuring heat flow of at least 6 mW, with provision for
less sensitive ranges.Multiple generations of DSCs from numerous commercial suppliers, as well as in-house custom apparatus,
utilizing a variety of sensor configurations may be available to the user. While all such apparatus capabilities may not be equivalent,
for purposes of this test method, any DSC that meets the following criteria should be able to generate acceptable results.
NOTE 2—In certain cases when the sample under study is of high volatility (for example, low molecular weight hydrocarbons), the use of pressures in
excess of 0.1 MPa (1 atmosphere) is needed. The operator is cautioned to verify (with apparatus designer) the maximum oxygen pressure at which the
apparatus may be safely operated. A PDSC is used in Method B.
7.1.1 A DSC test chamber composed of: a furnace to provide uniform controlled heating of a specimen and a reference to a
constant heating rate of at least 10 °C ⁄min within the applicable temperature range for these test methods; a temperature sensor
to provide an indication of the specimen temperature readable to 60.1 °C; a differential sensor to detect heat flow (power)
difference between the specimen and the reference to 0.1 mW; and the instrument should have the capability of measuring heat
flow of at least 6 mW, with provision for less sensitive ranges.
NOTE 2—In certain cases when the sample under study is of high volatility (e.g., low molecular weight hydrocarbons), the use of pressures ≥ 0.1 MPa
(1 atmosphere) is needed. The operator is cautioned to verify (with apparatus designer) the maximum oxygen pressure at which the apparatus may be
safely operated. A PDSC is used in Method B.
7.2 A Data Collection Device,data collection device to provide a means of acquiring, storing, and displaying measured or
calculated signals, or both. The minimum output signals required for DSC are heat flow, temperature, and time.
7.3 A high-pressure gas regulator or similar device to adjust the applied pressure in the test chamber to less than 65 %, including
any temperature dependence on the transducer, is used in Method B. (Warning—Use metal free of organic matter or fluoropolymer
tubing with oxygen rather than the commonly used rubber or polyvinyl chloride plastic tubing. There have been hazardous
situations with prolonged use of certain polymer tubing in oxygen service.with oxygen.)
NOTE 3—Gas delivery tubing should be kept as short as possible to minimize dead volume. The link between the test chamber and pressure transducer
should allow fast pressure equilibration to ensure accurate recording of the pressure above the specimen during testing.
7.4 Specimen containers are aluminum sample pans and should be inert to the specimen and reference material as well as the
oxidizing gas. The specimen containers should be of suitable structural shape and integrity to contain
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...