ASTM E2041-23
(Test Method)Standard Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels Method
Standard Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels Method
SIGNIFICANCE AND USE
6.1 This test method is useful in research and development.
6.2 The determination of the appropriate model for a chemical reaction or transformation and the values associated with its kinetic parameters may be used in the estimation of reaction performance at temperatures or time conditions not easily tested. This use, however, is not described in this test method.
SCOPE
1.1 This test method describes the determination of the kinetic parameters of activation energy, Arrhenius pre-exponential factor, and reaction order using the Borchardt and Daniels2 treatment of data obtained by differential scanning calorimetry. This test method is applicable to the temperature range from 170 K to 870 K (−100 °C to 600°C).
1.2 This treatment is applicable only to smooth exothermic reactions with no shoulders, discontinuous changes, or shifts in baseline. It is applicable only to reactions with reaction order n ≤ 2. It is not applicable to acceleratory reactions and, therefore, is not applicable to the determination of kinetic parameters for most thermoset curing reactions or to crystallization reactions.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jul-2023
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 15-Mar-2024
- Effective Date
- 15-Nov-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 15-Jan-2018
- Effective Date
- 01-Nov-2016
- Effective Date
- 15-Feb-2016
- Effective Date
- 01-May-2015
- Effective Date
- 01-Mar-2015
- Effective Date
- 01-Feb-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 15-Feb-2014
Overview
ASTM E2041-23: Standard Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels Method provides a robust, standardized approach for determining key kinetic parameters-activation energy, Arrhenius pre-exponential factor, and reaction order-from data collected by differential scanning calorimetry (DSC). Established by ASTM International, this method uses the Borchardt and Daniels model and is applicable to smooth exothermic reactions with no shoulders or discontinuities in the temperature range of 170 K to 870 K (−100 °C to 600 °C).
This method is widely utilized in research and development for characterizing reaction kinetics, especially in fields such as material science, chemistry, and pharmaceuticals. Accurate estimation of kinetic parameters enables researchers to better understand reaction mechanisms and to predict material performance under varying thermal conditions.
Key Topics
- Kinetic Parameter Estimation: The standard details how to calculate activation energy, reaction order (n ≤ 2), and Arrhenius pre-exponential factor from DSC data using the Borchardt and Daniels method.
- Applicable Reactions: Suitable for smooth, single-step exothermic reactions only. Not applicable to acceleratory reactions or complex behaviors like most thermoset curing or crystallization reactions.
- Experimental Requirements:
- Uses linear heating via DSC with specified apparatus and calibration requirements.
- Requires small, homogenous specimen sizes and low heating rates to ensure temperature equilibrium and measurement accuracy.
- Emphasizes the use of SI units throughout procedures and reporting.
- Data Analysis: Specifies both baseline construction and data treatment procedures, including the use of linear regression to extract kinetic parameters from integrated heat flow measurements.
Applications
Research and Development:
ASTM E2041-23 is critical for R&D teams investigating chemical reactions and material transformations. By enabling accurate determination of kinetic parameters, researchers can:
- Characterize new chemical substances for stability and reactivity.
- Investigate thermal behavior of advanced materials.
- Develop predictive models for reaction behavior outside easily testable temperature or time ranges (though this prediction use is beyond the standard's direct scope).
Material and Chemical Analysis:
This standard finds direct application in:
- Polymers and composites development (for qualifying reaction order and kinetics, provided reactions meet the method's criteria).
- Pharmaceutical studies requiring thermal stability or kinetic profiling.
- Quality control for chemicals sensitive to exothermic reactions.
Related Standards
Industry professionals using ASTM E2041-23 may also refer to the following standards for supporting measurements or alternative approaches:
- ASTM E473: Terminology Relating to Thermal Analysis and Rheology.
- ASTM E698: Test Method for Kinetic Parameters for Thermally Unstable Materials Using DSC and the Flynn/Wall/Ozawa Method.
- ASTM E537: Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry.
- ASTM E967: Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers.
- ASTM E1970: Practice for Statistical Treatment of Thermoanalytical Data.
- ASTM E3142: Test Method for Thermal Lag of Thermal Analysis Apparatus.
- ISO 11357-5: Determination of Temperature and/or Time of Reaction and Reaction Kinetics by DSC (an international method with additional provisions).
Practical Value
By following the ASTM E2041-23 standard, laboratories ensure consistent, reliable determination of kinetic parameters essential for safe chemical handling, product development, and regulatory compliance. This test method enhances comparability of results across different labs and instruments, increasing confidence in material characterization and process design. Applying this standard supports robust kinetic analysis, contributing to safer and more efficient research, manufacturing, and quality control processes.
Buy Documents
ASTM E2041-23 - Standard Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels Method
REDLINE ASTM E2041-23 - Standard Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels Method
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E2041-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels Method". This standard covers: SIGNIFICANCE AND USE 6.1 This test method is useful in research and development. 6.2 The determination of the appropriate model for a chemical reaction or transformation and the values associated with its kinetic parameters may be used in the estimation of reaction performance at temperatures or time conditions not easily tested. This use, however, is not described in this test method. SCOPE 1.1 This test method describes the determination of the kinetic parameters of activation energy, Arrhenius pre-exponential factor, and reaction order using the Borchardt and Daniels2 treatment of data obtained by differential scanning calorimetry. This test method is applicable to the temperature range from 170 K to 870 K (−100 °C to 600°C). 1.2 This treatment is applicable only to smooth exothermic reactions with no shoulders, discontinuous changes, or shifts in baseline. It is applicable only to reactions with reaction order n ≤ 2. It is not applicable to acceleratory reactions and, therefore, is not applicable to the determination of kinetic parameters for most thermoset curing reactions or to crystallization reactions. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 6.1 This test method is useful in research and development. 6.2 The determination of the appropriate model for a chemical reaction or transformation and the values associated with its kinetic parameters may be used in the estimation of reaction performance at temperatures or time conditions not easily tested. This use, however, is not described in this test method. SCOPE 1.1 This test method describes the determination of the kinetic parameters of activation energy, Arrhenius pre-exponential factor, and reaction order using the Borchardt and Daniels2 treatment of data obtained by differential scanning calorimetry. This test method is applicable to the temperature range from 170 K to 870 K (−100 °C to 600°C). 1.2 This treatment is applicable only to smooth exothermic reactions with no shoulders, discontinuous changes, or shifts in baseline. It is applicable only to reactions with reaction order n ≤ 2. It is not applicable to acceleratory reactions and, therefore, is not applicable to the determination of kinetic parameters for most thermoset curing reactions or to crystallization reactions. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2041-23 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2041-23 has the following relationships with other standards: It is inter standard links to ASTM E2781-24, ASTM E1445-08(2023), ASTM E473-23b, ASTM E1142-23b, ASTM E537-20, ASTM E3142-18, ASTM E2781-16, ASTM E1641-16, ASTM E1142-15, ASTM E1641-15, ASTM E1445-08(2015), ASTM E473-14, ASTM E1142-14b, ASTM E1142-14a, ASTM E1142-14. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2041-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2041 − 23
Standard Test Method for
Estimating Kinetic Parameters by Differential Scanning
Calorimeter Using the Borchardt and Daniels Method
This standard is issued under the fixed designation E2041; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method describes the determination of the
E473 Terminology Relating to Thermal Analysis and Rhe-
kinetic parameters of activation energy, Arrhenius pre-
ology
exponential factor, and reaction order using the Borchardt and
E537 Test Method for Thermal Stability of Chemicals by
Daniels treatment of data obtained by differential scanning
Differential Scanning Calorimetry
calorimetry. This test method is applicable to the temperature
E698 Test Method for Kinetic Parameters for Thermally
range from 170 K to 870 K (−100 °C to 600°C).
Unstable Materials Using Differential Scanning Calorim-
1.2 This treatment is applicable only to smooth exothermic
etry and the Flynn/Wall/Ozawa Method
reactions with no shoulders, discontinuous changes, or shifts in
E967 Test Method for Temperature Calibration of Differen-
baseline. It is applicable only to reactions with reaction order
tial Scanning Calorimeters and Differential Thermal Ana-
n ≤ 2. It is not applicable to acceleratory reactions and, lyzers
therefore, is not applicable to the determination of kinetic
E968 Practice for Heat Flow Calibration of Differential
parameters for most thermoset curing reactions or to crystalli- Scanning Calorimeters (Withdrawn 2023)
zation reactions. E1142 Terminology Relating to Thermophysical Properties
E1445 Terminology Relating to Hazard Potential of Chemi-
1.3 The values stated in SI units are to be regarded as
cals
standard. No other units of measurement are included in this
E1641 Test Method for Decomposition Kinetics by Thermo-
standard.
gravimetry Using the Ozawa/Flynn/Wall Method
E1970 Practice for Statistical Treatment of Thermoanalytical
1.4 This standard does not purport to address all of the
Data
safety concerns, if any, associated with its use. It is the
E2781 Practice for Evaluation of Methods for Determination
responsibility of the user of this standard to establish appro-
of Kinetic Parameters by Calorimetry and Differential
priate safety, health, and environmental practices and deter-
Scanning Calorimetry
mine the applicability of regulatory limitations prior to use.
E3142 Test Method for Thermal Lag of Thermal Analysis
1.5 This international standard was developed in accor-
Apparatus
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
3. Terminology
Development of International Standards, Guides and Recom-
3.1 Definitions—Specific technical terms used in this test
mendations issued by the World Trade Organization Technical
method are defined in Terminologies E473, E1142, and E1445,
Barriers to Trade (TBT) Committee.
including calibration, calorimeter, differential scanning
calorimetry, enthalpy, peak, reaction, repeatability,
reproducibility, and slope.
This test method is under the jurisdiction of ASTM Committee E37 on Thermal
Measurements and the direct responsibility of Subcommittee E37.01 on Calorimetry
and Mass Loss.
Current edition approved Aug. 1, 2023. Published August 2023. Originally For referenced ASTM standards, visit the ASTM website, www.astm.org, or
approved in 1999. Last previous edition approved in 2018 as E2041 – 13 (2018). contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
DOI: 10.1520/E2041-23. Standards volume information, refer to the standard’s Document Summary page on
Borchardt, H.J., Daniels, F., “The Application of Differential Thermal Analysis the ASTM website.
to the Study of Reaction Kinetics”, Journal of the American Chemical Society, Vol The last approved version of this historical standard is referenced on
79, 1957, pp. 41–46. www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2041 − 23
4. Summary of Test Method The resultant equation has the form z = a + bx + cy (where
z ≡ ln[dα/dt], ln[Z] ≡ a, b ≡ n, x ≡ ln[1 − α], c ≡ E/R, and y ≡
4.1 A test specimen is heated at a linear rate in a differential
l/T) and may be solved using multiple linear regression data
scanning calorimeter or other suitable calorimeter through a
treatment.
region of exothermic reaction behavior. The rate of heat
evolution, developed by a chemical reaction, is proportional to 5.6 The values for dα/dt, (1 − α) and T needed to solve Eq
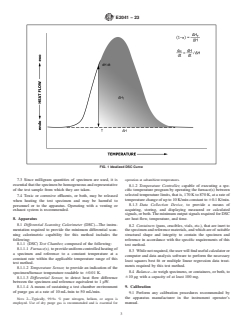
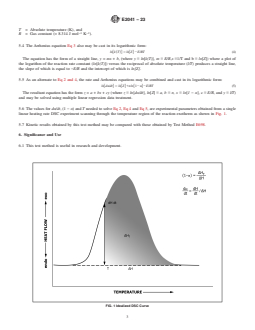
the rate of reaction. Integration of the heat flow as a function of 2, Eq 4 and Eq 5, are experimental parameters obtained from
time yields the total heat of a reaction. a single linear heating rate DSC experiment scanning through
the temperature region of the reaction exotherm as shown in
4.2 The Borchardt and Daniels data treatment is used to
Fig. 1.
derive the kinetic parameters of activation energy, Arrhenius
pre-exponential factor, and reaction order from the heat flow 5.7 Kinetic results obtained by this test method may be
and total heat of reaction information obtained in 4.1 (see compared with those obtained by Test Method E698.
Section 5).
6. Significance and Use
5. Basis of Methodology
6.1 This test method is useful in research and development.
5.1 Kinetic reactions may be modeled with a number of
6.2 The determination of the appropriate model for a chemi-
suitable equations. The Borchardt and Daniels method makes
cal reaction or transformation and the values associated with its
use of the rate equation to describe the dependence of the rate
kinetic parameters may be used in the estimation of reaction
of reaction on the amount of material present.
performance at temperatures or time conditions not easily
n
tested. This use, however, is not described in this test method.
dα/dt 5 k T 1 2 α (1)
~ ! ~ !
7. Interferences
where:
−1
dα/dt = reaction rate (min )
7.1 Because of its simplicity and ease of use, the Borchardt
α = fraction reacted (dimensionless),
and Daniels method is often the method of choice for
−1
k(T) = rate constant at temperature T (min ), and
characterization of the kinetic parameters of a reaction system.
n = reaction order (dimensionless).
The Borchardt and Daniels method, like all tools used to
evaluate kinetic parameters, is not applicable to all cases. The
5.2 For a reaction conducted at temperature (T), the rate
user of this test method is expressly advised to use this test
equation of Eq 1, may be cast in its logarithmic form:
method and its results with caution.
ln@dα/dt# 5 ln@k T #1nln@1 2 α# (2)
~ !
7.2 Tabulated below are some guidelines for the use of the
This equation has the form of a straight line, y = mx + b,
Borchardt and Daniels method.
where a plot of the logarithm of the reaction rate (ln[dα/dt])
7.2.1 The approach is applicable only to exothermic reac-
versus the logarithm of the fraction remaining ln[1 − α] yields
tions.
a straight line, the slope of which is equal to n and the intercept
NOTE 1—Endothermic reactions are controlled by the kinetics of the
is equal to ln[k(T)].
heat transfer of the apparatus and not by the kinetics of the reaction.
5.3 The Borchardt and Daniels model also makes use of the
7.2.2 The reaction under investigation must have a constant
Arrhenius equation to describe how the reaction rate changes
mechanism throughout the whole reaction process. In practice,
as a function of temperature:
this means that the reaction exotherm upon heating must be
·E/RT
k~T! 5 Z e (3)
smooth, well-shaped (as in Fig. 1) with no shoulders, multiple
peaks or discontinuous steps.
where:
−1 7.2.3 The reaction must be nth order. Confirmation of an nth
Z = Arrhenius pre-exponential factor (min ),
−1
order reaction may be made by an isothermal experiment such
E = Activation energy (J mol ),
as that described in Appendix X1.
T = Absolute temperature (K), and
−1 −1
7.2.4 Typical reactions which are not nth order and to which
R = Gas constant (= 8.314 J mol K ).
Borchardt and Daniels kinetic may not be applied for predic-
5.4 The Arrhenius equation Eq 3 also may be cast in its
tive purposes include many thermoset curing reactions and
logarithmic form:
crystallization transformations.
ln@k~T!# 5 ln@Z# 2 E/RT (4)
7.2.5 The nth order kinetic reactions anticipate that the
value of n will be small, non-zero integers, such as 1 or 2.
The equation has the form of a straight line, y = mx + b,
Values of n greater than 2 or that are not simple fractions, such
(where y ≡ ln[k(T)], m ≡ E/R, x ≡1/T and b ≡ ln[Z]) where a plot
as ⁄2 = 0.5, are highly unlikely and shall be viewed with
of the logarithm of the reaction rate constant (ln[k(T)]) versus
caution.
the reciprocal of absolute temperature (l/T) produces a straight
7.2.6 The Borchardt and Daniels method assumes tempera-
line, the slope of which is equal to −E/R and the intercept of
ture equilibrium throughout the whole test specimen. This
which is ln[Z].
means that low heating rates, (that is, <10 K/min), small
5.5 As an alternate to Eq 2 and 4, the rate and Arrhenius
specimen sizes (<5 mg) and highly conductive sealed specimen
equations may be combined and cast in its logarithmic form:
containers, for example, aluminum, gold, platinum, etc., should
ln dα/dt 5 ln Z 1nln 1 2 α 2 E/RT (5) be used.
@ # @ # @ #
E2041 − 23
FIG. 1 Idealized DSC Curve
operation at subambient temperatures.
7.3 Since milligram quantities of specimen are used, it is
essential that the specimen be homogeneous and representative
8.1.2 Temperature Controller, capable of executing a spe-
of the test sample from which they are taken.
cific temperature program by operating the furnace(s) between
selected temperature limits, that is, 170 K to 870 K, at a rate of
7.4 Toxic or corrosive effluents, or both, may be released
temperature change of up to 10 K/min constant to 60.1 K/min.
when heating the test specimen and may be harmful to
8.1.3 Data Collection Device, to provide a means of
personnel or to the apparatus. Operating with a venting or
acquiring, storing, and displaying measured or calculated
exhaust system is recommended.
signals, or both. The minimum output signals required for DSC
8. Apparatus
are heat flow, temperature, and time.
8.1 Differential Scanning Calorimeter (DSC)—The instru-
8.2 Containers (pans, crucibles, vials, etc.), that are inert to
mentation required to provide the minimum differential scan-
the specimen and reference materials, and which are of suitable
ning calorimetric capability for this method includes the
structural shape and integrity to contain the specimen and
following:
reference in accordance with the specific requirements of this
8.1.1 (DSC) Test Chamber, composed of the following:
test method.
8.1.1.1 Furnace(s), to provide uniform controlled heating of
8.3 While not required, the user will find useful calculator or
a specimen and reference to a constant temperature at a
computer and data analysis software to perform the necessary
constant rate within the applicable temperature range of this
least squares best fit or multiple linear regression data treat-
test method.
ments required by this test method.
8.1.1.2 Temperature Sensor, to provide an indication of the
8.4 Balance—to weigh specimens, or containers, or both, to
specimen/furnace temperature readable to 60.01 K.
610 μg with a capacity of at least 100 mg.
8.1.1.3 Differential Sensor, to detect heat flow difference
between the specimen and reference equivalent to 1 μW.
9. Calibration
8.1.1.4 A means of sustaining a test chamber environment
of purge gas at a rate of 10 mL ⁄min to 50 mL/min.
9.1 Perform any calibration procedures recommended by
the apparatus manufacturer in the instrument operator’s
NOTE 2—Typically, 99.9+ % pure nitrogen, helium, or argon is
employed. Use of dry purge gas is recommended and is essential for manual.
E2041 − 23
9.2 Calibrate the DSC temperature signal over the range of 11.5 Select a temperature interval which provides a mini-
the reaction using Test Method E967 (see Appendix X3). mum of ten equally-spaced values between the temperature
limits determined in 11.4.
9.3 Calibrate the DSC heat flow signal using Practice E968.
11.6 At each of the ten temperatures identified in 11.5,
NOTE 3—Committee E37 recommends calibration, or calibration
record the rate of reaction (dH/dt ) in mW, temperature (T) in
verification, of all signals at least annually.
K and heat of reaction remaining (ΔH ) in mJ as illustrated in
T
10. Procedure
Fig. 1.
10.1 Weigh 1 mg to 10 mg of test specimen to a precision of
NOTE 7—It is convenient to prepare a table of these values.
610 μg into a sample container and hermetically seal the
11.7 For each of the fractional areas obtained in 11.6,
container. Weigh the specimen and container to 610 μg. Load
determine the fraction remaining (1 − α) and the fractional rate
the test specimen into the apparatus using an equivalent empty
of reaction (dα/dt) using the following equation:
specimen container as the reference. Close the DSC sample
chamber and prepare the apparatus for an experimental run. ~1 2 α! 5 ΔH /ΔH (6)
T
dα/dt 5 dH/dt /ΔH (7)
~ !
NOTE 4—This test method is based upon a “non-self heating” assump-
NOTE 8—In this and all subsequent calculations, retain all available
tion. Combinations of specimen size and reaction kinetics that produce
heat flow greater than 8 mW fail this assumption and produce erroneous significant figures rounding only the final result to the number of
significant figures described in Section 13.
results. Small specimen sizes may be used to obtain this critical non-self
heating assumption. NOTE 9—The values for (1 − α) should range between 0.9 and 0.1
depending upon the values selected in 11.4 and 11.5
10.2 Equilibrate the specimen at a temperature 40 K below
11.8 Calculate the reciprocal of absolute temperature for
the first exothermic behavior.
each value determined in 11.6 and 11.7 (see Note 8).
NOTE 5—This temperature may be determined from a previously
recorded exploratory run using Test Method E537.
NOTE 10—Often, it is conve
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2041 − 13 (Reapproved 2018) E2041 − 23
Standard Test Method for
Estimating Kinetic Parameters by Differential Scanning
Calorimeter Using the Borchardt and Daniels Method
This standard is issued under the fixed designation E2041; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method describes the determination of the kinetic parameters of activation energy, Arrhenius pre-exponential factor,
and reaction order using the Borchardt and Daniels treatment of data obtained by differential scanning calorimetry. This test
method is applicable to the temperature range from 170170 K to 870 K (−100(−100 °C to 600°C).
1.2 This treatment is applicable only to smooth exothermic reactions with no shoulders, discontinuous changes, or shifts in
baseline. It is applicable only to reactions with reaction order
n ≤ 2. It is not applicable to acceleratory reactions and, therefore, is not applicable to the determination of kinetic parameters for
most thermoset curing reactions or to crystallization reactions.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This test method is similar, but not equivalent to, ISO 11357, Part 5, that contains provisions for additional information not
supplied by this test method.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E473 Terminology Relating to Thermal Analysis and Rheology
E537 Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry
E698 Test Method for Kinetic Parameters for Thermally Unstable Materials Using Differential Scanning Calorimetry and the
Flynn/Wall/Ozawa Method
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and the direct responsibility of Subcommittee E37.01 on Calorimetry and
Mass Loss.
Current edition approved April 1, 2018Aug. 1, 2023. Published May 2018August 2023. Originally approved in 1999. Last previous edition approved in 20132018 as
ε1
E2041 – 13 (2018). . DOI: 10.1520/E2041-13R18.10.1520/E2041-23.
Borchardt, H.J., Daniels, F., “The Application of Differential Thermal Analysis to the Study of Reaction Kinetics”, Journal of the American Chemical Society, Vol 79,
1957, pp. 41–46.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2041 − 23
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E968 Practice for Heat Flow Calibration of Differential Scanning Calorimeters (Withdrawn 2023)
E1142 Terminology Relating to Thermophysical Properties
E1445 Terminology Relating to Hazard Potential of Chemicals
E1641 Test Method for Decomposition Kinetics by Thermogravimetry Using the Ozawa/Flynn/Wall Method
E1970 Practice for Statistical Treatment of Thermoanalytical Data
E2781 Practice for Evaluation of Methods for Determination of Kinetic Parameters by Calorimetry and Differential Scanning
Calorimetry
E3142 Test Method for Thermal Lag of Thermal Analysis Apparatus
2.2 ISO Standards:
ISO 11357 Part 5: Determination of Temperature and/or Time of Reaction and Reaction Kinetics
3. Terminology
3.1 Definitions—Specific technical terms used in this test method are defined in Terminologies E473, E1142, and E1445, including
calibration, calorimeter, differential scanning calorimetry, enthalpy, peak, reaction, repeatability, reproducibility, and slope.
4. Summary of Test Method
4.1 A test specimen is heated at a linear rate in a differential scanning calorimeter or other suitable calorimeter through a region
of exothermic reaction behavior. The rate of heat evolution, developed by a chemical reaction, is proportional to the rate of
reaction. Integration of the heat flow as a function of time yields the total heat of a reaction.
4.2 The Borchardt and Daniels data treatment is used to derive the kinetic parameters of activation energy, Arrhenius
pre-exponential factor, and reaction order from the heat flow and total heat of reaction information obtained in 4.1 (see Section
5).
5. Basis of Methodology
5.1 Kinetic reactions may be modeled with a number of suitable equations. The Borchardt and Daniels method makes use of the
rate equation to describe the dependence of the rate of reaction on the amount of material present.
n
dα/dt 5 k~T! ~12 α! (1)
where:
−1
dα/dt = reaction rate (min )
α = fraction reacted (dimensionless),
−1
k(T) = rate constant at temperature T (min ), and
n = reaction order (dimensionless).
5.2 For a reaction conducted at temperature (T), the rate equation of Eq 1, may be cast in its logarithmic form:
ln dα/dt 5 ln k T 1nln 12α (2)
@ # @ ~ !# @ #
This equation has the form of a straight line, y = mx + b, where a plot of the logarithm of the reaction rate (ln[dα/dt]) versus
the logarithm of the fraction remaining ln[1 − α] yields a straight line, the slope of which is equal to n and the intercept is equal
to ln[k(T)].
5.3 The Borchardt and Daniels model also makes use of the Arrhenius equation to describe how the reaction rate changes as a
function of temperature:
·E/RT
k~T! 5 Z e (3)
where:
−1
Z = Arrhenius pre-exponential factor (min ),
−1
E = Activation energy (J mol ),
The last approved version of this historical standard is referenced on www.astm.org.
E2041 − 23
T = Absolute temperature (K), and
−1 −1
R = Gas constant (= 8.314 J mol K ).
5.4 The Arrhenius equation Eq 3 also may be cast in its logarithmic form:
ln@k~T!# 5 ln@Z# 2E/RT (4)
The equation has the form of a straight line, y = mx + b, (where y ≡ ln[k(T)], m ≡ E/R,x ≡1/T and b ≡ ln[Z]) where a plot of
the logarithm of the reaction rate constant (ln[k(T)]) versus the reciprocal of absolute temperature (l/T) produces a straight line,
the slope of which is equal to −E/R and the intercept of which is ln[Z].
5.5 As an alternate to Eq 2 and 4, the rate and Arrhenius equations may be combined and cast in its logarithmic form:
ln@dα/dt# 5 ln@Z#1nln@12α#2E/RT (5)
The resultant equation has the form z = a + bx + cy (where z ≡ ln[dα/dt], ln[Z] ≡ a, b ≡ n, x ≡ ln[1 − α], c ≡ E/R, and y ≡ l/T)
and may be solved using multiple linear regression data treatment.
5.6 The values for dα/dt, (1 − α) and T needed to solve Eq 2, Eq 4 and Eq 5, are experimental parameters obtained from a single
linear heating rate DSC experiment scanning through the temperature region of the reaction exotherm as shown in Fig. 1.
5.7 Kinetic results obtained by this test method may be compared with those obtained by Test Method E698.
6. Significance and Use
6.1 This test method is useful in research and development.
FIG. 1 Idealized DSC Curve
E2041 − 23
6.2 The determination of the appropriate model for a chemical reaction or transformation and the values associated with its kinetic
parameters may be used in the estimation of reaction performance at temperatures or time conditions not easily tested. This use,
however, is not described in this test method.
7. Interferences
7.1 Because of its simplicity and ease of use, the Borchardt and Daniels method is often the method of choice for characterization
of the kinetic parameters of a reaction system. The Borchardt and Daniels method, like all tools used to evaluate kinetic parameters,
is not applicable to all cases. The user of this test method is expressly advised to use this test method and its results with caution.
7.2 Tabulated below are some guidelines for the use of the Borchardt and Daniels method.
7.2.1 The approach is applicable only to exothermic reactions.
NOTE 1—Endothermic reactions are controlled by the kinetics of the heat transfer of the apparatus and not by the kinetics of the reaction.
7.2.2 The reaction under investigation must have a constant mechanism throughout the whole reaction process. In practice, this
means that the reaction exotherm upon heating must be smooth, well shaped well-shaped (as in Fig. 1) with no shoulders, multiple
peaks or discontinuous steps.
7.2.3 The reaction must be nth order. Confirmation of an nth order reaction may be made by an isothermal experiment such as
that described in Appendix X1.
7.2.4 Typical reactions which are not nth order and to which Borchardt and Daniels kinetic may not be applied for predictive
purposes include many thermoset curing reactions and crystallization transformations.
7.2.5 The nth order kinetic reactions anticipate that the value of n will be small, non-zero integers, such as 1 or 2. Values of n
greater than 2 or that are not simple fractions, such as ⁄2 = 0.5, are highly unlikely and shall be viewed with caution.
7.2.6 The Borchardt and Daniels method assumes temperature equilibrium throughout the whole test specimen. This means that
low heating rates, (that is, <10 K/min), small specimen sizes (<5 mg) and highly conductive sealed specimen containers, for
example, aluminum, gold, platinum, etc., should be used.
7.3 Since milligram quantities of specimen are used, it is essential that the specimen be homogeneous and representative of the
test sample from which they are taken.
7.4 Toxic or corrosive effluents, or both, may be released when heating the test specimen and may be harmful to personnel or to
the apparatus. Operating with a venting or exhaust system is recommended.
8. Apparatus
8.1 Differential Scanning Calorimeter (DSC)—(DSC)—The instrumentation required to provide the minimum differential
scanning calorimetric capability for this method includes the following:
8.1.1 DSC(DSC) Test Chamber, composed of the following:
8.1.1.1 Furnace(s), to provide uniform controlled heating of a specimen and reference to a constant temperature at a constant rate
within the applicable temperature range of this test method.
8.1.1.2 Temperature Sensor, to provide an indication of the specimen/furnace temperature readable to 60.01 K.
8.1.1.3 Differential Sensor, to detect heat flow difference between the specimen and reference equivalent to 1 μW.
8.1.1.4 A means of sustaining a test chamber environment of purge gas at a rate of 1010 mL ⁄min to 50 mL/min.
E2041 − 23
NOTE 2—Typically, 99.9+ % pure nitrogen, helium, or argon is employed. Use of dry purge gas is recommended and is essential for operation at
subambient temperatures.
8.1.2 Temperature Controller, capable of executing a specific temperature program by operating the furnace(s) between selected
temperature limits, that is, 170170 K to 870 K, at a rate of temperature change of up to 10 K/min constant to 60.1 K/min.
8.1.3 Data Collection Device, to provide a means of acquiring, storing, and displaying measured or calculated signals, or both.
The minimum output signals required for DSC are heat flow, temperature, and time.
8.2 Containers (pans, crucibles, vials, etc.), that are inert to the specimen and reference materials, and which are of suitable
structural shape and integrity to contain the specimen and reference in accordance with the specific requirements of this test
method.
8.3 While not required, the user will find useful calculator or computer and data analysis software to perform the necessary least
squares best fit or multiple linear regression data treatments required by this test method.
8.4 Balance—to weigh specimens, or containers, or both, to 610 μg with a capacity of at least 100 mg.
9. Calibration
9.1 Perform any calibration procedures recommended by the apparatus manufacturer in the instrument operator’s manual.
9.2 Calibrate the DSC temperature signal over the range of the reaction using Test Method E967. (see Appendix X3).
9.3 Calibrate the DSC heat flow signal using Practice E968.
NOTE 3—Committee E37 recommends calibration, or calibration verification, of all signals at least annually.
10. Procedure
10.1 Weigh 11 mg to 10 mg of test specimen to a precision of
610 μg into a sample container and hermetically seal the container. Weigh the specimen and container to 610 μg. Load the test
specimen into the apparatus using an equivalent empty specimen container as the reference. Close the DSC sample chamber and
prepare the apparatus for an experimental run.
NOTE 4—This test method is based upon a “non-self heating” assumption. Combinations of specimen size and reaction kinetics that produce heat flow
greater than 8 mW fail this assumption and produce erroneous results. Small specimen sizes may be used to obtain this critical non-self heating
assumption.
10.2 Equilibrate the specimen at a temperature 40 K below the first exothermic behavior.
NOTE 5—This temperature may be determined from a previously recorded exploratory run using Test Method E537.
10.3 Heat the test specimen at a rate of 5 K/min to a temperature 10 K higher than the completion of the exothermic reaction as
indicated by the return to baseline. Record the heat flow and sample temperature throughout this region.
NOTE 6—Other heating rates (<10 K/min) may be used but shall be indicated in the report. Agreement of results undertaken at several heating rates will
provide confidence in the method and efficacy of the results.
10.4 Cool the specimen container to ambient temperature and reweigh. Record and report any change in mass from that observed
in 10.1 prior to the test.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.Lyon, R. E., Safronava, N., Sense,
J., Stoliarov, S. I., “Thermokinetic Model of Sample Response in Nonisothermal Analysis,” Thermochimica Acta, Vol. 545, 2012, pp. 82 – 89.
----------------
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...