ASTM F2579-18
(Specification)Standard Specification for Amorphous Poly(lactide) and Poly(lactide-co-glycolide) Resins for Surgical Implants
Standard Specification for Amorphous Poly(lactide) and Poly(lactide-co-glycolide) Resins for Surgical Implants
ABSTRACT
This specification covers amorphous poly(lactide) and poly(lactide-co-glycolide) resins used in the manufacture of surgical implants. Materials covered by this specification are virgin poly(lactide) and poly(lactide-co-glycolide) resins that can be fully solvated at room temperature by methylene chloride (dichloromethane) or chloroform (trichloromethane). The poly(d,l-lactide) homopolymers are amorphous and shall be composed of meso-lactide or equimolar (racemic) combinations of d-lactide and l-lactide. The poly(d,l-lactide-co-glycolide) copolymers are amorphous and shall be composed of a combination of glycolide and either meso-lactide or a racemic combination of d-lactide and l-lactide. The resins shall be manufactured in pellet, granular, powder, flake, or other form and shall conform to the chemical and physical property requirements specified. Tests for chemical identification (by infrared, proton nuclear magnetic resonance, and carbon-13 nuclear magnetic resonance spectroscopy), specific rotation, molar mass, and residual monomer, residual solvent, and heavy metal content shall be performed and shall conform to the requirements specified. Additional tests for residual catalyst and residual water content may be performed as well.
SCOPE
1.1 This specification covers virgin amorphous poly(lactide) homopolymer and poly(lactide-co-glycolide) copolymer resins intended for use in surgical implants. The poly(dl-lactide) homopolymers covered by this specification are considered to be amorphous (that is, void of crystallinity) and are polymerized either from meso-lactide or from equimolar (racemic) combinations of d-lactide and l-lactide. The poly(dl-lactide-co-glycolide) copolymers covered by this specification are also considered to be amorphous and are co-polymerized from a combination of glycolide and either meso-lactide or racemic quantities of d-lactide and l-lactide, and typically possess nominal mole fractions that equal or exceed 50 % lactide.
1.2 Since poly(glycolide) is commonly abbreviated as PGA for poly(glycolic acid) and poly(lactide) is commonly abbreviated as PLA for poly(lactic acid), these polymers are commonly referred to as PGA, PLA, and PLA:PGA resins for the hydrolytic byproducts to which they respectively degrade. PLA is a term that carries no stereoisomeric specificity and therefore encompasses both the amorphous atactic/syndiotactic dl-lactide-based polymers and copolymers as well as the isotactic d-PLA and l-PLA moieties, each of which carries potential for crystallization. Therefore, specific reference to dl-PLA is essential to appropriately differentiate the amorphous atactic/syndiotactic dl-lactide-based polymers and copolymers covered by this specification. Thus, inclusion of stereoisomeric specificity within the lactic acid-based acronyms results in the following: poly(l-lactide) as PlLA for poly(l-lactic acid), poly(d-lactide) as PdLA for poly(d-lactic acid), and poly(dl-lactide) as PdlLA for poly(dl-lactic acid).
1.3 This specification covers virgin amorphous poly(lactide)-based resins able to be fully solvated at 30°C by either methylene chloride (dichloromethane) or chloroform (trichloromethane). This specification is not applicable to lactide-based polymers or copolymers that possess isotactic polymeric segments sufficient in size to carry potential for lactide-based crystallization, which are covered by Specification F1925 and typically possess nominal mole fractions that equal or exceed 50 % l-lactide. This specification is not applicable to lactide-co-glycolide copolymers that possess glycolide segments sufficient in size to deliver potential for glycolide-based crystallization, thereby requiring fluorinated solvents for complete dissolution under room temperature conditions. This specification is specifically not applicable to lactide-co-glycolide copolymers with glycolide mole fractions greater than or equal to 70 % (65.3 % in mass fraction), which are covere...
General Information
- Status
- Published
- Publication Date
- 14-Dec-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.11 - Polymeric Materials
Relations
- Effective Date
- 15-Dec-2018
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-May-2018
- Effective Date
- 01-Dec-2016
- Effective Date
- 01-Dec-2016
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Aug-2014
- Refers
ASTM E1994-09(2013) - Standard Practice for Use of Process Oriented AOQL and LTPD Sampling Plans - Effective Date
- 01-Apr-2013
- Effective Date
- 01-Dec-2012
- Effective Date
- 15-Nov-2011
- Effective Date
- 01-Dec-2010
- Refers
ASTM D1505-10 - Standard Test Method for Density of Plastics by the Density-Gradient Technique - Effective Date
- 01-Jul-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Jun-2009
Overview
ASTM F2579-18 specifies the requirements for virgin amorphous poly(lactide) (PDLLA) and poly(lactide-co-glycolide) (PLGA) resins used in the manufacture of surgical implants. These resins-commonly referenced in the medical device industry as PDLLA, PLA, PLGA, and PGA:PLA-are key materials in the production of absorbable surgical implants due to their controlled degradation and biocompatibility. The standard ensures that these raw polymers are clearly identified, chemically and physically characterized, and purity-controlled before use in implantable devices. This specification is intended for resin suppliers and manufacturers seeking compliance with recognized international standards for medical-grade raw materials.
Key Topics
Material Scope
- Covers only amorphous forms of poly(lactide) and poly(lactide-co-glycolide) resins.
- Excludes semi-crystalline and block copolymers, and copolymers with high glycolide fractions.
Resin Forms and Quality
- Resins may be supplied as pellets, granules, powders, flakes, or similar forms.
- All monomer components and contact materials must be suitable for medical use.
Chemical and Physical Property Requirements
- Chemical Identification: Polymer identity verified by infrared (IR) spectroscopy, proton nuclear magnetic resonance (1H-NMR), or carbon-13 nuclear magnetic resonance (13C-NMR).
- Specific Rotation: Must fall within –2.5° to +2.5°, confirming stereochemistry.
- Molar Mass: Established via inherent viscosity (IV) measurements; additional methods like gel permeation chromatography (GPC) may be used as agreed upon by supplier and purchaser.
- Residuals Control: Limits on residual monomer (<2.0% by mass), residual solvents (overall ≤1000 ppm), and elemental impurities (compliance assessed per USP and ICH guidelines).
- Optional Tests: Residual catalyst and moisture content may be assessed if requested.
Quality Assurance and Certification
- Each batch requires certification detailing composition, lot number, inherent viscosity, and residual content.
- Adheres to recognized quality management systems (e.g., ISO 9001, ISO 13485, FDA 21 CFR 820).
Applications
The key application area for amorphous PDLLA and PLGA resins is in absorbable surgical implants. These include:
- Orthopedic fixation devices
- Sutures
- Barriers and membranes for soft tissue regeneration
- Drug delivery devices
Benefits of Compliance:
- Patient Safety: Strict limits on impurities minimize risk of adverse tissue reactions and contamination.
- Predictable Degradation: Amorphous polymers provide controlled degradation rates, crucial for temporary implants.
- Regulatory Acceptance: Meeting ASTM F2579-18 facilitates market access and regulatory review by conforming to an internationally recognized standard for polymeric implant materials.
- Consistent Quality: Requirement for material certification and traceability ensures reliability in end-use applications.
Related Standards
ASTM F2579-18 interfaces with several other key standards and guidance documents, including:
- ASTM F1925: Specification for semi-crystalline poly(lactide) resins for surgical implants.
- ASTM F2313: Specification for polyglycolide and related copolymers with high glycolide content.
- ASTM F748: Selecting generic biological test methods for materials and devices.
- ISO 10993: Biological evaluation of medical devices.
- 21 CFR 820: FDA’s Quality System Regulation for medical device manufacturing.
- USP <232> and <233>: Elemental impurities limits and procedures.
- ICH Q3C and Q3D: Guidelines for residual solvents and elemental impurities in pharmaceutical and medical products.
Conclusion
Compliance with ASTM F2579-18 assures manufacturers of surgical implants that their starting polymeric materials meet the highest standards for purity, identity, and quality. By following this specification, both resin suppliers and medical device manufacturers can ensure the safety and effectiveness of absorbable implants, streamline regulatory submissions, and facilitate high-quality patient outcomes.
Keywords: ASTM F2579-18, PDLLA, PLGA, poly(lactide), poly(lactide-co-glycolide), medical implants, absorbable implants, surgical resin, biomaterials, quality assurance, biocompatibility.
Buy Documents
ASTM F2579-18 - Standard Specification for Amorphous Poly(lactide) and Poly(lactide-co-glycolide) Resins for Surgical Implants
REDLINE ASTM F2579-18 - Standard Specification for Amorphous Poly(lactide) and Poly(lactide-co-glycolide) Resins for Surgical Implants
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2579-18 is a technical specification published by ASTM International. Its full title is "Standard Specification for Amorphous Poly(lactide) and Poly(lactide-co-glycolide) Resins for Surgical Implants". This standard covers: ABSTRACT This specification covers amorphous poly(lactide) and poly(lactide-co-glycolide) resins used in the manufacture of surgical implants. Materials covered by this specification are virgin poly(lactide) and poly(lactide-co-glycolide) resins that can be fully solvated at room temperature by methylene chloride (dichloromethane) or chloroform (trichloromethane). The poly(d,l-lactide) homopolymers are amorphous and shall be composed of meso-lactide or equimolar (racemic) combinations of d-lactide and l-lactide. The poly(d,l-lactide-co-glycolide) copolymers are amorphous and shall be composed of a combination of glycolide and either meso-lactide or a racemic combination of d-lactide and l-lactide. The resins shall be manufactured in pellet, granular, powder, flake, or other form and shall conform to the chemical and physical property requirements specified. Tests for chemical identification (by infrared, proton nuclear magnetic resonance, and carbon-13 nuclear magnetic resonance spectroscopy), specific rotation, molar mass, and residual monomer, residual solvent, and heavy metal content shall be performed and shall conform to the requirements specified. Additional tests for residual catalyst and residual water content may be performed as well. SCOPE 1.1 This specification covers virgin amorphous poly(lactide) homopolymer and poly(lactide-co-glycolide) copolymer resins intended for use in surgical implants. The poly(dl-lactide) homopolymers covered by this specification are considered to be amorphous (that is, void of crystallinity) and are polymerized either from meso-lactide or from equimolar (racemic) combinations of d-lactide and l-lactide. The poly(dl-lactide-co-glycolide) copolymers covered by this specification are also considered to be amorphous and are co-polymerized from a combination of glycolide and either meso-lactide or racemic quantities of d-lactide and l-lactide, and typically possess nominal mole fractions that equal or exceed 50 % lactide. 1.2 Since poly(glycolide) is commonly abbreviated as PGA for poly(glycolic acid) and poly(lactide) is commonly abbreviated as PLA for poly(lactic acid), these polymers are commonly referred to as PGA, PLA, and PLA:PGA resins for the hydrolytic byproducts to which they respectively degrade. PLA is a term that carries no stereoisomeric specificity and therefore encompasses both the amorphous atactic/syndiotactic dl-lactide-based polymers and copolymers as well as the isotactic d-PLA and l-PLA moieties, each of which carries potential for crystallization. Therefore, specific reference to dl-PLA is essential to appropriately differentiate the amorphous atactic/syndiotactic dl-lactide-based polymers and copolymers covered by this specification. Thus, inclusion of stereoisomeric specificity within the lactic acid-based acronyms results in the following: poly(l-lactide) as PlLA for poly(l-lactic acid), poly(d-lactide) as PdLA for poly(d-lactic acid), and poly(dl-lactide) as PdlLA for poly(dl-lactic acid). 1.3 This specification covers virgin amorphous poly(lactide)-based resins able to be fully solvated at 30°C by either methylene chloride (dichloromethane) or chloroform (trichloromethane). This specification is not applicable to lactide-based polymers or copolymers that possess isotactic polymeric segments sufficient in size to carry potential for lactide-based crystallization, which are covered by Specification F1925 and typically possess nominal mole fractions that equal or exceed 50 % l-lactide. This specification is not applicable to lactide-co-glycolide copolymers that possess glycolide segments sufficient in size to deliver potential for glycolide-based crystallization, thereby requiring fluorinated solvents for complete dissolution under room temperature conditions. This specification is specifically not applicable to lactide-co-glycolide copolymers with glycolide mole fractions greater than or equal to 70 % (65.3 % in mass fraction), which are covere...
ABSTRACT This specification covers amorphous poly(lactide) and poly(lactide-co-glycolide) resins used in the manufacture of surgical implants. Materials covered by this specification are virgin poly(lactide) and poly(lactide-co-glycolide) resins that can be fully solvated at room temperature by methylene chloride (dichloromethane) or chloroform (trichloromethane). The poly(d,l-lactide) homopolymers are amorphous and shall be composed of meso-lactide or equimolar (racemic) combinations of d-lactide and l-lactide. The poly(d,l-lactide-co-glycolide) copolymers are amorphous and shall be composed of a combination of glycolide and either meso-lactide or a racemic combination of d-lactide and l-lactide. The resins shall be manufactured in pellet, granular, powder, flake, or other form and shall conform to the chemical and physical property requirements specified. Tests for chemical identification (by infrared, proton nuclear magnetic resonance, and carbon-13 nuclear magnetic resonance spectroscopy), specific rotation, molar mass, and residual monomer, residual solvent, and heavy metal content shall be performed and shall conform to the requirements specified. Additional tests for residual catalyst and residual water content may be performed as well. SCOPE 1.1 This specification covers virgin amorphous poly(lactide) homopolymer and poly(lactide-co-glycolide) copolymer resins intended for use in surgical implants. The poly(dl-lactide) homopolymers covered by this specification are considered to be amorphous (that is, void of crystallinity) and are polymerized either from meso-lactide or from equimolar (racemic) combinations of d-lactide and l-lactide. The poly(dl-lactide-co-glycolide) copolymers covered by this specification are also considered to be amorphous and are co-polymerized from a combination of glycolide and either meso-lactide or racemic quantities of d-lactide and l-lactide, and typically possess nominal mole fractions that equal or exceed 50 % lactide. 1.2 Since poly(glycolide) is commonly abbreviated as PGA for poly(glycolic acid) and poly(lactide) is commonly abbreviated as PLA for poly(lactic acid), these polymers are commonly referred to as PGA, PLA, and PLA:PGA resins for the hydrolytic byproducts to which they respectively degrade. PLA is a term that carries no stereoisomeric specificity and therefore encompasses both the amorphous atactic/syndiotactic dl-lactide-based polymers and copolymers as well as the isotactic d-PLA and l-PLA moieties, each of which carries potential for crystallization. Therefore, specific reference to dl-PLA is essential to appropriately differentiate the amorphous atactic/syndiotactic dl-lactide-based polymers and copolymers covered by this specification. Thus, inclusion of stereoisomeric specificity within the lactic acid-based acronyms results in the following: poly(l-lactide) as PlLA for poly(l-lactic acid), poly(d-lactide) as PdLA for poly(d-lactic acid), and poly(dl-lactide) as PdlLA for poly(dl-lactic acid). 1.3 This specification covers virgin amorphous poly(lactide)-based resins able to be fully solvated at 30°C by either methylene chloride (dichloromethane) or chloroform (trichloromethane). This specification is not applicable to lactide-based polymers or copolymers that possess isotactic polymeric segments sufficient in size to carry potential for lactide-based crystallization, which are covered by Specification F1925 and typically possess nominal mole fractions that equal or exceed 50 % l-lactide. This specification is not applicable to lactide-co-glycolide copolymers that possess glycolide segments sufficient in size to deliver potential for glycolide-based crystallization, thereby requiring fluorinated solvents for complete dissolution under room temperature conditions. This specification is specifically not applicable to lactide-co-glycolide copolymers with glycolide mole fractions greater than or equal to 70 % (65.3 % in mass fraction), which are covere...
ASTM F2579-18 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2579-18 has the following relationships with other standards: It is inter standard links to ASTM F2579-10, ASTM D5296-19, ASTM D4603-18, ASTM F2902-16, ASTM F2902-16e1, ASTM F748-16, ASTM E2977-14, ASTM E1994-09(2013), ASTM F2902-12, ASTM D4603-03(2011)e1, ASTM F2313-10, ASTM D1505-10, ASTM F748-06(2010), ASTM E1994-09, ASTM F1925-09. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2579-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2579 −18
Standard Specification for
Amorphous Poly(lactide) and Poly(lactide-co-glycolide)
Resins for Surgical Implants
This standard is issued under the fixed designation F2579; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope polymeric segments sufficient in size to carry potential for
lactide-based crystallization, which are covered by Specifica-
1.1 Thisspecificationcoversvirginamorphouspoly(lactide)
tion F1925 and typically possess nominal mole fractions that
homopolymer and poly(lactide-co-glycolide) copolymer resins
equal or exceed 50 % L-lactide. This specification is not
intended for use in surgical implants. The poly(DL-lactide)
applicable to lactide-co-glycolide copolymers that possess
homopolymers covered by this specification are considered to
glycolide segments sufficient in size to deliver potential for
be amorphous (that is, void of crystallinity) and are polymer-
glycolide-based crystallization, thereby requiring fluorinated
ized either from meso-lactide or from equimolar (racemic)
combinations of D-lactide and L-lactide. The poly(DL-lactide-
solvents for complete dissolution under room temperature
co-glycolide) copolymers covered by this specification are also
conditions. This specification is specifically not applicable to
considered to be amorphous and are co-polymerized from a
lactide-co-glycolide copolymers with glycolide mole fractions
combination of glycolide and either meso-lactide or racemic
greater than or equal to 70 % (65.3 % in mass fraction), which
quantities of D-lactide and L-lactide, and typically possess
are covered by Specification F2313. This specification is not
nominal mole fractions that equal or exceed 50 % lactide.
applicable to block copolymers or to polymers or copolymers
1.2 Since poly(glycolide) is commonly abbreviated as PGA synthesized from combinations of D-lactide and L-lactide that
for poly(glycolic acid) and poly(lactide) is commonly abbre- differ by more than 1.5 total mole percent (1.5 % of total
viated as PLA for poly(lactic acid), these polymers are com-
moles).
monly referred to as PGA, PLA, and PLA:PGA resins for the
1.4 This specification addresses material characteristics of
hydrolyticbyproductstowhichtheyrespectivelydegrade.PLA
both poly(DL-lactide) and poly(DL-lactide-co-glycolide) resins
isatermthatcarriesnostereoisomericspecificityandtherefore
intended for use in surgical implants and does not apply to
encompasses both the amorphous atactic/syndiotactic DL-
packaged and sterilized finished implants fabricated from these
lactide-based polymers and copolymers as well as the isotactic
materials.
D-PLAand L-PLAmoieties, each of which carries potential for
crystallization. Therefore, specific reference to DL-PLA is
1.5 As with any material, some characteristics may be
essential to appropriately differentiate the amorphous atactic/
altered by processing techniques (such as molding, extrusion,
syndiotactic DL-lactide-based polymers and copolymers cov-
machining, assembly, sterilization, and so forth) required for
ered by this specification. Thus, inclusion of stereoisomeric
the production of a specific part or device. Therefore, proper-
specificity within the lactic acid-based acronyms results in the
ties of fabricated forms of this resin should be evaluated
following: poly(L-lactide) as PLLA for poly(L-lactic acid),
independently using appropriate test methods to assure safety
poly(D-lactide) as PDLA for poly(D-lactic acid), and poly(DL-
and efficacy.
lactide) as PDLLA for poly(DL-lactic acid).
1.6 The values stated in SI units are to be regarded as
1.3 This specification covers virgin amorphous
standard. No other units of measurement are included in this
poly(lactide)-based resins able to be fully solvated at 30°C by
standard.
either methylene chloride (dichloromethane) or chloroform
(trichloromethane). This specification is not applicable to
1.7 This standard does not purport to address all of the
lactide-based polymers or copolymers that possess isotactic
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
This specification is under the jurisdiction of ASTM Committee F04 on
mine the applicability of regulatory limitations prior to use.
Medical and Surgical Materials and Devices and is the direct responsibility of
Subcommittee F04.11 on Polymeric Materials.
1.8 This international standard was developed in accor-
Current edition approved Dec. 15, 2018. Published February 2019. Originally
dance with internationally recognized principles on standard-
approved in 2006. Last previous edition approved in 2010 as F2579 – 10. DOI:
10.1520/F2579-18. ization established in the Decision on Principles for the
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2579 − 18
Development of International Standards, Guides and Recom- USP 781 United States Pharmacopeia: Physical Tests –
mendations issued by the World Trade Organization Technical Optical Rotation
Barriers to Trade (TBT) Committee. USP 788 United States Pharmacopeia: Particulate Matter in
Injections
2. Referenced Documents
2.5 Other Documents/Websites:
2.1 ASTM Standards: ICH Q3C International Conference on Harmonisation of
Technical Requirements for Registration of Pharmaceuti-
D1505 Test Method for Density of Plastics by the Density-
Gradient Technique cals for Human Use, Quality Guideline: Impurities: Re-
sidual Solvents
D2857 Practice for Dilute Solution Viscosity of Polymers
ICH Q3D International Conference on Harmonisation of
D4603 Test Method for Determining Inherent Viscosity of
Technical Requirements for Registration of Pharmaceuti-
Poly(Ethylene Terephthalate) (PET) by Glass Capillary
cals for Human Use: Guideline for Elemental Impurities
Viscometer
21 CFR 820 Code of Federal Regulations, Title 21, Part
D5296 Test Method for Molecular Weight Averages and
820, Quality System Regulation
Molecular Weight Distribution of Polystyrene by High
NIST Special Publication SP811 Guide for the Use of the
Performance Size-Exclusion Chromatography
International System of Units (SI)
E1252 Practice for General Techniques for Obtaining Infra-
FDAGuidance “Use of International Standard ISO 10993-1,
red Spectra for Qualitative Analysis
‘Biological evaluation of medical devices – Part 1: Evalu-
E1994 Practice for Use of Process Oriented AOQL and
ation and testing within a risk management process’ –
LTPD Sampling Plans
Guidance for Industry and Food and DrugAdministration
E2977 PracticeforMeasuringandReportingPerformanceof
Staff.
Fourier-Transform Nuclear Magnetic Resonance (FT-
NMR) Spectrometers for Liquid Samples
3. Terminology
F748 PracticeforSelectingGenericBiologicalTestMethods
for Materials and Devices
3.1 Definitions:
F1925 SpecificationforSemi-CrystallinePoly(lactide)Poly-
3.1.1 virgin polymer—the initially delivered form of a
mer and Copolymer Resins for Surgical Implants
polymer as synthesized from its monomers and prior to any
F2313 Specification for Poly(glycolide) and Poly(glycolide-
processing or fabrication into a medical device.
co-lactide) Resins for Surgical Implants with Mole Frac-
tions Greater Than or Equal to 70 % Glycolide
4. Materials and Manufacture
F2902 Guide for Assessment of Absorbable Polymeric Im-
4.1 All raw monomer components and other materials
plants
contacting either the raw monomer(s) or resin product shall be
2.2 ANSI Standards:
of a quality suitable to allow for use of such resin in the
ANSI/ISO/ASQ 13485 Medical devices -- Quality manage-
manufacture of an implantable medical product. Such quality
ment systems -- Requirements for regulatory purposes
includes adequate control of particles and other potential
ANSI/ISO/ASQ Q9000 Quality Management Systems, Fun-
contaminants that may affect either the toxicity of or the cell
damentals and Vocabulary
response to the as-implanted or degrading final product.
ANSI/ISO/ASQ Q9001 Quality Management Systems, Re-
4.2 All polymer manufacturing (including monomer
quirements
handling, synthesis, pelletization/grinding and all subsequent
2.3 ISO Standards:
steps) shall be undertaken under conditions suitable to allow
ISO 10993 Biological Evaluation of Medical Devices
for use of such resin in the manufacture of an implantable
ISO 80000-9 Quantities and units -- Part 9: Physical chem-
medical product.
istry and molecular physics
5. Chemical Composition
2.4 U. S. Pharmacopeia (USP) Standards:
USP 231 United States Pharmacopeia: Chemical Analysis –
5.1 The amorphous poly(DL-lactide) polymers covered by
Heavy Metals
this specification shall be composed either of meso -lactide or
USP 232 United States Pharmacopeia: Elemental Impurities
a racemic combination of D-lactide and L-lactide. The amor-
– Limits
phous poly(DL-lactide-co-glycolide) copolymers covered by
USP 233 United States Pharmacopeia: Elemental Impurities
this specification can be of variable copolymer ratios and shall
– Procedures
Available from ICH Secretariat, c/o IFPMA, 30 rue de St-Jean, P.O. Box 758,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or 1211 Geneva 13, Switzerland. Available online at http://www.ich.org/products/
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM guidelines/quality/article/quality-guidelines.html.
Standards volume information, refer to the standard’s Document Summary page on AvailablefromU.S.GovernmentPrintingOfficeSuperintendentofDocuments,
theASTM website. 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St., www.access.gpo.gov.
4th Floor, New York, NY 10036, http://www.ansi.org. Available from National Institute of Standards and Technology (NIST), 100
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, Bureau Dr., Stop 1070, Gaithersburg, MD 20899-1070, at http://physics.nist.gov/
MD 20852-1790, http://www.usp.org. cuu/Units/bibliography.html.
F2579 − 18
be composed of a combination of glycolide and either meso- 5.2.4.2 Additional spectral bands may be indicative of
lactide or a racemic combination of D-lactide and L-lactide known or unknown impurities, including residual solvents and
catalysts. Refer to the limits specified in Table 1.
where the glycolide mole fraction is less than 70 % (65.3 % in
mass fraction). To assure such composition and the attainment
5.3 Specific Rotation:
of the desired properties, the following tests are to be con-
5.3.1 The virgin homopolymer or copolymer shall have a
ducted.
specific rotation of –2.5 to +2.5 degrees when measured in
5.2 Chemical Identification:
either chloroform, methylene chloride, or tetrahydrofuran at
20°C using a polarimetry method equal to or equivalent to the
5.2.1 The identity of the virgin polymer shall be confirmed
1 13
Optical Rotation procedure described in USP <781>.
either by infrared, H-NMR, or C-NMR spectroscopy.
5.2.2 Infrared Identification:
5.4 Molar Mass:
5.2.2.1 Identity of either poly(lactide) homopolymer or
NOTE 1—The term molecular weight (abbreviated MW) is obsolete and
poly(lactide-co-glycolide) copolymer may be confirmed
should be replaced by the SI (Système Internationale) equivalent of either
through an infrared spectrum exhibiting major absorption
relative molecular mass (M ), which reflects the dimensionless ratio of the
r
bands only at the wavelengths that appear in a suitable
mass of a single molecule to an atomic mass unit [see ISO 80000-9], or
molar mass (M), which refers to the mass of a mole of a substance and is
reference spectrum. Analysis shall be conducted using infra-
typically expressed as grams/mole. For polymers and other
redspectroscopymethodssimilartothosedescribedinPractice
macromolecules,useofthesymbols M , M ,and M continue,referringto
w n z
E1252.Atypical infrared transmission reference spectrum and
mass-average molar mass, number-average molar mass, and z-average
a typical infrared absorption reference spectrum for DL-PLA
molar mass, respectively. For more information regarding proper utiliza-
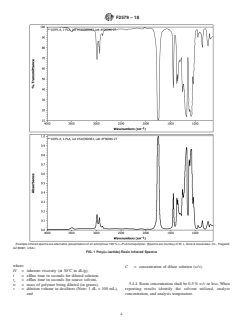
homopolymer are shown in Fig. 1, with example spectra for tion of SI units, see NIST Special Publication SP811.
copolymers presented in Fig. 2. While poly(lactide-co-
5.4.1 The molar mass of the virgin polymer shall be
glycolide) copolymers will each have their own respective
indicated by inherent viscosity (IV) in dilute solution. In
spectrum that will vary in response to copolymer ratio, this
addition to inherent viscosity (but not in place of), mass
analytic method typically lacks sensitivity sufficient for quan-
average molar mass and molar mass distributions maybe
tification of copolymer ratio as specified in 7.1.2.
determined by gel permeation chromatography (GPC) accord-
5.2.2.2 Additional or variable spectral bands may be indica-
ing to the general procedure described in Test Method D5296,
tive of sample crystallinity or either known or unknown
but using either chloroform or dichloromethane and appropri-
impurities, including residual monomer, solvents, and catalysts
ate calibration standards.
(refer to limits specified in Table 1).
NOTE2—Molarmasscalibrationstandards(forexample,polystyreneor
5.2.2.3 Since an infrared spectrum cannot distinguish be-
polymethylmethacrylate) provide relative values only, and are not to be
tweenthedifferentlactidestereoisomers,itisutilizedhereonly
confused with an absolute determination of a lactide-based polymer’s
as a means of identifying the non-stereospecific poly(lactide) molar mass.
component of a poly(lactide)-based polymer or copolymer.
5.4.2 Determine the inherent viscosity of the polymer pref-
5.2.3 Proton Nuclear Magnetic Resonance ( H-NMR) Iden-
erentially in chloroform at 30°C using procedures similar to
tification:
those described in Practice D2857 and Test Method D4603.
5.2.3.1 Identity of either poly(lactide) homopolymer or
Determination at a lower temperature of 25°C is allowable,
poly(lactide-co-glycolide) copolymer may be confirmed
provided the utilized equipment delivers the required thermal
through sample dissolution, H-NMR spectroscopy, and the
control and, if requested by the purchaser, an experimentally
use of a suitable reference spectrum. Sample dissolution is in
supported 30°C equivalent concentration-appropriate extrapo-
either deuterated chloroform, deuterated dichloromethane
lated result is also reported within the supplied certification. If
(methylene chloride) or other substantially proton-free solvent
the required sample of the subject copolymer ratio does not
able to fully solvate the specimen without inducing competing fully dissolve in chloroform, alternatively utilize dichloro-
spectral bands. Analysis shall be conducted using methods methane (methylene chloride) as the dissolution solvent. Note
similar to those described in Practice E2977. Typical proton that any incomplete sample dissolution, precipitation from
NMR reference spectra for 100 % DL-PLA homopolymer and solution, or the formation of gels will produce inconsistency
85 % DL -PLA:15 % PGA copolymer are shown in Fig. 3 and and variation in observed drop times.
Fig. 4, respectively.
NOTE 3—The IV test duration for each sample should be minimized to
5.2.3.2 Additional spectral bands may be indicative of
reduce risk of resin concentration changes due to evaporative loss of
known or unknown impurities, including residual monomer, solvent.
solvents, and catalysts (refer to limits specified in Table 1).
5.4.3 Inherent viscosity is determined utilizing the follow-
5.2.4 Carbon-13 Nuclear Magnetic Resonance ( C-NMR)
ing:
Identification:
ln t/t v
~ !
o
5.2.4.1 Identity of either poly(lactide) homopolymer or IV 5 (1)
w
poly(lactide-co-glycolide) copolymer may be confirmed in a
or
solid state through C-NMR spectroscopy and the use of a
suitable reference spectrum.Analysis shall be conducted using IV 5 ln~t/t !
o
(2)
methods similar to those described in Practice E2977. C
F2579 − 18
Example infrared spectra are alternative presentations of an amorphous 100 % DL-PLAhomopolymer. (Spectra are courtesy of W. L. Gore &Associates, Inc., Flagstaff,
AZ 86001, USA.)
FIG. 1 Poly(DL-lactide) Resin Infrared Spectra
where:
C = concentration of dilute solution (w/v).
IV = inherent viscosity (at 30°C in dL/g),
t = efflux time in seconds for diluted solution,
t = efflux time in seconds for source solvent,
o
5.4.4 Resin concentration shall be 0.5 % w/v or less. When
w = mass of polymer being diluted (in grams),
v = dilution volume in deciliters (Note: 1 dL = 100 mL), reporting results identify the solvent utilized, analyte
and concentration, and analysis temperature.
F2579 − 18
Exampleinfraredspectraarealternativepresentationsofanamorphous85% DL-PLA:15%PGA(moleratio)copolymer.(SpectraarecourtesyofW.L.Gore&Associates,
Inc., Flagstaff, AZ 86001, USA.)
FIG. 2 Poly(lactide-co-glycolide) Resin Infrared Spectra
5.5 Residual Monomer: content significantly less than 2 % to address processing or
5.5.1 The virgin polymer shall have a combined total intended end-use requirements, or both (see Section S1—
residual monomer content less than or equal to 2.0 % in mass Biocompatibility).
fraction. Residual monomer levels up to 3 % are acceptable if 5.5.2 Determine the mass fraction of residual monomer by
deemedbythepurchasertobesuitablefortheintendedend-use gas chromatography, HPLC, H-NMR spectroscopy (using
application. Alternatively, a purchaser may require monomer deuterated chloroform, deuterated dichoromethane or other
F2579 − 18
Supplied example infrared spectrum of an amorphous 50 % DL-PLA:50 % PGA (mole ratio) copolymer is courtesy of Lakeshore Biomaterials, 756 Tom Martin Dr.,
Birmingham, AL 35211, USA.
FIG. 2 Poly(lactide-co-glycolide) Resin Infrared Spectra (continued)
substantially proton-free solvent able to fully solvate the than 1000 ppm when combinations of more than one solvent
specimen), or other suitably sensitive analytic method as
are utilized (regardless of individual solvent toxicity).
agreed upon by the supplier and purchaser.
5.7 Elemental Impurities:
5.6 Residual Solvents:
5.7.1 The significance of Elemental Impurities within an
5.6.1 If any solvent is utilized in any resin manufacturing or
absorbablepolymerisultimatelydependentonthedimensional
purification step, determine the residual levels of any utilized
characteristics of the final product and the rate of release of
solvent(s) by gas chromatography or other suitable method as
those initially interstitial elements into the surrounding tissue
agreed upon by the supplier and purchaser.Acceptable residual
and extracelluar fluid. Thus, any risk assessment of such
levels of a particular solvent shall be reflective of toxicity, with
impurities will be dependent on the final product design and
a maximum acceptable limit consistent with ICH Q3C. The
intended application. Consequently, this raw material (not final
detection limit for the chosen analytic method shall be ad-
device) standard provides for appropriate reporting of Elemen-
equate to assure compliance with the applicable ICH guideline
tal Impurities values, but does not mandate any specific
and the determined residual(s) and applied concentration
performance requirements. More detailed and pharmaceutical
limit(s) shall be reported. If no ICH concentration guideline
oriented guidance regarding the appropriate means for both
has been established for a utilized solvent, an entry of “no ICH
monitoring and assessing relevant Elemental Impurities within
guidance available” shall be reported in instead of a limit.
5.6.2 To minimize potential for toxic interaction of solvent afinalproductcanbefoundinUSPChapters<232>and<233>
combinations, cumulative Total Solvent Combination Residu- andintheICHHARMONISEDGUIDELINEFORELEMEN-
als shall be limited to 1000 ppm (refer to the limit specified in
TAL IMPURITIES - Q3D.
Table1).ThislimithastheeffectofallowingICHQ3CQuality
Guidelines when a single solvent system is utilized and less
F2579 − 18
Supplied example NMR spectrum of an amorphous 100 % DL-PLA homopolymer is courtesy of W. L. Gore & Associates, Inc., Flagstaff, AZ 86001, USA.
FIG. 3 Poly(DL-lactide) Nuclear Magnetic Resonance Spectrum
5.7.2 DeterminetheconcentrationoftherespectiveElemen- implant product as well as its degradation and elemental
tal Impurities within the absorbable polymer by utilizing a elution rate into the surrounding tissue.
method as described in Chapter <233> of the U.S. Pharmaco-
5.7.4 For each element intentionally added as catalyst, the
peia. The specific 24 different Elemental Impurities of interest
concentration (in ppm, in accordance with USP <233> or
are outlined in both USP <232> and in TableA.2.2 of the ICH
equivalent) shall be both monitored and reported.
HARMONISED GUIDELINE FOR ELEMENTAL IMPURI-
5.8 Residual Catalyst:
TIES - Q3D (Dec 2014). Both of these documents include
risk-based approaches toward the assessment and control of
5.8.1 Determine the elemental concentration of residual
elemental impurities.
catalyst as described in USP <233>. If the utilized catalyst is
5.7.3 Except for elements intentionally added as catalysts,
not measurable via USP <233>, suitable methods to both
assess the obtained results for compliance with the Parenteral
determine and report
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2579 − 10 F2579 − 18
Standard Specification for
Amorphous Poly(lactide) and Poly(lactide-co-glycolide)
Resins for Surgical Implants
This standard is issued under the fixed designation F2579; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This specification covers virgin amorphous poly(lactide) homopolymer and poly(lactide-co-glycolide) copolymer resins
intended for use in surgical implants. The poly(DL-lactide) homopolymers covered by this specification are considered to be
amorphous (that is, void of crystallinity) and are polymerized either from meso-lactide or from equimolar (racemic) combinations
of D-lactide and L-lactide. The poly(DL-lactide-co-glycolide) copolymers covered by this specification are also considered to be
amorphous and are co-polymerized from a combination of glycolide and either meso-lactide or racemic quantities of D-lactide and
L-lactide, and typically possess nominal mole fractions that equal or exceed 50 % lactide.
1.2 Since poly(glycolide) is commonly abbreviated as PGA for poly(glycolic acid) and poly(lactide) is commonly abbreviated
as PLA for poly(lactic acid), these polymers are commonly referred to as PGA, PLA, and PLA:PGA resins for the hydrolytic
byproducts to which they respectively degrade. PLA is a term that carries no stereoisomeric specificity and therefore encompasses
both the amorphous atactic/syndiotactic DL-lactide-based polymers and copolymers as well as the isotactic D-PLA and L-PLA
moieties, each of which carries potential for crystallization. Therefore, specific reference to DL-PLA is essential to appropriately
differentiate the amorphous atactic/syndiotactic DL-lactide-based polymers and copolymers covered by this specification. Thus,
inclusion of stereoisomeric specificity within the lactic acid-based acronyms results in the following: poly(L-lactide) as PLLA for
poly(L-lactic acid), poly(D-lactide) as PDLA for poly(D-lactic acid), and poly(DL-lactide) as PDLLA for poly(DL-lactic acid).
1.3 This specification covers virgin amorphous poly(lactide)-based resins able to be fully solvated at 30°C by either methylene
chloride (dichloromethane) or chloroform (trichloromethane). This specification is not applicable to lactide-based polymers or
copolymers that possess isotactic polymeric segments sufficient in size to carry potential for lactide-based crystallization, which
are covered by Specification F1925 and typically possess nominal mole fractions that equal or exceed 50 % L-lactide. This
specification is not applicable to lactide-co-glycolide copolymers that possess glycolide segments sufficient in size to deliver
potential for glycolide-based crystallization, thereby requiring fluorinated solvents for complete dissolution under room
temperature conditions. This specification is specifically not applicable to lactide-co-glycolide copolymers with glycolide mole
fractions greater than or equal to 70 % (65.3 % in mass fraction), which are covered by Specification F2313. This specification is
not applicable to block copolymers or to polymers or copolymers synthesized from combinations of D-lactide and L-lactide that
differ by more than 1.5 total mole percent (1.5 % of total moles).
1.4 This specification addresses material characteristics of both poly(DL-lactide) and poly(DL-lactide-co-glycolide) resins
intended for use in surgical implants and does not apply to packaged and sterilized finished implants fabricated from these
materials.
1.5 As with any material, some characteristics may be altered by processing techniques (such as molding, extrusion, machining,
assembly, sterilization, and so forth) required for the production of a specific part or device. Therefore, properties of fabricated
forms of this resin should be evaluated independently using appropriate test methods to assure safety and efficacy.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
This specification is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.11 on Polymeric Materials.
Current edition approved Dec. 1, 2010Dec. 15, 2018. Published January 2011February 2019. Originally approved in 2006. Last previous edition approved in 20082010
as F2579 – 08.F2579 – 10. DOI: 10.1520/F2579-10.10.1520/F2579-18.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2579 − 18
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
F2579 − 18
2. Referenced Documents
2.1 ASTM Standards:
D1505 Test Method for Density of Plastics by the Density-Gradient Technique
D2857 Practice for Dilute Solution Viscosity of Polymers
D4603 Test Method for Determining Inherent Viscosity of Poly(Ethylene Terephthalate) (PET) by Glass Capillary Viscometer
D5296 Test Method for Molecular Weight Averages and Molecular Weight Distribution of Polystyrene by High Performance
Size-Exclusion Chromatography
E386 Practice for Data Presentation Relating to High-Resolution Nuclear Magnetic Resonance (NMR) Spectroscopy
(Withdrawn 2015)
E1252 Practice for General Techniques for Obtaining Infrared Spectra for Qualitative Analysis
E1994 Practice for Use of Process Oriented AOQL and LTPD Sampling Plans
E2977 Practice for Measuring and Reporting Performance of Fourier-Transform Nuclear Magnetic Resonance (FT-NMR)
Spectrometers for Liquid Samples
F748 Practice for Selecting Generic Biological Test Methods for Materials and Devices
F1925 Specification for Semi-Crystalline Poly(lactide) Polymer and Copolymer Resins for Surgical Implants
F2313 Specification for Poly(glycolide) and Poly(glycolide-co-lactide) Resins for Surgical Implants with Mole Fractions Greater
Than or Equal to 70 % Glycolide
F2902 Guide for Assessment of Absorbable Polymeric Implants
2.2 ANSI Standards:
ANSI/ISO/ASQ 13485 Medical devices -- Quality management systems -- Requirements for regulatory purposes
ANSI/ISO/ASQ Q9000 Quality Management Systems, Fundamentals and Vocabulary
ANSI/ISO/ASQ Q9001 Requirements Quality Management Systems, Requirements
2.3 ISO Standards:
ISO 31-8 Physical Chemistry and Molecular Physics, Part 8: Quantities and Units
ISO 10993 Biological Evaluation of Medical Devices
ISO 80000-9 Quantities and units -- Part 9: Physical chemistry and molecular physics
2.4 U. S. Pharmacopeia (USP) Standards:
USP30/NF25USP 231 United States Pharmacopeia (USP),Pharmacopeia: Chemical May 2, 2007Analysis – Heavy Metals
USP 232 United States Pharmacopeia: Elemental Impurities – Limits
USP 233 United States Pharmacopeia: Elemental Impurities – Procedures
USP 781 United States Pharmacopeia: Physical Tests – Optical Rotation
USP 788 United States Pharmacopeia: Particulate Matter in Injections
2.5 Other Documents/Websites:
ICH Q3C(R3)Q3C International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals
for Human Use, Quality Guideline: Impurities: Residual Solvents
ICH Q3D International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human
Use: Guideline for Elemental Impurities
21 CFR 820 Code of Federal Regulations, Title 21, Part 820, Quality System Regulation
NIST Special Publication SP811 Guide for the Use of the International System of Units (SI)
FDA Guidance “Use of International Standard ISO 10993-1, ‘Biological evaluation of medical devices – Part 1: Evaluation and
testing within a risk management process’ – Guidance for Industry and Food and Drug Administration Staff.
3. Terminology
3.1 Definitions:
3.1.1 virgin polymer—the initially delivered form of a polymer as synthesized from its monomers and prior to any processing
or fabrication into a medical device.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852-1790, http://www.usp.org.
Available from ICH Secretariat, c/o IFPMA, 30 rue de St-Jean, P.O. Box 758, 1211 Geneva 13, Switzerland. Available online at http://www.ich.org/LOB/media/
MEDIA423.pdfhttp://www.ich.org/products/guidelines/quality/article/quality-guidelines.html.
Available from U.S. Government Printing Office Superintendent of Documents, 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
www.access.gpo.gov.
Available from National Institute of Standards and Technology (NIST), 100 Bureau Dr., Stop 1070, Gaithersburg, MD 20899-1070, at http://physics.nist.gov/cuu/Units/
bibliography.html.
F2579 − 18
4. Materials and Manufacture
4.1 All raw monomer components and other materials contacting either the raw monomer(s) or resin product shall be of a
quality suitable to allow for use of such resin in the manufacture of an implantable medical product. Such quality includes adequate
control of particles and other potential contaminants that may affect either the toxicity of or the cell response to the as-implanted
or degrading final product.
4.2 All polymer manufacturing (including monomer handling, synthesis, pelletization/grinding and all subsequent steps) shall
be undertaken under conditions suitable to allow for use of such resin in the manufacture of an implantable medical product.
5. Chemical Composition
5.1 The amorphous poly(DL-lactide) polymers covered by this specification shall be composed either of meso -lactide or a
racemic combination of D-lactide and L-lactide. The amorphous poly(DL-lactide-co-glycolide) copolymers covered by this
specification can be of variable copolymer ratios and shall be composed of a combination of glycolide and either meso-lactide or
a racemic combination of D-lactide and L-lactide where the glycolide mole fraction is less than 70 % (65.3 % in mass fraction).
To assure such composition and the attainment of the desired properties, the following tests are to be conducted.
5.2 Chemical Identification:
1 13
5.2.1 The identity of the virgin polymer shall be confirmed either by infrared, H-NMR, or C-NMR spectroscopy.
5.2.2 Infrared Identification:
5.2.2.1 Identity of either poly(lactide) homopolymer or poly(lactide-co-glycolide) copolymer may be confirmed through an
infrared spectrum exhibiting major absorption bands only at the wavelengths that appear in a suitable reference spectrum. Analysis
shall be conducted using infra-red spectroscopy practicesmethods similar to those described in Practice E1252. A typical infrared
transmission reference spectrum and a typical infrared absorption reference spectrum for DL-PLA homopolymer are shown in Fig.
1, with example spectra for copolymers presented in Fig. 2. While poly(lactide-co-glycolide) copolymers will each have their own
respective spectrum that will vary in response to copolymer ratio, this analytic method typically lacks sensitivity sufficient for
quantification of copolymer ratio as specified in 7.1.2.
5.2.2.2 Additional or variable spectral bands may be indicative of sample crystallinity or either known or unknown impurities,
including residual monomer, solvents, and catalysts (refer to limits specified in Table 1).
5.2.2.3 Since an infrared spectrum cannot distinguish between the different lactide stereoisomers, it is utilized here only as a
means of identifying the non-stereospecific poly(lactide) component of a poly(lactide)-based polymer or copolymer.
5.2.3 Proton Nuclear Magnetic Resonance ( H-NMR) Identification:
5.2.3.1 Identity of either poly(lactide) homopolymer or poly(lactide-co-glycolide) copolymer may be confirmed through sample
dissolution, H-NMR spectroscopy, and the use of a suitable reference spectrum. Sample dissolution is in either deuterated
chloroform, deuterated dichloromethane (methylene chloride) or other substantially proton-free solvent able to fully solvate the
specimen without inducing competing spectral bands. Analysis shall be conducted using practicesmethods similar to those
described in Practice E386E2977. Typical proton NMR reference spectra for 100 % DL-PLA homopolymer and 85 % DL -PLA:15 %
PGA copolymer are shown in Fig. 3 and Fig. 4, respectively.
5.2.3.2 Additional spectral bands may be indicative of known or unknown impurities, including residual monomer, solvents, and
catalysts (refer to limits specified in Table 1).
5.2.4 Carbon-13 Nuclear Magnetic Resonance ( C-NMR) Identification:
5.2.4.1 Identity of either poly(lactide) homopolymer or poly(lactide-co-glycolide) copolymer may be confirmed in a solid state
through C-NMR spectroscopy and the use of a suitable reference spectrum. Analysis shall be conducted using practicesmethods
similar to those described in Practice E386E2977.
5.2.4.2 Additional spectral bands may be indicative of known or unknown impurities, including residual solvents and catalysts
(refercatalysts. Refer to the limits specified in Table 1).
5.3 Specific Rotation:
5.3.1 The virgin homopolymer or copolymer shall have a specific rotation of –2.5 to +2.5 degrees when measured in either
chloroform, methylene chloride, or tetrahydrofuran at 20°C using a polarimetry method equal to or equivalent to the Optical
Rotation procedure described in USP30/NF25USP <781>.
5.4 Molar Mass:
NOTE 1—The term molecular weight (abbreviated MW) is obsolete and should be replaced by the SI (Système Internationale) equivalent of either
relative molecular mass (M ), which reflects the dimensionless ratio of the mass of a single molecule to an atomic mass unit [see ISO 31-8],80000-9],
r
or molar mass (M), which refers to the mass of a mole of a substance and is typically expressed as grams/mole. For polymers and other macromolecules,
use of the symbols M ,M , and M continue, referring to mass-average molar mass, number-average molar mass, and z-average molar mass, respectively.
w n z
For more information regarding proper utilization of SI units, see NIST Special Publication SP811.
5.4.1 The molar mass of the virgin polymer shall be indicated by inherent viscosity (IV) in dilute solution (IV). solution. In
addition to inherent viscosity (but not in place of), mass average molar mass and molar mass distributions maybe determined by
gel permeation chromatography (GPC) according to the general procedure described in Test Method D5296, but using either
chloroform or dichloromethane and appropriate calibration standards.
F2579 − 18
Example infrared spectra are alternative presentations of an amorphous 100 % DL-PLA homopolymer. (Spectra are courtesy of W. L. Gore & Associates, Inc., Flagstaff,
AZ 86001, USA.)
FIG. 1 Poly(DL-lactide) Resin Infrared Spectra
NOTE 2—Molar mass calibration standards (for example, polystyrene or polymethylmethacrylate) provide relative values only, and are not to be
confused with an absolute determination of a lactide-based polymer’s molar mass.
5.4.2 Determine the inherent viscosity of the polymer preferentially in chloroform at 30°C using procedures similar to those
described in Practice D2857 and Test Method D4603. Determination at a lower temperature of 25°C is allowable, provided the
utilized equipment delivers the required thermal control and, if requested by the purchaser, an experimentally supported 30°C
equivalent concentration-appropriate extrapolated result is also reported within the supplied certification. If the required sample of
the subject copolymer ratio does not fully dissolve in chloroform, alternatively utilize dichloro-
methane (methylene chloride) as the dissolution solvent. Note that any incomplete sample dissolution, precipitation from solution,
or the formation of gels will produce inconsistency and variation in observed drop times.
F2579 − 18
Example infrared spectra are alternative presentations of an amorphous 85% DL-PLA:15% PGA (mole ratio) copolymer. (Spectra are courtesy of W. L. Gore & Associates,
Inc., Flagstaff, AZ 86001, USA.)
FIG. 2 Poly(lactide-co-glycolide) Resin Infrared Spectra
NOTE 3—The IV test duration for each sample should be minimized to reduce risk of resin concentration changes due to evaporative loss of solvent.
5.4.3 Inherent viscosity is determined utilizing the following:
ln~t/t !v
o
IV 5 (1)
w
or
IV 5 ln~t/t !
o
(2)
C
F2579 − 18
Supplied example infrared spectrum of an amorphous 50 % DL-PLA:50 % PGA (mole ratio) copolymer is courtesy of Lakeshore Biomaterials, 756 Tom Martin Dr.,
Birmingham, AL 35211, USA.
FIG. 2 Poly(lactide-co-glycolide) Resin Infrared Spectra (continued)
where:
IV = inherent viscosity (at 30°C in dL/g),
t = efflux time in seconds for diluted solution,
t = efflux time in seconds for source solvent,
o
w = mass of polymer being diluted (in grams),
v = dilution volume in deciliters (Note: 1 dL = 100 mL), and
C = concentration of dilute solution (w/v).
5.4.4 Resin concentration shall be 0.5 % w/v or less. When reporting results identify the solvent utilized, analyte concentration,
and analysis temperature.
5.5 Residual Monomer:
5.5.1 The virgin polymer shall have a combined total residual monomer content less than or equal to 2.0 % in mass fraction.
Residual monomer levels up to 3 % are acceptable if deemed by the purchaser to be suitable for the intended end-use application.
Alternatively, a purchaser may require monomer content significantly less than 2 % to address processing or intended end-use
requirements, or both (see Section S1—Biocompatibility).
5.5.2 Determine the mass fraction of residual monomer by gas chromatography, HPLC, H-NMR spectroscopy (using
deuterated chloroform, deuterated dichoromethane or other substantially proton-free solvent able to fully solvate the specimen),
or other suitably sensitive analytic method as agreed upon by the supplier and purchaser.
5.6 Residual Solvents:
5.6.1 If any solvent is utilized in any resin manufacturing or purification step, determine the residual levels of any utilized
solvent(s) by gas chromatography or other suitable method as agreed upon by the supplier and purchaser. Acceptable residual levels
of a particular solvent shall be reflective of toxicity, with a maximum acceptable limit consistent with ICH Q3C(R3).Q3C. The
F2579 − 18
Supplied example NMR spectrum of an amorphous 100 % DL-PLA homopolymer is courtesy of W. L. Gore & Associates, Inc., Flagstaff, AZ 86001, USA.
FIG. 3 Poly(DL-lactide) Nuclear Magnetic Resonance Spectrum
detection limit for the chosen analytic method mustshall be adequate to assure compliance with the applicable ICH guideline and
the determined residual(s) and applied concentration limit(s) shall be reported. If no ICH concentration guideline has been
established for a utilized solvent, an entry of “no ICH guidance available” shall be reported in lieuinstead of a limit.
5.6.2 To minimize potential for toxic interaction of solvent combinations, cumulative Total Solvent Combination Residuals shall
be limited to 1000 ppm (refer to the limit specified in Table 1). This limit carrieshas the effect of allowing ICH Q3C(R3)Q3C
Quality Guidelines when a single solvent system is utilized and less than 1000 ppm when combinations of more than one solvent
are utilized (regardless of individual solvent toxicity).
5.7 Heavy Metals:Elemental Impurities:
5.7.1 The significance of Elemental Impurities within an absorbable polymer is ultimately dependent on the dimensional
characteristics of the final product and the rate of release of those initially interstitial elements into the surrounding tissue and
extracelluar fluid. Thus, any risk assessment of such impurities will be dependent on the final product design and intended
application. Consequently, this raw material (not final device) standard provides for appropriate reporting of Elemental Impurities
values, but does not mandate any specific performance requirements. More detailed and pharmaceutical oriented guidance
regarding the appropriate means for both monitoring and assessing relevant Elemental Impurities within a final product can be
found in USP Chapters <232> and <233> and in the ICH HARMONISED GUIDELINE FOR ELEMENTAL IMPURITIES - Q3D.
5.7.2 Determine residual Heavy Metals per Method II, Chapter 231 of U.S. Pharmacopeia.the concentration of the respective
Elemental Impurities within the absorbable polymer by utilizing a method as described in Chapter <233> of the U.S.
Pharmacopeia. The specific 24 different Elemental Impurities of interest are outlined in both USP <232> and in Table A.2.2 of the
ICH HARMONISED GUIDELINE FOR ELEMENTAL IMPURITIES - Q3D (Dec 2014). Both of these documents include
risk-based approaches toward the assessment and control of elemental impurities.
5.7.3 Heavy metals generally refers to divalent cations of the elements cadmium (Cd), copper (Cu), mercury (Hg), and lead
(Pb), to the trivalent cations of antimony (Sb), arsenic (As), and bismuth (Bi), and to tetravalent (stannic) tin (SnExcept for
4+
elements intentionally added as catalysts, assess the obtained results for compliance ) that form complexes with sulfide under
slightly acidic conditions.with the Parenteral Concentration limits described Since stannous tin (Snwithin the Individual
2+
Component ) can also form tin (II) sulfide and therefore can potentially influence test results, the excess amount ascertained by
alternative analytic means to be directly attributable to both stannic and stannous tin may be ignored, provided that the cumulative
lead (Pb) equivalent total of the remaining listed heavy metals elements determined through the same alternative analytic means
(see discussion and calculations Option of USP <232>, Table 3 (derived from ICH-Q3D Option 1, Table A.2.2). If all listed
elements, except for those added as catalysts, can be assured to be maintained within the Parenteral Concentration - Individual
Component Option limits, the resin “complies” with the USP <232> ELEMENTAL IMPURITIES – LIMITS (except catalyst). If
any listed element (other than added catalyst) cannot be controlled to be maintained within the described <232> limits, the resin
F2579 − 18
Supplied example NMR spectra of amorphous 85 % DL-PLA:15% PGA and 50 % DL-PLA:50 % PGA (mole ratio) copolymers are courtesy of W. L. Gore & Associates,
Inc., Flagstaff, AZ 86001, USA.
FIG. 4 Poly(DL-lactide-co-glycolide) Nuclear Magnetic Resonance Spectra
does not comply with the USP <232> ELEMENTAL IMPURITIES – LIMITS (except catalyst) and the concentration (in ppm, in
X2.5) remains below a 10 ppm as lead (Pb) limit.accordance with USP <233> or equivalent) of each uncontrolled element is to
be both monitored and reported.
5.7.3.1 The Elemental Impurities thresholds for the Individual Component Option of USP <232>, Table 3, provide specific
elemental daily dosage limits for parenteral drug products. These daily Elemental Impurity limits (including those applied to
catalyst concentrations) should be considered as conservative thresholds
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...