ASTM F3294-18
(Guide)Standard Guide for Performing Quantitative Fluorescence Intensity Measurements in Cell-based Assays with Widefield Epifluorescence Microscopy
Standard Guide for Performing Quantitative Fluorescence Intensity Measurements in Cell-based Assays with Widefield Epifluorescence Microscopy
SIGNIFICANCE AND USE
5.1 Overview of Measurement System—Relative intensity measurements made by widefield epifluorescence microscopy are used as part of cell-based assays to quantify attributes such as the abundance of probe molecules (see ASTM F2997), fluorescently labeled antibodies, or fluorescence protein reporter molecules. The general procedure for quantifying relative intensities is to acquire digital images, then to perform image analysis to segment objects and compute intensities. The raw digital images acquired by epifluorescence microscopy are not typically amenable to relative intensity quantification because of the factors listed in 4.2. This guide offers a checklist of potential sources of bias that are often present in fluorescent microscopy images and suggests approaches for storing and normalizing raw image data to assure that computations are unbiased.
5.2 Areas of Application—Widefield fluorescence microscopy is frequently used to measure the location and abundance of fluorescent probe molecules within or between cells. In instances where RIM comparisons are made between a region of interest (ROI) and another ROI, accurate normalization procedures are essential to the measurement process to minimize biased results. Example use cases where this guidance document may be applicable include:
5.2.1 Characterization of cell cycle distribution by quantifying the abundance of DNA in individual cells (1).7
5.2.2 Measuring the area of positively stained mineralized deposits in cell cultures (ASTM F2997).
5.2.3 Quantifying the spread area of fixed cells (ASTM F2998).
5.2.4 Determining DNA damage in eukaryotic cells using the comet assay (ASTM E2186).
5.2.5 The quantitation of a secondary fluorescent marker that provides information related to the genotype, phenotype, biological activity, or biochemical features of a colony or cell (ASTM F2944).
SCOPE
1.1 This guidance document has been developed to facilitate the collection of microscopy images with an epifluorescence microscope that allow quantitative fluorescence measurements to be extracted from the images. The document is tailored to cell biologists that often use fluorescent staining techniques to visualize components of a cell-based experimental system. Quantitative comparison of the intensity data available in these images is only possible if the images are quantitative based on sound experimental design and appropriate operation of the digital array detector, such as a charge coupled device (CCD) or a scientific complementary metal oxide semiconductor (sCMOS) or similar camera. Issues involving the array detector and controller software settings including collection of dark count images to estimate the offset, flat-field correction, background correction, benchmarking of the excitation lamp and the fluorescent collection optics are considered.
1.2 This document is developed around epifluorescence microscopy, but it is likely that many of the issues discussed here are applicable to quantitative imaging in other fluorescence microscopy systems such as fluorescence confocal microscopy. This guide is developed around single-color fluorescence microscopy imaging or multi-color imaging where the measured fluorescence is spectrally well separated.
1.3 Fluorescence intensity is a relative measurement and does not in itself have an associated SI unit. This document does discuss metrology issues related to relative measurements and experimental designs that may be required to ensure quantitative fluorescence measurements are comparable after changing microscope, sample, and lamp configurations.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was ...
General Information
- Status
- Published
- Publication Date
- 30-Sep-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.46 - Cell Signaling
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Mar-2018
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Nov-2013
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Jul-2012
- Effective Date
- 01-May-2012
- Effective Date
- 01-Mar-2012
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2009
- Effective Date
- 01-Jun-2009
- Effective Date
- 15-Apr-2009
- Effective Date
- 01-Jan-2009
Overview
ASTM F3294-18 is a standard guide developed by ASTM International to support quantitative fluorescence intensity measurements in cell-based assays using widefield epifluorescence microscopy. This guideline is particularly relevant for cell biologists and researchers employing fluorescence staining techniques to visualize and quantify cellular components. By addressing potential sources of measurement bias and outlining best practices for image acquisition and analysis, the standard ensures reliable and reproducible relative intensity measurements in fluorescence microscopy.
Quantitative analysis of fluorescence intensity is essential in many biological applications, such as assessing the abundance of probe molecules, fluorescently labeled antibodies, or reporter proteins in cells. The standard emphasizes the importance of sound experimental design and proper management of digital imaging systems-such as CCD, sCMOS, or similar cameras-to obtain valid and comparable results.
Key Topics
- Quantitative Fluorescence Measurements: Guidance on extracting relative intensity data from epifluorescence microscopy images, vital for analyzing cellular and molecular phenomena.
- Image Acquisition and Normalization: Recommendations for acquiring digital images, including the correction of biases such as CCD bias, flat-field correction for field non-uniformities, and ensuring linearity in detector response.
- Data Storage and Compression: Emphasizes the importance of using lossless image formats (such as TIFF) to prevent distortion of intensity data and avoid biases introduced by lossy compression.
- Instrument Calibration and Validation: Addresses the need for periodic benchmarking of microscope illumination, detector settings, and optical components to ensure consistency and accuracy.
- Areas of Application: Applicable to diverse assays, including cell cycle analysis, DNA damage assays, quantification of mineralized deposits, and spread area measurements.
- Metrology Considerations: Discusses the relative nature of fluorescence intensity, the challenges in comparing data across different instruments, and the importance of experimental controls and instrument calibration.
Applications
Proper implementation of ASTM F3294-18 makes quantitative fluorescence microscopy a robust, reproducible technique for cell-based studies. Key applications include:
- Cell Cycle Assessment: Quantifying DNA content in single cells for cell cycle analysis.
- Mineralization Studies: Measuring areas of stained mineralized deposits in progenitor cell cultures.
- Cell Morphology Analysis: Quantifying cell spread areas in fixed cell assays.
- DNA Damage Detection: Facilitating the comet assay for measuring DNA single-strand breaks in eukaryotic cells.
- Multiparameter Analysis: Determining genotype, phenotype, biological activity, or other features using secondary fluorescent markers.
By following this standard, laboratories can enhance the precision of their quantitative fluorescence assays, reduce measurement variability, and ensure that imaging data is scientifically valuable over time.
Related Standards
ASTM F3294-18 interconnects with several other international and ASTM standards related to microscopy and digital imaging, including:
- ASTM F2997 - Practice for Quantification of Calcium Deposits in Osteogenic Culture of Progenitor Cells Using Fluorescent Image Analysis
- ASTM F2998 - Guide for Using Fluorescence Microscopy to Quantify the Spread Area of Fixed Cells
- ASTM E2186 - Guide for Determining DNA Single-Strand Damage in Eukaryotic Cells Using the Comet Assay
- ASTM F2944 - Test Method for Automated Colony Forming Unit (CFU) Assays-Image Acquisition and Analysis Method
- ASTM E2719 - Guide for Fluorescence-Instrument Calibration and Qualification
- ISO 13653 - Measurement of relative irradiance in the image field
- ISO/IEC 10918-1 - Digital compression and coding of continuous-tone still images (JPEG)
- EMVA 1288 - Standard for Characterization and Presentation of Specification Data for Image Sensors and Cameras
Practical Value
Implementing ASTM F3294-18 enhances the reliability of cell-based fluorescence assays by providing a clear, standardized approach for image correction, normalization, and data management. This drives higher data integrity in research, supports regulatory compliance, and facilitates methodologically sound scientific publications-all critical for advancing biomedical research and diagnostics leveraging widefield epifluorescence microscopy.
Buy Documents
ASTM F3294-18 - Standard Guide for Performing Quantitative Fluorescence Intensity Measurements in Cell-based Assays with Widefield Epifluorescence Microscopy
Frequently Asked Questions
ASTM F3294-18 is a guide published by ASTM International. Its full title is "Standard Guide for Performing Quantitative Fluorescence Intensity Measurements in Cell-based Assays with Widefield Epifluorescence Microscopy". This standard covers: SIGNIFICANCE AND USE 5.1 Overview of Measurement System—Relative intensity measurements made by widefield epifluorescence microscopy are used as part of cell-based assays to quantify attributes such as the abundance of probe molecules (see ASTM F2997), fluorescently labeled antibodies, or fluorescence protein reporter molecules. The general procedure for quantifying relative intensities is to acquire digital images, then to perform image analysis to segment objects and compute intensities. The raw digital images acquired by epifluorescence microscopy are not typically amenable to relative intensity quantification because of the factors listed in 4.2. This guide offers a checklist of potential sources of bias that are often present in fluorescent microscopy images and suggests approaches for storing and normalizing raw image data to assure that computations are unbiased. 5.2 Areas of Application—Widefield fluorescence microscopy is frequently used to measure the location and abundance of fluorescent probe molecules within or between cells. In instances where RIM comparisons are made between a region of interest (ROI) and another ROI, accurate normalization procedures are essential to the measurement process to minimize biased results. Example use cases where this guidance document may be applicable include: 5.2.1 Characterization of cell cycle distribution by quantifying the abundance of DNA in individual cells (1).7 5.2.2 Measuring the area of positively stained mineralized deposits in cell cultures (ASTM F2997). 5.2.3 Quantifying the spread area of fixed cells (ASTM F2998). 5.2.4 Determining DNA damage in eukaryotic cells using the comet assay (ASTM E2186). 5.2.5 The quantitation of a secondary fluorescent marker that provides information related to the genotype, phenotype, biological activity, or biochemical features of a colony or cell (ASTM F2944). SCOPE 1.1 This guidance document has been developed to facilitate the collection of microscopy images with an epifluorescence microscope that allow quantitative fluorescence measurements to be extracted from the images. The document is tailored to cell biologists that often use fluorescent staining techniques to visualize components of a cell-based experimental system. Quantitative comparison of the intensity data available in these images is only possible if the images are quantitative based on sound experimental design and appropriate operation of the digital array detector, such as a charge coupled device (CCD) or a scientific complementary metal oxide semiconductor (sCMOS) or similar camera. Issues involving the array detector and controller software settings including collection of dark count images to estimate the offset, flat-field correction, background correction, benchmarking of the excitation lamp and the fluorescent collection optics are considered. 1.2 This document is developed around epifluorescence microscopy, but it is likely that many of the issues discussed here are applicable to quantitative imaging in other fluorescence microscopy systems such as fluorescence confocal microscopy. This guide is developed around single-color fluorescence microscopy imaging or multi-color imaging where the measured fluorescence is spectrally well separated. 1.3 Fluorescence intensity is a relative measurement and does not in itself have an associated SI unit. This document does discuss metrology issues related to relative measurements and experimental designs that may be required to ensure quantitative fluorescence measurements are comparable after changing microscope, sample, and lamp configurations. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was ...
SIGNIFICANCE AND USE 5.1 Overview of Measurement System—Relative intensity measurements made by widefield epifluorescence microscopy are used as part of cell-based assays to quantify attributes such as the abundance of probe molecules (see ASTM F2997), fluorescently labeled antibodies, or fluorescence protein reporter molecules. The general procedure for quantifying relative intensities is to acquire digital images, then to perform image analysis to segment objects and compute intensities. The raw digital images acquired by epifluorescence microscopy are not typically amenable to relative intensity quantification because of the factors listed in 4.2. This guide offers a checklist of potential sources of bias that are often present in fluorescent microscopy images and suggests approaches for storing and normalizing raw image data to assure that computations are unbiased. 5.2 Areas of Application—Widefield fluorescence microscopy is frequently used to measure the location and abundance of fluorescent probe molecules within or between cells. In instances where RIM comparisons are made between a region of interest (ROI) and another ROI, accurate normalization procedures are essential to the measurement process to minimize biased results. Example use cases where this guidance document may be applicable include: 5.2.1 Characterization of cell cycle distribution by quantifying the abundance of DNA in individual cells (1).7 5.2.2 Measuring the area of positively stained mineralized deposits in cell cultures (ASTM F2997). 5.2.3 Quantifying the spread area of fixed cells (ASTM F2998). 5.2.4 Determining DNA damage in eukaryotic cells using the comet assay (ASTM E2186). 5.2.5 The quantitation of a secondary fluorescent marker that provides information related to the genotype, phenotype, biological activity, or biochemical features of a colony or cell (ASTM F2944). SCOPE 1.1 This guidance document has been developed to facilitate the collection of microscopy images with an epifluorescence microscope that allow quantitative fluorescence measurements to be extracted from the images. The document is tailored to cell biologists that often use fluorescent staining techniques to visualize components of a cell-based experimental system. Quantitative comparison of the intensity data available in these images is only possible if the images are quantitative based on sound experimental design and appropriate operation of the digital array detector, such as a charge coupled device (CCD) or a scientific complementary metal oxide semiconductor (sCMOS) or similar camera. Issues involving the array detector and controller software settings including collection of dark count images to estimate the offset, flat-field correction, background correction, benchmarking of the excitation lamp and the fluorescent collection optics are considered. 1.2 This document is developed around epifluorescence microscopy, but it is likely that many of the issues discussed here are applicable to quantitative imaging in other fluorescence microscopy systems such as fluorescence confocal microscopy. This guide is developed around single-color fluorescence microscopy imaging or multi-color imaging where the measured fluorescence is spectrally well separated. 1.3 Fluorescence intensity is a relative measurement and does not in itself have an associated SI unit. This document does discuss metrology issues related to relative measurements and experimental designs that may be required to ensure quantitative fluorescence measurements are comparable after changing microscope, sample, and lamp configurations. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was ...
ASTM F3294-18 is classified under the following ICS (International Classification for Standards) categories: 07.100.01 - Microbiology in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3294-18 has the following relationships with other standards: It is inter standard links to ASTM E2186-02a(2023), ASTM E2825-18, ASTM E2186-02a(2016), ASTM E284-13b, ASTM E284-13a, ASTM E284-13, ASTM E284-12, ASTM E2825-12, ASTM F2944-12, ASTM E131-10, ASTM E2186-02a(2010), ASTM E2719-09, ASTM E284-09a, ASTM E2642-09, ASTM E284-09. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3294-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F3294 −18
Standard Guide for
Performing Quantitative Fluorescence Intensity
Measurements in Cell-based Assays with Widefield
Epifluorescence Microscopy
This standard is issued under the fixed designation F3294; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.5 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.1 Thisguidancedocumenthasbeendevelopedtofacilitate
ization established in the Decision on Principles for the
the collection of microscopy images with an epifluorescence
Development of International Standards, Guides and Recom-
microscope that allow quantitative fluorescence measurements
mendations issued by the World Trade Organization Technical
to be extracted from the images. The document is tailored to
Barriers to Trade (TBT) Committee.
cell biologists that often use fluorescent staining techniques to
visualize components of a cell-based experimental system.
2. Referenced Documents
Quantitativecomparisonoftheintensitydataavailableinthese
2.1 ASTM Standards:
images is only possible if the images are quantitative based on
sound experimental design and appropriate operation of the E131Terminology Relating to Molecular Spectroscopy
E284Terminology of Appearance
digital array detector, such as a charge coupled device (CCD)
E2186Guide for Determining DNA Single-Strand Damage
or a scientific complementary metal oxide semiconductor
in Eukaryotic Cells Using the Comet Assay
(sCMOS)orsimilarcamera.Issuesinvolvingthearraydetector
E2642Terminology for Scientific Charge-Coupled Device
and controller software settings including collection of dark
(CCD) Detectors
count images to estimate the offset, flat-field correction,
E2719Guide for Fluorescence—Instrument Calibration and
background correction, benchmarking of the excitation lamp
Qualification
and the fluorescent collection optics are considered.
E2825Guide for Forensic Digital Image Processing
1.2 This document is developed around epifluorescence
F2944Test Method for Automated Colony Forming Unit
microscopy, but it is likely that many of the issues discussed
(CFU) Assays—Image Acquisition and Analysis Method
here are applicable to quantitative imaging in other fluores-
forEnumeratingandCharacterizingCellsandColoniesin
cence microscopy systems such as fluorescence confocal
Culture
microscopy. This guide is developed around single-color fluo-
F2997Practice for Quantification of Calcium Deposits in
rescencemicroscopyimagingormulti-colorimagingwherethe
Osteogenic Culture of Progenitor Cells Using Fluorescent
measured fluorescence is spectrally well separated.
Image Analysis
1.3 Fluorescence intensity is a relative measurement and
F2998Guide for Using Fluorescence Microscopy to Quan-
does not in itself have an associated SI unit. This document
tify the Spread Area of Fixed Cells
doesdiscussmetrologyissuesrelatedtorelativemeasurements
2.2 ISO Standards:
and experimental designs that may be required to ensure
ISO 13653Measurement of relative irradiance in the image
quantitative fluorescence measurements are comparable after
field
changing microscope, sample, and lamp configurations.
ISO/IEC 10918-1:1994Digital compression and coding of
1.4 This standard does not purport to address all of the
continuous-tonestillimages:Requirementsandguidelines
safety concerns, if any, associated with its use. It is the
ISO/TR12033:2009Guidancefortheselectionofdocument
responsibility of the user of this standard to establish appro-
image compression methods
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This guide is under the jurisdiction ofASTM Committee F04 on Medical and contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Surgical Materials and Devices and is the direct responsibility of Subcommittee Standards volume information, refer to the standard’s Document Summary page on
F04.46 on Cell Signaling. the ASTM website.
Current edition approved Oct. 1, 2018. Published October 2018. DOI: 10.1520/ Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
F3294-18. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3294−18
2.3 Other Documents: 3.1.9 full well capacity, n—the maximum number of photo-
SWGDE/SWGIT Glossary SWGDE and SWGIT Digital & electrons that can be collected on a single pixel in the image
Multimedia Evidence Glossary,updated June 8, 2012 area or in the horizontal register of a CCD. It is typically
U.S. Food and DrugAdministration (FDA) Guidance,Tech- specified in terms of number of electrons. E2642
nical Performance Assessment of Digital Pathology
3.1.10 irradiance, E, E,n—the radiant flux incident per
5 e
Whole Slide Imaging Devices
unit area. E284
European Machine Vision Association (EMVA) Standard
3.1.11 lossless compression, n—compression in which no
1288Standard for Characterization and Presentation of
dataarelostandalldatacanberetrievedintheiroriginalform.
Specification Data for Image Sensors and Cameras
SWGDE/SWGIT Glossary
3. Terminology
3.1.12 lossy compression, n—compressioninwhichdataare
3.1 Definitions of Terms Specific to This Standard:
lost and cannot be retrieved in their original form.
3.1.1 CCD bias, n—theminimumanalogoffsetaddedtothe
SWGDE/SWGIT Glossary
signal before the A/D converter to ensure a positive digital
3.1.13 photobleaching, n—loss of emission or absorption
output each time a signal is read out. E2642
intensity by a sample as a result of exposure to optical
3.1.2 charge-coupled device (CCD), n—a silicon-based
radiation. E2719
semiconductor chip consisting of a two-dimensional matrix of
3.1.14 pixel, n—abbreviation for picture element. The
photo sensors or pixels. E2642
smallest unit in an optical device in which charge is collected
3.1.3 complementary metal oxide semiconductor (CMOS),
as a signal. CCD detectors typically have 26 µm square pixels;
n—technology widely used to manufacture electronic devices
however, pixel sizes of 8, 13, 16, and 20 µm square are also
and image sensors similar to CCDs. In a CMOS sensor, each
available. E2642
pixel has its own charge-to-voltage conversion circuit, and the
3.1.15 radiant energy, n—energytransmittedaselectromag-
sensor often also includes amplifiers, noise correction, and
netic radiation. E284
digitization circuits. Due to the additional components associ-
ated with each pixel, the sensitivity to light is lower than with
3.1.16 radiant flux, Φ,n—the time rate of flow of radiant
a CCD, the signal is noisier, and the uniformity is lower. But
energy; radiant power. E284
the sensor can be built to require less off-chip circuitry for
3.1.17 region of interest (ROI), n—user-defined portion of
basic operation. E2642
the image area in which data will be acquired. The remainder
3.1.4 dynamic range, n—the ratio of the full well saturation
of the image area will be discarded. E2642
charge to the system noise level. It represents the ratio of the
brightestanddarkestsignalsadetectorcanmeasureinasingle
4. Summary of Guide
measurement. E2642
4.1 Wide-field fluorescence microscopy is an optical imag-
3.1.5 electron-multiplyingCCD(EMCCD),n—typeofCCD
ing technique that relies on illumination of the entire field of
that has a two-way readout register, that is, the shift register
view of a fluorescence microscope and simultaneous detection
andthegainregister,eachwithitsownoutputamplifier.When
oftheemittedfluorescencefromallorasub-regionofthefield
the charge is read out through the shift register, the detector
of view using a camera. The emitted fluorescence can be
works like a standard CCD detector, and when the charge is
measured as an intensity value in fluorescence microscopy,
read out through the gain register, it undergoes charge ampli-
which is computed by summing together the intensity values
fication as a result of a different electrode structure embedded
from a group of individual pixels in a digital image acquired
underneath the pixels of this register. E2642
usingadigitalcamera,suchasaCCD,sCMOS,orEMCCD.A
3.1.6 exposure time, n—the length of time for which a CCD
relativeintensitymeasurement(RIM)isdeterminedastheratio
accumulated charge. E2642
of one intensity measurement to another, the result of which
should be an accurate estimate of the ratio of the irradiance
3.1.7 fluorescence, n—the emission of radiant energy from
from part or all of a specimen to the irradiance from part or all
anatom,molecule,orionresultingfromabsorptionofaphoton
of the same or another specimen.
and a subsequent transition to the ground state without a
change in total spin quantum number. E131
4.2 The quantitative comparison of RIMs can be compro-
3.1.8 frame, n—one full image that is read out of a CCD.
mised or invalidated by many possible factors including the
E2642
non-uniformity of intensities across the field of view of the
microscope, the presence of an offset in the pixel values in the
Available from Scientific Working Group on Imaging Technology (SWGIT),
recorded digital image, the intensity signals in the image
http://www.swgit.org
exceeding the linear dynamic range of the camera, or the
Available from U.S. Department of Health and Human Services, Food and
inaccurate recording of the pixel values in image data files due
DrugAdministration (FDA), Center for Devices and Radiological Health (CDRH).
tofactorssuchasalossycompressionoperationorunexpected
https://www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/
GuidanceDocuments/UCM435355.pdf. You may also send an e-mail request to
modification of the pixel bit depth when saving each file. The
CDRH-Guidance@fda.hhs.gov to receive a copy of the guidance. Please use the
quantitative comparison of RIMs can also be compromised by
document number 1400053 to identify the guidance you are requesting.
low signal-to-noise ratio of the measured light intensities or
Available from European Machine Vision Association. http://www.emva.org/
standards-technology/emva-1288/. instability in the optical power of the illumination source.
F3294−18
4.3 This guide provides a list of corrections and normaliza- called the CCD bias (a.k.a. bias current offset or dark counts),
tions that are required so that RIMs can be accurately com- is often not zero. The CCD bias is an offset that is added to
pared within and between images. This guidance document each pixel in the digital image. Accurate determination of the
also includes a list of references to peer-reviewed, published valueoftheCCDbiasiscriticalasitwillneedtobesubtracted
methods that can aid in the qualification of instrument perfor- from the raw image.
mance in Appendix X1. The collection of these resources
6.1.1.1 A CCD bias that is less than zero is problematic.
should be useful in the design of robust cell-based assays that
Digital images are typically saved in files that store only
used quantitative fluorescence microscopy for data collection.
positive integer values. If the CCD bias is less than zero, an
unknownoffsetissubtractedfromeachpixelvalueandrelative
5. Significance and Use
intensity comparisons will not be possible.
5.1 Overview of Measurement System—Relative intensity
6.1.2 Linear Dynamic Range—Images of the specimen
measurements made by widefield epifluorescence microscopy
under evaluation must be collected with the signal within the
are used as part of cell-based assays to quantify attributes such
linear dynamic range of the detector. Signals that are at or
as the abundance of probe molecules (see ASTM F2997), below the noise floor of the camera will not be detected.
fluorescently labeled antibodies, or fluorescence protein re-
Similarly, signals that are above the detector saturation are no
porter molecules. The general procedure for quantifying rela- longer in the linear range of the camera and cannot be used in
tive intensities is to acquire digital images, then to perform
relative intensity evaluations.
imageanalysistosegmentobjectsandcomputeintensities.The
6.1.3 Non-Uniform Field—The intensities measured from a
rawdigitalimagesacquiredbyepifluorescencemicroscopyare
uniformly fluorescent sample are typically not uniform across
not typically amenable to relative intensity quantification
the field of view. Field non-uniformities can arise from many
becauseofthefactorslistedin4.2.Thisguideoffersachecklist
factors, including non-uniform illumination, vignetting, and
ofpotentialsourcesofbiasthatareoftenpresentinfluorescent
non-uniformities in the detector (ISO 13653 and (3, 4)). This
microscopy images and suggests approaches for storing and
meansthatthemeasuredfluorescenceintensityisdependenton
normalizing raw image data to assure that computations are
its position within the field of view. If measurements are to be
unbiased.
madeinaregionofthefieldofviewwithunevenillumination,
aflatfieldcorrectionshouldbeapplied.Anappropriateflatfield
5.2 Areas of Application—Widefield fluorescence micros-
field correction will result in measured intensities that are not
copy is frequently used to measure the location and abundance
dependent on their location within the field of view and can be
of fluorescent probe molecules within or between cells. In
compared.
instances where RIM comparisons are made between a region
6.1.4 Save Raw Images or Use Lossless Compression—
of interest (ROI) and another ROI, accurate normalization
Many of the software packages that are used for controlling
procedures are essential to the measurement process to mini-
image capture from digital cameras offer the opportunity to
mize biased results. Example use cases where this guidance
save images in a lossy compression format (e.g. jpeg). This
document may be applicable include:
formofcompressioncanaltertheintensitydatainanon-linear
5.2.1 Characterization of cell cycle distribution by quanti-
fashion, leading to unpredictable biases in relative intensity
fying the abundance of DNA in individual cells (1).
measurements. It is best to save raw, non-compressed image
5.2.2 Measuring the area of positively stained mineralized
data or a lossless compression format (e.g. tiff) for images that
deposits in cell cultures (ASTM F2997).
are intended to be used to make relative intensity measure-
5.2.3 Quantifying the spread area of fixed cells (ASTM
F2998). ments.Additionalmodificationstothepixelvaluesofanimage
can occur due to unexpected settings on the image analysis
5.2.4 Determining DNA damage in eukaryotic cells using
software. For example, bit truncation or bit depth conversion
the comet assay (ASTM E2186).
can occur on a saved image. It is worthwhile to evaluate the
5.2.5 The quantitation of a secondary fluorescent marker
histogram of pixel intensities on the saved image and compare
that provides information related to the genotype, phenotype,
it to the histogram of the collected image to ensure the image
biological activity, or biochemical features of a colony or cell
is appropriately saved. A good resource for information on
(ASTM F2944).
lossless and lossy image compression formats can be found in
6. Measurement Bias
ASTM E2825-12, ISO/TR 12033:2009, and ISO/IEC 10918-
1:1994.
6.1 Sources of bias in relative intensity measurements are
listed below:
7. Normalization Strategies for Sources of Bias
6.1.1 CCD Bias—The detectors used in widefield fluores-
cencemicroscopyaretypicallyscientificcomplementarymetal
7.1 CCD Bias—To measure the CCD bias, a dark frame
oxide semiconductor (sCMOS) sensors, charge-coupled de-
mustbecollectedintheabsenceofilluminationincidentonthe
vices (CCDs), electron multiplying (EM)-CCDs, or similar
detector. This estimate for the CCD bias will only be accurate
typesofarrayedphotodetectors (2).Regardlessofthedetector,
for the detector settings for which the image was taken. If any
the recorded digital signal in the absence of incident light,
detector settings are changed, such as the temperature, gain, or
binning, the CCD bias estimate may no longer be valid and
may need to be remeasured. The CCD bias may change over
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
this standard. thecourseofadataacquisitionruncaused,forexample,bythe
F3294−18
camera reaching a steady-state operating temperature during ment would include the offset associated with the CCD bias as
startup.Therefore,considerationshouldbegiventomeasuring wellasanybackgroundintensitycomingfromthesamplesuch
theCCDbiasatthebeginningandendofadataacquisitionrun
asfluorescencefromthesubstrateorthemediasurroundingthe
to assure that no change in the CCD bias has occurred. cells.
7.1.1 A simple approach for estimating the CCD bias is to
7.2 Linear Dynamic Range—Electronic image sensors are
turn off the microscope epi-fluorescent illumination and ac-
expected to be linear over a limited range.The upper bound of
quire at least 5 images at the same exposure time and same
the linear dynamic range, saturation, can be determined by
microscope settings (objective, filter sets, etc.) as used for the
acquiring images from a uniformly fluorescent specimen at
quantitative fluorescence images of the samples.
increasing exposure times (5). By taking two identical image
7.1.2 Amore general approach is to turn off the microscope
framesandsubtractingthempixelbypixel,theresultantimage
epi-fluorescent illumination and acquire at least 5 images at 5
produced is composed only of noise related to the intensity
or more exposure times. Plotting the mean intensity for each
measurementbyeachpixel.AsillustratedinFig.2,plottingthe
dark frame versus exposure time can reveal the presence of
mean frame intensity versus the pixel variance can be used to
background light that is incident on the detector. A regression
identify the intensity value at which the sensor begins to
analysisofthemeanintensityversusexposuretimecanbeused
saturate. The pixel variance increases with the mean frame
to estimate the CCD bias value for any exposure time. An
intensity until saturation is reached. On microscopes equipped
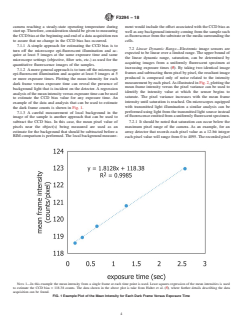
example of the data and analysis that can be used to estimate
with transmitted light illumination a similar analysis can be
the dark frame counts is shown in Fig. 1.
performedusinglightfromthetransmittedlightsourceinstead
7.1.3 A careful measurement of local background in the
offluorescenceemittedfromauniformlyfluorescentspecimen.
image of the sample is another approach that can be used to
subtract the CCD bias. In this case, the mean pixel value of 7.2.1 It should be noted that saturation can occur below the
pixels near the object(s) being measured are used as an maximum pixel range of the camera. As an example, for an
estimate for the background that should be subtracted before a array detector that records each pixel value as a 12-bit integer
RIMcomparisonisperformed.Thelocalbackgroundmeasure- each pixel value will range from 0 to 4095.The recorded pixel
NOTE 1—In this example the mean intensity from a single frame at each time point is used. Least squares regression of the mean intensities is used
to estimate the CCD bias = 118.38 counts. The data shown in the above plot is take from Halter et al. (5), where further details describing the data
acquisition can be found.
FIG. 1Example Plot of the Mean Intensity for Each Dark Frame Versus Exposure Time
F3294−18
NOTE 1—Images were acquired from a stably fluorescent glass at increasing exposure times. As the intensity increases, the pixel variance increases
at first, reaches a maximum, then decreases. The data shown in the above plot are taken from Halter et al. (5), where further details describing the data
acquisition can be found.
FIG. 2Example Plot of the Pixel Variance in Each Frame versus Mean Intensity
value may not be proportional to the incident light intensity is used. Both plots in Fig. 3A and Fig. 3B used data from the
over this entire range as the response of the detector becomes same experiment, so it is clear that the analysis method in Fig.
non-linear at values less than 4095.
2 or Fig. 3A is superior.
7.2.2 Thismethodfordeterminingthesaturationofadigital
7.3 Non-Uniform Field—The ideal correction for a non-
camera attached to a microscope is very similar to typical
uniform field would yield intensity measurements that are
approach (the photon-transfer-technique) that is used for de-
independent of an object’s location in the field. Therefore, any
terminingimagesensorandcamerasaturationthatoccurswhen
correction scheme can be evaluated by moving the sample to
the well is filled to capacity (6, 7), and is part of the
different locations and measuring the intensity of the same
methodology of EMVA Standard 1288.
object (8). During such an evaluation, one must be aware that
7.2.3 CCD and sCMOS detectors are known to exhibit a
photodegradation (i.e. photobleaching) of the sample may
sharp decrease in the image variance when the sensor reaches
change the fluorescence emission from the sample. Measure-
full well capacity. This behavior can provide a well defined
intensity level at which the detector saturates. An example of ments that demonstrate a small level of photodegradation can
the sharp decrease in variance is demonstrated with the data in
be used to support claims that the flatfield correction scheme
the plot in Fig. 2. The saturation can be defined as the
removes biases due to non-uniform field effects.
maximum of the pixel variance versus intensity plot.
7.3.1 A straightforward approach for estimating the non-
7.2.4 Plotting intensity versus exposure may also be an
uniformityacrossthefieldistoimagethefluorescenceemitted
acceptable method for determining saturation. The plot takes
from a uniformly fluorescent reference sample. Imaging of the
on a very similar shape (Fig. 3A) to the mean frame intensity
uniformly fluorescent reference sample must be performed
versus variance (Fig. 2). It is straightforward to determine the
under the same imaging conditions as the sample, since any
saturationpointwherethecurveplateaus.Fig.3Bdemonstrates
change in the optical path, spectral filters, and detection sensor
that the saturation point for the full well capacity can be
overestimatedifthemeanframeintensityversusexposuretime
F3294−18
NOTE 1—(A) As the exposure time increases, the variance increases at first, reaches a maximum, then decreases.
NOTE 2—(B) As the exposure time increases, the mean intensity increases, then gradually plateaus at a mean intensity in the range of 3000 to 3300
counts/pixel.
NOTE3—ThedatashownintheaboveplotaretakenfromHalteretal. (5),wherefurtherdetailsdescribingthedataacquisitioncanbefound.Allimages
were acquired from a stably fluorescent glass at increasing exposure times.
FIG. 3(A) Example Plot of the Pixel Variance in Each Frame versus Exposure Time,
(B) Example Plot of the Mean Pixel Intensity in Each Frame versus Exposure Time
may change the flatfield correction. Candidate reference mate- carbocyanine dissolved in dimethyl formamide (10) in a glass
rials for estimating the flatfield correction include, but are not bottom dish mounted on a depression slide.
limited to: 7.3.1.4 Fluorescent plastic slides (11).
7.3.1.1 High concentration solutions of sodium fluorescein 7.3.1.5 Fluorescent silver nanoclusters embedded in glass
for green fluorescence emission, Rose Bengal orAcid Fuchsin (12).
for red fluorescent specimens, or Acid Blue 9 for far red 7.3.1.6 Fluorescent polymeric material (13).
fluorescent specimens (3, 9). 7.3.2 Implementation of the empirical methods above for
7.3.1.2 Fluorescein embedded in a thin polymer. This thin estimating the flatfield correction need to be optimized by
polymer sample is used to quantify the photobleaching rate at adjusting the camera exposure time to use the full dynamic
each location of an imaged field, which in turn permits range of the camera without saturation. Acquiring several
computation of the flatfield correction (4). images from different fields and averaging the images in the
7.3.1.3 Solutions of 7-dimethylamino-4-methylcoumarin, image acquisition/processing software can reduce the effect of
oxacarbocyanine (diO), indo-carbo cyanine (diI), and indodi- minor artifacts (e.g. from debris) in the real sample and
F3294−18
averagingalsoreducestheeffectofnoise,resultinginanimage almost certainly change the absolute value of the pixel
that represents the illumination pattern in the microscope. In intensities, making comparisons between experiments chal-
addition, under high numerical aperture conditions candidate lenging. They can also misrepresent the linear relationship
reference materials for estimating the flatfield correction betweentheintensities,makingitdifficulttocomparedifferent
shouldincludeamicroscopecoverslipinthelightpathbetween
intensities even within the same field of view. Even without
the objective lens and the sample. This is important when compression, it is possible that unexpected software settings
coverslip-corrected objectives are used (i.e. when ∞/0.17 is
result in saving of modified images that are not quantitative
marked on the barrel of the objective lens). Otherwise, spheri- representations of the image collected. It is important to
cal aberrations that will add biases and uncertainties to
directly compare the histogram of pixel intensities in the
subsequentRIMcomparisonsappearintheflatfieldcorrection. collected image and the saved image to ensure that they are
7.3.3 Thecorrectionforanon-uniformfieldisperformedas: identi
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...