ASTM E2810-23
(Practice)Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units
Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units
SIGNIFICANCE AND USE
4.1 The methodology was originally developed (1-4)6 for use in drug content uniformity and dissolution but has general application to any multistage test with multiple acceptance criteria. Practice E2709 summarizes the statistical aspects of this methodology. This practice applies the general methodology of Practice E2709 specifically to the UDU test.
4.1.1 While other methods can be used to estimate the probability of passing the UDU test, they are outside the scope of this practice.
4.2 The UDU test procedure describes a two-stage sampling test, where at each stage one can pass or continue testing, and the decision to fail is deferred until the second stage. At each stage there are acceptance criteria on the test results as outlined in Table 1.
4.3 The UDU test is a market standard. The USP General Notices include the following statement about compendial standards. “The similarity to statistical procedures may seem to suggest an intent to make inference to some larger group of units, but in all cases, statements about whether the compendial standard is met apply only to the units tested.” Therefore, the UDU procedure is not intended for inspecting uniformity of finished product for lot/batch release or as a lot inspection procedure.
4.3.1 The UDU test defines a product requirement to be met at release and throughout the shelf-life of the product.
4.3.2 Passing the UDU test once does not provide statistical assurance that a batch of drug product meets specified statistical quality control criteria.
4.4 This practice provides a practical specification that may be applied when uniformity of dosage units is required. An acceptance region for the mean and standard deviation of a set of test results from the lot is defined such that, at a prescribed confidence level, the probability that a future sample from the lot will pass the UDU test is greater than or equal to a prespecified lower probability bound. Having test results fall in the acceptance r...
SCOPE
1.1 This practice provides a general procedure for evaluating the capability to comply with the Uniformity of Dosage Units (UDU) test. This test is given in General Chapter Uniformity of Dosage Units of the USP, in 2.9.40 Uniformity of Dosage Units of the Ph. Eur., and in 6.02 Uniformity of Dosage Units of the JP, and these versions are virtually interchangeable. For this multiple-stage test, the procedure computes a lower bound on the probability of passing the UDU test, based on statistical estimates made at a prescribed confidence level from a sample of dosage units.
1.2 This methodology can be used to generate an acceptance limit table, which defines a set of sample means and standard deviations that assures passing the UDU test for a prescribed lower probability bound, confidence level, and sample size.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2023
- Technical Committee
- E55 - Manufacture of Pharmaceutical and Biopharmaceutical Products
- Drafting Committee
- E55.14 - Measurement Systems and Analysis
Relations
- Effective Date
- 01-Dec-2023
Overview
ASTM E2810-23 is the Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units (UDU), developed by ASTM International. This standard provides a systematic statistical procedure to assess and demonstrate whether pharmaceutical products meet the uniformity of dosage unit criteria as defined in regulatory pharmacopeias, including the United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.), and Japanese Pharmacopoeia (JP). The methodology ensures products maintain consistent dosage unit content, which is essential for pharmaceutical quality control and regulatory compliance.
Key Topics
- Uniformity of Dosage Units (UDU): The degree of consistency in drug content among individual dosage units within a batch, critical for patient safety and product efficacy.
- Two-stage Sampling Test: E2810-23 describes a two-stage statistical sampling plan for UDU, with specific acceptance criteria at each stage, allowing either immediate passing, continuation, or deferral of rejection.
- Acceptance Region and Limit: Acceptance regions for the mean and standard deviation of batch samples are established. Results falling within these regions demonstrate, at a prescribed confidence level, that future samples will likely pass the UDU test.
- Probability and Confidence Level: The procedure estimates a lower bound on the probability that a batch will pass the UDU test, typically at common industry benchmarks (e.g., 90%, 95%, or 99%) and confidence levels (commonly 95%).
- General Applicability: While initially intended for drug content uniformity and dissolution, the methodology is applicable to any multistage test with multiple acceptance criteria.
Applications
ASTM E2810-23 is widely used in pharmaceutical manufacturing and quality control laboratories for:
- Process Validation: Demonstrating a process’s ability to consistently produce units that meet UDU requirements.
- Continuous Process Verification: Providing evidence during ongoing manufacturing that dosage uniformity remains within specification.
- In-Process Testing: Supporting in-line or at-line testing strategies for uniformity throughout production.
- Lot Release Assessments: Supplementing batch evaluation prior to product release, ensuring regulatory requirements are met. However, the methodology is not intended as a substitute for lot or batch release decisions applicable to finished product inspections.
- Regulatory Submissions: Documentation of UDU capability according to globally recognized standards, facilitating compliance with US FDA, EU EMA, and Japanese PMDA regulations.
By applying acceptance limit tables based on sample size, mean, and standard deviation, manufacturers can assert with statistical confidence that future samples from a batch meet UDU criteria.
Related Standards
- ASTM E2709: Practice for Demonstrating Capability to Comply with an Acceptance Procedure - summarizes the statistical fundamentals underpinning E2810 and other similar methodologies.
- USP <905> Uniformity of Dosage Units: The United States Pharmacopeia’s regulatory chapter outlining UDU requirements.
- Ph. Eur. 2.9.40: The European Pharmacopoeia’s parallel chapter on UDU.
- JP 6.02: The Japanese Pharmacopoeia’s corresponding UDU testing standard.
- ASTM E2363: Terminology relating to pharmaceutical and biopharmaceutical manufacturing, guiding definitions used in E2810.
Practical Value
Implementing ASTM E2810-23 enables pharmaceutical organizations to:
- Assure product quality and dosing consistency, thereby enhancing patient safety.
- Meet rigorous, harmonized global standards for dosage unit uniformity, supporting international market access.
- Use data-driven, statistically robust methods to establish, monitor, and demonstrate control over product uniformity throughout the product lifecycle.
- Facilitate transparent, well-documented quality assurance processes for regulatory inspections and audits.
By adhering to ASTM E2810-23, stakeholders in pharmaceutical quality assurance can reliably demonstrate compliance with UDU requirements, underpinning commercial success and regulatory confidence.
Buy Documents
ASTM E2810-23 - Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units
REDLINE ASTM E2810-23 - Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E2810-23 is a standard published by ASTM International. Its full title is "Standard Practice for Demonstrating Capability to Comply with the Test for Uniformity of Dosage Units". This standard covers: SIGNIFICANCE AND USE 4.1 The methodology was originally developed (1-4)6 for use in drug content uniformity and dissolution but has general application to any multistage test with multiple acceptance criteria. Practice E2709 summarizes the statistical aspects of this methodology. This practice applies the general methodology of Practice E2709 specifically to the UDU test. 4.1.1 While other methods can be used to estimate the probability of passing the UDU test, they are outside the scope of this practice. 4.2 The UDU test procedure describes a two-stage sampling test, where at each stage one can pass or continue testing, and the decision to fail is deferred until the second stage. At each stage there are acceptance criteria on the test results as outlined in Table 1. 4.3 The UDU test is a market standard. The USP General Notices include the following statement about compendial standards. “The similarity to statistical procedures may seem to suggest an intent to make inference to some larger group of units, but in all cases, statements about whether the compendial standard is met apply only to the units tested.” Therefore, the UDU procedure is not intended for inspecting uniformity of finished product for lot/batch release or as a lot inspection procedure. 4.3.1 The UDU test defines a product requirement to be met at release and throughout the shelf-life of the product. 4.3.2 Passing the UDU test once does not provide statistical assurance that a batch of drug product meets specified statistical quality control criteria. 4.4 This practice provides a practical specification that may be applied when uniformity of dosage units is required. An acceptance region for the mean and standard deviation of a set of test results from the lot is defined such that, at a prescribed confidence level, the probability that a future sample from the lot will pass the UDU test is greater than or equal to a prespecified lower probability bound. Having test results fall in the acceptance r... SCOPE 1.1 This practice provides a general procedure for evaluating the capability to comply with the Uniformity of Dosage Units (UDU) test. This test is given in General Chapter Uniformity of Dosage Units of the USP, in 2.9.40 Uniformity of Dosage Units of the Ph. Eur., and in 6.02 Uniformity of Dosage Units of the JP, and these versions are virtually interchangeable. For this multiple-stage test, the procedure computes a lower bound on the probability of passing the UDU test, based on statistical estimates made at a prescribed confidence level from a sample of dosage units. 1.2 This methodology can be used to generate an acceptance limit table, which defines a set of sample means and standard deviations that assures passing the UDU test for a prescribed lower probability bound, confidence level, and sample size. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The methodology was originally developed (1-4)6 for use in drug content uniformity and dissolution but has general application to any multistage test with multiple acceptance criteria. Practice E2709 summarizes the statistical aspects of this methodology. This practice applies the general methodology of Practice E2709 specifically to the UDU test. 4.1.1 While other methods can be used to estimate the probability of passing the UDU test, they are outside the scope of this practice. 4.2 The UDU test procedure describes a two-stage sampling test, where at each stage one can pass or continue testing, and the decision to fail is deferred until the second stage. At each stage there are acceptance criteria on the test results as outlined in Table 1. 4.3 The UDU test is a market standard. The USP General Notices include the following statement about compendial standards. “The similarity to statistical procedures may seem to suggest an intent to make inference to some larger group of units, but in all cases, statements about whether the compendial standard is met apply only to the units tested.” Therefore, the UDU procedure is not intended for inspecting uniformity of finished product for lot/batch release or as a lot inspection procedure. 4.3.1 The UDU test defines a product requirement to be met at release and throughout the shelf-life of the product. 4.3.2 Passing the UDU test once does not provide statistical assurance that a batch of drug product meets specified statistical quality control criteria. 4.4 This practice provides a practical specification that may be applied when uniformity of dosage units is required. An acceptance region for the mean and standard deviation of a set of test results from the lot is defined such that, at a prescribed confidence level, the probability that a future sample from the lot will pass the UDU test is greater than or equal to a prespecified lower probability bound. Having test results fall in the acceptance r... SCOPE 1.1 This practice provides a general procedure for evaluating the capability to comply with the Uniformity of Dosage Units (UDU) test. This test is given in General Chapter Uniformity of Dosage Units of the USP, in 2.9.40 Uniformity of Dosage Units of the Ph. Eur., and in 6.02 Uniformity of Dosage Units of the JP, and these versions are virtually interchangeable. For this multiple-stage test, the procedure computes a lower bound on the probability of passing the UDU test, based on statistical estimates made at a prescribed confidence level from a sample of dosage units. 1.2 This methodology can be used to generate an acceptance limit table, which defines a set of sample means and standard deviations that assures passing the UDU test for a prescribed lower probability bound, confidence level, and sample size. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2810-23 is classified under the following ICS (International Classification for Standards) categories: 03.120.30 - Application of statistical methods; 11.120.99 - Other standards related to pharmaceutics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2810-23 has the following relationships with other standards: It is inter standard links to ASTM E2810-19. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2810-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2810 − 23
Standard Practice for
Demonstrating Capability to Comply with the Test for
Uniformity of Dosage Units
This standard is issued under the fixed designation E2810; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E2709 Practice for Demonstrating Capability to Comply
with an Acceptance Procedure
1.1 This practice provides a general procedure for evaluat-
ing the capability to comply with the Uniformity of Dosage 2.2 Other Documents:
Units (UDU) test. This test is given in General Chapter <905> JP Japanese Pharmacopoeia
Uniformity of Dosage Units of the USP, in 2.9.40 Uniformity Ph. Eur. European Pharmacopoeia
of Dosage Units of the Ph. Eur., and in 6.02 Uniformity of USP United States Pharmacopeia
Dosage Units of the JP, and these versions are virtually
interchangeable. For this multiple-stage test, the procedure
3. Terminology
computes a lower bound on the probability of passing the UDU
3.1 Definitions—See Terminology E2363 for a more exten-
test, based on statistical estimates made at a prescribed
sive listing of terms in ASTM Committee E55 standards.
confidence level from a sample of dosage units.
3.2 Definitions of Terms Specific to This Standard:
1.2 This methodology can be used to generate an acceptance
3.2.1 acceptable parameter region, n—the set of values of
limit table, which defines a set of sample means and standard
parameters characterizing the distribution of test results for
deviations that assures passing the UDU test for a prescribed
which the probability of passing the lot acceptance procedure
lower probability bound, confidence level, and sample size.
is greater than a prescribed lower bound.
1.3 This standard does not purport to address all of the
3.2.2 acceptance limit, n—the boundary of the acceptance
safety concerns, if any, associated with its use. It is the
region, for example, the maximum sample standard deviation
responsibility of the user of this standard to establish appro-
for a given sample mean or minimum and maximum sample
priate safety, health, and environmental practices and deter-
mean for given standard deviations.
mine the applicability of regulatory limitations prior to use.
3.2.2.1 Discussion—The coefficient of variation (relative
1.4 This international standard was developed in accor-
standard deviation) may be substituted for the standard devia-
dance with internationally recognized principles on standard-
tion where applicable.
ization established in the Decision on Principles for the
3.2.3 acceptance region, n—the set of values of parameter
Development of International Standards, Guides and Recom-
estimates (that is, sample mean and standard deviation) where
mendations issued by the World Trade Organization Technical
confidence limits attain a prescribed lower bound on the
Barriers to Trade (TBT) Committee.
probability of passing a lot acceptance procedure.
2. Referenced Documents
3.2.4 confidence level, C, n—the prescribed overall level for
2.1 ASTM Standards: calculating the uncertainty region of the parameters from the
E2363 Terminology Relating to Manufacturing of Pharma-
sample estimates.
ceutical and Biopharmaceutical Products in the Pharma- 3.2.4.1 Discussion—The preset confidence level is stated as
ceutical and Biopharmaceutical Industry
a percentage, for example, 100 (1 – α) = 95 %, where α is a
risk that is allocated to the two parameters being estimated.
This practice is under the jurisdiction of ASTM Committee E55 on Manufac-
ture of Pharmaceutical and Biopharmaceutical Products and is the direct responsi-
bility of Subcommittee E55.14 on Measurement Systems and Analysis. Available from the Pharmaceuticals and Medical Devices Agency (PMDA),
Current edition approved Dec. 1, 2023. Published December 2023. Originally Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013,
approved in 2011. Last previous edition approved in 2019 as E2810 – 19. DOI: Japan, https://www.pmda.go.jp.
10.1520/E2810-23. Available from the European Directorate for the Quality of Medicines and
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Health Care (EDQM), Council of Europe, 7 allée Kastner, CS 30026, F-67081
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Strasbourg, France, http://www.edqm.eu.
Standards volume information, refer to the standard’s Document Summary page on Available from U.S. Pharmacopeial Convention (USP), 12601 Twinbrook
the ASTM website. Pkwy., Rockville, MD 20852-1790, http://www.usp.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2810 − 23
3.2.5 lower probability bound, LB, n—the nominal probabil- 4. Significance and Use
ity of passing the UDU test for a given set of parameter 6
4.1 The methodology was originally developed (1-4) for
estimates.
use in drug content uniformity and dissolution but has general
3.2.6 multiple-stage acceptance procedure, n—a procedure
application to any multistage test with multiple acceptance
that involves more than one stage of sampling and testing a
criteria. Practice E2709 summarizes the statistical aspects of
given quality characteristic with one or more acceptance
this methodology. This practice applies the general methodol-
criteria per stage.
ogy of Practice E2709 specifically to the UDU test.
4.1.1 While other methods can be used to estimate the
3.2.7 representative sample, n—a sample that consists of a
probability of passing the UDU test, they are outside the scope
number of units that are drawn based on rational criteria such
of this practice.
as random, stratified, or systematic sampling and intended to
assure that the sample accurately portrays the material being
4.2 The UDU test procedure describes a two-stage sampling
sampled.
test, where at each stage one can pass or continue testing, and
the decision to fail is deferred until the second stage. At each
3.2.8 sampling plan, n—scheme for selecting dosage units
stage there are acceptance criteria on the test results as outlined
from locations within a batch for testing purposes.
in Table 1.
3.2.8.1 Discussion—In this standard, either a single dosage
unit is selected from each batch location (called Sampling Plan
4.3 The UDU test is a market standard. The USP General
1) or an equal but greater than one dosage unit is selected from
Notices include the following statement about compendial
each batch location (called Sampling Plan 2).
standards. “The similarity to statistical procedures may seem to
3.2.9 uniformity of dosage units, UDU, n—the degree of suggest an intent to make inference to some larger group of
uniformity in the amount of the drug substance among dosage units, but in all cases, statements about whether the compendial
units. standard is met apply only to the units tested.” Therefore, the
3.2.9.1 Discussion—The requirements of the UDU test ap-
ply to each drug substance in dosage units containing one or
more drug substances, unless otherwise specified. The unifor- 6
The boldface numbers in parentheses refer to a list of references at the end of
mity improves as the variability decreases. this standard.
TABLE 1 Uniformity of Dosage Units Test Procedure
NOTE 1—All measurements of dosage units and criteria values are in
percentage label claim (%LC). At each stage calculate the sample average,
X—, and the sample standard deviation, s.
Number
Stage Pass Stage If:
Tested
¯
S 10 |M – X| + 2.4 s ≤ 15.0, where M is de-
fined below.
¯
S 20 (1) |M – X| + 2.0 s ≤ 15.0, using all 30
results (S + S ).
1 2
(2) No dosage unit is outside the
maximum allowed range of 0.75 * M
to 1.25 * M.
M is defined as follows:
If T is less than or equal to 101.5 %LC, and
¯
(1) If X is less than 98.5 %LC, then
M = 98.5 %LC.
¯
(2) If X is between 98.5 and 101.5 %LC,
¯
then M = X.
¯
(3) If X is greater than 101.5 %LC, then
M = 101.5 %LC.
If T is greater than 101.5 %LC, and
¯
(1) If X is less than 98.5 %LC, then
M = 98.5 %LC.
¯
(2) If X is between 98.5 and T, then
¯
M = X.
¯
(3) If X is greater than T, then M = T.
T is the target content per dosage unit at the time of manufacture, expressed as
%LC. Unless otherwise specified in the individual monograph, T is 100.0 %LC.
E2810 − 23
UDU procedure is not intended for inspecting uniformity of literature (4). A simplified description on the construction and
finished product for lot/batch release or as a lot inspection use of these tables is given in this section. A computer program
procedure. is required to generate the tables given a target T as a
4.3.1 The UDU test defines a product requirement to be met percentage of label claim (LC), a lower probability bound LB,
at release and throughout the shelf-life of the product. a confidence level C, and a sample size n.
4.3.2 Passing the UDU test once does not provide statistical 5.1.2 The first step is to determine the acceptable parameter
assurance that a batch of drug product meets specified statis-
region. On a two-dimensional content space consisting of the
tical quality control criteria. true mean (μ) on the horizontal axis and standard deviation (σ)
on the vertical axis the upper 95 % LB boundary of this region
4.4 This practice provides a practical specification that may
is defined by a contour, a curve that is concave downward and
be applied when uniformity of dosage units is required. An
depicted by the solid curve in Fig. 1. The simulated 95 %
acceptance region for the mean and standard deviation of a set
boundary for the actual probability of passing the USP UDU
of test results from the lot is defined such that, at a prescribed
test is defined by the dashed contour that shows the effect of
confidence level, the probability that a future sample from the
generating a lower bound. The LB contour is determined by the
lot will pass the UDU test is greater than or equal to a
LB probability (95 %) and the target under the assumption that
prespecified lower probability bound. Having test results fall in
the dosage unit content is normally distributed. The acceptable
the acceptance region provides assurance that a sample would
parameter region is the set of points on or below the contour.
pass the UDU test with at least the specified lower bound
Any (μ, σ) pair in the acceptable region would pass the UDU
probability. This procedure does not account for any decrease
test with a probability of at least the LB.
in potency during the shelf life, which could affect the ability
5.1.3 The second step is to generate the acceptance limit
to meet the UDU test requirements.
curve that defines the acceptance region.
4.5 This practice can be used as an element for process
demonstration or validation, continuous process verification,
SAMPLING PLAN 1
¯
in-process testing, or lot release (acceptance). As the circum-
5.1.4 As described in Practice E2709, the sample mean (X)
stances and available information vary in these different
and sample standard deviation (s) estimate the population
application areas, this practice does not prescribe a specific
parameters μ and σ within C % confidence limits as chosen by
target, sample size, lower probability bound, or confidence
the user. The joint confidence region for μ and σ (5) has the
¯
level. These must be prospectively selected by the user and
shape of an inverted triangle around a (X, s) pair as depicted in
¯
may be different from those used in the acceptance limit tables
Fig. 2 with the lowest vertex at (X, 0).
¯
provided in this practice.
5.1.4.1 To create an acceptance region, a value of X is
selected starting with s = 0, then the confidence region is
5. Procedure
expanded by increasing s until one of the upper vertices just
5.1 Generating The Acceptance Limit Table: touches the acceptable parameter region. The size of the
5.1.1 The general procedure that generates the acceptance confidence region is determined by C and n. This value of s
¯
limit tables is described in Practice E2709 and the specific defines a point on the acceptance limit curve at (X, s).
¯
procedure for application to the UDU test is described in the Additional selections of X then generate the acceptance limit
FIG. 1 Example of 95 % Lower Bound and Simulated Probabilities of Passing USP UDU <905>
E2810 − 23
NOTE 1—All points below the lower bound contour have higher than a 95 % chance of passing UDU test if mean and standard deviation are known.
All points below the acceptance region contours pass the associated acceptance limit table for n = 100 and n = 10.
ULS is the upper confidence limit for σ.
Z is a standard normal critical value.
FIG. 2 Example of Simultaneous Confidence Interval with 95 % Lower Bound and Acceptance Regions
curve, as depicted as dotted lines in Fig. 2. Acceptance limit 5.1.6.1 Table 5 is provided at C = 90 % and LB = 95 % for
curves are shown for n = 10 and n = 100, illustrating that the comparison with Table 3 to demonstrate the effect of a lower
acceptance limits approach the acceptable parameter region
confidence level.
with increasing sample size.
NOTE 1—Tables can also be generated for other choices for ranges of
5.1.5 Computer programs have been developed for generat-
means, such as 85.1 to 114.9 %LC, or for other sample sizes.
ing acceptance limit tables, but these may not be available to
all practitioners. This practice contains four acceptance limit
5.1.7 A representative sample of n dosage units is taken
tables for many practical use situations.
from the batch and the active content is measured on each unit,
5.1.6 Using the Acceptance Limit Tables in This Practice—
taken as a random sample or as a systematic sample (selecting
Various acceptance limit tables are given in Tables 2-5. In each
and testing one unit at equally spaced time intervals of
table acceptance limits on the standard deviation are given for
manufacture across the batch). Other sampling plans are
means ranging 90 % to 110 % of LC in increments of 0.2 %LC
outside the scope of this practice.
for sample sizes ranging from n = 10 to n = 500. In all tables,
¯
5.1.8 The sample average (X) and sample standard deviation
the target is set at T = 100 %LC, so the acceptance limits for
(s) are calculated. Enter the table row with the sample average
standard deviations are symmetrical around 100 %LC. This
and the table column with the sample size to find the
target is also required for interchangeability across the ICH
acceptance limit on the standard deviation. If the sample
regions (6). At the confidence level of C = 95 % used often in
standard deviation is at or below the acceptance limit then it
the regulatory arena, three levels of the probability lower
can be asserted at the C % confidence level that the batch will
bound are provided: LB = 90 % (Table 2), LB = 95 % (Table 3)
have an LB % chance of passing the UDU test. Some interpo-
and LB = 99 % (Table 4). These provide 90 %, 95 %, and 99 %
lation may be required for sample sizes and sample means not
coverage, respectively, of the population of dosage units under
consideration. The usual coverage is 95 %. listed in the table.
E2810 − 23
TABLE 2 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 95 %, LB = 90 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 2.91 4.36 4.65 4.84 4.99 5.19 5.33 5.43 5.54 5.66 5.93
99.8 or 100.2 2.88 4.31 4.59 4.79 4.94 5.14 5.28 5.38 5.50 5.62 5.91
99.6 or 100.4 2.84 4.26 4.54 4.74 4.89 5.09 5.24 5.34 5.45 5.58 5.88
99.4 or 100.6 2.81 4.21 4.49 4.69 4.83 5.04 5.18 5.29 5.40 5.53 5.84
99.2 or 100.8 2.77 4.16 4.43 4.63 4.77 4.98 5.13 5.23 5.35 5.48 5.79
99.0 or 101.0 2.74 4.10 4.38 4.57 4.72 4.92 5.07 5.17 5.29 5.43 5.74
98.8 or 101.2 2.70 4.05 4.32 4.52 4.66 4.86 5.01 5.11 5.23 5.37 5.69
98.6 or 101.4 2.67 4.00 4.27 4.46 4.60 4.80 4.94 5.05 5.17 5.30 5.63
98.4 or 101.6 2.63 3.95 4.21 4.40 4.54 4.74 4.88 4.99 5.10 5.24 5.56
98.2 or 101.8 2.60 3.89 4.16 4.34 4.48 4.68 4.82 4.92 5.04 5.17 5.49
98.0 or 102.0 2.56 3.84 4.10 4.28 4.42 4.62 4.75 4.86 4.97 5.10 5.43
97.8 or 102.2 2.53 3.79 4.05 4.22 4.36 4.55 4.69 4.79 4.90 5.03 5.35
97.6 or 102.4 2.49 3.74 3.99 4.17 4.30 4.49 4.62 4.72 4.84 4.97 5.28
97.4 or 102.6 2.46 3.68 3.93 4.11 4.24 4.43 4.56 4.66 4.77 4.90 5.21
97.2 or 102.8 2.42 3.63 3.88 4.05 4.18 4.36 4.50 4.59 4.70 4.83 5.13
97.0 or 103.0 2.39 3.58 3.82 3.99 4.12 4.30 4.43 4.53 4.63 4.76 5.06
96.8 or 103.2 2.35 3.53 3.77 3.93 4.06 4.24 4.37 4.46 4.56 4.69 4.99
96.6 or 103.4 2.32 3.48 3.71 3.87 4.00 4.18 4.30 4.39 4.50 4.62 4.91
96.4 or 103.6 2.28 3.42 3.65 3.81 3.94 4.11 4.23 4.33 4.43 4.55 4.84
96.2 or 103.8 2.24 3.37 3.60 3.76 3.88 4.05 4.17 4.26 4.36 4.48 4.77
96.0 or 104.0 2.21 3.32 3.54 3.70 3.82 3.99 4.10 4.19 4.29 4.41 4.69
95.8 or 104.2 2.17 3.26 3.48 3.64 3.76 3.92 4.04 4.13 4.23 4.34 4.62
95.6 or 104.4 2.14 3.21 3.43 3.58 3.70 3.86 3.98 4.06 4.16 4.27 4.54
95.4 or 104.6 2.10 3.16 3.37 3.52 3.63 3.80 3.91 3.99 4.09 4.20 4.47
95.2 or 104.8 2.07 3.11 3.31 3.46 3.57 3.73 3.84 3.93 4.02 4.13 4.39
95.0 or 105.0 2.03 3.05 3.26 3.40 3.51 3.67 3.78 3.86 3.95 4.06 4.32
94.8 or 105.2 2.00 3.00 3.20 3.35 3.45 3.61 3.71 3.79 3.88 3.99 4.24
94.6 or 105.4 1.96 2.95 3.15 3.29 3.39 3.54 3.65 3.73 3.82 3.92 4.17
94.4 or 105.6 1.93 2.89 3.09 3.23 3.33 3.48 3.58 3.66 3.75 3.85 4.10
94.2 or 105.8 1.89 2.84 3.03 3.17 3.27 3.42 3.52 3.59 3.68 3.78 4.02
94.0 or 106.0 1.86 2.79 2.98 3.11 3.21 3.35 3.45 3.53 3.61 3.71 3.95
93.8 or 106.2 1.82 2.74 2.92 3.05 3.15 3.29 3.39 3.46 3.54 3.64 3.87
93.6 or 106.4 1.79 2.68 2.86 2.99 3.09 3.23 3.32 3.39 3.47 3.57 3.80
93.4 or 106.6 1.75 2.63 2.81 2.93 3.03 3.16 3.25 3.33 3.40 3.50 3.72
93.2 or 106.8 1.72 2.58 2.75 2.87 2.97 3.10 3.19 3.26 3.34 3.43 3.65
93.0 or 107.0 1.68 2.52 2.69 2.81 2.90 3.03 3.12 3.19 3.27 3.36 3.57
92.8 or 107.2 1.65 2.47 2.64 2.75 2.84 2.97 3.06 3.13 3.20 3.28 3.50
92.6 or 107.4 1.61 2.42 2.58 2.70 2.78 2.91 2.99 3.06 3.13 3.21 3.42
92.4 or 107.6 1.57 2.36 2.52 2.64 2.72 2.84 2.93 2.99 3.06 3.14 3.35
92.2 or 107.8 1.54 2.31 2.47 2.58 2.66 2.78 2.86 2.92 2.99 3.07 3.27
92.0 or 108.0 1.50 2.26 2.41 2.52 2.60 2.71 2.80 2.86 2.92 3.00 3.20
91.8 or 108.2 1.47 2.20 2.35 2.46 2.54 2.65 2.73 2.79 2.86 2.93 3.12
91.6 or 108.4 1.43 2.15 2.30 2.40 2.48 2.59 2.66 2.72 2.79 2.86 3.05
91.4 or 108.6 1.40 2.10 2.24 2.34 2.42 2.52 2.60 2.65 2.72 2.79 2.97
91.2 or 108.8 1.36 2.04 2.18 2.28 2.35 2.46 2.53 2.59 2.65 2.72 2.89
91.0 or 109.0 1.33 1.99 2.13 2.22 2.29 2.39 2.47 2.52 2.58 2.65 2.82
90.8 or 109.2 1.29 1.94 2.07 2.16 2.23 2.33 2.40 2.45 2.51 2.58 2.74
90.6 or 109.4 1.26 1.89 2.01 2.10 2.17 2.27 2.33 2.39 2.44 2.51 2.67
90.4 or 109.6 1.22 1.83 1.96 2.04 2.11 2.20 2.27 2.32 2.37 2.44 2.59
90.2 or 109.8 1.19 1.78 1.90 1.98 2.05 2.14 2.20 2.25 2.30 2.37 2.52
90.0 or 110.0 1.15 1.73 1.84 1.93 1.99 2.08 2.14 2.18 2.24 2.30 2.44
5.1.8.1 Example 1—A sample of n = 60 dosage units pro- Acceptance limit 5 4.181@~4.36 2 4.18! ~70 2 60! ⁄ ~80 2 60!#
¯
vided dosage content statistics of X = 98.6 %LC and
5 4.181 0.18 10 ⁄ 20 5 4.27%LC
@~ !~ ! ~ !#
s = 3.91 %LC. Since s was below the upper limit of 4.41 %LC
Since s = 4.29 %LC was above the upper limit of 4.27 %LC,
(using Table 3), we can state with 95 % confidence there is at
we cannot state with 95 % confidence there is at least a 95 %
least a 95 % probability that a future sample taken from the
probability that a future sample taken from the batch will meet
batch will meet the UDU test.
the UDU test.
5.1.8.2 Example 2—A sample of n = 70 dosage units pro-
¯
vided dosage content statistics of X = 97.8 %LC and 5.1.8.3 Example 3—A sample of n = 60 dosage units pro-
¯
s = 4.29 %LC. Since n = was not provided in the Table 3, we vided dosage content statistics of X = 98.6 %LC and
must interpolate between entry n = 60 with the upper limit s = 3.91 %LC at time of manufacture. At the end of shelf life
4.18 %LC and entry n = 80 with the upper limit 4.36 %LC as the dosage units are predicted to have lost potency such that the
¯
follows: effective X = 96.2 %LC. Since s = 3.91 %LC was above the
E2810 − 23
TABLE 3 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 95 %, LB = 95 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 2.81 4.18 4.46 4.64 4.78 4.98 5.11 5.21 5.31 5.43 5.69
99.8 or 100.2 2.77 4.13 4.41 4.59 4.73 4.93 5.07 5.16 5.27 5.39 5.67
99.6 or 100.4 2.74 4.08 4.35 4.54 4.68 4.88 5.02 5.12 5.23 5.35 5.64
99.4 or 100.6 2.70 4.04 4.30 4.49 4.63 4.83 4.96 5.07 5.18 5.30 5.60
99.2 or 100.8 2.67 3.99 4.26 4.43 4.57 4.77 4.91 5.01 5.12 5.25 5.55
99.0 or 101.0 2.64 3.94 4.20 4.39 4.51 4.71 4.85 4.95 5.07 5.20 5.49
98.8 or 101.2 2.60 3.88 4.15 4.33 4.47 4.65 4.79 4.89 5.01 5.14 5.44
98.6 or 101.4 2.57 3.84 4.09 4.27 4.41 4.59 4.73 4.83 4.94 5.07 5.38
98.4 or 101.6 2.54 3.79 4.03 4.21 4.35 4.54 4.67 4.77 4.88 5.01 5.31
98.2 or 101.8 2.50 3.74 3.99 4.16 4.29 4.48 4.61 4.70 4.82 4.94 5.25
98.0 or 102.0 2.47 3.68 3.93 4.11 4.23 4.42 4.55 4.65 4.75 4.88 5.18
97.8 or 102.2 2.43 3.64 3.88 4.05 4.18 4.36 4.49 4.58 4.68 4.81 5.11
97.6 or 102.4 2.40 3.58 3.83 3.99 4.12 4.29 4.42 4.52 4.63 4.74 5.05
97.4 or 102.6 2.37 3.54 3.77 3.93 4.06 4.24 4.36 4.45 4.56 4.68 4.97
97.2 or 102.8 2.33 3.49 3.71 3.88 4.00 4.18 4.30 4.39 4.49 4.62 4.90
97.0 or 103.0 2.30 3.43 3.67 3.82 3.95 4.11 4.24 4.33 4.43 4.55 4.83
96.8 or 103.2 2.26 3.39 3.61 3.77 3.88 4.06 4.17 4.27 4.37 4.48 4.76
96.6 or 103.4 2.23 3.33 3.55 3.71 3.83 4.00 4.12 4.20 4.30 4.41 4.69
96.4 or 103.6 2.20 3.29 3.50 3.65 3.77 3.93 4.05 4.14 4.23 4.35 4.62
96.2 or 103.8 2.16 3.23 3.44 3.60 3.71 3.88 3.99 4.07 4.17 4.28 4.55
96.0 or 104.0 2.13 3.18 3.40 3.54 3.66 3.81 3.93 4.01 4.10 4.21 4.47
95.8 or 104.2 2.09 3.13 3.34 3.49 3.59 3.76 3.86 3.95 4.04 4.15 4.41
95.6 or 104.4 2.06 3.08 3.29 3.43 3.54 3.69 3.80 3.88 3.98 4.08 4.34
95.4 or 104.6 2.03 3.03 3.23 3.38 3.48 3.63 3.74 3.82 3.91 4.02 4.26
95.2 or 104.8 1.99 2.98 3.18 3.31 3.42 3.57 3.67 3.75 3.85 3.94 4.19
95.0 or 105.0 1.96 2.93 3.13 3.26 3.36 3.52 3.61 3.70 3.78 3.88 4.12
94.8 or 105.2 1.92 2.87 3.07 3.20 3.31 3.45 3.56 3.63 3.72 3.81 4.05
94.6 or 105.4 1.89 2.83 3.02 3.15 3.24 3.39 3.49 3.57 3.65 3.75 3.98
94.4 or 105.6 1.86 2.78 2.96 3.09 3.19 3.33 3.43 3.50 3.58 3.68 3.91
94.2 or 105.8 1.82 2.72 2.91 3.03 3.13 3.27 3.36 3.44 3.51 3.61 3.83
94.0 or 106.0 1.79 2.67 2.85 2.98 3.07 3.21 3.30 3.37 3.45 3.54 3.77
93.8 or 106.2 1.75 2.63 2.80 2.92 3.01 3.14 3.24 3.31 3.39 3.47 3.70
93.6 or 106.4 1.72 2.57 2.74 2.86 2.96 3.09 3.17 3.25 3.32 3.41 3.62
93.4 or 106.6 1.69 2.52 2.69 2.81 2.90 3.03 3.11 3.18 3.25 3.34 3.55
93.2 or 106.8 1.65 2.47 2.64 2.75 2.84 2.96 3.05 3.11 3.19 3.27 3.48
93.0 or 107.0 1.62 2.42 2.59 2.70 2.78 2.90 2.99 3.05 3.12 3.21 3.40
92.8 or 107.2 1.58 2.37 2.52 2.64 2.72 2.84 2.92 2.99 3.05 3.14 3.33
92.6 or 107.4 1.55 2.32 2.47 2.58 2.67 2.78 2.86 2.93 2.99 3.07 3.27
92.4 or 107.6 1.52 2.26 2.42 2.52 2.61 2.72 2.80 2.86 2.93 3.00 3.20
92.2 or 107.8 1.48 2.21 2.36 2.47 2.54 2.66 2.74 2.79 2.86 2.93 3.13
92.0 or 108.0 1.45 2.16 2.31 2.41 2.48 2.59 2.68 2.73 2.80 2.87 3.05
91.8 or 108.2 1.41 2.11 2.26 2.35 2.43 2.53 2.61 2.66 2.73 2.80 2.97
91.6 or 108.4 1.38 2.06 2.20 2.30 2.37 2.47 2.55 2.60 2.67 2.73 2.90
91.4 or 108.6 1.35 2.01 2.15 2.24 2.31 2.41 2.49 2.54 2.60 2.67 2.83
91.2 or 108.8 1.31 1.96 2.09 2.18 2.25 2.35 2.43 2.47 2.54 2.60 2.76
91.0 or 109.0 1.28 1.91 2.04 2.13 2.19 2.29 2.36 2.41 2.47 2.53 2.69
90.8 or 109.2 1.24 1.86 1.98 2.07 2.13 2.23 2.30 2.34 2.40 2.46 2.62
90.6 or 109.4 1.21 1.81 1.93 2.01 2.07 2.17 2.23 2.28 2.34 2.39 2.55
90.4 or 109.6 1.18 1.75 1.87 1.95 2.02 2.11 2.17 2.21 2.27 2.33 2.48
90.2 or 109.8 1.14 1.70 1.82 1.90 1.96 2.05 2.11 2.16 2.20 2.26 2.41
90.0 or 110.0 1.11 1.66 1.76 1.85 1.90 1.99 2.04 2.09 2.13 2.20 2.33
upper limit of 3.71 %LC (using Table 3), we cannot state with
mated by = 1 ⁄ r *MSB1 1 2 1 ⁄ r MSE where MSB and
~~ ! ~ ! !
95 % confidence there is at least a 95 % probability that a
MSE are the between location and within location mean square
future sample taken from the batch will meet the UDU test
errors from the analysis of variance table. The confidence limit
throughout the shelf life.
for σ is determined using MSE and MSB (see Practice
E2709). The joint confidence region is as in Sampling Plan 1.
SAMPLING PLAN 2
For fixed values of the between and within location standard
5.1.9 As described in Practice E2709, Sampling Plan 2
¯
deviations, the sample mean (X) is set at 83.5. Then the
(two-stage sampling) divides the population into primary
confidence region is expanded by increasing the sample mean
sampling units (locations). L locations are selected and from
until the left vertex just touches the acceptable parameter
each of them a subsample of r items is taken for a total of N
5Lxr units. The variance of a single observation, σ , is the sum region. Then the sample mean is set at 116.5 and the confidence
of between-location and within-location variances. σ is esti- region is expanded by decreasing the sample mean until the
E2810 − 23
TABLE 4 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 95 %, LB = 99 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 2.62 3.88 4.13 4.31 4.43 4.61 4.73 4.82 4.92 5.03 5.27
99.8 or 100.2 2.59 3.84 4.09 4.26 4.39 4.57 4.69 4.78 4.88 4.99 5.25
99.6 or 100.4 2.56 3.79 4.04 4.21 4.34 4.52 4.64 4.73 4.84 4.95 5.22
99.4 or 100.6 2.53 3.75 3.99 4.16 4.29 4.47 4.59 4.68 4.78 4.90 5.17
99.2 or 100.8 2.49 3.70 3.94 4.11 4.23 4.41 4.54 4.63 4.73 4.85 5.13
99.0 or 101.0 2.46 3.65 3.89 4.06 4.18 4.36 4.48 4.57 4.68 4.79 5.07
98.8 or 101.2 2.43 3.61 3.84 4.01 4.13 4.31 4.43 4.52 4.62 4.74 5.02
98.6 or 101.4 2.40 3.56 3.79 3.95 4.08 4.25 4.37 4.46 4.56 4.68 4.96
98.4 or 101.6 2.37 3.51 3.74 3.90 4.02 4.19 4.31 4.40 4.50 4.62 4.90
98.2 or 101.8 2.34 3.47 3.69 3.85 3.97 4.14 4.26 4.34 4.44 4.56 4.83
98.0 or 102.0 2.31 3.42 3.64 3.80 3.92 4.08 4.20 4.29 4.38 4.50 4.77
97.8 or 102.2 2.27 3.37 3.59 3.75 3.86 4.03 4.14 4.23 4.32 4.44 4.70
97.6 or 102.4 2.24 3.33 3.54 3.70 3.81 3.97 4.09 4.17 4.27 4.38 4.64
97.4 or 102.6 2.21 3.28 3.49 3.64 3.76 3.92 4.03 4.11 4.21 4.31 4.58
97.2 or 102.8 2.18 3.23 3.45 3.59 3.70 3.86 3.97 4.05 4.15 4.25 4.51
97.0 or 103.0 2.15 3.19 3.40 3.54 3.65 3.81 3.91 4.00 4.09 4.19 4.45
96.8 or 103.2 2.12 3.14 3.35 3.49 3.60 3.75 3.86 3.94 4.03 4.13 4.38
96.6 or 103.4 2.08 3.09 3.30 3.44 3.54 3.69 3.80 3.88 3.97 4.07 4.32
96.4 or 103.6 2.05 3.05 3.24 3.38 3.49 3.64 3.74 3.82 3.91 4.01 4.25
96.2 or 103.8 2.02 3.00 3.20 3.33 3.43 3.58 3.68 3.76 3.85 3.95 4.19
96.0 or 104.0 1.99 2.95 3.14 3.28 3.38 3.53 3.63 3.70 3.79 3.88 4.12
95.8 or 104.2 1.96 2.91 3.10 3.23 3.33 3.47 3.57 3.64 3.73 3.82 4.06
95.6 or 104.4 1.93 2.86 3.05 3.18 3.27 3.41 3.51 3.58 3.67 3.76 3.99
95.4 or 104.6 1.89 2.81 2.99 3.12 3.22 3.36 3.45 3.52 3.61 3.70 3.92
95.2 or 104.8 1.86 2.76 2.94 3.07 3.16 3.30 3.40 3.47 3.55 3.64 3.86
95.0 or 105.0 1.83 2.72 2.89 3.02 3.11 3.25 3.34 3.41 3.48 3.58 3.79
94.8 or 105.2 1.80 2.67 2.84 2.97 3.06 3.19 3.28 3.35 3.43 3.51 3.73
94.6 or 105.4 1.77 2.62 2.79 2.91 3.00 3.13 3.22 3.29 3.36 3.45 3.66
94.4 or 105.6 1.73 2.58 2.74 2.86 2.95 3.08 3.16 3.23 3.30 3.39 3.60
94.2 or 105.8 1.70 2.53 2.69 2.81 2.90 3.02 3.11 3.17 3.24 3.33 3.53
94.0 or 106.0 1.67 2.48 2.64 2.76 2.84 2.96 3.05 3.11 3.18 3.26 3.46
93.8 or 106.2 1.64 2.43 2.59 2.70 2.79 2.91 2.99 3.05 3.12 3.20 3.40
93.6 or 106.4 1.61 2.39 2.54 2.65 2.73 2.85 2.93 2.99 3.06 3.14 3.33
93.4 or 106.6 1.58 2.34 2.49 2.60 2.68 2.79 2.87 2.93 3.00 3.08 3.27
93.2 or 106.8 1.54 2.29 2.44 2.55 2.62 2.74 2.82 2.87 2.94 3.02 3.20
93.0 or 107.0 1.51 2.24 2.39 2.49 2.57 2.68 2.76 2.82 2.88 2.95 3.14
92.8 or 107.2 1.48 2.20 2.34 2.44 2.52 2.63 2.70 2.76 2.82 2.89 3.07
92.6 or 107.4 1.45 2.15 2.29 2.39 2.46 2.57 2.64 2.70 2.76 2.83 3.00
92.4 or 107.6 1.42 2.10 2.24 2.34 2.41 2.51 2.58 2.64 2.70 2.77 2.94
92.2 or 107.8 1.38 2.06 2.19 2.28 2.36 2.46 2.53 2.58 2.64 2.71 2.87
92.0 or 108.0 1.35 2.01 2.14 2.23 2.30 2.40 2.47 2.52 2.58 2.64 2.81
91.8 or 108.2 1.32 1.96 2.09 2.18 2.25 2.34 2.41 2.46 2.52 2.58 2.74
91.6 or 108.4 1.29 1.91 2.04 2.13 2.19 2.29 2.35 2.40 2.46 2.52 2.67
91.4 or 108.6 1.26 1.87 1.99 2.07 2.14 2.23 2.29 2.34 2.39 2.46 2.61
91.2 or 108.8 1.23 1.82 1.94 2.02 2.08 2.17 2.24 2.28 2.34 2.39 2.54
91.0 or 109.0 1.19 1.77 1.89 1.97 2.03 2.12 2.18 2.22 2.27 2.33 2.48
90.8 or 109.2 1.16 1.73 1.84 1.92 1.98 2.06 2.12 2.16 2.21 2.27 2.41
90.6 or 109.4 1.13 1.68 1.79 1.86 1.92 2.00 2.06 2.11 2.15 2.21 2.34
90.4 or 109.6 1.10 1.63 1.74 1.81 1.87 1.95 2.00 2.04 2.09 2.15 2.28
90.2 or 109.8 1.07 1.58 1.69 1.76 1.81 1.89 1.95 1.99 2.03 2.08 2.21
90.0 or 110.0 1.03 1.54 1.64 1.71 1.76 1.84 1.89 1.93 1.97 2.02 2.15
right vertex just touches the acceptable parameter region. The 5.1.11 Using the Acceptance Limit Tables in This Practice—
size of the confidence region is determined by C, L, and r.
For a given sampling plan with L locations with r dosage units
tested at each location and a specified confidence level and
5.1.10 Computer programs have been developed for gener-
probability of passing the USP UDU test, the acceptance limit
ating acceptance limit tables, but these may not be available to
table provides a lower and upper range for the overall L × r
all practitioners. The International Society for Pharmaceutical
mean based on the pooled within location standard deviation
Engineering (ISPE) has made available information for com-
(SE) and the standard deviation of the L location means (not
mon tables for Sampling Plans 1 and 2, a SAS program, and
the between location variance component). The pooled loca-
other useful tools.
tion standard deviation is the square root of the average L
within location variances.
https://www.ispe.org/initiatives/blend-uniformity-content-uniformity/tools.
E2810 − 23
TABLE 5 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 90 %, LB = 95 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 3.21 4.47 4.71 4.88 5.00 5.16 5.27 5.35 5.44 5.54 5.76
99.8 or 100.2 3.17 4.42 4.67 4.83 4.95 5.12 5.23 5.32 5.41 5.51 5.74
99.6 or 100.4 3.13 4.37 4.61 4.78 4.90 5.07 5.19 5.27 5.36 5.47 5.71
99.4 or 100.6 3.09 4.32 4.56 4.72 4.85 5.02 5.14 5.22 5.32 5.42 5.67
99.2 or 100.8 3.06 4.27 4.50 4.67 4.79 4.96 5.08 5.17 5.26 5.37 5.63
99.0 or 101.0 3.02 4.21 4.45 4.61 4.73 4.91 5.02 5.11 5.21 5.32 5.58
98.8 or 101.2 2.98 4.16 4.39 4.55 4.67 4.84 4.96 5.05 5.15 5.26 5.52
98.6 or 101.4 2.94 4.11 4.34 4.50 4.62 4.78 4.90 4.99 5.08 5.20 5.46
98.4 or 101.6 2.90 4.05 4.28 4.44 4.55 4.72 4.84 4.93 5.02 5.13 5.40
98.2 or 101.8 2.86 4.00 4.22 4.38 4.50 4.66 4.78 4.86 4.96 5.07 5.33
98.0 or 102.0 2.82 3.95 4.17 4.32 4.44 4.60 4.71 4.80 4.89 5.00 5.26
97.8 or 102.2 2.78 3.89 4.11 4.26 4.37 4.54 4.65 4.73 4.82 4.93 5.19
97.6 or 102.4 2.75 3.84 4.05 4.20 4.31 4.47 4.58 4.67 4.76 4.86 5.12
97.4 or 102.6 2.71 3.79 4.00 4.14 4.25 4.41 4.52 4.60 4.69 4.80 5.05
97.2 or 102.8 2.67 3.73 3.94 4.08 4.19 4.35 4.45 4.53 4.63 4.73 4.98
97.0 or 103.0 2.63 3.68 3.88 4.03 4.13 4.29 4.39 4.47 4.56 4.66 4.91
96.8 or 103.2 2.59 3.62 3.82 3.97 4.07 4.22 4.33 4.40 4.49 4.59 4.84
96.6 or 103.4 2.55 3.57 3.77 3.91 4.01 4.16 4.26 4.34 4.42 4.52 4.76
96.4 or 103.6 2.51 3.52 3.71 3.85 3.95 4.10 4.20 4.27 4.36 4.46 4.69
96.2 or 103.8 2.47 3.46 3.65 3.79 3.89 4.03 4.13 4.21 4.29 4.39 4.62
96.0 or 104.0 2.43 3.41 3.60 3.73 3.83 3.97 4.07 4.14 4.22 4.32 4.55
95.8 or 104.2 2.40 3.35 3.54 3.67 3.77 3.91 4.00 4.08 4.16 4.25 4.48
95.6 or 104.4 2.36 3.30 3.48 3.61 3.71 3.84 3.94 4.01 4.09 4.18 4.40
95.4 or 104.6 2.32 3.24 3.43 3.55 3.65 3.78 3.87 3.95 4.02 4.11 4.33
95.2 or 104.8 2.28 3.19 3.37 3.49 3.58 3.72 3.81 3.88 3.96 4.04 4.26
95.0 or 105.0 2.24 3.13 3.31 3.43 3.52 3.66 3.75 3.81 3.89 3.97 4.19
94.8 or 105.2 2.20 3.08 3.25 3.37 3.46 3.59 3.68 3.75 3.82 3.91 4.11
94.6 or 105.4 2.16 3.03 3.20 3.31 3.40 3.53 3.61 3.68 3.75 3.84 4.04
94.4 or 105.6 2.12 2.97 3.14 3.25 3.34 3.47 3.55 3.61 3.69 3.77 3.97
94.2 or 105.8 2.09 2.92 3.08 3.20 3.28 3.40 3.49 3.55 3.62 3.70 3.90
94.0 or 106.0 2.05 2.86 3.02 3.13 3.22 3.34 3.42 3.48 3.55 3.63 3.82
93.8 or 106.2 2.01 2.81 2.97 3.08 3.16 3.27 3.36 3.42 3.48 3.56 3.75
93.6 or 106.4 1.97 2.75 2.91 3.02 3.10 3.21 3.29 3.35 3.42 3.49 3.68
93.4 or 106.6 1.93 2.70 2.85 2.96 3.04 3.15 3.23 3.28 3.35 3.42 3.61
93.2 or 106.8 1.89 2.65 2.79 2.90 2.97 3.08 3.16 3.22 3.28 3.36 3.53
93.0 or 107.0 1.85 2.59 2.74 2.84 2.91 3.02 3.10 3.15 3.21 3.29 3.46
92.8 or 107.2 1.81 2.54 2.68 2.78 2.85 2.96 3.03 3.09 3.15 3.22 3.39
92.6 or 107.4 1.77 2.48 2.62 2.72 2.79 2.89 2.96 3.02 3.08 3.15 3.32
92.4 or 107.6 1.74 2.43 2.56 2.66 2.73 2.83 2.90 2.95 3.01 3.08 3.24
92.2 or 107.8 1.70 2.37 2.51 2.60 2.67 2.77 2.84 2.89 2.94 3.01 3.17
92.0 or 108.0 1.66 2.32 2.45 2.54 2.61 2.70 2.77 2.82 2.88 2.94 3.10
91.8 or 108.2 1.62 2.26 2.39 2.48 2.55 2.64 2.71 2.75 2.81 2.87 3.02
91.6 or 108.4 1.58 2.21 2.33 2.42 2.48 2.58 2.64 2.69 2.74 2.80 2.95
91.4 or 108.6 1.54 2.16 2.27 2.36 2.42 2.51 2.58 2.62 2.67 2.73 2.88
91.2 or 108.8 1.50 2.10 2.22 2.30 2.36 2.45 2.51 2.55 2.61 2.66 2.81
91.0 or 109.0 1.46 2.05 2.16 2.24 2.30 2.39 2.44 2.49 2.54 2.60 2.73
90.8 or 109.2 1.42 1.99 2.10 2.18 2.24 2.32 2.38 2.42 2.47 2.53 2.66
90.6 or 109.4 1.38 1.94 2.04 2.12 2.18 2.26 2.31 2.36 2.40 2.46 2.59
90.4 or 109.6 1.34 1.88 1.99 2.06 2.12 2.19 2.25 2.29 2.34 2.39 2.51
90.2 or 109.8 1.31 1.83 1.93 2.00 2.05 2.13 2.18 2.22 2.27 2.32 2.44
90.0 or 110.0 1.27 1.77 1.87 1.94 1.99 2.07 2.12 2.16 2.20 2.25 2.37
5.1.11.1 Two examples of acceptance limit tables for Sam- tables were created using a confidence level of C = 90 % and
pling Plan 2 are provided below. These two sampling plans
a LB = 95 % and assume a target value of 100 %LC. Table 6
were selected based on material given in two papers (7, 8)
applies to a sampling plan with 20 locations with 3 results per
published by an ISPE Blend Uniformity/Content Uniformity
location (20 × 3) and Table 7 applies to 40 locations with 3
(BUCU) group that became active after the FDA withdrew a
results per location. The between location and within location
process validation guidance. ISPE has made available infor-
standard deviations start at 0.1 %LC and increment by
mation regarding various activities of the group. Table 6 and
0.1 %LC up to the maximum possible range on the overall
Table 7 contain information available from the ISPE. Both
mean.
https://ispe.org/search?keys=BUCU.
https://ispe.org/initiatives/blend-uniformity-content-uniformity/tools/sampling.
E2810 − 23
TABLE 6 Sampling Plan 2 (20 Locations: 3 Results at Each Location) Acceptance Limits on Overall Mean (%LC) for
T = 100 %LC, C = 90 %, LB = 95 %LC
Standard Deviation of Location Means (%LC)
SE
0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8
(%LC)
LL UL LL UL LL UL LL UL LL UL LL UL LL UL LL UL
0.1 84.0 116.0 84.4 115.6 84.8 115.2 85.2 114.8 85.6 114.4 86.0 114.0 86.4 113.6 86.8 113.2
0.2 84.2 115.8 84.5 115.5 84.8 115.2 85.2 114.8 85.6 114.4 86.0 114.0 86.4 113.6 86.8 113.2
0.3 84.4 115.6 84.6 115.4 84.9 115.1 85.3 114.7 85.7 114.3 86.1 113.9 86.5 113.5 86.8 113.2
0.4 84.7 115.3 84.8 115.2 85.1 114.9 85.4 114.6 85.8 114.2 86.1 113.9 86.5 113.5 86.9 113.1
0.5 84.9 115.1 85.0 115.0 85.3 114.7 85.5 114.5 85.9 114.1 86.2 113.8 86.6 113.4 87.0 113.0
0.6 85.1 114.9 85.3 114.7 85.5 114.5 85.7 114.3 86.0 114.0 86.3 113.7 86.7 113.3 87.1 112.9
0.7 85.4 114.6 85.5 114.5 85.7 114.3 85.9 114.1 86.2 113.8 86.5 113.5 86.8 113.2 87.2 112.8
0.8 85.6 114.4 85.7 114.3 85.9 114.1 86.1 113.9 86.3 113.7 86.6 113.4 86.9 113.1 87.3 112.7
0.9 85.9 114.1 86.0 114.0 86.1 113.9 86.3 113.7 86.5 113.5 86.8 113.2 87.1 112.9 87.4 112.6
1.0 86.1 113.9 86.2 113.8 86.4 113.6 86.5 113.5 86.7 113.3 87.0 113.0 87.2 112.8 87.5 112.5
1.1 86.4 113.6 86.5 113.5 86.6 113.4 86.8 113.2 86.9 113.1 87.2 112.8 87.4 112.6 87.7 112.3
1.2 86.6 113.4 86.7 113.3 86.8 113.2 87.0 113.0 87.2 112.8 87.4 112.6 87.6 112.4 87.9 112.1
1.3 86.9 113.1 87.0 113.0 87.1 112.9 87.2 112.8 87.4 112.6 87.6 112.4 87.8 112.2 88.1 111.9
1.4 87.1 112.9 87.2 112.8 87.3 112.7 87.5 112.5 87.6 112.4 87.8 112.2 88.0 112.0 88.2 111.8
1.5 87.4 112.6 87.5 112.5 87.6 112.4 87.7 112.3 87.9 112.1 88.0 112.0 88.2 111.8 88.4 111.6
1.6 87.6 112.4 87.7 112.3 87.8 112.2 87.9 112.1 88.1 111.9 88.2 111.8 88.4 111.6 88.6 111.4
1.7 87.9 112.1 88.0 112.0 88.1 111.9 88.2 111.8 88.3 111.7 88.5 111.5 88.7 111.3 88.9 111.1
1.8 88.1 111.9 88.2 111.8 88.3 111.7 88.4 111.6 88.6 111.4 88.7 111.3 88.9 111.1 89.1 110.9
1.9 88.4 111.6 88.5 111.5 88.6 111.4 88.7 111.3 88.8 111.2 88.9 111.1 89.1 110.9 89.3 110.7
2.0 88.7 111.3 88.7 111.3 88.8 111.2 88.9 111.1 89.0 111.0 89.2 110.8 89.3 110.7 89.5 110.5
2.1 88.9 111.1 89.0 111.0 89.1 110.9 89.2 110.8 89.3 110.7 89.4 110.6 89.6 110.4 89.7 110.3
2.2 89.2 110.8 89.2 110.8 89.3 110.7 89.4 110.6 89.5 110.5 89.7 110.3 89.8 110.2 90.0 110.0
2.3 89.4 110.6 89.5 110.5 89.6 110.4 89.7 110.3 89.8 110.2 89.9 110.1 90.0 110.0 90.2 109.8
2.4 89.7 110.3 89.7 110.3 89.8 110.2 89.9 110.1 90.0 110.0 90.1 109.9 90.3 109.7 90.4 109.6
2.5 89.9 110.1 90.0 110.0 90.1 109.9 90.2 109.8 90.3 109.7 90.4 109.6 90.5 109.5 90.7 109.3
2.6 90.2 109.8 90.2 109.8 90.3 109.7 90.4 109.6 90.5 109.5 90.6 109.4 90.8 109.2 90.9 109.1
2.7 90.4 109.6 90.5 109.5 90.6 109.4 90.7 109.3 90.8 109.2 90.9 109.1 91.0 109.0 91.1 108.9
2.8 90.7 109.3 90.7 109.3 90.8 109.2 90.9 109.1 91.0 109.0 91.1 108.9 91.2 108.8 91.4 108.6
2.9 90.9 109.1 91.0 109.0 91.1 108.9 91.2 108.8 91.3 108.7 91.4 108.6 91.5 108.5 91.6 108.4
3.0 91.2 108.8 91.2 108.8 91.3 108.7 91.4 108.6 91.5 108.5 91.6 108.4 91.7 108.3 91.9 108.1
3.1 91.4 108.6 91.5 108.5 91.6 108.4 91.7 108.3 91.8 108.2 91.9 108.1 92.0 108.0 92.1 107.9
3.2 91.7 108.3 91.7 108.3 91.8 108.2 91.9 108.1 92.0 108.0 92.1 107.9 92.2 107.8 92.3 107.7
3.3 91.9 108.1 92.0 108.0 92.1 107.9 92.2 107.8 92.3 107.7 92.4 107.6 92.5 107.5 92.6 107.4
3.4 92.2 107.8 92.2 107.8 92.3 107.7 92.4 107.6 92.5 107.5 92.6 107.4 92.7 107.3 92.8 107.2
3.5 92.4 107.6 92.5 107.5 92.6 107.4 92.7 107.3 92.8 107.2 92.9 107.1 93.0 107.0 93.1 106.9
3.6 92.7 107.3 92.7 107.3 92.8 107.2 92.9 107.1 93.0 107.0 93.1 106.9 93.2 106.8 93.3 106.7
3.7 92.9 107.1 93.0 107.0 93.1 106.9 93.2 106.8 93.3 106.7 93.4 106.6 93.5 106.5 93.6 106.4
3.8 93.2 106.8 93.3 106.7 93.3 106.7 93.4 106.6 93.5 106.5 93.6 106.4 93.7 106.3 93.8 106.2
3.9 93.4 106.6 93.5 106.5 93.6 106.4 93.7 106.3 93.8 106.2 93.8 106.2 94.0 106.0 94.1 105.9
4.0 93.7 106.3 93.8 106.2 93.8 106.2 93.9 106.1 94.0 106.0 94.1 105.9 94.2 105.8 94.3 105.7
4.1 93.9 106.1 94.0 106.0 94.1 105.9 94.2 105.8 94.3 105.7 94.3 105.7 94.5 105.5 94.6 105.4
4.2 94.2 105.8 94.3 105.7 94.3 105.7 94.4 105.6 94.5 105.5 94.6 105.4 94.7 105.3 94.8 105.2
4.3 94.4 105.6 94.5 105.5 94.6 105.4 94.7 105.3 94.8 105.2 94.9 105.1 95.0 105.0 95.1 104.9
4.4 94.7 105.3 94.8 105.2 94.8 105.2 94.9 105.1 95.0 105.0 95.1 104.9 95.2 104.8 95.3 104.7
4.5 95.0 105.0 95.0 105.0 95.1 104.9 95.2 104.8 95.3 104.7 95.4 104.6 95.5 104.5 95.6 104.4
4.6 95.2 104.8 95.3 104.7 95.4 104.6 95.4 104.6 95.5 104.5 95.6 104.4 95.7 104.3 95.8 104.2
4.7 95.5 104.5 95.5 104.5 95.6 104.4 95.7 104.3 95.8 104.2 95.9 104.1 96.0 104.0 96.1 103.9
4.8 95.7 104.3 95.8 104.2 95.9 104.1 95.9 104.1 96.0 104.0 96.1 103.9 96.2 103.8 96.3 103.7
4.9 96.0 104.0 96.0 104.0 96.1 103.9 96.2 103.8 96.3 103.7 96.4 103.6 96.5 103.5 96.6 103.4
5.0 96.2 103.8 96.3 103.7 96.4 103.6 96.4 103.6 96.5 103.5 96.6 103.4 96.7 103.3 96.8 103.2
5.1 96.5 103.5 96.6 103.4 96.6 103.4 96.7 103.3 96.8 103.2 96.9 103.1 97.0 103.0 97.1 102.9
5.2 96.7 103.3 96.8 103.2 96.9 103.1 97.0 103.0 97.0 103.0 97.1 102.9 97.2 102.8 97.3 102.7
5.3 97.0 103.0 97.1 102.9 97.1 102.9 97.2 102.8 97.3 102.7 97.4 102.6 97.5 102.5 97.6 102.4
5.4 97.3 102.7 97.3 102.7 97.4 102.6 97.5 102.5 97.6 102.4 97.6 102.4 97.7 102.3 97.8 102.2
5.5 97.5 102.5 97.6 102.4 97.7 102.3 97.7 102.3 97.8 102.2 97.9 102.1 98.0 102.0 98.1 101.9
5.6 97.8 102.2 97.8 102.2 97.9 102.1 98.0 102.0 98.1 101.9 98.2 101.8 98.3 101.7 98.4 101.6
5.7 98.1 101.9 98.1 101.9 98.2 101.8 98.3 101.7 98.4 101.6 98.4 101.6 98.5 101.5 98.6 101.4
5.8 98.4 101.6 98.4 101.6 98.5 101.5 98.6 101.4 98.7 101.3 98.7 101.3 98.8 101.2 98.9 101.1
5.9 98.7 101.3 98.8 101.2 98.8 101.2 98.9 101.1 99.0 101.0 99.1 100.9 99.2 100.8 99.3 100.7
6.0 99.1 100.9 99.2 100.8 99.3 100.7 99.4 100.6 99.5 100.5 99.6 100.4 99.7 100.3 99.9 100.1
E2810 − 23
TABLE 6 Continued
Standard Deviation of Location Means (%LC)
SE
0.9 1.0 1.1 1.2 1.3 1.4 1.5 1.6
(%LC)
LL UL LL UL LL UL LL UL LL UL LL UL LL UL LL UL
0.1 87.2 112.8 87.6 112.4 88.0 112.0 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 90.0 110.0
0.2 87.2 112.8 87.6 112.4 88.0 112.0 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 90.0 110.0
0.3 87.2 112.8 87.6 112.4 88.0 112.0 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 90.0 110.0
0.4 87.3 112.7 87.7 112.3 88.1 111.9 88.5 111.5 88.9 111.1 89.3 110.7 89.7 110.3 90.1 109.9
0.5 87.4 112.6 87.7 112.3 88.1 111.9 88.5 111.5 88.9 111.1 89.3 110.7 89.7 110.3 90.1 109.9
0.6 87.4 112.6 87.8 112.2 88.2 111.8 88.6 111.4 89.0 111.0 89.4 110.6 89.7 110.3 90.1 109.9
0.7 87.5 112.5 87.9 112.1 88.3 111.7 88.6 111.4 89.0 111.0 89.4 110.6 89.8 110.2 90.2 109.8
0.8 87.6 112.4 88.0 112.0 88.3 111.7 88.7 111.3 89.1 110.9 89.5 110.5 89.9 110.1 90.2 109.8
0.9 87.7 112.3 88.1 111.9 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 89.9 110.1 90.3 109.7
1.0 87.9 112.1 88.2 111.8 88.5 111.5 88.9 111.1 89.3 110.7 89.6 110.4 90.0 110.0 90.4 109.6
1.1 88.0 112.0 88.3 111.7 88.7 111.3 89.0 111.0 89.4 110.6 89.7 110.3 90.1 109.9 90.5 109.5
1.2 88.2 111.8 88.5 111.5 88.8 111.2 89.1 110.9 89.5 110.5 89.8 110.2 90.2 109.8 90.6 109.4
1.3 88.3 111.7 88.6 111.4 88.9 111.1 89.3 110.7 89.6 110.4 89.9 110.1 90.3 109.7 90.7 109.3
1.4 88.5 111.5 88.8 111.2 89.1 110.9 89.4 110.6 89.7 110.3 90.1 109.9 90.4 109.6 90.8 109.2
1.5 88.7 111.3 89.0 111.0 89.2 110.8 89.5 110.5 89.9 110.1 90.2 109.8 90.5 109.5 90.9 109.1
1.6 88.9 111.1 89.1 110.9 89.4 110.6 89.7 110.3 90.0 110.0 90.3 109.7 90.6 109.4 91.0 109.0
1.7 89.1 110.9 89.3 110.7 89.6 110.4 89.9 110.1 90.2 109.8 90.5 109.5 90.8 109.2 91.1 108.9
1.8 89.3 110.7 89.5 110.5 89.8 110.2 90.0 110.0 90.3 109.7 90.6 109.4 90.9 109.1 91.2 108.8
1.9 89.5 110.5 89.7 110.3 90.0 110.0 90.2 109.8 90.5 109.5 90.8 109.2 91.1 108.9 91.4 108.6
2.0 89.7 110.3 89.9 110.1 90.1 109.9 90.4 109.6 90.7 109.3 90.9 109.1 91.2 108.8 91.5 108.5
2.1 89.9 110.1 90.1 109.9 90.3 109.7 90.6 109.4 90.8 109.2 91.1 108.9 91.4 108.6 91.7 108.3
2.2 90.1 109.9 90.3 109.7 90.6 109.4 90.8 109.2 91.0 109.0 91.3 108.7 91.6 108.4 91.9 108.1
2.3 90.4 109.6 90.6 109.4 90.8 109.2 91.0 109.0 91.2 108.8 91.5 108.5 91.7 108.3 92.0 108.0
2.4 90.6 109.4 90.8 109.2 91.0 109.0 91.2 108.8 91.4 108.6 91.7 108.3 91.9 108.1 92.2 107.8
2.5 90.8 109.2 91.0 109.0 91.2 108.8 91.4 108.6 91.6 108.4 91.9 108.1 92.1 107.9 92.4 107.6
2.6 91.1 108.9 91.2 108.8 91.4 108.6 91.6 108.4 91.8 108.2 92.1 107.9 92.3 107.7 92.6 107.4
2.7 91.3 108.7 91.5 108.5 91.6 108.4 91.8 108.2 92.0 108.0 92.3 107.7 92.5 107.5 92.7 107.3
2.8 91.5 108.5 91.7 108.3 91.9 108.1 92.0 108.0 92.2 107.8 92.5 107.5 92.7 107.3 92.9 107.1
2.9 91.8 108.2 91.9 108.1 92.1 107.9 92.3 107.7 92.5 107.5 92.7 107.3 92.9 107.1 93.1 106.9
3.0 92.0 108.0 92.2 107.8 92.3 107.7 92.5 107.5 92.7 107.3 92.9 107.1 93.1 106.9 93.3 106.7
3.1 92.2 107.8 92.4 107.6 92.5 107.5 92.7 107.3 92.9 107.1 93.1 106.9 93.3 106.7 93.5 106.5
3.2 92.5 107.5 92.6 107.4 92.8 107.2 92.9 107.1 93.1 106.9 93.3 106.7 93.5 106.5 93.7 106.3
3.3 92.7 107.3 92.9 107.1 93.0 107.0 93.2 106.8 93.4 106.6 93.5 106.5 93.7 106.3 93.9 106.1
3.4 93.0 107.0 93.1 106.9 93.3 106.7 93.4 106.6 93.6 106.4 93.8 106.2 94.0 106.0 94.2 105.8
3.5 93.2 106.8 93.3 106.7 93.5 106.5 93.6 106.4 93.8 106.2 94.0 106.0 94.2 105.8 94.4 105.6
3.6 93.4 106.6 93.6 106.4 93.7 106.3 93.9 106.1 94.0 106.0 94.2 105.8 94.4 105.6 94.6 105.4
3.7 93.7 106.3 93.8 106.2 94.0 106.0 94.1 105.9 94.3 105.7 94.4 105.6 94.6 105.4 94.8 105.2
3.8 93.9 106.1 94.1 105.9 94.2 105.8 94.4 105.6 94.5 105.5 94.7 105.3 94.8 105.2 95.0 105.0
3.9 94.2 105.8 94.3 105.7 94.4 105.6
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2810 − 19 E2810 − 23
Standard Practice for
Demonstrating Capability to Comply with the Test for
Uniformity of Dosage Units
This standard is issued under the fixed designation E2810; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice provides a general procedure for evaluating the capability to comply with the Uniformity of Dosage Units (UDU)
test. This test is given in General Chapter <905> Uniformity of Dosage Units of the USP, in 2.9.40 Uniformity of Dosage Units
of the Ph. Eur., and in 6.02 Uniformity of Dosage Units of the JP, and these versions are virtually interchangeable. For this
multiple-stage test, the procedure computes a lower bound on the probability of passing the UDU test, based on statistical estimates
made at a prescribed confidence level from a sample of dosage units.
1.2 This methodology can be used to generate an acceptance limit table, which defines a set of sample means and standard
deviations that assures passing the UDU test for a prescribed lower probability bound, confidence level, and sample size.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E2363 Terminology Relating to Manufacturing of Pharmaceutical and Biopharmaceutical Products in the Pharmaceutical and
Biopharmaceutical Industry
This practice is under the jurisdiction of ASTM Committee E55 on Manufacture of Pharmaceutical and Biopharmaceutical Products and is the direct responsibility of
Subcommittee E55.14 on Measurement Systems and Analysis.
Current edition approved April 1, 2019Dec. 1, 2023. Published August 2019December 2023. Originally approved in 2011. Last previous edition approved in 20172019
as E2810 – 11 (2017). DOI: 10.1520/E2810-19.19. DOI: 10.1520/E2810-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2810 − 23
E2709 Practice for Demonstrating Capability to Comply with an Acceptance Procedure
2.2 Other Documents:
JP Japanese Pharmacopoeia
Ph. Eur. European Pharmacopoeia
USP United States Pharmacopeia
3. Terminology
3.1 Definitions—See Terminology E2363 for a more extensive listing of terms in ASTM Committee E55 standards.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 acceptable parameter region, n—the set of values of parameters characterizing the distribution of test results for which the
probability of passing the lot acceptance procedure is greater than a prescribed lower bound.
3.2.2 acceptance limit, n—the boundary of the acceptance region, for example, the maximum sample standard deviation for a
given sample mean or minimum and maximum sample mean for given standard deviations.
3.2.2.1 Discussion—
The coefficient of variation (relative standard deviation) may be substituted for the standard deviation where applicable.
3.2.3 acceptance region, n—the set of values of parameter estimates (that is, sample mean and standard deviation) where
confidence limits attain a prescribed lower bound on the probability of passing a lot acceptance procedure.
3.2.4 confidence level, C, n—the prescribed overall level for calculating the uncertainty region of the parameters from the sample
estimates.
3.2.4.1 Discussion—
The preset confidence level is stated as a percentage, for example, 100 (1 – α) = 95 %, where α is a risk that is allocated to the two
parameters being estimated.
3.2.5 lower probability bound, LB, n—the nominal probability of passing the UDU test for a given set of parameter estimates.
3.2.6 multiple-stage acceptance procedure, n—a procedure that involves more than one stage of sampling and testing a given
quality characteristic with one or more acceptance criteria per stage.
3.2.7 representative sample, n—a sample that consists of a number of units that are drawn based on rational criteria such as
random, stratified, or systematic sampling and intended to assure that the sample accurately portrays the material being sampled.
3.2.8 sampling plan, n—scheme for selecting dosage units from locations within a batch for testing purposes.
3.2.8.1 Discussion—
In this standard, either a single dosage unit is selected from each batch location (called Sampling Plan 1) or an equal but greater
than one dosage unit is selected from each batch location (called Sampling Plan 2).
3.2.9 uniformity of dosage units, UDU, n—the degree of uniformity in the amount of the drug substance among dosage units.
3.2.9.1 Discussion—
The requirements of the UDU test apply to each drug substance in dosage units containing one or more drug substances, unless
otherwise specified. The uniformity improves as the variability decreases.
4. Significance and Use
4.1 The methodology was originally developed (1-4) for use in drug content uniformity and dissolution but has general
Available from the Pharmaceuticals and Medical Devices Agency (PMDA), Shin-Kasumigaseki Building, 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan,
https://www.pmda.go.jp.
Available from the European Directorate for the Quality of Medicines and Health Care (EDQM), Council of Europe, 7 allée Kastner, CS 30026, F-67081 Strasbourg,
France, http://www.edqm.eu.
Available from U.S. Pharmacopeial Convention (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852-1790, http://www.usp.org.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
E2810 − 23
application to any multistage test with multiple acceptance criteria. Practice E2709 summarizes the statistical aspects of this
methodology. This practice applies the general methodology of Practice E2709 specifically to the UDU test.
4.1.1 While other methods can be used to estimate the probability of passing the UDU test, they are outside the scope of this
practice.
4.2 The UDU test procedure describes a two-stage sampling test, where at each stage one can pass or continue testing, and the
decision to fail is deferred until the second stage. At each stage there are acceptance criteria on the test results as outlined in Table
1.
4.3 The UDU test is a market standard. The USP General Notices include the following statement about compendial standards.
“The similarity to statistical procedures may seem to suggest an intent to make inference to some larger group of units, but in all
cases, statements about whether the compendial standard is met apply only to the units tested.” Therefore, the UDU procedure is
not intended for inspecting uniformity of finished product for lot/batch release or as a lot inspection procedure.
4.3.1 The UDU test defines a product requirement to be met at release and throughout the shelf-life of the product.
4.3.2 Passing the UDU test once does not provide statistical assurance that a batch of drug product meets specified statistical
quality control criteria.
4.4 This practice provides a practical specification that may be applied when uniformity of dosage units is required. An acceptance
region for the mean and standard deviation of a set of test results from the lot is defined such that, at a prescribed confidence level,
the probability that a future sample from the lot will pass the UDU test is greater than or equal to a prespecified lower probability
bound. Having test results fall in the acceptance region provides assurance that a sample would pass the UDU test with at least
the specified lower bound probability. This procedure does not account for any decrease in potency during the shelf life, which
could affect the ability to meet the UDU test requirements.
4.5 This practice can be used as an element for process demonstration or validation, continuous process verification, in-process
testing, or lot release (acceptance). As the circumstances and available information vary in these different application areas, this
TABLE 1 Uniformity of Dosage Units Test Procedure
NOTE 1—All measurements of dosage units and criteria values are in
percentage label claim (%LC). At each stage calculate the sample average,
X—, and the sample standard deviation, s.
Number
Stage Pass Stage If:
Tested
S 10 |M – X¯| + 2.4 s ≤ 15.0, where M is de-
fined below.
S 20 (1) |M – X¯| + 2.0 s ≤ 15.0, using all 30
results (S + S ).
1 2
(2) No dosage unit is outside the
maximum allowed range of 0.75 * M
to 1.25 * M.
M is defined as follows:
If T is less than or equal to 101.5 %LC, and
(1) If X¯ is less than 98.5 %LC, then
M = 98.5 %LC.
(2) If X¯ is between 98.5 and
101.5 %LC, then M = X¯.
(3) If X¯ is greater than 101.5 %LC, then
M = 101.5 %LC.
If T is greater than 101.5 %LC, and
(1) If X¯ is less than 98.5 %LC, then
M = 98.5 %LC.
(2) If X¯ is between 98.5 and T, then
M = X¯.
(3) If X¯ is greater than T, then M = T.
T is the target content per dosage unit at the time of manufacture, expressed as
%LC. Unless otherwise specified in the individual monograph, T is 100.0 %LC.
E2810 − 23
practice does not prescribe a specific target, sample size, lower probability bound, or confidence level. These must be prospectively
selected by the user and may be different from those used in the acceptance limit tables provided in this practice.
5. Procedure
5.1 Generating The Acceptance Limit Table:
5.1.1 The general procedure that generates the acceptance limit tables is described in Practice E2709 and the specific procedure
for application to the UDU test is described in the literature (4). A simplified description on the construction and use of these tables
is given in this section. A computer program is required to generate the tables given a target T as a percentage of label claim (LC),
a lower probability bound LB, a confidence level C, and a sample size n.
5.1.2 The first step is to determine the acceptable parameter region. On a two-dimensional content space consisting of the true
mean (μ) on the horizontal axis and standard deviation (σ) on the vertical axis the upper 95 % LB boundary of this region is defined
by a contour, a curve that is concave downward and depicted by the solid curve in Fig. 1. The simulated 95 % boundary for the
actual probability of passing the USP UDU test is defined by the dashed contour that shows the effect of generating a lower bound.
The LB contour is determined by the LB probability (95 %) and the target under the assumption that the dosage unit content is
normally distributed. The acceptable parameter region is the set of points on or below the contour. Any (μ, σ) pair in the acceptable
region would pass the UDU test with a probability of at least the LB.
5.1.3 The second step is to generate the acceptance limit curve that defines the acceptance region.
SAMPLING PLAN 1
5.1.4 As described in Practice E2709, the sample mean (X¯) and sample standard deviation (s) estimate the population parameters
μ and σ within C % confidence limits as chosen by the user. The joint confidence region for μ and σ (5) has the shape of an inverted
triangle around a (X¯,s) pair as depicted in Fig. 2 with the lowest vertex at (X¯, 0).
5.1.4.1 To create an acceptance region, a value of X¯ is selected starting with s = 0, then the confidence region is expanded by
increasing s until one of the upper vertices just touches the acceptable parameter region. The size of the confidence region is
determined by C and n. This value of s defines a point on the acceptance limit curve at (X¯,s). Additional selections of X¯ then
generate the acceptance limit curve, as depicted as dotted lines in Fig. 2. Acceptance limit curves are shown for n = 10 and n = 100,
illustrating that the acceptance limits approach the acceptable parameter region with increasing sample size.
5.1.5 Computer programs have been developed for generating acceptance limit tables, but these may not be available to all
practitioners. This practice contains four acceptance limit tables for many practical use situations.
FIG. 1 Example of 95 % Lower Bound and Simulated Probabilities of Passing USP UDU <905>
E2810 − 23
NOTE 1—All points below the lower bound contour have higher than a 95 % chance of passing UDU test if mean and standard deviation are known.
All points below the acceptance region contours pass the associated acceptance limit table for n = 100 and n = 10.
ULS is the upper confidence limit for σ.
Z is a standard normal critical value.
FIG. 2 Example of Simultaneous Confidence Interval with 95 % Lower Bound and Acceptance Regions
5.1.6 Using the Acceptance Limit Tables in This Practice—Various acceptance limit tables are given in Tables 2-5. In each table
acceptance limits on the standard deviation are given for means ranging 90–110 % 90 % to 110 % of LC in increments of 0.2 %LC
for sample sizes ranging from n = 10 to n = 500. In all tables, the target is set at T = 100 %LC, so the acceptance limits for standard
deviations are symmetrical around 100 %LC. This target is also required for interchangeability across the ICH regions (6). At the
confidence level of C = 95 % used often in the regulatory arena, three levels of the probability lower bound are provided:
LB = 90 % (Table 2), LB = 95 % (Table 3) and LB = 99 % (Table 4). These provide 90 %, 95 %, and 99 % coverage, respectively,
of the population of dosage units under consideration. The usual coverage is 95 %.
5.1.6.1 Table 5 is provided at C = 90 % and LB = 95 % for comparison with Table 3 to demonstrate the effect of a lower
confidence level.
NOTE 1—Tables can also be generated for other choices for ranges of means, such as 85.1 to 114.9 %LC, or for other sample sizes.
5.1.7 A representative sample of n dosage units is taken from the batch and the active content is measured on each unit, taken as
a random sample or as a systematic sample (selecting and testing one unit at equally spaced time intervals of manufacture across
the batch). Other sampling plans are outside the scope of this practice.
5.1.8 The sample average (X¯) and sample standard deviation (s) are calculated. Enter the table row with the sample average and
the table column with the sample size to find the acceptance limit on the standard deviation. If the sample standard deviation is
at or below the acceptance limit then it can be asserted at the C % confidence level that the batch will have an LB % chance of
passing the UDU test. Some interpolation may be required for sample sizes and sample means not listed in the table.
E2810 − 23
TABLE 2 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 95 %, LB = 90 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 2.91 4.36 4.65 4.84 4.99 5.19 5.33 5.43 5.54 5.66 5.93
99.8 or 100.2 2.88 4.31 4.59 4.79 4.94 5.14 5.28 5.38 5.50 5.62 5.91
99.6 or 100.4 2.84 4.26 4.54 4.74 4.89 5.09 5.24 5.34 5.45 5.58 5.88
99.4 or 100.6 2.81 4.21 4.49 4.69 4.83 5.04 5.18 5.29 5.40 5.53 5.84
99.2 or 100.8 2.77 4.16 4.43 4.63 4.77 4.98 5.13 5.23 5.35 5.48 5.79
99.0 or 101.0 2.74 4.10 4.38 4.57 4.72 4.92 5.07 5.17 5.29 5.43 5.74
98.8 or 101.2 2.70 4.05 4.32 4.52 4.66 4.86 5.01 5.11 5.23 5.37 5.69
98.6 or 101.4 2.67 4.00 4.27 4.46 4.60 4.80 4.94 5.05 5.17 5.30 5.63
98.4 or 101.6 2.63 3.95 4.21 4.40 4.54 4.74 4.88 4.99 5.10 5.24 5.56
98.2 or 101.8 2.60 3.89 4.16 4.34 4.48 4.68 4.82 4.92 5.04 5.17 5.49
98.0 or 102.0 2.56 3.84 4.10 4.28 4.42 4.62 4.75 4.86 4.97 5.10 5.43
97.8 or 102.2 2.53 3.79 4.05 4.22 4.36 4.55 4.69 4.79 4.90 5.03 5.35
97.6 or 102.4 2.49 3.74 3.99 4.17 4.30 4.49 4.62 4.72 4.84 4.97 5.28
97.4 or 102.6 2.46 3.68 3.93 4.11 4.24 4.43 4.56 4.66 4.77 4.90 5.21
97.2 or 102.8 2.42 3.63 3.88 4.05 4.18 4.36 4.50 4.59 4.70 4.83 5.13
97.0 or 103.0 2.39 3.58 3.82 3.99 4.12 4.30 4.43 4.53 4.63 4.76 5.06
96.8 or 103.2 2.35 3.53 3.77 3.93 4.06 4.24 4.37 4.46 4.56 4.69 4.99
96.6 or 103.4 2.32 3.48 3.71 3.87 4.00 4.18 4.30 4.39 4.50 4.62 4.91
96.4 or 103.6 2.28 3.42 3.65 3.81 3.94 4.11 4.23 4.33 4.43 4.55 4.84
96.2 or 103.8 2.24 3.37 3.60 3.76 3.88 4.05 4.17 4.26 4.36 4.48 4.77
96.0 or 104.0 2.21 3.32 3.54 3.70 3.82 3.99 4.10 4.19 4.29 4.41 4.69
95.8 or 104.2 2.17 3.26 3.48 3.64 3.76 3.92 4.04 4.13 4.23 4.34 4.62
95.6 or 104.4 2.14 3.21 3.43 3.58 3.70 3.86 3.98 4.06 4.16 4.27 4.54
95.4 or 104.6 2.10 3.16 3.37 3.52 3.63 3.80 3.91 3.99 4.09 4.20 4.47
95.2 or 104.8 2.07 3.11 3.31 3.46 3.57 3.73 3.84 3.93 4.02 4.13 4.39
95.0 or 105.0 2.03 3.05 3.26 3.40 3.51 3.67 3.78 3.86 3.95 4.06 4.32
94.8 or 105.2 2.00 3.00 3.20 3.35 3.45 3.61 3.71 3.79 3.88 3.99 4.24
94.6 or 105.4 1.96 2.95 3.15 3.29 3.39 3.54 3.65 3.73 3.82 3.92 4.17
94.4 or 105.6 1.93 2.89 3.09 3.23 3.33 3.48 3.58 3.66 3.75 3.85 4.10
94.2 or 105.8 1.89 2.84 3.03 3.17 3.27 3.42 3.52 3.59 3.68 3.78 4.02
94.0 or 106.0 1.86 2.79 2.98 3.11 3.21 3.35 3.45 3.53 3.61 3.71 3.95
93.8 or 106.2 1.82 2.74 2.92 3.05 3.15 3.29 3.39 3.46 3.54 3.64 3.87
93.6 or 106.4 1.79 2.68 2.86 2.99 3.09 3.23 3.32 3.39 3.47 3.57 3.80
93.4 or 106.6 1.75 2.63 2.81 2.93 3.03 3.16 3.25 3.33 3.40 3.50 3.72
93.2 or 106.8 1.72 2.58 2.75 2.87 2.97 3.10 3.19 3.26 3.34 3.43 3.65
93.0 or 107.0 1.68 2.52 2.69 2.81 2.90 3.03 3.12 3.19 3.27 3.36 3.57
92.8 or 107.2 1.65 2.47 2.64 2.75 2.84 2.97 3.06 3.13 3.20 3.28 3.50
92.6 or 107.4 1.61 2.42 2.58 2.70 2.78 2.91 2.99 3.06 3.13 3.21 3.42
92.4 or 107.6 1.57 2.36 2.52 2.64 2.72 2.84 2.93 2.99 3.06 3.14 3.35
92.2 or 107.8 1.54 2.31 2.47 2.58 2.66 2.78 2.86 2.92 2.99 3.07 3.27
92.0 or 108.0 1.50 2.26 2.41 2.52 2.60 2.71 2.80 2.86 2.92 3.00 3.20
91.8 or 108.2 1.47 2.20 2.35 2.46 2.54 2.65 2.73 2.79 2.86 2.93 3.12
91.6 or 108.4 1.43 2.15 2.30 2.40 2.48 2.59 2.66 2.72 2.79 2.86 3.05
91.4 or 108.6 1.40 2.10 2.24 2.34 2.42 2.52 2.60 2.65 2.72 2.79 2.97
91.2 or 108.8 1.36 2.04 2.18 2.28 2.35 2.46 2.53 2.59 2.65 2.72 2.89
91.0 or 109.0 1.33 1.99 2.13 2.22 2.29 2.39 2.47 2.52 2.58 2.65 2.82
90.8 or 109.2 1.29 1.94 2.07 2.16 2.23 2.33 2.40 2.45 2.51 2.58 2.74
90.6 or 109.4 1.26 1.89 2.01 2.10 2.17 2.27 2.33 2.39 2.44 2.51 2.67
90.4 or 109.6 1.22 1.83 1.96 2.04 2.11 2.20 2.27 2.32 2.37 2.44 2.59
90.2 or 109.8 1.19 1.78 1.90 1.98 2.05 2.14 2.20 2.25 2.30 2.37 2.52
90.0 or 110.0 1.15 1.73 1.84 1.93 1.99 2.08 2.14 2.18 2.24 2.30 2.44
5.1.8.1 Example 1—A sample of n = 60 dosage units provided dosage content statistics of X¯ = 98.6 %LC and s = 3.91 %LC. Since
s was below the upper limit of 4.41 %LC (using Table 3), we can state with 95 % confidence there is at least a 95 % probability
that a future sample taken from the batch will meet the UDU test.
5.1.8.2 Example 2—A sample of n = 70 dosage units provided dosage content statistics of X¯ = 97.8 %LC and s = 4.29 %LC. Since
n = was not provided in the Table 3, we must interpolate between entry n = 60 with the upper limit 4.18 %LC and entry n = 80
with the upper limit 4.36 %LC as follows:
acceptance limit 5 4.181 4.36 2 4.18 80 2 70 / 80 2 60
@~ !~ ! ~ !#
54.181@ 0.18 10 / 20 # 5 4.27 %LC
~ !~ ! ~ !
Acceptancelimit5 4.181@~4.36 2 4.18! ~70 2 60! ⁄ ~80 2 60!# 5 4.181@~0.18!~10!⁄ ~20!# 5 4.27%LC
Since s = 4.29 %LC was above the upper limit of 4.27 %LC, we cannot state with 95 % confidence there is at least a 95 %
probability that a future sample taken from the batch will meet the UDU test.
E2810 − 23
TABLE 3 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 95 %, LB = 95 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 2.81 4.18 4.46 4.64 4.78 4.98 5.11 5.21 5.31 5.43 5.69
99.8 or 100.2 2.77 4.13 4.41 4.59 4.73 4.93 5.07 5.16 5.27 5.39 5.67
99.6 or 100.4 2.74 4.08 4.35 4.54 4.68 4.88 5.02 5.12 5.23 5.35 5.64
99.4 or 100.6 2.70 4.04 4.30 4.49 4.63 4.83 4.96 5.07 5.18 5.30 5.60
99.2 or 100.8 2.67 3.99 4.26 4.43 4.57 4.77 4.91 5.01 5.12 5.25 5.55
99.0 or 101.0 2.64 3.94 4.20 4.39 4.51 4.71 4.85 4.95 5.07 5.20 5.49
98.8 or 101.2 2.60 3.88 4.15 4.33 4.47 4.65 4.79 4.89 5.01 5.14 5.44
98.6 or 101.4 2.57 3.84 4.09 4.27 4.41 4.59 4.73 4.83 4.94 5.07 5.38
98.4 or 101.6 2.54 3.79 4.03 4.21 4.35 4.54 4.67 4.77 4.88 5.01 5.31
98.2 or 101.8 2.50 3.74 3.99 4.16 4.29 4.48 4.61 4.70 4.82 4.94 5.25
98.0 or 102.0 2.47 3.68 3.93 4.11 4.23 4.42 4.55 4.65 4.75 4.88 5.18
97.8 or 102.2 2.43 3.64 3.88 4.05 4.18 4.36 4.49 4.58 4.68 4.81 5.11
97.6 or 102.4 2.40 3.58 3.83 3.99 4.12 4.29 4.42 4.52 4.63 4.74 5.05
97.4 or 102.6 2.37 3.54 3.77 3.93 4.06 4.24 4.36 4.45 4.56 4.68 4.97
97.2 or 102.8 2.33 3.49 3.71 3.88 4.00 4.18 4.30 4.39 4.49 4.62 4.90
97.0 or 103.0 2.30 3.43 3.67 3.82 3.95 4.11 4.24 4.33 4.43 4.55 4.83
96.8 or 103.2 2.26 3.39 3.61 3.77 3.88 4.06 4.17 4.27 4.37 4.48 4.76
96.6 or 103.4 2.23 3.33 3.55 3.71 3.83 4.00 4.12 4.20 4.30 4.41 4.69
96.4 or 103.6 2.20 3.29 3.50 3.65 3.77 3.93 4.05 4.14 4.23 4.35 4.62
96.2 or 103.8 2.16 3.23 3.44 3.60 3.71 3.88 3.99 4.07 4.17 4.28 4.55
96.0 or 104.0 2.13 3.18 3.40 3.54 3.66 3.81 3.93 4.01 4.10 4.21 4.47
95.8 or 104.2 2.09 3.13 3.34 3.49 3.59 3.76 3.86 3.95 4.04 4.15 4.41
95.6 or 104.4 2.06 3.08 3.29 3.43 3.54 3.69 3.80 3.88 3.98 4.08 4.34
95.4 or 104.6 2.03 3.03 3.23 3.38 3.48 3.63 3.74 3.82 3.91 4.02 4.26
95.2 or 104.8 1.99 2.98 3.18 3.31 3.42 3.57 3.67 3.75 3.85 3.94 4.19
95.0 or 105.0 1.96 2.93 3.13 3.26 3.36 3.52 3.61 3.70 3.78 3.88 4.12
94.8 or 105.2 1.92 2.87 3.07 3.20 3.31 3.45 3.56 3.63 3.72 3.81 4.05
94.6 or 105.4 1.89 2.83 3.02 3.15 3.24 3.39 3.49 3.57 3.65 3.75 3.98
94.4 or 105.6 1.86 2.78 2.96 3.09 3.19 3.33 3.43 3.50 3.58 3.68 3.91
94.2 or 105.8 1.82 2.72 2.91 3.03 3.13 3.27 3.36 3.44 3.51 3.61 3.83
94.0 or 106.0 1.79 2.67 2.85 2.98 3.07 3.21 3.30 3.37 3.45 3.54 3.77
93.8 or 106.2 1.75 2.63 2.80 2.92 3.01 3.14 3.24 3.31 3.39 3.47 3.70
93.6 or 106.4 1.72 2.57 2.74 2.86 2.96 3.09 3.17 3.25 3.32 3.41 3.62
93.4 or 106.6 1.69 2.52 2.69 2.81 2.90 3.03 3.11 3.18 3.25 3.34 3.55
93.2 or 106.8 1.65 2.47 2.64 2.75 2.84 2.96 3.05 3.11 3.19 3.27 3.48
93.0 or 107.0 1.62 2.42 2.59 2.70 2.78 2.90 2.99 3.05 3.12 3.21 3.40
92.8 or 107.2 1.58 2.37 2.52 2.64 2.72 2.84 2.92 2.99 3.05 3.14 3.33
92.6 or 107.4 1.55 2.32 2.47 2.58 2.67 2.78 2.86 2.93 2.99 3.07 3.27
92.4 or 107.6 1.52 2.26 2.42 2.52 2.61 2.72 2.80 2.86 2.93 3.00 3.20
92.2 or 107.8 1.48 2.21 2.36 2.47 2.54 2.66 2.74 2.79 2.86 2.93 3.13
92.0 or 108.0 1.45 2.16 2.31 2.41 2.48 2.59 2.68 2.73 2.80 2.87 3.05
91.8 or 108.2 1.41 2.11 2.26 2.35 2.43 2.53 2.61 2.66 2.73 2.80 2.97
91.6 or 108.4 1.38 2.06 2.20 2.30 2.37 2.47 2.55 2.60 2.67 2.73 2.90
91.4 or 108.6 1.35 2.01 2.15 2.24 2.31 2.41 2.49 2.54 2.60 2.67 2.83
91.2 or 108.8 1.31 1.96 2.09 2.18 2.25 2.35 2.43 2.47 2.54 2.60 2.76
91.0 or 109.0 1.28 1.91 2.04 2.13 2.19 2.29 2.36 2.41 2.47 2.53 2.69
90.8 or 109.2 1.24 1.86 1.98 2.07 2.13 2.23 2.30 2.34 2.40 2.46 2.62
90.6 or 109.4 1.21 1.81 1.93 2.01 2.07 2.17 2.23 2.28 2.34 2.39 2.55
90.4 or 109.6 1.18 1.75 1.87 1.95 2.02 2.11 2.17 2.21 2.27 2.33 2.48
90.2 or 109.8 1.14 1.70 1.82 1.90 1.96 2.05 2.11 2.16 2.20 2.26 2.41
90.0 or 110.0 1.11 1.66 1.76 1.85 1.90 1.99 2.04 2.09 2.13 2.20 2.33
5.1.8.3 Example 3—A sample of n = 60 dosage units provided dosage content statistics of X¯ = 98.6 %LC and s = 3.91 %LC at
time of manufacture. At the end of shelf life the dosage units are predicted to have lost potency such that the effective
X¯ = 96.2 %LC. Since s = 3.91 %LC was above the upper limit of 3.71 %LC (using Table 3), we cannot state with 95 % confidence
there is at least a 95 % probability that a future sample taken from the batch will meet the UDU test throughout the shelf life.
SAMPLING PLAN 2
5.1.9 As described in Practice E2709, Sampling Plan 2 (two-stage sampling) divides the population into primary sampling units
(locations). L locations are selected and from each of them a subsample of r items is taken for a total of N5Lxr units. The variance
of a single observation, σ , is the sum of between-location and within-location variances. σ is estimated by 1 ⁄ r *MSB1 1
=~~ ! ~
2 1 ⁄ r!MSE! where MSB and MSE are the between location and within location mean square errors from the analysis of variance
table. The confidence limit for σ is determined using MSE and MSB (see Practice E2709). The joint confidence region is as in
E2810 − 23
TABLE 4 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 95 %, LB = 99 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 2.62 3.88 4.13 4.31 4.43 4.61 4.73 4.82 4.92 5.03 5.27
99.8 or 100.2 2.59 3.84 4.09 4.26 4.39 4.57 4.69 4.78 4.88 4.99 5.25
99.6 or 100.4 2.56 3.79 4.04 4.21 4.34 4.52 4.64 4.73 4.84 4.95 5.22
99.4 or 100.6 2.53 3.75 3.99 4.16 4.29 4.47 4.59 4.68 4.78 4.90 5.17
99.2 or 100.8 2.49 3.70 3.94 4.11 4.23 4.41 4.54 4.63 4.73 4.85 5.13
99.0 or 101.0 2.46 3.65 3.89 4.06 4.18 4.36 4.48 4.57 4.68 4.79 5.07
98.8 or 101.2 2.43 3.61 3.84 4.01 4.13 4.31 4.43 4.52 4.62 4.74 5.02
98.6 or 101.4 2.40 3.56 3.79 3.95 4.08 4.25 4.37 4.46 4.56 4.68 4.96
98.4 or 101.6 2.37 3.51 3.74 3.90 4.02 4.19 4.31 4.40 4.50 4.62 4.90
98.2 or 101.8 2.34 3.47 3.69 3.85 3.97 4.14 4.26 4.34 4.44 4.56 4.83
98.0 or 102.0 2.31 3.42 3.64 3.80 3.92 4.08 4.20 4.29 4.38 4.50 4.77
97.8 or 102.2 2.27 3.37 3.59 3.75 3.86 4.03 4.14 4.23 4.32 4.44 4.70
97.6 or 102.4 2.24 3.33 3.54 3.70 3.81 3.97 4.09 4.17 4.27 4.38 4.64
97.4 or 102.6 2.21 3.28 3.49 3.64 3.76 3.92 4.03 4.11 4.21 4.31 4.58
97.2 or 102.8 2.18 3.23 3.45 3.59 3.70 3.86 3.97 4.05 4.15 4.25 4.51
97.0 or 103.0 2.15 3.19 3.40 3.54 3.65 3.81 3.91 4.00 4.09 4.19 4.45
96.8 or 103.2 2.12 3.14 3.35 3.49 3.60 3.75 3.86 3.94 4.03 4.13 4.38
96.6 or 103.4 2.08 3.09 3.30 3.44 3.54 3.69 3.80 3.88 3.97 4.07 4.32
96.4 or 103.6 2.05 3.05 3.24 3.38 3.49 3.64 3.74 3.82 3.91 4.01 4.25
96.2 or 103.8 2.02 3.00 3.20 3.33 3.43 3.58 3.68 3.76 3.85 3.95 4.19
96.0 or 104.0 1.99 2.95 3.14 3.28 3.38 3.53 3.63 3.70 3.79 3.88 4.12
95.8 or 104.2 1.96 2.91 3.10 3.23 3.33 3.47 3.57 3.64 3.73 3.82 4.06
95.6 or 104.4 1.93 2.86 3.05 3.18 3.27 3.41 3.51 3.58 3.67 3.76 3.99
95.4 or 104.6 1.89 2.81 2.99 3.12 3.22 3.36 3.45 3.52 3.61 3.70 3.92
95.2 or 104.8 1.86 2.76 2.94 3.07 3.16 3.30 3.40 3.47 3.55 3.64 3.86
95.0 or 105.0 1.83 2.72 2.89 3.02 3.11 3.25 3.34 3.41 3.48 3.58 3.79
94.8 or 105.2 1.80 2.67 2.84 2.97 3.06 3.19 3.28 3.35 3.43 3.51 3.73
94.6 or 105.4 1.77 2.62 2.79 2.91 3.00 3.13 3.22 3.29 3.36 3.45 3.66
94.4 or 105.6 1.73 2.58 2.74 2.86 2.95 3.08 3.16 3.23 3.30 3.39 3.60
94.2 or 105.8 1.70 2.53 2.69 2.81 2.90 3.02 3.11 3.17 3.24 3.33 3.53
94.0 or 106.0 1.67 2.48 2.64 2.76 2.84 2.96 3.05 3.11 3.18 3.26 3.46
93.8 or 106.2 1.64 2.43 2.59 2.70 2.79 2.91 2.99 3.05 3.12 3.20 3.40
93.6 or 106.4 1.61 2.39 2.54 2.65 2.73 2.85 2.93 2.99 3.06 3.14 3.33
93.4 or 106.6 1.58 2.34 2.49 2.60 2.68 2.79 2.87 2.93 3.00 3.08 3.27
93.2 or 106.8 1.54 2.29 2.44 2.55 2.62 2.74 2.82 2.87 2.94 3.02 3.20
93.0 or 107.0 1.51 2.24 2.39 2.49 2.57 2.68 2.76 2.82 2.88 2.95 3.14
92.8 or 107.2 1.48 2.20 2.34 2.44 2.52 2.63 2.70 2.76 2.82 2.89 3.07
92.6 or 107.4 1.45 2.15 2.29 2.39 2.46 2.57 2.64 2.70 2.76 2.83 3.00
92.4 or 107.6 1.42 2.10 2.24 2.34 2.41 2.51 2.58 2.64 2.70 2.77 2.94
92.2 or 107.8 1.38 2.06 2.19 2.28 2.36 2.46 2.53 2.58 2.64 2.71 2.87
92.0 or 108.0 1.35 2.01 2.14 2.23 2.30 2.40 2.47 2.52 2.58 2.64 2.81
91.8 or 108.2 1.32 1.96 2.09 2.18 2.25 2.34 2.41 2.46 2.52 2.58 2.74
91.6 or 108.4 1.29 1.91 2.04 2.13 2.19 2.29 2.35 2.40 2.46 2.52 2.67
91.4 or 108.6 1.26 1.87 1.99 2.07 2.14 2.23 2.29 2.34 2.39 2.46 2.61
91.2 or 108.8 1.23 1.82 1.94 2.02 2.08 2.17 2.24 2.28 2.34 2.39 2.54
91.0 or 109.0 1.19 1.77 1.89 1.97 2.03 2.12 2.18 2.22 2.27 2.33 2.48
90.8 or 109.2 1.16 1.73 1.84 1.92 1.98 2.06 2.12 2.16 2.21 2.27 2.41
90.6 or 109.4 1.13 1.68 1.79 1.86 1.92 2.00 2.06 2.11 2.15 2.21 2.34
90.4 or 109.6 1.10 1.63 1.74 1.81 1.87 1.95 2.00 2.04 2.09 2.15 2.28
90.2 or 109.8 1.07 1.58 1.69 1.76 1.81 1.89 1.95 1.99 2.03 2.08 2.21
90.0 or 110.0 1.03 1.54 1.64 1.71 1.76 1.84 1.89 1.93 1.97 2.02 2.15
¯
Sampling Plan 1. For fixed values of the between and within location standard deviations, the sample mean (X) is set at 83.5. Then
the confidence region is expanded by increasing the sample mean until the left vertex just touches the acceptable parameter region.
Then the sample mean is set at 116.5 and the confidence region is expanded by decreasing the sample mean until the right vertex
just touches the acceptable parameter region. The size of the confidence region is determined by C, L, and r.
5.1.10 Computer programs have been developed for generating acceptance limit tables, but these may not be available to all
practitioners. The International Society for Pharmaceutical Engineering (ISPE) has made available information for common tables
for Sampling Plans 1 and 2, a SAS program, and other useful tools.
5.1.11 Using the Acceptance Limit Tables in This Practice—For a given sampling plan with L locations with r dosage units tested
at each location and a specified confidence level and probability of passing the USP UDU test, the acceptance limit table provides
a lower and upper range for the overall L × r mean based on the pooled within location standard deviation (SE) and the standard
https://www.ispe.org/initiatives/blend-uniformity-content-uniformity/toolshttps://www.ispe.org/initiatives/blend-uniformity-content-uniformity/tools.
E2810 − 23
TABLE 5 Acceptance Limits on Sample Standard Deviation (%LC) for
T = 100 %LC, C = 90 %, LB = 95 %LC
Sample Size (n)
Sample Average
(%LC)
10 30 40 50 60 80 100 120 150 200 500
100.0 3.21 4.47 4.71 4.88 5.00 5.16 5.27 5.35 5.44 5.54 5.76
99.8 or 100.2 3.17 4.42 4.67 4.83 4.95 5.12 5.23 5.32 5.41 5.51 5.74
99.6 or 100.4 3.13 4.37 4.61 4.78 4.90 5.07 5.19 5.27 5.36 5.47 5.71
99.4 or 100.6 3.09 4.32 4.56 4.72 4.85 5.02 5.14 5.22 5.32 5.42 5.67
99.2 or 100.8 3.06 4.27 4.50 4.67 4.79 4.96 5.08 5.17 5.26 5.37 5.63
99.0 or 101.0 3.02 4.21 4.45 4.61 4.73 4.91 5.02 5.11 5.21 5.32 5.58
98.8 or 101.2 2.98 4.16 4.39 4.55 4.67 4.84 4.96 5.05 5.15 5.26 5.52
98.6 or 101.4 2.94 4.11 4.34 4.50 4.62 4.78 4.90 4.99 5.08 5.20 5.46
98.4 or 101.6 2.90 4.05 4.28 4.44 4.55 4.72 4.84 4.93 5.02 5.13 5.40
98.2 or 101.8 2.86 4.00 4.22 4.38 4.50 4.66 4.78 4.86 4.96 5.07 5.33
98.0 or 102.0 2.82 3.95 4.17 4.32 4.44 4.60 4.71 4.80 4.89 5.00 5.26
97.8 or 102.2 2.78 3.89 4.11 4.26 4.37 4.54 4.65 4.73 4.82 4.93 5.19
97.6 or 102.4 2.75 3.84 4.05 4.20 4.31 4.47 4.58 4.67 4.76 4.86 5.12
97.4 or 102.6 2.71 3.79 4.00 4.14 4.25 4.41 4.52 4.60 4.69 4.80 5.05
97.2 or 102.8 2.67 3.73 3.94 4.08 4.19 4.35 4.45 4.53 4.63 4.73 4.98
97.0 or 103.0 2.63 3.68 3.88 4.03 4.13 4.29 4.39 4.47 4.56 4.66 4.91
96.8 or 103.2 2.59 3.62 3.82 3.97 4.07 4.22 4.33 4.40 4.49 4.59 4.84
96.6 or 103.4 2.55 3.57 3.77 3.91 4.01 4.16 4.26 4.34 4.42 4.52 4.76
96.4 or 103.6 2.51 3.52 3.71 3.85 3.95 4.10 4.20 4.27 4.36 4.46 4.69
96.2 or 103.8 2.47 3.46 3.65 3.79 3.89 4.03 4.13 4.21 4.29 4.39 4.62
96.0 or 104.0 2.43 3.41 3.60 3.73 3.83 3.97 4.07 4.14 4.22 4.32 4.55
95.8 or 104.2 2.40 3.35 3.54 3.67 3.77 3.91 4.00 4.08 4.16 4.25 4.48
95.6 or 104.4 2.36 3.30 3.48 3.61 3.71 3.84 3.94 4.01 4.09 4.18 4.40
95.4 or 104.6 2.32 3.24 3.43 3.55 3.65 3.78 3.87 3.95 4.02 4.11 4.33
95.2 or 104.8 2.28 3.19 3.37 3.49 3.58 3.72 3.81 3.88 3.96 4.04 4.26
95.0 or 105.0 2.24 3.13 3.31 3.43 3.52 3.66 3.75 3.81 3.89 3.97 4.19
94.8 or 105.2 2.20 3.08 3.25 3.37 3.46 3.59 3.68 3.75 3.82 3.91 4.11
94.6 or 105.4 2.16 3.03 3.20 3.31 3.40 3.53 3.61 3.68 3.75 3.84 4.04
94.4 or 105.6 2.12 2.97 3.14 3.25 3.34 3.47 3.55 3.61 3.69 3.77 3.97
94.2 or 105.8 2.09 2.92 3.08 3.20 3.28 3.40 3.49 3.55 3.62 3.70 3.90
94.0 or 106.0 2.05 2.86 3.02 3.13 3.22 3.34 3.42 3.48 3.55 3.63 3.82
93.8 or 106.2 2.01 2.81 2.97 3.08 3.16 3.27 3.36 3.42 3.48 3.56 3.75
93.6 or 106.4 1.97 2.75 2.91 3.02 3.10 3.21 3.29 3.35 3.42 3.49 3.68
93.4 or 106.6 1.93 2.70 2.85 2.96 3.04 3.15 3.23 3.28 3.35 3.42 3.61
93.2 or 106.8 1.89 2.65 2.79 2.90 2.97 3.08 3.16 3.22 3.28 3.36 3.53
93.0 or 107.0 1.85 2.59 2.74 2.84 2.91 3.02 3.10 3.15 3.21 3.29 3.46
92.8 or 107.2 1.81 2.54 2.68 2.78 2.85 2.96 3.03 3.09 3.15 3.22 3.39
92.6 or 107.4 1.77 2.48 2.62 2.72 2.79 2.89 2.96 3.02 3.08 3.15 3.32
92.4 or 107.6 1.74 2.43 2.56 2.66 2.73 2.83 2.90 2.95 3.01 3.08 3.24
92.2 or 107.8 1.70 2.37 2.51 2.60 2.67 2.77 2.84 2.89 2.94 3.01 3.17
92.0 or 108.0 1.66 2.32 2.45 2.54 2.61 2.70 2.77 2.82 2.88 2.94 3.10
91.8 or 108.2 1.62 2.26 2.39 2.48 2.55 2.64 2.71 2.75 2.81 2.87 3.02
91.6 or 108.4 1.58 2.21 2.33 2.42 2.48 2.58 2.64 2.69 2.74 2.80 2.95
91.4 or 108.6 1.54 2.16 2.27 2.36 2.42 2.51 2.58 2.62 2.67 2.73 2.88
91.2 or 108.8 1.50 2.10 2.22 2.30 2.36 2.45 2.51 2.55 2.61 2.66 2.81
91.0 or 109.0 1.46 2.05 2.16 2.24 2.30 2.39 2.44 2.49 2.54 2.60 2.73
90.8 or 109.2 1.42 1.99 2.10 2.18 2.24 2.32 2.38 2.42 2.47 2.53 2.66
90.6 or 109.4 1.38 1.94 2.04 2.12 2.18 2.26 2.31 2.36 2.40 2.46 2.59
90.4 or 109.6 1.34 1.88 1.99 2.06 2.12 2.19 2.25 2.29 2.34 2.39 2.51
90.2 or 109.8 1.31 1.83 1.93 2.00 2.05 2.13 2.18 2.22 2.27 2.32 2.44
90.0 or 110.0 1.27 1.77 1.87 1.94 1.99 2.07 2.12 2.16 2.20 2.25 2.37
deviation of the L location means (not the between location variance component). The pooled location standard deviation is the
square root of the average L within location variances.
5.1.11.1 Two examples of acceptance limit tables for Sampling Plan 2 are provided below. These two sampling plans were selected
based on material given in two papers (7, 8) published by an ISPE Blend Uniformity/Content Uniformity (BUCU) group that
became active after the FDA withdrew a process validation guidance. ISPE has made available information regarding various
8 9
activities of the group. Table 6 and Table 7 contain information available from the ISPE. Both tables were created using a
confidence level of C = 90 % and a LB = 95 % and assume a target value of 100 %LC. Table 6 applies to a sampling plan with
20 locations with 3 results per location (20 × 3) and Table 7 applies to 40 locations with 3 results per location. The between location
and within location standard deviations start at 0.1 %LC 0.1 %LC and increment by 0.1 %LC 0.1 %LC up to the maximum
possible range on the overall mean.
https://ispe.org/search?keys=BUCUhttps://ispe.org/search?keys=BUCU.
https://ispe.org/initiatives/blend-uniformity-content-uniformity/tools/samplinghttps://ispe.org/initiatives/blend-uniformity-content-uniformity/tools/sampling.
E2810 − 23
TABLE 6 Sampling Plan 2 (20 Locations: 3 Results at Each Location) Acceptance Limits on Overall Mean (%LC) for
T = 100 %LC, C = 90 %, LB = 95 %LC
Standard Deviation of Location Means (%LC)
SE
0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8
(%LC)
LL UL LL UL LL UL LL UL LL UL LL UL LL UL LL UL
0.1 84.0 116.0 84.4 115.6 84.8 115.2 85.2 114.8 85.6 114.4 86.0 114.0 86.4 113.6 86.8 113.2
0.2 84.2 115.8 84.5 115.5 84.8 115.2 85.2 114.8 85.6 114.4 86.0 114.0 86.4 113.6 86.8 113.2
0.3 84.4 115.6 84.6 115.4 84.9 115.1 85.3 114.7 85.7 114.3 86.1 113.9 86.5 113.5 86.8 113.2
0.4 84.7 115.3 84.8 115.2 85.1 114.9 85.4 114.6 85.8 114.2 86.1 113.9 86.5 113.5 86.9 113.1
0.5 84.9 115.1 85.0 115.0 85.3 114.7 85.5 114.5 85.9 114.1 86.2 113.8 86.6 113.4 87.0 113.0
0.6 85.1 114.9 85.3 114.7 85.5 114.5 85.7 114.3 86.0 114.0 86.3 113.7 86.7 113.3 87.1 112.9
0.7 85.4 114.6 85.5 114.5 85.7 114.3 85.9 114.1 86.2 113.8 86.5 113.5 86.8 113.2 87.2 112.8
0.8 85.6 114.4 85.7 114.3 85.9 114.1 86.1 113.9 86.3 113.7 86.6 113.4 86.9 113.1 87.3 112.7
0.9 85.9 114.1 86.0 114.0 86.1 113.9 86.3 113.7 86.5 113.5 86.8 113.2 87.1 112.9 87.4 112.6
1.0 86.1 113.9 86.2 113.8 86.4 113.6 86.5 113.5 86.7 113.3 87.0 113.0 87.2 112.8 87.5 112.5
1.1 86.4 113.6 86.5 113.5 86.6 113.4 86.8 113.2 86.9 113.1 87.2 112.8 87.4 112.6 87.7 112.3
1.2 86.6 113.4 86.7 113.3 86.8 113.2 87.0 113.0 87.2 112.8 87.4 112.6 87.6 112.4 87.9 112.1
1.3 86.9 113.1 87.0 113.0 87.1 112.9 87.2 112.8 87.4 112.6 87.6 112.4 87.8 112.2 88.1 111.9
1.4 87.1 112.9 87.2 112.8 87.3 112.7 87.5 112.5 87.6 112.4 87.8 112.2 88.0 112.0 88.2 111.8
1.5 87.4 112.6 87.5 112.5 87.6 112.4 87.7 112.3 87.9 112.1 88.0 112.0 88.2 111.8 88.4 111.6
1.6 87.6 112.4 87.7 112.3 87.8 112.2 87.9 112.1 88.1 111.9 88.2 111.8 88.4 111.6 88.6 111.4
1.7 87.9 112.1 88.0 112.0 88.1 111.9 88.2 111.8 88.3 111.7 88.5 111.5 88.7 111.3 88.9 111.1
1.8 88.1 111.9 88.2 111.8 88.3 111.7 88.4 111.6 88.6 111.4 88.7 111.3 88.9 111.1 89.1 110.9
1.9 88.4 111.6 88.5 111.5 88.6 111.4 88.7 111.3 88.8 111.2 88.9 111.1 89.1 110.9 89.3 110.7
2.0 88.7 111.3 88.7 111.3 88.8 111.2 88.9 111.1 89.0 111.0 89.2 110.8 89.3 110.7 89.5 110.5
2.1 88.9 111.1 89.0 111.0 89.1 110.9 89.2 110.8 89.3 110.7 89.4 110.6 89.6 110.4 89.7 110.3
2.2 89.2 110.8 89.2 110.8 89.3 110.7 89.4 110.6 89.5 110.5 89.7 110.3 89.8 110.2 90.0 110.0
2.3 89.4 110.6 89.5 110.5 89.6 110.4 89.7 110.3 89.8 110.2 89.9 110.1 90.0 110.0 90.2 109.8
2.4 89.7 110.3 89.7 110.3 89.8 110.2 89.9 110.1 90.0 110.0 90.1 109.9 90.3 109.7 90.4 109.6
2.5 89.9 110.1 90.0 110.0 90.1 109.9 90.2 109.8 90.3 109.7 90.4 109.6 90.5 109.5 90.7 109.3
2.6 90.2 109.8 90.2 109.8 90.3 109.7 90.4 109.6 90.5 109.5 90.6 109.4 90.8 109.2 90.9 109.1
2.7 90.4 109.6 90.5 109.5 90.6 109.4 90.7 109.3 90.8 109.2 90.9 109.1 91.0 109.0 91.1 108.9
2.8 90.7 109.3 90.7 109.3 90.8 109.2 90.9 109.1 91.0 109.0 91.1 108.9 91.2 108.8 91.4 108.6
2.9 90.9 109.1 91.0 109.0 91.1 108.9 91.2 108.8 91.3 108.7 91.4 108.6 91.5 108.5 91.6 108.4
3.0 91.2 108.8 91.2 108.8 91.3 108.7 91.4 108.6 91.5 108.5 91.6 108.4 91.7 108.3 91.9 108.1
3.1 91.4 108.6 91.5 108.5 91.6 108.4 91.7 108.3 91.8 108.2 91.9 108.1 92.0 108.0 92.1 107.9
3.2 91.7 108.3 91.7 108.3 91.8 108.2 91.9 108.1 92.0 108.0 92.1 107.9 92.2 107.8 92.3 107.7
3.3 91.9 108.1 92.0 108.0 92.1 107.9 92.2 107.8 92.3 107.7 92.4 107.6 92.5 107.5 92.6 107.4
3.4 92.2 107.8 92.2 107.8 92.3 107.7 92.4 107.6 92.5 107.5 92.6 107.4 92.7 107.3 92.8 107.2
3.5 92.4 107.6 92.5 107.5 92.6 107.4 92.7 107.3 92.8 107.2 92.9 107.1 93.0 107.0 93.1 106.9
3.6 92.7 107.3 92.7 107.3 92.8 107.2 92.9 107.1 93.0 107.0 93.1 106.9 93.2 106.8 93.3 106.7
3.7 92.9 107.1 93.0 107.0 93.1 106.9 93.2 106.8 93.3 106.7 93.4 106.6 93.5 106.5 93.6 106.4
3.8 93.2 106.8 93.3 106.7 93.3 106.7 93.4 106.6 93.5 106.5 93.6 106.4 93.7 106.3 93.8 106.2
3.9 93.4 106.6 93.5 106.5 93.6 106.4 93.7 106.3 93.8 106.2 93.8 106.2 94.0 106.0 94.1 105.9
4.0 93.7 106.3 93.8 106.2 93.8 106.2 93.9 106.1 94.0 106.0 94.1 105.9 94.2 105.8 94.3 105.7
4.1 93.9 106.1 94.0 106.0 94.1 105.9 94.2 105.8 94.3 105.7 94.3 105.7 94.5 105.5 94.6 105.4
4.2 94.2 105.8 94.3 105.7 94.3 105.7 94.4 105.6 94.5 105.5 94.6 105.4 94.7 105.3 94.8 105.2
4.3 94.4 105.6 94.5 105.5 94.6 105.4 94.7 105.3 94.8 105.2 94.9 105.1 95.0 105.0 95.1 104.9
4.4 94.7 105.3 94.8 105.2 94.8 105.2 94.9 105.1 95.0 105.0 95.1 104.9 95.2 104.8 95.3 104.7
4.5 95.0 105.0 95.0 105.0 95.1 104.9 95.2 104.8 95.3 104.7 95.4 104.6 95.5 104.5 95.6 104.4
4.6 95.2 104.8 95.3 104.7 95.4 104.6 95.4 104.6 95.5 104.5 95.6 104.4 95.7 104.3 95.8 104.2
4.7 95.5 104.5 95.5 104.5 95.6 104.4 95.7 104.3 95.8 104.2 95.9 104.1 96.0 104.0 96.1 103.9
4.8 95.7 104.3 95.8 104.2 95.9 104.1 95.9 104.1 96.0 104.0 96.1 103.9 96.2 103.8 96.3 103.7
4.9 96.0 104.0 96.0 104.0 96.1 103.9 96.2 103.8 96.3 103.7 96.4 103.6 96.5 103.5 96.6 103.4
5.0 96.2 103.8 96.3 103.7 96.4 103.6 96.4 103.6 96.5 103.5 96.6 103.4 96.7 103.3 96.8 103.2
5.1 96.5 103.5 96.6 103.4 96.6 103.4 96.7 103.3 96.8 103.2 96.9 103.1 97.0 103.0 97.1 102.9
5.2 96.7 103.3 96.8 103.2 96.9 103.1 97.0 103.0 97.0 103.0 97.1 102.9 97.2 102.8 97.3 102.7
5.3 97.0 103.0 97.1 102.9 97.1 102.9 97.2 102.8 97.3 102.7 97.4 102.6 97.5 102.5 97.6 102.4
5.4 97.3 102.7 97.3 102.7 97.4 102.6 97.5 102.5 97.6 102.4 97.6 102.4 97.7 102.3 97.8 102.2
5.5 97.5 102.5 97.6 102.4 97.7 102.3 97.7 102.3 97.8 102.2 97.9 102.1 98.0 102.0 98.1 101.9
5.6 97.8 102.2 97.8 102.2 97.9 102.1 98.0 102.0 98.1 101.9 98.2 101.8 98.3 101.7 98.4 101.6
5.7 98.1 101.9 98.1 101.9 98.2 101.8 98.3 101.7 98.4 101.6 98.4 101.6 98.5 101.5 98.6 101.4
5.8 98.4 101.6 98.4 101.6 98.5 101.5 98.6 101.4 98.7 101.3 98.7 101.3 98.8 101.2 98.9 101.1
5.9 98.7 101.3 98.8 101.2 98.8 101.2 98.9 101.1 99.0 101.0 99.1 100.9 99.2 100.8 99.3 100.7
6.0 99.1 100.9 99.2 100.8 99.3 100.7 99.4 100.6 99.5 100.5 99.6 100.4 99.7 100.3 99.9 100.1
E2810 − 23
TABLE 6 Continued
Standard Deviation of Location Means (%LC)
SE
0.9 1.0 1.1 1.2 1.3 1.4 1.5 1.6
(%LC)
LL UL LL UL LL UL LL UL LL UL LL UL LL UL LL UL
0.1 87.2 112.8 87.6 112.4 88.0 112.0 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 90.0 110.0
0.2 87.2 112.8 87.6 112.4 88.0 112.0 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 90.0 110.0
0.3 87.2 112.8 87.6 112.4 88.0 112.0 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 90.0 110.0
0.4 87.3 112.7 87.7 112.3 88.1 111.9 88.5 111.5 88.9 111.1 89.3 110.7 89.7 110.3 90.1 109.9
0.5 87.4 112.6 87.7 112.3 88.1 111.9 88.5 111.5 88.9 111.1 89.3 110.7 89.7 110.3 90.1 109.9
0.6 87.4 112.6 87.8 112.2 88.2 111.8 88.6 111.4 89.0 111.0 89.4 110.6 89.7 110.3 90.1 109.9
0.7 87.5 112.5 87.9 112.1 88.3 111.7 88.6 111.4 89.0 111.0 89.4 110.6 89.8 110.2 90.2 109.8
0.8 87.6 112.4 88.0 112.0 88.3 111.7 88.7 111.3 89.1 110.9 89.5 110.5 89.9 110.1 90.2 109.8
0.9 87.7 112.3 88.1 111.9 88.4 111.6 88.8 111.2 89.2 110.8 89.6 110.4 89.9 110.1 90.3 109.7
1.0 87.9 112.1 88.2 111.8 88.5 111.5 88.9 111.1 89.3 110.7 89.6 110.4 90.0 110.0 90.4 109.6
1.1 88.0 112.0 88.3 111.7 88.7 111.3 89.0 111.0 89.4 110.6 89.7 110.3 90.1 109.9 90.5 109.5
1.2 88.2 111.8 88.5 111.5 88.8 111.2 89.1 110.9 89.5 110.5 89.8 110.2 90.2 109.8 90.6 109.4
1.3 88.3 111.7 88.6 111.4 88.9 111.1 89.3 110.7 89.6 110.4 89.9 110.1 90.3 109.7 90.7 109.3
1.4 88.5 111.5 88.8 111.2 89.1 110.9 89.4 110.6 89.7 110.3 90.1 109.9 90.4 109.6 90.8 109.2
1.5 88.7 111.3 89.0 111.0 89.2 110.8 89.5 110.5 89.9 110.1 90.2 109.8 90.5 109.5 90.9 109.1
1.6 88.9 111.1 89.1 110.9 89.4 110.6 89.7 110.3 90.0 110.0 90.3 109.7 90.6 109.4 91.0 109.0
1.7 89.1 110.9 89.3 110.7 89.6 110.4 89.9 110.1 90.2 109.8 90.5 109.5 90.8 109.2 91.1 108.9
1.8 89.3 110.7 89.5 110.5 89.8 110.2 90.0 110.0 90.3 109.7 90.6 109.4 90.9 109.1 91.2 108.8
1.9 89.5 110.5 89.7 110.3 90.0 110.0 90.2 109.8 90.5 109.5 90.8 109.2 91.1 108.9 91.4 108.6
2.0 89.7 110.3 89.9 110.1 90.1 109.9 90.4 109.6 90.7 109.3 90.9 109.1 91.2 108.8 91.5 108.5
2.1 89.9 110.1 90.1 109.9 90.3 109.7 90.6 109.4 90.8 109.2 91.1 108.9 91.4 108.6 91.7 108.3
2.2 90.1 109.9 90.3 109.7 90.6 109.4 90.8 109.2 91.0 109.0 91.3 108.7 91.6 108.4 91.9 108.1
2.3 90.4 109.6 90.6 109.4 90.8 109.2 91.0 109.0 91.2 108.8 91.5 108.5 91.7 108.3 92.0 108.0
2.4 90.6 109.4 90.8 109.2 91.0 109.0 91.2 108.8 91.4 108.6 91.7 108.3 91.9 108.1 92.2 107.8
2.5 90.8 109.2 91.0 109.0 91.2 108.8 91.4 108.6 91.6 108.4 91.9 108.1 92.1 107.9 92.4 107.6
2.6 91.1 108.9 91.2 108.8 91.4 108.6 91.6 108.4 91.8 108.2 92.1 107.9 92.3 107.7 92.6 107.4
2.7 91.3 108.7 91.5 108.5 91.6 108.4 91.8 108.2 92.0 108.0 92.3 107.7 92.5 107.5 92.7 107.3
2.8 91.5 108.5 91.7 108.3 91.9 108.1 92.0 108.0 92.2 107.8 92.5 107.5 92.7 107.3 92.9 107.1
2.9 91.8 108.2 91.9 108.1 92.1 107.9 92.3 107.7 92.5 107.5 92.7 107.3 92.9 107.1 93.1 106.9
3.0 92.0 108.0 92.2 107.8 92.3 107.7 92.5 107.5 92.7 107.3 92.9 107.1 93.1 106.9 93.3 106.7
3.1 92.2 107.8 92.4 107.6 92.5 107.5 92.7 107.3 92.9 107.1 93.1 106.9 93.3 106.7 93.5 106.5
3.2 92.5 107.5 92.6 107.4 92.8 107.2 92.9 107.1 93.1 106.9 93.3 106.7 93.5 106.5 93.7 106.3
3.3 92.7 107.3 92.9 107.1 93.0 107.0 93.2 106.8 93.4 106.6 93.5 106.5 93.7 106.3 93.9 106.1
...


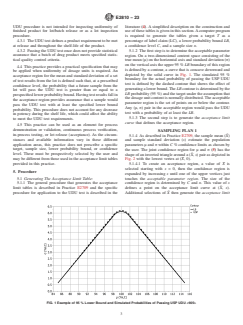
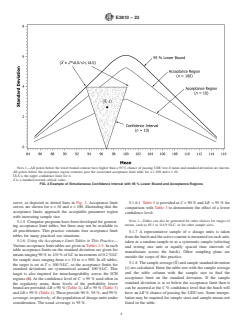



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...