ASTM F3090-20
(Test Method)Standard Test Method for Fatigue Testing of Acetabular Devices for Total Hip Replacement
Standard Test Method for Fatigue Testing of Acetabular Devices for Total Hip Replacement
SIGNIFICANCE AND USE
5.1 This in-vitro test method includes the use of cyclic forces to evaluate the fatigue strength of acetabular shells or monoblock acetabular devices used in THR.
5.2 Fracture or cracking of acetabular shells or monoblock acetabular devices in THR, although rare, does occur.
SCOPE
1.1 This test method is intended to evaluate the fatigue strength of metallic acetabular shells with hemispheric outer surfaces.
1.2 This test method, as described, is not intended to evaluate the following: the strength of components that may be mated with the acetabular shells (for example, augments, acetabular liners); attributes of the shells not related to strength (for example, fixation, coating adhesion); the strength of acetabular shell features away from, or loaded differently than, the primary load bearing region of the shell (for example, screws, spikes, flanges); non-hemispherical shells (for example, patient-matched geometries); or corrosion between modular components.
1.3 Modifications to this test method (for example, different support medium, different size/position of unsupported region, different testing environment) may result in a method appropriate to evaluate the characteristics listed in 1.2. Such modification must have adequate justification.
1.4 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison of acetabular device designs under repeated forces.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Feb-2009
- Effective Date
- 15-Dec-2008
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Jan-2007
- Effective Date
- 01-Nov-2003
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Apr-2003
- Effective Date
- 10-Nov-2002
- Effective Date
- 10-Apr-2001
- Effective Date
- 10-Apr-2001
- Effective Date
- 10-Oct-1997
Overview
ASTM F3090-20: Standard Test Method for Fatigue Testing of Acetabular Devices for Total Hip Replacement outlines a laboratory procedure for evaluating the fatigue strength of metallic acetabular shells and monoblock acetabular devices used in total hip replacement (THR). This in-vitro standard is crucial for assessing the ability of these orthopedic devices to withstand cyclic forces and repeated mechanical loading similar to those experienced in real-world activities. The standard helps manufacturers and researchers benchmark device durability, minimize fracture risk, and compare different acetabular designs in a controlled environment.
Key Topics
- Fatigue Strength Testing: Defines the process for subjecting acetabular shells or devices to repeated cyclic forces to determine resistance to cracking or fracture.
- Applicability: Specifically targets metallic acetabular shells with hemispherical outer surfaces. The method does not address the strength of liners, non-load-bearing features, non-hemispherical shells, or corrosion testing.
- Support and Loading Conditions: Devices are partially supported and loaded simulating an extreme physiological position, focusing stress on the primary load-bearing shell region.
- Specimen Selection and Preparation: Recommends testing a worst-case device configuration, considering variables such as liner, head size, and device orientation (“clocking”) to ensure relevant evaluation.
- Test Parameters: Includes procedures for preparation, force application, temperature and environment controls, and use of SI measurement units.
- Post-Test Evaluation: Guidelines for visual or dye penetrant inspection of devices for cracks or fractures, and for proper reporting of results and conditions.
Applications
ASTM F3090-20 is essential for various stakeholders in the orthopedic medical device field:
- Manufacturers: Enables design optimization and verification of acetabular shells for improved durability and patient safety.
- Research and Development: Offers a consistent method to compare new or existing acetabular device designs for fatigue life, supporting innovation.
- Regulatory and Quality Assurance: Assists in fulfilling regulatory requirements and demonstrating device safety based on standardized, internationally credible test data.
- Clinical Risk Assessment: Supports orthopedic surgeons and healthcare professionals by ensuring devices are rigorously tested against structural failure.
The method is particularly valuable in identifying potential fatigue-related failures, which, while rare, can have critical clinical implications. By simulating demanding real-life loading scenarios, it ensures that only robust devices reach the market or clinical trials.
Related Standards
Many standards complement or relate to ASTM F3090-20 in orthopedic device testing:
- ASTM E4: Practices for Force Verification of Testing Machines - ensures test machines are accurate and reliable.
- ASTM F1820: Method for Determining Forces for Disassembly of Modular Acetabular Devices.
- ASTM F2345: Methods for Determination of Static and Cyclic Fatigue Strength of Ceramic Modular Femoral Heads.
- ISO 4287 & ISO 4288: Provide guidance on geometrical product specifications and assessment of surface texture.
- ISO 7206-4: Implants for surgery-Determination of endurance properties and performance of stemmed femoral components.
Conclusion
ASTM F3090-20 provides a robust, internationally recognized method for fatigue testing of acetabular devices used in total hip arthroplasty. Use of this standard helps ensure high performance, reliability, and patient safety for hip replacement components through rigorous mechanical evaluation and data-driven decision-making.
Keywords: ASTM F3090-20, acetabular device, hip replacement, fatigue testing, orthopedic standards, cyclic loading, total hip arthroplasty, metallic acetabular shells, device durability, medical device testing.
Buy Documents
ASTM F3090-20 - Standard Test Method for Fatigue Testing of Acetabular Devices for Total Hip Replacement
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3090-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Fatigue Testing of Acetabular Devices for Total Hip Replacement". This standard covers: SIGNIFICANCE AND USE 5.1 This in-vitro test method includes the use of cyclic forces to evaluate the fatigue strength of acetabular shells or monoblock acetabular devices used in THR. 5.2 Fracture or cracking of acetabular shells or monoblock acetabular devices in THR, although rare, does occur. SCOPE 1.1 This test method is intended to evaluate the fatigue strength of metallic acetabular shells with hemispheric outer surfaces. 1.2 This test method, as described, is not intended to evaluate the following: the strength of components that may be mated with the acetabular shells (for example, augments, acetabular liners); attributes of the shells not related to strength (for example, fixation, coating adhesion); the strength of acetabular shell features away from, or loaded differently than, the primary load bearing region of the shell (for example, screws, spikes, flanges); non-hemispherical shells (for example, patient-matched geometries); or corrosion between modular components. 1.3 Modifications to this test method (for example, different support medium, different size/position of unsupported region, different testing environment) may result in a method appropriate to evaluate the characteristics listed in 1.2. Such modification must have adequate justification. 1.4 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison of acetabular device designs under repeated forces. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This in-vitro test method includes the use of cyclic forces to evaluate the fatigue strength of acetabular shells or monoblock acetabular devices used in THR. 5.2 Fracture or cracking of acetabular shells or monoblock acetabular devices in THR, although rare, does occur. SCOPE 1.1 This test method is intended to evaluate the fatigue strength of metallic acetabular shells with hemispheric outer surfaces. 1.2 This test method, as described, is not intended to evaluate the following: the strength of components that may be mated with the acetabular shells (for example, augments, acetabular liners); attributes of the shells not related to strength (for example, fixation, coating adhesion); the strength of acetabular shell features away from, or loaded differently than, the primary load bearing region of the shell (for example, screws, spikes, flanges); non-hemispherical shells (for example, patient-matched geometries); or corrosion between modular components. 1.3 Modifications to this test method (for example, different support medium, different size/position of unsupported region, different testing environment) may result in a method appropriate to evaluate the characteristics listed in 1.2. Such modification must have adequate justification. 1.4 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison of acetabular device designs under repeated forces. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3090-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3090-20 has the following relationships with other standards: It is inter standard links to ASTM E4-14, ASTM E4-10, ASTM E4-09a, ASTM E4-09, ASTM F1820-97(2009), ASTM F2345-03(2008), ASTM E4-08, ASTM E4-07, ASTM F2345-03, ASTM E4-03, ASTM F1820-97(2003), ASTM E4-02, ASTM E4-99, ASTM E4-01, ASTM F1820-97. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3090-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3090 − 20
Standard Test Method for
Fatigue Testing of Acetabular Devices for Total Hip
Replacement
This standard is issued under the fixed designation F3090; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.1 This test method is intended to evaluate the fatigue
strength of metallic acetabular shells with hemispheric outer
2. Referenced Documents
surfaces.
2.1 ASTM Standards:
1.2 This test method, as described, is not intended to
E4Practices for Force Verification of Testing Machines
evaluatethefollowing:thestrengthofcomponentsthatmaybe
F1820Test Method for Determining the Forces for Disas-
mated with the acetabular shells (for example, augments,
sembly of Modular Acetabular Devices
acetabularliners);attributesoftheshellsnotrelatedtostrength
F2345Test Methods for Determination of Static and Cyclic
(for example, fixation, coating adhesion); the strength of
Fatigue Strength of Ceramic Modular Femoral Heads
acetabular shell features away from, or loaded differently than,
2.2 ISO Standards:
the primary load bearing region of the shell (for example,
ISO 4287 Geometrical Product Specifications (GPS)—
screws, spikes, flanges); non-hemispherical shells (for
Surface texture: Profile method—Terms, definitions and
example, patient-matched geometries); or corrosion between
surface texture parameters
modular components.
ISO 4288 Geometrical Product Specifications (GPS—
Surface texture: Profile method—Rules and procedures
1.3 Modificationstothistestmethod(forexample,different
for the assessment of surface texture
support medium, different size/position of unsupported region,
ISO 7206-4Implants for surgery—Partial and total hip joint
different testing environment) may result in a method appro-
prostheses—Part 4: Determination of endurance proper-
priate to evaluate the characteristics listed in 1.2. Such modi-
ties and performance of stemmed femoral components
fication must have adequate justification.
1.4 Although the methodology described does not replicate
3. Terminology
all physiological force conditions, it is a means of in vitro
3.1 Definitions of Terms Specific to This Standard:
comparisonofacetabulardevicedesignsunderrepeatedforces.
3.1.1 acetabular liner, n—portion of a modular acetabular
1.5 The values stated in SI units are to be regarded as
device with a concave spherical shape intended to articulate
standard. No other units of measurement are included in this
withtheheadofafemoralprosthesis;theexternalgeometryof
standard.
this component interfaces directly with the acetabular shell
through a locking mechanism which may be integral to the
1.6 This standard does not purport to address all of the
design of the liner and shell or may rely upon additional
safety concerns, if any, associated with its use. It is the
components or fixation methods (for example a metal ring or
responsibility of the user of this standard to establish appro-
bone cement).
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
3.1.2 acetabular shell, n—the metallic external, concave
1.7 This international standard was developed in accor-
structurethatmechanicallysupportstheacetabularliner,whose
dance with internationally recognized principles on standard-
external features interface with the bones of the pelvic socket
ization established in the Decision on Principles for the
(forexample,throughbonecement,intimatepress-fit,coatings
Development of International Standards, Guides and Recom-
forattachmenttobonecementortissue,integralscrewthreads,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
ThistestmethodisunderthejurisdictionofASTMCommitteeF04onMedical contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
andSurgicalMaterialsandDevicesandisthedirectresponsibilityofSubcommittee Standards volume information, refer to the standard’s Document Summary page on
F04.22 on Arthroplasty. the ASTM website.
Current edition approved Oct. 1, 2020. Published November 2020. DOI: Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
10.1520/F3090-20. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3090 − 20
anchoringscrews,pegs,andsoforth);theacetabularshellmay 4. Summary of Test Method
have hole(s) for fixation or instrumentation, or have no holes.
4.1 Amodular or monoblock acetabular device is fixed in a
3.1.3 clocking reference point, n—a location on the shell
polymer block with a portion of the acetabular device unsup-
face that can be used to control shell clocking; in cases where
ported. This construct is placed in a fatigue test machine in
the shell face does not have any clear features to use as a
such a way that the polar axis of the acetabular device is at a
reference point it may be necessary to mark a location. The
55° angle relative to the line of force application of the test
location of the clocking reference point to any other shell
machine, as illustrated in Figs. 1 and 2. A head of the
features shall be noted.
appropriate diameter for the acetabular device shall be used to
apply cyclic forces to the acetabular device.
3.1.4 modular acetabular device, n—the acetabular compo-
nent of a total hip replacement (THR) system with multiple
4.2 After completion of fatigue testing, the acetabular liner
components including acetabular liner component(s) and ac-
may be axially disassembled according toTest Method F1820.
etabular shell component(s); it is possible that a modular
4.3 Following liner disassembly, the acetabular shell and
acetabular device might be pre-assembled in a manufacturing
linershallbeeitherexaminedopticallyatamagnificationinthe
process.
range of 10× to 30×, or subject to dye penetrant testing to
3.1.5 monoblock acetabular device, n—the acetabular com-
inspect for evidence of cracks.
ponent of a THR system that, excluding any exterior surface
5. Significance and Use
coatings intended for contact with bone cement or tissue, is
fabricated as a single piece.
5.1 This in-vitro test method includes the use of cyclic
forces to evaluate the fatigue strength of acetabular shells or
3.1.6 shell clocking, n—the rotational orientation of the
shell about the polar axis to the load axis or the center of the monoblock acetabular devices used in THR.
unsupported portion of the shell from a clocking reference
5.2 Fracture or cracking of acetabular shells or monoblock
point on the shell.
acetabular devices in THR, although rare, does occur.
3.1.7 shell face, n—the plane defined by the rim of the
6. Apparatus
acetabular shell that surrounds the opening that the acetabular
liner is placed into; for a precisely hemispherical shell, the 6.1 The specimens under test shall be supported on the
shell face is the equatorial plane. exterior surface of the acetabular device with a polymeric
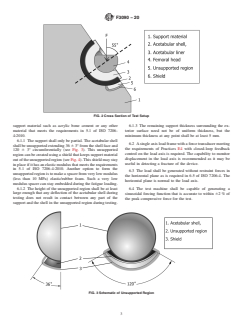
FIG. 1 Terminology and Schematic of Test Setup
F3090 − 20
FIG. 2 Cross Section of Test Setup
support material such as acrylic bone cement or any other 6.1.3 The remaining support thickness surrounding the ex-
material that meets the requirements in 5.1 of ISO 7206- terior surface need not be of uniform thickness, but the
4:2010.
minimum thickness at any point shall be at least 5 mm.
6.1.1 The support shall only be partial. The acetabular shell
6.2 Asingleaxisloadframewithaforcetransducermeeting
shallbeunsupportedextending36 63°fromtheshellfaceand
the requirements of Practices E4 with closed-loop feedback
120 6 3° circumferentially (see Fig. 3). This unsupported
control on the load axis is required. The capability to monitor
regioncanbecreatedusingashieldthatkeepssupportmaterial
displacement in the load axis is recommended as it may be
outoftheunsupportedregion(seeFig.4).Thisshieldmaystay
useful in detecting a fracture of the device.
inplaceifithasanelasticmodulusthatmeetstherequirements
in 5.1 of ISO 7206-4:2010. Another option to form the
6.3 The load shall be generated without restraint forces in
unsupportedregionistomakeaspacerfromverylowmodulus
the horizontal plane as is required in 6.5 of ISO 7206-4. The
(less than 10 MPa) elastic/rubber foam. Such a very low
horizontal plane is normal to the load axis.
modulus spacer can stay embedded during the fatigue loading.
6.1.2 The height of the unsupported region shall be at least
6.4 The test machine shall be capable of generating a
large enough that any deflection of the acetabular shell during
sinusoidal forcing function that is accurate to within 62%of
testing does not result in contact between any part of the
the peak compressive force for the test.
support and the shell in the unsupported region during testing.
FIG. 3 Schematic of Unsupported Region
F3090 − 20
FIG. 4 Views of Shield
7. Specimen Selection MethodF1820.Theregionbehindtheholeintheshellshallbe
freeofthepolymericsupportmaterialtopermitremovalofthe
7.1 If one particular acetabular device is being tested to
acetabular liner.
represent a range of acetabular devices (that is, worst-case
7.2.3 If the shell needs to be modified for liner removal, the
testing), it shall be the acetabular device that results in the
modification shall be modeled in the FE evaluation and
maximum stress in the acetabular shell.
compared to the unmodified version. The modified shell shall
7.1.1 The stress in the acetabular shell may be affected by
have the same peak stress location and magnitude as the
thelinersizeand/ormaterialandtheheadsizeand/ormaterial.
unmodified shell.
The worst-case liner and head shall be determined and justi-
fied.
8. Procedure
7.1.2 The worst-case acetabular device “clocking” orienta-
8.1 Specimen Preparation:
tion(s)shallbedeterminedandjustified.Acetabularshellsmay
8.1.1 If the acetabular liner mates to the shell by means of
have unique or periodic features around the shell. The posi-
a Morse taper, the surface condition of the contacting surfaces
tioning or clocking orientation of these features relative to the
of the liner and the shell may be documented by appropriate
unsupported region shall be evaluated to identify the clocking
microscopic techniques and quantitatively characterized by
orientation(s) that results in the worst case. For example, some
surface finish measurements according to ISO 4287 and ISO
modular shells have periodic scallops along the face of the
4288.
shell that act as anti-rotation features for the mating polyeth-
8.1.2 If the test components were exposed to a contaminat-
ylene liner. The acetabular construct should be evaluated to
ing environment following manufacturing cleaning processes,
determine whether positioning the scallop directly in line with
the components should be re-cleaned using manufacturing
the force or positioning the scallop towards the edge of the
process or cleaned following normal laboratory cleaning pro-
unsupported region results in a worst case. A second example
cedures to remove any debris or other surface contaminants as
would be the orientation of screw holes provided for screw
outlined in Appendix X2 of Test Methods F2345 prior to
fixation to the line of force. Determine a clocking reference
assembly.
point and describe its location.
8.1.3 Surgical assembly methods vary, and some involve
7.1.3 Finite element (FE) analyses of acetabular devices in
impactonabonebasewhichmayacttodampenimpactforces.
the specified test configuration and the consideration of the
Therefore,iftheacetabularlinermatestotheshellbymeansof
stress calculated to occur in the acetabular shell is one way to
a Morse taper, a static assembly witha2kN peak force (as
identify the worst-case specimen.
recommended by Test Methods F2345) applied with a head of
7.1.4 FE modeling of modular interfaces (for example,
the same diameter or the appropriate surgical tool for the
Morse taper) can be difficult and it may be necessary to model
assembly of the acetabular device should be performed. The
modular interfaces as fixed connections. The effect of the
force shall be applied at a loading rate of 500 6 100 N/s or a
interface modeling on the FE analysis should be considered.
displacement rate of 0.04 6 0.01 mm/s. For acetabular liners
7.2 All test specimens shall be of finished implant quality
that do not mate by means of a Morse taper (for example,
unless a justification is given for test samples that are not
polymeric liners with locking tabs), an assembly method
finished implant quality.
representativeoftherecommendedclinicalassemblytechnique
7.2.1 Formodularacetabulardevices,thereshallbeameans
shouldbeperformedfollowedbyanyrecommendedchecksfor
of removing the liner from the shell after completion of the complete assembly/locking.
test.
8.1.4 The acetabular device test specimens shall be fixed in
7.2.2 If the only way of removing the liner from the shell is polymeric support material such that the angle between the
to modify the shell, the shell shall be modified with a hole in forceandtheshellpolaraxisis55 62°(Fig.2).Themidpoint
thepolaraxisasshownintheaxialdisassemblymethodofTest of the unsupported portion of the acetabular devices shall be
F3090 − 20
aligned with the load axis 62° (that is, the 120° angle in Fig. 9.1.2 Record size information and provide the justification
3 shall be bisected by the load axis within 62°). fortheshell,liner,andheadsizecombination(s)chosenforthe
test.
8.1.5 The test specimen shall be placed in the test machine.
9.1.3 Provide the justification for the worst-case acetabular
8.2 Test Parameters:
device and “clocking” orientation(s) selected.
8.2.1 The head selected as worst case shall be used to load
9.1.4 Report the clocking reference point used in the test.
the acetabular device with a sinusoidal forcing function.
9.2 Test Apparatus and Methodology:
8.2.2 The test shall be conducted under force control using
9.2.1 Reportthetestequipmentandmethodusedforassem-
a minimum force corresp
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...