ASTM D6666-20
(Guide)Standard Guide for Evaluation of Aqueous Polymer Quenchants
Standard Guide for Evaluation of Aqueous Polymer Quenchants
SIGNIFICANCE AND USE
4.1 The significance and use of each test method will depend on the system in use and the purpose of the test method listed under Section 7. Use the most recent editions of the test methods.
SCOPE
1.1 This guide provides information, without specific limits, for selecting standard test methods for testing aqueous polymer quenchants for initial qualification, determining quality, and the effect of aging.
1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory requirements prior to use.
1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2020
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.L0.06 - Non-Lubricating Process Fluids
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-May-2019
- Effective Date
- 15-Dec-2016
- Effective Date
- 01-Jun-2015
- Effective Date
- 01-Aug-2014
- Effective Date
- 01-Jul-2014
- Effective Date
- 01-Jul-2014
- Effective Date
- 01-May-2014
Overview
ASTM D6666-20: Standard Guide for Evaluation of Aqueous Polymer Quenchants provides essential guidance for selecting and applying standard test methods for evaluating aqueous polymer quenchants. These fluid solutions are vital in metal heat treat processes, widely used for their ability to control cooling rates and achieve specific material properties in metals. The standard addresses key practices for initial qualification, ongoing quality control, and the assessment of aging effects in aqueous polymer quenchants. ASTM D6666-20 is internationally recognized and aligns with global standardization principles, making it relevant for a broad spectrum of industries that use polymer quenching.
Key Topics
- Selection of Test Methods: The guide offers information to help users select appropriate standard test methods for various stages, including qualification, routine monitoring, and evaluating long-term performance of quenchants.
- Safety and Environmental Considerations: Users are reminded that safety, health, and environmental practices are the user's responsibility when applying this guide, and that local regulatory requirements must be considered.
- Evaluation Parameters:
- Physical and Chemical Properties: Tests cover appearance, refractive index, viscosity, water content, pH, conductivity, and more.
- Contamination Checks: Methods to detect hydrocarbons, sludge, scale, and oil contamination are outlined.
- Performance Testing: Includes cooling curve analysis, assessment of corrosion inhibitors, foam testing, and evaluation of biodegradation and biodeterioration.
- Quality Monitoring: Recommends sampling protocols and testing at consistent locations and intervals for reliable results.
- Aging and Degradation: Procedures are recommended for monitoring changes that occur in polymer quenchants over time, including molecular weight analysis and cloud point testing.
Applications
ASTM D6666-20 is widely applied in industries requiring controlled quenching for metal treatment, including automotive, aerospace, toolmaking, and manufacturing of critical steel components. The guide is used in the following contexts:
- Heat Treatment Operations: For qualifying new quenchants and monitoring in-service baths to ensure consistent cooling performance and product quality.
- Laboratory Test Selection: Assists in choosing methods such as cooling curve analysis, viscosity determination, and refractive index measurement specific to the needs of the quenching operation.
- Routine Process Control: Used for regular checks of polymer quenchant properties such as water content (ASTM D95, D1744), viscosity (ASTM D445, D7042), pH (ASTM E70), ion contamination (ASTM D2624), and microbial stability (ASTM E979, E2275).
- Quality Assurance and Troubleshooting: Provides strategies to identify and remediate contamination, degradation, fouling, and loss of corrosion inhibitor efficacy.
- Environmental and Safety Compliance: Ensures users establish appropriate safety and environmental handling procedures.
Related Standards
ASTM D6666-20 references several key ASTM standards to define testing and evaluation methods for aqueous polymer quenchants, including:
- ASTM D95 – Water content by distillation
- ASTM D445 / D7042 – Kinematic and dynamic viscosity measurement
- ASTM D892 – Foaming characteristics of lubricating oils
- ASTM D1747 – Refractive index of viscous materials
- ASTM D1796 – Water and sediment in fuel oils (centrifuge method)
- ASTM D2624 – Electrical conductivity of fuels
- ASTM D3519 / D3601 – Foam in aqueous media
- ASTM D3867 / D4327 – Nitrite and anion analysis in water
- ASTM D5296 – Molecular weight analysis of polymers
- ASTM D6482 / D6549 – Cooling characteristics (cooling curve analysis)
- ASTM E70 – pH measurement
- ASTM E979 / E2275 – Microbial and biocide evaluation
Organizations applying ASTM D6666-20 benefit from a structured approach to polymer quenchant evaluation, achieve improved process consistency, and facilitate compliance with both industry and international requirements.
Keywords: aqueous polymer quenchants, ASTM D6666-20, cooling curve, viscosity, refractive index, corrosion inhibitor, biodegradation, heat treatment, quality control, polymer degradation, process fluids.
Buy Documents
ASTM D6666-20 - Standard Guide for Evaluation of Aqueous Polymer Quenchants
REDLINE ASTM D6666-20 - Standard Guide for Evaluation of Aqueous Polymer Quenchants
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D6666-20 is a guide published by ASTM International. Its full title is "Standard Guide for Evaluation of Aqueous Polymer Quenchants". This standard covers: SIGNIFICANCE AND USE 4.1 The significance and use of each test method will depend on the system in use and the purpose of the test method listed under Section 7. Use the most recent editions of the test methods. SCOPE 1.1 This guide provides information, without specific limits, for selecting standard test methods for testing aqueous polymer quenchants for initial qualification, determining quality, and the effect of aging. 1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory requirements prior to use. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The significance and use of each test method will depend on the system in use and the purpose of the test method listed under Section 7. Use the most recent editions of the test methods. SCOPE 1.1 This guide provides information, without specific limits, for selecting standard test methods for testing aqueous polymer quenchants for initial qualification, determining quality, and the effect of aging. 1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory requirements prior to use. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6666-20 is classified under the following ICS (International Classification for Standards) categories: 75.120 - Hydraulic fluids. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6666-20 has the following relationships with other standards: It is inter standard links to ASTM D6666-04(2019), ASTM D445-24, ASTM E70-24, ASTM D445-23, ASTM D892-23, ASTM D95-23, ASTM D5296-19, ASTM E2275-19, ASTM D1747-09(2019), ASTM D445-16, ASTM E70-07(2015), ASTM E2275-14, ASTM D445-14e1, ASTM D445-14, ASTM D7042-14. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6666-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6666 − 20
Standard Guide for
Evaluation of Aqueous Polymer Quenchants
This standard is issued under the fixed designation D6666; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* the Centrifuge Method (Laboratory Procedure)
D2624 Test Methods for Electrical Conductivity ofAviation
1.1 This guide provides information, without specific limits,
and Distillate Fuels
forselectingstandardtestmethodsfortestingaqueouspolymer
D3519 Test Method for Foam in Aqueous Media (Blender
quenchants for initial qualification, determining quality, and
Test) (Withdrawn 2013)
the effect of aging.
D3601 Test Method for Foam In Aqueous Media (Bottle
1.2 This standard does not purport to address all of the
Test) (Withdrawn 2013)
safety concerns, if any, associated with its use. It is the
D3867 Test Methods for Nitrite-Nitrate in Water
responsibility of the user of this standard to establish appro-
D4327 Test Method for Anions in Water by Suppressed Ion
priate safety, health, and environmental practices and deter-
Chromatography
mine the applicability of regulatory requirements prior to use.
D5296 Test Method for Molecular Weight Averages and
1.3 This international standard was developed in accor-
Molecular Weight Distribution of Polystyrene by High
dance with internationally recognized principles on standard-
Performance Size-Exclusion Chromatography
ization established in the Decision on Principles for the
D6482 Test Method for Determination of Cooling Charac-
Development of International Standards, Guides and Recom-
teristics of Aqueous Polymer Quenchants by Cooling
mendations issued by the World Trade Organization Technical
Curve Analysis with Agitation (Tensi Method)
Barriers to Trade (TBT) Committee.
D6549 Test Method for Determination of Cooling Charac-
teristics of Quenchants by Cooling Curve Analysis with
2. Referenced Documents
Agitation (Drayton Unit)
2.1 ASTM Standards:
D7042 Test Method for Dynamic Viscosity and Density of
D95 Test Method for Water in Petroleum Products and
Liquids by Stabinger Viscometer (and the Calculation of
Bituminous Materials by Distillation
Kinematic Viscosity)
D445 Test Method for Kinematic Viscosity of Transparent
E70 Test Method for pH of Aqueous Solutions With the
and Opaque Liquids (and Calculation of Dynamic Viscos-
Glass Electrode
ity)
E979 Practice for Evaluation of Antimicrobial Agents as
D892 Test Method for Foaming Characteristics of Lubricat-
Preservatives for Invert Emulsion and Other Water Con-
ing Oils
taining Hydraulic Fluids
D1744 Test Method for Determination of Water in Liquid
E2275 Practice for Evaluating Water-Miscible Metalwork-
Petroleum Products by Karl Fischer Reagent (Withdrawn
ing Fluid Bioresistance and Antimicrobial Pesticide Per-
2016)
formance
D1747 Test Method for Refractive Index of Viscous Mate-
3. Terminology
rials
D1796 Test Method for Water and Sediment in Fuel Oils by
3.1 Definitions of Terms Specific to This Standard:
3.1.1 austenite,n—solidsolutionofoneormoreelementsin
face-centered cubic iron (gamma iron) and unless otherwise
This guide is under the jurisdiction of ASTM Committee D02 on Petroleum 4
designated, the solute is generally assumed to be carbon (1).
Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcom-
mittee D02.L0.06 on Non-Lubricating Process Fluids. 3.1.2 austenitizing, n—forming austenite by heating a fer-
Current edition approved May 1, 2020. Published May 2020. Originally
rous alloy into the transformation range (partial austenitizing)
approved in 2001. Last previous edition approved in 2019 as D6666 – 04 (2019).
or above the transformation range (complete austenitizing).
DOI: 10.1520/D6666-20.
When used without qualification, the term implies complete
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
austenitizing (1).
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website.
The last approved version of this historical standard is referenced on The boldface numbers in parentheses refer to the list of references at the end of
www.astm.org. this standard.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6666 − 20
FIG. 1 Cooling Mechanisms of the Quenching Process
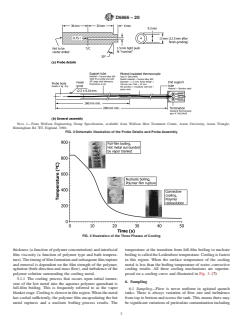
3.1.3 aqueous polymer quenchant, n—a solution containing 3.1.11 full-film boiling, n—upon initial immersion of hot
water, and one or more water-soluble polymers including steel into a quenchant solution, a vapor blanket surrounds the
poly(alkylene glycol), poly(vinyl pyrrolidone), poly(sodium metal surface resulting in full-film boiling as shown in Fig. 1.
acrylate), and poly(ethyl oxazoline) (2, 3) and additives for (5)
corrosion and foam control, if needed.
3.1.12 nucleate boiling, n—when the vapor blanket sur-
3.1.4 biodegradation, n—theprocessbywhichasubstrateis rounding the hot metal collapses and a nucleate boiling process
converted by biological, usually microbiological, agents into occurs as illustrated in Fig. 1. (5)
simple, environmentally acceptable derivatives. (4)
3.1.13 quenchant medium, n—any liquid or gas, or mixture,
3.1.5 biodeterioration, n—loss of product quality and per- usedtocontrolthecoolingofametaltofacilitatetheformation
formance and could be regarded as the initial stages of of the desired microstructure and properties. (1)
biodegradation (see 3.1.4), but in the wrong place at the wrong
3.1.14 quench severity, n—the ability of a quenchant me-
time, that is when the product is stored or in use. (4)
dium to extract heat from hot metal. (6)
3.1.6 convective cooling, n—after continued cooling, and
3.1.15 transformation temperatures, n—characteristic tem-
the interfacial temperature between the cooling metal and the
peratures that are important in the formation of martensitic
aqueous polymer quenchant is less than the boiling point of the
microstructure of steel including: A —equilibrium austeniti-
e1
water in the quenchant solution at which point cooling occurs
zation phase change temperature; M —temperature at which
S
by a convective cooling process. For convective cooling, fluid
transformation of austenite to martensite starts during cooling
motion is due to density differences and the action of gravity
and M—temperature at which transformation of austenite to
f
and includes both natural motion and forced circulation (1, 5).
martensite is completed during cooling. (1)
This process is illustrated in Fig. 1.
3.1.7 cooling curve, n—a graphical representation of the 4. Significance and Use
coolingtime(t)—temperature(T)responseoftheprobesuchas
4.1 The significance and use of each test method will
that shown in Fig. 1. (5)
depend on the system in use and the purpose of the test method
3.1.8 cooling curve analysis, n—the process of quantifying
listed under Section 7. Use the most recent editions of the test
thecoolingcharacteristicsofaquenchantmediumbasedonthe
methods.
temperature versus time profile obtained by cooling a pre-
heated metal probe assembly (see Fig. 2) under specified
5. Quenching Process
conditions which include: probe alloy and dimensions, probe
5.1 Aqueous Polymer Quenchant Cooling Mechanisms—
and bath temperature, agitation rate, and aqueous polymer
Upon initial immersion of a heated metal into a solution of an
quenchant concentration.
aqueous polymer quenchant, an insulating polymer film, which
3.1.9 cooling rate curve, n—obtained by calculating the first
controlstheheattransferratefromthehotmetalintothecooler
derivative (dT/dt) of the cooling time-temperature curve as
quenchant solution, forms around the hot metal which is
illustrated in Fig. 1. (5)
separated by a vapor film (Fig. 3) (7) for the quenching process
3.1.10 dragout, n—solution carried out of a bath on the in a poly(alkylene glycol) quenchant. The overall heat transfer
metal being quenched and associated handling equipment. (1) mediating properties of the film are dependent on both the film
D6666 − 20
NOTE 1—From Wolfson Engineering Group Specification, available from Wolfson Heat Treatment Centre, Aston University, Aston Triangle,
Birmingham B4 7ET, England, 1980.
FIG. 2 Schematic Illustration of the Probe Details and Probe Assembly
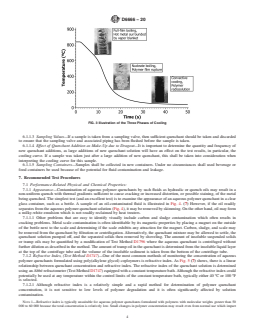
FIG. 3 Illustration of the Three Phases of Cooling
thickness (a function of polymer concentration) and interfacial temperature at the transition from full-film boiling to nucleate
film viscosity (a function of polymer type and bath tempera- boiling is called the Leidenfrost temperature. Cooling is fastest
ture).The timing of film formation and subsequent film rupture in this region. When the surface temperature of the cooling
and removal is dependent on the film strength of the polymer, metal is less than the boiling temperature of water, convective
agitation (both direction and mass flow), and turbulence of the cooling results. All three cooling mechanisms are superim-
polymer solution surrounding the cooling metal. posed on a cooling curve and illustrated in Fig. 3. (7)
5.1.1 The cooling process that occurs upon initial immer-
6. Sampling
sion of the hot metal into the aqueous polymer quenchant is
full-film boiling. This is frequently referred to as the vapor 6.1 Sampling—Flow is never uniform in agitated quench
blanketstage.Coolingisslowestinthisregion.Whenthemetal tanks. There is always variation of flow rate and turbulence
has cooled sufficiently, the polymer film encapsulating the hot from top to bottom and across the tank. This means there may
metal ruptures and a nucleate boiling process results. The be significant variations of particulate contamination including
D6666 − 20
(A) New aqueous polymer quenchant solution.
(B) Used quenchant solution with oil contamination (see separated upper layer).
FIG. 4 Sample of Oil Contaminated Aqueous Polymer Quenchant
carbon from the heat treating process and metal scale. For excellent test) is to examine the appearance of an aqueous
uniform sampling, a number of sampling recommendations
polymer quenchant in a clear glass container, such as a bottle.
have been developed. A sample of an oil-contaminated fluid is illustrated in Fig. 4.
6.1.1 Sampling Recommendations:
(7) However, if the oil readily separates from the aqueous
6.1.1.1 Minimum Sampling Time—The circulation pumps
polymer quenchant solution (Fig. 4), it may be removed by
shall be in operation for at least 1 h prior to taking a sample
skimming. On the other hand, oil may form a milky-white
from the quench system.
emulsion which is not readily reclaimed by heat treaters.
6.1.1.2 Sampling Position—For each system, the well-
7.1.1.1 Other problems that are easy to identify visually
mixed sample shall be taken from the same position each time
includecarbonandsludgecontaminationwhichoftenresultsin
that system is sampled. The position in the tank where the
cracking problems. Metal scale contamination is often identi-
sample is taken shall be recorded.
fiable by its magnetic properties by placing a magnet on the
6.1.1.3 Sampling Values—If a sample is taken from a
outside of the bottle next to the scale and determining if the
sampling valve, then sufficient quenchant should be taken and
scale exhibits any attraction for the magnet. Carbon, sludge,
discarded to ensure that the sampling valve and associated
and scale may be removed from the quenchant by filtration or
piping has been flushed before the sample is taken.
centrifugation. Alternatively, the quenchant mixture may be
6.1.1.4 Effect of Quenchant Addition as Make-Up due to
allowed to settle, the quenchant solution pumped off, and the
Dragout—It is important to determine the quantity and fre-
separated solids then removed by shoveling. The amount of
quency of new quenchant additions, as large additions of new
insoluble suspended solids or tramp oils may be quantified by
quenchant solution will have an effect on the test results, in
a modification of Test Method D1796 where the aqueous
particular, the cooling curve. If a sample was taken just after a
quenchant is centrifuged without further dilution as described
large addition of new quenchant, this shall be taken into
in the method. The amount of tramp oil in the quenchant is
consideration when interpreting the cooling curve for this
determined from the insoluble liquid layer at the top of the
sample.
centrifuge tube and the volume of the insoluble sediment is
6.1.1.5 Sampling Containers—Samples shall be collected in
taken from the bottom of the centrifuge tube.
newcontainers.Undernocircumstancesshallusedbeverageor
7.1.2 Refractive Index, (Test Method D1747)—One of the
food containers be used because of the potential for fluid
contamination and leakage. most common methods of monitoring the concentration of
aqueous polymer quenchants formulated using poly(alkylene
7. Recommended Test Procedures
glycol) coploymers is refractive index. As Fig. 5 (7) shows,
there is a linear relationship between quenchant concentration
7.1 Performance-Related Physical and Chemical Proper-
and refractive index. The refractive index of the quenchant
ties:
7.1.1 Appearance—Contamination of aqueous polymer solution is determined using an Abbé refractometer (Test
Method D1747) equipped with a constant temperature bath.
quenchants by such fluids as hydraulic or quench oils may
result in a non-uniform quench with thermal gradients suffi- Although the refractive index could potentially be used at any
cient to cause cracking or increased distortion, or possible temperature within the control limits of the constant tempera-
staining,ofthemetalbeingquenched.Thesimplesttest(andan ture bath, typically either 40 ºC or 100 ºF is selected.
D6666 − 20
FIG. 5 Illustration of the Linear Relationship Between Refractive Index and Concentration
7.1.2.1 Although refractive index is a relatively simple and If the absolute value of the difference in delta is greater than
a rapid method for determination of polymer quenchant
6-8, the source of this difference, contamination or
concentration, it is not sensitive to low levels of polymer
degradation, should be determined.
degradation and it is often significantly affected by solution
7.1.5 Water Content (Test Methods D95 and D1744)—
contamination.
Aqueous polymer quenchants are composed of water, a water
soluble polymer and an additive package to provide corrosion
NOTE 1—Refractive index is typically unsuitable for aqueous polymer
quenchantsformulatedwithpolymerswithmolecularweightsgreaterthan
inhibition, foam control, and so forth.Therefore, determination
50 000 to 60 000 because the total concentration is relatively low. Small
of water content is necessary to establish the concentration of
changes in polymer concentration may result even from normal use which
the quenchant in a way that is relatively insensitive to polymer
impart significant process effects but the corresponding variation in
degradation.
refractive index may not be detectable.
NOTE 2—Although it is most desirable to use an Abbé refractometer
7.1.5.1 Water content may be determined by Karl Fisher
becauseofitssensitivity,thisisonlypracticalinalaboratoryenvironment.
analysis (Test Method D1744). The advantage of Karl Fisher
In the heat treating industry, for tankside monitoring and control, a
analysis is that it is a direct measure of water content, whereas
temperature-compensated handheld refractometer (similar to the one
illustrated in Fig. 6) is used. The hand-held refractometer is self-
refractive index and viscosity are both indirect measurements
compensated for temperatures between 60 ºF and 100 ºF. Although there
that are substantially affected by either contamination (refrac-
are various models available, the most common models provide arbitrary
tive index) or degradation (viscosity). In some cases, interfer-
refractiveindexreadingsinBrixunitsovera0ºto30ºrange.Typically,the
ences may arise with Karl Fisher analysis and an alternative
smallest scale that can be read directly is in divisions of 0.2º as shown in
Fig. 7. A concentration-refractive index curve obtained by a hand-held
procedure is necessary such as distillation from toluene or
refractometer is shown in Fig. 8. (7) Hand-held refractometers are
some other water-insoluble solvent (Test Method D95), or
available whose scale readings correlate directly to the concentration of
evaporation.
the polymer quenchant being used. This is particularly convenient for
industrial tank-side use. However, since refractive index varies with
7.1.6 pH Determination, (Test Method E70)—The perfor-
contamination (such as dissolved salts) that may accumulate from
mance of an aqueous polymer quench bath may be critically
evaporation of hard water, the actual quenchant concentration shall be
dependent on its pH. The pH of a quenchant solution may be
verified periodically by other methods, and appropriate correction factors
determined by Test Method E70. There are many excellent
applied.Inthiscase,therefractometerreadingmultipliedbythecorrection
factor equals actual concentration.
commercially available sources of pH meters and glass elec-
trodes. The choice of the instrument will be primarily affected
7.1.3 Viscosity, (Test Method D445 or D7042)—Aqueous
by the desired precision of measurement. Electrodes used for
polymer quenchant viscosity depends on the quenchant con-
pH measurement are designed for specific pH ranges and
centration and temperature as shown in Fig. 9. (7)
temperature; therefore, the solution pH and temperature shall
7.1.4 Comparison of Concentration by Refractive Index and
Viscosity—A useful procedure for monitoring variations in be considered when the electrodes are selected for use.
aqueous polymer quenchants, particularly poly(alkylene gly-
7.1.6.1 F
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6666 − 04 (Reapproved 2019) D6666 − 20
Standard Guide for
Evaluation of Aqueous Polymer Quenchants
This standard is issued under the fixed designation D6666; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This guide provides information, without specific limits, for selecting standard test methods for testing aqueous polymer
quenchants for initial qualification, determining quality, and the effect of aging.
1.2 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory requirements prior to use.
1.3 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D95 Test Method for Water in Petroleum Products and Bituminous Materials by Distillation
D445 Test Method for Kinematic Viscosity of Transparent and Opaque Liquids (and Calculation of Dynamic Viscosity)
D892 Test Method for Foaming Characteristics of Lubricating Oils
D1744 Test Method for Determination of Water in Liquid Petroleum Products by Karl Fischer Reagent (Withdrawn 2016)
D1747 Test Method for Refractive Index of Viscous Materials
D1796 Test Method for Water and Sediment in Fuel Oils by the Centrifuge Method (Laboratory Procedure)
D2624 Test Methods for Electrical Conductivity of Aviation and Distillate Fuels
D3519 Test Method for Foam in Aqueous Media (Blender Test) (Withdrawn 2013)
D3601 Test Method for Foam In Aqueous Media (Bottle Test) (Withdrawn 2013)
D3867 Test Methods for Nitrite-Nitrate in Water
D4327 Test Method for Anions in Water by Suppressed Ion Chromatography
D5296 Test Method for Molecular Weight Averages and Molecular Weight Distribution of Polystyrene by High Performance
Size-Exclusion Chromatography
D6482 Test Method for Determination of Cooling Characteristics of Aqueous Polymer Quenchants by Cooling Curve Analysis
with Agitation (Tensi Method)
D6549 Test Method for Determination of Cooling Characteristics of Quenchants by Cooling Curve Analysis with Agitation
(Drayton Unit)
D7042 Test Method for Dynamic Viscosity and Density of Liquids by Stabinger Viscometer (and the Calculation of Kinematic
Viscosity)
E70 Test Method for pH of Aqueous Solutions With the Glass Electrode
E979 Practice for Evaluation of Antimicrobial Agents as Preservatives for Invert Emulsion and Other Water Containing
Hydraulic Fluids
E2275 Practice for Evaluating Water-Miscible Metalworking Fluid Bioresistance and Antimicrobial Pesticide Performance
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
This guide is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.L0.06 on Non-Lubricating Process Fluids.
Current edition approved May 1, 2019May 1, 2020. Published July 2019May 2020. Originally approved in 2001. Last previous edition approved in 20142019 as
D6666 – 04 (2014).(2019). DOI: 10.1520/D6666-04R19.10.1520/D6666-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6666 − 20
FIG. 1 Cooling Mechanisms of the Quenching Process
3.1.1 austenite, n—solid solution of one or more elements in face-centered cubic iron (gamma iron) and unless otherwise
designated, the solute is generally assumed to be carbon (1).
3.1.2 austenitizing, n—forming austenite by heating a ferrous alloy into the transformation range (partial austenitizing) or above
the transformation range (complete austenitizing). When used without qualification, the term implies complete austenitizing (1).
3.1.3 aqueous polymer quenchant, n—a solution containing water, and one or more water-soluble polymers including
poly(alkylene glycol), poly(vinyl pyrrolidone), poly(sodium acrylate), and poly(ethyl oxazoline) (2, 3) and additives for corrosion
and foam control, if needed.
3.1.4 biodegradation, n—the process by which a substrate is converted by biological, usually microbiological, agents into
simple, environmentally acceptable derivatives. (4)
3.1.5 biodeterioration, n—loss of product quality and performance and could be regarded as the initial stages of biodegradation
(see 3.1.4), but in the wrong place at the wrong time, that is when the product is stored or in use. (4)
3.1.6 convective cooling, n—after continued cooling, and the interfacial temperature between the cooling metal and the aqueous
polymer quenchant is less than the boiling point of the water in the quenchant solution at which point cooling occurs by a
convective cooling process. For convective cooling, fluid motion is due to density differences and the action of gravity and includes
both natural motion and forced circulation (1, 5). This process is illustrated in Fig. 1.
3.1.7 cooling curve, n—a graphical representation of the cooling time (t)—temperature (T) response of the probe such as that
shown in Fig. 1. (5)
3.1.8 cooling curve analysis, n—the process of quantifying the cooling characteristics of a quenchant medium based on the
temperature versus time profile obtained by cooling a preheated metal probe assembly (see Fig. 2) under specified conditions which
include: probe alloy and dimensions, probe and bath temperature, agitation rate, and aqueous polymer quenchant concentration.
3.1.9 cooling rate curve, n—obtained by calculating the first derivative (dT/dt) of the cooling time-temperature curve as
illustrated in Fig. 1. (5)
3.1.10 dragout, n—solution carried out of a bath on the metal being quenched and associated handling equipment. (1)
3.1.11 full-film boiling, n—upon initial immersion of hot steel into a quenchant solution, a vapor blanket surrounds the metal
surface resulting in full-film boiling as shown in Fig. 1. (5)
3.1.12 nucleate boiling, n—when the vapor blanket surrounding the hot metal collapses and a nucleate boiling process occurs
as illustrated in Fig. 1. (5)
3.1.13 quenchant medium, n—any liquid or gas, or mixture, used to control the cooling of a metal to facilitate the formation
of the desired microstructure and properties. (1)
3.1.14 quench severity, n—the ability of a quenchant medium to extract heat from hot metal. (6)
The boldface numbers in parentheses refer to the list of references at the end of this standard.
D6666 − 20
NOTE 1—From Wolfson Engineering Group Specification, available from Wolfson Heat Treatment Centre, Aston University, Aston Triangle,
Birmingham B4 7ET, England, 1980.
FIG. 2 Schematic Illustration of the Probe Details and Probe Assembly
3.1.15 transformation temperatures, n—characteristic temperatures that are important in the formation of martensitic
microstructure of steel including: A —equilibrium austenitization phase change temperature; M —temperature at which
e1 S
transformation of austenite to martensite starts during cooling and M —temperature at which transformation of austenite to
f
martensite is completed during cooling. (1)
4. Significance and Use
4.1 The significance and use of each test method will depend on the system in use and the purpose of the test method listed under
Section 7. Use the most recent editions of the test methods.
5. Quenching Process
5.1 Aqueous Polymer Quenchant Cooling Mechanisms—Upon initial immersion of a heated metal into a solution of an aqueous
polymer quenchant, an insulating polymer film, which controls the heat transfer rate from the hot metal into the cooler quenchant
solution, forms around the hot metal which is separated by a vapor film (Fig. 3) (7) for the quenching process in a poly(alkylene
glycol) quenchant. The overall heat transfer mediating properties of the film are dependent on both the film thickness (a function
of polymer concentration) and interfacial film viscosity (a function of polymer type and bath temperature). The timing of film
formation and subsequent film rupture and removal is dependent on the film strength of the polymer, agitation (both direction and
mass flow), and turbulence of the polymer solution surrounding the cooling metal.
5.1.1 The cooling process that occurs upon initial immersion of the hot metal into the aqueous polymer quenchant is full-film
boiling. This is frequently referred to as the vapor blanket stage. Cooling is slowest in this region. When the metal has cooled
sufficiently, the polymer film encapsulating the hot metal ruptures and a nucleate boiling process results. The temperature at the
transition from full-film boiling to nucleate boiling is called the Leidenfrost temperature. Cooling is fastest in this region. When
the surface temperature of the cooling metal is less than the boiling temperature of water, convective cooling results. All three
cooling mechanisms are superimposed on a cooling curve and illustrated in Fig. 3. (7)
6. Sampling
6.1 Sampling—Flow is never uniform in agitated quench tanks. There is always variation of flow rate and turbulence from top
to bottom and across the tank. This means there may be significant variations of particulate contamination including carbon from
the heat treating process and metal scale. For uniform sampling, a number of sampling recommendations have been developed.
6.1.1 Sampling Recommendations:
6.1.1.1 Minimum Sampling Time—The circulation pumps shall be in operation for at least 1 h prior to taking a sample from the
quench system.
6.1.1.2 Sampling Position—For each system, the well-mixed sample shall be taken from the same position each time that system
is sampled. The position in the tank where the sample is taken shall be recorded.
D6666 − 20
FIG. 3 Illustration of the Three Phases of Cooling
6.1.1.3 Sampling Values—If a sample is taken from a sampling valve, then sufficient quenchant should be taken and discarded
to ensure that the sampling valve and associated piping has been flushed before the sample is taken.
6.1.1.4 Effect of Quenchant Addition as Make-Up due to Dragout—It is important to determine the quantity and frequency of
new quenchant additions, as large additions of new quenchant solution will have an effect on the test results, in particular, the
cooling curve. If a sample was taken just after a large addition of new quenchant, this shall be taken into consideration when
interpreting the cooling curve for this sample.
6.1.1.5 Sampling Containers—Samples shall be collected in new containers. Under no circumstances shall used beverage or
food containers be used because of the potential for fluid contamination and leakage.
7. Recommended Test Procedures
7.1 Performance-Related Physical and Chemical Properties:
7.1.1 Appearance—Contamination of aqueous polymer quenchants by such fluids as hydraulic or quench oils may result in a
non-uniform quench with thermal gradients sufficient to cause cracking or increased distortion, or possible staining, of the metal
being quenched. The simplest test (and an excellent test) is to examine the appearance of an aqueous polymer quenchant in a clear
glass container, such as a bottle. A sample of an oil-contaminated fluid is illustrated in Fig. 4. (7) However, if the oil readily
separates from the aqueous polymer quenchant solution (Fig. 4), it may be removed by skimming. On the other hand, oil may form
a milky-white emulsion which is not readily reclaimed by heat treaters.
7.1.1.1 Other problems that are easy to identify visually include carbon and sludge contamination which often results in
cracking problems. Metal scale contamination is often identifiable by its magnetic properties by placing a magnet on the outside
of the bottle next to the scale and determining if the scale exhibits any attraction for the magnet. Carbon, sludge, and scale may
be removed from the quenchant by filtration or centrifugation. Alternatively, the quenchant mixture may be allowed to settle, the
quenchant solution pumped off, and the separated solids then removed by shoveling. The amount of insoluble suspended solids
or tramp oils may be quantified by a modification of Test Method D1796 where the aqueous quenchant is centrifuged without
further dilution as described in the method. The amount of tramp oil in the quenchant is determined from the insoluble liquid layer
at the top of the centrifuge tube and the volume of the insoluble sediment is taken from the bottom of the centrifuge tube.
7.1.2 Refractive Index, (Test Method D1747)—One of the most common methods of monitoring the concentration of aqueous
polymer quenchants formulated using poly(alkylene glycol) coploymers is refractive index. As Fig. 5 (7) shows, there is a linear
relationship between quenchant concentration and refractive index. The refractive index of the quenchant solution is determined
using an Abbé refractometer (Test Method D1747) equipped with a constant temperature bath. Although the refractive index could
potentially be used at any temperature within the control limits of the constant temperature bath, typically either 40 ºC or 100 ºF
is selected.
7.1.2.1 Although refractive index is a relatively simple and a rapid method for determination of polymer quenchant
concentration, it is not sensitive to low levels of polymer degradation and it is often significantly affected by solution
contamination.
NOTE 1—Refractive index is typically unsuitable for aqueous polymer quenchants formulated with polymers with molecular weights greater than 50
000 to 60 000 because the total concentration is relatively low. Small changes in polymer concentration may result even from normal use which impart
D6666 − 20
(A) New aqueous polymer quenchant solution.
(B) Used quenchant solution with oil contamination (see separated upper layer).
FIG. 4 Sample of Oil Contaminated Aqueous Polymer Quenchant
FIG. 5 Illustration of the Linear Relationship Between Refractive Index and Concentration
significant process effects but the corresponding variation in refractive index may not be detectable.
NOTE 2—Although it is most desirable to use an Abbé refractometer because of its sensitivity, this is only practical in a laboratory environment. In
the heat treating industry, for tankside monitoring and control, a temperature-compensated handheld refractometer (similar to the one illustrated in Fig.
6) is used. The hand-held refractometer is self-compensated for temperatures between 60 ºF and 100 ºF. Although there are various models available, the
most common models provide arbitrary refractive index readings in Brix units over a 0º to 30º range. Typically, the smallest scale that can be read directly
is in divisions of 0.2º as shown in Fig. 7. A concentration-refractive index curve obtained by a hand-held refractometer is shown in Fig. 8. (7) Hand-held
refractometers are available whose scale readings correlate directly to the concentration of the polymer quenchant being used. This is particularly
convenient for industrial tank-side use. However, since refractive index varies with contamination (such as dissolved salts) that may accumulate from
evaporation of hard water, the actual quenchant concentration shall be verified periodically by other methods, and appropriate correction factors applied.
In this case, the refractometer reading multiplied by the correction factor equals actual concentration.
7.1.3 Viscosity, (Test Method D445 or D7042)—Aqueous polymer quenchant viscosity depends on the quenchant concentration
and temperature as shown in Fig. 9. (7) Viscosity is readily determined using a Cannon-Fenske tube (see Fig. 10), stopwatch and
constant temperature bath as described in Test Method D445.
7.1.4 Comparison of Concentration by Refractive Index and Viscosity—A useful procedure for monitoring variations in aqueous
polymer quenchants, particularly poly(alkylene glycol) quenchants, is to compare the difference (delta) in the quenchant
concentration value obtained by refractive index (C ) and viscosity (C ). (8)
R V
Δ5 C 2 C (1)
R V
D6666 − 20
(A) Application of the aqueous polymer quenchant to the refractometer.
(B) Visual reading of the refractometer scale to determine refractance value.
FIG. 6 Typical Hand-Held Refractometer
FIG. 7 Illustration of the Degrees Brix Refractive Index Scale Used for the Hand-Held Refractometer
If the absolute value of the difference in delta is greater than 6-8, the source of this difference, contamination or degradation,
should be determined.
7.1.5 Water Content (Test Methods D95 and D1744)—Aqueous polymer quenchants are composed of water, a water soluble
polymer and an additive package to provide corrosion inhibition, foam control, and so forth. Therefore, determination of water
content is necessary to establish the concentration of the quenchant in a way that is relatively insensitive to polymer degradation.
D6666 − 20
FIG. 8 Typical Refractive Index (Degrees Brix) Versus Quenchant Concentration Relationship
FIG. 9 Quenchant Viscosity as a Function of Concentration and
Temperature
7.1.5.1 Water content may be determined by Karl Fisher analysis (Test Method D1744). The advantage of Karl Fisher analysis
is that it is a direct measure of water content, whereas refractive index and viscosity are both indirect measurements that are
substantially affected by either cont
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...