ASTM F3259-17
(Guide)Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds
Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds
General Information
- Status
- Published
- Publication Date
- 30-Apr-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 01-Aug-2020
- Effective Date
- 15-Nov-2018
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Jun-2009
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Nov-2004
- Effective Date
- 01-May-2017
- Effective Date
- 01-May-2017
Overview
ASTM F3259-17: Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds is an international standard developed by ASTM International. This guide provides a comprehensive framework for the micro-computed tomography (microCT) imaging and analysis of tissue engineered scaffolds, particularly porous scaffolds designed for tissue repair and regeneration. The document outlines best practices for sample preparation, imaging parameter selection, post-processing, and data interpretation for various scaffold materials including synthetic polymers, natural substances, and mammalian or human-derived materials.
MicroCT offers a non-destructive, three-dimensional imaging approach ideal for characterizing the microarchitecture and tissue ingrowth within biomaterial scaffolds, making it a valuable tool in tissue engineering research and development.
Key Topics
- Application Scope
- Suitable for in vitro and ex vivo analysis of porous scaffolds but does not provide detailed protocols for direct in vivo imaging.
- Applicable to a range of scaffold materials such as synthetic polymers (e.g., polyesters), natural materials (e.g., calcium phosphates), and matrixes derived from mammalian or human sources (e.g., demineralized bone).
- Imaging Considerations
- Guidance is given for optimal sample preparation, including considerations for scaffold dimension, hydration, and alignment to ensure high-quality image acquisition.
- Techniques for selecting voxel size suitable for the resolution of microstructural features like pore size and strut thickness.
- Image Acquisition and Quality
- Recommendations for adjusting microCT scanner parameters (energy, intensity, voxel size, scan time) according to material radiodensity and scaffold features.
- Emphasis on using consistent parameters across all specimens for data comparability, with additional notes on the use of phantoms and flat field corrections for calibration.
- Artifact Management
- Discussion of common imaging artifacts such as beam-hardening, motion, metal, and partial volume effects, with practical advice for mitigation.
Applications
- Quality Assurance and Product Development
- Utilized to quantify microstructural features of scaffolds (such as strut thickness and pore connectivity), assess manufacturing consistency, and compare as-designed versus as-produced properties.
- Ideal for monitoring scaffold degradation, both in vitro and ex vivo after implantation in animal models.
- Assessment of Tissue Ingrowth
- Supports quantitative measurement of tissue ingrowth within scaffolds (most useful for high-radiodensity tissues like bone), critical for evaluating scaffold performance and biocompatibility.
- Design Optimization
- Aids in evaluating the impact of scaffold architecture on cell adhesion, proliferation, and phenotypic differentiation, which are key to successful tissue engineering outcomes.
Related Standards
- ASTM F2450 – Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products. Offers a complementary methodology for microstructure analysis, especially for polymeric scaffolds.
- ASTM F2603 – Guide for Interpreting Images of Polymeric Tissue Scaffolds. Provides detailed instructions on interpreting imaging results from tissue scaffold microCT analyses.
Summary of Benefits
- Non-destructive, repeatable assessment of scaffold architecture, enabling longitudinal studies and continual quality control without damaging the specimen.
- Enhanced 3D visualization of both scaffold structure and tissue integration, overcoming the limitations of traditional 2D imaging methodologies.
- Standardization of methods for sample preparation, imaging, and data analysis, promoting reproducibility and regulatory compliance in tissue engineering research.
Conclusion
By following ASTM F3259-17, researchers and developers can ensure robust, comparable, and high-quality microCT assessments of tissue engineered scaffolds. This standard supports advancements in scaffold-based medical devices and regenerative therapies by providing clear guidelines for consistent imaging and analysis practices. For optimal results, users should also consider complementary ASTM standards related to polymeric scaffolds and image interpretation.
Buy Documents
ASTM F3259-17 - Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3259-17 is a guide published by ASTM International. Its full title is "Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds". This standard covers: Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds
Standard Guide for Micro-computed Tomography of Tissue Engineered Scaffolds
ASTM F3259-17 is classified under the following ICS (International Classification for Standards) categories: 11.100.99 - Other standards related to laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3259-17 has the following relationships with other standards: It is inter standard links to ASTM F2603-06(2020), ASTM F2450-18, ASTM F2603-06(2012), ASTM F2450-10, ASTM F2450-09, ASTM F2603-06, ASTM F2450-04, ASTM F2952-22, ASTM F3510-21. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3259-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3259 − 17
Standard Guide for
Micro-computed Tomography of Tissue Engineered
Scaffolds
This standard is issued under the fixed designation F3259; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.6 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 Thisguideisaresourceforconductingmicro-computed
responsibility of the user of this standard to establish appro-
tomography (microCT) imaging and analysis of porous scaf-
priate safety and health practices and determine the applica-
folds for tissue engineering applications. Considerations are
bility of regulatory limitations prior to use.
provided for sample preparation, image acquisition parameter
1.7 This international standard was developed in accor-
selection, post-processing, and data interpretation.
dance with internationally recognized principles on standard-
1.2 The information in this guide is intended to be appli-
ization established in the Decision on Principles for the
cabletoproductsthatincludeaporousscaffoldcomponentand
Development of International Standards, Guides and Recom-
are designed for tissue engineering repair strategies. The
mendations issued by the World Trade Organization Technical
scaffolds may be fabricated from synthetic polymers (e.g.,
Barriers to Trade (TBT) Committee.
absorbable polyesters) or natural materials (e.g., calcium
phosphates), mammalian or human derived materials (e.g.,
2. Referenced Documents
demineralized bone) or combinations of these. While some
2.1 ASTM Standards:
considerationsareprovidedforimagingofmaterialsthatareof
F2450Guide for Assessing Microstructure of Polymeric
moderate to high radiodensity, specific guidelines are not
Scaffolds for Use in Tissue-Engineered Medical Products
provided for imaging metallic scaffolds.
F2603Guide for Interpreting Images of Polymeric Tissue
1.3 Applicability of the guidelines herein will depend on
Scaffolds
scaffold material type and the user’s application (e.g., experi-
mental design, as manufactured characterization) as appropri-
3. Terminology
ate.
3.1 Definitions of Terms Specific to This Standard:
1.4 The guidelines for microCT discussed herein are most
3.1.1 microarchitecture, n—the set of structural features of
suitable for specimen scanning in vitro. Specific guidelines
an object defined at the microscale.
relevant to direct in vivo imaging of scaffolds are not included
3.1.2 volume of interest (VOI), n—a 3D sub-volume inside
because the imaging parameters will be dependent on the
an image that contains the features to be analyzed.
implantation site, animal size, breathing etc. In addition,
consensus recommendations for in vivo imaging are provided
4. Significance and Use
in Bouxsein et al 2010 (1). While the specific imaging
4.1 X-ray microcomputed tomography (microCT) is a non-
parameters and processing recommendations discussed in
destructivethree-dimensionalimagingmethodthatcanbeused
Bouxsein et al are specific to bone imaging, many of the
to reconstruct the microarchitecture of a tissue engineered
considerations and precautions are also applicable for in vivo
medical product (TEMP) scaffold that may or may not contain
scaffold imaging.
ingrowntissue.MicroCTwasfirstdevelopedtostudyceramics
1.5 The values stated in SI units are to be regarded as
for the auto-industry and adapted for bone morphology at the
standard. No other units of measurement are included in this
microscale (Feldkamp et al 1989) (2). More recently, the
standard.
imaging method has been adapted for in vivo applications and
studies of multiple natural and synthetic materials.
This guide is under the jurisdiction ofASTM Committee F04 on Medical and
Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.42 on Biomaterials and Biomolecules for TEMPs.
Current edition approved May 1, 2017. Published September 2017. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/F3259-17. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3259 − 17
4.2 Alternate characterization methods for assessing scaf- nique as a way to measure numerous architectural parameters
fold microarchitecture and tissue ingrowth are limited by their quantitatively and to track them at progressive time points.
two dimensional nature (e.g., microscopy) and low depth of Specific indices and types of architectural indices that can be
penetration (e.g., optical coherence tomography), even though tracked for scaffolds are discussed in Section 10.
their resolution may be increased over microCT. However,
5.2 The objective of a microCT assessment of tissue engi-
microCT is an ideal imaging choice for studying scaffold
neered scaffolds is dependent on numerous factors and con-
microarchitecture and tissue ingrowth because it is non-
trolled by the investigators. Some considerations for defining
destructive, provides scaffold assessments based on direct
the objective of the study include the need for point-in-time vs
measurements rather than stereological methods, offers the
longitudinal assessments, quality assurance, and monitoring
ability to perform longitudinal imaging, and can be conducted
tissuegrowthvsscaffolddegradation.Thisguideissuitablefor
at length scales relevant to cells and cell attachment (i.e., 1
the following experimental objectives when performing mi-
micron to hundreds of microns).
croCT assessment of a tissue engineered scaffold:
4.3 The microarchitecture of tissue engineered scaffolds
5.2.1 Quantification of microstructural features (e.g., strut
plays a critical role in providing structural support and/or thickness) in the scaffold. This type of assessment may be
facilitating cell adhesion, proliferation, and phenotype as well
performed as part of a quality assurance characterization, (e.g.
as matrix deposition. These parameters are essential elements
to test the degree of agreement between design and
of the tissue engineering strategy. During scaffold degradation,
production), or to characterize the microstructural features.
either in vitro or in vivo, changes to the microarchitecture
5.2.1.1 These analyses are most typically performed on
continue to influence the eventual tissue repair.Therefore, it is
scaffoldsthathavenotbeenexposedtoan in vivoenvironment.
critical to characterize the microarchitecture over time. Such
5.2.1.2 The same set of analyses can be applied to scaffolds
characterizationcanaidtheoptimaldesignofTEMPscaffolds,
that have undergone simulated degradation in vitro. MicroCT
establishmentofmanufacturingconsistency,andmonitoringof
providesasimpleandnon-destructivewaytotrackmicrostruc-
scaffold structure and/or tissue response.
tural and physical changes to the scaffold during degradation.
Since the technique provides a three-dimensional image of the
4.4 This guide provides a compendium of information
scaffold, it can be used to determine potential areas of
related to the use of microCT for the structural assessment of
non-uniform degradation or other structural features.
scaffold microarchitecture and tissue ingrowth. While the
5.2.1.3 While assessment of these indices is most com-
microarchitecture of tissue engineered scaffolds, as well as
monly completed following all manufacturing processes, simi-
changes to it over time, can be assessed using multiple
lar considerations would apply if assessing the scaffold at
methods, (e.g., such as those described in Guide F2450), this
interimpointsinthemanufacturingprocessorafterexposureto
guide focuses on unique considerations for conducting the
shelf life aging.
microCT analyses.
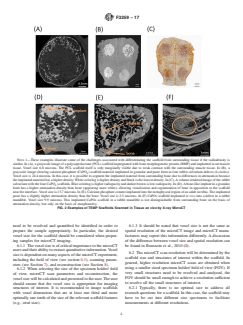
5.2.1.4 Examples of scaffolds imaged with microCT appear
4.5 The user of this guide is provided with considerations
in Fig. 1 to illustrate the type of information that may be
for each aspect of a complete microCTstudy including sample
gathered and the heterogeneity of visual representation for
preparation, image acquisition, assessing image quality and
TEMP scaffolds.
artifacts, post-processing, and image interpretation based on
5.2.2 Ex vivo characterization of changes to the scaffold
the specific application.
microarchitecturefollowingdegradationandtissueingrowth in
4.6 This standard provides imaging and analysis consider-
vivo. This type of assessment includes removal of the scaffold
ations for the following broad types of applications: (a)
from the animal model prior to imaging. This characterization
scaffold microarchitecture analysis in vitro either before or
would provide the user with information on geometric altera-
after different stages of degradation, (b) ex vivo analysis of
tions to the structure of the scaffold over time following
scaffold microarchitecture following partial degradation in an
implantation in an animal model.
in vivo animal model, (c) deriving microarchitectural informa-
5.2.2.1 Performing microCT of the scaffold after implanta-
tion when multiple materials are used in the scaffold, and (d)
tioninananimalmodelhasuniquechallengesasillustratedby
differentiating between scaffold microarchitectural changes
the images in Fig. 2.
and new tissue ingrowth.
5.2.2.2 Specific considerations for each aspect of perform-
4.7 The information provided in this standard guide is not ingamicroCTstudyofascaffoldwhileimplantedinan in vivo
intendedasatestmethodformicroCTcharacterizationbecause animalmodel,however,arebeyondthescopeofthisguideand
the user’s specific application and experimental design will are covered in Bouxsein et al 2010 (1).
significantly influence the imaging methodology and interpre-
5.2.3 Ex vivo characterization of tissue ingrowth. In
tation. addition, to understanding how the scaffold degrades and is
altered following implantation, microCT can be used to quan-
5. MicroCT Characterization Objectives
tify the extent of tissue ingrowth and provide some basic
5.1 A significant amount of tissue engineering research is information on the type of tissue regenerated.
focused on developing optimal scaffold microstructure to 5.2.3.1 While in theory, using microCT to quantify the
facilitatetissueingrowth,modulatecellphenotype,andcontrol amount of tissue ingrowth in the presence of a tissue engi-
the repair response. Due to the non-destructive nature of neered scaffold is feasible, it is limited by the ability of the
microCT, many investigators have utilized this imaging tech- microCT to differentiate the radiodensity of scaffold material
F3259 − 17
FIG. 1 Examples of TEMP Scaffolds Scanned Alone in vitro by X-ray MicroCT
as compared to tissue. In practice, this has been most readily 5.4.1 Blank scaffolds that are stable and do not change their
achieved by quantifying the production (Peyrin 2011) (3) of architecture (i.e., without any cells or degradation).
bone since this tissue type has a much higher density than that
5.4.2 For applications where the tissue engineered scaffold
of many synthetic absorbable polymeric scaffolds.
of interest is designed from multiple materials, the microCT
experiment may necessitate imaging of different scaffolds,
5.3 MicroCT characterizations of tissue engineered scaf-
each manufactured with only one of the pure materials. These
folds may be completed on structures that are fabricated from
additional images may be used to aid threshold selection.
one or multiple materials. The ability to differentiate multiple
materials within a scaffold will be dependent on the composi-
6. Sample Preparation
tion of those materials and their radiodensity.
5.4 Someapplicationsmaynecessitatedesigningtheexperi- 6.1 Scaffold dimensions and/or design are important when
mentinordertoincludevarioustypesofcontrols.Examplesof preparing TEMPs scaffolds for microCT imaging. During
controls which may be used to facilitate microCT image sample preparation, it is recommended that the key features of
analysis and/or interpretation may include the following: the scaffold (e.g. pore size, strut thickness, density, etc) which
F3259 − 17
NOTE 1—These examples illustrate some of the challenges associated with differentiating the scaffold from surrounding tissue if the radiodensity is
similar.In(A),agrayscaleimageofapolycaprolactone(PCL)scaffoldimpregnatedwithbonemorphogeneticprotein(BMP)andimplantedinratmuscle
tissue. Voxel size 6.8 microns. The PCL scaffold itself is only marginally visible due to weak contrast with the surrounding muscle tissue. In (B), a
grayscale image showing calcium phosphate (CaPO ) scaffold material implanted in granular and paste form in four rabbit calvarium defects (4 circles).
Voxel size is 24.4 microns. In this case, it is possible to segment the implanted material from surrounding bone due to differences in attenuation because
theimplantedmaterialhasahigherdensity.Whitecoloringishigherdensityandblackisthelowestdensity.In(C),Avolumerenderedimageoftherabbit
calvariumwiththefourCaPO scaffolds.Bluecoloringishigherradiopacityanddarkerbrownislowradiopacity.In(D),Abone-likeimplantinagranular
form has a higher attenuation density than bone (appearing more white), allowing visualisation and segmentation of bone in apposition to the scaffold
neartheinterface.Voxelsizeis13.7microns.In(E),Calciumphosphatecementimplantedintothemetaphysealregionofanadultrattibia.Theimplanted
paste has a slightly higher attenuation density than the bone. Voxel size is 2.5 microns. In (F) CaPO scaffold implanted in vivo into a defect in a rabbit
mandible. Voxel size 9.9 microns. This implanted CaPO scaffold in a rabbit mandible is not distinguishable from surrounding bone on the basis of
attenuation density, but only on the basis of morphometry.
FIG. 2 Examples of TEMP Scaffolds Scanned in Tissue ex vivo by X-ray MicroCT
need to be resolved and quantified be identified in order to 6.1.3 It should be noted that voxel size is not the same as
prepare the sample appropriately. In particular, the desired spatial resolution of the microCT image and microCT manu-
voxel size for the scaffold should be considered when prepar-
facturers may report this information differently. A discussion
ing samples for microCT imaging.
of the difference between voxel size and spatial resolution can
6.1.1 ThevoxelsizeisofcriticalimportancetothemicroCT
be found in Bousxein et al., 2010 (1).
usersandtheirabilitytoextractquantitativeinformation.Voxel
6.2 The microCT scan resolution will be determined by the
size is dependent on many aspects of the microCTexperiment,
scaffold size and structures of interest within the scaffold. In
including the field of view (see section 6.3), scanning param-
general, higher resolution microCT scans are obtained when
eters (see Section 7), and reconstruction (see Section 8).
using a smaller sized specimen holder/ field of view (FOV). If
6.1.2 When selecting the size of the specimen holder/ field
very small structures need to be resolved and analyzed, the
of view, microCT scan parameters and reconstruction, the
FOV should be small enough to achieve a resolution sufficient
voxelsizewillbecalculatedandpresentedtotheuser.Theuser
to resolve all the small structures of interest.
should ensure that the voxel size is appropriate for imaging
6.2.1 Typically, there is no optimal size to address all
structures of interest. It is recommended to image scaffolds
with voxel dimensions that are at least one third and more research questions for a scaffold. In this case, the scaffold may
have to be cut into different size specimens to facilitate
optimally one tenth of the size of the relevant scaffold features
(e.g., strut size). measurements at different resolutions.
F3259 − 17
6.2.2 Visual inspection and binary segmentation of struc- use of contrast agents or particles of high radiodensity may
tures for analysis provide a good indication of whether result in thicker struts due to swelling, accumulation, and/or
sufficientresolution,homogeneityandimagequalityhavebeen partial volume artifacts. In these cases, validation of the
attained in a microCT scan. technique is recommended.
6.2.3 Ideally, the scaffold size, specimen holder size, and
6.6.1 An example of the effect that a contrast agent may
therefore, the FOV should be consistent for all scaffolds
have on scaffold visualization with microCT is shown in
imaged within a study.
Appendix X1, Fig. X1.1.
6.3 Some small VOIs taken from a larger scaffold with a
6.7 For longitudinal scans of the same specimen, e.g.,
porous cellular structure may not be a representative selection
imaging a process or changes to the scaffold, maintaining
due to differences in the scale of features.Aparameter termed
consistentspecimenalignmentbetweenscansisrecommended,
theRepresentativeVolumeElement(RVE)hasbeendeveloped
particularly if spatial information is compared across multiple
to address this issue (Bachmat et al 1987) (4) and has
scans (e.g. bone ingrowth).
demonstrated some utility in the microCT analysis of snow
6.7.1 Registration markers may also aid in maintaining
(Srivastava et al 2010) (5). If a material with cellular structure
consistent alignment.
is essentially homogeneous, then the VOI can be selected
6.7.2 In addition, software registration can be used to align
randomly within the material.
longitudinal scans.
6.3.1 If the VOI is sufficiently large, that is, larger than the
RVE, then measured parameters of microarchitecture and 6.8 Additional considerations may be needed for sample
porosity will be very similar wherever the VOI is situated. preparationapplicationswherethescaffoldhasbeenimplanted
6.3.2 However if the VOI size falls below the RVE, then into an animal model and subsequently explanted for microCT
stochastic differences in the measured structural parameters analyses.
increase between VOIs at different locations, since the VOI
6.8.1 If possible, the scaffold with repair tissue should
becomestoosmalltoadequatelysamplethecellularstructures.
remain hydrated for the microCT scan in order to accurately
measure scaffold and tissue features. Hydration of the scaffold
6.4 It is critical to secure scaffolds firmly in position to
duringmicroCTimagingmayalsobenecessaryforsubsequent
prevent motion artifact (i.e. relative movement between the
analyses (e.g. mechanical testing, histology).
specimen holder and scaffold) during the scan. Due to the low
6.8.2 Some of the external tissue may need to be trimmed
radiopacity of many TEMPs scaffolds, it is recommended to
awayfromtherepairsitepriortoimagingtoaidindetermining
uselowdensityfoam(e.g.,polystyreneorpolyurethanefoams)
physical boundaries of the construct and accurately assessing
or another low-attenuating material to secure the scaffold
the effects that occur within the scaffold.
within holder. The securement material should have a lower
radiopacity than that of the scaffold itself.
6.8.3 Ifpossible,sampleorientationshouldremainthesame
6.4.1 For very low radiodense scaffolds, it may not be
between samples to allow for ease of segmentation of the VOI
feasible to use a lower radiopacity material. In those instances,
during analysis.
a higher radiodense material (e.g. spacer) above and/or below
6.8.4 Landmarks from radiopaque material (staples, screws,
the scaffold may provide adequate compression to secure the
etc) may be used and/or added to help distinguish boundaries
scaffold and prevent motion artifact.
between materials with similar radiopacities.
6.4.2 Other techniques to consider for securing specimens
include using 3D printed spacers/cartridges of specific
7. Image Acquisition
geometries, putty or solvent resistant, double sided tape.
7.1 MicroCTs are complex imaging systems that require a
6.5 In general, the voxel intensity of the scanning medium
careful selection of the scanning parameters.These parameters
should ideally be significantly lower than that of the scaffold
depend on the sample material and on the microCT system
and homogeneous in spatial density distribution. This ensures
used. It is therefore not possible to fix a protocol that is valid
that scaffold features can be differentiated and resolved.
forallscaffoldtypesorapplications.Solidunderstandingofthe
6.5.1 Scanning in air provides the highest contrast between
physics behind microCT is essential to acquire the best
scanning medium and scaffold.
possibleimages.However,foragivenapplicationandmaterial
6.5.2 For scaffolds where hydration is important, other
type,theoptimalparametersmustbedeterminedonlyonceand
media may be used (e.g., deionized water, saline, phosphate
then they can be used for all studies using the same scaffold
buffered saline, ethanol, and neutral buffered formalin); how-
materialandapplicationtype.InTable1,themostrelevantand
ever it is important to verify whether X-ray attenuation or
critical scanning parameters of tissue engineered scaffolds
morphometric properties are significantly affected by the
using X-ray tube based microCT systems are summarized and
medium.
their effect on scaffold image quality is highlighted.
6.5.3 The same scanning medium should be used for all
7.1.1 To set the energy to an appropriate value, it is helpful
samples in a microCT study for consistent, quantitative com-
to know the chemical composition of the scaffold material, as
parisons.
thisdeterminestheattenuationoftheX-raybeam.Ingeneralit
6.6 In situations where contrast agents are needed to en- can be said that the higher the atomic number, the greater the
hance images of scaffolds with very low radiodensity, care attenuation and thus, the higher the energy needs to be to
should be taken to prevent image artifacts. For example, the penetrate the material.
F3259 − 17
A
TABLE 1 General Description of Parameters That Define a Scan
Parameter Description Effects
Energy The energy that can be set in microCT systems refers to The higher the energy of the photons, the better the
the peak voltage applied between the cathode and the penetration through dense materials. However, materials
anode of the X-ray tube to accelerate the electrons. This with similar attenuation coefficients can be separated
peak energy and the anode material determine the more effectively at lower energies. Therefore, the energy
photon spectrum and the maximal energy of the chosen should be as low as possible but also high
converted photons. However, due to the spread of enough to ensure that the photons can penetrate all
energies emitted from the source, the mean photon materials in the specimen. When tissue engineered
energy is typically about 1/3rd of the peak energy. scaffolds are fabricated from low density absorbable
polymers, the optimal energies used for imaging
scaffolds alone are typically not consistent with those
used to image ceramics or metals. However, higher
energies may be necessary if calcified tissue, such as
bone, has ingrown into the scaffold.
Filter Filters are typically metal plates that can be inserted in Beam hardening artifacts (see Section 8) can be
the photon beam to modify the energy spectrum. Filtering mitigated by narrower photon spectrums. Narrow photon
narrows the photon energy spectrum and reduces the spectrums also aid density resolution. To separate
overall intensity. different material types with similar attenuation values
within a scaffold it can thus be beneficial to narrow the
photon spectrum by using a filter. However, the filters
reduce the overall number of photons, which
necessitates an increase in measurement time in order
to obtain a comparable signal-to-noise ratio (SNR).
Power, Intensity, Intensity or current refers to the current of the The higher the intensity, the more photons are produced
Current accelerated electrons. For a given energy, the power is and the better the SNR for a given measurement time.
proportional to the current. Whether power, intensity, or For a given energy, an increase in intensity is in general
current can be set depends on the microCT model. related to an increase in focal spot size.
Focal spot The size of the emission spot from the X-ray source. The focal spot size is a critical parameter limiting the
This parameter might not be set independent of intensity. resolution for objects measured close to the source. The
smaller the focal spot, the better resolution that can be
achieved. However, a small focal spot is also related to a
low intensity which may necessitate long measurement
times. If a scaffold has very thin struts and the goal is to
measure the strut thickness, it is essential to work with
the smallest possible spot size. However, if contrast
differences are of higher importance, a larger spot size
may yield better results.
Integration time The integration time, sometimes referred to as exposure The longer the integration time, the better the SNR but
(Exposure time) time, is the time to acquire an individual projection frame. the longer the measurement time. In order to obtain a
It might be limited to not oversaturate the detector. The reasonable SNR, the integration time may need to be
maximal integration time depends on the energy, longer for scaffolds with a low attenuation coefficient as
intensity, and filtering. compared to scaffolds with high attenuation coefficient. In
general, if different materials with comparable attenuation
coefficients should be separable in an image (e.g.
scaffold in bone), integration time should be selected
longer in order to increase SNR.
Frame averaging Number of times a frame (projection) is acquired at a Frame averaging improves SNR and can overcome a
given position in order to get an averaged frame. potential limitation in integration time. Frame averaging is
proportional to the measurement time, but can
significantly aid SNR for low density materials and small
structures (e.g., scaffold struts with small thickness and
width).
Number of projections The number of projections taken over 180 degrees The number of projections is a trade-off between image
quality and measurement time. For fast overview scans,
a relatively low number of projections might be sufficient.
However, if the number chosen is too low, aliasing
artifacts may distort the image. For standard scans, the
values set by the manufacturer should yield reasonable
results for most scaffold imaging. Typically, the
projections are acquired over 180 degrees, however,
some situations may require the user to take projections
over 360 degrees. In these situations, the total number
of projections and the scan angle should be reported.
A
For more detailed information on microCT parameters, please refer to Stauber M & Muller R, 2008 (6).
F3259 − 17
7.1.1.1 A good resource to estimate the attenuation of a X-ray tube or other system parts. Therefore, it is essential that
given material or composition is the “XCOM: Photon Cross the environmental conditions be kept as stable as possible
Sections Database” (http://www.nist.gov/pml/data/xcom/ throughout imaging.
index.cfm).
7.4 Some situations may necessitate scanning of the same
7.1.2 When scanning multi-phase materials, the selection of
specimen at different locations and subsequent combination of
energy and filter is determined by the total density of the
thereconstructedimages.Forexample,thissituationmayarise
sample. The energy should be set high enough to penetrate all
if the user desires very high resolution images such that the
materials sufficiently.
entirespecimendoesnotfitinthefieldofview.Thisisahighly
7.1.2.1 In the case of scaffolds with multiple materials, the
specialized method and specific guidance on how to conduct
total density tends to be dominated by the maximum density
the scanning and conduct the reconstruction is outside the
phase. However, if that phase has only a very small percent
scope of this document.
volume, then it may not be dominant.
7.1.2.2 If the phases are similar in density, the attenuation
7.5 Thesamescanparametersshouldbeusedwhenimaging
coefficients typically lie in a similar range as well. In this case,
all scaffolds in a study.
itisrecommendedtopickanenergyaslowaspossiblebecause
the attenuation curves exhibit better separation for lower
8. Image Reconstruction and Artifact
energies.
8.1 In a microCT measurement, the acquisition results in
7.2 Table 2 provides some examples of common materials
raw data that has to be reconstructed to obtain a three-
used in TEMP scaffolds that have been evaluated with
dimensional image. In this step, the 2D X-ray projections are
microCT, a qualitative ranking of their radiodensity, and some
reconstructedtoa3Dvolume,whichisreferredtoastheimage
basic guidelines for measurement parameters. The parameters
dataset. This dataset is used for subsequent analysis of the 3D
are also dependent on the size of the object or the relative
structural properties of the scaffold. The reconstruction meth-
composition of the object materials. This information may be
ods as well as the corrections applied during reconstruction
useful as a starting point when defining scan settings.
significantly determine the quality of the reconstructed data. It
7.3 Apartfromtheparametersthatcanbesetdirectlyonthe is therefore essential that all specimens be reconstructed in the
same way within a given study.As this is a critical step, most
microCT software, there are some indirect parameters that are
relevant for high-quality microCT acquisition. These include manufacturers use ideal parameters to reconstruct all scanned
temperature,humidity,andvibrationsduetotheireffectsonthe data in exactly the same way and no user interaction is
TABLE 2 Examples of Common Scaffold Materials Evaluated with MicroCT
Material TEMPs Application Radiodensity Basic Imaging Parameters
Synthetic polymers
Polylactic acid (PLA) Bone/cartilage Low Filter: none
Energy: 30-50kV
Polyglycolic acid (PGA) Bone/cartilage Low Filter: none
Energy: 30-50kV
Polycaprolactone (PCL) Bone/cartilage Low Filter: none
Energy: 30-50kV
Poly propylene(fumarate) (PPF) Bone/cardiovascular Low Filter: none
Energy: 30-50kV
Poly methyl methacrylate (PMMA) Bone Medium Filter: Al 0.5mm
Energy: 40-60kV
Natural materials
Collagen Bone Low Filter: none
Energy: 30-50kV
Fibrin Cardiovascular Low Filter: none
Energy: 30-50kV
Chitosan Bone/Cartilage Low Filter: none
Energy: 30-50kV
Alginate Bone/Cartilage Very low Filter: none
Energy: 30-50kV
Silk Bone/cartilage Low Filter: none
Energy: 30-50kV
Hydroxyapatite (HA) Bone Medium Filter: Al 0.5mm
Energy: 40-70kV
Tri-Calcium Phosphate (TCP) Bone Medium Filter: Al 0.5mm
Energy: 40-70kV
Titanium Bone Very high Filter: Al 0.5mm or
Cu 0.1mm
Energy: 70-100+
Mammalian or human derived materials
Demineralized Bone Bone Low Filter: Al 0.5mm
Energy: 50-70
--
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...