ASTM F2450-18
(Guide)Standard Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products
Standard Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products
SIGNIFICANCE AND USE
5.1 The ability to culture functional tissue to repair damaged or diseased tissues within the body offers a viable alternative to xenografts or heterografts. Using the patient’s own cells to produce the new tissue offers significant benefits by limiting rejection by the immune system. Typically, cells harvested from the intended recipient are cultured in vitro using a temporary housing or scaffold. The microstructure of the scaffold can be defined by the existence, type, size distribution, interconnectivity, and directionality of pores – all of which are critical for cell migration, growth, and proliferation (Appendix X1). Optimizing the design of tissue scaffolds is a complex task, given the range of available materials, different manufacturing routes, and processing conditions. All of these factors can, and will, affect the surface texture, surface chemistry, and microstructure of the resultant scaffolds. Surface texture, surface chemistry, and microstructure of the scaffolds may or may not be significant variables depending on the characteristics of a given cell type at any given time (that is, changes in cell behavior due to the number of passages, mechanical stimulation, and culture conditions).
5.2 Tissue scaffolds are typically assessed using an overall value for scaffold porosity and a range of pore sizes, though the distribution of sizes is rarely quantified. Published mean pore sizes and distributions are usually obtained from electron microscopy images and quoted in the micrometer range. Tissue scaffolds are generally complex structures that are not easily interpreted in terms of pore shape and size, especially in three dimensions. Therefore, it is difficult to quantifiably assess the batch-to-batch variance in microstructure or to make a systematic investigation of the role that the mean pore size and pore size distribution has on influencing cell behavior based solely on electron micrographs (Tomlins et al, (1)).4
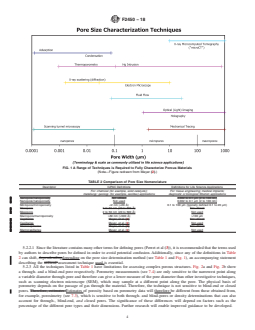
5.2.1 Fig. 1 gives an indication...
SCOPE
1.1 This guide covers an overview of test methods that may be used to obtain information relating to the dimensions of pores, the pore size distribution, the degree of porosity, interconnectivity, and measures of permeability for porous materials used as polymeric scaffolds in the development and manufacture of tissue-engineered medical products (TEMPs). This information is key to optimizing the structure for a particular application, developing robust manufacturing routes, and providing reliable quality control data.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Nov-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 15-Nov-2018
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Feb-2018
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Jul-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Dec-2005
- Effective Date
- 01-May-2005
- Effective Date
- 01-Jul-2004
Overview
ASTM F2450-18, “Standard Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products,” is an internationally recognized guide from ASTM International. This standard outlines recommended test methods and approaches for characterizing the microstructure of polymeric scaffolds that are essential components in tissue-engineered medical products (TEMPs). Detailed microstructural analysis is key for optimizing scaffold performance, supporting robust manufacturing routes, and ensuring reliable quality control. This guide plays a valuable role in advancing regenerative medicine by helping manufacturers and researchers assess critical scaffold properties-such as porosity, pore size, and connectivity-that directly influence cell migration, tissue growth, and the clinical effectiveness of medical implants.
Key Topics
- Microstructure Characterization: The guide identifies test methodologies to measure dimensions of pores, pore size distribution, overall porosity, pore interconnectivity, and permeability in polymeric scaffolds.
- Imaging Techniques: Recommends imaging techniques such as electron microscopy, optical microscopy (including confocal microscopy and optical coherence tomography), X-ray micro-computed tomography, and magnetic resonance imaging. Each technique provides complementary data on scaffold structure at different scales and depth resolutions.
- Physical Properties: Addresses the importance of measuring physical properties, such as density and permeability, to quantify the accessible pore space and the ability of scaffolds to facilitate fluid or cell migration.
- Terminology: Clarifies essential definitions for bioactive agents, different pore types (open, closed, blind-end), porosity levels (nanoporosity, microporosity, macroporosity), and manufacturing approaches.
- Method Limitations: Notes that each test method has inherent limitations based on scaffold material properties, such as stiffness and hydration state. The choice of characterization technique should align with scaffold composition and target application.
Applications
- Development of Tissue-Engineered Medical Products (TEMPs): Enables accurate quality assessment and refinement of scaffolds used for culturing functional tissue, such as for regenerative repair of bones, cartilage, or soft tissues.
- Optimization of Scaffold Design: Provides a framework for selecting appropriate characterization methods during the design phase to optimize pore architecture for specific cell types and intended applications.
- Manufacturing and Quality Control: Facilitates batch-to-batch consistency by guiding the physical and microstructural evaluation of polymeric scaffold materials during production.
- Support for Regulatory Submissions: By using standardized assessment methods, manufacturers can generate reliable, comparable data for regulatory filings and product approvals.
- Research and Innovation: Assists researchers in the systematic investigation of how scaffold microstructure affects cell behavior, tissue integration, and long-term performance in vivo.
Related Standards
ASTM F2450-18 references and complements several other ASTM standards relevant to porous medical materials:
- ASTM F2150 - Guide for Characterization and Testing of Biomaterial Scaffolds Used in Tissue-Engineered Medical Products
- ASTM F2603 - Guide for Interpreting Images of Polymeric Tissue Scaffolds
- ASTM D4404 - Test Method for Determination of Pore Volume and Pore Volume Distribution of Soil and Rock by Mercury Intrusion Porosimetry
- ASTM E128 / E1294 / F316 - Test methods for determining pore size characteristics and permeability of porous materials
- ASTM E1441 - Guide for Computed Tomography (CT) Imaging
These related standards collectively support comprehensive assessment of polymeric scaffold microstructure and are integral to developing safe, effective, and high-performing tissue-engineered medical products.
Keywords: ASTM F2450-18, polymeric scaffold assessment, tissue-engineered medical products, microstructural characterization, porosity, pore size distribution, scaffold quality control, imaging techniques, regenerative medicine standards.
Buy Documents
ASTM F2450-18 - Standard Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products
REDLINE ASTM F2450-18 - Standard Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2450-18 is a guide published by ASTM International. Its full title is "Standard Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products". This standard covers: SIGNIFICANCE AND USE 5.1 The ability to culture functional tissue to repair damaged or diseased tissues within the body offers a viable alternative to xenografts or heterografts. Using the patient’s own cells to produce the new tissue offers significant benefits by limiting rejection by the immune system. Typically, cells harvested from the intended recipient are cultured in vitro using a temporary housing or scaffold. The microstructure of the scaffold can be defined by the existence, type, size distribution, interconnectivity, and directionality of pores – all of which are critical for cell migration, growth, and proliferation (Appendix X1). Optimizing the design of tissue scaffolds is a complex task, given the range of available materials, different manufacturing routes, and processing conditions. All of these factors can, and will, affect the surface texture, surface chemistry, and microstructure of the resultant scaffolds. Surface texture, surface chemistry, and microstructure of the scaffolds may or may not be significant variables depending on the characteristics of a given cell type at any given time (that is, changes in cell behavior due to the number of passages, mechanical stimulation, and culture conditions). 5.2 Tissue scaffolds are typically assessed using an overall value for scaffold porosity and a range of pore sizes, though the distribution of sizes is rarely quantified. Published mean pore sizes and distributions are usually obtained from electron microscopy images and quoted in the micrometer range. Tissue scaffolds are generally complex structures that are not easily interpreted in terms of pore shape and size, especially in three dimensions. Therefore, it is difficult to quantifiably assess the batch-to-batch variance in microstructure or to make a systematic investigation of the role that the mean pore size and pore size distribution has on influencing cell behavior based solely on electron micrographs (Tomlins et al, (1)).4 5.2.1 Fig. 1 gives an indication... SCOPE 1.1 This guide covers an overview of test methods that may be used to obtain information relating to the dimensions of pores, the pore size distribution, the degree of porosity, interconnectivity, and measures of permeability for porous materials used as polymeric scaffolds in the development and manufacture of tissue-engineered medical products (TEMPs). This information is key to optimizing the structure for a particular application, developing robust manufacturing routes, and providing reliable quality control data. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The ability to culture functional tissue to repair damaged or diseased tissues within the body offers a viable alternative to xenografts or heterografts. Using the patient’s own cells to produce the new tissue offers significant benefits by limiting rejection by the immune system. Typically, cells harvested from the intended recipient are cultured in vitro using a temporary housing or scaffold. The microstructure of the scaffold can be defined by the existence, type, size distribution, interconnectivity, and directionality of pores – all of which are critical for cell migration, growth, and proliferation (Appendix X1). Optimizing the design of tissue scaffolds is a complex task, given the range of available materials, different manufacturing routes, and processing conditions. All of these factors can, and will, affect the surface texture, surface chemistry, and microstructure of the resultant scaffolds. Surface texture, surface chemistry, and microstructure of the scaffolds may or may not be significant variables depending on the characteristics of a given cell type at any given time (that is, changes in cell behavior due to the number of passages, mechanical stimulation, and culture conditions). 5.2 Tissue scaffolds are typically assessed using an overall value for scaffold porosity and a range of pore sizes, though the distribution of sizes is rarely quantified. Published mean pore sizes and distributions are usually obtained from electron microscopy images and quoted in the micrometer range. Tissue scaffolds are generally complex structures that are not easily interpreted in terms of pore shape and size, especially in three dimensions. Therefore, it is difficult to quantifiably assess the batch-to-batch variance in microstructure or to make a systematic investigation of the role that the mean pore size and pore size distribution has on influencing cell behavior based solely on electron micrographs (Tomlins et al, (1)).4 5.2.1 Fig. 1 gives an indication... SCOPE 1.1 This guide covers an overview of test methods that may be used to obtain information relating to the dimensions of pores, the pore size distribution, the degree of porosity, interconnectivity, and measures of permeability for porous materials used as polymeric scaffolds in the development and manufacture of tissue-engineered medical products (TEMPs). This information is key to optimizing the structure for a particular application, developing robust manufacturing routes, and providing reliable quality control data. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2450-18 is classified under the following ICS (International Classification for Standards) categories: 11.100 - Laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2450-18 has the following relationships with other standards: It is inter standard links to ASTM F2450-10, ASTM F316-03(2019), ASTM E128-99(2019), ASTM D4404-18, ASTM F2150-13, ASTM F2603-06(2012), ASTM E128-99(2011), ASTM E1441-11, ASTM F316-03(2011), ASTM D4404-10, ASTM F2150-07, ASTM F2603-06, ASTM E1441-00(2005), ASTM E128-99(2005), ASTM D4404-84(2004). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2450-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2450 − 18
Standard Guide for
Assessing Microstructure of Polymeric Scaffolds for Use in
Tissue-Engineered Medical Products
This standard is issued under the fixed designation F2450; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D4404 Test Method for Determination of Pore Volume and
Pore Volume Distribution of Soil and Rock by Mercury
1.1 This guide covers an overview of test methods that may
Intrusion Porosimetry
be used to obtain information relating to the dimensions of
E128 Test Method for Maximum Pore Diameter and Perme-
pores, the pore size distribution, the degree of porosity,
ability of Rigid Porous Filters for Laboratory Use
interconnectivity, and measures of permeability for porous
E1294 Test Method for Pore Size Characteristics of Mem-
materials used as polymeric scaffolds in the development and
brane Filters UsingAutomated Liquid Porosimeter (With-
manufacture of tissue-engineered medical products (TEMPs).
drawn 2008)
This information is key to optimizing the structure for a
E1441 Guide for Computed Tomography (CT) Imaging
particularapplication,developingrobustmanufacturingroutes,
F316 Test Methods for Pore Size Characteristics of Mem-
and providing reliable quality control data.
brane Filters by Bubble Point and Mean Flow Pore Test
1.2 The values stated in SI units are to be regarded as
F2150 Guide for Characterization and Testing of Biomate-
standard. No other units of measurement are included in this
rial Scaffolds Used in Tissue-Engineered Medical Prod-
standard.
ucts
1.3 This standard does not purport to address all of the
F2603 Guide for Interpreting Images of Polymeric Tissue
safety concerns, if any, associated with its use. It is the
Scaffolds
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
3. Terminology
mine the applicability of regulatory limitations prior to use.
3.1 Definitions:
1.4 This international standard was developed in accor-
3.1.1 bioactive agent, n—any molecular component in, on,
dance with internationally recognized principles on standard-
or within the interstices of a device that is intended to elicit a
ization established in the Decision on Principles for the
desired tissue or cell response.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
3.1.1.1 Discussion—Growth factors and antibiotics are typi-
Barriers to Trade (TBT) Committee.
cal examples of bioactive agents. Device structural compo-
nents or degradation byproducts that evoke limited localized
2. Referenced Documents
bioactivity are not bioactive agents.
2.1 ASTM Standards:
3.1.2 blind (end)-pore, n—a pore that is in contact with an
D2873 Test Method for Interior Porosity of Poly(Vinyl
exposed internal or external surface through a single orifice
Chloride) (PVC) Resins by Mercury Intrusion Porosim-
smaller than the pore’s depth.
etry (Withdrawn 2003)
3.1.3 closed cell, n—a void isolated within a solid, lacking
any connectivity with an external surface. Synonym: closed
pore
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
Surgical Materials and Devices and is the direct responsibility of Subcommittee
3.1.4 hydrogel, n—a water-based open network of polymer
F04.42 on Biomaterials and Biomolecules for TEMPs.
chains that are cross-linked either chemically or through
Current edition approved Nov. 15, 2018. Published December 2018. Originally
approved in 2004. Last previous edition approved in 2010 as F2450 – 10. DOI: crystalline junctions or by specific ionic interactions.
10.1520/F2450-18.
3.1.5 macropore/macroporosity (life sciences),n—a struc-
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
ture (including void spaces) sized to allow substantially unre-
Standards volume information, refer to the standard’s Document Summary page on
stricted passage of chemicals, biomolecules, viruses, bacteria,
the ASTM website.
and mammalian cells. In implants with interconnecting pores,
The last approved version of this historical standard is referenced on
www.astm.org. macroporosity provides dimensions that allow for ready tissue
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2450 − 18
penetration and microvascularization after implantation. In- 3.1.15 scaffold, n—a support, delivery vehicle, or matrix for
cludes materials that contain voids with the potential to be facilitating the migration, binding, or transport of cells or
bioactive molecules used to replace, repair, or regenerate
observable to the naked eye (>100 µm).
tissues.
3.1.6 micropore/microporosity (life sciences),n—a struc-
3.1.16 through-pores, n—an inherent or induced network of
ture (including void spaces) sized to allow substantially unre-
voids or channels that permit flow of fluid (liquid or gas) from
stricted passage of chemicals, biomolecules, and viruses while
one side of the structure to the other.
sized to control or moderate the passage of bacteria, mamma-
3.1.17 tortuosity, n—a measure of the mean free path length
lian cells, and/or tissue. Includes materials with typical pore
of through-pores relative to the sample thickness. Alternative
sizes greater than 0.1 µm (100 nm) and less than about 100 µm
definition: The squared ratio of the mean free path to the
(100 000 nm), with a common microporous context encom-
minimum possible path length.
passing the range of 20 µm or less for the filtration of cells
ranging from bacteria to common mammalian cells and above
4. Summary of Guide
30 µm for the ingrowth of tissue. Objects in this size range
4.1 The microstructure, surface chemistry, and surface mor-
typically can be observed by conventional light microscopy.
phology of polymer-based tissue scaffolds plays a key role in
3.1.7 nanopore/nanoporosity (life sciences),n—a structure
encouraging cell adhesion, migration, growth, and prolifera-
inclusive of void spaces sized to control or moderate the
tion.The intention of this guide is to provide a compendium of
passage of chemicals, biomolecules, and viruses while sized to
techniques for characterizing this microstructure. The breadth
substantially exclude most bacteria and all mammalian cells.
of the techniques described reflects the practical difficulties of
Includes materials with typical pore sizes of less than 100 nm quantifying pore sizes and pore size distributions over length
(0.1µm),withcommonnanoporouscontextintherangeof~20 scales ranging from nanometres to sub-millimetres and the
porosity of materials that differ widely in terms of their
nm or less for the filtration of viruses.
mechanical properties.
3.1.8 permeability, n—a measure of fluid, particle, or gas
4.2 These microstructural data when used in conjunction
flow through an open pore structure.
with other characterization methods, for example, chemical
3.1.9 polymer, n—a long chain molecule composed of
analysis of the polymer (to determine parameters such as the
monomers including both natural and synthetic materials.
molecular mass (molecular weight) and its distribution), will
Examples include collagen and polycaprolactone.
aid in the optimization of scaffolds for tissue-engineered
medical products (TEMPs). Adequate characterization is also
3.1.10 pore, n—a fluid (liquid or gas) filled externally
critical to ensure the batch-to-batch consistency of scaffolds;
connecting channel, void, or open space within an otherwise
eithertoassessbasematerialssuppliedbydifferentsuppliersor
solid or gelatinous material (for example, textile meshes
to develop robust manufacturing procedures for commercial
composed of many or single fibers (textile based scaffolds),
production.
open cell foams, hydrogels). Synonyms: open-pore, through-
pore. 4.3 Application of the techniques described in this guide
will not guarantee that the scaffold will perform the functions
3.1.11 porogen, n—a material used to create pores within an
for which it is being developed but they may help to identify
inherently solid material.
the reasons for success or failure.
3.1.11.1 Discussion—For example, a polymer dissolved in
4.4 This guide does not suggest that all listed tests be
anorganicsolventispouredoverawater-solublepowder.After
conducted. The choice of technique will depend on the
evaporation of the solvent, the porogen is leached out, usually
information that is required and on the scaffold’s physical
by water, to leave a porous structure. The percentage of
properties; for example, mercury porosimetry will not yield
porogenneedstobehighenoughtoensurethatalltheporesare
meaningful data if used to characterize soft materials that
interconnected.
deform during the test and cannot be used for hydrated
scaffolds.
3.1.12 porometry, n—the determination of the distribution
of open pore diameters relative to the direction of fluid flow by
4.5 Table 1 provides guidance for users of this guide by
thedisplacementofanon-volatilewettingfluidasafunctionof
providing a brief overview of the applicability of a range of
pressure.
different measurement techniques that can be used to physi-
cally characterize tissue scaffolds.This list of techniques is not
3.1.13 porosimetry, n—the determination of the pore vol-
definitive.
umeandporesizedistributionthroughtheuseofanon-wetting
liquid (typically mercury) intrusion into a porous material as a
5. Significance and Use
function of pressure.
5.1 Theabilitytoculturefunctionaltissuetorepairdamaged
3.1.14 porosity, n—property of a solid which contains an
ordiseasedtissueswithinthebodyoffersaviablealternativeto
inherent or induced network of channels and open spaces.
xenografts or heterografts. Using the patient’s own cells to
Porosity can be determined by measuring the ratio of pore produce the new tissue offers significant benefits by limiting
(void) volume to the apparent (total) volume of a porous
rejection by the immune system. Typically, cells harvested
material and is commonly expressed as a percentage. from the intended recipient are cultured in vitro using a
F2450 − 18
TABLE 1 A Guide to the Physical / Microstructural Characterization of Tissue Scaffolds
Generic Technique Information Available Section
Microscopy Pore shape, size and size distribution; porosity. 6.1 (Electron microscopy)
6.2 (Optical microscopy)
6.2.3 (Confocal microscopy)
6.2.4 (Optical coherence tomography)
6.2.5 (Optical coherence microscopy)
X-Ray micro-computed Tomography Pore shape, size and size distribution; porosity. 6.3
(MicroCT)
Magnetic Resonance Imaging Pore shape, size and size distribution; porosity. 6.4
Measurement of density Porosity, pore volume 7.2
Porosimetry Porosity, total pore surface area, pore diameter, pore size distribution 7.3
Porometry Median pore diameter (assuming cylindrical geometry), through-pore 7.4
size distribution
Diffusion of markers Permeability 8.2
NMR Pore size and distribution 7.5
temporary housing or scaffold. The microstructure of the lifescienceapplications,whichincludebothimplantandtissue
scaffold can be defined by the existence, type, size distribution, engineering applications.
interconnectivity, and directionality of pores – all of which are 5.2.2.1 Since the literature contains many other terms for
critical for cell migration, growth, and proliferation (Appendix defining pores (Perret et al (3)), it is recommended that the
X1). Optimizing the design of tissue scaffolds is a complex terms used by authors to describe pores be defined in order to
task, given the range of available materials, different manufac- avoid potential confusion. Additionally, since any of the
turing routes, and processing conditions. All of these factors definitions in Table 2 can shift, depending on the pore size
can, and will, affect the surface texture, surface chemistry, and determination method (see Table 1 and Fig. 1), an accompa-
microstructure of the resultant scaffolds. Surface texture, sur- nying statement describing the assessment technique used is
face chemistry, and microstructure of the scaffolds may or may essential.
not be significant variables depending on the characteristics of 5.2.3 AllthetechniqueslistedinTable1havelimitationsfor
a given cell type at any given time (that is, changes in cell assessing complex porous structures. Fig. 2a and Fig. 2b show
behavior due to the number of passages, mechanical a through- and a blind-end pore respectively. Porometry
stimulation, and culture conditions).
measurements (see 7.4) are only sensitive to the narrowest
point along a variable diameter through-pore and therefore can
5.2 Tissue scaffolds are typically assessed using an overall
give a lower measure of the pore diameter than other investi-
valueforscaffoldporosityandarangeofporesizes,thoughthe
gative techniques, such as scanning electron microscope
distribution of sizes is rarely quantified. Published mean pore
(SEM), which may sample at a different point along the pore.
sizes and distributions are usually obtained from electron
The physical basis of porometry depends on the passage of gas
microscopyimagesandquotedinthemicrometerrange.Tissue
through the material. Therefore, the technique is not sensitive
scaffolds are generally complex structures that are not easily
to blind-end or closed pores. Estimates of porosity based on
interpreted in terms of pore shape and size, especially in three
porometry data will therefore be different from those obtained
dimensions. Therefore, it is difficult to quantifiably assess the
from, for example, porosimetry (see 7.3), which is sensitive to
batch-to-batch variance in microstructure or to make a system-
both through- and blind-pores or density determinations that
atic investigation of the role that the mean pore size and pore
canalsoaccountforthrough-,blind-end, andclosedpores.The
size distribution has on influencing cell behavior based solely
4 significance of these differences will depend on factors such as
on electron micrographs (Tomlins et al, (1)).
the percentage of the different pore types and their dimensions.
5.2.1 Fig. 1 gives an indication of potential techniques that
Further research will enable improved guidance to be devel-
can be used to characterize the structure of porous tissue
oped.
scaffolds and the length scale that they can measure. Clearly a
5.2.4 Polymer scaffolds range from mechanically rigid
range of techniques must be used if the scaffold is to be
structures to soft hydrogels. The methods currently used to
characterized in detail.
manufacture these structures include, but are not limited to:
5.2.2 The classification and terminology of pore sizes, such
5.2.4.1 Casting a polymer, dissolved in an organic solvent,
as those given in Table 2, has yet to be standardized, with
overawater-solubleparticulateporogen,followedbyleaching.
definitions of terms varying widely (as much as three orders of
5.2.4.2 Melt mixing of immiscible polymers followed by
magnitude) between differing applications and industries. Both
leaching of the water-soluble component.
Table 2 and the supporting detailed discussion included within
5.2.4.3 Dissolution of supercritical carbon dioxide under
Appendix X2 describe differences that exist between IUPAC
pressure into an effectively molten polymer, a phenomenon
(International Union of Pure and Applied Chemistry) defini-
attributed to the dramatic reduction in the glass transition
tions and the common terminology currently used within most
temperature which occurs, followed by a reduction in pressure
that leads to the formation of gas bubbles and solidification.
5.2.4.4 Controlled deposition of molten polymer to produce
The boldface numbers in parentheses refer to the list of references at the end of
this standard. a well-defined three-dimensional lattice.
F2450 − 18
FIG. 1 A Range of Techniques is Required to Fully Characterize Porous Materials
(Note—Figure redrawn from Meyer (2).)
TABLE 2 Comparison of Pore Size Nomenclature
Descriptor IUPAC Definitions Definitions for Life Science Applications
For: chemical (for example, solid catalysts); For: tissue engineering; medical implants;
metallurgy; geology (for example, zeolites) applications diagnostic or biological filtration applications
Nanopore/nanoporosity Not used 0.002 to 0.1 µm (2 to 100 nm)
Micropore/microporosity <2 nm (<20 Å) 0.1 to 100 µm (typically defined 0.1 to 20 µm)
Mesopore 2 to 50 nm (20 to 500 Å) Not used
Macropore/macroporosity >50 nm (>500 Å) >100 µm
Capillaries Meyer, et al. (2) Not used
Macrocapillaries Meyer, et al. (2) Not used
FIG. 2 A through-pore showing a variation of pore diameter, D (a); and an example of a blind-pore (b).
F2450 − 18
5.2.4.5 The manufacture of three-dimensional fibrous subsequently obtained are only of value when comparing
weaves, knits, or non-woven structures. microstructures between different samples. However, the re-
5.2.4.6 Chemical or ionic cross-linking of a polymeric sults are less useful for characterizing expected in vivo micro-
matrix.
structure due to sample distortion.
5.2.5 Considerations have been given to the limitations of
6.2 Optical Microscopy-Based Methods:
these methods in Appendix X1.
6.2.1 Optical methods can be used, providing sufficient
5.2.6 This guide focuses on the specific area of character-
contrast exists between the structure and surrounding media to
ization of polymer-based porous scaffolds and is an extension
enablesurfacefeaturestobestudiedinaminimallypreparedor
of an earlier ASTM guide, Guide F2150.
natural state (that is, the specimen does not need to be stained
or sectioned.) The disadvantage of this approach is that
6. Imaging
penetration of light into the sample can be limited, particularly
6.1 Electron Microscopy—Both transmission and scanning
for porous matrices, due to scattering. In practice, this limits
electron microscopy can be used to image intact or fractured
the use of confocal microscopy and optical coherence tomog-
surfaces or sections cut from tissue scaffolds. The resultant
raphy to depths that are typically less than 0.5 mm.
images can be interpreted using image analysis software
6.2.2 Optical(Light)Microscopy—Imagesofthesurfacesof
packages to generate data concerning the shape of pores within
tissue scaffolds can be obtained using an optical microscope.
the scaffold, their mean size, and their distribution. Estimates
Differential focus can used to collect images at different depths
of both permeability and tortuosity can be made from three-
within semi-transparent specimens. These deep view images
dimensional virtual images generated from transmission elec-
canbeusedtotrackthepathofinterconnectedporeswithinthe
tron microscopic images of serially sectioned samples.
sample.
6.1.1 Thereislikelytobeahighdegreeofuncertaintyinthe
6.2.3 Confocal Microscopy—Substantial improvements in
reliability of quantitative data derived from electron micro-
scopic examination of soft or especially highly hydrated soft the quality of ‘optically’ sectioned samples can be made by
polymer-basedscaffoldsduetothepresenceofartifactscreated either exploiting the natural fluorescent properties that the
during sample preparation. Highly hydrated scaffolds need to scaffold may have or by using a fluorescent stain such as
be freeze-dried before examination under vacuum in a conven- fluorescein. Confocal microscopy can capture well resolved
tional scanning electron microscope (SEM). This process, if
images at different depths because of its shallow depth of field
carried out in liquid nitrogen, usually results in a significant
and elimination of out-of-focus light.Alaser is usually used as
amount of ice damage due to the relatively slow cooling rates
apointlightsourceinpreferencetoaconventionallampandin
that are encountered due to the thin layer of insulating nitrogen
most modern instruments, several lasers are used. This capa-
gas that forms around the sample as it is frozen. Freezing
bilityisusedtoimprovecontrastwithintheimageandtoexcite
samples in slush nitrogen can reduce ice damage by enabling
stains that bind to different structural elements and fluoresce at
fastercoolingrates,apparentlybyreducingthethicknessofthe
different wavelengths. Laser scanning confocal microscopy
insulating gas layer.
(LSCM) can be used in reflection or transmission modes. The
6.1.2 Cryogenic SEM may also be used to reduce artifacts
size of the pinhole and the numerical aperture of the objective
introduced as a result of conventional freezing. In this
primarily determine the resolution in the thickness or axial
technique, a rapidly frozen specimen is fractured whilst frozen
direction. Generally, smaller holes give better resolution but at
within the cryo-SEM unit and sputter coated with gold-
the expense of reduced intensity.
palladium after allowing some of the ice to sublime away. The
6.2.3.1 Some work on scaffold characterization using laser
amount of sublimation that occurs can be controlled through
scanningconfocalmicroscopy(LSCM)hasbeenreported(Tjia
exposure time. With this technique, any freeze-drying of the
and Moghe, (4), Birla and Matthew (5)).
sample is minimized. Experimentally validating the results
6.2.4 Optical Coherence Tomography (OCT)—Optical co-
obtained from this technique to ensure that they are artifact-
herence tomography is a reflectance optical imaging technique
free is difficult.
that uses interferometric rejection of out-of-plane scattering of
6.1.3 Polymer-based scaffolds often lack sufficient electron
photons rather than a pinhole as in LSCM to determine axial
density to provide suitable levels of contrast; this can be
resolution. Briefly, OCT uses a low coherence source with a
overcome by staining using a high electron density material
bandwidth of anywhere from 30 to 200 nm and an
such as osmium tetroxide that has a high affinity for carbon-
interferometer, usually of Michelson type, that generates pro-
carbon double bonds.
files of back-reflected light for any one transverse position. For
6.1.4 Most polymer-based scaffolds can be mounted in
acompletedescriptionofOCTanditsapplications,seeRef (6).
epoxy resin using standard procedures and subsequently sec-
An analogous technique is ultrasound A-scanning. In the
tioned for serial examination in the transmission electron
Michelson configuration, the material is the fixed arm of the
microscope. This method is less appropriate for investigating
interferometer rather than a mirror. A low numerical aperture
hydrogels that can dehydrate. However, this concern can be
lens is used to achieve a large axial sampling volume and
partially mitigated by gradual dehydration of the scaffold by
using a series of alcohol solutions before embedding in reflections from heterogeneities within the sample are mapped
as a function of depth for any one position. Like LSCM,
resin.This procedure tends to reduce the size of the water-filled
pores within the sample. Thus, the quantifiable pore size data transverseresolutionisdeterminedbygeometricoptics.Unlike
F2450 − 18
LSCM, axial resolution is inversely proportional to the band- scaffold.Afull three-dimensional image can be generated from
width of the source, and a typical value for axial resolution is a series of two-dimensional slices obtained at different heights
10 mm.
within the sample. Typical resolution of such an image is
around 5 to 10 µm, but instruments that can resolve 50 nm are
6.2.4.1 The advantage of OCT is that it is highly sensitive,
commercially available. The success of the technique relies on
typically 90 dB. OCT has been extensively used to image the
there being sufficient contrast, that is, differences in electron
human retina (Hee et al (7)), skin and blood vessels (Barton et
density between the solid material and a fluid (typically air or
al (8)), and the functioning circulatory systems of small live
water) within the pores.
animals (Boppart et al (9)) with excellent clarity. In the late
1990s, the potential for optical coherence tomography in the
6.3.1 The technique does not suffer from the same penetra-
area of materials science was first seen. The first published
tion depth limitations that optical tomographic methods suffer
OCT images of a tissue-engineering scaffold were of a hydro-
from, providing a more complete picture of the scaffold
gel and demonstrated the depth to which images can be
structure. Further information can be found in Guide E1441.
obtained (McDonough et al (10)). The depth of field of the
The non-destructive approach has been used to investigate the
image is limited by scattering from the pores and any crystal-
structureofboneandothermaterials(Mulleretal, (12),Muller
litesthatarepresent.Itcanvaryfromapproximately100µmto
et al, (13)) to validate the design of bone scaffolds (Van
several millimetres depending on the difference in refractive
Oosterwijck et al, (14)) and to investigate polymeric scaffolds
index between the material and its surroundings, the level of
(Maspero et al (15), Lin et al (16)).
porosity, and the pore size distribution. The penetration depth
6.4 Magnetic Resonance Imaging—Many polymers contain
can be improved by filling the pores with a fluid of similar
magnetic resonance (MR) active nuclei (for example, 1H,
refractive index to the scaffold material. In practice, this is
13C), but the relaxation times of nuclei on the polymer
usually a substitution of water for air or oil for water. This
backbone are too short for routine imaging applications. Thus,
procedurecanresultinadditionalproblemsduetopoorwetting
to study the three-dimensional morphology of polymeric
and trapped air. OCT images of porous materials tend to be
scaffolds, the pore space must be filled with a fluid, which is
noisy due to multiply scattered photons that contribute to the
visible in a magnetic resonance imaging (MRI) experiment.
signal. A related technique, optical coherence microscopy,
The ideal fluid must contain MR active nuclei, which are
overcomes the issues related to the fidelity of imaging tissue-
naturally abundant, have a high receptivity, and have a well-
engineering scaffolds.
resolved nuclear magnetic resonance (NMR) spectrum of
6.2.5 Optical Coherence Microscopy—Optical coherence
narrow lines. Moreover, it needs to have a low viscosity to
microscopy is a combination of optical coherence tomography
infiltrate the pore space and must have appropriate relaxation
and confocal microscopy. Optical coherence microscopy is
properties to provide a large signal, after the application of the
highly suited for imaging of optically opaque materials such as
imaging gradients. Fortunately, immersion in water will suffice
tissue-engineering scaffolds because it can attain axial and
for most polymeric scaffolds.
transverse resolution on the order of a micrometer and still
maintain high background rejection. The confocal enhance-
6.4.1 The theoretical limit in spatial resolution for MRI
ment is done in the usual manner by the addition of a high
experiments is typically the distance (~10 µm) a water mol-
numerical aperture objective and a pinhole, which is usually
ecule diffuses during the time it takes to acquire the MRI
the open aperture of the sample arm fiber. For more informa-
signal. Thus, polymeric scaffolds with large pores (50 to 100
tion on optical coherence microscopy, see Ref (6). The key to
µm) can be spatially resolved with this technique. In MR
the technique is the axial point spread functions (PSF) of the
images, the water-filled pores appear bright and the polymer
confocal and coherence techniques. For the confocal
mesh dark.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2450 − 10 F2450 − 18
Standard Guide for
Assessing Microstructure of Polymeric Scaffolds for Use in
Tissue-Engineered Medical Products
This standard is issued under the fixed designation F2450; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide covers an overview of test methods that may be used to obtain information relating to the dimensions of pores,
the pore size distribution, the degree of porosity, interconnectivity, and measures of permeability for porous materials used as
polymeric scaffolds in the development and manufacture of tissue-engineered medical products (TEMPs). This information is key
to optimizing the structure for a particular application, developing robust manufacturing routes, and providing reliable quality
control data.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This guidestandard does not purport to address all of the safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and to
determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D2873 Test Method for Interior Porosity of Poly(Vinyl Chloride) (PVC) Resins by Mercury Intrusion Porosimetry (Withdrawn
2003)
D4404 Test Method for Determination of Pore Volume and Pore Volume Distribution of Soil and Rock by Mercury Intrusion
Porosimetry
E128 Test Method for Maximum Pore Diameter and Permeability of Rigid Porous Filters for Laboratory Use
E1294 Test Method for Pore Size Characteristics of Membrane Filters Using Automated Liquid Porosimeter (Withdrawn 2008)
E1441 Guide for Computed Tomography (CT) Imaging
F316 Test Methods for Pore Size Characteristics of Membrane Filters by Bubble Point and Mean Flow Pore Test
F2150 Guide for Characterization and Testing of Biomaterial Scaffolds Used in Tissue-Engineered Medical Products
F2603 Guide for Interpreting Images of Polymeric Tissue Scaffolds
3. Terminology
3.1 Definitions:
3.1.1 bioactive agent, n—any molecular component in, on, or within the interstices of a device that is intended to elicit a desired
tissue or cell response.
3.1.1.1 Discussion—
This guide is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.42
on Biomaterials and Biomolecules for TEMPs.
Current edition approved March 1, 2010Nov. 15, 2018. Published April 2010December 2018. Originally approved in 2004. Last previous edition approved in 20092010
as F2450 – 09.F2450 – 10. DOI: 10.1520/F2450-10.10.1520/F2450-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2450 − 18
Growth factors and antibiotics are typical examples of bioactive agents. Device structural components or degradation byproducts
that evoke limited localized bioactivity are not bioactive agents.
3.1.2 blind (end)-pore, n—a pore that is in contact with an exposed internal or external surface through a single orifice smaller
than the pore’s depth.
3.1.3 closed cell, n—a void isolated within a solid, lacking any connectivity with an external surface. Synonym: closed pore
3.1.4 hydrogel, n—a water-based open network of polymer chains that are cross-linked either chemically or through crystalline
junctions or by specific ionic interactions.
3.1.5 macropore/macroporosity (life sciences) , n—a structure (including void spaces) sized to allow substantially unrestricted
passage of chemicals, biomolecules, viruses, bacteria, and mammalian cells. In implants with interconnecting pores, macroporosity
provides dimensions that allow for ready tissue penetration and microvascularization after implantation. Includes materials that
contain voids with the potential to be observable to the naked eye (>100 μm).
3.1.6 micropore/microporosity (life sciences) , n—a structure (including void spaces) sized to allow substantially unrestricted
passage of chemicals, biomolecules, and viruses while sized to control or moderate the passage of bacteria, mammalian cells,
and/or tissue. Includes materials with typical pore sizes of greater than 0.1 μm (100 nm) and less than about 100 μm (100 000 nm),
with a common microporous context encompassing the range of 20 μm or less for the filtration of cells ranging from bacteria to
common mammalian cells and above 30 μm for the ingrowth of tissue. Objects in this size range typically can be observed by
conventional light microscopy.
3.1.7 nanopore/nanoporosity (life sciences),n—a structure inclusive of void spaces sized to control or moderate the passage of
chemicals, biomolecules, and viruses while sized to substantially exclude most bacteria and all mammalian cells. Includes
materials with typical pore sizes of less than 100 nm (0.1 μm), with common nanoporous context in the range of ~20 nm or less
for the filtration of viruses.
3.1.8 permeability, n—a measure of fluid, particle, or gas flow through an open pore structure.
3.1.9 polymer, n—a long chain molecule composed of monomers including both natural and synthetic materials. Examples
include collagen and polycaprolactone.
3.1.10 pore, n—a fluid (liquid or gas) filled externally connecting channel, void, or open space within an otherwise solid or
gelatinous material (for example, textile meshes composed of many or single fibers (textile based scaffolds), open cell foams,
(hydrogels)).hydrogels). Synonyms: open-pore, through-pore.
3.1.11 porogen, n—a material used to create pores within an inherently solid material.
3.1.11.1 Discussion—
For example, a polymer dissolved in an organic solvent is poured over a water-soluble powder. After evaporation of the solvent,
the porogen is leached out, usually by water, to leave a porous structure. The percentage of porogen needs to be high enough to
ensure that all the pores are interconnected.
3.1.12 porometry, n—the determination of the distribution of open pore diameters relative to the direction of fluid flow by the
displacement of a non-volatile wetting fluid as a function of pressure.
3.1.13 porosimetry, n—the determination of the pore volume and pore size distribution through the use of a non-wetting liquid
(typically mercury) intrusion into a porous material as a function of pressure.
3.1.14 porosity, n—property of a solid which contains an inherent or induced network of channels and open spaces. Porosity
can be determined by measuring the ratio of pore (void) volume to the apparent (total) volume of a porous material and is
commonly expressed as a percentage.
3.1.15 scaffold, n—a support, delivery vehicle, or matrix for facilitating the migration, binding, or transport of cells or bioactive
molecules used to replace, repair, or regenerate tissues.
3.1.16 through-pores, n—an inherent or induced network of voids or channels that permit flow of fluid (liquid or gas) from one
side of the structure to the other.
3.1.17 tortuosity, n—a measure of the mean free path length of through-pores relative to the sample thickness. Alternative
definition: The squared ratio of the mean free path to the minimum possible path length.
4. Summary of Guide
4.1 The microstructure, surface chemistry, and surface morphology of polymer-based tissue scaffolds plays a key role in
encouraging cell adhesion, migration, growth, and proliferation. The intention of this guide is to provide a compendium of
techniques for characterizing this microstructure. The breadth of the techniques described reflects the practical difficulties of
quantifying pore sizes and pore size distributions over length scales ranging from nanometres to sub-millimetres and the porosity
of materials that differ widely in terms of their mechanical properties.
F2450 − 18
4.2 These microstructural data when used in conjunction with other characterization methods, for example, chemical analysis
of the polymer (to determine parameters such as the molecular weight mass (molecular weight) and its distribution), will aid in
the optimization of scaffolds for tissue-engineered medical products (TEMPs). Adequate characterization is also critical to ensure
the batch-to-batch consistency of scaffolds; either to assess base materials supplied by different suppliers or to develop robust
manufacturing procedures for commercial production.
4.3 Application of the techniques described in this guide will not guarantee that the scaffold will perform the functions for which
it is being developed but they may help to identify the reasons for success or failure.
4.4 This guide does not suggest that all listed tests be conducted. The choice of technique will depend on the information that
is required and on the scaffold’s physical properties; for example, mercury porosimetry will not yield meaningful data if used to
characterize soft materials that deform during the test and cannot be used for hydrated scaffolds.
4.5 Table 1 provides guidance for users of this guide by providing a brief overview of the applicability of a range of different
measurement techniques that can be used to physically characterize tissue scaffolds. This list of techniques is not definitive.
5. Significance and Use
5.1 The ability to culture functional tissue to repair damaged or diseased tissues within the body offers a viable alternative to
xenografts or heterografts. Using the patient’s own cells to produce the new tissue offers significant benefits by limiting rejection
by the immune system. Typically, cells harvested from the intended recipient are cultured in vitro using a temporary housing or
scaffold. The microstructure of the scaffold, that is, its porosity, the mean size, and size distribution of pores and their
interconnectivity is scaffold can be defined by the existence, type, size distribution, interconnectivity, and directionality of pores
– all of which are critical for cell migration, growth, and proliferation (Appendix X1). Optimizing the design of tissue scaffolds
is a complex task, given the range of available materials, different manufacturing routes, and processing conditions. All of these
factors can, and will, affect the surface texture, surface chemistry, and microstructure of the resultant scaffolds. Factors that Surface
texture, surface chemistry, and microstructure of the scaffolds may or may not be significant variables dependdepending on the
characteristics of a given cell type at any given time (that is, changes in cell behavior due to the number of passages, mechanical
stimulation, and culture conditions).
5.2 Tissue scaffolds are typically assessed using an overall value for scaffold porosity and a range of pore sizes, though the
distribution of sizes is rarely quantified. Published mean pore sizes and distributions are usually obtained from electron microscopy
images and quoted in the micrometer range. Tissue scaffolds are generally complex structures that are not easily interpreted in
terms of pore shape and size, especially in three dimensions. Therefore, it is difficult to quantifiably assess the batch-to-batch
variance in microstructure or to make a systematic investigation of the role that the mean pore size and pore size distribution has
on influencing cell behavior based solely on electron micrographs (Tomlins et al, (1)).
5.2.1 Fig. 1 gives an indication of potential techniques that can be used to characterize the structure of porous tissue scaffolds
and the length scale that they can measure. Clearly a range of techniques must be utilizedused if the scaffold is to be characterized
in detail.
5.2.2 The classification and terminology of pore sizes, such as those given in Table 2, has yet to be standardized, with definitions
of terms varying widely (as much as three orders of magnitude) between differing applications and industries. Both Table 2 and
the supporting detailed discussion included within Appendix X2 describe differences that exist between IUPAC (International
Union of Pure and Applied Chemistry) definitions and the common terminology currently utilizedused within most life science
applications, which include both implant and tissue engineering applications.
TABLE 1 A Guide to the Physical / Microstructural Characterization of Tissue Scaffolds
Generic Technique Information Available Section
Microscopy Pore shape, size and size distribution; porosity. 6.1 (Electron microscopy)
6.2 (Optical microscopy)
6.2.3 (Confocal microscopy)
6.2.4 (Optical coherence tomography)
6.2.5 (Optical coherence microscopy)
X-Ray micro-computed Tomography Pore shape, size and size distribution; porosity. 6.3
(MicroCT)
Magnetic Resonance Imaging Pore shape, size and size distribution; porosity. 6.4
Measurement of density Porosity, pore volume 7.2
Porosimetry Porosity, total pore surface area, pore diameter, pore size distribution 7.3
Porometry Median pore diameter (assuming cylindrical geometry), through-pore 7.4
size distribution
Diffusion of markers Permeability 8.2
NMR Pore size and distribution 7.5
The boldface numbers in parentheses refer to the list of references at the end of this standard.
F2450 − 18
FIG. 1 A Range of Techniques is Required to Fully Characterize Porous Materials
(Note—Figure redrawn from Meyer (2).)
TABLE 2 Comparison of Pore Size Nomenclature
Descriptor IUPAC Definitions Definitions for Life Science Applications
For: chemical (for example, solid catalysts); For: tissue engineering; medical implants;
metallurgy; geology (for example, zeolites) applications diagnostic or biological filtration applications
Nanopore/nanoporosity Not utilized 0.002 to 0.1 μm (2 to 100 nm)
Nanopore/nanoporosity Not used 0.002 to 0.1 μm (2 to 100 nm)
Micropore/microporosity <2 nm (<20 Å) 0.1 to 100 μm (typically defined 0.1 to 20 μm)
Mesopore 2 to 50 nm (20 to 500 Å) Not utilized
Mesopore 2 to 50 nm (20 to 500 Å) Not used
Macropore/macroporosity >50 nm (>500 Å) >100 μm
Capillaries Meyer, et al. (2) Not utilized
Capillaries Meyer, et al. (2) Not used
Macrocapillaries Meyer, et al. (2) Not utilized
Macrocapillaries Meyer, et al. (2) Not used
5.2.2.1 Since the literature contains many other terms for defining pores (Perret et al (3)), it is recommended that the terms used
by authors to describe pores be defined in order to avoid potential confusion. Additionally, since any of the definitions in Table
2 can shift, dependendingdepending on the pore size determination method (see Table 1 and Fig. 1), an accompanying statement
describing the utilized assessment technique used is essential.
5.2.3 All the techniques listed in Table 1 have limitations for assessing complex porous structures. Fig. 2a and Fig. 2b show
a through- and a blind-end pore respectively. Porometry measurements (see 7.4) are only sensitive to the narrowest point along
a variable diameter through-pore and therefore can give a lower measure of the pore diameter than other investigative techniques,
such as scanning electron microscope (SEM), which may sample at a different point along the pore. The physical basis of
porometry depends on the passage of gas through the material. Therefore, the technique is not sensitive to blind-end or closed
pores. Therefore, estimates Estimates of porosity based on porometry data will therefore be different from those obtained from,
for example, porosimetry (see 7.3), which is sensitive to both through- and blind-pores or density determinations that can also
account for through-, blind-end, and closed pores. The significance of these differences will depend on factors such as the
percentage of the different pore types and their dimensions. Further research will enable improved guidance to be developed.
F2450 − 18
FIG. 2 A through-pore showing a variation of pore diameter, D (a); and an example of a blind-pore (b).
5.2.4 Polymer scaffolds range from mechanically rigid structures to soft hydrogels. The methods currently used to manufacture
these structures include, but are not limited to:
5.2.4.1 Casting a polymer, dissolved in an organic solvent, over a water-soluble particulate porogen, followed by leaching.
5.2.4.2 Melt mixing of immiscible polymers followed by leaching of the water-soluble component.
5.2.4.3 Dissolution of supercritical carbon dioxide under pressure into an effectively molten polymer, a phenomenon attributed
to the dramatic reduction in the glass transition temperature which occurs, followed by a reduction in pressure that leads to the
formation of gas bubbles and solidification.
5.2.4.4 Controlled deposition of molten polymer to produce a well-defined three-dimensional lattice.
5.2.4.5 The manufacture of three-dimensional fibrous weaves, knits, or non-woven structures.
5.2.4.6 Chemical or ionic cross-linking of a polymeric matrix.
5.2.5 Considerations have been given to the limitations of these methods in Appendix X1.
5.2.6 This guide focuses on the specific area of characterization of polymer-based porous scaffolds and is an extension of an
earlier ASTM guide, Guide F2150.
6. Imaging
6.1 Electron Microscopy—Both transmission and scanning electron microscopy can be used to image intact or fractured surfaces
or sections cut from tissue scaffolds. The resultant images can be interpreted using image analysis software packages to generate
data concerning the shape of pores within the scaffold, their mean size, and their distribution. Estimates of both permeability and
tortuosity can be made from three-dimensional virtual images generated from transmission electron microscopic images of serially
sectioned samples.
6.1.1 There is likely to be a high degree of uncertainty in the reliability of quantitative data derived from electron microscopic
examination of soft or especially highly hydrated soft polymer-based scaffolds due to the presence of artifacts created during
sample preparation. Highly hydrated scaffolds need to be freeze-dried before examination under vacuum in a conventional
scanning electron microscope (SEM). This process, if carried out in liquid nitrogen, usually results in a significant amount of ice
damage due to the relatively slow cooling rates that are encountered due to the thin layer of insulating nitrogen gas that forms
around the sample as it is frozen. Freezing samples in slush nitrogen can reduce ice damage by enabling faster cooling rates,
apparently by reducing the thickness of the insulating gas layer.
6.1.2 The relatively new technique of cryogenic Cryogenic SEM may also be used to reduce artifacts. artifacts introduced as
a result of conventional freezing. In this technique, a rapidly frozen specimen can be is fractured whilst frozen within the cryo-SEM
unit and sputter coated with gold-palladium after allowing some of the ice to sublime away. The amount of sublimation that occurs
can be controlled through exposure time. With this technique, any freeze-drying of the sample is minimized. Experimentally
validating the results obtained from this technique to ensure that they are artifact-free is difficult.
6.1.3 Polymer-based scaffolds often lack sufficient electron density to provide suitable levels of contrast; this can be overcome
by staining using a high electron density material such as osmium tetroxide that has a high affinity for carbon-carbon double bonds.
6.1.4 Most polymer-based scaffolds can be mounted in epoxy resin using standard procedures and subsequently sectioned for
serial examination in the transmission electron microscope. This method is less appropriate for investigating hydrogels that can
dehydrate. However, this concern can be partially mitigated by gradual dehydration of the scaffold by using a series of alcohol
solutions before embedding in resin. However, this resin.This procedure tends to reduce the size of the water-filled pores within
the sample. Thus, the quantifiable pore size data subsequently obtained are only of value when comparing microstructures between
different samples. However, the results are less useful for characterizing expected in vivo microstructure due to sample distortion.
6.2 Optical Microscopy-Based Methods:
F2450 − 18
6.2.1 Optical methods can be utilized,used, providing sufficient contrast exists between the structure and surrounding media to
enable surface features to be studied in a minimally prepared or natural state (that is, the specimen does not need to be stained or
sectioned.) The disadvantage of this approach is that penetration of light into the sample can be limited, particularly for porous
matrices, due to scattering. In practice, this limits the use of confocal microscopy and optical coherence tomography to depths that
are typically less than 0.5 mm.
6.2.2 Optical (Light) Microscopy—Images of the surfaces of tissue scaffolds can be obtained using an optical microscope.
Differential focus can used to collect images at different depths within semi-transparent specimens. These deep view images can
be used to track the path of interconnected pores within the sample.
6.2.3 Confocal Microscopy—Substantial improvements in the quality of ‘optically’ sectioned samples can be made by either
exploiting the natural fluorescent properties that the scaffold may have or by using a fluorescent stain such as fluorescein. Confocal
microscopy can capture well-resolved well resolved images at different depths because of its shallow depth of field and elimination
of out-of-focus light. A laser is usually used as a point light source in preference to a conventional lamp and in most modern
instruments, several lasers are used. This capability is used to improve contrast within the image and to excite stains that bind to
different structural elements and fluoresce at different wavelengths. Laser scanning confocal microscopy (LSCM) can be
utilizedused in reflection or transmission modes. The size of the pinhole and the numerical aperture of the objective primarily
determine the resolution in the thickness or axial direction. Generally, smaller holes give better resolution but at the expense of
reduced intensity.
6.2.3.1 Some work on scaffold characterization using laser scanning confocal microscopy (LSCM) has been reported (Tjia and
Moghe, (4), Birla and Matthew (5)).
6.2.4 Optical Coherence Tomography—Tomography (OCT)—Optical coherence tomography is a reflectance optical imaging
technique that uses interferometric rejection of out-of-plane scattering of photons rather than a pinhole as in LSCM to determine
axial resolution. Briefly, optical coherence tomography OCT uses a low coherence source with a bandwidth of anywhere from 30
to 200 nm and an interferometer, usually of Michelson type, that generates profiles of back-reflected light for any one transverse
position. For a complete description of optical coherence tomography OCT and its applications, see Ref (6). An analogous
technique is ultrasound A-scanning. In the Michelson configuration, the material is the fixed arm of the interferometer rather than
a mirror. A low numerical aperture lens is used to achieve a large axial sampling volume and reflections from heterogeneities within
the sample are mapped as a function of depth for any one position. Like LSCM, transverse resolution is determined by geometric
optics. Unlike LSCM, axial resolution is inversely proportional to the bandwidth of the source, and a typical value for axial
resolution is 10 mm.
6.2.4.1 The advantage of optical coherence tomography OCT is that it is highly sensitive, typically 90 dB. Optical coherence
tomography OCT has been extensively used to image the human retina (Hee et al (7)), skin and blood vessels (Barton et al (8)),
and the functioning circulatory systems of small live animals (Boppart et al (9)) with excellent clarity. In the late 1990s, the
potential for optical coherence tomography in the area of materials science was first seen. The first published optical coherence
tomography OCT images of a tissue-engineering scaffold were of a hydrogel and demonstrated the depth to which images can be
obtained (McDonough et al (10)). The depth of field of the image is limited by scattering from the pores and any crystallites that
are present. It can vary from approximately 100 μm to several millimetres depending on the difference in refractive index between
the material and its surroundings, the level of porosity, and the pore size distribution. The penetration depth can be improved by
filling the pores with a fluid of similar refractive index to the scaffold material. In practice, this is usually a substitution of water
for air or oil for water. This procedure can result in additional problems due to poor wetting and trapped air. Optical coherence
tomography OCT images of porous materials tend to be noisy due to multiply scattered photons that contribute to the signal. A
related technique, optical coherence microscopy, overcomes the issues related to the fidelity of imaging tissue-engineering
scaffolds.
6.2.5 Optical Coherence Microscopy—Optical coherence microscopy is a combination of optical coherence tomography and
confocal microscopy. Optical coherence microscopy is highly suited for imaging of optically opaque materials such as
tissue-engineering scaffolds because it can attain axial and transverse resolution on the order of a micrometer and still maintain
high background rejection. The confocal enhancement is done in the usual manner by the addition of a high numerical aperture
objective and a pinhole, which is usually the open aperture of the sample arm fiber. For more information on optical coherence
microscopy, see Ref (6). The key to the technique is the axial point spread functions (PSF) of the confocal and coherence
techniques. For the confocal component, the Lorentzian axial PSF results in a finite collection efficiency even far out of the focus
plane, and this limits its use in highly scattering media such as TEMPs. For the coherence component, the Gaussian PSF drops
off far from the focal plane much more rapidly than that of confocal microscopy. Hence, the confocal component contributes to
the high resolution near the focus and the coherence component contributes to the high background rejection, two qualities needed
for effective imaging of TEMPs (Dunkers et al (11)).
6.3 X-Ray Micro-computed Tomography (MicroCT)—X-rays can be used to generate three-dimensional images of tissue
scaffolds from which information on pore size and shape, porosity, and interconnectivity can be obtained. The principle of the
method is to position the scaffold between an X-ray source and a detector. The sample is rotated and the X-ray attenuation is
recorded at a number of different angles. These data can then be analyzed using reconstruction algorithms to produce an image
of a two-dimensional slice through the scaffold. A full three-dimensional image can be generated from a series of two-dimensional
F2450 − 18
slices obtained at different heights within the sample. Typical resolution of such an image is around 5 to 10 μm, but instruments
that can resolve 50 nm are now commercially available. The success of the technique relies on there being sufficient contrast, that
is, differences in electron density between the solid material and a fluid (typically air or water) within the pores.
6.3.1 The technique does not suffer from the same penetration depth limitations that optical tomographic methods suffer from,
providing a more complete picture of the scaffold structure. Further information can be found in Guide E1441. The non-destructive
approach has been used to investigate the
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...