ASTM D2879-23
(Test Method)Standard Test Method for Vapor Pressure-Temperature Relationship and Initial Decomposition Temperature of Liquids by Isoteniscope
Standard Test Method for Vapor Pressure-Temperature Relationship and Initial Decomposition Temperature of Liquids by Isoteniscope
SIGNIFICANCE AND USE
5.1 The vapor pressure of a substance as determined by isoteniscope reflects a property of the sample as received including most volatile components, but excluding dissolved fixed gases such as air. Vapor pressure, per se, is a thermodynamic property which is dependent only upon composition and temperature for stable systems. The isoteniscope method is designed to minimize composition changes which may occur during the course of measurement.
SCOPE
1.1 This test method covers the determination of the vapor pressure of pure liquids, the vapor pressure exerted by mixtures in a closed vessel at 40 % ± 5 % ullage, and the initial thermal decomposition temperature of pure and mixed liquids. It is applicable to liquids that are compatible with borosilicate glass and that have a vapor pressure between 133 Pa (1.0 torr) and 101.3 kPa (760 torr) at the selected test temperatures. The test method is suitable for use over the range from ambient to 623 K. The temperature range may be extended to include temperatures below ambient provided a suitable constant-temperature bath for such temperatures is used.
Note 1: The isoteniscope is a constant-volume apparatus and results obtained with it on other than pure liquids differ from those obtained in a constant-pressure distillation.
1.2 Most petroleum products boil over a fairly wide temperature range, and this fact shall be recognized in discussion of their vapor pressures. Even an ideal mixture following Raoult's law will show a progressive decrease in vapor pressure as the lighter component is removed, and this is vastly accentuated in complex mixtures such as lubricating oils containing traces of dewaxing solvents, etc. Such a mixture may well exert a pressure in a closed vessel of as much as 100 times that calculated from its average composition, and it is the closed vessel which is simulated by the isoteniscope. For measurement of the apparent vapor pressure in open systems, Test Method D2878, is recommended.
1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard.
1.4 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use caution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national law. Users must determine legality of sales in their location.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see 6.10, 6.12, and Annex A2.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2023
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
Relations
- Effective Date
- 01-Dec-2023
- Refers
ASTM D4175-23a - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 15-Dec-2023
- Refers
ASTM D4175-23e1 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
- Referred By
ASTM D7863-22 - Standard Guide for Evaluation of Convective Heat Transfer Coefficient of Liquids - Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
Overview
ASTM D2879-23, "Standard Test Method for Vapor Pressure-Temperature Relationship and Initial Decomposition Temperature of Liquids by Isoteniscope," is a widely recognized method published by ASTM International. This standard specifies the procedure for determining the vapor pressure-temperature relationship and initial thermal decomposition temperature of liquids, both pure and mixed, using an isoteniscope apparatus. Measurement of vapor pressure and decomposition temperature is essential for evaluating the thermodynamic behavior, storage, and safety of liquid samples, especially those used in the chemical, petroleum, and materials industries.
The test method applies to liquids compatible with borosilicate glass and covers vapor pressures ranging from 133 Pa (1.0 torr) to 101.3 kPa (760 torr) over temperatures from below ambient to 623 K, subject to temperature bath capabilities.
Key Topics
Vapor Pressure Determination
The standard outlines the procedure for measuring the vapor pressure of liquids in a controlled, closed system, which reflects the sample’s composition and its most volatile components. Exclusion of dissolved fixed gases, such as air, ensures accurate vapor pressure data.Isoteniscope Technique
Utilizes a constant-volume isoteniscope apparatus to minimize changes in sample composition during measurement and simulates conditions found in closed containers or sealed vessels.Initial Decomposition Temperature
The test identifies the temperature at which the sample begins to thermally decompose, which is indicated by deviation from the linear relationship in a vapor pressure versus reciprocal temperature plot.Sample Compatibility
The method is suitable for a range of organic liquids, including many petroleum products and mixtures, provided they do not react adversely with borosilicate glass.Precision and Safety
Emphasizes the importance of methodical sample preparation, avoidance of mercury hazards, and adherence to safety protocols when handling vacuum, high temperature, and potentially hazardous substances.
Applications
ASTM D2879-23 serves critical functions in various sectors:
Petrochemical Industry
Determining vapor pressure of lubricating oils, fuels, solvents, and blends to ensure compliant storage, blending, and handling practices.Chemical Manufacturing
Assessing thermal stability and safe process temperatures for raw materials and formulations.Materials Research
Evaluating boiling range, volatility, and decomposition limits for development of new materials and specialty fluids.Regulatory Compliance and Safety
Supporting classification of liquids for transport, storage, and workplace regulations by providing accurate vapor pressure and decomposition data.
The method’s ability to analyze products that boil over a broad temperature range, such as complex petroleum mixtures, is a significant advantage for industries dealing with non-ideal solutions.
Related Standards
For comprehensive or comparative analysis, consider these related ASTM standards:
ASTM D2878 – Test Method for Estimating Apparent Vapor Pressures and Molecular Weights of Lubricating Oils
Suitable for open system vapor pressure estimation where D2879 simulates closed vessel conditions.ASTM D4175 – Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
Provides definitions and terminology used within D2879-23.ASTM E230 – Specification for Temperature-Electromotive Force Tables for Standardized Thermocouples
Supports accurate temperature measurement in isoteniscope setups.
ASTM D2879-23 is an internationally recognized procedure supporting critical quality, safety, and compliance needs associated with vapor pressure and decomposition temperature evaluation of liquids. Adherence to this standard ensures reliable and repeatable results across diverse applications.
Buy Documents
ASTM D2879-23 - Standard Test Method for Vapor Pressure-Temperature Relationship and Initial Decomposition Temperature of Liquids by Isoteniscope
REDLINE ASTM D2879-23 - Standard Test Method for Vapor Pressure-Temperature Relationship and Initial Decomposition Temperature of Liquids by Isoteniscope
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D2879-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Vapor Pressure-Temperature Relationship and Initial Decomposition Temperature of Liquids by Isoteniscope". This standard covers: SIGNIFICANCE AND USE 5.1 The vapor pressure of a substance as determined by isoteniscope reflects a property of the sample as received including most volatile components, but excluding dissolved fixed gases such as air. Vapor pressure, per se, is a thermodynamic property which is dependent only upon composition and temperature for stable systems. The isoteniscope method is designed to minimize composition changes which may occur during the course of measurement. SCOPE 1.1 This test method covers the determination of the vapor pressure of pure liquids, the vapor pressure exerted by mixtures in a closed vessel at 40 % ± 5 % ullage, and the initial thermal decomposition temperature of pure and mixed liquids. It is applicable to liquids that are compatible with borosilicate glass and that have a vapor pressure between 133 Pa (1.0 torr) and 101.3 kPa (760 torr) at the selected test temperatures. The test method is suitable for use over the range from ambient to 623 K. The temperature range may be extended to include temperatures below ambient provided a suitable constant-temperature bath for such temperatures is used. Note 1: The isoteniscope is a constant-volume apparatus and results obtained with it on other than pure liquids differ from those obtained in a constant-pressure distillation. 1.2 Most petroleum products boil over a fairly wide temperature range, and this fact shall be recognized in discussion of their vapor pressures. Even an ideal mixture following Raoult's law will show a progressive decrease in vapor pressure as the lighter component is removed, and this is vastly accentuated in complex mixtures such as lubricating oils containing traces of dewaxing solvents, etc. Such a mixture may well exert a pressure in a closed vessel of as much as 100 times that calculated from its average composition, and it is the closed vessel which is simulated by the isoteniscope. For measurement of the apparent vapor pressure in open systems, Test Method D2878, is recommended. 1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard. 1.4 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use caution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national law. Users must determine legality of sales in their location. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see 6.10, 6.12, and Annex A2. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The vapor pressure of a substance as determined by isoteniscope reflects a property of the sample as received including most volatile components, but excluding dissolved fixed gases such as air. Vapor pressure, per se, is a thermodynamic property which is dependent only upon composition and temperature for stable systems. The isoteniscope method is designed to minimize composition changes which may occur during the course of measurement. SCOPE 1.1 This test method covers the determination of the vapor pressure of pure liquids, the vapor pressure exerted by mixtures in a closed vessel at 40 % ± 5 % ullage, and the initial thermal decomposition temperature of pure and mixed liquids. It is applicable to liquids that are compatible with borosilicate glass and that have a vapor pressure between 133 Pa (1.0 torr) and 101.3 kPa (760 torr) at the selected test temperatures. The test method is suitable for use over the range from ambient to 623 K. The temperature range may be extended to include temperatures below ambient provided a suitable constant-temperature bath for such temperatures is used. Note 1: The isoteniscope is a constant-volume apparatus and results obtained with it on other than pure liquids differ from those obtained in a constant-pressure distillation. 1.2 Most petroleum products boil over a fairly wide temperature range, and this fact shall be recognized in discussion of their vapor pressures. Even an ideal mixture following Raoult's law will show a progressive decrease in vapor pressure as the lighter component is removed, and this is vastly accentuated in complex mixtures such as lubricating oils containing traces of dewaxing solvents, etc. Such a mixture may well exert a pressure in a closed vessel of as much as 100 times that calculated from its average composition, and it is the closed vessel which is simulated by the isoteniscope. For measurement of the apparent vapor pressure in open systems, Test Method D2878, is recommended. 1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard. 1.4 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use caution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national law. Users must determine legality of sales in their location. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see 6.10, 6.12, and Annex A2. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D2879-23 is classified under the following ICS (International Classification for Standards) categories: 13.300 - Protection against dangerous goods. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D2879-23 has the following relationships with other standards: It is inter standard links to ASTM D2879-18, ASTM D4175-23a, ASTM D4175-23e1, ASTM D7863-22, ASTM E1194-17, ASTM E2071-21, ASTM D7665-22, ASTM D8046-21. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D2879-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D2879 − 23
Standard Test Method for
Vapor Pressure-Temperature Relationship and Initial
Decomposition Temperature of Liquids by Isoteniscope
This standard is issued under the fixed designation D2879; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope* onstrated to be hazardous to health and corrosive to materials.
Use caution when handling mercury and mercury-containing
1.1 This test method covers the determination of the vapor
products. See the applicable product Safety Data Sheet (SDS)
pressure of pure liquids, the vapor pressure exerted by mixtures
for additional information. The potential exists that selling
in a closed vessel at 40 % 6 5 % ullage, and the initial thermal
mercury or mercury-containing products, or both, is prohibited
decomposition temperature of pure and mixed liquids. It is
by local or national law. Users must determine legality of sales
applicable to liquids that are compatible with borosilicate glass
in their location.
and that have a vapor pressure between 133 Pa (1.0 torr) and
1.5 This standard does not purport to address all of the
101.3 kPa (760 torr) at the selected test temperatures. The test
safety concerns, if any, associated with its use. It is the
method is suitable for use over the range from ambient to
responsibility of the user of this standard to establish appro-
623 K. The temperature range may be extended to include
priate safety, health, and environmental practices and deter-
temperatures below ambient provided a suitable constant-
mine the applicability of regulatory limitations prior to use.
temperature bath for such temperatures is used.
For specific warning statements, see 6.10, 6.12, and Annex A2.
NOTE 1—The isoteniscope is a constant-volume apparatus and results
1.6 This international standard was developed in accor-
obtained with it on other than pure liquids differ from those obtained in a
dance with internationally recognized principles on standard-
constant-pressure distillation.
ization established in the Decision on Principles for the
1.2 Most petroleum products boil over a fairly wide tem-
Development of International Standards, Guides and Recom-
perature range, and this fact shall be recognized in discussion
mendations issued by the World Trade Organization Technical
of their vapor pressures. Even an ideal mixture following
Barriers to Trade (TBT) Committee.
Raoult’s law will show a progressive decrease in vapor
pressure as the lighter component is removed, and this is vastly
2. Referenced Documents
accentuated in complex mixtures such as lubricating oils
2.1 ASTM Standards:
containing traces of dewaxing solvents, etc. Such a mixture
D2878 Test Method for Estimating Apparent Vapor Pres-
may well exert a pressure in a closed vessel of as much as 100
sures and Molecular Weights of Lubricating Oils
times that calculated from its average composition, and it is the
D4175 Terminology Relating to Petroleum Products, Liquid
closed vessel which is simulated by the isoteniscope. For
Fuels, and Lubricants
measurement of the apparent vapor pressure in open systems,
E230 Specification for Temperature-Electromotive Force
Test Method D2878, is recommended.
(emf) Tables for Standardized Thermocouples
1.3 The values stated in SI units are to be regarded as
standard. The values given in parentheses after SI units are
3. Terminology
provided for information only and are not considered standard.
3.1 Definitions:
1.4 WARNING—Mercury has been designated by many
3.1.1 For definitions of terms used in this test method, refer
regulatory agencies as a hazardous substance that can cause
to Terminology D4175.
serious medical issues. Mercury, or its vapor, has been dem-
3.2 Definitions of Terms Specific to This Standard:
3.2.1 ullage, n—that percentage of a closed system which is
filled with vapor.
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.L0.07 on Engineering Sciences of High Performance Fluids and
Solids (Formally D02.1100). For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2023. Published December 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1970. Last previous edition approved in 2018 as D2879 – 18. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D2879-23. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D2879 − 23
NOTE 2—Vapor pressures less than 133 Pa (1.0 torr), but greater than
3.2.1.1 Discussion—Specifically, on Fig. 1, that portion of
13.3 Pa (0.1 torr) at a selected test temperature can be determined directly
the volume of the isoteniscope to the right of point A that is
with reduced accuracy. In some cases the tendency of the sample to retain
filled with vapor.
dissolved or occluded air may prevent direct determinations of vapor
pressure in this range. In such cases, data points obtained at higher
3.3 Symbols:
pressures can be extrapolated to yield approximate vapor pressures in this
range.
C = temperature, °C,
K = temperature, K,
5. Significance and Use
p = pressure, Pa or torr,
P = experimentally measured total system pressure,
e
5.1 The vapor pressure of a substance as determined by
P = partial pressure due to fixed gases dissolved in sample,
a
isoteniscope reflects a property of the sample as received
P = corrected vapor pressure, Pa or torr.
c
including most volatile components, but excluding dissolved
t = time, s,
fixed gases such as air. Vapor pressure, per se, is a thermody-
K 5 C1273.15 (1) namic property which is dependent only upon composition and
temperature for stable systems. The isoteniscope method is
4. Summary of Test Method
designed to minimize composition changes which may occur
during the course of measurement.
4.1 Dissolved and entrained fixed gases are removed from
the sample in the isoteniscope by heating a thin layer of a
sample at reduced pressure, removing in this process the 6. Apparatus
minimum amount of volatile constituents from the sample.
6.1 Isoteniscope (Fig. 1).
4.2 The vapor pressure of the sample at selected tempera-
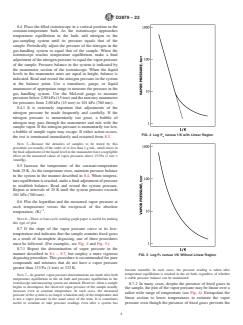
6.2 Constant-Temperature Air Bath—(Fig. 2) for use over
tures is determined by balancing the pressure due to the vapor
the temperature range from ambient to 623 K, controlled to
of the sample against a known pressure of an inert gas. The
62 K in the zone occupied by the isoteniscope beyond point
manometer section of the isoteniscope is used to determine
“A” (Fig. 1).
pressure equality.
6.3 Temperature Controller.
4.3 The initial decomposition temperature is determined
6.4 Vacuum and Gas Handling System (Fig. 3).
from a plot of the logarithm of the vapor pressure versus the
reciprocal of absolute temperature. The initial decomposition
6.5 Pressure Measurement Instrumentation—Pressure trans-
temperature is taken as that temperature at which the plot first ducers of suitable ranges are the preferred means for the
departs from linearity as a result of the decomposition of the
measurement of pressure in the gas handling system. Alterna-
sample. An optional method provides for the use of isothermal tively bourdon-type vacuum gauges or liquid manometers may
rates of pressure rise for this purpose (see Annex A1). These
be used. Note that more than one gauge or transducer may be
are measured at several temperatures and the logarithm of the required for use over the range of 2.00 kPa (15 torr) to 101 kPa
rate of pressure rise is plotted versus the reciprocal of absolute (760 torr) for pressures.
temperature. The decomposition temperature of the sample is
6.6 McLeod Vacuum Gauge—0 kPa to 2.00 kPa (0 torr to
taken to be that temperature at which the rate of increase of
15 torr), vertical primary standard type.
pressure is sufficient to produce a rise of 185 Pa
6.7 Mechanical Two-Stage Vacuum Pump.
(0.0139 torr ⁄s).
6.8 Direct Temperature Readout, either potentiometric or
electronic.
6.9 Thermocouple—in accordance with American National
Standard for Temperature Measurement Thermocouples (ANSI
C96.1) from Specification and Temperature Electromotive
Force Tables E230.
6.10 Nitrogen—pre-purified grade. (Warning—
Compressed gas under high pressure. Gas reduces oxygen
available for breathing. See A2.1.)
6.11 Nitrogen Pressure Regulator—single-stage, 0 kPa to
345 kPa gauge (0 psig to 50 psig).
6.12 Alcohol Lamp—(Warning—Flammable. Denatured
alcohol cannot be made nontoxic. See A2.2.)
7. Hazards
7.1 The apparatus includes a vacuum system and a Dewar
flask (constant temperature air bath) that is subjected to
elevated temperatures. Suitable means should be employed to
FIG. 1 Isoteniscope protect the operator from implosion of these systems. These
D2879 − 23
FIG. 3 Vacuum and Gas Handling System
(Warning—Compressed gas under high pressure. Gas reduces
oxygen available for breathing. See A1.2.). Repeat the evacu-
ation and purge of the system twice to remove residual oxygen.
8.2 Place the filled isoteniscope in a horizontal position so
that the sample spreads out into a thin layer in the sample bulb
and manometer section. Reduce the system pressure to 133 Pa
A Dewar, strip silvered, 110 mm ID by 400 mm deep.
(1 torr). Remove dissolved fixed gases by gently warming the
B Borosilicate glass tube, 90 mm OD by 320 mm long.
sample with an alcohol lamp until it just boils (Warning—
C Glass rod, ⁄8 in. in diameter by 310 mm long. Three of these heater element
holders are fused along their entire length to the outer surface of Tube B at
Flammable. Denatured alcohol cannot be made nontoxic. See
120 deg intervals. Slots cut into the fused glass rods on ⁄8 in. centers serve
A2.2.). Continue for 1 min.
as guides for the heating wire D.
D Resistance wire, B. and S. No. 21 gauge, spirally wrapped around Tube B
NOTE 3—During the initial evacuation of the system, it may be
and its attached guides.
necessary to cool volatile samples to prevent boiling or loss of volatiles.
E Glass wool pad.
NOTE 4—If the sample is a pure compound, complete removal of fixed
F Glass wool pad for centering Tube B and sealing annular opening.
gases may readily be accomplished by vigorous boiling at 13.3 Pa
G Lower plate of insulated isoteniscope holder.
(0.1 torr). For samples that consist of mixtures of substances differing in
Transite disk ⁄8 in. thick, loose fit in Tube B.
With hole for isoteniscope.
vapor pressure, this procedure is likely to produce an error due to the loss
H Upper plate of insulated isoteniscope holder.
of volatile components. Gentle boiling is to be preferred in such cases. The
Transite disk ⁄8 in. thick, loose fit in Dewar A.
rate of boiling during degassing may be controlled by varying both the
With hole for isoteniscope.
pressure at which the procedure is carried out and the amount of heating.
J Glass wool insulation between plates G and H.
In most cases, satisfactory degassing can be obtained at 133 Pa (1 torr).
K Plate spacer rods.
However, extremely viscous materials may require degassing at lower
L Heater leads connected to power output of temperature controller.
pressures. Samples of high volatility may have to be degassed at higher
T Temperature-control thermocouple affixed to inside wall of Tube B.
pressures. In the event that the vapor pressure data indicate that the
T Temperature-indicating thermocouple affixed to isoteniscope.
degassing procedure has not completely removed all dissolved gases, it
may be necessary to apply a correction to the data or to disregard data
FIG. 2 Constant-Temperature Air Bath
points that are so affected (see 8.7). The degassing procedure does not
prevent the loss of volatile sample components completely. However, the
means include wrapping of vacuum vessels, use of safety
described procedure minimizes such losses, so that for most purposes the
shield in front of Dewar flask, and use of safety glasses by the degassed sample can be considered to be representative of the original
sample less the fixed gases that have been removed.
operator.
8.3 After the sample has been degassed, close the vacuum
8. Procedure
line valve and turn the isoteniscope to return the sample to the
8.1 Add to the isoteniscope a quantity of sample sufficient to bulb and short leg of the manometer so that both are entirely
fill the sample bulb and the short leg of the manometer section filled with the liquid. Create a vapor-filled, nitrogen-free space
(Warning—Poison. Can be harmful or fatal if inhaled or between the bulb and the manometer in the following manner:
swallowed. Vapor harmful; emits toxic fumes when heated. maintain the pressure in the isoteniscope at the same pressure
Vapor pressure at normal room temperature exceeds threshold used f
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D2879 − 18 D2879 − 23
Standard Test Method for
Vapor Pressure-Temperature Relationship and Initial
Decomposition Temperature of Liquids by Isoteniscope
This standard is issued under the fixed designation D2879; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope*
1.1 This test method covers the determination of the vapor pressure of pure liquids, the vapor pressure exerted by mixtures in a
closed vessel at 40 % 6 5 % ullage, and the initial thermal decomposition temperature of pure and mixed liquids. It is applicable
to liquids that are compatible with borosilicate glass and that have a vapor pressure between 133 Pa (1.0 torr) and 101.3 kPa
(760 torr) at the selected test temperatures. The test method is suitable for use over the range from ambient to 623 K. The
temperature range may be extended to include temperatures below ambient provided a suitable constant-temperature bath for such
temperatures is used.
NOTE 1—The isoteniscope is a constant-volume apparatus and results obtained with it on other than pure liquids differ from those obtained in a
constant-pressure distillation.
1.2 Most petroleum products boil over a fairly wide temperature range, and this fact shall be recognized in discussion of their
vapor pressures. Even an ideal mixture following Raoult’s law will show a progressive decrease in vapor pressure as the lighter
component is removed, and this is vastly accentuated in complex mixtures such as lubricating oils containing traces of dewaxing
solvents, etc. Such a mixture may well exert a pressure in a closed vessel of as much as 100 times that calculated from its average
composition, and it is the closed vessel which is simulated by the isoteniscope. For measurement of the apparent vapor pressure
in open systems, Test Method D2878, is recommended.
1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for
information only and are not considered standard.
1.4 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious
medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use
Cautioncaution when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for
additional information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or
national law. Users must determine legality of sales in their location.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. For specific warning statements, see 6.10, 6.12, and Annex A2.
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.L0.07 on Engineering Sciences of High Performance Fluids and Solids (Formally D02.1100).
Current edition approved Dec. 1, 2018Dec. 1, 2023. Published December 2018December 2023. Originally approved in 1970. Last previous edition approved in 20102018
as D2879 – 10.D2879 – 18. DOI: 10.1520/D2879-18.10.1520/D2879-23.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D2879 − 23
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D2878 Test Method for Estimating Apparent Vapor Pressures and Molecular Weights of Lubricating Oils
D4175 Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
E230 Specification for Temperature-Electromotive Force (emf) Tables for Standardized Thermocouples
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this test method, refer to Terminology D4175.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 ullage—ullage, n—that percentage of a closed system which is filled with vapor.
3.2.1.1 Discussion—
Specifically, on Fig. 1, that portion of the volume of the isoteniscope to the right of point A that is filled with vapor.
FIG. 1 Isoteniscope
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
D2879 − 23
3.3 Symbols:
C = temperature, °C,
K = temperature, K,
p = pressure, Pa or torr,
P = experimentally measured total system pressure,
e
P = partial pressure due to fixed gases dissolved in sample,
a
P = corrected vapor pressure, Pa or torr.
c
t = time, s,
K 5 C1273.15 (1)
4. Summary of Test Method
4.1 Dissolved and entrained fixed gases are removed from the sample in the isoteniscope by heating a thin layer of a sample at
reduced pressure, removing in this process the minimum amount of volatile constituents from the sample.
4.2 The vapor pressure of the sample at selected temperatures is determined by balancing the pressure due to the vapor of the
sample against a known pressure of an inert gas. The manometer section of the isoteniscope is used to determine pressure equality.
4.3 The initial decomposition temperature is determined from a plot of the logarithm of the vapor pressure versus the reciprocal
of absolute temperature. The initial decomposition temperature is taken as that temperature at which the plot first departs from
linearity as a result of the decomposition of the sample. An optional method provides for the use of isothermal rates of pressure
rise for this purpose (see Annex A1). These are measured at several temperatures and the logarithm of the rate of pressure rise is
plotted versus the reciprocal of absolute temperature. The decomposition temperature of the sample is taken to be that temperature
at which the rate of increase of pressure is sufficient to produce a rise of 185 Pa (0.0139 torr ⁄s).
NOTE 2—Vapor pressures less than 133 Pa (1.0 torr), but greater than 13.3 Pa (0.1 torr) at a selected test temperature can be determined directly with
reduced accuracy. In some cases the tendency of the sample to retain dissolved or occluded air may prevent direct determinations of vapor pressure in
this range. In such cases, data points obtained at higher pressures can be extrapolated to yield approximate vapor pressures in this range.
5. Significance and Use
5.1 The vapor pressure of a substance as determined by isoteniscope reflects a property of the sample as received including most
volatile components, but excluding dissolved fixed gases such as air. Vapor pressure, per se, is a thermodynamic property which
is dependent only upon composition and temperature for stable systems. The isoteniscope method is designed to minimize
composition changes which may occur during the course of measurement.
6. Apparatus
6.1 Isoteniscope (Fig. 1).
6.2 Constant-Temperature Air Bath—(Fig. 2) for use over the temperature range from ambient to 623 K, controlled to 62 K in
the zone occupied by the isoteniscope beyond point “A” (Fig. 1).
6.3 Temperature Controller.
6.4 Vacuum and Gas Handling System (Fig. 3).
6.5 Pressure Measurement Instrumentation—Pressure transducers of suitable ranges are the preferred means for the measurement
of pressure in the gas handling system. Alternatively bourdon-type vacuum gauges or liquid manometers may be used. Note that
more than one gauge or transducer may be required for use over the range of 2.00 kPa (15 torr) to 101 kPa (760 torr) for pressures.
6.6 McLeod Vacuum Gauge—0 kPa to 2.00 kPa (0 torr to 15 torr), vertical primary standard type.
6.7 Mechanical Two-Stage Vacuum Pump.
D2879 − 23
A Dewar, strip silvered, 110 mm ID by 400 mm deep.
B Borosilicate glass tube, 90 mm OD by 320 mm long.
C Glass rod, ⁄8 in. in diameter by 310 mm long. Three of these heater element
holders are fused along their entire length to the outer surface of Tube B at
120 deg intervals. Slots cut into the fused glass rods on ⁄8 in. centers serve
as guides for the heating wire D.
D Resistance wire, B. and S. No. 21 gauge, spirally wrapped around Tube B
and its attached guides.
E Glass wool pad.
F Glass wool pad for centering Tube B and sealing annular opening.
G Lower plate of insulated isoteniscope holder.
Transite disk ⁄8 in. thick, loose fit in Tube B.
With hole for isoteniscope.
H Upper plate of insulated isoteniscope holder.
Transite disk ⁄8 in. thick, loose fit in Dewar A.
With hole for isoteniscope.
J Glass wool insulation between plates G and H.
K Plate spacer rods.
L Heater leads connected to power output of temperature controller.
T Temperature-control thermocouple affixed to inside wall of Tube B.
T Temperature-indicating thermocouple affixed to isoteniscope.
FIG. 2 Constant-Temperature Air Bath
6.8 Direct Temperature Readout, either potentiometric or electronic.
6.9 Thermocouple—in accordance with American National Standard for Temperature Measurement Thermocouples (ANSI C96.1)
from Specification and Temperature Electromotive Force Tables E230.
6.10 Nitrogen—pre-purified grade. (Warning—Compressed gas under high pressure. Gas reduces oxygen available for breathing.
See A2.1.)
6.11 Nitrogen Pressure Regulator —Regulator—single-stage, 0 kPa to 345 kPa gauge (0 psig to 50 psig).
6.12 Alcohol Lamp—(Warning—Flammable. Denatured alcohol cannot be made nontoxic. See A2.2.)
D2879 − 23
FIG. 3 Vacuum and Gas Handling System
7. Hazards
7.1 The apparatus includes a vacuum system and a Dewar flask (constant temperature air bath) that is subjected to elevated
temperatures. Suitable means should be employed to protect the operator from implosion of these systems. These means include
wrapping of vacuum vessels, use of safety shield in front of Dewar flask, and use of safety glasses by the operator.
8. Procedure
8.1 Add to the isoteniscope a quantity of sample sufficient to fill the sample bulb and the short leg of the manometer section
(Warning—Poison. Can be harmful or fatal if inhaled or swallowed. Vapor harmful; emits toxic fumes when heated. Vapor
pressure at normal room temperature exceeds threshold limit value for occupational exposure. See A1.1.) to point A of Fig. 1.
Attach the isoteniscope to the vacuum system as shown in Fig. 3, and evacuate both the system and the filled isoteniscope to a
pressure of 13.3 Pa (0.1 torr) as measured on the McLeod gauge. Break the vacuum with nitrogen (Warning—Compressed gas
under high pressure. Gas reduces oxygen available for breathing. See A1.2.). Repeat the evacuation and purge of the system twice
to remove residual oxygen.
8.2 Place the filled isoteniscope in a horizontal position so that the sample spreads out into a thin layer in the sample bulb and
manometer section. Reduce the system pressure to 133 Pa (1 torr). Remove dissolved fixed gases by gently warming the sample
with an alcohol lamp until it just boils (Warning—Flammable. Denatured alcohol cannot be made nontoxic. See A2.2.). Continue
for 1 min.
NOTE 3—During the initial evacuation of the system, it may be necessary to cool volatile samples to prevent boiling or loss of volatiles.
NOTE 4—If the sample is a pure compound, complete removal of fixed gases may readily be accomplished by vigorous boiling at 13.3 Pa (0.1 torr). For
samples that consist of mixtures of substances differing in vapor pressure, this procedure is likely to produce an error due to the loss of volatile
components. Gentle boiling is to be preferred in such cases. The rate of boiling during degassing may be controlled by varying both the pressure at which
the procedure is carried out and the amount of heating. In most cases, satisfactory degassing can be obtained at 133 Pa (1 torr). However, extremely
viscous materials may require degassing at lower pressures. Samples of high volatility may have to be degassed at higher pressures. In the event that the
vapor pressure data indicate that the degassing procedure has not completely removed all dissolved gases, it may be necessary to apply a correction to
the data or to disregard data points that are so affected (see 8.7). The degassing procedure does not prevent the loss of volatile sample components
completely. However, the described procedure minimizes such losses, so that for most purposes the degassed sample can be considered to be
representative of the original sample less the fixed gases that have been removed.
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...