ASTM D3558-15(2023)
(Test Method)Standard Test Methods for Cobalt in Water
Standard Test Methods for Cobalt in Water
SIGNIFICANCE AND USE
4.1 Most waters rarely contain more than trace concentrations of cobalt from natural sources. Although trace amounts of cobalt seem to be essential to the nutrition of some animals, large amounts have pronounced toxic effects on both plant and animal life.
SCOPE
1.1 These test methods cover the determination of dissolved and total recoverable cobalt in water and wastewater 2 by atomic absorption spectrophotometry. Three test methods are included as follows:
Concentration Range
Sections
Test Method A—Atomic Absorption, Direct
0.1 mg/L to 10 mg/L
7 to 16
Test Method B—Atomic Absorption, Chelation-Extraction
10 μg/L to 1000 μg/L
17 to 26
Test Method C—Atomic Absorption, Graphite Furnace
5 μg/L to 100 μg/L
27 to 36
1.2 Test Method A has been used successfully with reagent water, potable water, river water, and wastewater. Test Method B has been used successfully with reagent water, potable water, river water, sea water and brine. Test Method C was successfully evaluated in reagent water, artificial seawater, river water, tap water, and a synthetic brine. It is the analyst's responsibility to ensure the validity of these test methods for other matrices.
1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see 11.8.1, 21.12, and 23.10.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2023
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jun-2015
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
Overview
ASTM D3558-15(2023) - Standard Test Methods for Cobalt in Water specifies reliable procedures for detecting and quantifying cobalt in various water matrices. Developed by ASTM International, this standard outlines three analytical approaches using atomic absorption spectrophotometry to determine both dissolved and total recoverable cobalt in water and wastewater samples. The accurate measurement of cobalt is essential given that, while trace amounts are nutritionally important to some animals, higher concentrations are toxic to both plant and animal life. As most natural waters contain cobalt only at trace levels, adhering to recognized testing methods ensures data consistency, regulatory compliance, and environmental safety.
Key Topics
Test Methods Covered:
- Direct Atomic Absorption (Method A): For concentrations from 0.1 mg/L to 10 mg/L.
- Chelation-Extraction Method (Method B): Sensitive detection from 10 µg/L to 1000 µg/L.
- Graphite Furnace Atomic Absorption (Method C): Highly sensitive, suitable for 5 µg/L to 100 µg/L.
Water Types Assessed:
- Method A: Reagent, potable, river, and wastewater
- Method B: Reagent, potable, river, sea water, and brine
- Method C: Reagent water, artificial seawater, river water, tap water, and synthetic brine
Ensuring Accuracy and Precision:
- Use of calibration standards, method blanks, laboratory control samples, and independent reference materials
- Addressing interferences (e.g., effects of nitrate and other metals)
- Quality control protocols including calibration verification and matrix spike recoveries
Safety and Compliance:
- Recommendations for appropriate safety and environmental practices
- Sample preservation and preparation guidance to preserve integrity and reliability of results
Applications
Environmental Monitoring:
- Assessment of surface waters, groundwaters, and effluents to comply with environmental regulations for trace metals.
- Monitoring of industrial discharges and municipal wastewater for cobalt content.
Drinking Water Quality:
- Ensures safe levels of cobalt in potable water sources, supporting public health mandates.
Industrial and Process Waters:
- Quality control for process water in industries where cobalt contamination might be a concern.
- Essential for industries such as mining, metal plating, and electronics manufacturing.
Research and Laboratory Analysis:
- Provides reproducible, standardized test methods for academic and commercial laboratory workflows requiring cobalt quantification.
Related Standards
Sampling and Terminology:
- ASTM D3370: Practices for Sampling Water from Flowing Process Streams
- ASTM D1066: Practice for Sampling Steam
- ASTM D1129: Terminology Relating to Water
Trace Elements in Water:
- ASTM D1886: Test Methods for Nickel in Water
- ASTM D1687: Test Methods for Chromium in Water
- ASTM D3557: Test Methods for Cadmium in Water
- ASTM D1688: Test Methods for Copper in Water
- ASTM D3559: Test Methods for Lead in Water
- ASTM D1691: Test Methods for Zinc in Water
- ASTM D1068: Test Methods for Iron in Water
- ASTM D858: Test Methods for Manganese in Water
Quality Control and Analytical Practices:
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3919: Practice for Measuring Trace Elements in Water by Graphite Furnace Atomic Absorption Spectrophotometry
- ASTM D4841: Practice for Estimation of Holding Time for Water Samples
- ASTM D5810: Guide for Spiking into Aqueous Samples
- ASTM D5847: Practice for Writing Quality Control Specifications for Water Analysis
Practical Value
The ASTM D3558-15(2023) standard enables laboratories, regulatory agencies, and industries to measure cobalt in water through validated, internationally recognized methodologies. This supports regulatory compliance, environmental protection, and public health initiatives by ensuring trace cobalt levels in water can be accurately monitored and managed. With its flexible options for detection range and water matrix compatibility, this standard is a critical resource for any water testing laboratory or facility handling industrial water analysis involving trace cobalt determination.
Buy Documents
ASTM D3558-15(2023) - Standard Test Methods for Cobalt in Water
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D3558-15(2023) is a standard published by ASTM International. Its full title is "Standard Test Methods for Cobalt in Water". This standard covers: SIGNIFICANCE AND USE 4.1 Most waters rarely contain more than trace concentrations of cobalt from natural sources. Although trace amounts of cobalt seem to be essential to the nutrition of some animals, large amounts have pronounced toxic effects on both plant and animal life. SCOPE 1.1 These test methods cover the determination of dissolved and total recoverable cobalt in water and wastewater 2 by atomic absorption spectrophotometry. Three test methods are included as follows: Concentration Range Sections Test Method A—Atomic Absorption, Direct 0.1 mg/L to 10 mg/L 7 to 16 Test Method B—Atomic Absorption, Chelation-Extraction 10 μg/L to 1000 μg/L 17 to 26 Test Method C—Atomic Absorption, Graphite Furnace 5 μg/L to 100 μg/L 27 to 36 1.2 Test Method A has been used successfully with reagent water, potable water, river water, and wastewater. Test Method B has been used successfully with reagent water, potable water, river water, sea water and brine. Test Method C was successfully evaluated in reagent water, artificial seawater, river water, tap water, and a synthetic brine. It is the analyst's responsibility to ensure the validity of these test methods for other matrices. 1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see 11.8.1, 21.12, and 23.10. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Most waters rarely contain more than trace concentrations of cobalt from natural sources. Although trace amounts of cobalt seem to be essential to the nutrition of some animals, large amounts have pronounced toxic effects on both plant and animal life. SCOPE 1.1 These test methods cover the determination of dissolved and total recoverable cobalt in water and wastewater 2 by atomic absorption spectrophotometry. Three test methods are included as follows: Concentration Range Sections Test Method A—Atomic Absorption, Direct 0.1 mg/L to 10 mg/L 7 to 16 Test Method B—Atomic Absorption, Chelation-Extraction 10 μg/L to 1000 μg/L 17 to 26 Test Method C—Atomic Absorption, Graphite Furnace 5 μg/L to 100 μg/L 27 to 36 1.2 Test Method A has been used successfully with reagent water, potable water, river water, and wastewater. Test Method B has been used successfully with reagent water, potable water, river water, sea water and brine. Test Method C was successfully evaluated in reagent water, artificial seawater, river water, tap water, and a synthetic brine. It is the analyst's responsibility to ensure the validity of these test methods for other matrices. 1.3 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see 11.8.1, 21.12, and 23.10. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D3558-15(2023) is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3558-15(2023) has the following relationships with other standards: It is inter standard links to ASTM D3558-15, ASTM D1129-13(2020)e1, ASTM D1129-13(2020)e2, ASTM D3559-15, ASTM D1068-15, ASTM D1687-17, ASTM D4382-18, ASTM D3557-17, ASTM D857-17, ASTM D1886-14(2021)e1, ASTM D1688-17, ASTM D1971-16(2021)e1, ASTM D858-17, ASTM D1691-17, ASTM D3645-15(2023). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3558-15(2023) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D3558 − 15 (Reapproved 2023)
Standard Test Methods for
Cobalt in Water
This standard is issued under the fixed designation D3558; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 These test methods cover the determination of dissolved
D858 Test Methods for Manganese in Water
and total recoverable cobalt in water and wastewater by
D1066 Practice for Sampling Steam
atomic absorption spectrophotometry. Three test methods are
D1068 Test Methods for Iron in Water
included as follows:
D1129 Terminology Relating to Water
Concentration Range Sections
Test Method A—Atomic Absorption, 0.1 mg ⁄L to 10 mg ⁄L 7 to 16 D1193 Specification for Reagent Water
Direct
D1687 Test Methods for Chromium in Water
Test Method B—Atomic Absorption, 10 μg ⁄L to 1000 μg ⁄L 17 to 26
D1688 Test Methods for Copper in Water
Chelation-Extraction
D1691 Test Methods for Zinc in Water
Test Method C—Atomic Absorption, 5 μg ⁄L to 100 μg/L 27 to 36
Graphite Furnace
D1886 Test Methods for Nickel in Water
D2777 Practice for Determination of Precision and Bias of
1.2 Test Method A has been used successfully with reagent
Applicable Test Methods of Committee D19 on Water
water, potable water, river water, and wastewater. Test Method
D3370 Practices for Sampling Water from Flowing Process
B has been used successfully with reagent water, potable water,
Streams
river water, sea water and brine. Test Method C was success-
D3557 Test Methods for Cadmium in Water
fully evaluated in reagent water, artificial seawater, river water,
D3559 Test Methods for Lead in Water
tap water, and a synthetic brine. It is the analyst’s responsibility
D3919 Practice for Measuring Trace Elements in Water by
to ensure the validity of these test methods for other matrices.
Graphite Furnace Atomic Absorption Spectrophotometry
1.3 The values stated in SI units are to be regarded as
D4841 Practice for Estimation of Holding Time for Water
standard. The values given in parentheses are mathematical
Samples Containing Organic and Inorganic Constituents
conversions to inch-pound units that are provided for informa-
D5673 Test Method for Elements in Water by Inductively
tion only and are not considered standard.
Coupled Plasma—Mass Spectrometry
1.4 This standard does not purport to address all of the D5810 Guide for Spiking into Aqueous Samples
safety concerns, if any, associated with its use. It is the D5847 Practice for Writing Quality Control Specifications
responsibility of the user of this standard to establish appro- for Standard Test Methods for Water Analysis
priate safety, health, and environmental practices and deter-
3. Terminology
mine the applicability of regulatory limitations prior to use.
For specific hazard statements, see 11.8.1, 21.12, and 23.10.
3.1 Definitions:
1.5 This international standard was developed in accor- 3.1.1 For definitions of terms used in these test methods,
refer to Terminology D1129.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the 3.2 Definitions of Terms Specific to This Standard:
Development of International Standards, Guides and Recom- 3.2.1 total recoverable cobalt, n—a descriptive term relating
mendations issued by the World Trade Organization Technical to the cobalt forms recovered in the acid-digestion procedure
Barriers to Trade (TBT) Committee. specified in these test methods.
4. Significance and Use
4.1 Most waters rarely contain more than trace concentra-
These test methods are under the jurisdiction of ASTM Committee D19 on
tions of cobalt from natural sources. Although trace amounts of
Water and are the direct responsibility of Subcommittee D19.05 on Inorganic
Constituents in Water.
Current edition approved Dec. 1, 2023. Published December 2023. Originally
approved in 1977. Last previous edition approved in 2015 as D3558 – 15. DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/D3558-15R23. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Platte, J. A., and March, V. M., “A New Tool for the Water Chemist,” Industrial Standards volume information, refer to the standard’s Document Summary page on
Water Engineering, May 1965. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3558 − 15 (2023)
cobalt seem to be essential to the nutrition of some animals, following hydrochloric-nitric acid digestion and filtration. The
large amounts have pronounced toxic effects on both plant and same digestion procedure may be used to determine total
animal life. recoverable nickel (Test Methods D1886), chromium (Test
Methods D1687), cadmium (Test Methods D3557), copper
5. Purity of Reagents
(Test Methods D1688), iron (Test Methods D1068), lead (Test
5.1 Reagent grade chemicals shall be used in all tests. Methods D3559), manganese (Test Methods D858), zinc (Test
Methods D1691).
Unless otherwise indicated, it is intended that all reagents shall
conform to the specifications of the Committee on Analytical
9. Interferences
Reagents of the American Chemical Society where such
9.1 Nitrate reportedly interferes at 1 mg/L by suppressing
specifications are available. Other grades may be used, pro-
the absorption of the cobalt. This interference can be elimi-
vided it is first ascertained that the reagent is of sufficiently
nated by adding 18 000 mg/L of ammonium chloride to
high purity to permit its use without lessening the accuracy of
blanks, standards, and samples.
the determination.
9.2 Other metals usually do not interfere in the determina-
5.2 Unless otherwise indicated, reference to water shall be
understood to mean reagent water conforming to Specification tion of cobalt by increasing or decreasing the amount of
absorbed radiation. The most common interference is caused
D1193, Type I. Other reagent water types may be used,
provided it is first ascertained that the water is of sufficiently by a chemical reaction in the flame that prevents conversion of
the cobalt to the atomic state.
high purity to permit its use without lessening the bias and
precision of the determination. Type II water specified at the
9.3 Sodium, potassium, and sulfate, magnesium (4500 mg/
time of the round-robin testing of this test method.
L), iron (4000 mg/L), and nickel, lead, copper, zinc, cadmium,
and chromium (10 mg/L each) do not interfere.
6. Sampling
9.4 Background correction or a chelation-extraction proce-
6.1 Collect the samples in accordance with Practices D3370
dure (see Test Method B) may be necessary to determine low
and Practice D1066, as applicable.
levels of cobalt in some water.
6.2 Preserve samples with HNO (sp gr 1.42) to a pH of 2 or
NOTE 2—Instrument manufacturers’ instructions for use of the specific
less immediately at the time of collection, normally about
correction technique should be followed.
2 mL ⁄L. If only dissolved cobalt is to be determined, filter the
sample through a 0.45 μm membrane filter before acidification. 10. Apparatus
The holding time for samples may be calculated in accordance
10.1 Atomic Absorption Spectrophotometer, for use at
with Practice D4841.
240.7 nm.
NOTE 1—Alternatively, the pH may be adjusted in the laboratory if the
NOTE 3—The manufacturer’s instructions should be followed for all
sample is returned within 14 days. However, acid must be added at least
instrumental parameters. A wavelength other than 240.7 nm may be used
24 hours before analysis to dissolve any metals that adsorb to the container
if it has been determined to be equally suitable.
walls. This could reduce hazards of working with acids in the field when
appropriate. 10.2 Cobalt Hollow-Cathode Lamps—Multielement
hollow-cathode lamps.
TEST METHOD A—ATOMIC ABSORPTION, DIRECT
10.3 Pressure-Reducing Valves—The supplies of fuel and
7. Scope
oxidant shall be maintained at pressures somewhat higher than
the controlled operating pressure of the instrument by suitable
7.1 This test method covers the determination of dissolved
valves.
and total recoverable cobalt in most waters and waste waters.
It is the user’s responsibility to ensure the validity of this test
11. Reagents and Materials
method in a particular matrix.
11.1 Cobalt Solution, Stock (1 mL = 1.0 mg Co)—
7.2 This test method is applicable in the range from
Commercially purchase or dissolve 4.0372 g of cobaltous
0.1 mg ⁄L to 10 mg ⁄L of cobalt. The range may be extended to
chloride (CoCl ·6H O) in reagent water and dilute to 1 L. A
2 2
concentrations greater than 10 mg/L by dilution of the sample.
purchased cobalt stock solution of appropriate known purity is
also acceptable.
8. Summary of Test Method
11.2 Cobalt Solution, Standard (1 mL = 0.1 mg Co)—Dilute
8.1 Cobalt is determined by atomic absorption spectropho-
100.0 mL of the stock cobalt solution to 1 L with water.
tometry. Dissolved cobalt is determined by aspirating a portion
11.3 Filter Paper—Purchase suitable filter paper. Typically
of the filtered sample directly with no pretreatment. Total
the filter papers have a pore size of 0.45 μm membrane.
recoverable cobalt is determined by aspirating the sample
Material such as fine-textured, acid-washed, ashless paper, or
glass fiber paper are acceptable. The user must first ascertain
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
that the filter paper is of sufficient purity to use without
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by the American Chemical adversely affecting the bias and precision of the test method.
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
11.4 Hydrochloric Acid (sp gr 1.19)—Concentrated hydro-
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. chloric acid (HCl).
D3558 − 15 (2023)
NOTE 4—If a high reagent blank is obtained, distill the HCl or use NOTE 7—For samples with high levels of suspended matter, the amount
spectrograde acid. When HCl is distilled, an azeotrophic mixture is of reduction in volume is left to the discretion of the analyst.
obtained (approximately 6 N HCl is formed). Therefore, whenever NOTE 8—Many laboratories have found block digestion systems a
concentrated HCl is specified for the preparation of a reagent or in the useful way to digest samples for trace metals analysis. Systems typically
procedure, use double the volume specified if distilled acid is used. consist of either a metal or graphite block with wells to hold digestion
tubes. The block temperature controller must be able to maintain unifor-
11.5 Nitric Acid (sp gr 1.42)—Concentrated nitric acid
mity of temperature across all positions of the block. For trace metals
(HNO ).
analysis, the digestion tubes should be constructed of polypropylene and
have a volume accuracy of at least 0.5 %. All lots of tubes should come
NOTE 5—If a high reagent blank is obtained, distill the HNO or use a
with a certificate of analysis to demonstrate suitability for their intended
spectrograde acid.
purpose.
11.6 Nitric Acid (1 + 499)—Add 1 volume of HNO (sp gr
13.4 Cool and filter the samples through a suitable filter
1.42) to 499 volumes of water.
(11.3), such as fine-texture, acid-washed, ashless paper, into
11.7 Oxidant:
100 mL volumetric flasks. Wash the filter paper two or three
11.7.1 Air, which has been passed through a suitable filter to
times with water and bring to volume.
remove oil, water, and other foreign substances is the usual
13.5 Atomize each filtered and acidified sample and deter-
oxidant.
mine its absorbance or concentration. Atomize HNO (1 + 499)
11.8 Fuel:
between samples.
11.8.1 Acetylene—Standard, commercially available acety-
14. Calculation
lene is the usual fuel. Acetone, always present in acetylene
cylinders can affect analytical results. The cylinder should be
14.1 Calculate the concentration of cobalt in the sample, in
replaced at 345 kPa (50 psi). (“Purified” grade acetylene
milligrams per litre, using an analytical curve or, alternatively,
containing a special proprietary solvent rather than acetone
read directly in concentration (see 12.4).
should not be used with poly(vinyl chloride) tubing as weak-
ening of the tubing walls can cause a potentially hazardous 15. Precision and Bias
situation.)
15.1 The single-operator and overall precision of this test
method within its designated range for eleven laboratories,
12. Standardization
which include a total of sixteen operators (at 1.0 mg ⁄L,
12.1 Prepare 100 mL each of a blank and at least four
4.0 mg ⁄L, and 8.0 mg ⁄L) analyzing each sample on three
standard solutions to bracket the expected cobalt concentration
different days varies with the quantity being tested in accor-
range of the samples to be analyzed by diluting the standard
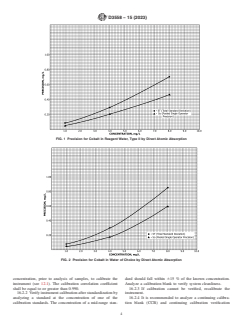
dance with Fig. 1 and Fig. 2.
cobalt solution (see 11.2) with HNO (1 + 499). Analyze at
15.2 Recoveries of known amounts of cobalt in a series of
least four working standards containing concentrations of
prepared standards for the same laboratories were as shown in
cobalt that bracket the expected sample concentration, prior to
Table 1.
analysis of samples, to calibrate the instrument. Prepare the
standards each time the test is to be performed or as determined 15.3 These precision and bias data may not apply to waters
of different matrices. The waters used in evaluations were
by Practice D4841.
reagent water, potable water, river water, and wastewater. It is
12.2 When determining total recoverable cobalt, add 0.5 mL
the user’s responsibility to ensure the validity of this test
of HNO (sp gr 1.42) (11.5) and proceed as directed in 13.2
method for waters of untested matrices.
through 13.4. When determining dissolved cobalt, proceed
15.4 This section on precision and bias conforms to Practice
with 12.3.
D2777 – 77 which was in place at the time of collaborative
12.3 Aspirate the blank and the standards and record the
testing. Under the allowances made in 1.4 of Practice
instrument readings. Aspirate HNO (1 + 499) between stan-
D2777 – 13, these precision and bias data do meet existing
dards.
requirements of interlaboratory studies of Committee D19 test
12.4 Prepare an analytical curve by plotting the absorbance
methods.
versus concentration for each standard on linear graph paper.
Alternatively, read directly in concentration if this capability is
16. Quality Control
provided with the instrument.
16.1 In order to be certain that analytical values obtained
using these test methods are valid and accurate within the
13. Procedure
confidence limits of the test, the following QC procedures must
13.1 Measure 100.0 mL of a well-mixed acidified sample
be followed when analyzing cobalt.
into a 125 mL beaker or flask.
16.2 Calibration and Calibration Verification:
NOTE 6—If only dissolved cobalt is to be determined, proceed from
16.2.1 Analyze at least four working standards containing
13.5.
concentrations of cobalt that bracket the expected sample
13.2 Add 5 mL of HCl (sp gr 1.19) (11.4) to each sample.
13.3 Heat the samples on a steam bath or hotplate in a
Supporting data have been filed at ASTM International Headquarters and may
well-ventilated hood until the volume has been reduced to
be obtained by requesting Research Report RR:D19-1032. Contact ASTM Customer
15 mL to 20 mL, making certain that the samples do not boil. Service at service@astm.org.
D3558 − 15 (2023)
FIG. 1 Precision for Cobalt in Reagent Water, Type II by Direct Atomic Absorption
FIG. 2 Precision for Cobalt in Water of Choice by Direct Atomic Absorption
concentration, prior to analysis of samples, to calibrate the dard should fall within 615 % of the known concentration.
instrument (see 12.1). The calibration correlation coefficient Analyze a calibration blank to verify system cleanliness.
shall be equal to or greater than 0.990. 16.2.3 If calibration cannot be verified, recalibrate the
16.2.2 Verify instrument calibration after standardization by instrument.
analyzing a standard at the concentration of one of the 16.2.4 It is recommended to analyze a continuing calibra-
calibration standards. The concentration of a mid-range stan- tion blank (CCB) and continuing calibration verification
D3558 − 15 (2023)
TABLE 1 Recoveries of Known Amounts of Cobalt Atomic
level, analysis of samples is halted until the contamination is
Absorption, Direct
eliminated, and a blank shows no contamination at or above
Statistically
this level, or the results must be qualified with an indication
Amount Amount Significant
that they do not fall within the performance criteria of the test
Added, Found, Bias, % (95 %
method.
mg/L mg/L Confidence
Level)
16.6 Matrix Spike (MS):
Reagent water, Type II 1.00 1.03 +3.00 Yes
4.00 3.98 −0.50 No
16.6.1 To check for interferences in the specific matrix
8.00 8.00 0.00 No
being tested, perform a MS on at least one sample from each
Water of choice 1.00 1.01 +1.00 No
laboratory-defined batch by spiking an aliquot of the sample
4.00 4.01 +0.25 No
8.00 8.12 +1.50 No
with a known concentration of cobalt and taking it through the
analytical method.
16.6.2 The spike concentration plus the background concen-
(CCV) at a 10 % frequency. The results should fall within the
tration of cobalt must not exceed the high calibration standard.
expected precision of the method or 615 % of the known
The spike must produce a concentration in the spiked sample
concentration.
that is 2 to 5 times the analyte concentration in the unspiked
16.3 Initial Demonstration of Laboratory Capability: sample, or 10 to 50 times the detection limit of the test method,
whichever is greater.
16.3.1 If a laboratory has not performed the test before, or if
there has been a major change in the measurement system, for 16.6.3 Calculate the percent recovery of the spike (P) using
the following calculation:
example, new analyst, new instrument, and so forth, a precision
and bias study must be performed to demonstrate laboratory
P 5 @A~V 1V! 2 BV #/CV (1)
s s
capability.
where:
16.3.2 Analyze seven replicates of a standard solution
prepared from an Independent Reference Material containing a A = analyte known concentration (mg/L) in spiked sample,
B = analyte known concentration (mg/L) in unspiked
midrange concentration of cobalt. The matrix and chemistry of
sample,
the solution should be equivalent to the solution used in the
C = known concentration (mg/L) of analyte in spiking
collaborative study. Each replicate must be taken through the
solution,
complete analytical test method including any sample preser-
V = volume (mL) of sample used, and
vation and pretreatment steps. s
V = volume (mL) of spiking solution added.
16.3.3 Calculate the mean and standard deviation of the
seven values and compare to the acceptable ranges of bias in
16.6.4 The percent recovery of the spike shall fall within the
Table 1. This study should be repeated until the recoveries are
limits, based on the analyte concentration, listed in Guide
within the limits given in Table 1. If a concentration other than
D5810, Table 1. If the percent recovery is not within these
the recommended concentration is used, refer to Practice
limits, a matrix interference may be present in the sample
D5847 for information on applying the F test and t test in
selected for spiking. Under these circumstances, one of the
evaluating the acceptability of the mean and standard devia-
following remedies must be employed: the matrix interference
tion.
must be removed, all samples in the batch must be analyzed by
a test method not affected by the matrix interference, or the
16.4 Laboratory Control Sample (LCS):
results must be qualified with an indication that they do not fall
16.4.1 To ensure that the test method is in control, prepare
within the performance criteria of the test method.
and analyze a LCS containing a known concentration of cobalt
with each batch (laboratory-defined or twenty samples). The
NOTE 9—Acceptable spike recoveries are dependent on the concentra-
laboratory control samples for a large batch should cover the
tion of the component of interest. See Guide D5810 for additional
information.
analytical range when possible. The LCS must be taken
through all of the steps of the analytical method including
16.7 Duplicate:
sample preservation and pretreatment. The result obtained for
16.7.1 To check the precision of sample analyses, analyze a
a mid-range LCS shall fall within 615 % of the known
sample in duplicate with each laboratory-defined batch. If the
concentration.
concentration of the analyte is less than five times the detection
16.4.2 If the result is not within these limits, analysis of
limit for the analyte, a matrix spike duplicate (MSD) should be
samples is halted until the problem is corrected, and either all
used.
the samples in the batch must be reanalyzed, or the results must
16.7.2 Calculate the standard deviation of the duplicate
be qualified with an indication that they do not fall within the
values and compare to the precision in the collaborative study
performance criteria of the test method.
using an F test. Refer to 6.4.4 of Practice D5847 for informa-
16.5 Method Blank: tion on applying the F test.
16.5.1 Analyze a reagent water test blank with each 16.7.3 If the result exceeds the precision limit, the batch
laboratory-defined batch. The concentration of cobalt found in must be reanalyzed or the results must be qualified with an
the blank should be less than 0.5 times the lowest calibration indication that they do not fall within the performance criteria
standard. If the concentration of cobalt is found above this of the test method.
D3558 − 15 (2023)
16.8 Independent Reference Material (IRM): 21.6 Cobalt Solution, Stock (1.0 mL = 1.0 mg Co)—See
16.8.1 In order to verify the quantitative value produced by 11.1.
the test method, analyze an Independent Reference Material
21.7 Filter Paper—See 11.3.
(IRM) submitted as a regular sample (if practical) to the
21.8 Hydrochloric Acid (sp gr 1.19)—Concentrated hydro-
laboratory at least once per quarter. The concentration of the
chloric acid (HCl) (“Purified” grade acetylene containing a
IRM should be in the concentration mid-range for the method
special proprietary solvent rather than acetone should not be
chosen. The value obtained must fall within the control limits
used with poly(vinyl chloride) tubing as weakening of the
established by the laboratory.
tubing walls can cause a potentially hazardous situation).
TEST METHOD B—ATOMIC
21.9 Hydrochloric Acid (1 + 2)—Add 1 volume of HCl (sp
ABSORPTION, CHELATION-EXTRACTION
gr 1.19) to 2 volumes of water.
21.10 Hydrochloric Acid (1 + 49)—Add 1 volume of HCl
17. Scope
(sp gr 1.19) to 49 volumes of water.
17.1 This test method covers the determination of dissolved
21.11 Nitric Acid (sp gr 1.42)—Concentrated nitric acid
and total recoverable cobalt in most waters and brines. It is the
(HNO ) (“Purified” grade acetylene containing a special pro-
user’s responsibility to ensure the validity of this test method in
prietary solvent rather than acetone should not be used with
a particular matrix.
poly(vinyl chloride) tubing as weakening of the tubing walls
17.2 This test method is applicable in the range from
can cause a potentially hazardous situation).
10 μg ⁄L to 1000 μg ⁄L of cobalt. The range may be extended to
21.12 Pyrrolidine Dithiocarbamic Acid-Chloroform
concentrations greater than 1000 μg/L by dilution of the
Reagent—Add 36 mL of pyrrolidine to 1 L of CHCl . Cool the
samples.
solution and add 30 mL of CS (21.2) in small portions
swirling between additions. Dilute to 2 L with CHCl . The
18. Summary of Test Method
reagent can be used for several months if stored in a cool, dark
18.1 Cobalt is determined by atomic absorption spectropho-
place. (All components of this reagent are highly toxic. Carbon
tometry. The element, either dissolved or total recoverable, is
disulfide is also flammable. Prepare and use in a well-
chelated with pyrrolidine dithiocarbamic acid and extracted
ventilated hood. Avoid inhalation and direct contact.)
with chloroform. The extract is evaporated to dryness, treated
with HCl, and diluted to a specified volume with water. A 21.13 Sodium Hydroxide Solution (100 g/L)—Dissolve
portion of the resulting solution is then aspirated into the
100 g of sodium hydroxide (NaOH) in water and dilute to 1 L.
air-acetylene flame of the spectrophotometer. The digestion (Warning—This is a very exothermic reaction.)
procedure summarized in 8.1 is used to determine total
21.14 Materials—See 11.7 and 11.8.
recoverable cobalt. The same chelation-extraction procedure
may be used to determine nickel (Test Methods D1886),
22. Standardization
chromium (Test Methods D1687), copper (Test Methods
22.1 Prepare 100 mL each of a blank and sufficient stan-
D1688), iron (Test Methods D1068), lead (Test Methods
dards containing from 0.0 μg to 100 μg of cobalt from the
D3559), and zinc (Test Methods D1691).
cobalt standard solution (see 21.5) by making appropriate
dilutions with water. Analyze at least three working standards
19. Interferences
containing concentrations of cobalt that bracket the expected
19.1 See Section 9.
sample concentration, prior to analysis of samples, to calibrate
the instrument. Prepare standards immediately prior to use or
20. Apparatus
as determined by Practice D4841.
20.1 All items of apparatus described in Section 10 are
22.2 When determining total recoverable cobalt use 125 mL
required.
beakers or flasks. Add 0.5 mL of HNO (sp gr 1.42) and
proceed as directed in 23.2 to 23.15.
21. Reagents and Materials
22.3 Prepare an analytical curve by plotting the absorbances
21.1 Bromphenol Blue Indicator Solution (1 g/L)—Dissolve
of standards versus micrograms of cobalt. Alternatively, read
0.1 g of bromphenol blue in 100 mL of 50 % ethanol or
directly in concentration if this capability is provided with the
2-propanol.
instrument.
21.2 Carbon Disulfide (CS ).
21.3 Chloroform (CHCl ).
23. Procedure
21.4 Cobalt Solution, Intermediate (1.0 mL = 100 μg Co)— 23.1 Measure a volume of a well-mixed acidified sample
See 11.2.
containing less than 100 μg of cobalt (100 mL maximum) into
a 125 mL beaker or flask and adjust the volume to 100 mL with
21.5 Cobalt Solution, Standard (1.0 mL = 1 μg Co)—Dilute
water.
10 mL of cobalt solution, intermediate, and 1 mL of HNO (sp
gr 1.42) to 1 L with water. This standard is used to prepare
NOTE 10—If only dissolved cobalt is to be determined measure a
working standards at the time of analysis. volume of filtered (21.7) and acidified sample containing less than 100 μg
D3558 − 15 (2023)
of cobalt (100 mL maximum) into a 250 mL separatory f
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...