ASTM E2548-16
(Guide)Standard Guide for Sampling Seized Drugs for Qualitative and Quantitative Analysis
Standard Guide for Sampling Seized Drugs for Qualitative and Quantitative Analysis
SIGNIFICANCE AND USE
3.1 This guide provides information for the sampling of seized-drug submissions.
3.2 The principal purpose of sampling in the context of this guide is to answer relevant questions about a population by examination of a portion of the population. For example:
What is the net weight of the population?
What portion of the units of a population can be said to contain a given drug at a given level of confidence?
3.3 By developing a sampling strategy and implementing appropriate sampling schemes, as illustrated in Fig. 1, a laboratory will minimize the total number of required analytical determinations, while ensuring that all relevant legal and scientific requirements are met.
SCOPE
1.1 This guide covers minimum considerations for sampling of seized drugs for qualitative and quantitative analysis.
1.2 This guide cannot replace knowledge, skill, or ability acquired through appropriate education, training, and experience and should be used in conjunction with sound professional judgment.
General Information
- Status
- Published
- Publication Date
- 29-Feb-2016
- Technical Committee
- E30 - Forensic Sciences
- Drafting Committee
- E30.01 - Criminalistics
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 15-Jan-2019
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Jun-2018
- Effective Date
- 15-May-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Dec-2014
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Apr-2013
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Oct-2011
Overview
ASTM E2548-16: Standard Guide for Sampling Seized Drugs for Qualitative and Quantitative Analysis provides minimum considerations for the sampling of seized drugs intended for forensic qualitative and quantitative analysis. Developed by ASTM International’s Committee E30 on Forensic Sciences, this guide assists laboratories and professionals in establishing robust and defensible sampling strategies that meet both scientific and legal requirements. It emphasizes proper sampling to ensure that analytical results are representative and that the total number of required analytical determinations is minimized.
Key Topics
- Purpose of Sampling: The primary objective of sampling seized drug submissions is to answer pertinent questions about the whole population by examining only a portion. Key considerations include determining the net weight and the proportion of units containing a specific drug at a certain confidence level.
- Sampling Strategies: Laboratories must develop their own sampling strategies to meet specific investigative, legal, and customer requirements. The use of statistical or non-statistical sampling plans should be considered based on the intended inference and applicable law.
- Statistical vs Non-Statistical Sampling:
- Statistical sampling is used when results must be extrapolated to the entire population, requiring a probability-based approach and appropriate statistical analysis.
- Non-statistical sampling may be adequate if no inference about the whole population is required or if driven by specific legal directives.
- Population Determination: The sampling scheme must account for all typical forms and quantities in which seized drugs may appear-whether as a single unit or multiple units. Homogeneity of bulk samples must be addressed.
- Random Sampling: Random samples ensure unbiased selection and are key for statistical inference. Methods such as random number tables or the “black box” approach can be used to achieve this.
- Analysis and Reporting: Each selected sample must be analyzed according to standardized analytical practices. Reports must clearly state the basis of inference-whether statistical or limited to tested units.
- Documentation: All steps from population determination to sample selection and analysis must be thoroughly documented to support legal scrutiny and scientific rigor.
Applications
ASTM E2548-16 is essential for forensic laboratories, law enforcement agencies, and professionals handling seized drug analysis. By following this guide, organizations can:
- Ensure that sampling methods are scientifically valid and legally defensible.
- Reduce analytical workload while maintaining confidence in results.
- Support judicial processes by providing robust, well-documented evidence about seized substances.
- Address varying scenarios such as bulk seizures or multiple container submissions with appropriate sampling strategies.
- Facilitate compliance with legal mandates, especially where particular methods of sampling or reporting are required by regulation.
The guide is particularly valuable for those seeking to balance efficiency with the need for high-confidence results in forensic drug analysis.
Related Standards
For comprehensive implementation, ASTM E2548-16 references several important standards:
- ASTM E105 - Practice for Probability Sampling of Materials
- ASTM E122 - Practice for Calculating Sample Size
- ASTM E141 - Practice for Acceptance of Evidence Based on Probability Sampling Results
- ASTM E1732 - Terminology Relating to Forensic Science
- ASTM E2329 - Practice for Identification of Seized Drugs
- ASTM E2334 - Practice for Setting Upper Confidence Bounds (Zero Response Sample Data)
- ISO 3534-1 & ISO 3534-2 - Statistics Vocabulary and Symbols
By aligning with these referenced documents, laboratories strengthen the integrity of their sampling procedures and bolster overall forensic quality assurance.
Keywords: seized drugs, forensic analysis, sampling guide, qualitative analysis, quantitative analysis, random sampling, statistical sampling, ASTM E2548-16, drug evidence, forensic science standards.
Buy Documents
ASTM E2548-16 - Standard Guide for Sampling Seized Drugs for Qualitative and Quantitative Analysis
REDLINE ASTM E2548-16 - Standard Guide for Sampling Seized Drugs for Qualitative and Quantitative Analysis
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E2548-16 is a guide published by ASTM International. Its full title is "Standard Guide for Sampling Seized Drugs for Qualitative and Quantitative Analysis". This standard covers: SIGNIFICANCE AND USE 3.1 This guide provides information for the sampling of seized-drug submissions. 3.2 The principal purpose of sampling in the context of this guide is to answer relevant questions about a population by examination of a portion of the population. For example: What is the net weight of the population? What portion of the units of a population can be said to contain a given drug at a given level of confidence? 3.3 By developing a sampling strategy and implementing appropriate sampling schemes, as illustrated in Fig. 1, a laboratory will minimize the total number of required analytical determinations, while ensuring that all relevant legal and scientific requirements are met. SCOPE 1.1 This guide covers minimum considerations for sampling of seized drugs for qualitative and quantitative analysis. 1.2 This guide cannot replace knowledge, skill, or ability acquired through appropriate education, training, and experience and should be used in conjunction with sound professional judgment.
SIGNIFICANCE AND USE 3.1 This guide provides information for the sampling of seized-drug submissions. 3.2 The principal purpose of sampling in the context of this guide is to answer relevant questions about a population by examination of a portion of the population. For example: What is the net weight of the population? What portion of the units of a population can be said to contain a given drug at a given level of confidence? 3.3 By developing a sampling strategy and implementing appropriate sampling schemes, as illustrated in Fig. 1, a laboratory will minimize the total number of required analytical determinations, while ensuring that all relevant legal and scientific requirements are met. SCOPE 1.1 This guide covers minimum considerations for sampling of seized drugs for qualitative and quantitative analysis. 1.2 This guide cannot replace knowledge, skill, or ability acquired through appropriate education, training, and experience and should be used in conjunction with sound professional judgment.
ASTM E2548-16 is classified under the following ICS (International Classification for Standards) categories: 11.100.30 - Analysis of blood and urine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2548-16 has the following relationships with other standards: It is inter standard links to ASTM E1732-24, ASTM E141-10(2023), ASTM E1732-19, ASTM E1732-18b, ASTM E1732-18a, ASTM E1732-18, ASTM E141-10(2018), ASTM E1732-17, ASTM E2329-14, ASTM E2334-09(2013)e2, ASTM E2334-09(2013), ASTM E2334-09(2013)e1, ASTM E1732-12, ASTM E1732-11b, ASTM E1732-11a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2548-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2548 − 16 An American National Standard
Standard Guide for
Sampling Seized Drugs for Qualitative and Quantitative
Analysis
This standard is issued under the fixed designation E2548; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope ISO 3534-2 Statistics – Vocabulary and Symbols – Part 2:
Statistical Quality Control
1.1 Thisguidecoversminimumconsiderationsforsampling
of seized drugs for qualitative and quantitative analysis.
3. Significance and Use
1.2 This guide cannot replace knowledge, skill, or ability
3.1 This guide provides information for the sampling of
acquired through appropriate education, training, and experi-
seized-drug submissions.
ence and should be used in conjunction with sound profes-
sional judgment.
3.2 The principal purpose of sampling in the context of this
guide is to answer relevant questions about a population by
1.3 This international standard was developed in accor-
examination of a portion of the population. For example:
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
What is the net weight of the population?
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
What portion of the units of a population can be said to contain a
given drug at a given level of confidence?
Barriers to Trade (TBT) Committee.
3.3 By developing a sampling strategy and implementing
2. Referenced Documents
appropriate sampling schemes, as illustrated in Fig. 1,a
2.1 ASTM Standards: laboratory will minimize the total number of required analyti-
cal determinations, while ensuring that all relevant legal and
E105 Guide for Probability Sampling of Materials
E122 Practice for Calculating Sample Size to Estimate,With scientific requirements are met.
Specified Precision, the Average for a Characteristic of a
Lot or Process 4. Sampling Strategy
E141 Practice for Acceptance of Evidence Based on the
4.1 Anappropriatesamplingstrategyishighlydependenton
Results of Probability Sampling
the purpose of the investigation, the customer’s request, and
E1732 Terminology Relating to Forensic Science
theanticipateduseoftheresults.Lawsandlegalpracticesform
E2329 Practice for Identification of Seized Drugs
the foundation of most strategies and shall be taken into
E2334 Practice for Setting an Upper Confidence Bound for a
account when designing a sampling scheme. Therefore, spe-
Fraction or Number of Non-Conforming items, or a Rate
cific sampling strategies are not defined in this guide.
of Occurrence for Non-Conformities, Using Attribute
4.2 The laboratory has the responsibility to develop its own
Data, When There is a Zero Response in the Sample
strategiesconsistentwiththisguide.Itisrecommendedthatthe
2.2 ISO Standards:
following key points be addressed:
ISO 3534-1 Statistics – Vocabulary and Symbols – Part 1:
4.2.1 Sampling may be statistical or non-statistical.
Probability and General Statistical Terms
NOTE 1—For the purpose of this guide, the use of the term statistical is
meant to include the notion of an approach that is probability-based.
This guide is under the jurisdiction of ASTM Committee E30 on Forensic
4.2.1.1 In many cases, a non-statistical approach may suf-
Sciences and is the direct responsibility of Subcommittee E30.01 on Criminalistics.
fice. The sampling plan shall provide an adequate basis for
Current edition approved March 1, 2016. Published April 2016. Originally
answering questions of applicable law. For example,
approved in 2007. Last previous version approved in 2011 as E2548– 11. DOI:
10.1520/E2548-16.
Is there a drug present in the population?
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Are statutory enhancement levels satisfied by the analysis of a
Standards volume information, refer to the standard’s Document Summary page on
specified number of units?
the ASTM website.
4.2.1.2 If an inference about the whole population is to be
Available from International Organization for Standardization (ISO), 1 rue de
Varembé, Case postale 56, CH-1211, Geneva 20, Switzerland, http://www.iso.ch. drawn from a sample, then the plan shall be either statistically
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2548 − 16
FIG. 1 Relationship of Various Levels Required in Sampling
based or have an appropriate statistical analysis completed and
The probability that a given percentage of the population contains
the drug of interest or is positive for a given characteristic.
limits of the inference shall be documented.
4.2.2 Selected units shall be analyzed to meet Practice
The total net weight of the population is to be extrapolated from the
E2329 if statistical inferences are to be made about the whole
weight of a sample.
population.
5.5.1.1 Published examples of statistical approaches involv-
ing general considerations:
5. Sampling Scheme
(1) Practice E105.
5.1 The sampling scheme is an overall approach that in-
(2) Practice E141.
cludespopulationdetermination,selectionofthesamplingplan
(3) Terminology E1732.
andprocedureand,whenappropriate,samplereductionpriorto
(4)Guidelines on Representative Drug Sampling.
analysis (Fig. 2).
(5) ISO 3534-1.
(6) ISO 3534-2.
5.2 Population Determination:
5.5.1.2 Published examples of statistical approaches involv-
5.2.1 The population determination shall take into account
ing the hypergeometric, normal, and other distributions from a
all typical forms and quantities in which exhibits may appear.
frequentist perspective:
5.2.2 A population can consist of a single unit or multiple
(1) Frank, et al., Journal of Forensic Sciences.
units.
(2)Guidelines on Representative Drug Sampling.
5.2.3 A multiple unit population shall consist of items that
(3) Practice E2334.
are similar in relevant visual characteristics (for example, size,
(4) Practice E122.
color, shape, etc.).
5.5.1.3 Published examples of statistical approaches involv-
SAMPLING PLAN
ing the hypergeometric, nor
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: E2548 − 11 E2548 − 16
Standard Guide for
Sampling Seized Drugs for Qualitative and Quantitative
Analysis
This standard is issued under the fixed designation E2548; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Editorial corrections were made to Sections 1 and 8 in December 2011.
1. Scope
1.1 This guide covers minimum considerations for sampling of seized drugs for qualitative and quantitative analysis.
1.2 This guide cannot replace knowledge, skill, or ability acquired through appropriate education, training, and experience and
should be used in conjunction with sound professional judgment.
2. Referenced Documents
2.1 ASTM Standards:
E105 Practice for Probability Sampling of Materials
E122 Practice for Calculating Sample Size to Estimate, With Specified Precision, the Average for a Characteristic of a Lot or
Process
E141 Practice for Acceptance of Evidence Based on the Results of Probability Sampling
E1732 Terminology Relating to Forensic Science
E2329 Practice for Identification of Seized Drugs
E2334 Practice for Setting an Upper Confidence Bound For a Fraction or Number of Non-Conforming items, or a Rate of
Occurrence for Non-conformities, Using Attribute Data, When There is a Zero Response in the Sample
2.2 ISO Standards:
ISO 3534-1 Statistics – Vocabulary and Symbols – Part 1: Probability and General Statistical Terms
ISO 3534-2 Statistics – Vocabulary and Symbols – Part 2: Statistical Quality Control
3. Significance and Use
3.1 This guide provides information for the sampling of seized-drug submissions.
3.2 The principal purpose of sampling in the context of this guide is to answer relevant questions about a population by
examination of a portion of the population. For example:
What is the net weight of the population?
What portion of the units of a population can be said to contain a
given drug at a given level of confidence?
3.3 By developing a sampling strategy and implementing appropriate sampling schemes, as illustrated in Fig. 1, a laboratory
will minimize the total number of required analytical determinations, while ensuring that all relevant legal and scientific
requirements are met.
4. Sampling Strategy
4.1 AAn appropriate sampling strategy is highly dependent on the purpose of the investigation, the original question,customer’s
request, and the ultimateanticipated use of the results. Laws and legal practices form the foundation of most strategies and shall
be taken into account when designing a sampling scheme. Therefore, specific sampling strategies are not defined in this guide.
This guide is under the jurisdiction of ASTM Committee E30 on Forensic Sciences and is the direct responsibility of Subcommittee E30.01 on Criminalistics.
Current edition approved Sept. 1, 2011March 1, 2016. Published October 2011April 2016. Originally approved in 2007. Last previous version approved in 20072011 as
E2548– 07.– 11. DOI: 10.1520/E2548-11E01.10.1520/E2548-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from International Organization for Standardization (ISO), 1 rue de Varembé, Case postale 56, CH-1211, Geneva 20, Switzerland, http://www.iso.ch.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2548 − 16
FIG. 1 Relationship of Various Levels Required in Sampling
4.2 The laboratory has the responsibility to develop its own strategies consistent with these recommendations.this guide. It is
recommended that the following key points be addressed:
4.2.1 Sampling may be statistical or non-statistical.
NOTE 1—For the purpose of this guide, the use of the term statistical is meant to include the notion of an approach that is probability-based.
4.2.1.1 In many cases, a non-statistical approach may suffice. The sampling plan shall provide an adequate basis for answering
questions of applicable law. For example,
Is there a drug present in the population?
Are statutory enhancement levels satisfied by the analysis of a
specified number of units?
4.2.1.2 If an inference about the whole population is to be drawn from a sample, then the plan shall be statistically based either
statistically based or have an appropriate statistical analysis completed and limits of the inference shall be documented.
4.2.2 Statistically selected Selected units shall be analyzed to meet Practice E2329 if statistical inferences are to be made about
the whole population.
5. Sampling Scheme
5.1 The sampling scheme is an overall approach that includes population determination, selection of the sampling plan and
procedure and, when appropriate, sample reduction prior to analysis (Fig. 2).
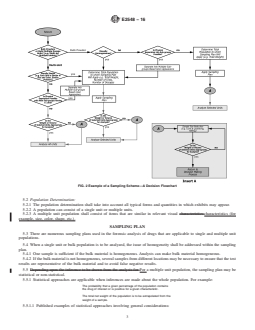
E2548 − 16
FIG. 2 Example of a Sampling Scheme—A Decision Flowchart
5.2 Population Determination:
5.2.1 The population determination shall take into account all typical forms and quantities in which exhibits may appear.
5.2.2 A population can consist of a single unit or multiple units.
5.2.3 A multiple unit population shall consist of items that are similar in relevant visual characteristics.characteristics (for
example, size, color, shape, etc.).
SAMPLING PLAN
5.3 There are numerous sampling plans used in the forensic analysis of drugs that are applicable to single and multiple unit
populations.
5.4 When a single unit or bulk population is to be analyzed, the issue of homogeneity shall be addressed within the sampling
plan.
5.4.1 One sample is sufficient if the bulk material is homogeneous. Analysts can make bulk material homogeneous.
5.4.2 If the bulk material is not homogeneous, several samples from different locations may be necessary to ensure that the test
results are representative of the bulk material and to avoid false negative results.
5.5 Depending upon the inference to be drawn from the analysis for For a multiple unit population, the sampling plan may be
statistical or non-statistical.
5.5.1 Statistical approaches are applicable when inferences are made abo
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...