ASTM F750-20
(Practice)Standard Practice for Evaluating Acute Systemic Toxicity of Material Extracts by Systemic Injection in the Mouse
Standard Practice for Evaluating Acute Systemic Toxicity of Material Extracts by Systemic Injection in the Mouse
SIGNIFICANCE AND USE
4.1 This practice is intended to help assess the biocompatibility of materials used in medical devices. It is an acute toxicological test designed to detect the presence of injurious leachable substances.
4.2 This practice may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the materials being tested, their potential applications, and the recommendations contained in Practice F748.
4.3 The only limitation applicable is the extract preparation. Refer to Sections 4.3 and 4.4 of Practice F619 for a description of this limitation.
SCOPE
1.1 This practice covers a nonspecific, acute toxicity test used for detecting leachables from materials used in medical devices.
1.2 The liquids injected into the mouse are those obtained by Practice F619 where the extraction vehicles are saline, vegetable oil, or other liquids simulating human body fluids.
1.3 Two procedures are outlined: Method A for intravenous injection and Method B for intraperitoneal injection.
1.4 This practice is one of several developed for the assessment of the biocompatibility of materials. Practice F748 may provide guidance for the selection of appropriate methods for testing materials for a specific application.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.16 - Biocompatibility Test Methods
Relations
- Effective Date
- 01-Jun-2020
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Aug-2008
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-May-2004
- Effective Date
- 01-Nov-2003
- Effective Date
- 10-Apr-2002
- Effective Date
- 10-Aug-1998
- Effective Date
- 10-Dec-1997
- Effective Date
- 01-Jun-2020
- Effective Date
- 01-Jun-2020
Overview
ASTM F750-20 - Evaluating Acute Systemic Toxicity of Material Extracts by Systemic Injection in the Mouse is an internationally recognized standard developed by ASTM International. The purpose of this practice is to assess the acute systemic toxicity of leachables from materials used in medical devices. By utilizing this procedure, laboratories and manufacturers can determine the biocompatibility and safety profile of materials through the observation of test mice injected with extracts derived from candidate medical device materials. This standard is especially significant in medical device development, where ensuring material biocompatibility is critical to patient safety.
Key Topics
- Acute Toxicity Testing: Measures potential toxic effects of material extracts when administered systemically to mice, providing a model for detecting injurious substances leachable from device materials.
- Extract Preparation: Follows ASTM F619, using extraction vehicles such as saline or vegetable oil to mimic body fluids. Extracts are injected intravenously (Method A) or intraperitoneally (Method B) to assess toxicity.
- Biocompatibility Evaluation: F750-20 supports the broader objective of verifying that medical device materials do not introduce unsafe substances into the body.
- Observation Protocol: Test mice are observed over a 72-hour period for clinical signs, including changes in body condition, behavior, physiological responses, and mortality.
- Results Interpretation: Criteria are established for acceptable outcomes and adverse responses. Results guide regulatory and quality decisions on the use of materials in medical devices.
- Reporting Requirements: Comprehensive documentation of sample characteristics, extraction conditions, animal data, and observed effects is required for traceability and compliance.
Applications
The ASTM F750-20 standard plays a crucial role in the preclinical safety assessment of materials used in medical devices. Common applications include:
- Medical Device Development: Ensures that polymers, plastics, and other materials used in device components are free from acutely toxic leachables before they reach clinical trials or the market.
- Regulatory Submissions: F750-20 data is frequently requested by regulatory agencies to demonstrate compliance with biocompatibility and safety requirements for new or modified medical devices.
- Material Screening: Used by manufacturers and suppliers to screen raw materials and additives for acute toxicity prior to selection for product use.
- Quality Assurance: Supports ongoing quality control and risk management activities in the medical device supply chain.
By following this standard, organizations can demonstrate due diligence in material selection and contribute to safer medical products for patient care.
Related Standards
Several other important ASTM standards are referenced and complement F750-20:
- ASTM F619: Practice for Extraction of Medical Plastics-Defines procedures for preparing extracts from medical-grade materials, forming the foundation for the systemic injection test.
- ASTM F748: Practice for Selecting Generic Biological Test Methods for Materials and Devices-Offers guidance on selecting appropriate biocompatibility evaluation methods, including when to use F750-20.
- International Guidelines: The standard aligns with principles from the World Trade Organization's Technical Barriers to Trade (TBT) Committee, fostering international acceptance in regulatory frameworks.
Keywords: acute toxicity, biocompatibility, medical device materials, systemic injection, mouse model, leachables, ASTM F750-20, material safety testing, intravenous injection, intraperitoneal injection.
Buy Documents
ASTM F750-20 - Standard Practice for Evaluating Acute Systemic Toxicity of Material Extracts by Systemic Injection in the Mouse
REDLINE ASTM F750-20 - Standard Practice for Evaluating Acute Systemic Toxicity of Material Extracts by Systemic Injection in the Mouse
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F750-20 is a standard published by ASTM International. Its full title is "Standard Practice for Evaluating Acute Systemic Toxicity of Material Extracts by Systemic Injection in the Mouse". This standard covers: SIGNIFICANCE AND USE 4.1 This practice is intended to help assess the biocompatibility of materials used in medical devices. It is an acute toxicological test designed to detect the presence of injurious leachable substances. 4.2 This practice may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the materials being tested, their potential applications, and the recommendations contained in Practice F748. 4.3 The only limitation applicable is the extract preparation. Refer to Sections 4.3 and 4.4 of Practice F619 for a description of this limitation. SCOPE 1.1 This practice covers a nonspecific, acute toxicity test used for detecting leachables from materials used in medical devices. 1.2 The liquids injected into the mouse are those obtained by Practice F619 where the extraction vehicles are saline, vegetable oil, or other liquids simulating human body fluids. 1.3 Two procedures are outlined: Method A for intravenous injection and Method B for intraperitoneal injection. 1.4 This practice is one of several developed for the assessment of the biocompatibility of materials. Practice F748 may provide guidance for the selection of appropriate methods for testing materials for a specific application. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This practice is intended to help assess the biocompatibility of materials used in medical devices. It is an acute toxicological test designed to detect the presence of injurious leachable substances. 4.2 This practice may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the materials being tested, their potential applications, and the recommendations contained in Practice F748. 4.3 The only limitation applicable is the extract preparation. Refer to Sections 4.3 and 4.4 of Practice F619 for a description of this limitation. SCOPE 1.1 This practice covers a nonspecific, acute toxicity test used for detecting leachables from materials used in medical devices. 1.2 The liquids injected into the mouse are those obtained by Practice F619 where the extraction vehicles are saline, vegetable oil, or other liquids simulating human body fluids. 1.3 Two procedures are outlined: Method A for intravenous injection and Method B for intraperitoneal injection. 1.4 This practice is one of several developed for the assessment of the biocompatibility of materials. Practice F748 may provide guidance for the selection of appropriate methods for testing materials for a specific application. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F750-20 is classified under the following ICS (International Classification for Standards) categories: 11.220 - Veterinary medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F750-20 has the following relationships with other standards: It is inter standard links to ASTM F750-87(2012), ASTM F748-16, ASTM F748-06(2010), ASTM F619-03(2008), ASTM F748-06, ASTM F748-04, ASTM F619-03, ASTM F619-02, ASTM F748-98, ASTM F619-79(1997)e1, ASTM E3219-20, ASTM F2148-18. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F750-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F750 − 20
Standard Practice for
Evaluating Acute Systemic Toxicity of Material Extracts by
Systemic Injection in the Mouse
ThisstandardisissuedunderthefixeddesignationF750;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F748 PracticeforSelectingGenericBiologicalTestMethods
for Materials and Devices
1.1 This practice covers a nonspecific, acute toxicity test
used for detecting leachables from materials used in medical
3. Summary of Practice
devices.
3.1 The extract liquid is prepared in accordance with Prac-
1.2 The liquids injected into the mouse are those obtained
tice F619. The extraction vehicles are saline and vegetable oil,
by Practice F619 where the extraction vehicles are saline,
or other extraction vehicles, as described in Practice F619.The
vegetable oil, or other liquids simulating human body fluids.
extract liquid is injected into mice, and the animals are
1.3 Two procedures are outlined: MethodAfor intravenous
observed at regular intervals for 72 h for reactions, survival,
injection and Method B for intraperitoneal injection.
etc.
1.4 This practice is one of several developed for the
4. Significance and Use
assessment of the biocompatibility of materials. Practice F748
may provide guidance for the selection of appropriate methods 4.1 This practice is intended to help assess the biocompat-
for testing materials for a specific application. ibility of materials used in medical devices. It is an acute
toxicological test designed to detect the presence of injurious
1.5 The values stated in SI units are to be regarded as
leachable substances.
standard. No other units of measurement are included in this
standard. 4.2 This practice may not be appropriate for all types of
implant applications. The user is cautioned to consider the
1.6 This standard does not purport to address all of the
appropriateness of the method in view of the materials being
safety concerns, if any, associated with its use. It is the
tested, their potential applications, and the recommendations
responsibility of the user of this standard to establish appro-
contained in Practice F748.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
4.3 The only limitation applicable is the extract preparation.
1.7 This international standard was developed in accor-
Refer to Sections 4.3 and 4.4 of Practice F619 for a description
dance with internationally recognized principles on standard-
of this limitation.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom- 5. Apparatus
mendations issued by the World Trade Organization Technical
5.1 Mice—The mice shall be albino-type, healthy, and not
Barriers to Trade (TBT) Committee.
previously used, and shall weigh between 17 and 23 g.Animal
care shall be in accordance with the “Guide for the Care and
2. Referenced Documents
Use of Laboratory Animals.” Age, sex, and weight shall be
2.1 ASTM Standards:
recorded and reported.All the mice for each extraction vehicle
F619 Practice for Extraction of Medical Plastics
shall be from the same source. For each extraction vehicle, a
minimum of ten mice are used in the test. If the results of this
first test group are inconclusive, then 20 more mice will be
ThispracticeisunderthejurisdictionofASTMCommitteeF04onMedicaland
needed to complete the test of one extraction vehicle for one
Surgical Materials and Devices and is the direct responsibility of Subcommittee
plastic.
F04.16 on Biocompatibility Test Methods.
Current edition approved June 1, 2020. Published August 2020. Originally
5.1.1 During the test the mice shall be fed normally with
approved in 1982. Last previous edition approved in 2012 as F750 – 87 (2012).
commercially available feed and tap water.
DOI: 10.1520/F0750-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on NationalResearchCouncil,GuidefortheCareandUseofLaboratoryAnimals,
the ASTM website. National Academy Press, Washington, DC, 2011.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F750 − 20
5.2 Cages—There shall be one cage for the five mice Weigh all mice, and record their weights. Use a system of
exposed to one extract liquid. Each mouse in a cage shall be marking to identify each individual mouse within each group
uniquely identified, and this identification shall be recorded. of five.
Male and female mice shall be housed separately, and their 8.1.4 Inject the predetermined amount (see 7.2.1)ofthe
cages positioned in a manner which prevents the accidental sample extract liquid into the tail vein of each of the five mice.
transfer of feces or bedding from cage to cage. Inject the blank extract liquid in the same way into five other
mice.Theuseofwarmwateroraheatlampmayhelpdilatethe
5.3 Syringe—Sterile syringes, not greater than 3 mL in
tail veins for ease of injection.
volume, with a precision of 60.10 mL shall be used.
8.1.5 Observe all animals immediately after injection, again
5.3.1 Method A—Sterile needles of 25 to 27 ⁄2 gauge shall
4 h after injection, and then at 24, 48, and 72 h, respectively,
be used.
after injection for clinical signs. Cage-side observations should
5.3.2 Method B—Sterile needles of 21 to 26 gauge shall be
include, but not be limited to, changes in body condition,
used.
skin/hair coat, gait/posture, eyes, respiratory, gastrointestinal,
and consciousness/mentation using the descriptors provided in
6. Sampling
Table 1. Record the observations. Measure and record the body
weights of all animals at 24, 48, and 72 h post-injection.
6.1 Sample in accordance with Practice F619.
8.2 Method B, Intraperitoneal:
7. Sample and Test Specimen
8.2.1 Method B shall be used with vegetable oil and similar
extraction vehicles designated for intraperitoneal injection.
7.1 General—The sample is the plastic or other material
8.2.2 Agitate each extract liquid vigorously prior to with-
exposed to the extraction procedure. As a result of the
drawal of each injection dose to ensure even distribution of
extraction in Practice F619, for each extraction vehicle there
extracted matter. If the extract liquid contains particulates,
shall be available: (1) a sample extract liquid, and (2) a blank
record and report observations.
extract liquid. These extract liquids shall be injected into the
8.2.3 For each extraction vehicle use ten mice, five for the
test animals within 24 h of the end of the extraction procedure.
sample extract liquid and five for the blank extract liquid.
Record the storage conditions if the liquid extract is not used
Weigh all mice, and record their weight. Use a system of
immediately after preparation.
marking to identify each individual mouse within each group
7.1.1 There are usually two extract liquids (a blank and a
of five.
sample)preparedfromanextractionvehicle.Samplesbasedon
8.2.4 Inject the predetermined amount (see 7.3)ofthe
other extraction vehicles may be available, as described in
sample extract liquid intraperitoneally into each of the five
Practice F619, or as required by the standard for the medical
mice. Inject the blank extract liquid in the same way into five
device.
other mice.
7.2 Method A, Intravenous:
8.2.5 Observe all animals immediately after injection, again
7.2.1 The extract liquid is usually prepared from a saline
4 h after injection, and then not earlier than 24, 48, and 72 h,
extraction vehicle. The dose of the extract liquid is 50 mL/kg
respectively, after injection for clinical signs. Cage-side obser-
of body weight for each mouse, injected at a steady rate of not
vations should include, but not be limited to, changes in body
more than 0.1 mL/s.
condition, skin/hair coat, gait/posture, eyes, respiratory,
7.2.2 Aqueous extract liquids shall be nominally isotonic
gastrointestinal, and consciousness/mentation using the de-
before injection. For example, sodium chloride may be added
scriptors provided in Table 1. Record the observations.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F750 − 87 (Reapproved 2012) F750 − 20
Standard Practice for
Evaluating Acute Systemic Toxicity of Material Extracts by
Systemic Injection in the Mouse
This standard is issued under the fixed designation F750; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers a nonspecific, acute toxicity test used for detecting leachables from materials used in medical devices.
1.2 The liquids injected into the mouse are those obtained by Practice F619 where the extraction vehicles are saline, vegetable
oil, or other liquids simulating human body fluids.
1.3 Two procedures are outlined: Method A for intravenous injection and Method B for intraperitoneal injection.
1.4 This practice is one of several developed for the assessment of the biocompatibility of materials. Practice F748 may provide
guidance for the selection of appropriate methods for testing materials for a specific application.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
F619 Practice for Extraction of Medical Plastics
F748 Practice for Selecting Generic Biological Test Methods for Materials and Devices
3. Summary of Practice
3.1 The extract liquid is prepared in accordance with Practice F619. The extraction vehicles are saline and vegetable oil, or other
extraction vehicles, as described in Practice F619. The extract liquid is injected into mice, and the animals are observed at regular
intervals for 72 h for reactions, survival, etc.
4. Significance and Use
4.1 This practice is intended to help assess the biocompatibility of materials used in medical devices. It is an acute toxicological
test designed to detect the presence of injurious leachable substances.
4.2 This practice may not be appropriate for all types of implant applications. The user is cautioned to consider the
appropriateness of the method in view of the materials being tested, their potential applications, and the recommendations
contained in Practice F748.
4.3 The only limitation applicable is the extract preparation. Refer to Sections 4.3 and 4.4 of Practice F619 for a description
of this limitation.
This practice is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.16
on Biocompatibility Test Methods.
Current edition approved Oct. 1, 2012June 1, 2020. Published October 2012August 2020. Originally approved in 1982. Last previous edition approved in 20072012 as
ε1
F750 – 87 (2007)(2012). . DOI: 10.1520/F0750-87R12.10.1520/F0750-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F750 − 20
5. Apparatus
5.1 Mice—The mice shall be albino-type, healthy, and not previously used, and shall weigh between 17 and 23 g. Animal care
shall be in accordance with the “Guide for the Care and Use of Laboratory Animals.” Age, sex, and weight shall be recorded and
reported. All the mice for each extraction vehicle shall be from the same source. For each extraction vehicle, a minimum of ten
mice are used in the test. If the results of this first test group are inconclusive, then 20 more mice will be needed to complete the
test of one extraction vehicle for one plastic.
5.1.1 During the test the mice shall be fed normally with commercially available feed and tap water.
5.2 Cages—There shall be one cage for the five mice exposed to one extract liquid. Each mouse in a cage shall be uniquely
identified, and this identification shall be recorded. Male and female mice shall be housed separately, and their cages are positioned
in a manner which prevents the accidental transfer of feces or bedding from cage to cage.
5.3 Syringe—Sterile syringes, not greater than 3 mL in volume, with a precision of 60.10 mL shall be used.
5.3.1 Method A—Sterile needles of 25 to 27 ⁄2 gagegauge shall be used.
5.3.2 Method B—Sterile needles of 21 to 26 gagegauge shall be used.
6. Sampling
6.1 Sample in accordance with Practice F619.
7. Sample and Test Specimen
7.1 General—The sample is the plastic or other material exposed to the extraction procedure. As a result of the extraction in
Practice F619, for each extraction vehicle there are shall be available: (1) a sample extract liquid, and (2) a blank extract liquid.
These extract liquids are to shall be injected into the test animals within 24 h of the end of the extraction procedure. Record the
storage conditions if the liquid extract is not used immediately after preparation.
7.1.1 There are usually two extract liquids (a blank and a sample) prepared from an extraction vehicle. Samples based on other
extraction vehicles may be available, as described in Practice F619, or as required by the standard for the medical device.
7.2 Method A, Intravenous:
7.2.1 The extract liquid is usually prepared from a saline extraction vehicle. The dose of the extract liquid is 50 mL/kg of body
weight for each mouse, injected at a steady rate of not more than 0.1 mL/s. If a hypotonic or hypertonic extract liquid is used, then
the injection rate shall be adjusted appropriately.
7.2.2 Aqueous extract liquids shall be nominally isosmoticisotonic before injection. For example, sodium chloride may be
added to distilled water extracts.
7.3 Method B, Intraperitoneal—The extract liquid is prepared from a vegetable oil extraction vehicle. The dose of the extract
liquid is 50 mL/kg of body weight for each mouse.
8. Procedure
8.1 Method A, Intravenous:
8.1.1 Use saline,saline and similar extraction vehicles designated for intravenous injection.
8.1.2 Agitate each extract liquid vigorously prior to withdrawal of each injection dose to ensure even distribution of the
extracted matter. If particulates are clearly present, then the extract liquids shall be injected allow the particulates to settle. If they
do not settle, consider injecting them by the intraperitoneal method. Optionally, the pH may be measured and recorded.route and
provide justification.
NOTE 1—pH may be measured and recorded as particulate formation may result in a pH change.
8.1.3 For each extraction vehicle, use ten mice, five for the sample extract liquid and five for the blank extract liquid. Weigh
all mice, and record their weights. Use a system of marking to identify each individual mouse within each group of five.
8.1.4 Inject the predetermined amount (see 7.2.1) of the sample extract liquid into the tail vein of each of the five mice. Inject
the blank extract liquid in the same way into five other mice. The use of warm water or a heat lamp may help dilate the tail veins
for ease of injection.
8.1.5 Observe all animals immediately after injection, again 4 h after injection, and then at 24, 48, and 72 h, respectively, after
injection for symptoms of slight, moderate, or marked toxicity or death (clinical signs. Cage-side observations should include, but
not be limited to, changes in body condition, skin/hair coat, gait/posture, eyes, respiratory, gastrointestinal, and consciousness/
mentation using the descriptors provided in Table 1). Record the observations. Measure and record the body weights of all animals
at 24, 48, and 72 h post-injection.
8.2 Method B, Intraperitoneal:
U.S. Department of Health, Education, and Welfare, National Research Council, Guide for the Care and Use of Laboratory Animals, Publication No. NIH 78-23,
Bethesda, MD, 1978.National Academy Press, Washington, DC, 2011.
F750 − 20
A,B,C
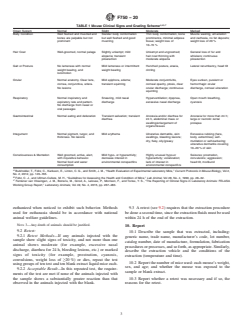
TABLE 1 Response to Systemic Injection Assay Mouse Clinical Signs and Grading Scheme
Response Description
Organ System Normal Slight Moderate Marked
Normal, no symptoms Mouse exhibits no adverse
physical symptoms after
injection.
Body Condition Well fleshed and muscled and Slender body conformation but Thin body conformation, bone Muscle wasting, emaciation and
bones are palpable but not well fleshed and good muscling prominence, minimal adipose cachexia, no fat deposits;
prominent tissue; weight loss of 10–15 % weight loss of 20 %
Slight Mouse exhibits slight but
noticeable symptoms of
hypokinesia, dyspnea, or
abdominal irritation after
injection.
Hair Coat Well-groomed, normal pelage Slightly unkempt; mild alopecia; Unkempt and ungroomed; hair General loss of fur and
transient piloerection coat thinning with moderate whiskers; continuous
alopecia piloerection
Gait or
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...