ASTM D7892-22
(Test Method)Standard Test Method for Determination of Total Organic Halides, Total Non-Methane Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas Chromatography/Mass Spectrometry
Standard Test Method for Determination of Total Organic Halides, Total Non-Methane Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas Chromatography/Mass Spectrometry
SIGNIFICANCE AND USE
5.1 Low operating temperature fuel cells such as PEMFCs require high purity hydrogen for optimal performance and longevity. Organic halides and formaldehyde can react with catalyst in PEMs and non-methane hydrocarbons degrade PEM stack performance.
SCOPE
1.1 The gas chromatography/mass spectrometry (GC/MS) procedure described in this test method is used to determine concentrations of total organic halides and total non-methane hydrocarbons (TNMHC) in hydrogen by measurement of individual target halocarbons (Table 1) and hydrocarbons (including formaldehyde, Table 1 and Table 2), respectively.
1.2 Mention of trade names in this test method does not constitute endorsement or recommendation for use. Other manufacturers’ equipment or equipment models can be used.
1.3 Units—The values stated in SI units are to be regarded as standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2022
- Technical Committee
- D03 - Gaseous Fuels
- Drafting Committee
- D03.14 - Hydrogen and Fuel Cells
Relations
- Effective Date
- 15-Dec-2019
- Effective Date
- 01-Nov-2017
- Effective Date
- 01-Jul-2016
- Effective Date
- 01-Mar-2011
- Effective Date
- 01-Dec-2008
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Jun-2000
Overview
ASTM D7892-22 is the internationally recognized standard test method for determining the concentration of total organic halides, total non-methane hydrocarbons (TNMHCs), and formaldehyde in hydrogen fuel. Using gas chromatography/mass spectrometry (GC/MS), this standard supports the quality control of hydrogen supplied for use in fuel cell vehicles, especially those utilizing proton exchange membrane fuel cells (PEMFCs). High-purity hydrogen is essential for these applications because trace contaminants such as organic halides and formaldehyde can adversely affect fuel cell catalysts, while non-methane hydrocarbons degrade stack performance and longevity.
Key Topics
- Hydrogen Fuel Quality: Focuses on assessing the purity of hydrogen used in fuel cell vehicles, meeting strict thresholds for contaminants.
- Target Compounds: Measures a comprehensive list of organic halides and non-methane hydrocarbons, including formaldehyde, as specified in the standard’s tables.
- GC/MS Analytical Method: Employs advanced gas chromatography and mass spectrometry techniques for sensitive, selective detection and quantification.
- Reporting Limit: Achieves low detection limits (down to 0.001 μmole/mole for most compounds), supporting regulatory and industry requirements.
- Standardized Procedures: Describes detailed procedures for sampling, calibration, system cleaning, and data analysis, ensuring reliable, reproducible results.
Applications
ASTM D7892-22 is essential in several contexts within the hydrogen and fuel cell industry, including:
- Hydrogen Fuel Supply Chain: Used by hydrogen producers and suppliers to verify product quality before distribution for automotive use.
- Fuel Cell Vehicle Infrastructure: Critical at hydrogen refueling stations to ensure fuel quality aligns with SAE J2719 guidelines and prevents contamination of on-board fuel cells.
- Laboratory Quality Control: Provides a reproducible method for laboratories to detect trace levels of organic and hydrocarbon impurities, supporting research and development, as well as commercial testing.
- Regulatory Compliance: Supports compliance with international hydrogen fuel standards, helping organizations and laboratories meet legal and technical certification needs.
By employing this standard, stakeholders can reduce the risk of catalyst poisoning, maintain high efficiency, extend the operational life of PEMFC stacks, and foster broader adoption of hydrogen-powered vehicles.
Related Standards
The following standards are commonly referenced in conjunction with ASTM D7892-22, further supporting quality control across the hydrogen value chain:
- SAE J2719 - Hydrogen Fuel Quality for Fuel Cell Vehicles: Sets the impurity limits for hydrogen used in PEM fuel cells, closely tied to the measurements produced by this test method.
- ASTM D4150 - Terminology Relating to Gaseous Fuels: Provides key definitions and terminology relevant to hydrogen fuel testing.
- ASTM D7606 - Practice for Sampling of High Pressure Hydrogen and Related Fuel Cell Feed Gases: Describes best practices for collecting representative hydrogen samples for analysis.
- ISO Standards: Many international standards for hydrogen quality reference or align with ASTM methods, ensuring global applicability.
Practical Value
Adoption of ASTM D7892-22 empowers hydrogen producers, laboratories, and fuel cell infrastructure providers to:
- Ensure compliance with stringent hydrogen fuel quality standards.
- Protect valuable fuel cell systems from degradation due to contaminants.
- Guarantee consistent fuel performance for end-users and OEMs.
- Support the transition towards sustainable, hydrogen-powered transportation by meeting regulatory and industry expectations for hydrogen purity.
By following the latest edition of this standard, organizations uphold technical accuracy, build stakeholder confidence, and contribute to the reliable adoption of hydrogen fuel technologies worldwide.
Buy Documents
ASTM D7892-22 - Standard Test Method for Determination of Total Organic Halides, Total Non-Methane Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas Chromatography/Mass Spectrometry
REDLINE ASTM D7892-22 - Standard Test Method for Determination of Total Organic Halides, Total Non-Methane Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas Chromatography/Mass Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

DNV
DNV is an independent assurance and risk management provider.

Lloyd's Register
Lloyd's Register is a global professional services organisation specialising in engineering and technology.

DNV Energy Systems
Energy and renewable energy certification.
Sponsored listings
Frequently Asked Questions
ASTM D7892-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Total Organic Halides, Total Non-Methane Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas Chromatography/Mass Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 Low operating temperature fuel cells such as PEMFCs require high purity hydrogen for optimal performance and longevity. Organic halides and formaldehyde can react with catalyst in PEMs and non-methane hydrocarbons degrade PEM stack performance. SCOPE 1.1 The gas chromatography/mass spectrometry (GC/MS) procedure described in this test method is used to determine concentrations of total organic halides and total non-methane hydrocarbons (TNMHC) in hydrogen by measurement of individual target halocarbons (Table 1) and hydrocarbons (including formaldehyde, Table 1 and Table 2), respectively. 1.2 Mention of trade names in this test method does not constitute endorsement or recommendation for use. Other manufacturers’ equipment or equipment models can be used. 1.3 Units—The values stated in SI units are to be regarded as standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Low operating temperature fuel cells such as PEMFCs require high purity hydrogen for optimal performance and longevity. Organic halides and formaldehyde can react with catalyst in PEMs and non-methane hydrocarbons degrade PEM stack performance. SCOPE 1.1 The gas chromatography/mass spectrometry (GC/MS) procedure described in this test method is used to determine concentrations of total organic halides and total non-methane hydrocarbons (TNMHC) in hydrogen by measurement of individual target halocarbons (Table 1) and hydrocarbons (including formaldehyde, Table 1 and Table 2), respectively. 1.2 Mention of trade names in this test method does not constitute endorsement or recommendation for use. Other manufacturers’ equipment or equipment models can be used. 1.3 Units—The values stated in SI units are to be regarded as standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7892-22 is classified under the following ICS (International Classification for Standards) categories: 27.075 - Hydrogen technologies. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7892-22 has the following relationships with other standards: It is inter standard links to ASTM D4150-19, ASTM D7606-17, ASTM D4150-08(2016), ASTM D7606-11, ASTM D4150-08, ASTM D4150-03, ASTM D4150-00. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7892-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7892 − 22
Standard Test Method for
Determination of Total Organic Halides, Total Non-Methane
Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas
Chromatography/Mass Spectrometry
This standard is issued under the fixed designation D7892; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2.2 SAE Standard:
SAE J2719 Hydrogen Fuel Quality for Fuel Cell Vehicles
1.1 The gas chromatography/mass spectrometry (GC/MS)
procedure described in this test method is used to determine
3. Terminology
concentrations of total organic halides and total non-methane
3.1 Definitions—For definitions of general terms used in
hydrocarbons (TNMHC) in hydrogen by measurement of
D03 Gaseous Fuels standards, refer to Terminology D4150.
individual target halocarbons (Table 1) and hydrocarbons
(including formaldehyde, Table 1 and Table 2), respectively.
3.2 Abbreviations:
3.2.1 GC—gas chromatograph
1.2 Mention of trade names in this test method does not
3.2.2 m/e—mass to charge ratio
constitute endorsement or recommendation for use. Other
manufacturers’ equipment or equipment models can be used.
3.2.3 MS—mass spectrometer
1.3 Units—The values stated in SI units are to be regarded 3.2.4 ppb(v)—parts per billion as a volume
as standard.
3.2.5 ppm(v)—parts per million as a volume
1.4 This standard does not purport to address all of the
3.2.6 UHP—ultra high purity
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro- 4. Summary of Test Method
priate safety, health, and environmental practices and deter-
4.1 The target compounds in Table 1 and Table 2, which
mine the applicability of regulatory limitations prior to use.
may be contained in a 400 mLhydrogen sample, are cryogeni-
1.5 This international standard was developed in accor-
cally frozen or concentrated onto a glass bead trap at -150 °C.
dance with internationally recognized principles on standard-
The target compounds are slowly desorbed by warming to 10
ization established in the Decision on Principles for the
°C and transferred to a Tenax trap cooled to -60 °C using
Development of International Standards, Guides and Recom-
desorption flow rate of 10 mL/min. This process leaves water
mendations issued by the World Trade Organization Technical
in the glass bead trap and dehydrates the sample. The Tenax
Barriers to Trade (TBT) Committee.
trap is then desorbed by heating to 180 °C and the target
compounds cyro-focused at -170 °C at the entrance to a GC
2. Referenced Documents
column (see 6.5). The cyro-focusing section is then rapidly
heated up to 80 °C to release the cryo-focused target
2.1 ASTM Standards:
compounds, which are eventually eluted from the column and
D4150 Terminology Relating to Gaseous Fuels
analyzed using a mass spectrometer scanning from m/e 23 to
D7606 Practice for Sampling of High Pressure Hydrogen
100 for initial 4.5 min and from m/e 34 to 550 the remaining
and Related Fuel Cell Feed Gases
analytical time. The retention times of the target compounds
are listed in Table 1 and Table 2 under the chromatographic
conditions in 6.5.
ThistestmethodisunderthejurisdictionofASTMCommitteeD03onGaseous
Fuels and is the direct responsibility of Subcommittee D03.14 on Hydrogen and
5. Significance and Use
Fuel Cells.
CurrenteditionapprovedJune1,2022.PublishedJuly2022.Originallyapproved
5.1 Low operating temperature fuel cells such as PEMFCs
in 2015. Last previous edition approved in 2015 as D7892 – 15. DOI: 10.1520/
require high purity hydrogen for optimal performance and
D7892-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available from SAE International (SAE), 400 Commonwealth Dr.,Warrendale,
the ASTM website. PA 15096, http://www.sae.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7892 − 22
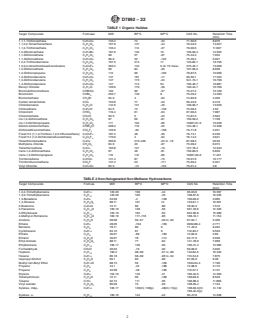
TABLE 1 Organic Halides
Target Compounds Formulas MW BP°C MP°C CAS No. Retention Time
(min)
1,1,1-Trichloroethane C H Cl 133.4 74 -33 71-55-6 8.876
2 3 3
1,1,2,2-Tetrachloroethane C H Cl 167.9 147 -44 79-34-5 14.627
2 2 4
1,1,2-Trichloroethane C H Cl 133.4 114 -37 79-00-5 11.607
2 3 3
1,2-Dibromoethane C H Br 187.9 132 10 106-93-4 12.555
2 4 2
1,1-Dichloroethane C H Cl 99 57 -97 75-34-3 7.034
2 4 2
1,1-Dichloroethene C H Cl 96.9 32 -122 75-35-4 5.927
2 2 2
1,2,4-Trichlorobenzene C H Cl 181.5 214 17 120-82-1 19.795
6 3 3
1,2,3,4-tetrachlorohexafluorobutane C Cl F 303.4 134 0 dl; 73 meso 375-45-1 13.008
4 4 6
1,2-Dichloroethane C H Cl 99 84 -35 107-06-2 8.658
2 4 2
1,2-Dichloropropane C H Cl 113 96 -100 78-87-5 10.006
3 6 2
1,2-Dichlorobenzene C H Cl 147 181 -17 95-50-1 17.334
6 4 2
1,3-Dichlorobenzene C H Cl 147 173 -24 541-73-1 16.799
6 4 2
1,4-Dichlorobenzene C H Cl 147 174 54 106-46-7 16.881
6 4 2
Benzyl Chloride C H Cl 126.6 179 -39 100-44-7 16.769
7 7
Bromodichloromethane CHBrCl 162 90 -57 75-27-4 10.189
Bromoform CHBr 252.7 149 8 75-25-2 14.303
Bromomethane CH Br 94.9 4 -94 74-83-9 4.326
Carbon tetrachloride CCl 153.8 77 -23 56-23-5 9.418
Chlorobenzene C H Cl 112.6 131 -45 108-90-7 13.626
6 5
Chloroethane C H Cl 64.5 12 -139 75-00-3 4.52
2 5
Chloroform CHCl 119.4 61 -64 67-66-3 7.987
Chloromethane CH Cl 50.5 5 -24 74-87-3 3.504
cis-1,2-dichloroethene C H Cl 97 60 -81 156-59-2 7.728
2 2 2
cis-1,3-Dichloropropene C H Cl 111 104 -85 10061-01-5 10.948
3 4 2
Dibromochloromethane CHBr Cl 208.3 119 -22 124-48-1 12.308
Dichlorodifluoromethane CCl F 120.9 -30 -158 75-71-8 3.251
2 2
Freon113 (1,1,2-Trichloro-1,2,2-trifluoroethane) C Cl F 187.4 48 -35 76-13-1 6.239
2 3 3
Freon114 (1,2-Dichlorotetrafluoroethane) C Cl F 170.9 4 -94 76-14-2 3.641
2 2 4
Hexachlorobutadiene C Cl 260.8 210–220 -22 to -19 87-68-3 20.56
4 6
Methylene chloride CH Cl 84.9 40 -97 75-09-2 6.015
2 2
Tetrachloroethene C Cl 165.8 121 -19 127-18-4 12.943
2 4
trans-1,2-dichloroethene C H Cl 97 48 -81 156-60-5 6.839
2 2 2
trans-1,3-Dichloropropene C H Cl 110 112 -85 10061-02-6 11.401
3 4 2
Trichloroethene C HCl 131.4 87 -73 79-01-6 10.177
2 3
Trichlorofluoromethane CCl F 137.4 23 -111 75-69-4 5.321
Vinyl Chloride C H Cl 62.5 -13 -154 75-01-4 3.8
2 2 2
TABLE 2 Non-Halogenated Non-Methane Hydrocarbons
Target Compounds Formula MW BP°C MP°C CAS No. Retention Time
(min)
1,2,4-Trimethylbenzene C H 120.20 169 -44 95-63-6 16.557
9 12
1,3,5-Trimethylbenzene C H 120.20 165 -45 108-67-8 16.045
9 12
1,3-Butadiene C H 54.09 -4 -109 106-99-0 3.985
4 6
1,4-Dioxane C H O 88.11 101 12 123-91-1 10.601
4 8 2
2-Butanone C H O 72.11 80 -86 78-93-3 7.516
4 8
2-Hexanone C H O 100.16 128 -56 591-78-6 12.325
6 12
4-Ethyltoluene C H 120.19 162 -62 622-96-8 15.969
9 12
4-Methyl-2-Pentanone C H O 100.16 117–118 -85 108-10-1 11.154
6 12
Acetone C H O 58.08 56–57 -95 to -93 67-64-1 5.356
3 6
Ethene C H 28.05 -104 -169 9002-88-4 2.771
2 4
Benzene C H 78.11 80 6 71-43-2 9.294
6 6
Cyclohexane C H 84.16 81 6 110-82-7 9.529
6 12
Ethane C H 30.07 -89 -183 74-84-0 2.82
2 6
Ethanol C H O 46.07 78 -114 64-17-5 5.556
2 6
Ethyl Acetate C H O 88.11 77 -84 141-78-6 7.958
4 8 2
Ethylbenzene C H 106.17 136 -95 100-41-4 13.962
8 10
Formaldehyde CH O 30.03 -19 -92 50-00-0 3.025
Heptane C H 100.2 98–99 -91 to -90 142-82-5 10.342
7 16
Hexane C H 86.18 68–69 -96 to -94 110-54-3 7.875
6 14
Isopropyl Alcohol C H O 60.1 83 -89 67-63-0 6.38
3 8
Methyl tert-Butyl Ether C H O 88.15 55 -109 1634-04-4 7.199
5 12
Propane C H 44.1 -42 -188 74-98-6 3.173
3 8
Propene C H 42.08 -48 -185 115-07-1 3.137
3 6
Styrene C H 104.16 145 -31 100-42-5 14.503
8 8
Tetrahydrofuran C H O 72.11 66 -108 109-99-9 8.529
4 8
Toluene C H 92.15 111 -95 108-88-3 11.866
7 8
Vinyl acetate C H O 86.09 73 -93 108-05-4 7.134
4 6 2
Xylenes, m&p- C H 106.17 139(m) 138(p) -48(m) 13(p) 108-38-3(m) 14.132
8 10
106-42-3(p)
Xylenes, o- C H 106.16 144 -24 95-47-6 14.638
8 10
D7892 − 22
longevity. Organic halides and formaldehyde can react with long as accuracy and stability can be verified and satisfy
catalystinPEMsandnon-methanehydrocarbonsdegradePEM application requirements.
stack performance.
7.2 Carrier Gas—UHP hydrogen is used; however, other
UHP gases, such as helium, can also be used provided
6. Apparatus
application requirements are met. No target compounds in
6.1 Sample Concentration System—The sample concentra- Table 1 and Table 2 are present at greater than reporting limits
in the carrier gas.
tion system and GC/MS system described in this test method
(4) are commercially available.
7.3 Sample Transferring Gas for Sample Concentration—
UHP hydrogen is used; however, other UHP gases, such as
6.2 Data Acquisition—A computer or other data recorder,
loaded with appropriate software for data acquisition, reduc- helium, can also be used provided application requirements are
met. See 10.3.
tion and reporting, possessing the following capabilities is
required.
7.4 Liquid Nitrogen—Required for cryogenic cooling.
6.2.1 Graphic presentation of the chromatogram.
6.2.2 Digital display of chromatographic peak areas.
8. Equipment Preparation
6.2.3 Identification of peaks by retention time or relative
8.1 GC/MS and Sample Concentration System—Placed into
retention time.
service in accordance to the manufacturer’s instructions with
6.2.4 Calculation using of response factors.
performance of daily GC/MS mass calibration using perfluo-
6.2.5 Internal standard calculation and data presentation.
rotributylamine (FC-43).
6.3 Hydrogen Fuel Sample Container—Any sample con-
8.2 Liquid Nitrogen Dewar—A160 to 230 Lliquid nitrogen
tainerwithworkingpressuresupto12.4MPa(1800psi)canbe
Dewar with a head pressure of 0.15 MPa (22 psi) is used for
used. A sample container fitting the requirements of Practice
cryogenic cooling.
D7606 with internal surface coated with silicon has been used
in the application of this method. The sample container should
9. Hazards
demonstrate the absence of each organic halide or hydrocarbon
9.1 Warning—Mercury has been designated by many regu-
at less than the reporting limit before it is used for sample
latory agencies as a hazardous material that can cause serious
collection.
medical issues. Mercury, or its vapor, has been demonstrated to
6.4 Carrier Gas Control—Constant flow control of carrier
be hazardous to health and corrosive to materials. Caution
gas is critical for optimal and consistent analytical perfor-
should be taken when handling mercury and mercury contain-
mance. Control is achieved by use of two-stage pressure
ing products. See the applicable product Safety Data Sheet
regulators, fixed flow restrictors, mass flow controller, or
(SDS) for additional information. Users should be aware that
electronic pressure controllers.
selling mercury and/or mercury containing products into your
6.5 Chromatographic Column and Conditions—A60 m, state or country may be prohibited by law.
0.32 mm ID, 3 µm dimethylpolysiloxane stationary phase or
10. Laboratory Procedures
similar fused silica column is used with a flow rate of
1 mL⁄min. An initial column temperature of 35 °C (1.5 min)
10.1 Sampling Procedures—See Practice D7606.
followed by ramping to 110 °C at 9 °C/min then to 210 °C
10.2 Sample Receipt—Examine the overall condition of
(4 min) at 11 °C/min has been successfully used in perfor-
each sample container; perform leak checks and record obser-
mance of this test method.
vations in a dedicated logbook. Each container should possess
6.6 Sample Container Cleaning System—For simultaneous
anattachedsampleidentificationtagthatincludestheweightof
cleaning of several sample containers, the following is recom-
hydrogen sampled, pressure of hydrogen in the container, the
mended.
sampling place, date and time of sample collection.
6.6.1 Vacuum Pump—capable of evacuating sample ves-
10.3 Sample Concentration and Analysis—The four steps of
sel(s) to an absolute pressure of less than 0.05 mmHg.
sample concentration are described below.
6.6.2 Vacuum Gauge—capable of measuring vacuum down
10.3.1 Concentration Using a Glass Bead Trap—Flow
to an absolute pressure of 0.01 mmHg or less.
20 mL of the standard containing 100 ppb(v) of each internal
6.6.3 Isothermal Oven—used to heat containers to 80 °C
standard and surrogate at a flow rate of 100 mL⁄min through a
during cleaning.
glass bead trap cooled to -150 °C. Then, pass 400 mL of a
hydrogen sample (10.3.1.1) or known volume of gaseous
7. Compressed Gas Standards
calibration standards, at the same flow rate, throug
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7892 − 15 D7892 − 22
Standard Test Method for
Determination of Total Organic Halides, Total Non-Methane
Hydrocarbons, and Formaldehyde in Hydrogen Fuel by Gas
Chromatography/Mass Spectrometry
This standard is issued under the fixed designation D7892; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 The gas chromatography/mass spectrometry (GC/MS) procedure described in this test method is used to determine
concentrations of total organic halides and total non-methane hydrocarbons (TNMHC) in hydrogen by measurement of individual
target halocarbons (Table 1) and hydrocarbons (including formaldehyde, Table 1 and Table 2), respectively. Measurement of these
substances is required for application of SAE J2719 to hydrogen fuel quality where this fuel is intended for use in fuel cell vehicles.
SAE 2719 states hydrogen fuel is expected to contain less than 0.05 μmole/mole total halogenates (including organic halides), 2
μmole/mole total non-methane hydrocarbons (C1 Basis, 3.2.16) and 0.01 μmole/mole formaldehyde.
1.2 Based upon the GC/MS/full scan analysis of a 400 mL hydrogen sample, the reporting limit (RL) is 0.001 μmole/mole for each
target compound listed in Table 1 and Table 2, with the exception of 0.002 μmole/mole for ethane and 0.002 μmole/mole for ethene.
1.2 Mention of trade names in this standard test method does not constitute endorsement or recommendation for use. Other
manufacturers’ equipment or equipment models can be used.
1.3 Units—The values stated in SI units are to be regarded as standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D4150 Terminology Relating to Gaseous Fuels
D7606 Practice for Sampling of High Pressure Hydrogen and Related Fuel Cell Feed Gases
This test method is under the jurisdiction of ASTM Committee D03 on Gaseous Fuels and is the direct responsibility of Subcommittee D03.14 on Hydrogen and Fuel
Cells.
Current edition approved June 1, 2015June 1, 2022. Published July 2015July 2022. Originally approved in 2015. Last previous edition approved in 2015 as D7892 – 15.
DOI: 10.1520/D7892-15.10.1520/D7892-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7892 − 22
TABLE 1 Organic Halides
Target Compounds Formulas MW BP°C MP°C CAS No. Retention Time
(min)
1,1,1-Trichloroethane C H Cl 133.4 74 -33 71-55-6 8.876
2 3 3
1,1,2,2-Tetrachloroethane C H Cl 167.9 147 -44 79-34-5 14.627
2 2 4
1,1,2-Trichloroethane C H Cl 133.4 114 -37 79-00-5 11.607
2 3 3
1,2-Dibromoethane C H Br 187.9 132 10 106-93-4 12.555
2 4 2
1,1-Dichloroethane C H Cl 99 57 -97 75-34-3 7.034
2 4 2
1,1-Dichloroethene C H Cl 96.9 32 -122 75-35-4 5.927
2 2 2
1,2,4-Trichlorobenzene C H Cl 181.5 214 17 120-82-1 19.795
6 3 3
1,2,3,4-tetrachlorohexafluorobutane C Cl F 303.4 134 0 dl; 73 meso 375-45-1 13.008
4 4 6
1,2-Dichloroethane C H Cl 99 84 -35 107-06-2 8.658
2 4 2
1,2-Dichloropropane C H Cl 113 96 -100 78-87-5 10.006
3 6 2
1,2-Dichlorobenzene C H Cl 147 181 -17 95-50-1 17.334
6 4 2
1,3-Dichlorobenzene C H Cl 147 173 -24 541-73-1 16.799
6 4 2
1,4-Dichlorobenzene C H Cl 147 174 54 106-46-7 16.881
6 4 2
Benzyl Chloride C H Cl 126.6 179 -39 100-44-7 16.769
7 7
Bromodichloromethane CHBrCl 162 90 -57 75-27-4 10.189
Bromoform CHBr 252.7 149 8 75-25-2 14.303
Bromomethane CH Br 94.9 4 -94 74-83-9 4.326
Carbon tetrachloride CCl 153.8 77 -23 56-23-5 9.418
Chlorobenzene C H Cl 112.6 131 -45 108-90-7 13.626
6 5
Chloroethane C H Cl 64.5 12 -139 75-00-3 4.52
2 5
Chloroform CHCl 119.4 61 -64 67-66-3 7.987
Chloromethane CH Cl 50.5 5 -24 74-87-3 3.504
cis-1,2-dichloroethene C H Cl 97 60 -81 156-59-2 7.728
2 2 2
cis-1,3-Dichloropropene C H Cl 111 104 -85 10061-01-5 10.948
3 4 2
Dibromochloromethane CHBr Cl 208.3 119 -22 124-48-1 12.308
Dichlorodifluoromethane CCl F 120.9 -30 -158 75-71-8 3.251
2 2
Freon113 (1,1,2-Trichloro-1,2,2-trifluoroethane) C Cl F 187.4 48 -35 76-13-1 6.239
2 3 3
Freon114 (1,2-Dichlorotetrafluoroethane) C Cl F 170.9 4 -94 76-14-2 3.641
2 2 4
Hexachlorobutadiene C Cl 260.8 210–220 -22 to -19 87-68-3 20.56
4 6
Methylene chloride CH Cl 84.9 40 -97 75-09-2 6.015
2 2
Tetrachloroethene C Cl 165.8 121 -19 127-18-4 12.943
2 4
trans-1,2-dichloroethene C H Cl 97 48 -81 156-60-5 6.839
2 2 2
trans-1,3-Dichloropropene C H Cl 110 112 -85 10061-02-6 11.401
3 4 2
Trichloroethene C HCl 131.4 87 -73 79-01-6 10.177
2 3
Trichlorofluoromethane CCl F 137.4 23 -111 75-69-4 5.321
Vinyl Chloride C H Cl 62.5 -13 -154 75-01-4 3.8
2 2 2
TABLE 2 Non-Halogenated Non-Methane Hydrocarbons
Target Compounds Formula MW BP°C MP°C CAS No. Retention Time
(min)
1,2,4-Trimethylbenzene C H 120.20 169 -44 95-63-6 16.557
9 12
1,3,5-Trimethylbenzene C H 120.20 165 -45 108-67-8 16.045
9 12
1,3-Butadiene C H 54.09 -4 -109 106-99-0 3.985
4 6
1,4-Dioxane C H O 88.11 101 12 123-91-1 10.601
4 8 2
2-Butanone C H O 72.11 80 -86 78-93-3 7.516
4 8
2-Hexanone C H O 100.16 128 -56 591-78-6 12.325
6 12
4-Ethyltoluene C H 120.19 162 -62 622-96-8 15.969
9 12
4-Methyl-2-Pentanone C H O 100.16 117–118 -85 108-10-1 11.154
6 12
Acetone C H O 58.08 56–57 -95 to -93 67-64-1 5.356
3 6
Ethene C H 28.05 -104 -169 9002-88-4 2.771
2 4
Benzene C H 78.11 80 6 71-43-2 9.294
6 6
Cyclohexane C H 84.16 81 6 110-82-7 9.529
6 12
Ethane C H 30.07 -89 -183 74-84-0 2.82
2 6
Ethanol C H O 46.07 78 -114 64-17-5 5.556
2 6
Ethyl Acetate C H O 88.11 77 -84 141-78-6 7.958
4 8 2
Ethylbenzene C H 106.17 136 -95 100-41-4 13.962
8 10
Formaldehyde CH O 30.03 -19 -92 50-00-0 3.025
Heptane C H 100.2 98–99 -91 to -90 142-82-5 10.342
7 16
Hexane C H 86.18 68–69 -96 to -94 110-54-3 7.875
6 14
Isopropyl Alcohol C H O 60.1 83 -89 67-63-0 6.38
3 8
Methyl tert-Butyl Ether C H O 88.15 55 -109 1634-04-4 7.199
5 12
Propane C H 44.1 -42 -188 74-98-6 3.173
3 8
Propene C H 42.08 -48 -185 115-07-1 3.137
3 6
Styrene C H 104.16 145 -31 100-42-5 14.503
8 8
Tetrahydrofuran C H O 72.11 66 -108 109-99-9 8.529
4 8
Toluene C H 92.15 111 -95 108-88-3 11.866
7 8
Vinyl acetate C H O 86.09 73 -93 108-05-4 7.134
4 6 2
Xylenes, m&p- C H 106.17 139(m) 138(p) -48(m) 13(p) 108-38-3(m) 14.132
8 10
106-42-3(p)
Xylenes, o- C H 106.16 144 -24 95-47-6 14.638
8 10
D7892 − 22
2.2 Other Standards:SAE Standard:
SAE J2719 Information Report on the Development of a Hydrogen Quality Guideline Hydrogen Fuel Quality for Fuel Cell
Vehicles
3. Terminology
3.1 Definitions—For definitions of general terms use in this test method, used in D03 Gaseous Fuels standards, refer to
Terminology D4150.
3.2 Definitions:
3.2.1 absolute pressure—pressure measured with reference to absolute zero pressure usually expressed as kPa, mm Hg, bar or psi.
3.2.2 constituent—component (or compound) found within a hydrogen fuel mixture.
3.2.3 contaminant—contaminant as defined in this application is an impurity that adversely affects the components within a fuel
cell system or hydrogen storage system.
3.2.4 cryogen—a refrigerant is used to obtain very low temperatures. The cryogen used in this method is liquid nitrogen (bp -196
°C).
3.2.5 dynamic calibration—Calibration of an analytical system uses gaseous calibration standard generated by diluting a known
concentration of compressed gaseous standard with a diluent gas.
3.2.6 fuel cell grade hydrogen—hydrogen satisfying the specifications in SAE J2719.
3.2.7 hydrogen fuel—hydrogen sampled at a vehicle fueling nozzle, without change of composition by drying, sampling, etc.
3.2.8 internal standard—material added to samples in a known amount to serve as a reference measurement.
3.2.9 internal standard calibration—calibration performed using internal standards to compensate for variation of GC/MS
sensitivity. In this test method, 0.005 μmole/mole each of 1,4-difluorobenzene, and D5-chlorobenzene are added during a
GC/MS/full scan analysis for the target compounds listed in Table 1 and Table 2.
3.2.10 poisoning—process by which the catalysts inside a PEMFC are made inoperative due to the activity of contaminants that
can bind to or chemically alter the catalyst used in a fuel cell.
3.2.11 reporting limit, RL—the lowest level of an analyte that an individual laboratory can confidently report for a particular
matrix.
3.2.12 qualitative accuracy—the ability of an analytical system to correctly identify compounds.
3.2.13 quantitative accuracy—the ability of an analytical system to correctly measure the concentration of an identified compound.
3.2.14 static calibration—calibration of an analytical system using standards in a form, matrix, state, or manner different from
samples to be analyzed.
3.2.15 surrogate—a pure analyte, which is extremely unlikely to be found in a sample that is added to a sample aliquot in a known
amount. It is measured using the same procedure(s) used to measure the target compounds in the sample. The purpose of a
surrogate analyte is to monitor the method performance with each sample.
3.2.15.1 Discussion—
In this method, 0.005 μmole/mole each of bromochloromethane and 4-bromofluorobenzene are added to every sample or standard
during analysis. The surrogate recoveries are expected to be within 70 % and 130 %.
Available from SAE International (SAE), 400 Commonwealth Dr., Warrendale, PA 15096, http://www.sae.org.
D7892 − 22
3.2.16 total non-methane hydrocarbons (C basis)—the concentration of total non-methane hydrocarbons (C basis) is defined by
1 1
the following formula:
Total non-methane hydrocarbons ~C basis! 5
all
Σ concentration of found non-methane hydrocarbon
~ !
i51 i
3 its carbon numbers (1)
i
3.2 Acronyms:Abbreviations:
3.3.1 EIC—extracted ion chromatogram
3.3.2 FCV—fuel cell vehicle
3.2.1 GC—gas chromatograph
3.3.4 IS—internal standard
3.3.5 ISO—International Organization for Standardization
3.2.2 m/e—mass to charge ratio
3.2.3 MS—mass spectrometer
3.3.8 NIST—National Institute of Standards and Technology
3.3.9 NTC—non-target compound
3.2.4 ppb(v) (μL/mppb(v)— )—parts per billion as a volume/volume ratiovolume
3.2.5 ppm(v) (μL/L)—ppm(v)—parts per million as a volume/volume ratiovolume
3.3.12 PEMFC—proton exchange membrane fuel cell
3.3.13 RL—reporting limits
3.3.14 SIM—selected ion monitoring
3.3.15 TC—target compounds
3.3.16 TIC—total ion current
3.2.6 UHP—ultra high purity (99.999%)
3.3.18 UOM—unit of measure
3.3.19 US EPA or EPA—The United States of America Environmental Protection Agency
4. Summary of Test Method
4.1 The target compounds in Table 1 and Table 2, which may be contained in a 400 mL hydrogen sample, are cryogenically frozen
or concentrated onto a glass bead trap at -150 °C. The target compounds are slowly desorbed by warming to 10 °C and transferred
to a Tenax trap cooled to -60 °C using desorption flow rate of 10 mL/min. This process leaves water in the glass bead trap and
dehydrates the sample. The Tenax trap is then desorbed by heating to 180 °C and the target compounds cyro-focused at -170 °C
at the entrance to a GC column (see 6.5). The cyro-focusing section is then rapidly heated up to 80 °C to release the cryo-focused
D7892 − 22
target compounds, which are eventually eluted from the column and analyzed using a mass spectrometer scanning from m/e 23
to 100 for initial 4.5 min and from m/e 34 to 550 the remaining analytical time. The retention times of the target compounds are
listed in Table 1 and Table 2 under the chromatographic conditions in 6.5.
5. Significance and Use
5.1 Low operating temperature fuel cells such as PEMFCs require high purity hydrogen for optimal performance and longevity.
Organic halides and formaldehyde can react with catalyst in PEMs and non-methane hydrocarbons degrade PEM stack
performance.
6. Apparatus
6.1 Sample Concentration System—The sample concentration system and GC/MS system described in this test method (4) are
commercially available.
6.2 Data Acquisition—A computer or other data recorder, loaded with appropriate software for data acquisition, reduction and
reporting, possessing the following capabilities is required.
6.2.1 Graphic presentation of the chromatogram.
6.2.2 Digital display of chromatographic peak areas.
6.2.3 Identification of peaks by retention time or relative retention time.
6.2.4 Calculation using of response factors.
6.2.5 Internal standard calculation and data presentation.
6.3 Hydrogen Fuel Sample Container—Hydrogen Fuel Sample Container – Any sample container with working pressures up to
12.4 MPa (1800 psi) can be used. A sample container fitting the requirements of Practice D7606 with internal surface coated with
silicon has been used in the application of this method. The sample container should demonstrate the absence of each organic
halide or hydrocarbon at less than the reporting limit (see 1.2) before it is used for sample collection.
6.4 Carrier Gas Control—Constant flow control of carrier gas is critical for optimal and consistent analytical performance. Control
is achieved by use of two-stage pressure regulators, fixed flow restrictors, mass flow controller, or electronic pressure controllers.
6.5 Chromatographic Column and Conditions—A 60 m, 0.32 mm ID, 3 μm dimethylpolysiloxane stationary phase or similar fused
silica column is used with a flow rate of 1 mL ⁄min. An initial column temperature of 35 °C (1.5 min) followed by ramping to 110
°C at 9 °C/min then to 210 °C (4 min) at 11 °C/min has been successfully used in performance of this test method.
6.6 Sample Container Cleaning System—For simultaneous cleaning of several sample containers, the following is recommended.
6.6.1 Vacuum Pump—capable of evacuating sample vessel(s) to an absolute pressure of less than 0.05 mmHg.
6.6.2 Vacuum Gauge—capable of measuring vacuum down to an absolute pressure of 0.01 mmHg or less.
6.6.3 Isothermal Oven—used to heat containers to 80 °C 80 °C during cleaning.
7. Compressed Gas Standards
7.1 Compressed Gas Standards:
7.1.1 The gaseous calibration standards for target compounds listed in Table 1 and Table 2, internal calibration standards
(standards, 3.2.9), and surrogates (3.2.15) used in this meth
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...