ASTM E1782-22
(Test Method)Standard Test Method for Determining Vapor Pressure by Thermal Analysis
Standard Test Method for Determining Vapor Pressure by Thermal Analysis
SIGNIFICANCE AND USE
5.1 Vapor pressure is a fundamental thermophysical property of a liquid. Vapor pressure data are useful in process design and control, in establishing environmental regulations for safe handling and transport, for estimation of volatile organic content (VOC), and in deriving hazard assessments. Vapor pressure and boiling temperature data are required for Safety Data Sheets (SDS). The enthalpy of vaporization may also be estimated from the slope of the vapor pressure curve (see Practice E2071).
SCOPE
1.1 This test method describes a procedure for the determination of the vapor pressure of pure liquids or melts from boiling point measurements made using differential thermal analysis (DTA) or differential scanning calorimetry (DSC) instrumentation operated at different applied pressures.
1.2 This test method can be used for the temperature range 273 K to 773 K (0 °C to 500 °C) and for pressures between 0.2 kPa to 2 MPa. These ranges may differ depending upon the instrumentation used and the thermal stability of materials tested. Because a range of applied pressures is required by this test method, the analyst is best served by use of instrumentation referred to as high pressure differential thermal instrumentation (HPDSC or HPDTA).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. (See also IEEE/ASTM SI 10.)
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2022
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 15-Jan-2018
- Effective Date
- 01-May-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Aug-2011
- Effective Date
- 15-Jun-2011
Overview
ASTM E1782-22: Standard Test Method for Determining Vapor Pressure by Thermal Analysis is an international standard developed by ASTM for accurately determining the vapor pressure of pure liquids or melts. This test method utilizes differential thermal analysis (DTA) or differential scanning calorimetry (DSC) instrumentation, enabling measurements over a wide temperature (273 K to 773 K) and pressure range (0.2 kPa to 2 MPa). Vapor pressure data are essential for process design, safety data sheets (SDS), environmental regulations, and hazard assessment across chemical, pharmaceutical, materials, and related industries.
By employing high-pressure differential thermal instrumentation (HPDSC or HPDTA), this method ensures reliable, reproducible measurements for materials that exhibit a single, sharp boiling endotherm. All values are reported in SI units, supporting international standardization and regulatory alignment.
Key Topics
- Vapor Pressure Determination: Establishes a systematic approach for measuring vapor pressure as a function of temperature using DSC or DTA, fundamental for understanding thermophysical properties.
- Equipment Requirements: Details the use of DSC or DTA with appropriate temperature and pressure control, regular calibration, and suitable sample containers to ensure accurate thermal analysis.
- Pressure and Temperature Range: Applicable for measurements from 0 °C to 500 °C and pressures from 0.2 kPa up to 2 MPa, allowing flexible application for various substances.
- Data Analysis and Reporting: Emphasizes fitting observed pressure-temperature data to the Antoine vapor pressure equation, reporting precise boiling points, and providing documentation suitable for compliance and risk analysis.
- Quality Assurance: Addresses calibration routines, repeatability, reproducibility, and bias, aligning with ASTM practices for interlaboratory consistency.
- Safety and Precautions: Outlines responsibilities for safety, environmental protection, and proper handling of equipment, especially when operating at non-ambient pressures.
Applications
This widely-adopted test method is practical for:
- Process Design & Engineering: Enables chemical engineers to select appropriate materials and process conditions based on accurate vapor pressure data.
- Environmental and Regulatory Compliance: Supports determination of volatile organic content (VOC) and helps establish protocols for safe handling, storage, and transport of chemicals.
- Material Safety Data Sheets (SDS): Provides reliable vapor pressure and boiling point data mandated for product documentation and hazard communication.
- Hazard and Risk Assessment: Assists in evaluating the flammability, volatility, and thermal stability of substances, critical in chemical and pharmaceutical manufacturing.
- Research and Development: Facilitates advanced thermophysical property studies within materials science and thermal analysis laboratories.
Related Standards
For comprehensive thermal analysis and accurate vapor pressure measurement, the following ASTM standards and references are commonly used in conjunction with ASTM E1782-22:
- ASTM E177 – Practice for Use of the Terms Precision and Bias in ASTM Test Methods
- ASTM E473 – Terminology Relating to Thermal Analysis and Rheology
- ASTM E691 – Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM E967 – Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
- ASTM E1142 – Terminology Relating to Thermophysical Properties
- ASTM E2071 – Practice for Calculating Heat of Vaporization or Sublimation from Vapor Pressure Data
- ASTM E3142 – Test Method for Thermal Lag of Thermal Analysis Apparatus
- IEEE/ASTM SI 10 – Standard for Use of the International System of Units (SI), The Modern Metric System
Key terms: vapor pressure, differential scanning calorimetry, thermal analysis, boiling point measurement, process safety, regulatory compliance.
By following ASTM E1782-22, professionals ensure robust, internationally recognized practices for determining vapor pressure, supporting safe and efficient operations in the global chemical and materials industries.
Buy Documents
ASTM E1782-22 - Standard Test Method for Determining Vapor Pressure by Thermal Analysis
REDLINE ASTM E1782-22 - Standard Test Method for Determining Vapor Pressure by Thermal Analysis
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E1782-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Determining Vapor Pressure by Thermal Analysis". This standard covers: SIGNIFICANCE AND USE 5.1 Vapor pressure is a fundamental thermophysical property of a liquid. Vapor pressure data are useful in process design and control, in establishing environmental regulations for safe handling and transport, for estimation of volatile organic content (VOC), and in deriving hazard assessments. Vapor pressure and boiling temperature data are required for Safety Data Sheets (SDS). The enthalpy of vaporization may also be estimated from the slope of the vapor pressure curve (see Practice E2071). SCOPE 1.1 This test method describes a procedure for the determination of the vapor pressure of pure liquids or melts from boiling point measurements made using differential thermal analysis (DTA) or differential scanning calorimetry (DSC) instrumentation operated at different applied pressures. 1.2 This test method can be used for the temperature range 273 K to 773 K (0 °C to 500 °C) and for pressures between 0.2 kPa to 2 MPa. These ranges may differ depending upon the instrumentation used and the thermal stability of materials tested. Because a range of applied pressures is required by this test method, the analyst is best served by use of instrumentation referred to as high pressure differential thermal instrumentation (HPDSC or HPDTA). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. (See also IEEE/ASTM SI 10.) 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Vapor pressure is a fundamental thermophysical property of a liquid. Vapor pressure data are useful in process design and control, in establishing environmental regulations for safe handling and transport, for estimation of volatile organic content (VOC), and in deriving hazard assessments. Vapor pressure and boiling temperature data are required for Safety Data Sheets (SDS). The enthalpy of vaporization may also be estimated from the slope of the vapor pressure curve (see Practice E2071). SCOPE 1.1 This test method describes a procedure for the determination of the vapor pressure of pure liquids or melts from boiling point measurements made using differential thermal analysis (DTA) or differential scanning calorimetry (DSC) instrumentation operated at different applied pressures. 1.2 This test method can be used for the temperature range 273 K to 773 K (0 °C to 500 °C) and for pressures between 0.2 kPa to 2 MPa. These ranges may differ depending upon the instrumentation used and the thermal stability of materials tested. Because a range of applied pressures is required by this test method, the analyst is best served by use of instrumentation referred to as high pressure differential thermal instrumentation (HPDSC or HPDTA). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. (See also IEEE/ASTM SI 10.) 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1782-22 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1782-22 has the following relationships with other standards: It is inter standard links to ASTM E473-23b, ASTM E1142-23b, ASTM E3142-18, ASTM E1142-15, ASTM E473-14, ASTM E1142-14b, ASTM E177-14, ASTM E1142-14a, ASTM E1142-14, ASTM E691-13, ASTM E177-13, ASTM E1142-12, ASTM E691-11, ASTM E1142-11b, ASTM E1142-11a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1782-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1782 − 22
Standard Test Method for
Determining Vapor Pressure by Thermal Analysis
This standard is issued under the fixed designation E1782; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E473Terminology Relating to Thermal Analysis and Rhe-
ology
1.1 This test method describes a procedure for the determi-
E691Practice for Conducting an Interlaboratory Study to
nation of the vapor pressure of pure liquids or melts from
Determine the Precision of a Test Method
boiling point measurements made using differential thermal
E967Test Method for Temperature Calibration of Differen-
analysis (DTA) or differential scanning calorimetry (DSC)
tial Scanning Calorimeters and Differential ThermalAna-
instrumentation operated at different applied pressures.
lyzers
1.2 This test method can be used for the temperature range
E1142Terminology Relating to Thermophysical Properties
273K to 773 K (0°C to 500°C) and for pressures between
E2071Practice for Calculating Heat of Vaporization or
0.2kPato2MPa.Theserangesmaydifferdependinguponthe
Sublimation from Vapor Pressure Data
instrumentation used and the thermal stability of materials
E3142Test Method for Thermal Lag of Thermal Analysis
tested. Because a range of applied pressures is required by this
Apparatus
test method, the analyst is best served by use of instrumenta-
IEEE/ASTM SI10Standard for Use of the International
tionreferredtoashighpressuredifferentialthermalinstrumen-
System of Units (SI) The Modern Metric System
tation (HPDSC or HPDTA).
3. Terminology
1.3 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
3.1 Definitions:
standard. (See also IEEE/ASTM SI10.) 3.1.1 The following terms are applicable to this test method
and can be found in either Terminology E473 or Terminology
1.4 This standard does not purport to address all of the
E1142:boilingpressure,boilingtemperature,differentialscan-
safety concerns, if any, associated with its use. It is the
ning calorimetry (DSC), differential thermal analysis (DTA),
responsibility of the user of this standard to establish appro-
vapor pressure, vaporization point, vaporization temperature.
priate safety, health, and environmental practices and deter-
3.2 Symbols:
mine the applicability of regulatory limitations prior to use.
3.2.1 A, B, C—Antoine vapor pressure equation (1) con-
1.5 This international standard was developed in accor-
stants (log , kPa, K):
dance with internationally recognized principles on standard- 10
ization established in the Decision on Principles for the
Antoinevaporpressureequation:Log P 5 A 2 B/~T1C!
Development of International Standards, Guides and Recom-
where:
mendations issued by the World Trade Organization Technical
P = vapor pressure, kPa,
Barriers to Trade (TBT) Committee.
T = temperature, K, and
A, B, and C = constants.
2. Referenced Documents
2.1 ASTM Standards:
4. Summary of Test Method
E177Practice for Use of the Terms Precision and Bias in
4.1 In thermal analysis, a physical property of a material is
ASTM Test Methods
measured either as a function of time at a specified constant
temperature, or more frequently, as a function of temperature
1 under conditions of a fixed rate of temperature change. The
ThistestmethodisunderthejurisdictionofASTMCommitteeE37onThermal
Measurements and is the direct responsibility of Subcommittee E37.01 on Calo- measured property is the dependent variable and the measured
rimetry and Mass Loss.
temperature is the independent variable. A specimen in an
Current edition approved July 1, 2022. Published August 2022. Originally
appropriate container is heated at a constant rate within a DTA
approved in 1996. Last previous edition approved in 2014 as E1782–14. DOI:
orDSCinstrumentoperatedunderanappliedconstantvacuum/
10.1520/E1782-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on The boldface numbers in parentheses refer to a list of references at the end of
the ASTM website. this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1782 − 22
pressurebetween0.2kPaand2MPauntilaboilingendotherm
is recorded. Boiling is observed at the temperature where the
specimenpartialpressureequalsthepressureappliedtothetest
chamber. The pressure is recorded during observation of the
boiling endotherm and the boiling temperature is recorded as
the extrapolated onset temperature. This measurement is re-
peated using new specimens for each of five or more different
pressurescoveringthepressurerangeofinterest.Thepressure-
−1
temperature data are fitted as Log P and 1/T (K )tothe
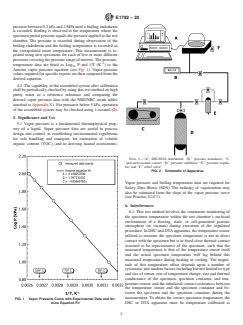
Antoine vapor pressure equation (see Fig. 1). Vapor pressure
valuesrequiredforspecificreportsarethencomputedfromthe
derived equation.
4.2 The capability of the assembled system after calibration
shallbeperiodicallycheckedbyusingthistestmethodonhigh
purity water as a reference substance and comparing the
derived vapor pressure data with the NBS/NRC steam tables
attachedasAppendixX1.Forpressuresbelow5kPa,operation
of the assembled system may be checked using 1-octanol (2).
5. Significance and Use
5.1 Vapor pressure is a fundamental thermophysical prop-
erty of a liquid. Vapor pressure data are useful in process
design and control, in establishing environmental regulations
for safe handling and transport, for estimation of volatile
organic content (VOC), and in deriving hazard assessments.
NOTE 1—“A”, DSC/DTA instrument; “B,” pressure transducer; “C,
”pressure/vacuum source; “D,” pressure stabilizer; “E,” pressure regula-
tor; and “F,” relief valve.
FIG. 2 Schematic of Apparatus
Vapor pressure and boiling temperature data are required for
Safety Data Sheets (SDS). The enthalpy of vaporization may
also be estimated from the slope of the vapor pressure curve
(see Practice E2071).
6. Interferences
6.1 This test method involves the continuous monitoring of
the specimen temperature within the test chamber’s enclosed
environment of a flowing, static, or self-generated gaseous
atmosphere (or vacuum) during execution of the stipulated
procedure. In DSC and DTAapparatus, the temperature sensor
utilized to measure the specimen temperature is not in direct
contact with the specimen but is in fixed close thermal contact
assumed to be representative of the specimen, such that the
measured temperature is that of the temperature sensor itself
and the actual specimen temperature will lag behind this
measured temperature during heating or cooling. The magni-
tude of this temperature offset depends upon a number of
systematicandrandomfactorsincludingbutnotlimitedtotype
andsizeofsensor,rateoftemperaturechange,sizeandthermal
conductance of the specimen, specimen container, and tem-
perature sensor, and the interfacial contact resistances between
the temperature sensor and the specimen container and be-
tween the specimen and the specimen container during the
measurement. To obtain the correct specimen temperature, the
FIG. 1 Vapor Pressure Curve with Experimental Data and An-
toine Equation Fit DSC or DTA apparatus must be temperature calibrated at
E1782 − 22
equivalent experimental conditions so that the recorded tem- 7.1.2.1 A pressure vessel, or similar means of sealing the
perature correctly indicates the specimen temperature. test chamber at any applied absolute pressure within the 0.2
kPa to 2 MPa range of this test method.
6.2 Temperature sensors are subject to degraded perfor-
7.1.2.2 Source of Pressurized Gas, or vacuum capable of
mancewithageandexposuretotheDSCorDTAtestchamber
sustaining a regulated inert gas pressure to the test chamber of
atmosphere. It is therefore imperative that the apparatus is
between 0.2 kPa and 2 MPa.
temperature calibrated regularly.
7.1.2.3 Pressure Transducer(s), to measure the pressure in
6.3 This test method is limited to materials that exhibit a
the test chamber to within 1% including any temperature
singlesharpboilingendothermundertheconditionsoutlinedin
dependence of the transducer(s) over the range of 0.2 kPa to
this test method.
2MPa.
6.4 Oxidation, pyrolysis, or polymerization of condensed
NOTE 1—Distance (or dead volume) between the pressure transducer
organic materials retained at temperatures above their ambient
and the specimen in the test chamber should be minimized to ensure
boilingpointmaybeencounteredatelevatedpressures.Thisis
accurate recording of the pressure at the time of boiling.
observed as an exotherm or a significantly broadened
7.1.2.4 Pressure Regulator, or similar device to adjust the
endotherm, or both, and shall not be considered a valid
applied pressure in the test chamber to 62% of the desired
pressure-temperature datum point. Use of an inert gas for
value.
elevated pressures or for back-filling after evacuation of the
7.1.2.5 Ballast, or similar means to maintain the applied
sample chamber is recommended to minimize the risk of
pressure in the test chamber constant to 61%.
oxidation.
7.1.2.6 Valves,tocontroldeliveryoftheinertgas/vacuumto
6.5 PartialblockageofthepinholeintheDSCcontainercan
the test chamber or to isolate components of the pressure/
occasionally be encountered. This may be observed as noise
vacuum system, or both. Valves shall be rated in excess of the
spikes on the boiling endotherm and shall not be considered a
2 MPa upper pressure limit of this test method.
valid pressure-temperature datum point
7.1.3 Containers, (pans, capillary tubes, etc.) that are inert
to the specimen and reference materials and which are of
7. Apparatus
suitable structural shape and integrity to contain the specimen
7.1 Differential Scanning Calorimeter (DSC) or Differential
and reference in accordance with the following specific re-
Thermal Analyzer (DTA)—Multiple generations of differential
quirements:
scanning calorimeters and differential thermal analyzers from
7.1.3.1 It is imperative that the containers used in this test
numerous commercial suppliers, as well as in-house custom
method are capable of retaining the specimen in a manner that
apparatus, utilizing a variety of temperature and heat flow
minimizes specimen loss through vaporization prior to boiling
sensors in various configurations may be available to the user.
andthatpromotesthedevelopmentofvapor-liquidequilibrium
Whileallsuchapparatuscapabilitiesmaynotbeequivalent,for
at boiling. When both conditions are met a sharp endotherm
purposesofthistestmethod,anyDSCorDTAinstrumentation
with minimal baseline curvature at the onset will be observed.
that meets the following criteria should be able to generate
NOTE2—StudiesbyASTMtaskgroupE37.01.05andothers (3-5)have
acceptable results.
determined glass cylindrical containers of 2mm to 4 mm inside diameter
7.1.1 The essential equipment required to provide the mini-
by 25 mm long are suitable for thermocouple inserted style DTA
mum instrument capability of this test method includes (see
instruments; and a hermetic sealable pan (approximately 40 µL vol) with
a single pinhole in the center of the lid is suitable for DSC instruments
Fig. 2):
with nominal heating rates of 5 K/min. Use of a progression of pinhole
7.1.1.1 DSC/DTA Test Chamber, composed of a furnace(s)
sizes ranging from approximately 50µm to 350 µm is recommended in
to provide uniform controlled heating of a specimen and
ordertoretainboilingendothermsharpnessoverthefullpressurerangeof
reference at a constant rate within the 273K to 773 K
thismethod.Typically,thesharpestboilingendothermforasamplewillbe
temperature range of this test method; a temperature sensor to produced at atmospheric pressure with a small (50µm to 75 µm) pinhole.
As pressure is reduced, increasingly larger pinholes should be used to
provide an indication of the specimen/furnace temperature to
minimize endotherm broadening. Use of large pinholes (350 µm) at
60.01 K; a differential sensor to detect a difference (tempera-
pressuresaslowas0.2kPahasbeenshowntoproduceboilingendotherms
ture or heat flow) between the specimen and reference equiva-
of comparable sharpness to atmospheric pressure endotherms. Use of
lentto5mW;andameansofsustaininganinertgasorvacuum
heating rates other than 5 K/min are not recommended for this test
test chamber environment at pressures above and below method. Higher rates may result in some self-pressurization of the
specimen and lesser rates will extend measurement times and will tend to
ambient.
promote preboiling vaporization.
7.1.1.2 Temperature Controller, capable of executing a
7.2 Auxiliary equipment considered useful in conducting
specific temperature program by operating the furnace(s)
this test method include:
between selected temperature limits to 61 K at a rate of
temperature change of 5 K/min constant within 61%.
7.2.1 Acoolantsystemthatcanbecoupleddirectlywiththe
controller to the furnace to hasten its recovery from elevated
7.1.1.3 Recording Device, to provide a means of acquiring,
storing and displaying measured or calculated signals or both. temperatures or to sustain a subambient temperature to within
61 K of a lower limit temperature.
The minimum output signals are heat flow, temperature and
time.
7.2.2 Abalancetoweighspecimensorspecimencontainers,
7.1.2 Pressure/Vacuum System, consisting of: or both, to 60.1 mg.
E1782 − 22
7.2.3 Asyringeormicropipettodeliverliquidspecimensof relationship is the thermal lag and its determination permits
1µLto5µL 610%. temperature calibration determined at one rate of temperature
7.2.4 Pressure relief valve to prevent accidental overpres- change to be adjusted to that of another rate of temperature
surizationinthepressuresystem.Aratingof10%inexcessof change. Using the same calibrant material, purge gas and flow
theupperusepressureissuggestedprovideditdoesnotexceed rate as that used to calibrate the temperature axis of the
the maximum working pressure rating of any individual instrument, repeat for two additional rates of temperature
component in the system. changefollowingtheproceduregiveninTestMethodE3142to
determine the thermal offset.
8. Precautions
10.3 Regular calibration of the temperature axis and deter-
8.1 Safety Precautions:
mination of the thermal lag is necessary for making good
8.1.1 Pressures in addition to ambient are employed in this temperature based measurements using DSC or DTA. The
test method. Ensure that the pressure/vacuum system is certi-
interval between calibrations is dependent upon a variety of
fied for operation at the extremes of pressure encountered with factors. Follow the instrument manufacturer’s guidelines. For
this test method. Incorporation of a pressure relief device is
testing requiring parameters outside those routinely used for
recommended. calibration and thermal lag determination or if an apparent
8.1.2 Adequate provisions shall be available for retention
changeintheequipmentisnoted,itisnecessarytotemperature
and disposal of any spilled mercury if mercury-containing calibrate the apparatus. Use the same variables during calibra-
pressure devices are employed.
tion that will be used for the subsequent testing.
10.4 Calibrate the pressure transducer according to the
9. Sampling
recommendations of the manufacturer or similar appropriate
9.1 Typica
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1782 − 14 E1782 − 22
Standard Test Method for
Determining Vapor Pressure by Thermal Analysis
This standard is issued under the fixed designation E1782; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method describes a procedure for the determination of the vapor pressure of pure liquids or melts from boiling point
measurements made using differential thermal analysis (DTA) or differential scanning calorimetry (DSC) instrumentation operated
at different applied pressures.
1.2 This test method maycan be used for the temperature range 273273 K to 773 K (0(0 °C to 500°C)500 °C) and for pressures
between 0.2 kPa 0.2 kPa to 2 MPa. These ranges may differ depending upon the instrumentation used and the thermal stability of
materials tested. Because a range of applied pressures is required by this test method, the analyst is best served by use of
instrumentation referred to as high pressure differential thermal instrumentation (HPDSC or HPDTA).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. (See
also IEEE/ASTM SI 10.)
1.4 There is no ISO standard equivalent to this test method.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E473 Terminology Relating to Thermal Analysis and Rheology
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E1142 Terminology Relating to Thermophysical Properties
E2071 Practice for Calculating Heat of Vaporization or Sublimation from Vapor Pressure Data
E3142 Test Method for Thermal Lag of Thermal Analysis Apparatus
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.01 on Calorimetry
and Mass Loss.
Current edition approved March 15, 2014July 1, 2022. Published April 2014August 2022. Originally approved in 1996. Last previous edition approved in 20082014 as
E1782 – 08.E1782 – 14. DOI: 10.1520/E1782-14.10.1520/E1782-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1782 − 22
IEEE/ASTM SI 10 Standard for Use of the International System of Units (SI) The Modern Metric System
3. Terminology
3.1 Definitions:
3.1.1 The following terms are applicable to this test method and can be found in either Terminology E473 or Terminology E1142:
boiling pressure, boiling temperature, differential scanning calorimetry (DSC), differential thermal analysis (DTA), vapor pressure,
vaporization point, vaporization temperature.
3.2 Symbols:
3.2.1 A, B, C—Antoine vapor pressure equation (1) constants (log , kPa, K):
Antoine vapor pressure equation:Log P 5 A 2 B/ T1C
~ !
Antoine vapor pressure equation :Log P 5 A 2 B/~T1C!
where:
P = vapor pressure, kPa, and
T = temperature, K.
P = vapor pressure, kPa,
T = temperature, K, and
A, B, and C = constants.
4. Summary of Test Method
4.1 In thermal analysis, a physical property of a material is measured either as a function of time at a specified constant
temperature, or more frequently, as a function of temperature under conditions of a fixed rate of temperature change. The measured
property is the dependent variable and the measured temperature is the independent variable. A specimen in an appropriate
container is heated at a constant rate within a DTA or DSC instrument operated under an applied constant vacuum/pressure between
0.2 kPa and 2 MPa until a boiling endotherm is recorded. Boiling is observed at the temperature where the specimen partial
pressure equals the pressure applied to the test chamber. The pressure is recorded during observation of the boiling endotherm and
the boiling temperature is recorded as the extrapolated onset temperature. This measurement is repeated using new specimens for
each of five or more different pressures covering the pressure range of interest. The pressure-temperature data are fitted as Log
−1
P and 1/T (K ) to the Antoine vapor pressure equation (see Fig. 1). Vapor pressure values required for specific reports are then
computed from the derived equation.
4.2 The capability of the assembled system after calibration shouldshall be periodically checked by using this test method on pure
high purity water as a reference substance and comparing the derived vapor pressure data with the NBS/NRC steam tables attached
as Appendix X1. For pressures below 5 kPa, operation of the assembled system may be checked using 1-octanol (2).
5. Significance and Use
5.1 Vapor pressure is a fundamental thermophysical property of a liquid. Vapor pressure data are useful in process design and
control, in establishing environmental regulations for safe handling and transport, for estimation of volatile organic content (VOC),
and in deriving hazard assessments. Vapor pressure and boiling temperature data are required for Material Safety Data Sheets
(MSDS).(SDS). The enthalpy of vaporization may also be estimated from the slope of the vapor pressure curve (see Practice
E2071).
6. Interferences
6.1 This test method involves the continuous monitoring of the specimen temperature within the test chamber’s enclosed
environment of a flowing, static, or self-generated gaseous atmosphere (or vacuum) during execution of the stipulated procedure.
In DSC and DTA apparatus, the temperature sensor utilized to measure the specimen temperature is not in direct contact with the
specimen but is in fixed close thermal contact assumed to be representative of the specimen, such that the measured temperature
is that of the temperature sensor itself and the actual specimen temperature will lag behind this measured temperature during
heating or cooling. The magnitude of this temperature offset depends upon a number of systematic and random factors including
The boldface numbers in parentheses refer to a list of references at the end of this standard.
E1782 − 22
FIG. 1 Vapor Pressure Curve with Experimental Data and Antoine Equation Fit
but not limited to type and size of sensor, rate of temperature change, size and thermal conductance of the specimen, specimen
container, and temperature sensor, and the interfacial contact resistances between the temperature sensor and the specimen
container and between the specimen and the specimen container during the measurement. To obtain the correct specimen
temperature, the DSC or DTA apparatus must be temperature calibrated at equivalent experimental conditions so that the recorded
temperature correctly indicates the specimen temperature.
6.2 Temperature sensors are subject to degraded performance with age and exposure to the DSC or DTA test chamber atmosphere.
It is therefore imperative that the apparatus is temperature calibrated regularly.
6.3 This test method is limited to materials that exhibit a single sharp boiling endotherm under the conditions outlined in this test
method.
6.4 Oxidation, pyrolysis, or polymerization of condensed organic materials retained at temperatures above their ambient boiling
point may be encountered at the elevated pressures of this method. This will be elevated pressures. This is observed as an exotherm
or a significantly broadened endotherm, or both, and shall not be considered a valid pressure-temperature datum point. Use of an
inert gas for elevated pressures or for back-filling after evacuation of the sample chamber is recommended to minimize the risk
of oxidation.
6.5 Partial blockage of the pinhole in the DSC containers couldcontainer can occasionally be encountered. This may be observed
as noise spikes on the boiling endotherm and shall not be considered a valid pressure-temperature datum point
7. Apparatus
7.1 Differential Scanning Calorimeter (DSC) or Differential Thermal Analyzer (DTA)—The essential equipment required to
provide the minimum instrument capability Multiple generations of differential scanning calorimeters and differential thermal
E1782 − 22
NOTE 1—“A”, DSC/DTA instrument; “B,” pressure transducer; “C,”pressure/vacuum source; “D,” pressure stabilizer; “E,” pressure regulator; and “F,”
relief valve.
FIG. 2 Schematic of Apparatus
analyzers from numerous commercial suppliers, as well as in-house custom apparatus, utilizing a variety of temperature and heat
flow sensors in various configurations may be available to the user. While all such apparatus capabilities may not be equivalent,
for purposes of this test method includes (seemethod, any DSC Fig. 2): or DTA instrumentation that meets the following criteria
should be able to generate acceptable results.
7.1.1 Differential Scanning Calorimeter (DSC) or Differential Thermal Analyzer (DTA),The essential equipment required to
provide the minimum instrument consisting of: capability of this test method includes (see Fig. 2):
7.1.1.1 DSC/DTA Test Chamber, composed of a furnace(s) to provide uniform controlled heating of a specimen and reference at
a constant rate within the 273273 K to 773 K temperature range of this test method; a temperature sensor to provide an indication
of the specimen/furnace temperature to 6160.01 K; a differential sensor to detect a difference (temperature or heat flow) between
the specimen and reference equivalent to 5 mW; and a means of sustaining an inert gas or vacuum test chamber environment at
pressures above and below ambient.
7.1.1.2 Temperature Controller, capable of executing a specific temperature program by operating the furnace(s) between selected
temperature limits to 61 K at a rate of temperature change of 5 K/min constant within 61 %.
7.1.1.3 Recording Device, to provide a means of acquiring, storing and displaying measured or calculated signals or both. The
minimum output signals are heat flow, temperature and time.
7.1.2 Pressure/Vacuum System, consisting of:
7.1.2.1 A pressure vessel, or similar means of sealing the test chamber at any applied absolute pressure within the 0.2 kPa to 2
MPa range of this test method.
7.1.2.2 Source of Pressurized Gas, or vacuum capable of sustaining a regulated inert gas pressure to the test chamber of between
0.2 kPa and 2 MPa.
E1782 − 22
7.1.2.3 Pressure Transducer(s), to measure the pressure in the test chamber to within 1 % including any temperature dependence
of the transducer(s) over the range of 0.2 kPa to 2 MPa.2 MPa.
NOTE 1—Distance (or dead volume) between the pressure transducer and the specimen in the test chamber should be minimized to ensure accurate
recording of the pressure at the time of boiling.
7.1.2.4 Pressure Regulator, or similar device to adjust the applied pressure in the test chamber to 62 % of the desired value.
7.1.2.5 Ballast, or similar means to maintain the applied pressure in the test chamber constant to 61 %.
7.1.2.6 Valves, to control delivery of the inert gas/vacuum to the test chamber or to isolate components of the pressure/vacuum
system, or both. Valves shall be rated in excess of the 2 MPa upper pressure limit of this test method.
7.1.3 Containers, (pans, capillary tubes, etc.) that are inert to the specimen and reference materials and which are of suitable
structural shape and integrity to contain the specimen and reference in accordance with the following specific requirements:
7.1.3.1 It is imperative that the containers used in this test method beare capable of retaining the specimen in a manner that
minimizes samplespecimen loss through vaporization prior to boiling and that promotes the development of vapor-liquid
equilibrium at boiling. When both conditions are met a sharp endotherm with little or no minimal baseline curvature at the onset
will be observed.
NOTE 2—Studies by ASTM task group E37.01.05 and others (3-5) have determined glass cylindrical containers of 22 mm to 4 mm inside diameter by
25 mm long are suitable for thermocouple inserted style DTA instruments; and a hermetic sealable pan (approximately 40 μL vol) with a single pinhole
in the center of the lid is suitable for DSC instruments with nominal heating rates of 5 K/min. Use of a progression of pinhole sizes ranging from
approximately 5050 μm to 350 μm is recommended in order to retain boiling endotherm sharpness over the full pressure range of this method. Typically,
the sharpest boiling endotherm for a sample will be produced at atmospheric pressure with a small (50(50 μm to 75 μm) pinhole. As pressure is reduced,
increasingly larger pinholes should be used to minimize endotherm broadening. Use of large pinholes (350 μm) at pressures as low as 0.2 kPa has been
shown to produce boiling endotherms of comparable sharpness to atmospheric pressure endotherms. Use of heating rates other than 5 K/min are not
recommended for this test method. Higher rates may result in some self-pressurization of the specimen and lesser rates will extend measurement times
and will tend to promote preboiling vaporization.
7.2 Auxiliary equipment considered useful in conducting this test method include:
7.2.1 A coolant system that can be coupled directly with the controller to the furnace to hasten its recovery from elevated
temperatures or to sustain a subambient temperature to within 61 K of a lower limit temperature.
7.2.2 A balance to weigh specimens or specimen containers, or both, to 60.1 mg.
7.2.3 A syringe or micropipet to deliver liquid specimens of 11 μL to 5 μL 610 %.
7.2.4 Pressure relief valve to prevent accidental overpressurization in the pressure system. A rating of 10 % in excess of the upper
use pressure is suggested provided it does not exceed the maximum working pressure rating of any individual component in the
system.
8. Precautions
8.1 Safety Precautions:
8.1.1 Pressures in addition to ambient are employed in this test method. Ensure that the pressure/vacuum system is certified for
operation at the extremes of pressure encountered with this test method. Incorporation of a pressure relief device is recommended.
8.1.2 Adequate provisions shall be available for retention and disposal of any spilled mercury if mercury-containing pressure
devices are employed.
9. Sampling
9.1 Typical specimen sizes used for individual pressure measurements are 11 mg to 5 mg of solid or 11 μL to 5 μL of liquid.
Similar size specimens should be used for each individual measurement of a given sample.
E1782 − 22
9.2 Samples are assumed to be tested as received. Report any special sampling or pretreatment used with this test method.
10. Calibration
10.1 Prior to installation, the temperature sensors used in DSC or DTA apparatus are calibrated under isothermal conditions where
static equilibrium is achieved. Thermal analysis apparatus temperature calibration is performed under assumed linear rates of
temperature change where dynamic equilibrium is obtained between the test specimen and the temperature sensor. Such
temperature calibration comp
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...