ASTM D1976-20
(Test Method)Standard Test Method for Elements in Water by Inductively-Coupled Plasma Atomic Emission Spectroscopy
Standard Test Method for Elements in Water by Inductively-Coupled Plasma Atomic Emission Spectroscopy
SIGNIFICANCE AND USE
5.1 This test method is useful for the determination of element concentrations in many natural waters and wastewaters. It has the capability for the simultaneous determination of up to 29 elements. High sensitivity analysis and larger dynamic range can be achieved for some elements that are difficult to determine by other techniques such as Flame Atomic Absorption.
5.2 The test method is useful for multi-element analysis of domestic and commercial well produced drinking water for metals and nonmetals for use in baseline analysis and monitoring during exploration, hydraulic fracturing, production, closure and reclamation activities related to oil and gas operations (see Guide D8006).
5.2.1 Minimum analyses include arsenic, barium, iron, magnesium, sodium, calcium, manganese, and lead.
5.2.2 Boron, potassium, chromium, selenium, cadmium, and strontium may be required on a site specific basis.
5.2.3 The most abundant elements in oil and gas produced water are sodium, potassium, lithium, magnesium, calcium, strontium, iron, silica, phosphorus, and sulfur.
5.3 The test method is useful for multi-element analysis of acid rock drainage and other major and some trace elements in mining influenced water.
5.4 Where low quantitation limits are required, Test Method D5673 may be applicable.
5.5 The test method is also useful for testing leachates and effluents for ore and mining and metallurgical waste characterization tests including Test Methods D6234, E2242, D5744, and solutions from the Biological Acid Production Potential and Peroxide Acid Generation Methods in the Appendix of Test Methods E1915.
SCOPE
1.1 This test method covers the determination of dissolved, total-recoverable, or total elements in drinking water, ground water, surface water, domestic, commercial or industrial wastewaters,2,3 within the following concentration ranges of Table 1.
1.2 It is the user’s responsibility to ensure the validity of the test method for waters of untested matrices.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Note 2 and Section 9.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2020
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 15-Feb-2024
- Effective Date
- 01-May-2020
- Effective Date
- 01-Sep-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Mar-2016
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Sep-2013
- Effective Date
- 01-Sep-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Dec-2012
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-Jun-2012
Overview
ASTM D1976-20 is the recognized standard test method for determining elements in water by Inductively-Coupled Plasma Atomic Emission Spectroscopy (ICP-AES). Developed by ASTM International, this method is widely used in environmental testing, industrial operations, and resource management for accurate and efficient elemental analysis. The standard covers the determination of dissolved, total-recoverable, or total elements in various water matrices, including drinking water, groundwater, surface water, and wastewaters associated with domestic, commercial, and industrial processes.
This test method enables simultaneous or sequential detection of up to 29 elements, offering high sensitivity and a broad dynamic range. The method enhances the detection of elements that are difficult to analyze accurately using other techniques, such as Flame Atomic Absorption. Users benefit from detailed protocols for sample collection, preservation, calibration, and quality control in water analysis.
Key Topics
- Multi-element Analysis: Simultaneous measurement of metals and nonmetals, including arsenic, barium, iron, magnesium, sodium, calcium, manganese, lead, boron, potassium, chromium, selenium, cadmium, strontium, lithium, and others.
- Suitable Water Types: Applicable to drinking water, groundwater, surface water, and wastewaters from various sources.
- Sample Preparation: Procedures for sample collection, preservation (acidification), and digestion for different analytical needs (dissolved, total-recoverable, or total elements).
- Instrument Calibration: Guidance on calibration standards, blanks, matrix matching, and validation of calibration curves.
- Interferences: Addressing spectral, physical, and chemical interferences, with recommendations for sample dilution, use of alternative wavelengths, and background corrections.
- Quality Control: Requirements for calibration correlation, error limits, and confirmation with quality control samples.
- Detection Limits: Standard provides guidance on estimated detection limits and recommended wavelengths for each element.
Applications
ASTM D1976-20 finds broad application across environmental and industrial sectors. Key uses include:
- Environmental Monitoring: Baseline assessment and ongoing monitoring of water quality in natural and impacted environments, including acid rock drainage and mining-influenced waters.
- Oil and Gas Operations: Essential for analyzing produced water during exploration, hydraulic fracturing, production, closure, and reclamation. Supports studies of metals and nonmetals relevant to environmental compliance and resource management.
- Mining and Metallurgical Waste Characterization: Useful for analyzing leachates and effluents in ore, mining, and metallurgical waste, employing associated test methods for comprehensive assessment.
- Drinking Water Compliance: Multi-element testing for metals and nonmetals in municipal, domestic, and commercial well water, supporting regulatory compliance and public safety.
- Industrial Wastewater Testing: Analytical support for process control, waste treatment optimization, and identifying potential environmental contaminants in industrial effluents.
- Baseline Studies: Supports investigations into baseline water composition for impact assessment and regulatory submissions.
Related Standards
ASTM D1976-20 is often used in conjunction with other important standards to ensure comprehensive water analysis. Key related standards include:
- ASTM D5673: Test Method for Elements in Water by Inductively Coupled Plasma - Mass Spectrometry, offering lower quantitation limits.
- ASTM D8006: Guide for Sampling and Analysis of Water Supply Wells in Exploration and Production Operations.
- ASTM D6234, E2242, D5744, E1915: Standards supporting characterization and extraction methods for mining wastes and ore materials.
- USEPA Method 200.7: Determination of Metals and Trace Elements in Water by ICP-AES, frequently referenced for regulatory compliance.
- ASTM D1193: Specification for Reagent Water, ensuring sample and reagent purity across analytical applications.
These related standards help laboratories and industries maintain consistency, accuracy, and regulatory compliance when implementing water analysis programs.
Keywords: ASTM D1976-20, ICP-AES water analysis, elemental testing in water, multi-element analysis, environmental monitoring, produced water testing, metal analysis, industrial wastewater, standard methods for water, ASTM water quality standards, water testing compliance, oil and gas water analysis, mining effluent testing.
Buy Documents
ASTM D1976-20 - Standard Test Method for Elements in Water by Inductively-Coupled Plasma Atomic Emission Spectroscopy
REDLINE ASTM D1976-20 - Standard Test Method for Elements in Water by Inductively-Coupled Plasma Atomic Emission Spectroscopy
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D1976-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Elements in Water by Inductively-Coupled Plasma Atomic Emission Spectroscopy". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is useful for the determination of element concentrations in many natural waters and wastewaters. It has the capability for the simultaneous determination of up to 29 elements. High sensitivity analysis and larger dynamic range can be achieved for some elements that are difficult to determine by other techniques such as Flame Atomic Absorption. 5.2 The test method is useful for multi-element analysis of domestic and commercial well produced drinking water for metals and nonmetals for use in baseline analysis and monitoring during exploration, hydraulic fracturing, production, closure and reclamation activities related to oil and gas operations (see Guide D8006). 5.2.1 Minimum analyses include arsenic, barium, iron, magnesium, sodium, calcium, manganese, and lead. 5.2.2 Boron, potassium, chromium, selenium, cadmium, and strontium may be required on a site specific basis. 5.2.3 The most abundant elements in oil and gas produced water are sodium, potassium, lithium, magnesium, calcium, strontium, iron, silica, phosphorus, and sulfur. 5.3 The test method is useful for multi-element analysis of acid rock drainage and other major and some trace elements in mining influenced water. 5.4 Where low quantitation limits are required, Test Method D5673 may be applicable. 5.5 The test method is also useful for testing leachates and effluents for ore and mining and metallurgical waste characterization tests including Test Methods D6234, E2242, D5744, and solutions from the Biological Acid Production Potential and Peroxide Acid Generation Methods in the Appendix of Test Methods E1915. SCOPE 1.1 This test method covers the determination of dissolved, total-recoverable, or total elements in drinking water, ground water, surface water, domestic, commercial or industrial wastewaters,2,3 within the following concentration ranges of Table 1. 1.2 It is the user’s responsibility to ensure the validity of the test method for waters of untested matrices. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Note 2 and Section 9. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is useful for the determination of element concentrations in many natural waters and wastewaters. It has the capability for the simultaneous determination of up to 29 elements. High sensitivity analysis and larger dynamic range can be achieved for some elements that are difficult to determine by other techniques such as Flame Atomic Absorption. 5.2 The test method is useful for multi-element analysis of domestic and commercial well produced drinking water for metals and nonmetals for use in baseline analysis and monitoring during exploration, hydraulic fracturing, production, closure and reclamation activities related to oil and gas operations (see Guide D8006). 5.2.1 Minimum analyses include arsenic, barium, iron, magnesium, sodium, calcium, manganese, and lead. 5.2.2 Boron, potassium, chromium, selenium, cadmium, and strontium may be required on a site specific basis. 5.2.3 The most abundant elements in oil and gas produced water are sodium, potassium, lithium, magnesium, calcium, strontium, iron, silica, phosphorus, and sulfur. 5.3 The test method is useful for multi-element analysis of acid rock drainage and other major and some trace elements in mining influenced water. 5.4 Where low quantitation limits are required, Test Method D5673 may be applicable. 5.5 The test method is also useful for testing leachates and effluents for ore and mining and metallurgical waste characterization tests including Test Methods D6234, E2242, D5744, and solutions from the Biological Acid Production Potential and Peroxide Acid Generation Methods in the Appendix of Test Methods E1915. SCOPE 1.1 This test method covers the determination of dissolved, total-recoverable, or total elements in drinking water, ground water, surface water, domestic, commercial or industrial wastewaters,2,3 within the following concentration ranges of Table 1. 1.2 It is the user’s responsibility to ensure the validity of the test method for waters of untested matrices. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Note 2 and Section 9. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D1976-20 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D1976-20 has the following relationships with other standards: It is inter standard links to ASTM D1976-19, ASTM D8006-24, ASTM D1129-13(2020)e2, ASTM D5744-18, ASTM D1066-18, ASTM D1066-18e1, ASTM D8006-16, ASTM D5673-15, ASTM D5744-13, ASTM D5744-13e1, ASTM D4841-88(2013), ASTM D4841-88(2013)e1, ASTM E2242-12a, ASTM D2777-12, ASTM E2242-12e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D1976-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D1976 − 20
Standard Test Method for
Elements in Water by Inductively-Coupled Plasma Atomic
Emission Spectroscopy
This standard is issued under the fixed designation D1976; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
1.1 This test method covers the determination of dissolved, 2.1 ASTM Standards:
total-recoverable, or total elements in drinking water, ground D1066 Practice for Sampling Steam
water, surface water, domestic, commercial or industrial D1129 Terminology Relating to Water
2,3
wastewaters, within the following concentration ranges of D1193 Specification for Reagent Water
Table 1. D2777 Practice for Determination of Precision and Bias of
Applicable Test Methods of Committee D19 on Water
1.2 It is the user’s responsibility to ensure the validity of the
D3370 Practices for Sampling Water from Flowing Process
test method for waters of untested matrices.
Streams
1.3 The values stated in SI units are to be regarded as
D4841 Practice for Estimation of Holding Time for Water
standard. No other units of measurement are included in this
Samples Containing Organic and Inorganic Constituents
standard.
D5673 Test Method for Elements in Water by Inductively
1.4 This standard does not purport to address all of the
Coupled Plasma—Mass Spectrometry
safety concerns, if any, associated with its use. It is the D5744 Test Method for Laboratory Weathering of Solid
responsibility of the user of this standard to establish appro-
Materials Using a Humidity Cell
priate safety, health, and environmental practices and deter- D5810 Guide for Spiking into Aqueous Samples
mine the applicability of regulatory limitations prior to use.
D5847 Practice for Writing Quality Control Specifications
For specific hazard statements, see Note 2 and Section 9. for Standard Test Methods for Water Analysis
1.5 This international standard was developed in accor-
D6234 Test Method for Shake Extraction of Mining Waste
dance with internationally recognized principles on standard-
by the Synthetic Precipitation Leaching Procedure
ization established in the Decision on Principles for the D8006 Guide for Sampling and Analysis of Residential and
Development of International Standards, Guides and Recom-
Commercial Water Supply Wells in Areas of Exploration
mendations issued by the World Trade Organization Technical and Production (E&P) Operations
Barriers to Trade (TBT) Committee.
E1915 Test Methods forAnalysis of Metal Bearing Ores and
Related Materials for Carbon, Sulfur, and Acid-Base
This test method is under the jurisdiction of ASTM Committee D19 on Water Characteristics
and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
E2242 Test Method for Column Percolation Extraction of
in Water.
Mine Rock by the Meteoric Water Mobility Procedure
Current edition approved May 1, 2020. Published June 2020. Originally
2.2 USEPA Standards:
approved in 1991. Last previous edition approved in 2019 as D1976 – 19. DOI:
10.1520/D1976-20.
Method 200.7 Determination of Metals and Trace Elements
The detailed report of EPA Method Study 27, Method 200.7 is available from
in Water and Wastes by Inductively Coupled Plasma-
theNationalTechnicalInformationService,5285PortRoyalRoad,Springfield,VA.
Atomic Emission Spectrometry
A summary of the project is available from the U.S. Environmental Protection
Agency, Environmental Monitoring and Support Laboratory, Cincinnati, OH,
http://www.epa.gov.
3 4
Fishman, M. J. and Friedman, L., “Methods for Determination of Inorganic For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Substances in Water and Fluvial Sediments,” U.S. Geological Survey Techniques of contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Water-Resources Investigations, Book 5, Chapter D1066, Open File Report 85-495, Standards volume information, refer to the standard’s Document Summary page on
1985, p. 659–671. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D1976 − 20
TABLE 1 Summary of Tested Concentration Ranges
4.2 A background correction technique may be used to
Element From To Unit compensate for variable background contribution from high
Aluminum 0.083 1.43 µg/mL concentrations of major and trace elements.
Antimony 0.411 1.41 µg/mL
Arsenic 0.083 0.943 µg/mL
5. Significance and Use
Barium 0.030 250 µg/mL
Beryllium 0.017 0.076 µg/mL
5.1 This test method is useful for the determination of
Boron 0.330 1.18 µg/mL
element concentrations in many natural waters and wastewa-
Cadmium 0.018 0.776 µg/mL
Calcium 0.400 1100 µg/mL ters. It has the capability for the simultaneous determination of
Chromium 0.025 0.47 µg/mL
upto29elements.Highsensitivityanalysisandlargerdynamic
Cobalt 0.058 0.843 µg/mL
range can be achieved for some elements that are difficult to
Copper 0.017 0.189 µg/mL
Iron 0.074 2.34 µg/mL determine by other techniques such as Flame Atomic Absorp-
Lead 0.085 0.943 µg/mL
tion.
Lithium 0.800 450 µg/mL
Magnesium 0.073 4.62 µg/mL
5.2 The test method is useful for multi-element analysis of
Manganese 0.017 0.94 µg/mL
domestic and commercial well produced drinking water for
Molybdenum 0.073 1.09 µg/mL
metals and nonmetals for use in baseline analysis and moni-
Nickel 0.043 0.943 µg/mL
Phosphorus 10.0 310 µg/mL
toring during exploration, hydraulic fracturing, production,
Potassium 8.00 5200 µg/mL
closure and reclamation activities related to oil and gas
Selenium 0.083 0.755 µg/mL
operations (see Guide D8006).
Silica 1.00 3000 µg/mL
Silver 0.017 0.189 µg/mL
5.2.1 Minimum analyses include arsenic, barium, iron,
Sodium 5.00 3500 µg/mL
magnesium, sodium, calcium, manganese, and lead.
Strontium 0.500 500 µg/mL
5.2.2 Boron, potassium, chromium, selenium, cadmium,
Sulfur 2.00 600 µg/mL
Thallium 0.126 0.953 µg/mL
and strontium may be required on a site specific basis.
Vanadium 0.041 1.877 µg/mL
5.2.3 The most abundant elements in oil and gas produced
Zinc 0.068 0.759 µg/mL
water are sodium, potassium, lithium, magnesium, calcium,
strontium, iron, silica, phosphorus, and sulfur.
3. Terminology
5.3 The test method is useful for multi-element analysis of
3.1 Definitions:
acid rock drainage and other major and some trace elements in
3.1.1 For definitions of terms used in this standard, refer to
mining influenced water.
Terminology D1129.
5.4 Where low quantitation limits are required,Test Method
3.2 Definitions of Terms Specific to This Standard:
D5673 may be applicable.
3.2.1 calibration blank, n—a volume of water containing
5.5 The test method is also useful for testing leachates and
the same acid matrix as the calibration standards (see 11.1).
effluents for ore and mining and metallurgical waste charac-
3.2.2 calibration standards, n—a series of known standard
terization tests including Test Methods D6234, E2242, D5744,
solutions used by the analyst for calibration of the instrument
and solutions from the Biological Acid Production Potential
(preparation of the analytical curve) (see 8.9).
andPeroxideAcidGenerationMethodsintheAppendixofTest
Methods E1915.
3.2.3 instrumental detection limit, n—the concentration
equivalent to a signal, due to the analyte, that is equal to three
6. Interferences
times the standard deviation of a series of ten replicate
measures of a reagent-blank signal at the same wavelength.
6.1 Several types of interference effects may contribute to
inaccuracies in the determination of trace elements. These
3.2.4 laboratory control sample, n—a solution with the
interferences can be summarized as follows:
certified concentration(s) of the analytes.
6.1.1 Spectral interferences can be categorized as (1) over-
3.2.5 method blank, n—a volume of water carried through
lap of a spectral line from another element; (2) unresolved
the entire sample preparation, preservation, and analytical
overlapofmolecularbandspectra;(3)backgroundcontribution
procedure.
from continuous or recombination phenomena; and (4) back-
3.2.6 reagent blank, n—a volume of water containing the
groundcontributionfromstraylightfromlineemissionofhigh
same matrix as the calibration standards, carried through the
concentration elements.
entire analytical procedure.
6.1.1.1 The effects described in 6.1.1 can be compensated
3.2.7 total,n—theconcentrationdeterminedonanunfiltered
forbyutilizingacomputercorrectionoftherawdata,requiring
sample following vigorous digestion (see 12.3).
the monitoring and measurement of the interfering element.
The second effect may require selection of an alternate wave-
3.2.8 total-recoverable, adj—determinable by the digestion
length.Thethirdandfourtheffectscanusuallybecompensated
method that is included in this procedure (see 12.2).
for by a background correction adjacent to the analyte line.
4. Summary of Test Method
6.1.1.2 Table 2 lists some interference effects for the rec-
4.1 Elements are determined, either sequentially or ommended wavelengths given in Table 2. The data in Table 2
simultaneously, by inductively-coupled plasma atomic emis- are intended for use only as a rudimentary guide for the
sion spectroscopy. indication of potential spectral interferences. For this purpose,
D1976 − 20
A
TABLE 2 Analyte Concentration Equivalents, mg/L, Arising from Interferents at the 100 mg/L Level
Interferent
Wavelength,
Analyte
nm
Al Ca Cr Cu Fe Mg Mn Ni Ti V
Aluminum 308.215 . . . . . . 0.21 . . 1.4
Antimony 206.833 0.47 . 2.9 . 0.08 . . . 0.25 0.45
Arsenic 193.696 1.3 . 0.44 . . . . . . 1.1
Barium 455.403 . . . . . . . . . .
Beryllium 313.042 . . . . . . . . 0.04 0.05
Boron 249.773 0.04 . . . 0.32 . . . . .
Cadmium 226.502 . . . . 0.03 . . 0.02 . .
Calcium 317.933 . . 0.08 . 0.01 0.01 0.04 . 0.03 0.03
Chromium 267.716 . . . . 0.003 . 0.04 . . 0.04
Cobalt 228.616 . . 0.03 . 0.005 . . 0.03 0.15 .
Copper 324.754 . . . . 0.003 . . . 0.05 0.02
Iron 259.940 . . . . . 0.12 0.12 . . .
Lead 220.353 0.17 . . . . . . . . .
Magnesium 279.079 . 0.02 0.11 . 0.13 0.002 0.25 . 0.07 0.12
Manganese 257.610 0.005 . 0.01 . 0.002 . . . . .
Molybdenum 202.030 0.05 . . . 0.03 . . . . .
Nickel 231.604 . . . . . . . . . .
Selenium 196.026 0.23 . . . 0.09 . . . . .
Silicon 288.158 . . 0.07 . . . . . . 0.01
Sodium 588.995 . . . . . . . . 0.08 .
Thallium 190.864 0.30 . . . . . . . . .
Vanadium 292.402 . . 0.05 . 0.005 . . . 0.02 .
Zinc 213.856 . . . 0.14 . . . 0.29 . .
A
See Table 4 for concentrations used.
TABLE 4 Interferent Elemental Concentrations for Analytes
A
Tested
linear relations between concentration and intensity for the
Interferents mg/L
analytes and the interferents can be assumed.
Al 1000
6.1.1.3 Only those interferents listed in Table 2 were inves-
Ca 1000
tigated for the analytes in Table 3. The blank spaces in Table 2 Cr 200
Cu 200
indicate that measurable interferences were not observed for
Fe 1000
the interferent concentrations listed in Table 4. Generally,
Mg 1000
interferences were considered as discernible if the interferent Mn 200
Ni 200
produced interference peaks or background shifts that corre-
Ti 200
sponded to 2 to 5 % of the peaks generated by the analyte
V 200
concentrations listed in Table 3.
A
This table indicates concentrations used for interference measurements in Table
2.
TABLE 3 Analyte Elemental Concentrations Tested for
A
Interferents
6.1.2 Physical interferences are generally considered to be
Analytes mg/L
effects associated with the sample nebulization and transport
Al 10
As 10 processes. Such properties as change in viscosity and surface
B10
tension can cause significant inaccuracies, especially in
Ba 1
samples that may contain high dissolved solids or acid
Be 1
Ca 1 concentrations, or both. The use of a peristaltic pump may
Cd 10
lessen these interferences. If these types of interferences are
Co 1
operative,theymustbereducedbydilutionofthesesamplesor
Cr 1
Cu 1 utilization of standard addition techniques, or both.
Fe 1
6.1.2.1 Salt buildup at the tip of the nebulizer is another
Mg 1
problem that can occur from high dissolved solids. This salt
Mn 1
Na 10 buildup affects aerosol flow rate that can cause instrumental
Ni 10
drift. To control this problem, wet the argon prior to
Pb 10
nebulization, use a tip washer, or dilute the sample.
Sb 10
Se 10
NOTE 1—Periodic inspection and cleaning of the nebulizer and torch
Si 1
components are highly recommended.
Tl 10
V1
6.1.2.2 Reports indicate that better control of the argon flow
Zn 10
rate improves instrument performance. This control of the
A
This table indicates concentrations used for interference measurements in Table
argon flow rate can be accomplished with the use of mass flow
2.
controllers.
D1976 − 20
6.1.3 Chemicalinterferencesarecharacterizedbymolecular may also vary. In time, other elements may be added as more
compound formation, ionization effects, and solute vaporiza- information becomes available and as required.
tioneffects.Normallytheseeffectsarenotpronouncedwiththe 7.1.1 Use of a vacuum or purged path is necessary for
inductively coupled plasma (ICP) technique; however, if determination of sulfur.
observed, they can be minimized by careful selection of 7.1.2 Use of glass in the sample path may not be acceptable
operating conditions (incident power, plasma observation for silica, use of an inert material is recommended to avoid
position, and so forth), by buffering the sample, by matrix silica contamination.
matching, and by standard addition procedures. These types of
8. Reagents and Materials
interferences can be highly dependent on matrix type and the
specific analyte.
8.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
6.2 Analysis for silica precludes the use of borosilicate
reagents shall conform to the specifications of the Committee
glassware due to potential contamination.
on Analytical Reagents of the American Chemical Society.
The high sensitivity of inductively-coupled argon plasma
7. Apparatus
atomic emission spectrometry may require reagents of higher
7.1 See the manufacturer’s instruction manual for installa-
purity. Stock standard solutions are prepared from high purity
tion and operation of inductively-coupled argon plasma spec-
metals, oxides, or nonhydroscopic reagent grade salts using
trometers. Table 5 lists elements for which this test method
Types I, II, and III reagent water, and ultrapure acids. Other
applies, with recommended wavelengths and typical estimated
grades may be used, provided it is first ascertained that the
instrumental detection limits using conventional pneumatic
reagentisofsufficientpuritytopermititsusewithoutlessening
nebulization. Actual working detection limits are sample de-
the accuracy of the determination.
pendent and as the sample matrix varies, these detection limits
8.2 Purity of Water—Unless otherwise indicated, reference
towatershallbeunderstoodtomeanreagentwaterconforming
to Type I, II, or III of Specification D1193. It is the analyst’s
TABLE 5 Suggested Wavelengths and Estimated Detection
responsibilitytoassurethatwaterisfreeofinterferences.Other
A
Limits
reagent water types may be used provided it is first ascertained
Wavelength, Estimated detection limit,
Element
B C
that the water is of sufficiently high purity to permit its use
nm µg/L
without adversely affecting the precision and bias of the test
Aluminum 308.215 45
Arsenic 193.696 53
method. Type II water was specified at the time of round robin
Antimony 206.833 32
testing of this test method.
Barium 455.403 2
Beryllium 313.042 0.3
8.3 Aqua Regia—Mix three parts hydrochloric acid (sp gr
Boron 249.773 5
1.19) and one part concentrated nitric acid (sp gr 1.42) just
Cadmium 226.502 4
Calcium 317.933 10
before use.
Chromium 267.716 7
Cobalt 228.616 7 NOTE 2—Exercise caution when mixing this reagent, use of a fume
Copper 324.754 6
hood is recommended.
Iron 259.940 7
8.4 Argon—Welding grade equivalent or better.
Lead 220.353 42
Lithium 670.784 4
8.5 Hydrochloric Acid (1+1)—Add 1 volume of hydro-
Magnesium 279.079 30
chloric acid (sp gr 1.19) ultrapure or equivalent to 1 volume of
Manganese 257.610 2
Molybdenum 202.030 8
water.
Nickel 231.604 15
Phosphorous 214.914 76 8.6 Nitric Acid(1+1)—Add 1 volume of nitric acid (sp gr
Potassium 766.491 700
1.42) ultrapure or equivalent to 1 volume of water.
Selenium 196.026 75
Silica 288.158 27
8.7 Nitric Acid (1 + 499)—Add 1 volume of nitric acid (sp
Silver 328.068 7
gr 1.42) ultrapure or equivalent to 499 volumes of water.
Sodium 588.995 29
Strontium 421.552 0.77
8.8 Stock Solutions—Preparationofexamplestocksolutions
Sulfur 182.037 3
for each element is listed in Table 6. Use of commercially
Thallium 190.864 40
Vanadium 292.402 8 prepared certified stock solutions is recommended.
Zinc 213.856 2
8.9 Mixed Calibration Standard Solutions—Prepare mixed
A
Winge, R. K., Fassel, V. A., Peterson, V. J., and Floyd, M. A., “Inductively
calibration standard solutions by combining appropriate vol-
Coupled Plasma-Atomic Emission Spectroscopy,” An Atlas of Spectral Information,
Elsevier Science Publishing Co., Inc., New York, NY, 1985. umes of the stock solutions in volumetric flasks (see Note 3).
B
The wavelengths listed are recommended because of their sensitivity and overall
acceptance. Other wavelengths may be substituted if they can provide the needed
sensitivity and are treated with the same corrective techniques for spectral
Reagent Chemicals, American Chemical Society Specifications, American
interference (see 6.1.1).
C A
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
The estimated detection limits as shown are taken from Winge et al., USEPA
Method 200.7, or task group data. They are given as a guide for approximate
listed by the American Chemical Society, see Annual Standards for Laboratory
detection limits for the listed wavelengths. The actual test method instrumental
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
detection limits are sample-dependent and may vary as the sample matrix varies
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
(see 3.2.3).
MD.
D1976 − 20
A,B
TABLE 6 Preparation of Example Element Stock Solutions
10.1.1 Analysis for silica precludes the use of borosilicate
Element (Compound) Weight, g Solvent
glassware due to potential contamination.
Al 0.1000 HCl (1 + 1)
10.2 Preservethesamplesbyimmediatelyaddingnitricacid
Sb 0.1000 Aqua regia
C
As O 0.1320 Water + 0.4 g NaOH to adjust the pH to 2 at the time of collection. Normally, 2 mL
2 3
E
BaCl 0.1516 HCl (1 + 1)
of HNO is required per L of sample. If only dissolved
Be 0.1000 Aqua regia
elements are to be determined, filter the sample through a
H BO 0.5716 Water
3 3
Cd 0.1000 HNO (sp gr 1.42) 0.45-µm membrane filter before acidification (see Note 4).The
F
CaCO 0.2498 Water + HCl (1 + 1)
holding time for the sample may be calculated in accordance
Cr 0.1000 HCl (1 + 1)
with Practice D4841.
Co 0.1000 HNO (1+1)
Cu 0.1000 HNO (1+1)
NOTE 4—Depending on the manufacturer, some filters have been found
Fe 0.1000 HNO (sp gr 1.42)
to be contaminated to various degrees with heavy metals. Care should be
Pb 0.1000 HNO (sp gr 1.42)
exercised in selecting a source for these filters. It is good practice to wash
Li CO 0.5323 HNO (1+1)
2 3 3
the filters with dilute nitric acid and a small portion of the sample before
Mg 0.1000 HNO (1+1)
Mn 0.1000 HNO (1+1)
filtering.
Ni 0.1000 HNO (sp gr 1.42)
NH H PO 0.3745 HCl (1 + 9) 11. Calibration and Standardization
4 2 4
KCl 0.1907 Water
11.1 Calibrate the instrument over a suitable concentration
(NH ) MoO 0.2043 Water
4 2 4
D
Na SeO 0.2393 Water
range for the elements chosen by atomizing the calibration
2 4
Na SiO ·5H O 0.3531 Water
2 3 2
blank and mixed standard solutions and recording their con-
Ag 0.1000 HNO (sp gr 1.42)
centrations and signal intensities.
NaCl 0.2542 Water
SrCO 0.1685 HCl (1 + 9)
3 11.1.1 Multiple-point calibration standards may be used,
Na SO 0.4431 Water
2 4
and it is the user’s responsibility to ensure the validity of the
TlNO 0.1303 Water
test method. Regardless of the calibration procedure used,
NH VO 0.2297 HNO (1+1)
4 3 3
Zn 0.1000 HNO (1+1)
3 appropriate quality control (QC) is required to verify the
A
Example element stock solutions, 1.00 mL = 100 µg of metal. Dissolve the listed
calibration curve at the anticipated concentration range(s)
weights of each compound or metal in 20 mL of specified solvent and dilute to 1
before proceeding to the sample analysis. It is recommended
L. The metals may require heat to increase rate of dissolution.
B
that the calibration blank and standard(s) be matrix matched
Where water is used as the solvent, acidify with 10 mL of HNO (sp gr 1.42) and
dilute to 1 L. See Section 8 for concentration of acids. Commercially available
with the same acid concentration contained in the samples.
standards may be used. Alternative salts or oxides may also be used.
C
Add 2 mL of HNO (sp gr 1.42) and dilute to 1 L.
3 12. Procedure
D
Add 1 mL of HNO (sp gr 1.42) and dilute to 1 L.
E
Dry for 1 h at 180°C. 12.1 To determine dissolved elements, proceed with 12.4.
F
Dryfor1hat 180°C. Add to approximately 600 mL of water and dissolve
12.2 When determining total-recoverable elements for a
cautiously with a minimum of dilute HCl. Dilute to 1 L with water.
two-point calibration, choose a volume of a well mixed,
acid-preserved sample appropriate for the expected level of
Prior to preparing mixed standards, each stock solution should
elements.
be analyzed separately to determine possible spectral interfer-
12.2.1 Transfer the sample to a beaker (use tetrafluoroeth-
ence or the presence of impurities. Care should be taken when
ylene or equivalent for silica analysis) and add 2 mLof HNO
preparing the mixed standards to ensure the elements are
(1 + 1) and 10 mL of HCl (1 + 1) and heat on a steam bath or
compatible and stable. It is common practice to have all or
hot plate until the volume has been reduced to near 25 mL,
nearly all elements in one mixed calibration standard.
making certain the sample does not boil. Cool the sample, and
if necessary filter or let insoluble material settle to avoid
NOTE 3—Mixed calibration standards will vary depending on the
clogging of the nebulizer. Adjust to the original sample
number of elements being determined. An example of mixed calibration
standards for the simultaneous determination of 20 elements is as follows: volume.Todeterminetotal-recoverableelements,proceedwith
12.4.
Mixed Standard Solution I—manganese, beryllium, cadmium, lead, and zinc
Mixed Standard Solution II—copper, vanadium, iron, and cobalt
12.3 When determining total elements (hard digestion),
Mixed Standard Solution III—molybdenum, arsenic, and selenium
Mixed Standard Solution IV—aluminum, chromium, and nickel
choose a volume of well mixed, acid-preserved sample appro-
Mixed Standard Solution V—antimony, boron, magnesium, silver, and
priate for the expected level of elements.
thallium
NOTE 5—Addition of HF acid may be required in order to effect
9. Hazards
complete dissolution of all siliceous material, so this digestion method
will result in low results for samples containing insoluble silica.
9.1 The toxicity or carcinogenicity of each reagent used in
this test method has not been precisely defined; however, each 12.3.1 Transfer the sample to a beaker (use tetrafluoroeth-
chemical should be treated as a potential health hazard. ylene or equivalent for silica analysis).Add 3 mLof HNO (sp
Adequate precautions should be taken to minimize personnel gr 1.42). Place the beaker on a hot plate and cautiously
exposure to chemicals used in this procedure. evaporate to near dryness, making certain that the sample does
not boil and that no area of the bottom of the beaker is allowed
10. Sampling
to go dry. Cool the beaker and add 5 mLof HNO (sp gr 1.42).
10.1 Collect the samples in accordance with Practices Cover the beaker with a watch glass (use tetrafluoroethylene or
D1066 or Practices D3370 as applicable. equivalent for silica analysis) and return it to the hot plate.
D1976 − 20
Increase the temperature of the hot plate so a gentle reflux 14. Precision and Bias
action occurs. Continue heating, adding additional acid as
14.1 The original precision and bias data for this test
necessary, until the digestion is complete (generally indicated
method are based on an interlaboratory study conducted by the
when the digestate is light in color or does not change in 2
U.S. Environmental Protection Agency.
appearance with continued refluxing).Again, evaporate to near
14.2 The test design of the study meets the requirements of
dryness and cool the beaker.Add 10 mLof HCl (1 + 1) and 15
Practice D2777 – 86 for elements listed in this test method
mLof water per 100 mLof final solution and warm the beaker
during the original testing.
gently for 15 min to dissolve any precipitate or residue
14.2.1 Theoriginalstudytestdesignwasbasedonaformof
resulting from evaporation.Allow the sample to cool, wash the
the analysis of variance applying the approach and methods of
beakerwallsandwatchglasswithwater,andifnecessary,filter
the Youden Unit block design. In the Youden nonreplicate
or let insoluble material settle to avoid clogging the nebulizer.
approachtodeterminingtheprecisionandbiasoftheanalytical
Adjust to the original sample volume. To determine total
method,pairsofsamplesofsimilarbutdifferentconcentrations
elements, proceed with 12.4.
are analyzed. The key in the Youden approach is to estimate
NOTE 6—Many laboratories have found block digestion systems a
precision from analyses of Youden pairs rather than through
useful way to digest samples for trace metals analysis. Systems typically
replicateanalyses.Inthereferencedstudy,fiveYoudenpairsof
consist of either a metal or graphite block with wells to hold digestion
spike materials were prepared (Guide D5810). Six water types
tubes. The block temperature controller must be able to maintain unifor-
were included. Only the data from reagent water and surface
mity of temperature across all positions of the block. For trace metals
analysis, the digestion tubes should be constructed of polypropylene and
water are presented here, from the original study but the data
have a volume accuracy of at least 0.5 %. All lots of tubes should come
for the four other water types can be found in the original
with a certificate of analysis to demonstrate suitability for their intended
interlaboratory study report. Each water type was spiked with
purpose.
three of the five Youden pairs with the exception of reagent
12.4 Atomize each solution to record its emission intensity
water,whichwasspikedwithallfiveYoudenpairs.Eachwater
or concentration.Asample rinse of HNO (1 + 499) is recom-
sample was prepared for analysis by both a total and a
mended between samples.
total-recoverable digestion procedure. A total of twelve labo-
ratories participated in the study.
13. Calculation
14.2.2 Type II water was specified for this round robin.
14.2.3 Twenty-sevendifferentelementswereincludedinthe
13.1 Include the blank in the calibration as the zero point.
original study and individual measurements of precision and
NOTE 7—The original interlaboratory study subtracted reagent blanks
bias were developed for each element and matrix tested. Bias
from all samples. This subtraction was particularly important for digested
was related to mean recovery of the analyte.The equation used
samples requiring large quantities of acids to complete the digestion.
13.2 If dilutions are required, apply the appropriate dilution
Supporting data have been filed at ASTM International Headquarters and may
factor to sample values.
be obtained by requesting Resea
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D1976 − 19 D1976 − 20
Standard Test Method for
Elements in Water by Inductively-Coupled Plasma Atomic
Emission Spectroscopy
This standard is issued under the fixed designation D1976; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of dissolved, total-recoverable, or total elements in drinking water, ground water,
2,3
surface water, domestic, commercial or industrial wastewaters, within the following concentration ranges of Table 1.
1.2 It is the user’s responsibility to ensure the validity of the test method for waters of untested matrices.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. For specific hazard statements, see Note 2 and Section 9.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1066 Practice for Sampling Steam
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Flowing Process Streams
D4841 Practice for Estimation of Holding Time for Water Samples Containing Organic and Inorganic Constituents
D5673 Test Method for Elements in Water by Inductively Coupled Plasma—Mass Spectrometry
D5744 Test Method for Laboratory Weathering of Solid Materials Using a Humidity Cell
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
D6234 Test Method for Shake Extraction of Mining Waste by the Synthetic Precipitation Leaching Procedure
D8006 Guide for Sampling and Analysis of Residential and Commercial Water Supply Wells in Areas of Exploration and
Production (E&P) Operations
E1915 Test Methods for Analysis of Metal Bearing Ores and Related Materials for Carbon, Sulfur, and Acid-Base Characteristics
E2242 Test Method for Column Percolation Extraction of Mine Rock by the Meteoric Water Mobility Procedure
2.2 USEPA Standards:
Method 200.7 Determination of Metals and Trace Elements in Water and Wastes by Inductively Coupled Plasma-Atomic
Emission Spectrometry
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.05 on Inorganic Constituents in Water.
Current edition approved Nov. 1, 2019May 1, 2020. Published December 2019June 2020. Originally approved in 1991. Last previous edition approved in 20182019 as
D1976 – 18.D1976 – 19. DOI: 10.1520/D1976-19.10.1520/D1976-20.
The detailed report of EPA Method Study 27, Method 200.7 is available from the National Technical Information Service, 5285 Port Royal Road, Springfield, VA. A
summary of the project is available from the U.S. Environmental Protection Agency, Environmental Monitoring and Support Laboratory, Cincinnati, OH, http://www.epa.gov.
Fishman, M. J. and Friedman, L., “Methods for Determination of Inorganic Substances in Water and Fluvial Sediments,” U.S. Geological Survey Techniques of
Water-Resources Investigations, Book 5, Chapter D1066, Open File Report 85-495, 1985, p. 659–671.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D1976 − 20
TABLE 1 Summary of Tested Concentration Ranges
Element From To Unit
Aluminum 0.083 1.43 μg/mL
Antimony 0.411 1.41 μg/mL
Arsenic 0.083 0.943 μg/mL
Barium 0.030 250 μg/mL
Beryllium 0.017 0.076 μg/mL
Boron 0.330 1.18 μg/mL
Cadmium 0.018 0.776 μg/mL
Calcium 0.400 1100 μg/mL
Chromium 0.025 0.47 μg/mL
Cobalt 0.058 0.843 μg/mL
Copper 0.017 0.189 μg/mL
Iron 0.074 2.34 μg/mL
Lead 0.085 0.943 μg/mL
Lithium 0.800 450 μg/mL
Magnesium 0.073 4.62 μg/mL
Manganese 0.017 0.94 μg/mL
Molybdenum 0.073 1.09 μg/mL
Nickel 0.043 0.943 μg/mL
Phosphorus 10.0 310 μg/mL
Potassium 8.00 5200 μg/mL
Selenium 0.083 0.755 μg/mL
Silica 1.00 3000 μg/mL
Silver 0.017 0.189 μg/mL
Sodium 5.00 3500 μg/mL
Strontium 0.500 500 μg/mL
Sulfur 2.00 600 μg/mL
Thallium 0.126 0.953 μg/mL
Vanadium 0.041 1.877 μg/mL
Zinc 0.068 0.759 μg/mL
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 calibration blank, n—a volume of water containing the same acid matrix as the calibration standards (see 11.1).
3.2.2 calibration standards, n—a series of known standard solutions used by the analyst for calibration of the instrument
(preparation of the analytical curve) (see 8.9).
3.2.3 instrumental detection limit, n—the concentration equivalent to a signal, due to the analyte, that is equal to three times the
standard deviation of a series of ten replicate measures of a reagent-blank signal at the same wavelength.
3.2.4 laboratory control sample, n—a solution with the certified concentration(s) of the analytes.
3.2.5 method blank, n—a volume of water carried through the entire sample preparation, preservation, and analytical procedure.
3.2.6 reagent blank, n—a volume of water containing the same matrix as the calibration standards, carried through the entire
analytical procedure.
3.2.7 total, n—the concentration determined on an unfiltered sample following vigorous digestion (see 12.3).
3.2.8 total-recoverable, adj—determinable by the digestion method that is included in this procedure (see 12.2).
4. Summary of Test Method
4.1 Elements are determined, either sequentially or simultaneously, by inductively-coupled plasma atomic emission spectros-
copy.
4.2 A background correction technique may be used to compensate for variable background contribution from high
concentrations of major and trace elements.
5. Significance and Use
5.1 This test method is useful for the determination of element concentrations in many natural waters and wastewaters. It has
the capability for the simultaneous determination of up to 29 elements. High sensitivity analysis and larger dynamic range can be
achieved for some elements that are difficult to determine by other techniques such as Flame Atomic Absorption.
5.2 The test method is useful for multi-element analysis of domestic and commercial well produced drinking water for metals
and nonmetals for use in baseline analysis and monitoring during exploration, hydraulic fracturing, production, closure and
reclamation activities related to oil and gas operations (see Guide D8006).
D1976 − 20
5.2.1 Minimum analyses include arsenic, barium, iron, magnesium, sodium, calcium, manganese, and lead.
5.2.2 Boron, potassium, chromium, selenium, cadmium, and strontium may be required on a site specific basis.
5.2.3 The most abundant elements in oil and gas produced water are sodium, potassium, lithium, magnesium, calcium,
strontium, iron, silica, phosphorus, and sulfur.
5.3 The test method is useful for multi-element analysis of acid rock drainage and other major and some trace elements in
mining influenced water.
5.4 Where low quantitation limits are required, Test Method D5673 may be applicable.
5.5 The test method is also useful for testing leachates and effluents for ore and mining and metallurgical waste characterization
tests including Test Methods D6234, E2242, D5744, and solutions from the Biological Acid Production Potential and Peroxide
Acid Generation Methods in the Appendix of Test Methods E1915.
6. Interferences
6.1 Several types of interference effects may contribute to inaccuracies in the determination of trace elements. These
interferences can be summarized as follows:
6.1.1 Spectral interferences can be categorized as (1) overlap of a spectral line from another element; (2) unresolved overlap
of molecular band spectra; (3) background contribution from continuous or recombination phenomena; and (4) background
contribution from stray light from line emission of high concentration elements.
6.1.1.1 The effects described in 6.1.1 can be compensated for by utilizing a computer correction of the raw data, requiring the
monitoring and measurement of the interfering element. The second effect may require selection of an alternate wavelength. The
third and fourth effects can usually be compensated for by a background correction adjacent to the analyte line.
6.1.1.2 Table 2 lists some interference effects for the recommended wavelengths given in Table 2. The data in Table 2 are
intended for use only as a rudimentary guide for the indication of potential spectral interferences. For this purpose, linear relations
between concentration and intensity for the analytes and the interferents can be assumed.
6.1.1.3 Only those interferents listed in Table 2 were investigated for the analytes in Table 3. The blank spaces in Table 2
indicate that measurable interferences were not observed for the interferent concentrations listed in Table 4. Generally,
interferences were considered as discernible if the interferent produced interference peaks or background shifts that corresponded
to 2 to 5 % of the peaks generated by the analyte concentrations listed in Table 3.
6.1.2 Physical interferences are generally considered to be effects associated with the sample nebulization and transport
processes. Such properties as change in viscosity and surface tension can cause significant inaccuracies, especially in samples that
may contain high dissolved solids or acid concentrations, or both. The use of a peristaltic pump may lessen these interferences.
If these types of interferences are operative, they must be reduced by dilution of these samples or utilization of standard addition
techniques, or both.
6.1.2.1 Salt buildup at the tip of the nebulizer is another problem that can occur from high dissolved solids. This salt buildup
affects aerosol flow rate that can cause instrumental drift. To control this problem, wet the argon prior to nebulization, use a tip
washer, or dilute the sample.
A
TABLE 2 Analyte Concentration Equivalents, mg/L, Arising from Interferents at the 100 mg/L Level
Interferent
Wavelength,
Analyte
nm
Al Ca Cr Cu Fe Mg Mn Ni Ti V
Aluminum 308.215 . . . . . . 0.21 . . 1.4
Antimony 206.833 0.47 . 2.9 . 0.08 . . . 0.25 0.45
Arsenic 193.696 1.3 . 0.44 . . . . . . 1.1
Barium 455.403 . . . . . . . . . .
Beryllium 313.042 . . . . . . . . 0.04 0.05
Boron 249.773 0.04 . . . 0.32 . . . . .
Cadmium 226.502 . . . . 0.03 . . 0.02 . .
Calcium 317.933 . . 0.08 . 0.01 0.01 0.04 . 0.03 0.03
Chromium 267.716 . . . . 0.003 . 0.04 . . 0.04
Cobalt 228.616 . . 0.03 . 0.005 . . 0.03 0.15 .
Copper 324.754 . . . . 0.003 . . . 0.05 0.02
Iron 259.940 . . . . . 0.12 0.12 . . .
Lead 220.353 0.17 . . . . . . . . .
Magnesium 279.079 . 0.02 0.11 . 0.13 0.002 0.25 . 0.07 0.12
Manganese 257.610 0.005 . 0.01 . 0.002 . . . . .
Molybdenum 202.030 0.05 . . . 0.03 . . . . .
Nickel 231.604 . . . . . . . . . .
Selenium 196.026 0.23 . . . 0.09 . . . . .
Silicon 288.158 . . 0.07 . . . . . . 0.01
Sodium 588.995 . . . . . . . . 0.08 .
Thallium 190.864 0.30 . . . . . . . . .
Vanadium 292.402 . . 0.05 . 0.005 . . . 0.02 .
Zinc 213.856 . . . 0.14 . . . 0.29 . .
A
See Table 4 for concentrations used.
D1976 − 20
TABLE 3 Analyte Elemental Concentrations Tested for
A
Interferents
Analytes mg/L
Al 10
As 10
B 10
Ba 1
Be 1
Ca 1
Cd 10
Co 1
Cr 1
Cu 1
Fe 1
Mg 1
Mn 1
Na 10
Ni 10
Pb 10
Sb 10
Se 10
Si 1
Tl 10
V 1
Zn 10
A
This table indicates concentrations used for interference measurements in Table
2.
TABLE 4 Interferent Elemental Concentrations for Analytes
A
Tested
Interferents mg/L
Al 1000
Ca 1000
Cr 200
Cu 200
Fe 1000
Mg 1000
Mn 200
Ni 200
Ti 200
V 200
A
This table indicates concentrations used for interference measurements in Table
2.
NOTE 1—Periodic inspection and cleaning of the nebulizer and torch components are highly recommended.
6.1.2.2 Reports indicate that better control of the argon flow rate improves instrument performance. This control of the argon
flow rate can be accomplished with the use of mass flow controllers.
6.1.3 Chemical interferences are characterized by molecular compound formation, ionization effects, and solute vaporization
effects. Normally these effects are not pronounced with the inductively coupled plasma (ICP) technique; however, if observed, they
can be minimized by careful selection of operating conditions (incident power, plasma observation position, and so forth), by
buffering the sample, by matrix matching, and by standard addition procedures. These types of interferences can be highly
dependent on matrix type and the specific analyte.
6.2 Analysis for silica precludes the use of borosilicate glassware due to potential contamination.
7. Apparatus
7.1 See the manufacturer’s instruction manual for installation and operation of inductively-coupled argon plasma spectrometers.
Table 5 lists elements for which this test method applies, with recommended wavelengths and typical estimated instrumental
detection limits using conventional pneumatic nebulization. Actual working detection limits are sample dependent and as the
sample matrix varies, these detection limits may also vary. In time, other elements may be added as more information becomes
available and as required.
7.1.1 Use of a vacuum or purged path is necessary for determination of sulfur.
7.1.2 Use of glass in the sample path may not be acceptable for silica, use of an inert material is recommended to avoid silica
contamination.
D1976 − 20
TABLE 5 Suggested Wavelengths and Estimated Detection
A
Limits
Wavelength, Estimated detection limit,
Element
B C
nm μg/L
Aluminum 308.215 45
Arsenic 193.696 53
Antimony 206.833 32
Barium 455.403 2
Beryllium 313.042 0.3
Boron 249.773 5
Cadmium 226.502 4
Calcium 317.933 10
Chromium 267.716 7
Cobalt 228.616 7
Copper 324.754 6
Iron 259.940 7
Lead 220.353 42
Lithium 670.784 4
Magnesium 279.079 30
Manganese 257.610 2
Molybdenum 202.030 8
Nickel 231.604 15
Phosphorous 214.914 76
Potassium 766.491 700
Selenium 196.026 75
Silica 288.158 27
Silver 328.068 7
Sodium 588.995 29
Strontium 421.552 0.77
Sulfur 182.037 3
Thallium 190.864 40
Vanadium 292.402 8
Zinc 213.856 2
A
Winge, R. K., Fassel, V. A., Peterson, V. J., and Floyd, M. A., “Inductively
Coupled Plasma-Atomic Emission Spectroscopy,” An Atlas of Spectral Information,
Elsevier Science Publishing Co., Inc., New York, NY, 1985.
B
The wavelengths listed are recommended because of their sensitivity and overall
acceptance. Other wavelengths may be substituted if they can provide the needed
sensitivity and are treated with the same corrective techniques for spectral
interference (see 6.1.1).
C A
The estimated detection limits as shown are taken from Winge et al., USEPA
Method 200.7, or task group data. They are given as a guide for approximate
detection limits for the listed wavelengths. The actual test method instrumental
detection limits are sample-dependent and may vary as the sample matrix varies
(see 3.2.3).
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society. The high
sensitivity of inductively-coupled argon plasma atomic emission spectrometry may require reagents of higher purity. Stock
standard solutions are prepared from high purity metals, oxides, or nonhydroscopic reagent grade salts using Types I, II, and III
reagent water, and ultrapure acids. Other grades may be used, provided it is first ascertained that the reagent is of sufficient purity
to permit its use without lessening the accuracy of the determination.
8.2 Purity of Water—Unless otherwise indicated, reference to water shall be understood to mean reagent water conforming to
Type I, II, or III of Specification D1193. It is the analyst’s responsibility to assure that water is free of interferences. Other reagent
water types may be used provided it is first ascertained that the water is of sufficiently high purity to permit its use without
adversely affecting the precision and bias of the test method. Type II water was specified at the time of round robin testing of this
test method.
8.3 Aqua Regia—Mix three parts hydrochloric acid (sp gr 1.19) and one part concentrated nitric acid (sp gr 1.42) just before
use.
NOTE 2—Exercise caution when mixing this reagent, use of a fume hood is recommended.
8.4 Argon—Welding grade equivalent or better.
8.5 Hydrochloric Acid (1 + 1)—Add 1 volume of hydrochloric acid (sp gr 1.19) ultrapure or equivalent to 1 volume of water.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For Suggestions on the testing of reagents not listed by
the American Chemical Society, see Annual Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D1976 − 20
8.6 Nitric Acid (1 + 1)—Add 1 volume of nitric acid (sp gr 1.42) ultrapure or equivalent to 1 volume of water.
8.7 Nitric Acid (1 + 499)—Add 1 volume of nitric acid (sp gr 1.42) ultrapure or equivalent to 499 volumes of water.
8.8 Stock Solutions—Preparation of example stock solutions for each element is listed in Table 6. Use of commercially prepared
certified stock solutions is recommended.
8.9 Mixed Calibration Standard Solutions—Prepare mixed calibration standard solutions by combining appropriate volumes of
the stock solutions in volumetric flasks (see Note 3). Prior to preparing mixed standards, each stock solution should be analyzed
separately to determine possible spectral interference or the presence of impurities. Care should be taken when preparing the mixed
standards to ensure the elements are compatible and stable. It is common practice to have all or nearly all elements in one mixed
calibration standard.
NOTE 3—Mixed calibration standards will vary depending on the number of elements being determined. An example of mixed calibration standards
for the simultaneous determination of 20 elements is as follows:
Mixed Standard Solution I—manganese, beryllium, cadmium, lead, and zinc
Mixed Standard Solution II—copper, vanadium, iron, and cobalt
Mixed Standard Solution III—molybdenum, arsenic, and selenium
Mixed Standard Solution IV—aluminum, chromium, and nickel
Mixed Standard Solution V—antimony, boron, magnesium, silver, and
thallium
9. Hazards
9.1 The toxicity or carcinogenicity of each reagent used in this test method has not been precisely defined; however, each
chemical should be treated as a potential health hazard. Adequate precautions should be taken to minimize personnel exposure to
chemicals used in this procedure.
A,B
TABLE 6 Preparation of Example Element Stock Solutions
Element (Compound) Weight, g Solvent
Al 0.1000 HCl (1 + 1)
Sb 0.1000 Aqua regia
C
As O 0.1320 Water + 0.4 g NaOH
2 3
E
BaCl 0.1516 HCl (1 + 1)
Be 0.1000 Aqua regia
H BO 0.5716 Water
3 3
Cd 0.1000 HNO (sp gr 1.42)
F
CaCO 0.2498 Water + HCl (1 + 1)
Cr 0.1000 HCl (1 + 1)
Co 0.1000 HNO (1 + 1)
Cu 0.1000 HNO (1 + 1)
Fe 0.1000 HNO (sp gr 1.42)
Pb 0.1000 HNO (sp gr 1.42)
Li CO 0.5323 HNO (1 + 1)
2 3 3
Mg 0.1000 HNO (1 + 1)
Mn 0.1000 HNO (1 + 1)
Ni 0.1000 HNO (sp gr 1.42)
NH H PO 0.3745 HCl (1 + 9)
4 2 4
KCl 0.1907 Water
(NH ) MoO 0.2043 Water
4 2 4
D
Na SeO 0.2393 Water
2 4
Na SiO ·5H O 0.3531 Water
2 3 2
Ag 0.1000 HNO (sp gr 1.42)
NaCl 0.2542 Water
SrCO 0.1685 HCl (1 + 9)
Na SO 0.4431 Water
2 4
TlNO 0.1303 Water
NH VO 0.2297 HNO (1 + 1)
4 3 3
Zn 0.1000 HNO (1 + 1)
A
Example element stock solutions, 1.00 mL = 100 μg of metal. Dissolve the listed
weights of each compound or metal in 20 mL of specified solvent and dilute to 1
L. The metals may require heat to increase rate of dissolution.
B
Where water is used as the solvent, acidify with 10 mL of HNO (sp gr 1.42) and
dilute to 1 L. See Section 8 for concentration of acids. Commercially available
standards may be used. Alternative salts or oxides may also be used.
C
Add 2 mL of HNO (sp gr 1.42) and dilute to 1 L.
D
Add 1 mL of HNO (sp gr 1.42) and dilute to 1 L.
E
Dry for 1 h at 180°C.
F
Dry for 1 h at 180°C. Add to approximately 600 mL of water and dissolve
cautiously with a minimum of dilute HCl. Dilute to 1 L with water.
D1976 − 20
10. Sampling
10.1 Collect the samples in accordance with Practices D1066 or Practices D3370 as applicable.
10.1.1 Analysis for silica precludes the use of borosilicate glassware due to potential contamination.
10.2 Preserve the samples by immediately adding nitric acid to adjust the pH to 2 at the time of collection. Normally, 2 mL of
HNO is required per L of sample. If only dissolved elements are to be determined, filter the sample through a 0.45-μm membrane
filter before acidification (see Note 4). The holding time for the sample may be calculated in accordance with Practice D4841.
NOTE 4—Depending on the manufacturer, some filters have been found to be contaminated to various degrees with heavy metals. Care should be
exercised in selecting a source for these filters. It is good practice to wash the filters with dilute nitric acid and a small portion of the sample before
filtering.
11. Calibration and Standardization
11.1 Calibrate the instrument over a suitable concentration range for the elements chosen by atomizing the calibration blank and
mixed standard solutions and recording their concentrations and signal intensities.
11.1.1 Multiple-point calibration standards may be used, and it is the user’s responsibility to ensure the validity of the test
method. Regardless of the calibration procedure used, appropriate quality control (QC) is required to verify the calibration curve
at the anticipated concentration range(s) before proceeding to the sample analysis. It is recommended that the calibration blank and
standard(s) be matrix matched with the same acid concentration contained in the samples.
12. Procedure
12.1 To determine dissolved elements, proceed with 12.4.
12.2 When determining total-recoverable elements for a two-point calibration, choose a volume of a well mixed, acid-preserved
sample appropriate for the expected level of elements.
12.2.1 Transfer the sample to a beaker (use tetrafluoroethylene or equivalent for silica analysis) and add 2 mL of HNO (1 + 1)
and 10 mL of HCl (1 + 1) and heat on a steam bath or hot plate until the volume has been reduced to near 25 mL, making certain
the sample does not boil. Cool the sample, and if necessary filter or let insoluble material settle to avoid clogging of the nebulizer.
Adjust to the original sample volume. To determine total-recoverable elements, proceed with 12.4.
12.3 When determining total elements (hard digestion), choose a volume of well mixed, acid-preserved sample appropriate for
the expected level of elements.
NOTE 5—Addition of HF acid may be required in order to effect complete dissolution of all siliceous material, so this digestion method will result in
low results for samples containing insoluble silica.
12.3.1 Transfer the sample to a beaker (use tetrafluoroethylene or equivalent for silica analysis). Add 3 mL of HNO (sp gr
1.42). Place the beaker on a hot plate and cautiously evaporate to near dryness, making certain that the sample does not boil and
that no area of the bottom of the beaker is allowed to go dry. Cool the beaker and add 5 mL of HNO (sp gr 1.42). Cover the beaker
with a watch glass (use tetrafluoroethylene or equivalent for silica analysis) and return it to the hot plate. Increase the temperature
of the hot plate so a gentle reflux action occurs. Continue heating, adding additional acid as necessary, until the digestion is
complete (generally indicated when the digestate is light in color or does not change in appearance with continued refluxing).
Again, evaporate to near dryness and cool the beaker. Add 10 mL of HCl (1 + 1) and 15 mL of water per 100 mL of final solution
and warm the beaker gently for 15 min to dissolve any precipitate or residue resulting from evaporation. Allow the sample to cool,
wash the beaker walls and watch glass with water, and if necessary, filter or let insoluble material settle to avoid clogging the
nebulizer. Adjust to the original sample volume. To determine total elements, proceed with 12.4.
NOTE 6—Many laboratories have found block digestion systems a useful way to digest samples for trace metals analysis. Systems typically consist of
either a metal or graphite block with wells to hold digestion tubes. The block temperature controller must be able to maintain uniformity of temperature
across all positions of the block. For trace metals analysis, the digestion tubes should be constructed of polypropylene and have a volume accuracy of
at least 0.5 %. All lots of tubes should come with a certificate of analysis to demonstrate suitability for their intended purpose.
12.4 Atomize each solution to record its emission intensity or concentration. A sample rinse of HNO (1 + 499) is recommended
between samples.
13. Calculation
13.1 Include the blank in the calibration as the zero point.
NOTE 7—The original interlaboratory study subtracted reagent blanks from all samples. This subtraction was particularly important for digested
samples requiring large quantities of acids to complete the digestion.
13.2 If dilutions are required, apply the appropriate dilution factor to sample values.
13.3 Report results in the calibration concentration units.
D1976 − 20
TABLE 7 Regression Equations for Bias and Precision, μg/L, Reagent Water versus Surface Water
(Aluminum, Antimony, Arsenic, Beryllium)
NOTE 1—X = mean recovery; C = true value for the concentration.
Water Type Aluminum Antimony Arsenic Beryllium
Total Digestion
Applicable concentration range (83 to 1434) (411 to 1406) (83 to 943) (17 to 76)
Reagent water, hard
Single-analyst precision S = 0.05X + 3.72 S = 0.23X − 50.17 S = 0.07X + 8.28 S = 0.02X + 0.18
o o o o
Overall precision S = 0.07X + 9.34 S = 0.21X − 24.02 S = 0.11X + 2.96 S = 0.02X + 0.91
t t t t
Bias X = 0.91C + 6.62 X = 0.74C + 2.27 X = 1.03C − 12.03 X = 1.02C − 1.92
Surface water, hard
Single-analyst precision S = 0.00X + 40.75 S = 0.11X − 0.14 S = 0.05X + 7.79 S = 0.00X + 0.85
o o o o
Overall precision S = 0.10X + 67.23 S = 0.07X + 35.71 S = 0.10X + 10.55 S = 0.09X − 0.47
t t t t
Bias X = 0.98C + 90.54 X = 0.88C − 55.19 X = 1.00C − 16.02 X = 1.00C − 0.89
Total-Recoverable Digestion
Applicable concentration range (83 to 1434) (411 to 1406) (83 to 943) (17 to 76)
Reagent water, soft
Single-analyst precision S = 0.05X + 25.05 S = 0.06X + 7.85 S = 0.07X + 6.12 S = 0.04X + 0.14
o o o o
Overall precision S = 0.10X + 28.72 S = 0.05X + 20.10 S = 0.12X + 2.99 S = 0.07X − 0.47
t t t t
Bias X = 0.93C + 28.40 X = 0.92C − 22.46 X = 1.01C − 2.08 X = 1.03C − 0.73
Surface water, soft
Single-analyst precision S = 0.01X + 34.72 S = 0.06X + 0.97 S = 0.05X + 9.29 S = 0.02X + 0.43
o o o o
Overall precision S = 0.10X + 74.75 S = 0.07X + 14.28 S = 0.11X + 1.82 S = 0.01X + 15.4
t t t t
Bias X = 1.02C + 40.42 X = 0.95C − 34.50 X = 1.06C − 7.00 X = 1.04C − 2.08
14. Precision and Bias
14.1 The original precision and bias data for this test method are based on an interlaboratory study conducted by the U.S.
Environmental Protection Agency.
14.2 The test design of the study meets the requirements of Practice D2777 – 86 for elements listed in this test method during
the original testing.
14.2.1 The original study test design was based on a form of the analysis of variance applying the approach and methods of
the Youden Unit block design. In the Youde
...


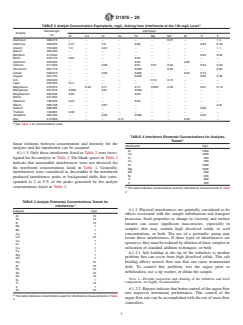





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...