ASTM D1072-23
(Test Method)Standard Test Method for Total Sulfur in Fuel Gases by Combustion and Barium Chloride Titration
Standard Test Method for Total Sulfur in Fuel Gases by Combustion and Barium Chloride Titration
ABSTRACT
This test method is for the determination of total sulfur in combustible fuel gases and is applicable to natural gases, manufactured gases, mixed gases, and other miscellaneous gaseous fuels. For the use of barium chloride titration following collection of sulfur dioxide by alternative procedures, ammonia, amines, substances producing water soluble cations, and fluorides will interfere with the titration. The apparatus includes the following: (1) burner, (2) chimneys, absorbers, and spray traps, (3) flow meter, (4) vacuum system, (5) air-purifying system, and (6) monometer. The schematic diagrams of the gas burner, combustion and absorption apparatus, suction system, and purified air system are provided. Reagent grade chemicals shall be used in all tests and include: (1) water, (2) denatured ethyl or isopropyl alcohol, (3) barium chloride, standard solution, (4) hydrochloric acid, (5) hydrogen peroxide, (6) iso-propanol, (7) potassium hydrogen phthalate, (8) phenolphthalein, (9) methyl orange indicator solution, (10) silver nitrate solution, (11) sodium carbonate solution, (12) sodium hydroxide solution, (13) sulfuric acid, (14) tetrahydroxyquinone indicator, and (15) thorin indicator. The procedure for the following are detailed: (1) calibration and standardization of sodium hydroxide, sulfuric acid, and barium chloride solutions, (2) preparation of apparatus, (3) sulfur determination, (4) analysis of absorbent, and (5) quality assurance. The formula of calculating the volume of gas in standard cubic feet burned during the determination and the concentration of sulfur from the results of titration are given.
SCOPE
1.1 This test method is for the determination of total sulfur in combustible fuel gases, when present in sulfur concentrations between approximately 25 and 700 mg/m3 (1 to 30 grains per 100 cubic feet). It is applicable to natural gases, manufactured gases, mixed gases, and other miscellaneous gaseous fuels.
1.2 The values stated in inch-pound units are to be regarded as standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-May-2023
- Technical Committee
- D03 - Gaseous Fuels
Relations
- Effective Date
- 15-Dec-2019
- Effective Date
- 01-Jul-2016
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Jun-2000
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM D1072-23 is the internationally recognized standard test method for determining total sulfur content in combustible fuel gases using combustion and barium chloride titration. Developed by ASTM International, this method applies to a variety of gaseous fuels, including natural gas, manufactured gas, mixed gases, and other miscellaneous fuel gases. Accurate measurement of sulfur concentration is essential for regulatory compliance, process control, and minimizing environmental impact.
The scope of ASTM D1072-23 covers sulfur concentrations in fuel gases between approximately 25 and 700 mg/m³ (1 to 30 grains per 100 cubic feet). Its methodology is widely adopted in industries where accurate sulfur analysis in fuel gases is necessary for both operational efficiency and regulatory conformity.
Key Topics
- Applicability: Suitable for natural, manufactured, and mixed fuel gases, as well as other miscellaneous combustible gases.
- Measurement Range: Effective for sulfur concentrations from 25 to 700 mg/m³.
- Combustion Method: A metered gas sample is combusted in a closed system with sulfur-free air to generate sulfur oxides.
- Absorption & Titration: Sulfur oxides produced during combustion are absorbed in sodium carbonate and oxidized to sulfate. The resulting sulfate is determined by titration with barium chloride solution, using indicators for endpoint detection.
- Accuracy & Reproducibility: Allows for precise and repeatable results due to standardized procedures for calibration, sample preparation, and titration.
- Interferences: The method is robust against most interferences, though substances such as ammonia, amines, water-soluble cations, and fluorides can interfere if alternative SO₂ collection procedures are used.
- Quality Assurance: Requires duplicate sampling and quality control checks with known standards to ensure validity and traceability of results.
- Safety Precautions: Users are responsible for establishing proper safety, health, and environmental protocols due to hazardous chemicals and combustion processes involved.
Applications
ASTM D1072-23 is vital for industries reliant on fuel gases, including:
- Natural Gas Utilities: Ensures fuel quality and regulatory compliance regarding sulfur emissions.
- Petrochemical and Refining Sectors: Monitors and controls sulfur content in feedstocks and process gases to protect equipment and minimize corrosion.
- Environmental Compliance: Provides sulfur data necessary for emissions reporting and conformance to air quality standards.
- Industrial Fuel Users: Supports process optimization by monitoring fuel gas purity.
- Quality Control Laboratories: Used for routine fuel gas analysis as part of quality management systems.
Routine implementation of this standard helps industries safeguard operations, support environmental stewardship, and optimize combustion efficiency by carefully monitoring and controlling total sulfur levels in fuel gases.
Related Standards
- ASTM D1193: Standard Specification for Reagent Water - details water quality for use in analytical procedures, including those in D1072-23.
- ASTM D4150: Terminology Relating to Gaseous Fuels - provides definitions relevant to the analysis of gaseous fuels.
- ISO and EN Standards: There are multiple international methods focusing on sulfur determination in gaseous fuels; users may reference these for additional guidance or regulatory alignment.
Keywords: total sulfur, fuel gases, combustion analysis, barium chloride titration, sulfur determination, natural gas, manufactured gas, ASTM D1072-23, gaseous fuel standards, industrial gas testing, sulfur emissions, environmental compliance.
Buy Documents
ASTM D1072-23 - Standard Test Method for Total Sulfur in Fuel Gases by Combustion and Barium Chloride Titration
REDLINE ASTM D1072-23 - Standard Test Method for Total Sulfur in Fuel Gases by Combustion and Barium Chloride Titration
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D1072-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Total Sulfur in Fuel Gases by Combustion and Barium Chloride Titration". This standard covers: ABSTRACT This test method is for the determination of total sulfur in combustible fuel gases and is applicable to natural gases, manufactured gases, mixed gases, and other miscellaneous gaseous fuels. For the use of barium chloride titration following collection of sulfur dioxide by alternative procedures, ammonia, amines, substances producing water soluble cations, and fluorides will interfere with the titration. The apparatus includes the following: (1) burner, (2) chimneys, absorbers, and spray traps, (3) flow meter, (4) vacuum system, (5) air-purifying system, and (6) monometer. The schematic diagrams of the gas burner, combustion and absorption apparatus, suction system, and purified air system are provided. Reagent grade chemicals shall be used in all tests and include: (1) water, (2) denatured ethyl or isopropyl alcohol, (3) barium chloride, standard solution, (4) hydrochloric acid, (5) hydrogen peroxide, (6) iso-propanol, (7) potassium hydrogen phthalate, (8) phenolphthalein, (9) methyl orange indicator solution, (10) silver nitrate solution, (11) sodium carbonate solution, (12) sodium hydroxide solution, (13) sulfuric acid, (14) tetrahydroxyquinone indicator, and (15) thorin indicator. The procedure for the following are detailed: (1) calibration and standardization of sodium hydroxide, sulfuric acid, and barium chloride solutions, (2) preparation of apparatus, (3) sulfur determination, (4) analysis of absorbent, and (5) quality assurance. The formula of calculating the volume of gas in standard cubic feet burned during the determination and the concentration of sulfur from the results of titration are given. SCOPE 1.1 This test method is for the determination of total sulfur in combustible fuel gases, when present in sulfur concentrations between approximately 25 and 700 mg/m3 (1 to 30 grains per 100 cubic feet). It is applicable to natural gases, manufactured gases, mixed gases, and other miscellaneous gaseous fuels. 1.2 The values stated in inch-pound units are to be regarded as standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This test method is for the determination of total sulfur in combustible fuel gases and is applicable to natural gases, manufactured gases, mixed gases, and other miscellaneous gaseous fuels. For the use of barium chloride titration following collection of sulfur dioxide by alternative procedures, ammonia, amines, substances producing water soluble cations, and fluorides will interfere with the titration. The apparatus includes the following: (1) burner, (2) chimneys, absorbers, and spray traps, (3) flow meter, (4) vacuum system, (5) air-purifying system, and (6) monometer. The schematic diagrams of the gas burner, combustion and absorption apparatus, suction system, and purified air system are provided. Reagent grade chemicals shall be used in all tests and include: (1) water, (2) denatured ethyl or isopropyl alcohol, (3) barium chloride, standard solution, (4) hydrochloric acid, (5) hydrogen peroxide, (6) iso-propanol, (7) potassium hydrogen phthalate, (8) phenolphthalein, (9) methyl orange indicator solution, (10) silver nitrate solution, (11) sodium carbonate solution, (12) sodium hydroxide solution, (13) sulfuric acid, (14) tetrahydroxyquinone indicator, and (15) thorin indicator. The procedure for the following are detailed: (1) calibration and standardization of sodium hydroxide, sulfuric acid, and barium chloride solutions, (2) preparation of apparatus, (3) sulfur determination, (4) analysis of absorbent, and (5) quality assurance. The formula of calculating the volume of gas in standard cubic feet burned during the determination and the concentration of sulfur from the results of titration are given. SCOPE 1.1 This test method is for the determination of total sulfur in combustible fuel gases, when present in sulfur concentrations between approximately 25 and 700 mg/m3 (1 to 30 grains per 100 cubic feet). It is applicable to natural gases, manufactured gases, mixed gases, and other miscellaneous gaseous fuels. 1.2 The values stated in inch-pound units are to be regarded as standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D1072-23 is classified under the following ICS (International Classification for Standards) categories: 75.160.30 - Gaseous fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D1072-23 has the following relationships with other standards: It is inter standard links to ASTM D4150-19, ASTM D4150-08(2016), ASTM D4150-08, ASTM D1193-06, ASTM D4150-03, ASTM D4150-00, ASTM D1193-99, ASTM D1193-99e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D1072-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D1072 − 23
Standard Test Method for
Total Sulfur in Fuel Gases by Combustion and Barium
Chloride Titration
This standard is issued under the fixed designation D1072; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope are absorbed in sodium carbonate solution, where they are
oxidized to sulfate. The sulfate in the absorbent solution is
1.1 This test method is for the determination of total sulfur
determined by titration with standardized barium chloride
in combustible fuel gases, when present in sulfur concentra-
3 solution, using tetra-hydroxy-quinone (THQ) as an indicator.
tions between approximately 25 and 700 mg/m (1 to 30 grains
per 100 cubic feet). It is applicable to natural gases, manufac-
5. Interferences
tured gases, mixed gases, and other miscellaneous gaseous
5.1 There are no known interferences for the determination
fuels.
of total sulfur in fuel gases when combustion is followed by
1.2 The values stated in inch-pound units are to be regarded
barium chloride titration. However, users employing barium
as standard.
chloride titration following collection of sulfur dioxide by
1.3 This standard does not purport to address all of the
alternative procedures are cautioned that ammonia, amines,
safety concerns, if any, associated with its use. It is the
substances producing water soluble cations, and fluorides will
responsibility of the user of this standard to establish appro-
interfere with the titration.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
6. Apparatus
1.4 This international standard was developed in accor-
6.1 Burner (Fig. 1), as specified in the Appendix X1.
dance with internationally recognized principles on standard-
6.2 Chimneys, Absorbers and Spray Traps, (Fig. 2), as
ization established in the Decision on Principles for the
specified in the Appendix X1.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
6.3 Flow meter—A calibrated capillary flow meter for
Barriers to Trade (TBT) Committee.
predetermining and indicating the rate of flow of gas to the
burner. The capillary selected should be of such size that at the
2. Referenced Documents
required rate of flow the differential pressure is at least 20 cm
2.1 ASTM Standards:
of water. A scale divided into millimeters will then provide a
D1193 Specification for Reagent Water
reading precision of 6 0.5 %. Other metering devices, includ-
D4150 Terminology Relating to Gaseous Fuels
ing but not limited to rotameters or dry displacement meters,
are suitable provided the reading precision is 6 0.5 % or better.
3. Terminology
A flow controlling valve is attached to the inlet connection of
3.1 For definitions of general terms used in D03 Gaseous
the flow meter.
Fuels standards, refer to Terminology D4150.
6.4 Vacuum System—A vacuum manifold equipped with a
vacuum regulating device, valves, and other necessary accou-
4. Summary of Test Method
terments. An example vacuum system capable of performing
4.1 A metered sample of gas is burned in a closed system in
multiple test measurements is shown in Fig. 3. Other vacuum
an atmosphere of sulfur-free air. The oxides of sulfur produced
system configurations can be used to perform this test method.
The vacuum system shall be connected to a vacuum pump
capable of providing a steady gas flow of 3 L of air per minute
This test method is under the jurisdiction of ASTM Committee D03 on Gaseous
Fuels and is the direct responsibility of D03.06.03 on Analysis by Spectroscopy.
through each absorber and capable of maintaining a constant
Current edition approved June 1, 2023. Published June 2023. Originally
manifold pressure of approximately 40 cm of water below
approved in 1954. Last previous edition approved in 2017 as D1072 – 06 (2017).
atmospheric pressure.
DOI: 10.1520/D1072-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
6.5 Air-Purifying System—A device supplying purified air
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
to the burner manifold at a constant pressure of approximately
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. 200 mm of water and to the chimney manifold at a pressure of
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D1072 − 23
7.5 Hydrochloric Acid (CAS No 7647-01-0)
(2.275-g HCl ⁄L)—Titrated against Na CO solution (see
2 3
7.15), using methyl orange indicator. Adjusted such that 1 mL
of HCl solution is equivalent to 1 mL of Na CO solution.
2 3
7.6 Hydrogen Peroxide (30 %) (H O ;CAS No: 7722-84-1).
2 2
7.7 iso-Propanol (CAS No. 67630)
7.8 Potassium Hydrogen Phthalate (KHP; CAS No 877-
24-7) —Dry use.
7.9 Phenolphthalein (CAS No 77-09-8)
7.10 Methyl Orange (CAS No 547-58-0) Indicator
Solution—Dissolve 0.1 g of methyl orange in 100 mL of water.
7.11 Silver Nitrate (CAS No 7761-88-8)Solution (17-g
NOTE 1—All dimensions in millimetres.
AgNO /L)—Dissolve 1.7 g of silver nitrate (AgNO ) in
3 3
FIG. 1 Gas Burner for Sulfur Determination
100 mL of water. Store in a brown bottle.
7.12 Sodium Carbonate (CAS No 5968-11-6) Solution
(3.306-g Na CO /L)—Dissolve 3.306 g of sodium carbonate
1 cm to 2 cm of water. An example system configuration for
2 3
(Na CO ) in water and dilute to 1 L.
multiple tests is illustrated in Fig. 4; however, other air- 2 3
purifying system configurations can be used to perform this test
7.13 Sodium Hydroxide Solution (CAS No 1310-73-2)
method. The tubing that connects the chimneys to the manifold
(100-g NaOH/L)—Dissolve 100 g of technical grade sodium
shall be of an internal diameter not smaller than 0.63 cm in
hydroxide (NaOH) pellets in water and dilute to 1 L. Standard-
order to prevent unnecessary restriction of airflow.
ize against potassium hydrogen phthalate (See 6.1)
6.6 Manometer—A water manometer for indicating the gas
7.14 Sulfuric Acid(CAS No 7664-93-9) (1 + 16)—Mix
pressure at the point of volume measurement. It is connected
60 mL of concentrated sulfuric acid (H SO , sp gr 1.84) with
2 4
between the flowmeter and the burner, with one leg open to the
960 mL of water.
atmosphere.
7.15 Tetrahydroxyquinone Indicator (THQ CAS No. 5676-
48-2), in powdered form.
7. Reagents and Materials
7.16 Thorin indicator— (CAS No. 132-33-2)
7.1 Reagents Purity—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
8. Calibration and Standardization
all reagents shall conform to the specifications of the Commit-
tee on Analytical Reagents of the American Chemical Society,
8.1 Sodium Hydroxide Solution Standardization— The fol-
where such specifications are available. Other grades may be
lowing provides an example procedure for standardization;
used, provided it is first ascertained that the reagent is of
other quantities of reagents, as convenient, can be used. Dry
sufficiently high purity to permit its use without adversely
and crushed potassium hydrogen phthalate (KHP) is heated in
impacting the accuracy of the determination. Warning—
an oven at 105 °C for 2 h and allowed to cool to room
Sodium hydroxide is corrosive and can cause severe damage to
temperature in a desiccator. KHP (950 mg 6 50 mg weighed to
eyes and skin. Inhalation will irritate the nose, throat and lungs.
the nearest 0.1 mg) is placed in an Erlenmeyer flask. Water (70
It reacts exothermically with water.
mL) and 2-4 drops of phenolphthalein are added. Titrate the
KHP solution with the sodium hydroxide solution prepared
7.2 Purity of Water—Unless otherwise indicated, references
to water shall be understood to mean reagent water conforming under 7.13 to a faint pink color. Repeat the titration using a
second portion of KHP. Titrate a 70 mL water blank containing
to Specification D1193.
1-4 drops of phenolphthalein to a faint pink color using the
7.3 Alcohol—Ethyl alcohol, denatured by Formula 30 or
sodium hydroxide solution prepared under 7.13. Repeat this
3-A, or isopropyl alcohol.
procedure and average the results. For both the water blank and
7.4 Barium Chloride,(CAS No: 10361-37-2), Standard So-
the KHP titration replicates should agree to 0.05 mL titrant. For
lution (1 mL = 1 mg S)—Dissolve 7.634 g of barium chloride
each KHP trial, independently calculate the normality for the
(BaCl ·2H O) in water and dilute to 1 L. The solution is
2 2
sodium hydroxide solution according to the following equa-
standardized gravimetrically by precipitation as barium sulfate
tion:
or by titration against sulfuric acid (see 7.12)
mg KHP/204.23
Normality of NaOH 5 (1)
mL NaOH 2 avg. mL blank
~ !
Values for the two KHP trials should agree within 6 0.5 %.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
If they do not, repeat the titrations or identify the cause for
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by the American Chemical the excessive discrepancy, or both.
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
8.2 Sulfuric Acid Standardization—The following provides
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. an example procedure for standardization; other quantities of
D1072 − 23
NOTE 1—In the case of those dimensions for which no specific tolerances are designated above, the permissible variation is 610 % to the nearest 1 mm,
provided, however, that in no case shall the deviation be greater than 5 mm.
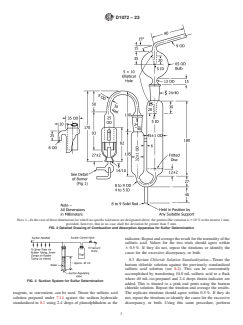
FIG. 2 Detailed Drawing of Combustion and Absorption Apparatus for Sulfur Determination
indicator. Repeat and average the result for the normality of the
sulfuric acid. Values for the two trials should agree within
6 0.5 %. If they do not, repeat the titrations or identify the
cause for the excessive discrepancy, or both.
8.3 Barium Chloride Solution Standardization—Titrate the
barium chloride solution against the previously standardized
sulfuric acid solution (see 8.2). This can be conveniently
accomplished by transferring 10.0 mL sulfuric acid to a flask
where 40 mL iso-propanol and 2-4 drops thorin indicator are
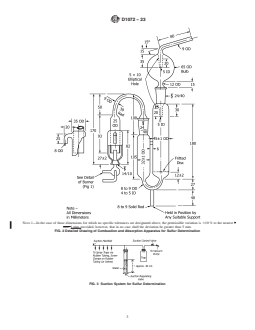
FIG. 3 Suction System for Sulfur Determination
added. This is titrated to a pink end point using the barium
chloride solution. Repeat the titration and average the results.
reagents, as convenient, can be used. Titrate the sulfuric acid The replicate titrations should agree within 0.5 %. If they do
solution prepared under 7.14 against the sodium hydroxide not, repeat the titrations or identify the cause for the excessive
standardized in 8.1 using 2-4 drops of phenolphthalein as the discrepancy, or both. Using this same procedure, perform
D1072 − 23
the air-inlet tube in the vacuum regulator by sliding it in a rubber sleeve.
9.4 When first assembling the apparatus, connect the gas
sample line using glass or aluminum tubing to the inlet of the
flowmeter. Connect the outlet of the flowmeter in a similar way
to the lower side arm of the burner. Adjust the valve for
controlling the rate of flow of gas so that gas is burned at a rate
to liberate approximately 250 Btu ⁄h to 500 Btu ⁄h (Note 2).
This rate should be indicated by two index marks on the
columns of the flowmeter U-tube or timing a rate-index device.
Make the primary air connection from the purified air line to
the upper side arm of the burner by means of rubber or plastic
tubing.
NOTE 2—Using this gas rate, the chimney and absorber should not
become overheated during a test. The appropriate volumetric rate of gas
flow will therefore depend on the heating value of the gas being tested.
9.5 Wash the spray trap, absorber, and chimney well with
water before each test. Charge the larger bulb of the absorber
with 10 mL of Na CO solution (Note 3) and 20 mL of water.
2 3
Attach the spray trap and chimney, and connect them,
FIG. 4 Purified Air System for Sulfur Determination
respectively, to the vacuum line and to the purified air line
using rubber or plastic tubing. Close the chimney opening
duplicate blank titrations using water in place of sulfuric acid
using a cork or other suitable plug.
solution. The replicate titrations should agree within 0.5 %.
NOTE 3—This quantity of Na CO solution is adequate to absorb the
Calculate the normality of the barium chloride solution accord- 2 3
SO from the combustion products of 1 ft of gas containing 15 grains of
ing to the following equation:
3 3 3
sulfur per 100 ft (0.03 m of gas containing 350 mg/m of sulfur.) For
higher concentrations of sulfur in the gas, the volume of Na CO solution
10.0 mL x N Sulfuri
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D1072 − 06 (Reapproved 2017) D1072 − 23
Standard Test Method for
Total Sulfur in Fuel Gases by Combustion and Barium
Chloride Titration
This standard is issued under the fixed designation D1072; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method is for the determination of total sulfur in combustible fuel gases, when present in sulfur concentrations
between approximately 25 and 700 mg/m (1 to 30 grains per 100 cubic feet). It is applicable to natural gases, manufactured gases,
mixed gases, and other miscellaneous gaseous fuels.
1.2 The values stated in inch-pound units are to be regarded as standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D4150 Terminology Relating to Gaseous Fuels
3. Terminology
3.1 For definitions of general terms used in D03 Gaseous Fuels standards, refer to Terminology D4150.
4. Summary of Test Method
4.1 A metered sample of gas is burned in a closed system in an atmosphere of sulfur-free air. The oxides of sulfur produced are
absorbed in sodium carbonate solution, where they are oxidized to sulfate. The sulfate in the absorbent solution is determined by
titration with standardized barium chloride solution, using tetra-hydroxy-quinone (THQ) as an indicator.
5. Interferences
5.1 There are no known interferences for the determination of total sulfur in fuel gases when combustion is followed by barium
This test method is under the jurisdiction of ASTM Committee D03 on Gaseous Fuels and is the direct responsibility of D03.05 on Determination of Special Constituents
of Gaseous Fuels.
Current edition approved Nov. 1, 2017June 1, 2023. Published December 2017June 2023. Originally approved in 1954. Last previous edition approved in 20122017 as
D1072 – 06 (2017).(2012). DOI: 10.1520/D1072-06R17.10.1520/D1072-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D1072 − 23
chloride titration. However, users employing barium chloride titration following collection of sulfur dioxide by alternative
procedures are cautioned that ammonia, amines, substances producing water soluble cations, and fluorides will interfere with the
titration.
6. Apparatus
6.1 Burner (Fig. 1), as specified in the Appendix X1.
6.2 Chimneys, Absorbers and Spray Traps, (Fig. 2), as specified in the Appendix X1.
6.3 Flow meter—A calibrated capillary flow meter for predetermining and indicating the rate of flow of gas to the burner. The
capillary selected should be of such size that at the required rate of flow the differential pressure is at least 20 cm of water. A scale
divided into millimeters will then provide a reading precision of 6 0.5 %. Other metering devices, including but not limited to
rotameters or dry displacement meters, are suitable provided the reading precision is 6 0.5 % or better. A flow controlling valve
is attached to the inlet connection of the flow meter.
6.4 Vacuum System—A vacuum manifold equipped with a vacuum regulating device, valves, and other necessary accouterments.
An example vacuum system capable of performing multiple test measurements is shown in Fig. 3. Other vacuum system
configurations can be used to perform this test method. The vacuum system shall be connected to a vacuum pump capable of
providing a steady gas flow of 3 L of air per minute through each absorber and capable of maintaining a constant manifold pressure
of approximately 40 cm of water below atmospheric pressure.
6.5 Air-Purifying System—A device supplying purified air to the burner manifold at a constant pressure of approximately 200 mm
of water and to the chimney manifold at a pressure of 1 to 2 cm 1 cm to 2 cm of water. An example system configuration for
multiple tests is illustrated in Fig. 4; however, other air-purifying system configurations can be used to perform this test method.
The tubing that connects the chimneys to the manifold shall be of an internal diameter not smaller than 0.63 cm in order to prevent
unnecessary restriction of airflow.
6.6 Manometer—A water manometer for indicating the gas pressure at the point of volume measurement. It is connected between
the flowmeter and the burner, with one leg open to the atmosphere.
7. Reagents and Materials
7.1 Reagents Purity—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all reagents
shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such
NOTE 1—All dimensions in millimetres.
FIG. 1 Gas Burner for Sulfur Determination
D1072 − 23
NOTE 1—In the case of those dimensions for which no specific tolerances are designated above, the permissible variation is 610 % to the nearest 1
mm, 1 mm, provided, however, that in no case shall the deviation be greater than 5 mm.
FIG. 2 Detailed Drawing of Combustion and Absorption Apparatus for Sulfur Determination
FIG. 3 Suction System for Sulfur Determination
D1072 − 23
FIG. 4 Purified Air System for Sulfur Determination
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without adversely impacting the accuracy of the determination. Warning— Sodium hydroxide is corrosive and
can cause severe damage to eyes and skin. Inhalation will irritate the nose, throat and lungs. It reacts exothermically with water.
7.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Specification D1193.
7.3 Alcohol—Ethyl alcohol, denatured by Formula 30 or 3-A, or isopropyl alcohol.
7.4 Barium Chloride,(CAS No: 10361-37-2), Standard Solution (1 mL = 1 mg S)—Dissolve 7.634 g of barium chloride
(BaCl ·2H O) in water and dilute to 1 L. The solution is standardized gravimetrically by precipitation as barium sulfate or by
2 2
titration against sulfuric acid (see 6.127.12)
7.5 Hydrochloric Acid (CAS No 7647-01-0)
(2.275-g (2.275-g HCl HCl/L)⁄L)—Titrated against Na CO solution (see 6.157.15), using methyl orange indicator. Adjusted such
2 3
that 1 mL of HCl solution is equivalent to 1 mL of Na CO solution.
2 3
7.6 Hydrogen Peroxide (30 %) (H O ;CAS No: 7722-84-1).
2 2
7.7 iso-Propanol (CAS No. 67630)
7.8 Potassium Hydrogen Phthalate (KHP; CAS No 877-24-7) —Dry use.
7.9 Phenolphthalein (CAS No 77-09-8)
7.10 Methyl Orange (CAS No 547-58-0) Indicator Solution—Dissolve 0.1 g of methyl orange in 100 mL of water.
7.11 Silver Nitrate (CAS No 7761-88-8)Solution (17-g AgNO /L)—Dissolve 1.7 g of silver nitrate (AgNO ) in 100 mL 100 mL
3 3
of water. Store in a brown bottle.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
D1072 − 23
7.12 Sodium Carbonate (CAS No 5968-11-6) Solution (3.306-g Na CO /L)—Dissolve 3.306 g of sodium carbonate (Na CO ) in
2 3 2 3
water and dilute to 1 L.
7.13 Sodium Hydroxide Solution (CAS No 1310-73-2) (100-g NaOH/L)—Dissolve 100 g of technical grade sodium hydroxide
(NaOH) pellets in water and dilute to 1 L. Standardize against potassium hydrogen phthalate (See 6.16.1))
7.14 Sulfuric Acid(CAS No 7664-93-9) (1 + 16)—Mix 60 mL 60 mL of concentrated sulfuric acid (H SO , sp gr 1.84) with 960
2 4
mL 960 mL of water.
7.15 Tetrahydroxyquinone Indicator (THQ CAS No. 5676-48-2), in powdered form.
7.16 Thorin indicator— (CAS No. 132-33-2)
8. Calibration and Standardization
8.1 Sodium Hydroxide Solution Standardization— The following provides an example procedure for standardization; other
quantities of reagents, as convenient, can be used. Dry and crushed potassium hydrogen phthalate (KHP) is heated in an oven at
105 °C for 2 hoursh and allowed to cool to room temperature in a desiccator. KHP (950 6 50 mg (950 mg 6 50 mg weighed to
the nearest 0.1 mg) is placed in an Erlenmeyer flask. Water (70 mL) and 2-4 drops of phenolphthalein are added. Titrate the KHP
solution with the sodium hydroxide solution prepared under 6.137.13 to a faint pink color. Repeat the titration using a second
portion of KHP. Titrate a 70 mL water blank containing 1-4 drops of phenolphthalein to a faint pink color using the sodium
hydroxide solution prepared under 6.137.13. Repeat this procedure and average the results. For both the water blank and the KHP
titration replicates should agree to 0.05 mL titrant. For each KHP trial, independently calculate the normality for the sodium
hydroxide solution according to the following equation:
mg KHP/204.23
Normality of NaOH 5 (1)
~ mL NaOH 2 avg. mL blank!
Values for the two KHP trials should agree within 6 0.5 percent. 0.5 %. If they do not, repeat the titrations or identify the
cause for the excessive discrepancy, or both.
8.2 Sulfuric Acid Standardization—The following provides an example procedure for standardization; other quantities of reagents,
as convenient, can be used. Titrate the sulfuric acid solution prepared under 6.147.14 against the sodium hydroxide standardized
in 7.18.1 using 2-4 drops of phenolphthalein as the indicator. Repeat and average the result for the normality of the sulfuric acid.
Values for the two trials should agree within 6 0.5 percent. 6 0.5 %. If they do not, repeat the titrations or identify the cause for
the excessive discrepancy, or both.
8.3 Barium Chloride Solution Standardization— – Titrate the barium chloride solution against the previously standardized sulfuric
acid solution (see 7.28.2). This can be conveniently accomplished by transferring 10.0 mL sulfuric acid to a flask where 40 mL
iso-propanol and 2-4 drops thorin indicator are added. This is titrated to a pink end point using the barium chloride solution. Repeat
the titration and average the results. The replicate titrations should agree within 0.5 percent. 0.5 %. If they do not, repeat the
titrations or identify the cause for the excessive discrepancy, or both. Using this same procedure, perform duplicate blank titrations
using water in place of sulfuric acid solution. The replicate titrations should agree within 0.5 percent. 0.5 %. Calculate the
normality of the barium chloride solution according to the following equation:
10.0 mL x N Sulfuric Acid
Normality of Barium Chloride 5 (2)
~avg. mL 2 avg. blank!
8.4 An auto titration can be used to determine the concentration of both sodium hydroxide and sulfuric acid.
9. Preparation of Apparatus
9.1 Place 300 to 400 mL 300 mL to 400 mL of NaOH solution in the first scrubber (Fig. 4) and the same amount of H O -H SO
2 2 2 4
solution (300 mL of water, 30 mL of H SO , and 30 mL of H O (30 % w/w)) in the second scrubber. Replace these solutions
2 4 2 2
whenever the volume becomes less than two thirds of the original.
9.2 When the apparatus is first assembled, adjust the valve between the vacuum manifold and the spray trap so that approximately
D1072 − 23
3 L of air per minute will be drawn through the absorber when the chimney outlet is open to the atmosphere, the absorber is charged
with 30 6 2 mL 30 mL 6 2 mL of water, and the pressure in the vacuum manifold is maintained at approximately 40 cm of water
below atmospheric. When all adjustments have been made, remove the water from the absorbers.
9.3 With the burner control valve closed, the valve to the vacuum regulator fully open, and the pressure in the vacuum manifold
adjusted to approximately 40 cm of water below atmospheric, turn on the purified air. Adjust the chimney manifold control valve
so that, at the required flow through the absorber, only a small stream of air escapes at the pressure-relief valve, a small stream
of air enters at the vacuum regulator, and the pressure in the chimney manifold is 1 to 2 cm 1 cm to 2 cm of water. Minor
adjustment of the vacuum regulator and vacuum control valve may be necessary to achieve this condition.
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...