ASTM D2622-21
(Test Method)Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive X-ray Fluorescence Spectrometry
Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive X-ray Fluorescence Spectrometry
SIGNIFICANCE AND USE
5.1 This test method provides rapid and precise measurement of total sulfur in petroleum and petroleum products with a minimum of sample preparation. A typical analysis time is 1 min to 2 min per sample.
5.2 The quality of many petroleum products is related to the amount of sulfur present. Knowledge of sulfur concentration is necessary for processing purposes. There are also regulations promulgated in federal, state, and local agencies that restrict the amount of sulfur present in some fuels.
5.3 This test method provides a means of determining whether the sulfur content of petroleum or a petroleum product meets specification or regulatory limits.
5.4 When this test method is applied to petroleum materials with matrices significantly different from the white oil calibration materials specified in this test method, the cautions and recommendations in Section 6 should be observed when interpreting results.
Note 2: The equipment specified for Test Method D2622 tends to be more expensive than that required for alternative test methods, such as Test Method D4294. Consult the Index to ASTM Standards for alternative test methods.
SCOPE
1.1 This test method covers the determination of total sulfur in petroleum and petroleum products that are single-phase and either liquid at ambient conditions, liquefiable with moderate heat, or soluble in hydrocarbon solvents. These materials can include diesel fuel, jet fuel, kerosene, other distillate oil, naphtha, residual oil, lubricating base oil, hydraulic oil, crude oil, unleaded gasoline, gasoline-ethanol blends, and biodiesel.
1.2 The range of this test method is between the PLOQ value (calculated by procedures consistent with Practice D6259) of 3 mg/kg total sulfur and the highest level sample in the round robin, 4.6 % by weight total sulfur.
Note 1: Instrumentation covered by this test method can vary in sensitivity. The applicability of the test method at sulfur concentrations below 3 mg/kg may be determined on an individual basis for WDXRF instruments capable of measuring lower levels, but precision in this test method does not apply.
1.2.1 The values of the limit of quantitation (LOQ) and method precision for a specific laboratory’s instrument depends on instrument source power (low or high power), sample type, and the practices established by the laboratory to perform the method.
1.3 Samples containing more than 4.6 % by mass sulfur should be diluted to bring the sulfur concentration of the diluted material within the scope of this test method. Samples that are diluted can have higher errors than indicated in Section 15 than non-diluted samples.
1.4 Volatile samples (such as high vapor pressure gasolines or light hydrocarbons) may not meet the stated precision because of selective loss of light materials during the analysis.
1.5 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix differences are accounted for (see 13.2). Matrix mismatch can be caused by C/H ratio differences between samples and standards or by the presence of other interfering heteroatoms or species (see Table 1).
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2021
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.03 - Elemental Analysis
Relations
- Refers
ASTM D4175-23a - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 15-Dec-2023
- Effective Date
- 01-Dec-2023
- Refers
ASTM D4175-23e1 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jun-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Jan-2016
- Effective Date
- 01-Dec-2014
- Effective Date
- 01-Oct-2013
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
- Effective Date
- 01-Jul-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Nov-2009
Overview
ASTM D2622-21: Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive X-ray Fluorescence Spectrometry (WDXRF) provides a reliable, rapid, and precise methodology for determining total sulfur content in various petroleum products. Developed by ASTM International, this standard employs Wavelength Dispersive X-ray Fluorescence (WDXRF) spectrometry, which offers minimal sample preparation and short analysis times (typically 1-2 minutes per sample). Effective sulfur analysis is critical for product quality and compliance with federal, state, and local regulatory sulfur limits.
Key Topics
Scope of Application: ASTM D2622-21 applies to single-phase petroleum products that are liquid at room temperature, liquefiable by moderate heating, or soluble in hydrocarbon solvents. This includes diesel, jet fuel, kerosene, naphtha, lubricating oils, crude oil, gasoline, gasoline-ethanol blends, and biodiesel.
Measurement Range: The method is validated for sulfur concentrations ranging from 3 mg/kg up to 4.6% by mass. For levels above 4.6%, samples should be diluted.
Precision and Rapid Results: The test method is known for high precision and repeatability, providing rapid results essential for refining, quality assurance, and regulatory compliance.
Sample Matrix Considerations: Accurate results depend on proper matrix matching between samples and calibration standards. Significant differences, such as the carbon/hydrogen ratio, or the presence of interfering species (like phosphorus, lead, or oxygenates) may require specific corrections or dilution.
Instrument Requirements: Analysis is performed on WDXRF spectrometers, which need to be operated and maintained according to the manufacturer’s specifications for optimal performance and safety. Strict procedures are stipulated for calibration, drift correction, and quality control.
Interferences: The standard gives action levels for matrix elements such as phosphorus, lead, zinc, and calcium to minimize interference in sulfur measurement.
Regulatory and Specification Compliance: The test method is valuable for both internal process control and for demonstrating compliance with ever-tightening regulatory sulfur limits in fuels.
Applications
- Petroleum Refinery Operations: Enables process optimization and quality assurance by quickly measuring sulfur content at different production stages.
- Product Quality Control: Ensures that fuels and oils meet required specifications regarding sulfur content, which is crucial for engine performance, emissions, and overall product quality.
- Regulatory Compliance: Used to verify conformity with domestic and international environmental regulations that restrict sulfur emissions in fuel products.
- Blending Control: Assesses sulfur levels in blending components such as biodiesel, ethanol blends, and additives to adhere to required limits.
- Research and Development: Supports product innovation and the development of low-sulfur fuels.
- Trade and Customs: Provides a recognized analytical standard for sulfur content in the global trade of petroleum and related products.
Related Standards
For alternative methods or supporting procedures, refer to the following ASTM standards:
- ASTM D4294: Test Method for Sulfur in Petroleum Products by Energy Dispersive X-ray Fluorescence Spectrometry (an often less expensive alternative for certain matrices).
- ASTM D4057 and D4177: Standards for manual and automatic sampling of petroleum products.
- ASTM D4927: Elemental Analysis of Lubricant and Additive Components (for sulfur >100 mg/kg).
- ASTM D6259: Practice for determining the pooled limit of quantitation (PLOQ).
- ASTM D7343: Practices for X-ray Fluorescence Spectrometry method optimization and calibration.
Practical Value
By standardizing sulfur determination through WDXRF, ASTM D2622-21 delivers a crucial tool for the petroleum industry to maintain product consistency, protect engine systems, and support regulatory compliance. Its rapid analysis and broad applicability across fuel and oil types make it an essential resource for refineries, testing laboratories, regulatory agencies, and bulk fuel distributors. Adherence to this method enables the industry to reliably monitor sulfur content, improve environmental outcomes, and meet marketplace demands for cleaner fuels.
Buy Documents
ASTM D2622-21 - Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive X-ray Fluorescence Spectrometry
REDLINE ASTM D2622-21 - Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive X-ray Fluorescence Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D2622-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Sulfur in Petroleum Products by Wavelength Dispersive X-ray Fluorescence Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 This test method provides rapid and precise measurement of total sulfur in petroleum and petroleum products with a minimum of sample preparation. A typical analysis time is 1 min to 2 min per sample. 5.2 The quality of many petroleum products is related to the amount of sulfur present. Knowledge of sulfur concentration is necessary for processing purposes. There are also regulations promulgated in federal, state, and local agencies that restrict the amount of sulfur present in some fuels. 5.3 This test method provides a means of determining whether the sulfur content of petroleum or a petroleum product meets specification or regulatory limits. 5.4 When this test method is applied to petroleum materials with matrices significantly different from the white oil calibration materials specified in this test method, the cautions and recommendations in Section 6 should be observed when interpreting results. Note 2: The equipment specified for Test Method D2622 tends to be more expensive than that required for alternative test methods, such as Test Method D4294. Consult the Index to ASTM Standards for alternative test methods. SCOPE 1.1 This test method covers the determination of total sulfur in petroleum and petroleum products that are single-phase and either liquid at ambient conditions, liquefiable with moderate heat, or soluble in hydrocarbon solvents. These materials can include diesel fuel, jet fuel, kerosene, other distillate oil, naphtha, residual oil, lubricating base oil, hydraulic oil, crude oil, unleaded gasoline, gasoline-ethanol blends, and biodiesel. 1.2 The range of this test method is between the PLOQ value (calculated by procedures consistent with Practice D6259) of 3 mg/kg total sulfur and the highest level sample in the round robin, 4.6 % by weight total sulfur. Note 1: Instrumentation covered by this test method can vary in sensitivity. The applicability of the test method at sulfur concentrations below 3 mg/kg may be determined on an individual basis for WDXRF instruments capable of measuring lower levels, but precision in this test method does not apply. 1.2.1 The values of the limit of quantitation (LOQ) and method precision for a specific laboratory’s instrument depends on instrument source power (low or high power), sample type, and the practices established by the laboratory to perform the method. 1.3 Samples containing more than 4.6 % by mass sulfur should be diluted to bring the sulfur concentration of the diluted material within the scope of this test method. Samples that are diluted can have higher errors than indicated in Section 15 than non-diluted samples. 1.4 Volatile samples (such as high vapor pressure gasolines or light hydrocarbons) may not meet the stated precision because of selective loss of light materials during the analysis. 1.5 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix differences are accounted for (see 13.2). Matrix mismatch can be caused by C/H ratio differences between samples and standards or by the presence of other interfering heteroatoms or species (see Table 1). 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method provides rapid and precise measurement of total sulfur in petroleum and petroleum products with a minimum of sample preparation. A typical analysis time is 1 min to 2 min per sample. 5.2 The quality of many petroleum products is related to the amount of sulfur present. Knowledge of sulfur concentration is necessary for processing purposes. There are also regulations promulgated in federal, state, and local agencies that restrict the amount of sulfur present in some fuels. 5.3 This test method provides a means of determining whether the sulfur content of petroleum or a petroleum product meets specification or regulatory limits. 5.4 When this test method is applied to petroleum materials with matrices significantly different from the white oil calibration materials specified in this test method, the cautions and recommendations in Section 6 should be observed when interpreting results. Note 2: The equipment specified for Test Method D2622 tends to be more expensive than that required for alternative test methods, such as Test Method D4294. Consult the Index to ASTM Standards for alternative test methods. SCOPE 1.1 This test method covers the determination of total sulfur in petroleum and petroleum products that are single-phase and either liquid at ambient conditions, liquefiable with moderate heat, or soluble in hydrocarbon solvents. These materials can include diesel fuel, jet fuel, kerosene, other distillate oil, naphtha, residual oil, lubricating base oil, hydraulic oil, crude oil, unleaded gasoline, gasoline-ethanol blends, and biodiesel. 1.2 The range of this test method is between the PLOQ value (calculated by procedures consistent with Practice D6259) of 3 mg/kg total sulfur and the highest level sample in the round robin, 4.6 % by weight total sulfur. Note 1: Instrumentation covered by this test method can vary in sensitivity. The applicability of the test method at sulfur concentrations below 3 mg/kg may be determined on an individual basis for WDXRF instruments capable of measuring lower levels, but precision in this test method does not apply. 1.2.1 The values of the limit of quantitation (LOQ) and method precision for a specific laboratory’s instrument depends on instrument source power (low or high power), sample type, and the practices established by the laboratory to perform the method. 1.3 Samples containing more than 4.6 % by mass sulfur should be diluted to bring the sulfur concentration of the diluted material within the scope of this test method. Samples that are diluted can have higher errors than indicated in Section 15 than non-diluted samples. 1.4 Volatile samples (such as high vapor pressure gasolines or light hydrocarbons) may not meet the stated precision because of selective loss of light materials during the analysis. 1.5 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix differences are accounted for (see 13.2). Matrix mismatch can be caused by C/H ratio differences between samples and standards or by the presence of other interfering heteroatoms or species (see Table 1). 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D2622-21 is classified under the following ICS (International Classification for Standards) categories: 75.080 - Petroleum products in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D2622-21 has the following relationships with other standards: It is inter standard links to ASTM D4175-23a, ASTM D6299-23a, ASTM D4175-23e1, ASTM D6299-17b, ASTM D6299-17a, ASTM D7343-12(2017), ASTM D6299-17, ASTM D4294-16, ASTM D4927-14, ASTM D6299-13e1, ASTM D4057-06(2011), ASTM D6259-98(2010), ASTM D6299-10e2, ASTM D6299-10, ASTM D6299-09. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D2622-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D2622 − 21
Standard Test Method for
Sulfur in Petroleum Products by Wavelength Dispersive
X-ray Fluorescence Spectrometry
This standard is issued under the fixed designation D2622; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope* match can be caused by C/H ratio differences between samples
and standards or by the presence of other interfering heteroa-
1.1 This test method covers the determination of total sulfur
toms or species (see Table 1).
in petroleum and petroleum products that are single-phase and
either liquid at ambient conditions, liquefiable with moderate 1.6 The values stated in SI units are to be regarded as
heat, or soluble in hydrocarbon solvents. These materials can standard. No other units of measurement are included in this
include diesel fuel, jet fuel, kerosene, other distillate oil, standard.
naphtha, residual oil, lubricating base oil, hydraulic oil, crude
1.7 This standard does not purport to address all of the
oil, unleaded gasoline, gasoline-ethanol blends, and biodiesel.
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
1.2 The range of this test method is between the PLOQ
priate safety, health, and environmental practices and deter-
value (calculated by procedures consistent with Practice
mine the applicability of regulatory limitations prior to use.
D6259) of 3 mg/kg total sulfur and the highest level sample in
1.8 This international standard was developed in accor-
the round robin, 4.6 % by weight total sulfur.
dance with internationally recognized principles on standard-
NOTE 1—Instrumentation covered by this test method can vary in
ization established in the Decision on Principles for the
sensitivity. The applicability of the test method at sulfur concentrations
Development of International Standards, Guides and Recom-
below 3 mg⁄kg may be determined on an individual basis for WDXRF
instruments capable of measuring lower levels, but precision in this test mendations issued by the World Trade Organization Technical
method does not apply.
Barriers to Trade (TBT) Committee.
1.2.1 The values of the limit of quantitation (LOQ) and
2. Referenced Documents
methodprecisionforaspecificlaboratory’sinstrumentdepends
on instrument source power (low or high power), sample type,
2.1 ASTM Standards:
and the practices established by the laboratory to perform the
D4057 Practice for Manual Sampling of Petroleum and
method.
Petroleum Products
1.3 Samples containing more than 4.6 % by mass sulfur D4175 Terminology Relating to Petroleum Products, Liquid
Fuels, and Lubricants
should be diluted to bring the sulfur concentration of the
diluted material within the scope of this test method. Samples D4177 Practice for Automatic Sampling of Petroleum and
Petroleum Products
thataredilutedcanhavehighererrorsthanindicatedinSection
D4294 Test Method for Sulfur in Petroleum and Petroleum
15 than non-diluted samples.
Products by Energy Dispersive X-ray Fluorescence Spec-
1.4 Volatile samples (such as high vapor pressure gasolines
trometry
or light hydrocarbons) may not meet the stated precision
D4927 Test Methods for Elemental Analysis of Lubricant
because of selective loss of light materials during the analysis.
and Additive Components—Barium, Calcium,
1.5 Afundamental assumption in this test method is that the
Phosphorus, Sulfur, and Zinc by Wavelength-Dispersive
standard and sample matrices are well matched, or that the
X-Ray Fluorescence Spectroscopy
matrix differences are accounted for (see 13.2). Matrix mis-
D6259 Practice for Determination of a Pooled Limit of
Quantitation for a Test Method
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.03 on Elemental Analysis. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2021. Published January 2022. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1967. Last previous edition approved in 2016 as D2622 – 16. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D2622-21. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D2622 − 21
TABLE 1 Concentrations of Interfering Species Test Method D4294. Consult the Index toASTM Standards for alternative
test methods.
Species Mass % Tolerated
Phosphorus 0.3
6. Interferences
Zinc 0.6
Barium 0.8
6.1 When the elemental composition (excluding sulfur) of
Lead 0.9
Calcium 1
samples differs significantly from the standards, errors in the
Chlorine 3
sulfur determination can result. For example, differences in the
Oxygen 2.8
carbon-hydrogen ratio of sample and calibration standards
FAME (see Note 16) 25
Ethanol (see Note 16) 8.6
introduce errors in the determination. Some other interferences
Methanol (see Note 16) 6
and action levels are listed in Table 1. If a sample is known
from its history or another analysis to contain any of the
species listed in Table 1 at or above the values listed there, that
sample should be diluted with blank sulfur solvent to reduce
D6299 Practice for Applying Statistical Quality Assurance
the interferent concentration below the value to mitigate the
and Control Charting Techniques to Evaluate Analytical
effect of this interference.
Measurement System Performance
NOTE 3—The concentrations of the first seven substances in Table 1
D7343 Practice for Optimization, Sample Handling,
were determined by the calculation of the sum of the mass absorption
Calibration, and Validation of X-ray Fluorescence Spec-
coefficients times mass fraction of each element present. This calculation
trometry Methods for Elemental Analysis of Petroleum was made for dilutions of representative samples containing approxi-
mately 3 % of interfering substances and 0.5 % sulfur. Refer to Note 16
Products and Lubricants
for additional information regarding FAME, ethanol, and methanol.
E29 Practice for Using Significant Digits in Test Data to
6.2 Fuels containing large quantities of FAME, ethanol, or
Determine Conformance with Specifications
methanol (see Table 1) have a high oxygen content leading to
3. Terminology
significant absorption of sulfur Kα radiation and low sulfur
results. Such fuels can, however, be analyzed using this test
3.1 For definitions of terms used in this test method refer to
method provided either that correction factors are applied to
Terminology D4175.
the results (when calibrating with white oils) or that the
calibration standards are prepared to match the matrix of the
4. Summary of Test Method
sample. See 12.5.
4.1 The sample is placed in the X-ray beam, and the peak
intensity of the sulfur K-L (Kα) line at 0.5373 nm is 6.3 In general, petroleum materials with compositions that
2,3
measured. The background intensity, measured at a recom- vary from white oils as specified in 10.1 can be analyzed with
mended wavelength of 0.5190 nm (0.5437 nm for a Rh target standards made from base materials that are of the same or
tube) is subtracted from the peak intensity. The resultant net similar composition. Thus a gasoline may be simulated by
mixing isooctane and toluene in a ratio that approximates the
counting rate is then compared to a previously prepared
calibration curve or equation to obtain the concentration of expected aromatic content of the samples to be analyzed.
Standards made from this simulated gasoline can produce
sulfur in milligrams per kilogram (mg/kg) or mass percent (see
Section 13). results that are more accurate than results obtained using white
oil standards.
5. Significance and Use
6.4 TestMethodD4927istherecommendedtestmethodfor
5.1 This test method provides rapid and precise measure-
the determination of sulfur >100 mg⁄kg in lubricating oils and
ment of total sulfur in petroleum and petroleum products with
lubricating oil additives because method D4927 implements
a minimum of sample preparation. A typical analysis time is
inter-element correction factors. Test Method D2622 is not
1 min to 2 min per sample.
suitable because it does not encompass the measurement of the
additional elements present in lubricating oils and their addi-
5.2 The quality of many petroleum products is related to the
tives making matrix correction impossible.
amount of sulfur present. Knowledge of sulfur concentration is
necessary for processing purposes. There are also regulations
7. Apparatus
promulgated in federal, state, and local agencies that restrict
the amount of sulfur present in some fuels.
7.1 Wavelength Dispersive X-Ray Fluorescence Spectrom-
eter (WDXRF),equippedforX-raydetectioninthewavelength
5.3 This test method provides a means of determining
whether the sulfur content of petroleum or a petroleum product range from about 0.52 nm to about 0.55 nm (specifically at
0.537 nm). For optimum sensitivity to sulfur, the instrument
meets specification or regulatory limits.
should be equipped with the following items:
5.4 When this test method is applied to petroleum materials
7.1.1 Optical Path, vendor specified, helium preferred, am-
with matrices significantly different from the white oil calibra-
bient air or nitrogen are inferior.
tion materials specified in this test method, the cautions and
7.1.2 Pulse-Height Analyzer, or other means of energy
recommendations in Section 6 should be observed when
discrimination.
interpreting results.
7.1.3 Detector, for the detection of X-rays with wavelengths
NOTE 2—The equipment specified for Test Method D2622 tends to be
more expensive than that required for alternative test methods, such as in the range of interest (from about 0.52 nm to about 0.55 nm).
D2622 − 21
7.1.4 Analyzing Crystal, suitable for the dispersion of sulfur
K-L (Kα) and background X-rays within the angular range
2,3
of the spectrometer employed. Germanium or pentaerythritol
(PET) are generally found to be acceptable. Other crystals may
be used, consult with the instrument vendor.
7.1.5 X-ray Tube, capable of exciting sulfur K-L (Kα)
2,3
radiation. Tubes with anodes of rhodium, chromium, and
scandium are most popular although other anodes can be used.
NOTE4—Exposuretoexcessivequantitiesofhighenergyradiationsuch
as those produced by X-ray spectrometers is injurious to health. The
operator needs to take appropriate actions to avoid exposing any part of
their body, not only to primary X-rays, but also to secondary or scattered
radiation that might be present. The X-ray spectrometer should be
operated in accordance with the regulations governing the use of ionizing
radiation.
FIG. 1 Relative Sulfur Sensitivity versus C/H Ratio
7.2 Analytical Balance, capable of weighing to the nearest
0.1 mg and up to 100 g.
8. Reagents
time of analysis (see 11.1). These counting rates are used to
8.1 Purity of Reagents—Reagent grade chemicals shall be
calculate a drift correction factor (see 12.1).
used in all tests. Unless otherwise indicated, it is intended that
8.3.1 Drift correction is usually implemented automatically
all reagents conform to the specifications of the Committee on
in software, although the calculation can readily be done
Analytical Reagents of the American Chemical Society where
manually. For X-ray instruments that are highly stable, the
such specifications are available. Other grades may be used,
magnitude of the drift correction factor may not differ signifi-
provided it is first ascertained that the reagent is of sufficiently
cantly from unity.
high purity to permit its use without lessening the accuracy of
8.4 Polysulfide Oil, generally nonyl polysulfides containing
the determination.
a known percentage of sulfur diluted in a hydrocarbon matrix.
8.2 Di-n-Butyl Sulfide (DBS), a high-purity material with a
(Warning—May cause allergic skin reactions. Warning—
certified analysis for sulfur content. Use the certified sulfur
Solutionspreparedinavolatileorun-stabilizedmatrixmaynot
content and the material purity when calculating the exact
be stable several months after preparation.)
concentrations of the calibration standards (see 10.1).
NOTE 6—Polysulfide oils are high molecular weight oils that contain
(Warning—Di-n-butyl sulfide is flammable and toxic.
high concentrations of sulfur, as high as 50 % by weight. They exhibit
Warning—Solutions prepared in a volatile or un-stabilized physical properties such as low viscosity, low volatility, and durable shelf
life while being completely miscible in white oil.The sulfur content of the
matrix may not be stable several months after preparation.)
polysulfide oil concentrate is determined via mass dilution in sulfur-free
NOTE 5—It is essential to know the concentration of sulfur in the
white oil followed by a direct comparison analysis against NIST (or other
di-n-butyl sulfide, not only the purity, since impurities may also be sulfur
primary standards body) reference materials.
containing compounds. The sulfur content may be determined via mass
dilution in sulfur-free white oil followed by a direct comparison analysis
8.5 Mineral Oil, White (MOW), ACS Reagent Grade con-
against NIST (or other primary standards body) reference materials.
taining less than 2 mg/kg sulfur or other suitable base material
8.3 Drift Correction Monitor(s) (Optional), Several differ-
containing less than 2 mg/kg sulfur. When low level
ent materials have been found to be suitable for use as drift
(<200 mg⁄kg) measurements are anticipated, then the sulfur
correction monitors. Appropriate drift monitor samples should
content, if any, of the base material needs to be included in the
be permanent materials that are stable with respect to repeated
calculation of calibration standard concentration (see 10.1).
exposure to X-rays. Stable liquids like polysulfide oils, glass,
When the sulfur content of the solvent or reagent is not
or metallic specimens are recommended. Liquids, pressed
certified, verify the absence of sulfur. Use the purest grades for
powders, and solid materials that degrade with repeated expo-
the preparation of calibration standards. It is also important to
sure to X-rays should not be used. Examples of sulfur
measure the C/H ratio (see Section 13 and Fig. 1).
containing materials that have been found to be suitable
8.6 X-ray Transparent Film—Any film that resists attack by
includearenewableliquidpetroleummaterial,ametalalloy,or
the sample, is free of sulfur, and is sufficiently X-ray transpar-
a fused glass disk.The monitor’s counting rate, in combination
ent can be used. Film types can include polyester,
with count time, shall be sufficient to give a relative counting
polypropylene, polycarbonate, and polyimide. However,
erroroflessthan1 %.Thecountingrateforthemonitorsample
samples of high aromatic content can dissolve polypropylene
is determined during calibration (see 10.4) and again at the
and polycarbonate films.
8.7 Helium Gas, minimum purity 99.9 %.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
8.8 Counting Gas, for instruments equipped with flow
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
proportional counters.The purity of the counting gas should be
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
in agreement with the specification provided by the instrument
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. manufacturer.
D2622 − 21
TABLE 2 Suggested Sulfur Standard Calibration Ranges
8.9 Sample Cells, compatible with the sample and the
geometry requirements of the spectrometer. Disposable cells 0–1000 0.10–1.00 1.0–5.0
mg/kg % mass % mass
are preferred over reusable ones for ultra low (<50 mg/kg)
A,B
0.0 0.100 1.0
sulfur levels.
B
5 0.250 2.0
B
10 0.500 3.0
8.10 Calibration Check Samples, portions of one or more
B
100 1.000 4.0
liquid petroleum or product standards of known or certified
250 5.0
sulfur content (including polysulfide oils, di-n-butyl sulfide,
thiophenes, etc.) and not used in the generation of the calibra-
tion curve. The check samples shall be used to determine the
A
Base material.
precision and accuracy of the initial calibration (see 10.5).
B
Analyze these standards in duplicate and use both values or the average value
in the calibration.
8.11 Quality Control Samples, stable petroleum or product
samples or solids representative of the samples of interest that
are run on a regular basis to verify that the system is in
standards are listed in Table 2 for the sulfur concentration
statistical control (see Section 14).
ranges of interest. Take into account any sulfur in the base
NOTE 7—Verification of system control through the use of QC samples
materialwhencalculatingtheconcentrationofstandardsbelow
and control charting is highly recommended. It is recognized that QC
0.02 % by mass (200 mg/kg), as shown in Eq 1:
procedures are the province of the individual laboratory.
NOTE 8—Suitable QC samples can often be prepared by combining
S 5 @~DBS 3 S !1~WO 3 S !#/~DBS1WO! (1)
DBS WO
retains of typical samples if they are stable. For monitors, solid materials
are recommended. QC samples must be stable over long periods.
where:
S = mass percent sulfur of the prepared standards,
9. Sampling and Specimen Preparation
DBS = actual mass of DBS, g,
9.1 Samples shall be taken in accordance with the instruc-
S = mass percent sulfur in DBS, typically 21.91 %,
DBS
tions in Practices D4057 or D4177 when applicable.
WO = actual mass of white oil, g, and
S = mass percent sulfur in the white oil.
9.2 Whenreusablesamplecellsareused,cleananddrycells WO
NOTE 9—If desired, additional standards can prepared and analyzed
before each use. Disposable sample cells shall not be reused.
with concentrations between those listed in Table 2, see 10.1.1.
For each sample, an unused piece of X-ray film is required for
10.1.1 Calibration standards can also be prepared by careful
the sample cell. Avoid touching the inside of the sample cell,
mixing of certified reference materials (CRM) of the same
the portion of the window film in the cell, or the instrument
matrix, so long as the sulfur values of the resulting blends and
window (if the instrument is so equipped) that is exposed to
their uncertainties are characterized by the certifying body.
X-rays. Oil from fingerprints can affect the reading when
10.1.2 Alternatively, standards may be prepared by mass
analyzing for low levels of sulfur. Wrinkles in the film will
serial dilution of polysulfide oils (Note 6) with sulfur-free
affect the intensity of the sulfur X-rays transmitted. Therefore,
white oil. A freshly prepared calibration curve should be
it is essential that the film be taut and clean to ensure reliable
verified using CRMs traceable to a national measurement
results. The analyzer may need recalibration if the type or
institution that has demonstrated proficiency for measuring
thickness of the window film is changed.After the sample cell
sulfur in the matrix of interest.
is filled, create a small vent hole except when the cell is of the
sealed type.
NOTE 10—Commercially available standards may be used provided
their sulfur concentrations are accurately known and they approximate the
9.3 Employ adequate storage, mixing, and sampling proce-
nominal concentrations listed in Table 2.
dures. Refrigerate gasolines or other similar volatile materials
10.2 Establish calibration curve data by carefully determin-
toretainsampleintegrityinstorage,butallowthemtoreturnto
ing the net intensity of the emitted sulfur radiation from each
room temperature before testing. Expose these materials to
ofthestandardsbytheproceduresdescribedinSections 11and
ambient conditions only as long as necessary to obtain a
12.
sample for analysis.
10.2.1 Analyze standards containing 100 mg/kg total sulfur
9.4 Impurities or thickness variations, which may affect the
or less in duplicate. Either both of the individual values or the
measurement of low levels of sulfur, have been found in
average value of these measurements may be used in the
polyester films and may vary from lot to lot. Therefore, the
calibration.All samples in this sulfur concentration range must
calibration should be checked after starting each new lot of
also be analyzed in duplicate, as in 11.12, and reported as in
film.
13.1.1.
9.5 See Practice D7343 for more detailed sample handling
10.3 Construct a calibration model by using the software
and preparation information.
and algorithms supplied by the equipment vendor. The calibra-
tion model typically takes one of the following forms (refer to
10. Calibration
the equipment vendor’s software documentation to determine
10.1 Prepare calibration standards by careful mass dilution
the exact form):
of the certified di-n-butyl sulfide with a sulfur-free white oil or
othersuitablebasematerial(see8.5).Theconcentrationsofthe
Kelly, W. R., MacDonald, B. S., and Leigh, S. D., “A Method for the
unknown samples must lie within the calibration range that is
Preparation of NIST Traceable Fossil Fuel Standards with Concentrations Interme-
used.Approximaterecommendednominalsulfurconcentration diate to SRM Values,” Journal of ASTM International, Vol 4, No. 2, 2007.
D2622 − 21
C 5 a1bI linear calibration (2) are set correctly. Before performing a calibration of the
S
goniometer angles carry out pulse height discriminator settings
C 5 a1bI 11 α C correction for matrix effects (3)
~ !~ !
S ( ij j
(PHD’s) for each element and background to be used. The
C 5 a1bI~11 α C ! alternative correction for matrix effects
S ( ij j angle should be checked first, then the PHD, then the angle
(4) re-checked if the PHD settings are changed significantly. A
2 very poor angle calibration may lead to bad PHD. The only
C 5 a1bI1cI 2nd order polynomial (5)
S
reasonable alternate line is the sulfur K-M (Kβ) with
2,3
where:
significantly less than 10 % of the sensitivity of the K-L
2,3
C = mass fraction of sulfur.The concentration units depend
(Kα); this will only be practical for samples with high sulfur
S
on the fitted calibration constants a, b and c,
concentrations.
I = measured net intensity for the sulfur radiation,
11.3 Account for observations of known instrument inter-
a = fitted calibration line offset (intercept),
ferences.These include crystal fluorescence, tube line overlaps
b = fitted calibration line slope,
and any element spectral contamination from the materials
c = fitted calibration line 2nd order polynomial,
within the instrument construction. Lead is a particularly bad
a = correctionfactorfortheeffectofaninterferingelement
ij
interference for sulfur measurement. A number of these inter-
(j) on sulfur (i). The interfering element can be sulfur
ferences can be avoided by careful selection of window
when “matrix eliminated alphas” or empirical correc-
settings during PHD set-up and for element interference the
tions are used, and
selection of an alternative line or minimizing overlap using
C = concentration of the interfering element (j).
j
NOTE 11—The a factors can be determined empirically through higher resolution collimators and crystal selection.
ij
multiple regression, or theoretically through fundamental parameters.
11.4 When the factor F' is used in Eq 8, regularly analyze a
Equipment vendors typically have provisions in their software for
blank sample to determine the factor F'. On a sulfur free
calculating theoretical a’s.
sample, such as the base material, determine the counting rate
10.3.1 Fit the calibration data over several ranges if
at the appropriate sulfur peak and background angles.
necessary, depending on the sulfur concentrations to be cov-
ered. For example (Table 2): 0 % to 0.10 % by mass sulfur,
11.5 Place the sample in an appropriate cell using tech-
0.10 % to 1.0 % by mass sulfur, and 1.0 % to 5.0 % by mass
niques consistent with good practice for the particular instru-
sulfur.
ment being used. Although incident radiation may penetrate a
significantdistanceintothesample,sulfurradiationwillescape
NOTE 12—Calibration curves are typically linear to about 0.10 % by
fromonlyasmalldistanceintothesample,andscatterfromthe
mass sulfur. The analyst should choose a linear calibration model when
fitting within this range. One of the other calibration models (correction sample cell and the sample can vary. Ensure that the sample
for matrix effects or a 2nd order polynominal) should be chosen when
cell is filled above a minimum depth, beyond which additional
fitting to higher concentrations of sulfur.
sample does not significantly affect the counting rate.
10.4 When using drift correction monitors, determine the Generally, fill the sample cell to a minimum of two-thirds of
intensity of the drift correction monitor sample(s) during the
the cell’s capacity. Provide a small vent hole in the sample cell
calibrationprocedure.Thevaluedeterminedcorrespondstothe unless using a sealed cell.
factor A in Eq 7 in 12.1.
11.6 Place the sample in the X-ray beam and allow the
10.5 Immediately after completing the calibration, deter-
X-ray optical path to come to equilibrium.
mine the sulfur concentration of one or more of the calibration
11.7 Determine the intensity of the sulfur K-L (Kα)
2,3
check samples (8.10). The differences between two measured
radiation at 0.5373 nm by making counting rate measurements
values shall be within the repeatability of this test method (see
at the precise angular settings for this wavelength.
15.1.1). When this is not the case, the stability of the instru-
NOTE 13—It is suggested that a sufficient number of counts be taken to
ment and the repeatability of the sample preparation are
satisfy an expected coefficient of variation (% rsd) of 1.0 % or less when
suspectandcorrectivemeasuresshouldbetaken.Thedegreeof
practical. When sensitivity or concentration, or both, make it impractical
matrix mismatch between samples and standards should also to collect a sufficient number of counts to achieve a 1.0 % coefficient of
variation, accepted techniques, which will allow the greatest statistical
be considered when evaluating a calibration. Statistical quality
precision in the time allotted for each analysis, should be used.
control charts may be prepared for these materials to establish
if the method is in statistical control, as described in Section 11.8 The coefficient of va
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D2622 − 16 D2622 − 21
Standard Test Method for
Sulfur in Petroleum Products by Wavelength Dispersive
X-ray Fluorescence Spectrometry
This standard is issued under the fixed designation D2622; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope*
1.1 This test method covers the determination of total sulfur in petroleum and petroleum products that are single-phase and either
liquid at ambient conditions, liquefiable with moderate heat, or soluble in hydrocarbon solvents. These materials can include diesel
fuel, jet fuel, kerosene, other distillate oil, naphtha, residual oil, lubricating base oil, hydraulic oil, crude oil, unleaded gasoline,
gasoline-ethanol blends, and biodiesel.
1.2 The range of this test method is between the PLOQ value (calculated by procedures consistent with Practice D6259) of 3
mg/kg total sulfur and the highest level sample in the round robin, 4.6 weight % 4.6 % by weight total sulfur.
NOTE 1—Instrumentation covered by this test method can vary in sensitivity. The applicability of the test method at sulfur concentrations below 3 mg ⁄kg
may be determined on an individual basis for WDXRF instruments capable of measuring lower levels, but precision in this test method does not apply.
1.2.1 The values of the limit of quantitation (LOQ) and method precision for a specific laboratory’s instrument depends on
instrument source power (low or high power), sample type, and the practices established by the laboratory to perform the method.
1.3 Samples containing more than 4.6 mass % 4.6 % by mass sulfur should be diluted to bring the sulfur concentration of the
diluted material within the scope of this test method. Samples that are diluted can have higher errors than indicated in Section 1415
than non-diluted samples.
1.4 Volatile samples (such as high vapor pressure gasolines or light hydrocarbons) may not meet the stated precision because of
selective loss of light materials during the analysis.
1.5 A fundamental assumption in this test method is that the standard and sample matrices are well matched, or that the matrix
differences are accounted for (see 12.213.2). Matrix mismatch can be caused by C/H ratio differences between samples and
standards or by the presence of other interfering heteroatoms or species (see Table 1).
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.03 on Elemental Analysis.
Current edition approved Jan. 1, 2016Dec. 1, 2021. Published February 2016January 2022. Originally approved in 1967. Last previous edition approved in 20102016 as
D2622 – 10.D2622 – 16. DOI: 10.1520/D2622-16.10.1520/D2622-21.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D2622 − 21
TABLE 1 Concentrations of Interfering Species
Species Mass % Tolerated
Phosphorus 0.3
Zinc 0.6
Barium 0.8
Lead 0.9
Calcium 1
Chlorine 3
Oxygen 2.8
FAME (see Note 16) 25
Ethanol (see Note 16) 8.6
Methanol (see Note 16) 6
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4175 Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
D4294 Test Method for Sulfur in Petroleum and Petroleum Products by Energy Dispersive X-ray Fluorescence Spectrometry
D4927 Test Methods for Elemental Analysis of Lubricant and Additive Components—Barium, Calcium, Phosphorus, Sulfur,
and Zinc by Wavelength-Dispersive X-Ray Fluorescence Spectroscopy
D6259 Practice for Determination of a Pooled Limit of Quantitation for a Test Method
D6299 Practice for Applying Statistical Quality Assurance and Control Charting Techniques to Evaluate Analytical Measure-
ment System Performance
D7343 Practice for Optimization, Sample Handling, Calibration, and Validation of X-ray Fluorescence Spectrometry Methods
for Elemental Analysis of Petroleum Products and Lubricants
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
3. Terminology
3.1 For definitions of terms used in this test method refer to Terminology D4175.
4. Summary of Test Method
4.1 The sample is placed in the X-ray beam, and the peak intensity of the sulfur KαK-L (Kα) line at 0.5373 nm is measured.
2,3
The background intensity, measured at a recommended wavelength of 0.5190 nm (0.5437 nm for a Rh target tube) is subtracted
from the peak intensity. The resultant net counting rate is then compared to a previously prepared calibration curve or equation
to obtain the concentration of sulfur in milligrams per kilogram (mg/kg) or mass percent (see Section 1213).
5. Significance and Use
5.1 This test method provides rapid and precise measurement of total sulfur in petroleum and petroleum products with a minimum
of sample preparation. A typical analysis time is 1 min to 2 min per sample.
5.2 The quality of many petroleum products is related to the amount of sulfur present. Knowledge of sulfur concentration is
necessary for processing purposes. There are also regulations promulgated in federal, state, and local agencies that restrict the
amount of sulfur present in some fuels.
5.3 This test method provides a means of determining whether the sulfur content of petroleum or a petroleum product meets
specification or regulatory limits.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
D2622 − 21
5.4 When this test method is applied to petroleum materials with matrices significantly different from the white oil calibration
materials specified in this test method, the cautions and recommendations in Section 56 should be observed when interpreting
results.
NOTE 2—The equipment specified for Test Method D2622 tends to be more expensive than that required for alternative test methods, such as Test Method
D4294. Consult the Index to ASTM Standards for alternative test methods.
6. Interferences
6.1 When the elemental composition (excluding sulfur) of samples differs significantly from the standards, errors in the sulfur
determination can result. For example, differences in the carbon-hydrogen ratio of sample and calibration standards introduce
errors in the determination. Some other interferences and action levels are listed in Table 1. If a sample is known from its history
or another analysis to contain any of the species listed in Table 1 at or above the values listed there, that sample should be diluted
with blank sulfur solvent to reduce the interferent concentration below the value to mitigate the effect of this interference.
NOTE 3—The concentrations of the first seven substances in Table 1 were determined by the calculation of the sum of the mass absorption coefficients
times mass fraction of each element present. This calculation was made for dilutions of representative samples containing approximately 3 % of
interfering substances and 0.5 % sulfur. Refer to Note 16 for additional information regarding FAME, ethanol, and methanol.
6.2 Fuels containing large quantities of FAME, ethanol, or methanol (see Table 1) have a high oxygen content leading to
significant absorption of sulfur Kα radiation and low sulfur results. Such fuels can, however, be analyzed using this test method
provided either that correction factors are applied to the results (when calibrating with white oils) or that the calibration standards
are prepared to match the matrix of the sample. See 11.512.5.
6.3 In general, petroleum materials with compositions that vary from white oils as specified in 9.110.1 can be analyzed with
standards made from base materials that are of the same or similar composition. Thus a gasoline may be simulated by mixing
isooctane and toluene in a ratio that approximates the expected aromatic content of the samples to be analyzed. Standards made
from this simulated gasoline can produce results that are more accurate than results obtained using white oil standards.
6.4 Test Method D4927 is the recommended test method for the determination of sulfur >100 mg ⁄kg in lubricating oils and
lubricating oil additives because method D4927 implements inter-element correction factors. Test Method D2622 is not suitable
because it does not encompass the measurement of the additional elements present in lubricating oils and their additives making
matrix correction impossible.
7. Apparatus
7.1 Wavelength Dispersive X-Ray Fluorescence Spectrometer (WDXRF), equipped for X-ray detection in the wavelength range
from about 0.52 nm to about 0.55 nm (specifically at 0.537 nm). For optimum sensitivity to sulfur, the instrument should be
equipped with the following items:
7.1.1 Optical Path, vendor specified, helium preferred, ambient air or nitrogen are inferior.
7.1.2 Pulse-Height Analyzer, or other means of energy discrimination.
7.1.3 Detector, for the detection of X-rays with wavelengths in the range of interest (from about 0.52 nm to about 0.55 nm).
7.1.4 Analyzing Crystal, suitable for the dispersion of sulfur KαK-L (Kα) and background X-rays within the angular range of
2,3
the spectrometer employed. Germanium or pentaerythritol (PET) are generally found to be acceptable. Other crystals may be used,
consult with the instrument vendor.
7.1.5 X-ray Tube, capable of exciting sulfur KαK-L (Kα) radiation. Tubes with anodes of rhodium, chromium, and scandium
2,3
are most popular although other anodes can be used.
NOTE 4—Exposure to excessive quantities of high energy radiation such as those produced by X-ray spectrometers is injurious to health. The operator
needs to take appropriate actions to avoid exposing any part of their body, not only to primary X-rays, but also to secondary or scattered radiation that
might be present. The X-ray spectrometer should be operated in accordance with the regulations governing the use of ionizing radiation.
D2622 − 21
7.2 Analytical Balance, capable of weighing to the nearest 0.1 mg and up to 100 g.
8. Reagents
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society where such
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without lessening the accuracy of the determination.
8.2 Di-n-Butyl Sulfide (DBS), a high-purity material with a certified analysis for sulfur content. Use the certified sulfur content and
the material purity when calculating the exact concentrations of the calibration standards (see 9.110.1). (Warning—Di-n-butyl
sulfide is flammable and toxic. Prepared solutions may not be stable several months after preparation.)Warning—Solutions
prepared in a volatile or un-stabilized matrix may not be stable several months after preparation.)
NOTE 5—It is essential to know the concentration of sulfur in the di-n-butyl sulfide, not only the purity, since impurities may also be sulfur containing
compounds. The sulfur content may be determined via mass dilution in sulfur-free white oil followed by a direct comparison analysis against NIST (or
other primary standards body) reference materials.
8.3 Drift Correction Monitor(s) (Optional), Several different materials have been found to be suitable for use as drift correction
monitors. Appropriate drift monitor samples should be permanent materials that are stable with respect to repeated exposure to
X-rays. Stable liquids like polysulfide oils, glass, or metallic specimens are recommended. Liquids, pressed powders, and solid
materials that degrade with repeated exposure to X-rays should not be used. Examples of sulfur containing materials that have been
found to be suitable include a renewable liquid petroleum material, a metal alloy, or a fused glass disk. The monitor’s counting
rate, in combination with count time, shall be sufficient to give a relative counting error of less than 1 %. The counting rate for
the monitor sample is determined during calibration (see 9.410.4) and again at the time of analysis (see 10.111.1). These counting
rates are used to calculate a drift correction factor (see 11.112.1).
8.3.1 Drift correction is usually implemented automatically in software, although the calculation can readily be done manually.
For X-ray instruments that are highly stable, the magnitude of the drift correction factor may not differ significantly from unity.
8.4 Polysulfide Oil, generally nonyl polysulfides containing a known percentage of sulfur diluted in a hydrocarbon matrix.
(Warning—May cause allergic skin reactions.)reactions. Warning—Solutions prepared in a volatile or un-stabilized matrix may
not be stable several months after preparation.)
NOTE 6—Polysulfide oils are high molecular weight oils that contain high concentrations of sulfur, as high as 50 weight %.50 % by weight. They exhibit
excellent physical properties such as low viscosity, low volatility, and durable shelf life while being completely miscible in white oil. Polysulfide oils are
readily available commercially. The sulfur content of the polysulfide oil concentrate is determined via mass dilution in sulfur-free white oil followed by
a direct comparison analysis against NIST (or other primary standards body) reference materials.
8.5 Mineral Oil, White (MOW), ACS Reagent Grade containing less than 2 mg/kg sulfur or other suitable base material containing
less than 2 mg/kg sulfur. When low level (<200 mg ⁄kg) measurements are anticipated, then the sulfur content, if any, of the base
material needs to be included in the calculation of calibration standard concentration (see 9.110.1). When the sulfur content of the
solvent or reagent is not certified, verify the absence of sulfur. Use the purest grades for the preparation of calibration standards.
It is also important to measure the C/H ratio (see Section 1213 and Fig. 1).
8.6 X-ray Transparent Film—Any film that resists attack by the sample, is free of sulfur, and is sufficiently X-ray transparent can
be used. Film types can include polyester, polypropylene, polycarbonate, and polyimide. However, samples of high aromatic
content can dissolve polypropylene and polycarbonate films.
8.7 Helium Gas, minimum purity 99.9 %.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D2622 − 21
FIG. 1 Relative Sulfur Sensitivity versus C/H Ratio
8.8 Counting Gas, for instruments equipped with flow proportional counters. The purity of the counting gas should be in
agreement with the specification provided by the instrument manufacturer.
8.9 Sample Cells, compatible with the sample and the geometry requirements of the spectrometer. Disposable cells are preferred
over reusable ones for ultra low (<50 mg/kg) sulfur levels.
8.10 Calibration Check Samples, portions of one or more liquid petroleum or product standards of known or certified sulfur
content (including polysulfide oils, di-n-butyl sulfide, thiophenes, etc.) and not used in the generation of the calibration curve. The
check samples shall be used to determine the precision and accuracy of the initial calibration (see 9.510.5).
8.11 Quality Control Samples, stable petroleum or product samples or solids representative of the samples of interest that are run
on a regular basis to verify that the system is in statistical control (see Section 1314).
NOTE 7—Verification of system control through the use of QC samples and control charting is highly recommended. It is recognized that QC procedures
are the province of the individual laboratory.
NOTE 8—Suitable QC samples can often be prepared by combining retains of typical samples if they are stable. For monitors, solid materials are
recommended. QC samples must be stable over long periods.
9. Sampling and Specimen Preparation
9.1 Samples shall be taken in accordance with the instructions in Practices D4057 or D4177 when applicable.
9.2 When reusable sample cells are used, clean and dry cells before each use. Disposable sample cells shall not be reused. For
each sample, an unused piece of X-ray film is required for the sample cell. Avoid touching the inside of the sample cell, the portion
of the window film in the cell, or the instrument window (if the instrument is so equipped) that is exposed to X-rays. Oil from
fingerprints can affect the reading when analyzing for low levels of sulfur. Wrinkles in the film will affect the intensity of the sulfur
X-rays transmitted. Therefore, it is essential that the film be taut and clean to ensure reliable results. The analyzer may need
recalibration if the type or thickness of the window film is changed. After the sample cell is filled, create a small vent hole except
when the cell is of the sealed type.
9.3 Employ adequate storage, mixing, and sampling procedures. Refrigerate gasolines or other similar volatile materials to retain
sample integrity in storage, but allow them to return to room temperature before testing. Expose these materials to ambient
conditions only as long as necessary to obtain a sample for analysis.
9.4 Impurities or thickness variations, which may affect the measurement of low levels of sulfur, have been found in polyester
films and may vary from lot to lot. Therefore, the calibration should be checked after starting each new lot of film.
9.5 See Practice D7343 for more detailed sample handling and preparation information.
D2622 − 21
TABLE 2 Suggested Sulfur Standard Calibration Ranges
0–1000 0.10–1.00 1.0–5.0
mg/kg mass %% mass mass %% mass
A,B
0.0 0.100 1.0
B
5 0.250 2.0
B
10 0.500 3.0
B
100 1.000 4.0
250 5.0
A
Base material.
B
Analyze these standards in duplicate and use both values or the average value
in the calibration.
10. Calibration
10.1 Prepare calibration standards by careful mass dilution of the certified di-n-butyl sulfide with a sulfur-free white oil or other
suitable base material (see 7.58.5). The concentrations of the unknown samples must lie within the calibration range that is used.
Approximate recommended nominal sulfur concentration standards are listed in Table 2 for the sulfur concentration ranges of
interest. Take into account any sulfur in the base material when calculating the concentration of standards below 0.02 mass %
0.02 % by mass (200 mg/kg), as shown in Eq 1:
S 5 DBS 3S 1 WO 3S / DBS1WO (1)
@~ ! ~ !# ~ !
DBS WO
where:
S = mass percent sulfur of the prepared standards,
DBS = actual mass of DBS, g,
S = mass percent sulfur in DBS, typically 21.91 %,
DBS
WO = actual mass of white oil, g, and
S = mass percent sulfur in the white oil.
WO
NOTE 9—If desired, additional standards can prepared and analyzed with concentrations between those listed in Table 2, see 9.1.110.1.1.
10.1.1 Calibration standards can also be prepared by careful mixing of certified reference materials (CRM) of the same matrix,
so long as the sulfur values of the resulting blends and their uncertainties are characterized by the certifying body.
10.1.2 Alternatively, standards may be prepared by mass serial dilution of polysulfide oils (Note 6) with sulfur-free white oil. A
freshly prepared polysulfide oil calibration curve should be verified using CRMs traceable to a national measurement institution
that has demonstrated proficiency for measuring sulfur in the matrix of interest. Once a polysulfide oil calibration curve is
established, the calibration standards are stored at room temperature, out of direct sunlight, and in amber glass bottles. Polysulfide
oil standards can be prepared over a wide concentration range from low ppm to high weight percent levels of sulfur. They are easily
prepared in quantity and make excellent quality control standards. Shaking polysulfide oil standards before fresh aliquots are taken
is recommended to ensure the standard is uniformly blended. The high molecular weight of these sulfur compounds results in a
very low vapor pressure that inhibits X-ray film diffusion. Therefore, an autosampler can be employed during the measurement
process. Calibration curves prepared from polysulfide oils demonstrate excellent linearity and help the analyst visualize the full
dynamic range of their analytical method.
NOTE 10—Commercially available standards canmay be used provided their sulfur concentrations are accurately known and they approximate the nominal
concentrations listed in Table 2.
10.2 Establish calibration curve data by carefully determining the net intensity of the emitted sulfur radiation from each of the
standards by the procedures described in Sections 1011 and 1112.
10.2.1 Standards Analyze standards containing 100 mg/kg total sulfur or less must be analyzed in duplicate. Either both of the
Kelly, W. R., MacDonald, B. S., and Leigh, S. D., “A Method for the Preparation of NIST Traceable Fossil Fuel Standards with Concentrations Intermediate to SRM
Values,” Journal of ASTM International, Vol 4, No. 2, 2007.
D2622 − 21
individual values or the average value of these measurements canmay be used in the calibration. All samples in this sulfur
concentration range must also be analyzed in duplicate, as in 10.1211.12, and reported as in 12.1.113.1.1.
10.3 Construct a calibration model by using the software and algorithms supplied by the equipment vendor. The calibration model
typically takes one of the following forms (refer to the equipment vendor’s software documentation to determine the exact form):
C 5 a1bI linear calibration (2)
S
C 5 a1bI ~11 α C ! correction for matrix effects (3)
~ !
S ij j
(
C 5 a1bI~11 α C ! alternative correction for matrix effects (4)
S ( ij j
C 5 a1bI1cI 2nd order polynomial (5)
S
where:
C = mass fraction of sulfur. The concentration units depend on the fitted calibration constants a,b and c,
S
I = measured net intensity for the sulfur radiation,
a = fitted calibration line offset (intercept),
b = fitted calibration line slope,
c = fitted calibration line 2nd order polynomial,
a = correction factor for the effect of an interfering element (j) on sulfur (i). The interfering element can be sulfur when “matrix
ij
eliminated alphas” or empirical corrections are used, and
C = concentration of the interfering element (j).
j
NOTE 11—The a factors can be determined empirically through multiple regression, or theoretically through fundamental parameters. Equipment vendors
ij
typically have provisions in their software for calculating theoretical a’s.
10.3.1 Fit the calibration data over several ranges if necessary, depending on the sulfur concentrations to be covered. For example
(Table 2): 0 mass % to 0.10 mass % sulfur, 0.10 mass % to 1.0 mass % sulfur, and 1.0 mass % to 5.0 mass % 0 % to 0.10 % by
mass sulfur, 0.10 % to 1.0 % by mass sulfur, and 1.0 % to 5.0 % by mass sulfur.
NOTE 12—Calibration curves are typically linear to about 0.10 mass % 0.10 % by mass sulfur. The analyst should choose a linear calibration model when
fitting within this range. One of the other calibration models (correction for matrix effects or a 2nd order polynominal) should be chosen when fitting
to higher concentrations of sulfur.
10.4 When using drift correction monitors, determine the intensity of the drift correction monitor sample(s) during the calibration
procedure. The value determined corresponds to the factor A in Eq 7 in 11.112.1.
10.5 Immediately after completing the calibration, determine the sulfur concentration of one or more of the calibration check
samples (7.108.10). The differences between two measured values shall be within the repeatability of this test method (see
14.1.115.1.1). When this is not the case, the stability of the instrument and the repeatability of the sample preparation are suspect
and corrective measures should be taken. The degree of matrix mismatch between samples and standards should also be considered
when evaluating a calibration. Statistical quality control charts may be prepared for these materials to establish if the method is
in statistical control, as described in Section 1314.
11. Procedure
11.1 Instrument Setup—Before using any WDXRF spectrometer, it is essential that the instrument is performing to the
manufacturer’s specifications. Consult with the manufacturer on how to perform spectrometer quality control checks. Practice
D7343, Section 7 also provides more detailed information in this area.
11.2 Place particular attention on goniometer settings for sequential instruments, that is, ensuring goniometer positions are set
correctly. Before performing a calibration of the goniometer angles carry out pulse height discriminator settings (PHD’s) for each
element and background to be used. The angle should be checked first, then the PHD, then the angle re-checked if the PHD settings
are changed significantly. A very poor angle calibration may lead to bad PHD. The only reasonable alternate line is the sulfur
KβK-M (Kβ) with significantly less than 10 % of the sensitivity of the Kα;K-L (Kα); this will only be practical for samples
2,3 2,3
with high sulfur concentrations.
11.3 Account for observations of known instrument interferences. These include crystal fluorescence, tube line overlaps and any
D2622 − 21
TABLE 3 2θ Angles for Most Common Crystals
Background
S Kα
2d (0.5373
(0.5190 (0.5437
Crystal
(nm) nm)
nm) nm)
Deg 2θ
Deg 2θ Deg 2θ
Pentaerythritol (002) 0.8742 75.85 72.84 76.92
GE (111) 0.6532 110.68 105.23 112.68
element spectral contamination from the materials within the instrument construction. Lead is a particularly bad interference for
sulfur measurement. A number of these interferences can be avoided by careful selection of window settings during PHD set-up
and for element interference the selection of an alternative line or minimizing overlap using higher resolution collimators and
crystal selection.
11.4 When the factor F' is used in Eq 8, regularly analyze a blank sample to determine the factor F'. On a sulfur free sample, such
as the base material, determine the counting rate at the appropriate sulfur peak and background angles.
11.5 Place the sample in an appropriate cell using techniques consistent with good practice for the particular instrument being
used. Although sulfurincident radiation willmay penetrate only a smallsignificant distance into the sample, it sulfur radiation will
escape from only a small distance into the sample, and scatter from the sample cell and the sample can vary. Ensure that the sample
cell is filled above a minimum depth, beyond which additional sample does not significantly affect the counting rate
...


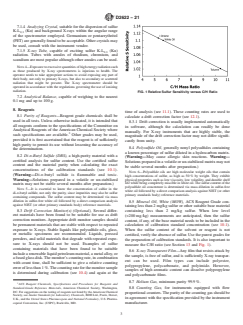





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...