ASTM F2706-18
(Test Method)Standard Test Methods for Occipital-Cervical and Occipital-Cervical-Thoracic Spinal Implant Constructs in a Vertebrectomy Model
Standard Test Methods for Occipital-Cervical and Occipital-Cervical-Thoracic Spinal Implant Constructs in a Vertebrectomy Model
SIGNIFICANCE AND USE
5.1 Occipital-cervical and occipital-cervical-thoracic spinal implants are generally composed of several components which, when connected together, form either an occipital-cervical spinal implant assembly or an occipital-cervical-thoracic spinal implant assembly. Occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are designed to provide some stability to the spine during the process of arthrodesis. These test methods outline standard materials and methods for the evaluation of different spinal implant assemblies to facilitate comparisons between different designs.
5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex loads of the occipital-cervical and occipital-cervical-thoracic spine.
5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these test methods. The results obtained here cannot be used directly to predict in vivo performance. The results can be used to compare different component designs in terms of the relative mechanical parameters.
5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for consistency. The effect of the environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency for this type of cyclic testing should be 5 Hz.
5.5 The location of the longitudinal elements is determined by the intended in vivo location of the ...
SCOPE
1.1 These test methods cover the materials and methods for the static and fatigue testing of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of occipital-cervical and occipital-cervical-thoracic spinal implant components can be specific depending on the intended location and intended method of attachment.
1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. They allow comparison of occipital-cervical and occipital-cervical-thoracic spinal implant constructs with different methods of application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a particular device.
1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and two fatigue tests for the comparative evaluation of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are defined.
1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness and strength of occipital-cervical or occipital-cervical-thoracic spinal implant assemblies.
1.5 It may not be possible to test some occipital-cervical and some occipital-cervical-thoracic spinal constructs in all test configurations.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine th...
General Information
- Status
- Published
- Publication Date
- 31-Mar-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 01-Apr-2018
- Effective Date
- 15-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-May-2015
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Feb-2013
- Effective Date
- 15-Dec-2012
- Effective Date
- 01-Dec-2012
- Effective Date
- 15-Nov-2012
- Effective Date
- 01-Sep-2012
Overview
ASTM F2706-18: Standard Test Methods for Occipital-Cervical and Occipital-Cervical-Thoracic Spinal Implant Constructs in a Vertebrectomy Model establishes standardized laboratory methods for evaluating spinal implant assemblies designed for the occipital-cervical and occipital-cervical-thoracic regions of the spine. Developed by ASTM International, this standard provides critical guidelines for assessing the static and fatigue mechanical properties of these spinal constructs using a vertebrectomy model, supporting comparative evaluations of different implant designs and materials.
Key Topics
- Scope and Purpose: ASTM F2706-18 covers test methods for static and fatigue testing of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. The aim is to provide a standardized approach for comparing mechanical performance among various designs.
- Testing Methods: The standard outlines procedures for three static load types (compression bending, tensile bending, and torsion) and two fatigue tests (compression bending fatigue and torsion fatigue).
- Vertebrectomy Model: Tests are conducted using polyacetal test blocks that represent the absence of a vertebral body. This approach removes variability associated with biological specimens and permits more consistent mechanical comparisons.
- Performance Metrics: The methods set guidelines for measuring displacements, determining yield loads, as well as stiffness and strength of implant assemblies.
- Comparative Evaluation: The results are designed to compare mechanical characteristics of different constructs under standardized conditions, though direct in vivo performance prediction is not intended.
Applications
ASTM F2706-18 serves manufacturers, researchers, and regulatory bodies by enabling:
- Mechanical Testing of Spinal Implants: Evaluate occipital-cervical and occipital-cervical-thoracic spinal implant constructs under controlled laboratory conditions.
- Design Validation: Facilitate the assessment of new implant designs and materials for mechanical strength and durability before clinical use.
- Comparative Assessment: Generate data to compare legacy, current, and emerging spinal implant systems, ensuring that new products perform as well as or better than existing options.
- Regulatory Submissions: Supply standardized mechanical data to support product registration and pre-market approval for spinal implant systems.
- Quality Assurance: Support consistent and repeatable testing protocols necessary for product development, quality control, and benchmarking within the orthopedic device industry.
Related Standards
For a comprehensive evaluation of spinal implant constructs, ASTM F2706-18 should be considered in conjunction with the following related standards:
- ASTM F1717: Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
- ASTM F2077: Test Methods for Intervertebral Body Fusion Devices
- ASTM E4: Practices for Force Verification of Testing Machines
- ASTM E6: Terminology Relating to Methods of Mechanical Testing
- ASTM E739: Practice for Statistical Analysis of Linear or Linearized Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
- ASTM F1582: Terminology Relating to Spinal Implants
- ASTM E1823: Terminology Relating to Fatigue and Fracture Testing
Practical Value
By standardizing the methodology for testing spinal implant constructs, ASTM F2706-18:
- Enables direct mechanical comparisons between different spinal implant systems, supporting innovation and safety.
- Increases the reliability of test data used by regulatory agencies during product evaluations.
- Reduces variability inherent to biological testing through the use of synthetic test models.
- Supports product development processes, ensuring new implant designs meet minimum mechanical criteria before clinical deployment.
For manufacturers and laboratories, adherence to ASTM F2706-18 assists in maintaining compliance, streamlining regulatory submissions, and enhancing confidence in spinal implant performance.
Buy Documents
ASTM F2706-18 - Standard Test Methods for Occipital-Cervical and Occipital-Cervical-Thoracic Spinal Implant Constructs in a Vertebrectomy Model
REDLINE ASTM F2706-18 - Standard Test Methods for Occipital-Cervical and Occipital-Cervical-Thoracic Spinal Implant Constructs in a Vertebrectomy Model
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2706-18 is a standard published by ASTM International. Its full title is "Standard Test Methods for Occipital-Cervical and Occipital-Cervical-Thoracic Spinal Implant Constructs in a Vertebrectomy Model". This standard covers: SIGNIFICANCE AND USE 5.1 Occipital-cervical and occipital-cervical-thoracic spinal implants are generally composed of several components which, when connected together, form either an occipital-cervical spinal implant assembly or an occipital-cervical-thoracic spinal implant assembly. Occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are designed to provide some stability to the spine during the process of arthrodesis. These test methods outline standard materials and methods for the evaluation of different spinal implant assemblies to facilitate comparisons between different designs. 5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex loads of the occipital-cervical and occipital-cervical-thoracic spine. 5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these test methods. The results obtained here cannot be used directly to predict in vivo performance. The results can be used to compare different component designs in terms of the relative mechanical parameters. 5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for consistency. The effect of the environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency for this type of cyclic testing should be 5 Hz. 5.5 The location of the longitudinal elements is determined by the intended in vivo location of the ... SCOPE 1.1 These test methods cover the materials and methods for the static and fatigue testing of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of occipital-cervical and occipital-cervical-thoracic spinal implant components can be specific depending on the intended location and intended method of attachment. 1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. They allow comparison of occipital-cervical and occipital-cervical-thoracic spinal implant constructs with different methods of application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a particular device. 1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and two fatigue tests for the comparative evaluation of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are defined. 1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness and strength of occipital-cervical or occipital-cervical-thoracic spinal implant assemblies. 1.5 It may not be possible to test some occipital-cervical and some occipital-cervical-thoracic spinal constructs in all test configurations. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine th...
SIGNIFICANCE AND USE 5.1 Occipital-cervical and occipital-cervical-thoracic spinal implants are generally composed of several components which, when connected together, form either an occipital-cervical spinal implant assembly or an occipital-cervical-thoracic spinal implant assembly. Occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are designed to provide some stability to the spine during the process of arthrodesis. These test methods outline standard materials and methods for the evaluation of different spinal implant assemblies to facilitate comparisons between different designs. 5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. The mechanical tests are conducted in vitro using simplified load schemes and do not attempt to mimic the complex loads of the occipital-cervical and occipital-cervical-thoracic spine. 5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in these test methods. The results obtained here cannot be used directly to predict in vivo performance. The results can be used to compare different component designs in terms of the relative mechanical parameters. 5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for consistency. The effect of the environment may be significant. Repeating all or part of these test methods in simulated body fluid, saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended frequency for this type of cyclic testing should be 5 Hz. 5.5 The location of the longitudinal elements is determined by the intended in vivo location of the ... SCOPE 1.1 These test methods cover the materials and methods for the static and fatigue testing of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of occipital-cervical and occipital-cervical-thoracic spinal implant components can be specific depending on the intended location and intended method of attachment. 1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. They allow comparison of occipital-cervical and occipital-cervical-thoracic spinal implant constructs with different methods of application to the spine. These test methods are not intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a particular device. 1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and two fatigue tests for the comparative evaluation of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are defined. 1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness and strength of occipital-cervical or occipital-cervical-thoracic spinal implant assemblies. 1.5 It may not be possible to test some occipital-cervical and some occipital-cervical-thoracic spinal constructs in all test configurations. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine th...
ASTM F2706-18 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2706-18 has the following relationships with other standards: It is inter standard links to ASTM F2706-17, ASTM E1823-24a, ASTM E1823-24, ASTM E1823-20, ASTM F2077-17, ASTM F1582-98(2016), ASTM F1717-15, ASTM F2077-14, ASTM E4-14, ASTM F1717-14, ASTM F1717-13, ASTM E1823-12e, ASTM F1717-12a, ASTM E1823-12d, ASTM E1823-12c. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2706-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2706 − 18
Standard Test Methods for
Occipital-Cervical and Occipital-Cervical-Thoracic Spinal
Implant Constructs in a Vertebrectomy Model
This standard is issued under the fixed designation F2706; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
1.1 These test methods cover the materials and methods for
mine the applicability of regulatory limitations prior to use.
thestaticandfatiguetestingofoccipital-cervicalandoccipital-
1.8 This international standard was developed in accor-
cervical-thoracic spinal implant assemblies in a vertebrectomy
dance with internationally recognized principles on standard-
model. The test materials for most combinations of occipital-
ization established in the Decision on Principles for the
cervical and occipital-cervical-thoracic spinal implant compo-
Development of International Standards, Guides and Recom-
nents can be specific depending on the intended location and
mendations issued by the World Trade Organization Technical
intended method of attachment.
Barriers to Trade (TBT) Committee.
1.2 These test methods are intended to provide a basis for
the mechanical comparison among past, present, and future 2. Referenced Documents
occipital-cervical and occipital-cervical-thoracic spinal im-
2.1 ASTM Standards:
plant assemblies. They allow comparison of occipital-cervical
E4Practices for Force Verification of Testing Machines
and occipital-cervical-thoracic spinal implant constructs with
E6Terminology Relating to Methods of Mechanical Testing
different methods of application to the spine. These test
E739PracticeforStatisticalAnalysisofLinearorLinearized
methodsarenotintendedtodefinelevelsofperformance,since
Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
sufficient knowledge is not available to predict the conse-
E1823TerminologyRelatingtoFatigueandFractureTesting
quences of the use of a particular device.
F1582Terminology Relating to Spinal Implants
1.3 These test methods set out guidelines for load types and F1717Test Methods for Spinal Implant Constructs in a
methods of applying loads. Methods for three static load types Vertebrectomy Model
and two fatigue tests for the comparative evaluation of F2077Test Methods for Intervertebral Body Fusion Devices
occipital-cervical and occipital-cervical-thoracic spinal im-
3. Terminology
plant assemblies are defined.
3.1 Definitions—For definitions of terms relating to these
1.4 These test methods establish guidelines for measuring
test methods, see Terminologies E6, F1582, and E1823.
displacements, determining the yield load, and evaluating the
stiffnessandstrengthofoccipital-cervicaloroccipital-cervical-
3.2 Definitions of Terms Specific to This Standard:
thoracic spinal implant assemblies. 3.2.1 active length of the longitudinal element, n—the
straight line distance between the centers of rotation of the test
1.5 Itmaynotbepossibletotestsomeoccipital-cervicaland
blocks.
some occipital-cervical-thoracic spinal constructs in all test
configurations. 3.2.2 block moment arm, n—the perpendicular to the ap-
pliedloadbetweentheinsertionpointofananchorandtheaxis
1.6 The values stated in SI units are to be regarded as
of the hinge pin.
standard. No other units of measurement are included in this
3.2.3 compressive or tensile bending stiffness (N/mm),
standard.
n—the compressive or tensile bending yield force divided by
1.7 This standard does not purport to address all of the
elastic displacement (see the initial slope of line BC in Fig. 1).
safety concerns, if any, associated with its use. It is the
3.2.4 compressive or tensile bending ultimate load (N),
n—the maximum compressive or tensile force in the X-Z plane
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of
Subcommittee F04.25 on Spinal Devices. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved April 1, 2018. Published May 2018. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2008. Last previous edition approved in 2017 as F2706 – 17. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F2706-18. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2706 − 18
FIG. 1 Typical Load Displacement Curve or Torque Angulation Curve
applied to an occipital-cervical or occipital-cervical-thoracic 3.2.10 failure, n—permanent deformation resulting from
spinalimplantassembly(seetheforceatPointEinFig.1).The fracture, plastic deformation, or loosening beyond the ultimate
ultimate load should be a function of the device and not of the displacementorlooseningthatrenderstheoccipital-cervicalor
load cell or testing machine. occipital-cervical-thoracic spinal implant assembly ineffective
or unable to adequately resist load.
3.2.5 compressive or tensile bending yield load (N), n—the
compressive or tensile bending force in the X-Z plane neces- 3.2.11 fatigue life, n—the number of loading cycles, N,ofa
sary to produce a permanent deformation equal to 0.020 times specified character that the occipital-cervical or occipital-
the active length of the longitudinal element (see the force at cervical-thoracic spinal implant assembly sustains before fail-
Point D in Fig. 1). ure of a specified nature occurs (see Terminology E1823).
3.2.6 coordinate system/axes, n—three orthogonal axes are 3.2.12 hinge pin, n—the cylindrical rod connecting a test
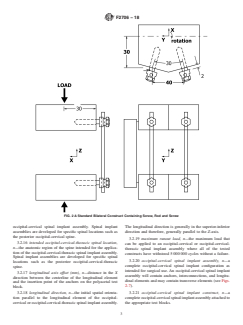
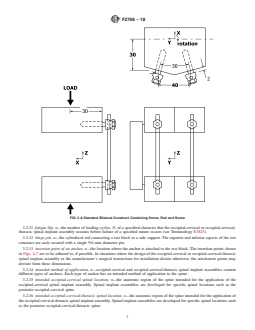
defined in Figs. 2 and 3. The anterior-posterior axis is X with blocktoasidesupport.Thesuperiorandinferioraspectsofthe
positive being anterior. The medial-lateral axis is Y with left test construct are each secured with a single 9.6-mm diameter
being positive when viewed posteriorly. The superior-inferior pin.
axis is Z with superior being positive.
3.2.13 insertion point of an anchor, n—the location where
3.2.7 displacement at 2 % offset yield (mm), n—the dis- the anchor is attached to the test block. The insertion points
placement of a construct measured via the actuator that shown in Figs. 4-7 are to be adhered to, if possible. In
produces a permanent deformation equal to 0.020 times the situations where the design of the occipital-cervical or
active length of the longitudinal element (distance OA in Fig. occipital-cervical-thoracic spinal implant assembly or the
1). manufacturer’s surgical instructions for installation dictate
otherwise, the attachment points may deviate from these
3.2.8 elasticangulardisplacement(degrees),n—theangular
dimensions.
displacement at 2% offset yield (see PointAin Fig. 1) minus
the2%offsetangulardisplacement(seePointBinFig.1)(that 3.2.14 intended method of application, n—occipital-cervical
is, the distance between Point A and Point B in Fig. 1). and occipital-cervical-thoracic spinal implant assemblies con-
tain different types of anchors. Each type of anchor has an
3.2.9 elastic displacement (mm), n—the displacement at
intended method of application to the spine.
2% offset yield (see Point A in Fig. 1) minus the 2% offset
displacement (see Point B in Fig. 1). (The distance between 3.2.15 intended occipital-cervical spinal location, n—the
Point A and Point B in Fig. 1.) anatomicregionofthespineintendedfortheapplicationofthe
F2706 − 18
FIG. 2 A Standard Bilateral Construct Containing Screw, Rod and Screw
occipital-cervical spinal implant assembly. Spinal implant The longitudinal direction is generally in the superior-inferior
assemblies are developed for specific spinal locations such as
direction and therefore, generally parallel to the Z-axis.
the posterior occipital-cervical spine.
3.2.19 maximum runout load, n—the maximum load that
3.2.16 intended occipital-cervical-thoracic spinal location,
can be applied to an occipital-cervical or occipital-cervical-
n—the anatomic region of the spine intended for the applica-
thoracic spinal implant assembly where all of the tested
tion of the occipital-cervical-thoracic spinal implant assembly.
constructs have withstood 5000000 cycles without a failure.
Spinal implant assemblies are developed for specific spinal
3.2.20 occipital-cervical spinal implant assembly, n—a
locations such as the posterior occipital-cervical-thoracic
complete occipital-cervical spinal implant configuration as
spine.
intended for surgical use. An occipital-cervical spinal implant
3.2.17 longitudinal axis offset (mm), n—distance in the X
assembly will contain anchors, interconnections, and longitu-
direction between the centerline of the longitudinal element
dinal elements and may contain transverse elements (see Figs.
and the insertion point of the anchors on the polyacetal test
2-7).
block.
3.2.21 occipital-cervical spinal implant construct, n—a
3.2.18 longitudinal direction, n—the initial spatial orienta-
completeoccipital-cervicalspinalimplantassemblyattachedto
tion parallel to the longitudinal element of the occipital-
cervical or occipital-cervical-thoracic spinal implant assembly. the appropriate test blocks.
F2706 − 18
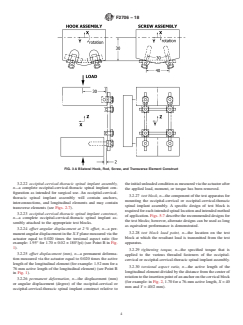
FIG. 3 A Bilateral Hook, Rod, Screw, and Transverse Element Construct
3.2.22 occipital-cervical-thoracic spinal implant assembly, theinitialunloadedconditionasmeasuredviatheactuatorafter
n—a complete occipital-cervical-thoracic spinal implant con-
the applied load, moment, or torque has been removed.
figuration as intended for surgical use. An occipital-cervical-
3.2.27 test block, n—thecomponentofthetestapparatusfor
thoracic spinal implant assembly will contain anchors,
mounting the occipital-cervical or occipital-cervical-thoracic
interconnections, and longitudinal elements and may contain
spinal implant assembly. A specific design of test block is
transverse elements (see Figs. 2-7).
requiredforeachintendedspinallocationandintendedmethod
3.2.23 occipital-cervical-thoracic spinal implant construct,
ofapplication.Figs.5-7describetherecommendeddesignsfor
n—a complete occipital-cervical-thoracic spinal implant as-
the test blocks; however, alternate designs can be used as long
sembly attached to the appropriate test blocks.
as equivalent performance is demonstrated.
3.2.24 offset angular displacement at 2 % offset, n—a per-
3.2.28 test block load point, n—the location on the test
manentangulardisplacementinthe X-Yplanemeasuredviathe
block at which the resultant load is transmitted from the test
actuator equal to 0.020 times the torsional aspect ratio (for
apparatus.
example: 1.95° for 1.70 × 0.02 × 180°/pi) (see Point B in Fig.
1).
3.2.29 tightening torque, n—the specified torque that is
3.2.25 offset displacement (mm), n—a permanent deforma-
applied to the various threaded fasteners of the occipital-
tion measured via the actuator equal to 0.020 times the active
cervical or occipital-cervical-thoracic spinal implant assembly.
lengthofthelongitudinalelement(forexample:1.52mmfora
3.2.30 torsional aspect ratio, n—the active length of the
76 mm active length of the longitudinal element) (see Point B
longitudinalelementdividedbythedistancefromthecenterof
in Fig. 1).
rotationtotheinsertionpointofananchoronthecervicalblock
3.2.26 permanent deformation, n—the displacement (mm)
(for example: in Fig. 2, 1.70 for a 76-mm active length, X=40
or angular displacement (degree) of the occipital-cervical or
mm and Y = 40/2 mm).
occipital-cervical-thoracic spinal implant construct relative to
F2706 − 18
FIG. 4 Occipital-Cervical Bilateral Construct Test Setup for Occipital Screws or Bolts
L L
3.2.34 yield displacement (distance OA—Fig. 6), n—the
A 5 5 (1)
2 2 1/2
D x 1y displacement (mm) or angular displacement (deg) when an
~ !
assembly has a permanent deformation equal to the offset
where:
displacement or the offset angular displacement.
L = active length of longitudinal element,
3.2.35 yield torque (N-m), n—the torque in the X-Y plane
D = distance to insertion point,
required to produce a permanent displacement of 0.020 times
x = x distance to insertion point, and
the torsional aspect ratio (the torque at Point D in Fig. 1).
y = y distance to insertion point.
3.2.36 zero displacement intercept (mm), n—theintersection
3.2.31 torsional stiffness (N-m/degree), n—the yield torque
of the straight line section of the load-displacement curve and
(N-m) divided by elastic angular displacement (degrees) (the
the zero load axis (the zero displacement reference Point 0 in
initial slope of line BC in Fig. 1).
Fig. 1).
3.2.32 torsional ultimate load (N-m), n—the maximum
torque in the X-Y plane applied to an occipital-cervical or
4. Summary of Test Methods
occipital-cervical-thoracic spinal implant assembly (the torque
4.1 Similar test methods are proposed for the mechanical
at Point E in Fig. 1). The ultimate torque should be a function
evaluation of all occipital-cervical and occipital-cervical-
of the device and not of the load cell or testing machine.
thoracic spinal implant assemblies (see Fig. 4).
3.2.33 ultimate displacement (mm), n—the displacement
associated with the ultimate load, ultimate bending load or 4.2 A vertebrectomy model is used for the evaluation of
ultimate torque (the displacement at Point F in Fig. 1). both occipital-cervical and occipital-cervical-thoracic systems.
F2706 − 18
FIG. 5 Occipital Bilateral Polyacetal Block for Occipital Screws or Bolts
The spinal hardware is attached at the superior and inferior Occipital-cervical and occipital-cervical-thoracic spinal im-
aspects to polyacetal homopolymer (polyacetal) test blocks
plant assemblies contain different types of anchors. Each type
separated by a large gap.The polyacetal homopolymer used to
of anchor has an intended method of application to the spine.
manufacture the test blocks should have a tensile breaking
For example, one assembly may include screws and rods (see
strengthnolessthan61MPa.Theuseofpolyacetaltestblocks
Fig. 2), while another assembly may contain screws, hooks,
(see Figs. 5-8) eliminates the effects of the variability of bone
rods, and transverse elements (see Fig. 3). The block moment
geometry and material properties associated with cadaveric
arm of a test configuration will be independent of the intended
testing.Alternate designs of test blocks may be used as long as
method of application of a spinal implant assembly, thereby
equivalent performance is demonstrated.
allowing the user to compare devices for a given load.
4.3 Threestaticmechanicaltestsandtwodynamictestswill However, it should be noted that the same load on different
evaluate the occipital-cervical or occipital-cervical-thoracic implant assemblies will result in different loading on the
spinalimplantassemblies.Thethreestaticmechanicaltestsare
implants as a function of implant design. The user should take
compression bending, tensile bending, and torsion. The dy- this into account when evaluating the performance of a device.
namic tests are compression bending fatigue and torsion
fatigue.
5. Significance and Use
4.4 A specific clinical indication generally requires a spe-
5.1 Occipital-cervical and occipital-cervical-thoracic spinal
cific occipital-cervical or occipital-cervical-thoracic spinal im-
implantsaregenerallycomposedofseveralcomponentswhich,
plant assembly. Occipital-cervical and occipital-cervical-
when connected together, form either an occipital-cervical
thoracic spinal implant assemblies will be evaluated with test
spinalimplantassemblyoranoccipital-cervical-thoracicspinal
configurations that simulate the clinical requirements for the
implant assembly. Occipital-cervical and occipital-cervical-
intended spinal location. The intended spinal location is the
thoracic spinal implant assemblies are designed to provide
posterior surface of the occipital-cervical or occipital-cervical-
some stability to the spine during the process of arthrodesis.
thoracic spine (see Fig. 4). The block moment arm for a test
These test methods outline standard materials and methods for
configuration depends on the intended spinal location. The
the evaluation of different spinal implant assemblies to facili-
block moment arm of the occipital-cervical or occipital-
tate comparisons between different designs.
cervical-thoracic spine configuration (see Fig. 4) varies de-
pendingontheoccipitalattachmentcomponents,butshouldbe
5.2 These test methods are used to quantify the static and
no less than the block moment arm specified in the cervical
dynamic mechanical characteristics of different designs of
spineconfiguration.Thecervicalspineconfiguration(seeFigs.
occipital-cervical and occipital-cervical-thoracic spinal im-
6 and 7) specifies the block moment arm.
plant assemblies. The mechanical tests are conducted in vitro
4.5 The intended method of application of the occipital- using simplified load schemes and do not attempt to mimic the
complex loads of the occipital-cervical and occipital-cervical-
cervical or occipital-cervical-thoracic spinal implant assembly
mayvaryforspecificanatomicregionsandclinicalindications. thoracic spine.
F2706 − 18
FIG. 6 Cervical Bilateral Polyacetal Block for Screws or Bolts
5.3 The loads applied to the spinal implant assemblies in thereby affect the relative performance of tested devices. This
vivo will, in general, differ from the loading configurations test should be initially performed dry (ambient room condi-
used in these test methods.The results obtained here cannot be
tions) for consistency. The effect of the environment may be
useddirectlytopredict in vivoperformance.Theresultscanbe
significant. Repeating all or part of these test methods in
used to compare different component designs in terms of the
simulated body fluid, saline (9 g NaCl per 1000 mL water), a
relative mechanical parameters.
saline drip, water, or a lubricant should be considered. The
5.4 Fatigue testing in a simulated body fluid or saline may maximum recommended frequency for this type of cyclic
cause fretting, corrosion, or lubricate the interconnections and testing should be 5 Hz.
F2706 − 18
FIG. 7 Cervical Bilateral Polyacetal Block for Hooks, Cables, Wires
5.5 The location of the longitudinal elements is determined block is independent of anchor-type for the cervical block, but
by the intended in vivo location of the anchors. The perpen- dependent on the design for the occipital test block. The
dicular distance to the load direction between the axis of a distance between the polyacetal block and the center of the
hinge pin and the anchor’s attachment points to a polyacetal longitudinal element is a function of the design of the implant.
F2706 − 18
the spinal implant assembly in a manner that simulates the
specific clinical indication at the intended spinal location.
6.2.4 Regardless of what testing method is employed, all
polyacetal screws are to be inserted 2 mm short of the depth
they would be inserted in vivo. In other words, the screws are
to be inserted to the point at which they would just come in
contact with the vertebra (that is, polyacetal blocks) and then
backed out 2 mm. Alternatively, insertion points for screws
may be counterbored at a minimum of 300% of the diameter
of the screw (depth of 2 mm) used to purchase the polyacetal.
Both methods effectively prevent biasing the test toward any
particulardesignduetoabuttressingeffectofthescrewagainst
thepolyacetal,whichcouldeffectivelystressshieldtheimplant
from failure that might be experienced in vivo.
6.3 The design of the occipital polyacetal blocks causes the
plane through the occipital implant assemblies to be angled at
23° from the X-Y plane. (This angle from the X-Y plane to the
occiputistheaverageoccipitocervicalneutralpositionangleas
determined from human radiological data, n=15.) The design
of the cervical polyacetal blocks causes the plane through the
spinal implant assemblies to be parallel to the plane (the Y-Z
Representative of how Figs. 2-7 can be modified to allow for unconstrained
plane) through the axes of the hinge pins. Align the superior
motion.
polyacetal block with the inferior polyacetal block. The center
FIG. 8 Alternate Lumbar Bilateral Polyacetal Block for Screws
axis of each hinge pin should be perpendicular (60.5°) to and
and Bolts
aligned (60.5 mm) with the load axis of the test machine.
Center the test apparatus in the test machine such that the line
6. Apparatus
throughthemid-point(0,0,Z1)oftheinferiorhingepin’saxis
and the mid-point (0, 0, Z2) of the superior hinge pin’s axis is
6.1 Test machines shall conform to the requirements of
collinearwithin 60.1mmoftheloadandrotationalaxisofthe
Practices E4.
test machine’s actuator. Or, in the case of spherical joint or
6.2 Thetestapparatusallowsmultipleloadingregimestobe
pin-slot gimbal mechanism, the points (0, 0, Z1) and (0, 0, Z2)
appliedtoallformsofoccipital-cervicalandoccipital-cervical-
are to be collinear (60.1 mm) with the test machine’s actuator.
thoracic spinal implant assemblies. Two pair of side supports
6.4 Alternativedesignsoftestblocksmaybeusedaslongas
are mounted on the test machine (see Fig. 4). One pair of side
equivalence is demonstrated. The solid polyacetal test blocks
supports attach to the actuator and the second to the load cell.
may be replaced with metal blocks with polyacetal inserts of
A mounting plate for one of the sets of side support plates
appropriate size. Any surface or component of the occipital-
should be free to rotate about the Z-axis for the compression
cervical or occipital-cervical-thoracic spinal assembly which
bending, tension bending and fatigue tests. Polyacetal blocks
would contact the solid polyacetal should also contact an
are connected to the side supports via hinge pins, as one
appropriate thickness of the polyacetal. If screws are used to
possible test method, to facilitate comparison to constructs
mount the spinal construct to the test blocks, then the screws
tested as in Test Methods F1717. However, it is recommended
shall be placed into polyacetal inserts in the alternate design of
that unconstrained motion be used as is specified in Test
testblock.Thediameterofthepolyacetalinsertsshallbeequal
Methods F2077 and is summarized as follows:
to or greater than three times the diameter of the screws.
6.2.1 Sphere joint superior-inferior blocks joint blocks,
made of polyacetal, are shown in Fig. 8. A 12.7 mm ( ⁄2 in.)
6.5 Ifthelocationsofthesuperioranchors,inferioranchors,
diameter spherical socket is shown to maintain the 12 mm
orbothsetsofanchorsaredictatedbythelongitudinalelement
longitudinaldistancethatwasusedforthepinnedblockinFig.
and are at different Z locations (a diagonal), then the set of
6. Spherical blocks are limited to conducting compression-
anchors should be centered above and below the standard
flexion tests.
location such that they maintain the average Z location. If the
6.2.2 A spherical gimbal superior block with push rod
anchors are secured into slots in the longitudinal element, then
system similar to that used in Test Methods F2077 could be
theyshouldbecentrallyplacedintheslotsandnotateitherend
used as an alternative to perform unconstrained torsion,
to produce a worst case scenario.
compression-flexion tests, and tensile-extension tests. See
6.6 Different spinal implant assemblies have different in-
Figure 4, Torsion Testing Configuration With Pin-Slot Gimbal
tended methods of application to the polyacetal blocks. The
in Test Methods F2077.
locations of the longitudinal elements are determined by the
6.2.3 All testing will simulate a vertebrectomy model via a
design of anchors and interconnections. The load capacity of
large gap between the two polyacetal blocks. Select the
appropriate design of the polyacetal blocks (see Figs. 2-7 and
Figures 2 and 4 of Test Methods F2077) to facilitate testing of Personal correspondence with John Kirkpatrick, M.D., September 21, 2007.
F2706 − 18
the spinal construct is a function of the designs of the 1431. (See Note 1.) Alternative configurations such as a 196
interconnections, anchors, and longitudinal elements but mm occipital-cervical-thoracic construct with the inferior
block fixed, rather than free to rotate, can be used with
should not be a function of the test apparatus, hence the
justification. The active length should be constant for all
addition of the 2 mm offset or counterbore when attaching the
constructsusedincomparativetesting.Inallcases,selectionof
construct to the blocks (see 6.2).
attachment method (hooks, screws, etc.) at all points on the
6.7 The hinge pin in the test configuration allows the same
construct should match the configuration expected in the
test apparatus to be used for the static compression bending
worst-caseconstructindicatedforuse.Forexample,constructs
test, static tensile bending test, and static torsion test as well as
thatareindicatedforscrewfixationtothethoracicspineshould
thecompressionbendingfatiguetest.Thepolyacetalblocksare
be tested with screws, rather than hooks, at the inferior test
allowed to rotate around the Y-axis of the hinge pin during the
block.
compression bending, tensile bending, and fatigue tests. NOTE1—The76mmisdefinedasthezdistancefromhingepintohinge
pin instead of the z distance from the occipital attachment point to the
6.8 Ifasuperiorsphericaljointisused,thetestconfiguration
cervical attachment point, thereby providing a fixed distance for all
implant assemblies.
allows the same test apparatus to be used for the static
compression bending test and the compression bending fatigue 6.14 The testing machine or the apparatus used in the static
test.The polyacetal blocks are free to rotate around the Y-axis, torsion test applies torque about the Z-axis without constrain-
ing displacement in the Z direction. The aluminum blocks are
Z-axis, and X-axis during the compression bending static and
placed in the apparatus to prevent rotation in the X-Z plane
fatigue tests.
during the static torsion tests.
6.9 If a pin-slot gimbal mechanism is used (Figure 4, Test
Methods F2077), the test configuration allows the same test
7. Sampling
apparatus to be used for the static compression bending test,
7.1 All components in the occipital-cervical or occipital-
static tensile bending test, and static torsion test as well as the
cervical-thoracic spinal implant assembly shall be previously
compression bending fatigue test. The polyacetal blocks are
unused parts. Implants shall not be retested.
free to rotate around the Y-axis of the hinge pin, Z-axis, and
7.2 Use the polyacetal test blocks for only one test. When
X-axis during the compression bending, tensile bending, and
alternate designs of test blocks are used, then all polyacetal
fatigue tests.
inserts should be replaced after each test.Alternate designs of
6.10 Modifiedbilateralpolyacetalblocks(seeFigs.4and5)
test bl
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2706 − 17 F2706 − 18
Standard Test Methods for
Occipital-Cervical and Occipital-Cervical-Thoracic Spinal
Implant Constructs in a Vertebrectomy Model
This standard is issued under the fixed designation F2706; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods cover the materials and methods for the static and fatigue testing of occipital-cervical and
occipital-cervical-thoracic spinal implant assemblies in a vertebrectomy model. The test materials for most combinations of
occipital-cervical and occipital-cervical-thoracic spinal implant components can be specific depending on the intended location and
intended method of attachment.
1.2 These test methods are intended to provide a basis for the mechanical comparison among past, present, and future
occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. They allow comparison of occipital-cervical and
occipital-cervical-thoracic spinal implant constructs with different methods of application to the spine. These test methods are not
intended to define levels of performance, since sufficient knowledge is not available to predict the consequences of the use of a
particular device.
1.3 These test methods set out guidelines for load types and methods of applying loads. Methods for three static load types and
two fatigue tests for the comparative evaluation of occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are
defined.
1.4 These test methods establish guidelines for measuring displacements, determining the yield load, and evaluating the stiffness
and strength of occipital-cervical or occipital-cervical-thoracic spinal implant assemblies.
1.5 It may not be possible to test some occipital-cervical and some occipital-cervical-thoracic spinal constructs in all test
configurations.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Verification of Testing Machines
E6 Terminology Relating to Methods of Mechanical Testing
E739 Practice for Statistical Analysis of Linear or Linearized Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
E1823 Terminology Relating to Fatigue and Fracture Testing
F1582 Terminology Relating to Spinal Implants
F1717 Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
F2077 Test Methods for Intervertebral Body Fusion Devices
These test methods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and are the direct responsibility of Subcommittee
F04.25 on Spinal Devices.
Current edition approved March 1, 2017April 1, 2018. Published April 2017May 2018. Originally approved in 2008. Last previous edition approved in 20142017 as F2706
– 08 (2014). 17. DOI: 10.1520/F2706-17.10.1520/F2706-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2706 − 18
3. Terminology
3.1 Definitions—For definitions of terms relating to these test methods, see Terminologies E6, F1582, and E1823.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 active length of the longitudinal element, n—the straight line distance between the centers of rotation of the test blocks.
3.2.2 block moment arm, n—the perpendicular to the applied load between the insertion point of an anchor and the axis of the
hinge pin.
3.2.3 compressive or tensile bending stiffness (N/mm), n—the compressive or tensile bending yield force divided by elastic
displacement (see the initial slope of line BC in Fig. 1).
3.2.4 compressive or tensile bending ultimate load (N), n—the maximum compressive or tensile force in the X-Z plane applied
to an occipital-cervical or occipital-cervical-thoracic spinal implant assembly (see the force at Point E in Fig. 1). The ultimate load
should be a function of the device and not of the load cell or testing machine.
3.2.5 compressive or tensile bending yield load (N), n—the compressive or tensile bending force in the X-Z plane necessary to
produce a permanent deformation equal to 0.020 times the active length of the longitudinal element (see the force at Point D in
Fig. 1).
3.2.6 coordinate system/axes, n—three orthogonal axes are defined in Figs. 2 and 3. The anterior-posterior axis is X with positive
being anterior. The medial-lateral axis is Y with left being positive when viewed posteriorly. The superior-inferior axis is Z with
superior being positive.
3.2.7 displacement at 2 % offset yield (mm), n—the displacement of a construct measured via the actuator that produces a
permanent deformation equal to 0.020 times the active length of the longitudinal element (distance OA in Fig. 1).
3.2.8 elastic angular displacement (degrees), n—the angular displacement at 2 % offset yield (see Point A in Fig. 1) minus the
2 % offset angular displacement (see Point B in Fig. 1) (that is, the distance between Point A and Point B in Fig. 1).
3.2.9 elastic displacement (mm), n—the displacement at 2 % offset yield (see Point A in Fig. 1) minus the 2 % offset
displacement (see Point B in Fig. 1). (The distance between Point A and Point B in Fig. 1.)
3.2.10 failure, n—permanent deformation resulting from fracture, plastic deformation, or loosening beyond the ultimate
displacement or loosening that renders the occipital-cervical or occipital-cervical-thoracic spinal implant assembly ineffective or
unable to adequately resist load.
FIG. 1 Typical Load Displacement Curve or Torque Angulation Curve
F2706 − 18
FIG. 2 A Standard Bilateral Construct Containing Screw, Rod and Screw
3.2.11 fatigue life, n—the number of loading cycles, N, of a specified character that the occipital-cervical or occipital-cervical-
thoracic spinal implant assembly sustains before failure of a specified nature occurs (see Terminology E1823).
3.2.12 hinge pin, n—the cylindrical rod connecting a test block to a side support. The superior and inferior aspects of the test
construct are each secured with a single 9.6-mm diameter pin.
3.2.13 insertion point of an anchor, n—the location where the anchor is attached to the test block. The insertion points shown
in Figs. 4-7 are to be adhered to, if possible. In situations where the design of the occipital-cervical or occipital-cervical-thoracic
spinal implant assembly or the manufacturer’s surgical instructions for installation dictate otherwise, the attachment points may
deviate from these dimensions.
3.2.14 intended method of application, n—occipital-cervical and occipital-cervical-thoracic spinal implant assemblies contain
different types of anchors. Each type of anchor has an intended method of application to the spine.
3.2.15 intended occipital-cervical spinal location, n—the anatomic region of the spine intended for the application of the
occipital-cervical spinal implant assembly. Spinal implant assemblies are developed for specific spinal locations such as the
posterior occipital-cervical spine.
3.2.16 intended occipital-cervical-thoracic spinal location, n—the anatomic region of the spine intended for the application of
the occipital-cervical-thoracic spinal implant assembly. Spinal implant assemblies are developed for specific spinal locations such
as the posterior occipital-cervical-thoracic spine.
F2706 − 18
FIG. 3 A Bilateral Hook, Rod, Screw, and Transverse Element Construct
3.2.17 longitudinal axis offset (mm), n—distance in the X direction between the centerline of the longitudinal element and the
insertion point of the anchors on the polyacetal test block.
3.2.18 longitudinal direction, n—the initial spatial orientation parallel to the longitudinal element of the occipital-cervical or
occipital-cervical-thoracic spinal implant assembly. The longitudinal direction is generally in the superior-inferior direction and
therefore, generally parallel to the Z-axis.
3.2.19 maximum runout load, n—the maximum load that can be applied to an occipital-cervical or occipital-cervical-thoracic
spinal implant assembly where all of the tested constructs have withstood 5 000 000 cycles without a failure.
3.2.20 occipital-cervical spinal implant assembly, n—a complete occipital-cervical spinal implant configuration as intended for
surgical use. An occipital-cervical spinal implant assembly will contain anchors, interconnections, and longitudinal elements and
may contain transverse elements (see Figs. 2-7).
3.2.21 occipital-cervical spinal implant construct, n—a complete occipital-cervical spinal implant assembly attached to the
appropriate test blocks.
3.2.22 occipital-cervical-thoracic spinal implant assembly, n—a complete occipital-cervical-thoracic spinal implant configu-
ration as intended for surgical use. An occipital-cervical-thoracic spinal implant assembly will contain anchors, interconnections,
and longitudinal elements and may contain transverse elements (see Figs. 2-7).
3.2.23 occipital-cervical-thoracic spinal implant construct, n—a complete occipital-cervical-thoracic spinal implant assembly
attached to the appropriate test blocks.
3.2.24 offset angular displacement at 2 % offset, n—a permanent angular displacement in the X-Y plane measured via the
actuator equal to 0.020 times the torsional aspect ratio (for example: 1.95° for 1.70 × 0.02 × 180°/pi) (see Point B in Fig. 1).
3.2.25 offset displacement (mm), n—a permanent deformation measured via the actuator equal to 0.020 times the active length
of the longitudinal element (for example: 1.52 mm for a 76 mm active length of the longitudinal element) (see Point B in Fig. 1).
3.2.26 permanent deformation, n—the displacement (mm) or angular displacement (degree) of the occipital-cervical or
occipital-cervical-thoracic spinal implant construct relative to the initial unloaded condition as measured via the actuator after the
applied load, moment, or torque has been removed.
F2706 − 18
FIG. 4 Occipital-Cervical Bilateral Construct Test Setup for Occipital Screws or Bolts
3.2.27 test block, n—the component of the test apparatus for mounting the occipital-cervical or occipital-cervical-thoracic spinal
implant assembly. A specific design of test block is required for each intended spinal location and intended method of application.
Figs. 5-7 describe the recommended designs for the test blocks; however, alternate designs can be used as long as equivalent
performance is demonstrated.
3.2.28 test block load point, n—the location on the test block at which the resultant load is transmitted from the test apparatus.
3.2.29 tightening torque, n—the specified torque that is applied to the various threaded fasteners of the occipital-cervical or
occipital-cervical-thoracic spinal implant assembly.
3.2.30 torsional aspect ratio, n—the active length of the longitudinal element divided by the distance from the center of rotation
to the insertion point of an anchor on the cervical block (for example: in Fig. 2, 1.70 for a 76-mm active length, X = 40 mm and
Y = 40/2 mm).
L L
A 5 5 (1)
2 2 1/2
D ~x 1y !
where:
L = active length of longitudinal element,
D = distance to insertion point,
x = x distance to insertion point, and
y = y distance to insertion point.
F2706 − 18
FIG. 5 Occipital Bilateral Polyacetal Block for Occipital Screws or Bolts
3.2.31 torsional stiffness (N-m/degree), n—the yield torque (N-m) divided by elastic angular displacement (degrees) (the initial
slope of line BC in Fig. 1).
3.2.32 torsional ultimate load (N-m), n—the maximum torque in the X-Y plane applied to an occipital-cervical or
occipital-cervical-thoracic spinal implant assembly (the torque at Point E in Fig. 1). The ultimate torque should be a function of
the device and not of the load cell or testing machine.
3.2.33 ultimate displacement (mm), n—the displacement associated with the ultimate load, ultimate bending load or ultimate
torque (the displacement at Point F in Fig. 1).
3.2.34 yield displacement (distance OA—Fig. 6), n—the displacement (mm) or angular displacement (deg) when an assembly
has a permanent deformation equal to the offset displacement or the offset angular displacement.
3.2.35 yield torque (N-m), n—the torque in the X-Y plane required to produce a permanent displacement of 0.020 times the
torsional aspect ratio (the torque at Point D in Fig. 1).
3.2.36 zero displacement intercept (mm), n—the intersection of the straight line section of the load-displacement curve and the
zero load axis (the zero displacement reference Point 0 in Fig. 1).
4. Summary of Test Methods
4.1 Similar test methods are proposed for the mechanical evaluation of all occipital-cervical and occipital-cervical-thoracic
spinal implant assemblies (see Fig. 4).
4.2 A vertebrectomy model is used for the evaluation of both occipital-cervical and occipital-cervical-thoracic systems. The
spinal hardware is attached at the superior and inferior aspects to polyacetal homopolymer (polyacetal) test blocks separated by
a large gap. The polyacetal homopolymer used to manufacture the test blocks should have a tensile breaking strength no less than
61 MPa. The use of polyacetal test blocks (see Figs. 5-8) eliminates the effects of the variability of bone geometry and material
properties associated with cadaveric testing. Alternate designs of test blocks may be used as long as equivalent performance is
demonstrated.
4.3 Three static mechanical tests and two dynamic tests will evaluate the occipital-cervical or occipital-cervical-thoracic spinal
implant assemblies. The three static mechanical tests are compression bending, tensile bending, and torsion. The dynamic tests are
compression bending fatigue and torsion fatigue.
4.4 A specific clinical indication generally requires a specific occipital-cervical or occipital-cervical-thoracic spinal implant
assembly. Occipital-cervical and occipital-cervical-thoracic spinal implant assemblies will be evaluated with test configurations
that simulate the clinical requirements for the intended spinal location. The intended spinal location is the posterior surface of the
occipital-cervical or occipital-cervical-thoracic spine (see Fig. 4). The block moment arm for a test configuration depends on the
intended spinal location. The block moment arm of the occipital-cervical or occipital-cervical-thoracic spine configuration (see Fig.
F2706 − 18
FIG. 6 Cervical Bilateral Polyacetal Block for Screws or Bolts
4) varies depending on the occipital attachment components, but should be no less than the block moment arm specified in the
cervical spine configuration. The cervical spine configuration (see Figs. 6 and 7) specifies the block moment arm.
4.5 The intended method of application of the occipital-cervical or occipital-cervical-thoracic spinal implant assembly may vary
for specific anatomic regions and clinical indications. Occipital-cervical and occipital-cervical-thoracic spinal implant assemblies
contain different types of anchors. Each type of anchor has an intended method of application to the spine. For example, one
assembly may include screws and rods (see Fig. 2), while another assembly may contain screws, hooks, rods, and transverse
elements (see Fig. 3). The block moment arm of a test configuration will be independent of the intended method of application
of a spinal implant assembly, thereby allowing the user to compare devices for a given load. However, it should be noted that the
F2706 − 18
FIG. 7 Cervical Bilateral Polyacetal Block for Hooks, Cables, Wires
same load on different implant assemblies will result in different loading on the implants as a function of implant design. The user
should take this into account when evaluating the performance of a device.
5. Significance and Use
5.1 Occipital-cervical and occipital-cervical-thoracic spinal implants are generally composed of several components which,
when connected together, form either an occipital-cervical spinal implant assembly or an occipital-cervical-thoracic spinal implant
F2706 − 18
Representative of how Figs. 2-7 can be modified to allow for unconstrained motion.
FIG. 8 Alternate Lumbar Bilateral Polyacetal Block for Screws and Bolts
assembly. Occipital-cervical and occipital-cervical-thoracic spinal implant assemblies are designed to provide some stability to the
spine during the process of arthrodesis. These test methods outline standard materials and methods for the evaluation of different
spinal implant assemblies to facilitate comparisons between different designs.
5.2 These test methods are used to quantify the static and dynamic mechanical characteristics of different designs of
occipital-cervical and occipital-cervical-thoracic spinal implant assemblies. The mechanical tests are conducted in vitro using
simplified load schemes and do not attempt to mimic the complex loads of the occipital-cervical and occipital-cervical-thoracic
spine.
5.3 The loads applied to the spinal implant assemblies in vivo will, in general, differ from the loading configurations used in
these test methods. The results obtained here cannot be used directly to predict in vivo performance. The results can be used to
compare different component designs in terms of the relative mechanical parameters.
5.4 Fatigue testing in a simulated body fluid or saline may cause fretting, corrosion, or lubricate the interconnections and thereby
affect the relative performance of tested devices. This test should be initially performed dry (ambient room conditions) for
consistency. The effect of the environment may be significant. Repeating all or part of these test methods in simulated body fluid,
saline (9 g NaCl per 1000 mL water), a saline drip, water, or a lubricant should be considered. The maximum recommended
frequency for this type of cyclic testing should be 5 Hz.
5.5 The location of the longitudinal elements is determined by the intended in vivo location of the anchors. The perpendicular
distance to the load direction between the axis of a hinge pin and the anchor’s attachment points to a polyacetal block is
independent of anchor-type for the cervical block, but dependent on the design for the occipital test block. The distance between
the polyacetal block and the center of the longitudinal element is a function of the design of the implant.
6. Apparatus
6.1 Test machines shall conform to the requirements of Practices E4.
6.2 The test apparatus allows multiple loading regimes to be applied to all forms of occipital-cervical and occipital-cervical-
thoracic spinal implant assemblies. Two pair of side supports are mounted on the test machine (see Fig. 4). One pair of side
supports attach to the actuator and the second to the load cell. A mounting plate for one of the sets of side support plates should
be free to rotate about the Z-axis for the compression bending, tension bending and fatigue tests. Polyacetal blocks are connected
to the side supports via hinge pins, as one possible test method, to facilitate comparison to constructs tested as in Test Methods
F1717. However, it is recommended that unconstrained motion be used as is specified in Test Methods F2077 and is summarized
as follows:
6.2.1 Sphere joint superior-inferior blocks joint blocks, made of polyacetal, are shown in Fig. 8. A 12.7 mm ( ⁄2 in.) diameter
spherical socket is shown to maintain the 12 mm longitudinal distance that was used for the pinned block in Fig. 6. Spherical blocks
are limited to conducting compression-flexion tests.
F2706 − 18
6.2.2 A spherical gimbal superior block with push rod system similar to that used in Test Methods F2077 could be used as an
alternative to perform unconstrained torsion, compression-flexion tests, and tensile-extension tests. See Figure 4, Torsion Testing
Configuration With Pin-Slot Gimbal in Test Methods F2077.
6.2.3 All testing will simulate a vertebrectomy model via a large gap between the two polyacetal blocks. Select the appropriate
design of the polyacetal blocks (see Figs. 2-7 and Figures 2 and 4 of Test Methods F2077) to facilitate testing of the spinal implant
assembly in a manner that simulates the specific clinical indication at the intended spinal location.
6.2.4 Regardless of what testing method is employed, all polyacetal screws are to be inserted 2 mm short of the depth they
would be inserted in vivo. In other words, the screws are to be inserted to the point at which they would just come in contact with
the vertebra (that is, polyacetal blocks) and then backed out 2 mm. Alternatively, insertion points for screws may be counterbored
at a minimum of 300 % of the diameter of the screw (depth of 2 mm) used to purchase the polyacetal. Both methods effectively
prevent biasing the test toward any particular design due to a buttressing effect of the screw against the polyacetal, which could
effectively stress shield the implant from failure that might be experienced in vivo.
6.3 The design of the occipital polyacetal blocks causes the plane through the occipital implant assemblies to be angled at 23°
from the X-Y plane. (This angle from the X-Y plane to the occiput is the average occipitocervical neutral position angle as
determined from human radiological data, n=15.) The design of the cervical polyacetal blocks causes the plane through the spinal
implant assemblies to be parallel to the plane (the Y-Z plane) through the axes of the hinge pins. Align the superior polyacetal block
with the inferior polyacetal block. The center axis of each hinge pin should be perpendicular (60.5°) to and aligned (60.5 mm)
with the load axis of the test machine. Center the test apparatus in the test machine such that the line through the mid-point (0,
0, Z1) of the inferior hinge pin’s axis and the mid-point (0, 0, Z2) of the superior hinge pin’s axis is collinear within 60.1 mm
of the load and rotational axis of the test machine’s actuator. Or, in the case of spherical joint or pin-slot gimbal mechanism, the
points (0, 0, Z1) and (0, 0, Z2) are to be collinear (60.1 mm) with the test machine’s actuator.
6.4 Alternative designs of test blocks may be used as long as equivalence is demonstrated. The solid polyacetal test blocks may
be replaced with metal blocks with polyacetal inserts of appropriate size. Any surface or component of the occipital-cervical or
occipital-cervical-thoracic spinal assembly which would contact the solid polyacetal should also contact an appropriate thickness
of the polyacetal. If screws are used to mount the spinal construct to the test blocks, then the screws shall be placed into polyacetal
inserts in the alternate design of test block. The diameter of the polyacetal inserts shall be equal to or greater than three times the
diameter of the screws.
6.5 If the locations of the superior anchors, inferior anchors, or both sets of anchors are dictated by the longitudinal element
and are at different Z locations (a diagonal), then the set of anchors should be centered above and below the standard location such
that they maintain the average Z location. If the anchors are secured into slots in the longitudinal element, then they should be
centrally placed in the slots and not at either end to produce a worst case scenario.
6.6 Different spinal implant assemblies have different intended methods of application to the polyacetal blocks. The locations
of the longitudinal elements are determined by the design of anchors and interconnections. The load capacity of the spinal construct
is a function of the designs of the interconnections, anchors, and longitudinal elements but should not be a function of the test
apparatus, hence the addition of the 2 mm offset or counterbore when attaching the construct to the blocks (see 6.2).
6.7 The hinge pin in the test configuration allows the same test apparatus to be used for the static compression bending test,
static tensile bending test, and static torsion test as well as the compression bending fatigue test. The polyacetal blocks are allowed
to rotate around the Y-axis of the hinge pin during the compression bending, tensile bending, and fatigue tests.
6.8 If a superior spherical joint is used, the test configuration allows the same test apparatus to be used for the static compression
bending test and the compression bending fatigue test. The polyacetal blocks are free to rotate around the Y-axis, Z-axis, and X-axis
during the compression bending static and fatigue tests.
6.9 If a pin-slot gimbal mechanism is used (Figure 4, Test Methods F2077), the test configuration allows the same test apparatus
to be used for the static compression bending test, static tensile bending test, and static torsion test as well as the compression
bending fatigue test. The polyacetal blocks are free to rotate around the Y-axis of the hinge pin, Z-axis, and X-axis during the
compression bending, tensile bending, and fatigue tests.
6.10 Modified bilateral polyacetal blocks (see Figs. 4 and 5) have been developed for testing occipital components (see Fig. 5).
6.11 Modified bilateral polyacetal blocks (see Fig. 7) have been developed for testing hooks, wires, or cables. Steel roll pins
are placed into the modified blocks such that the outer surfaces of the roll pins are parallel to the front surfaces of the standard
bilateral polyacetal block (see Fig. 7). Hooks, wires, and cables are not fully constrained (semi-rigid) fixation devices because they
cannot transfer bending moments in the three axes. The combination of the rotation of the modified polyacetal block and the
rotation of the hooks, wires, or cables around the steel roll pins means that the test configuration would be a mechanism. Therefore,
the testing of hooks, wires, and cables necessitates that the modified polyacetal block shall not rotate. Eliminate a degree of
freedom by inhibiting rotation about the Y-axis.
Personal correspondence with John Kirkpatrick, M.D., September 21, 2007.
F2706 − 18
6.12 The relative cephalad/caudad location of the hinge pin with respect to the anchor insertion point is intentionally selected
to minimize variation of the block moment arm. Orient the test blocks such that the hinge pins are internal to the anchors during
the tension bending test. Flip each polyacetal block over for the compression bending, torsion, and fatigue tests such that the hinge
pins are external to the anchors. (See Figs. 4-7.)
6.13 The recommended active length of the longitudinal element of the occipital-cervical or occipital-cervical-thoracic spinal
implant system is 76 mm as described in ASTM STP 1431. (See Note 1.) Alternative configurations such as a 196 mm
occipital-cervical-thoracic construct with the inferior block fixed, rather than free to rotate, can be used with justification. The
active length should be constant for all constructs used in comparative testing. In all cases, selection of attachment method (hooks,
screws, etc.) at all points on the construct should match the configuration expected in the worst-case construct indicated for use.
For example, constructs that are indicated for screw fixation to the thoracic
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...