ASTM E2890-21
(Test Method)Standard Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger and Farjas Methods
Standard Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger and Farjas Methods
SIGNIFICANCE AND USE
6.1 This test method is useful for research and development, quality assurance, regulatory compliance and specification-based acceptance.
6.2 The kinetic parameters determined by this method may be used to calculate thermal hazard figures-of-merit according to Practice E1231.
SCOPE
1.1 This test method describes the determination of the kinetic parameters of Arrhenius activation energy and pre-exponential factor using the Kissinger variable heating rate iso-conversion method (1, 2)2 and activation energy and reaction order by the Farjas method (3) for thermally unstable materials. The test method is applicable to the temperature range from 300 K to 900 K (27 °C to 627 °C).
1.2 Both nth order and accelerating reactions are addressed by this method over the range of 0.5 n p n is the nth order reaction order and p is the Avrami reaction order (4). Reaction orders n and p are determined by the Farjas method (3).
1.3 This test method uses the same experimental conditions as Test Method E698. The Flynn/Wall/Ozawa data treatment of Test Method E698 may be simultaneously applied to these experimental results.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2021
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Sep-2019
- Effective Date
- 01-Nov-2015
- Effective Date
- 01-Sep-2015
- Effective Date
- 01-May-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 15-Sep-2013
- Effective Date
- 15-Sep-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Mar-2013
Overview
ASTM E2890-21, titled the Standard Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger and Farjas Methods, is a critical international standard developed by ASTM. This test method provides precise guidance for determining kinetic parameters-such as activation energy, pre-exponential factor, and reaction order-of thermally sensitive materials over a temperature range of 300 K to 900 K (27 °C to 627 °C). The methodology uses Differential Scanning Calorimetry (DSC) with data treatment techniques developed by Kissinger and Farjas, accommodating both nth order and autocatalytic (Avrami) reactions.
This standard supports R&D, quality assurance, regulatory compliance, and specification-based acceptance, serving as a fundamental tool in chemical and material safety evaluation.
Key Topics
- Kinetic Parameter Determination: The standard outlines procedures for calculating Arrhenius activation energy and pre-exponential factors using the Kissinger variable heating rate iso-conversion method, and both activation energy and reaction order using the Farjas method.
- Applicability: Suitable for thermally unstable materials, addressing reactions from nth order (decomposition-style) to accelerating (autocatalytic/crystallization) types.
- Testing with DSC: Relies on differential scanning calorimetry to track exothermic behavior, measuring heat evolution as a function of temperature and time under controlled conditions.
- Data Analysis: Emphasizes linear regression strategies for data from multiple heating rates, ensuring statistical robustness and reproducibility when determining critical kinetic parameters.
- Precision and Reproducibility: Inter- and intra-laboratory precision are quantified, supporting comparable results across different labs and setups.
- Figures-of-Merit: Results may be used for calculating thermal hazard potential per ASTM Practice E1231.
Applications
Adopting ASTM E2890-21 brings practical value in several areas:
- Research & Development: Material scientists and chemists use this method to characterize new compounds, assess their stability, and inform product formulation.
- Quality Assurance: Manufacturers apply kinetic analysis to verify batch-to-batch consistency and stability of thermally reactive materials, ensuring safety and performance.
- Regulatory Compliance: The method aids in meeting legal and regulatory requirements for chemical handling, storage, and transport by providing standardized hazard figures.
- Process Safety: By understanding thermal decomposition and reaction kinetics, organizations can evaluate process risks, improve safety margins, and prevent incidents involving energetic materials.
- Specification & Acceptance Criteria: The test method underpins contractual and supply agreements that specify thermal stability and reactivity requirements for supplied chemicals or materials.
Related Standards
ASTM E2890-21 is part of a broader framework for thermal and kinetic analysis. Key related ASTM standards include:
- ASTM E698: Kinetic Parameters for Thermally Unstable Materials by DSC and the Flynn/Wall/Ozawa Method – Can be used in tandem with E2890.
- ASTM E537: Test Method for Thermal Stability of Chemicals by DSC.
- ASTM E967 & E968: Methods for temperature and heat flow calibration of DSC instruments.
- ASTM E473, E1142, E2161: Terminology standards for thermal analysis and rheology.
- ASTM E1231: Practice for Calculation of Hazard Potential Figures of Merit for Thermally Unstable Materials.
- ASTM E691 & E1970: Practices concerning statistical data treatment and interlaboratory studies.
By aligning with ASTM E2890-21 and its related standards, organizations ensure the adoption of globally recognized practices for the kinetic analysis of thermally unstable substances, leading to improved safety, compliance, and material performance.
Buy Documents
ASTM E2890-21 - Standard Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger and Farjas Methods
REDLINE ASTM E2890-21 - Standard Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger and Farjas Methods
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E2890-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Kinetic Parameters and Reaction Order for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger and Farjas Methods". This standard covers: SIGNIFICANCE AND USE 6.1 This test method is useful for research and development, quality assurance, regulatory compliance and specification-based acceptance. 6.2 The kinetic parameters determined by this method may be used to calculate thermal hazard figures-of-merit according to Practice E1231. SCOPE 1.1 This test method describes the determination of the kinetic parameters of Arrhenius activation energy and pre-exponential factor using the Kissinger variable heating rate iso-conversion method (1, 2)2 and activation energy and reaction order by the Farjas method (3) for thermally unstable materials. The test method is applicable to the temperature range from 300 K to 900 K (27 °C to 627 °C). 1.2 Both nth order and accelerating reactions are addressed by this method over the range of 0.5 n p n is the nth order reaction order and p is the Avrami reaction order (4). Reaction orders n and p are determined by the Farjas method (3). 1.3 This test method uses the same experimental conditions as Test Method E698. The Flynn/Wall/Ozawa data treatment of Test Method E698 may be simultaneously applied to these experimental results. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 6.1 This test method is useful for research and development, quality assurance, regulatory compliance and specification-based acceptance. 6.2 The kinetic parameters determined by this method may be used to calculate thermal hazard figures-of-merit according to Practice E1231. SCOPE 1.1 This test method describes the determination of the kinetic parameters of Arrhenius activation energy and pre-exponential factor using the Kissinger variable heating rate iso-conversion method (1, 2)2 and activation energy and reaction order by the Farjas method (3) for thermally unstable materials. The test method is applicable to the temperature range from 300 K to 900 K (27 °C to 627 °C). 1.2 Both nth order and accelerating reactions are addressed by this method over the range of 0.5 n p n is the nth order reaction order and p is the Avrami reaction order (4). Reaction orders n and p are determined by the Farjas method (3). 1.3 This test method uses the same experimental conditions as Test Method E698. The Flynn/Wall/Ozawa data treatment of Test Method E698 may be simultaneously applied to these experimental results. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2890-21 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2890-21 has the following relationships with other standards: It is inter standard links to ASTM E1142-23b, ASTM E473-23b, ASTM E537-20, ASTM E1231-19, ASTM E1231-15, ASTM E2161-15, ASTM E1142-15, ASTM E1142-14b, ASTM E473-14, ASTM E1142-14a, ASTM E1142-14, ASTM E1860-13, ASTM E2041-13e1, ASTM E691-13, ASTM E2161-13. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2890-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2890 − 21
Standard Test Method for
Determination of Kinetic Parameters and Reaction Order for
Thermally Unstable Materials by Differential Scanning
Calorimetry Using the Kissinger and Farjas Methods
This standard is issued under the fixed designation E2890; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.1 This test method describes the determination of the
kinetic parameters of Arrhenius activation energy and pre-
2. Referenced Documents
exponential factor using the Kissinger variable heating rate
2.1 ASTM Standards:
iso-conversion method (1, 2) and activation energy and
E473Terminology Relating to Thermal Analysis and Rhe-
reaction order by the Farjas method (3) for thermally unstable
ology
materials. The test method is applicable to the temperature
E537Test Method for Thermal Stability of Chemicals by
range from 300 K to 900 K (27 °C to 627 °C).
Differential Scanning Calorimetry
1.2 Both nth order and accelerating reactions are addressed
E691Practice for Conducting an Interlaboratory Study to
by this method over the range of 0.5 < n<4and1< p<4
Determine the Precision of a Test Method
where n is the nth order reaction order and p is the Avrami
E698Test Method for Kinetic Parameters for Thermally
reaction order (4). Reaction orders n and p are determined by
Unstable Materials Using Differential Scanning Calorim-
the Farjas method (3).
etry and the Flynn/Wall/Ozawa Method
E967Test Method for Temperature Calibration of Differen-
1.3 This test method uses the same experimental conditions
tial Scanning Calorimeters and Differential ThermalAna-
asTestMethodE698.TheFlynn/Wall/Ozawadatatreatmentof
lyzers
Test Method E698 may be simultaneously applied to these
E968Practice for Heat Flow Calibration of Differential
experimental results.
Scanning Calorimeters
1.4 The values stated in SI units are to be regarded as
E1142Terminology Relating to Thermophysical Properties
standard. No other units of measurement are included in this
E1231Practice for Calculation of Hazard Potential Figures
standard.
of Merit for Thermally Unstable Materials
1.5 This standard does not purport to address all of the
E1860Test Method for Elapsed Time Calibration of Ther-
safety concerns, if any, associated with its use. It is the
mal Analyzers
responsibility of the user of this standard to establish appro-
E1970PracticeforStatisticalTreatmentofThermoanalytical
priate safety, health, and environmental practices and deter-
Data
mine the applicability of regulatory limitations prior to use.
E2041Test Method for Estimating Kinetic Parameters by
1.6 This international standard was developed in accor-
Differential Scanning Calorimeter Using the Borchardt
dance with internationally recognized principles on standard-
and Daniels Method
ization established in the Decision on Principles for the
E2161Terminology Relating to Performance Validation in
Development of International Standards, Guides and Recom-
Thermal Analysis and Rheology
3. Terminology
ThistestmethodisunderthejurisdictionofASTMCommitteeE37onThermal
3.1 Technical terms used in this test method are defined in
Measurements and is the direct responsibility of Subcommittee E37.01 on Calo-
Terminologies E473, E1142, and E2161. Referenced terms
rimetry and Mass Loss.
Current edition approved Oct. 1, 2021. Published November 2021. Originally
approved in 2012. Last previous approval in 2018 as E2890 – 12 (2018). DOI: For referenced ASTM standards, visit the ASTM website, www.astm.org, or
10.1520/E2890-21. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2890 − 21
include Arrhenius equation, baseline, calibration, Celsius,
p = Avrami reaction order (dimensionless).
differential scanning calorimeter, endotherm, enthalpy, figure-
NOTE 1—There are a large number of conversion function expressions
of-merit, first-deviation-from baseline, full-width-at-half- for f(a) (5). Those described here are the more common ones but are not
the only functions suitable for this method. Eq 2 is known as the Law of
maximum, Kelvin, onset point, peak, peak value, relative
Mass Action (6) while Eq 3 is the Avrami equation (4).
standard deviation, standard deviation, thermal analysis, and
5.3 The Arrhenius equation (7) describes how the reaction
thermal curve.
rate changes as a function of temperature:
4. Summary of Test Method
2E⁄RT
k T 5Ze (4)
~ !
4.1 Aseries of thermally unstable test specimens are heated
where:
at a minimum of four different linear rates in a differential
-1
scanning calorimeter through a region of exothermic reaction Z = pre-exponential factor (s ),
-1
behavior. The rate of heat evolution, created by a chemical E = activation energy (J mol ),
T = absolute temperature (K),
reaction, is proportional to the rate of reaction and is measured
-1 -1
R = gas constant (8.314 J mol K ), and
as a function of temperature and time.
e = natural logarithm base (2.7182818).
4.2 The temperature corresponding to the maximum rate of
5.4 Eq1andEq4maybecombinedtoyieldthegeneralrate
reaction (measured at the heat flow maximum of the exother-
equation:
mic reaction peak) is recorded at each linear heating rate. This
2E⁄RT
observed temperature is corrected for instrument thermal
da⁄dt 5 f α Ze (5)
~ !
resistance. Activation energy and pre-exponential factor are
5.5 As the temperature increases, the rate of reaction will
derived from the linear regression of the natural logarithm of
increase until a maximum is reached and then the rate declines
the heating rate, normalized to the square of the absolute
back to “zero” as the reactant is consumed. When the rate of
temperature,versusthereciprocalabsolutetemperatureofheat
reaction is displayed as a function of increasing temperature,
flow at the peak maximum. The approach is known as the
theshapeofthisresponseiscalleda“peak”.Themathematical
Kissinger method (1, 2).
derivative of the reaction rate at the peak maximum equals
4.3 Areaction type is determined for the specimen from the
zero. Taking the derivative of Eq 5 over time at the maximum
shape of the reaction exotherm under isothermal temperature
point for the heating with constant rate β, then casting in
conditions.
logarithmic form and assuming that ln@d ~f ~α!! ⁄dt#50, leads
to Eq 6.
4.4 Onceareactiontypeisdeterminedkineticparametersof
order (either n or p) are determined using the shape of the
ln β ⁄ T 5 lnZR ⁄ E 2 E⁄RT (6)
@ # @ #
m m
reaction exotherm measured by the time at full-width-at-half-
where:
maximum (t ). This approach is known at the Farjas
FWHM
-1
method (3). The activation energy and reaction order are
β = heating rate (K s ), and
T = temperature a peak maximum (K).
derived from the linear regression of the natural logarithm of
m
NOTE 2—The assumption of ln d f α ⁄dt 50 holds strictly only
thetimeatfull-width-at-half-maximumversusthereciprocalof @ ~ ~ !! #
for 1st order reaction but is considered a “reasonable” approximation for
absolute temperature at maximum reaction rate (heat flow).
other nth order or Avrami reactions.
5. Basis of Methodology
5.6 Eq6isoftheform Y5mX1b.Ifln[β/T ]issetequalto
m
Yand1/T issetequaltoX,thenadisplayofYversusXyields
5.1 For reactions that are exothermic in nature, the rate of
m
heat evolution is proportional to the rate of the reaction. a slope (m ) equal to –E /R and an intercept (b ) equal to
K K K
Differentialscanningcalorimetrymeasurestheheatflowasthe ln[ZR/E ] where Z and E are the pre-exponential factor and
K K
dependent experimental parameter versus temperature (or theactivationenergy,respectively,determinedbytheKissinger
time) as the independent parameter. method.
5.2 Reactions may be modeled with a number of suitable 5.7 The shape of the reaction exothermic peak may be
equations of the form:
characterized by the time at full-width-at-half-maximum
(t ) (3).
FWHM
da⁄dt 5 k T f α (1)
~ ! ~ !
ln t 5 E ⁄RT 1ln t' ⁄ Z (7)
@ # @ #
FWHM F m
where:
-1
where:
da/dt = reaction rate (s ),
α = fraction reacted or conversion (dimensionless),
t = the full-width-at-half-maximum time (s), and
FWHM
k(T) = specific rate constant at temperature T, and
t’ = an arbitrary function.
f(α) = conversion function (dimensionless).
5.8 Eq 7 is of the form Y5mX1b.Ifln[t ] is set equal
FWHM
Commonly used functions include:
to Y and 1/T is set equal to X, then a display of Y versus X
m
n
yieldsaslope(m )equalto E /Randanintercept(b )equalto
f ~α! 5 ~1 2 α! (2)
1 F F F
ln[t’/Z] where E is the activation energy determined by the
~p 2 1!⁄p
F
f ~α! 5 p~1 2 α!@2 ln ~1 2 α!# (3)
Farjas method.
where:
5.9 The reaction order, n or p, is determined through an
n = nth reaction order (dimensionless), and
empirical relationship based on t’.
E2890 − 21
6. Significance and Use any) and that are of suitable structural shape and integrity to
contain the specimen (even under internal pressure developed
6.1 Thistestmethodisusefulforresearchanddevelopment,
during the reaction) and reference in accordance with the
quality assurance, regulatory compliance and specification-
specific requirements of this test method.
based acceptance.
NOTE3—Manyusersfindglass,goldorgoldcoatedhermeticallysealed
6.2 The kinetic parameters determined by this method may
containers of low headspace volume advantageous for testing with high
energy materials. The selected container shall meet the necessary internal
be used to calculate thermal hazard figures-of-merit according
pressure rating to withstand internal pressure buildup.
to Practice E1231.
8.3 Ameans, tool or device to close, encapsulate or seal the
7. Interferences
container of choice.
7.1 This test method assumes a single reaction mechanism
8.4 Analytical Balance with a capacity of at least 100 mg to
constant over the reaction conversion temperature range of the
weigh specimens or containers, or both to within 610 µg.
material under evaluation. Some overall reactions of interest
8.5 Auxiliary instrumentation considered useful but not
are known to include a series of competing reaction mecha-
essential for conducting this method would include cooling
nisms that lead to changes in reaction order with conversion
capability to hasten cooling to ambient temperature conditions
(8). This method addresses the reaction only at a single
at the end of the test.
conversion value at the maximum reaction rate — often about
0.7.
9. Hazards
7.2 Method precision is enhanced with the selection of the
9.1 This test method is used to determine the properties of
appropriate conversion function [f(α)]. The shape of the ther-
thermally reactive materials. The user of this test method shall
mal curve, as described in 11.2, may confirm the selection of
use the smallest quantity of material (typically a few milli-
the nth order or accelerating reaction models.
grams) needed to obtain the desired analytical results.
7.2.1 Typically nth reactions include many (but not all)
9.2 Special precautions shall be taken to protect personnel
decompositionreactionsorthosewhereoneoftheparticipating
andequipmentwhentheapparatusinuserequirestheinsertion
species is in excess.
ofspecimensintoaheatedfurnace.Typicalspecialprecautions
7.2.2 Typical accelerating (Avrami) reactions include ther-
include adequate shielding, ventilation of equipment and face
moset cure, crystallization, and some pyrotechnic reactions.
and hand protection for users.Asafety analysis prior to testing
7.3 Since this method uses milligram quantities of material,
is recommended.
it is essential for the test specimens to be homogeneous and
representative of the larger sample from which they are taken.
10. Calibration and Standardization
7.4 Acriticalliteratureevaluationofkineticmethodsreports
10.1 Perform any calibration procedures recommended by
that the Kissinger method is the most accurate method for
the manufacturer as described in the operator’s manual to
determining activation energy in many cases (9).
ensurethattheapparatusiscalibratedateachheatingrateused.
8. Apparatus
10.2 Calibrate the heat flow signal using 99.99+ % indium,
Practice E968, and the same type of specimen container to be
8.1 DifferentialScanningCalorimeter(DSC)—Theessential
used in the subsequent test for kinetic parameters.
instrumentation required to provide the minimum differential
scanning calorimetric capability for this method includes (a) a
10.3 Calibrate the temperature signal using 99.99+ %
furnace(s) to provide uniform controlled heating or cooling of
indium, Test Method E967, and the same type of specimen
a specimen and reference to a constant temperature or at a
containerandheatingratestobeusedinthesubsequenttestfor
constant rate over the range of 300 K to 900 K, (b) a
kinetic parameters.
temperature sensor for the range of 100 K to 900 K and
10.4 Calibrate the elapsed time signal using Test Method
readable to 0.01 K to measure the assumed temperature of the
E1860.
test specimen, (c) differential sensors to detect a heat flow
10.5 Determine the thermal resistance (φ) from the leading
difference between the specimen and reference with a range of
edge slope ~S 5 ∆ q ⁄ ∆ T! in (mW/K) of the indium melting
100 mW readable to 61µW, (d) a means of sustaining a test
endotherm as shown in Fig. 1 and 12.1.
chamber environment of inert purge gas at a purge rate of 10
mL/min to 100 mL/min within 65 mL/min, (e) a temperature
11. Procedure
controller,capableofexecutingaspecifictemperatureprogram
byoperatingthefurnace(s)betweenselectedtemperaturelimits 11.1 Scouting Experiment:
over the range of ambient to 900 K (627 °C) at a rate of 0.1 11.1.1 Using a 1mg to 5 mg test specimen, weighed to a
K/min to 20 K/min constant to 1% or at an isothermal precision of 60.1 mg, perform a scouting experiment using
temperature constant to 0.1 K, (f) a data collection device,to Test Method E537 to determine the temperature of first-
provideameansofacquiring,storing,anddisplayingmeasured deviation-from baseline (T ) and the heat of reaction (∆H).
o
or calculated signals or both. The minimum output signals
11.2 Determination of Reaction Type:
required are heat flow, temperature, and time.
11.2.1 Weighintoaspecimencontainer1mgto5mgofthe
8.2 Containers (pans, crucibles, vials, lids, closures, seals, test specimen, with a precision of 60.1 mg, and hermetically
etc.) that are inert to the specimen and reference materials (if seal the container. DO NOT load the test specimen into the
E2890 − 21
FIG. 1 Determination of Leading Edge Slope and Thermal Resistance
apparatus. Load an empty specimen container into the refer- equipment (such as heat resistant gloves and face shield) shall
ence chamber. Close the DSC chamber and prepare the be used to ensure the safety of the operator.)
apparatus for an experimental run.
11.2.5 Evaluate the shape of the resultant thermal curve and
11.2.2 Select an isothermal test temperature corresponding
determine whether the reaction is nth order or accelerating.An
to 10% peak area (∆H) from the scouting experiment per-
nth order reaction is likely when the heat flow curve reaches a
formed in 11.1. Equilibrate the apparatus for 1 min at this test
maximum within seconds (nearly immediately) after the speci-
temperature.
menisloadedintotheapparatusthenslowlyreturnstobaseline
11.2.3 Initiate the experiment, recording heat flow as a
heat flow as shown in Fig. 2.An accelerating reaction is likely
function of time.
when heat flow builds to a maximum (after tens of seconds)
11.2.4 Open the DSC sample chamber and quickly load the
and then decays, as shown in Fig. 3.
test specimen container into the apparatus. Immediately close
11.3 Procedure for Measuring Kinetic Parameters:
the sample chamber. Record the thermal curve for 20 min or
untiltheexothermiceventiscompleteandtherateofheatflow 11.3.1 Weigh 1 mg to 5 mg of the test specimen to a
precision of 610 µg into a tarred sample container and
approaches zero. (Warning—Burn hazard. The sample
chamber, heat shield and covers present a burn hazard to the hermetically seal the container. Weigh the specimen and
operator.Exercisegreatcareinthisoperation.Protectivesafety containerto 610µg.Loadthetestspecimenintotheapparatus.
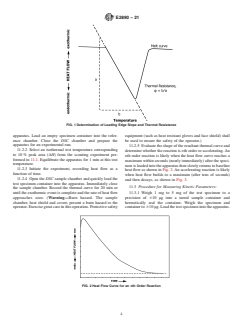
FIG. 2 Heat Flow Curve for an nth Order Reaction
E2890 − 21
FIG. 3 Heat Flow Curve for an Accelerating Reaction
Prepareanequivalentempty,sealedspecimencontainerforuse 11.3.8 Correct the observed temperature (T) using Eq 9 and
as the reference container. Close the DSC sample chamber and the thermal resistance (φ) value determined in 10.5 to deter-
prepare the apparatus for an experimental run. mine T .
m
11.3.2 Equilibrate the specimen for 1 min at a temperature 11.3.9 Determinetheheatingratethroughthereactionusing
(T ); that is, 10 min below the first-deviation-from-baseline T, T, t, and t from 11.3.6 and Eq 10.
s i f i f
temperature (T ) observed in 11.1. 11.3.10 Display the reaction exotherm and baseline of
o
11.3.6 versus time on the X-axis.
NOTE 4—Starting temperature T 5T 2~10 min 3 β! where β is
s o
the selected heating rate in K/min.
NOTE 7—Modern differential scanning calorimeters may easily trans-
form the thermal curve between temperature and time.
11.3.3 Heat the test specimen at a rate of 1 K/min to a
11.3.11 From the baseline, determine the heat flow (q )
temperature 10 min higher than the completion of the exother- max
corresponding to the maximum of the reaction exotherm.
mic reaction as indicated by the return to baseline. Record the
11.3.12 Determinetheelapsedtimebetweentheleadingand
heatflow,time,andsampletemperaturethroughoutthisregion.
trailing edges of the reaction exothermic peak (t ) corre-
FWHM
NOTE 5—If the heat flow at peak maximum is greater than 8 mW,
sponding to the value of 0.5 of the maximum heat flow (q )
max
discard the result and re-examine the material using a smaller specimen
as shown in Fig. 4.
size. Specimens with heat flow greater than 8 mW may not be uniform in
11.3.13 Repeat steps 11.3.1 to 11.3.12 using at least three
temperature and may produce erroneous results.
NOTE 6—Other heating rates may be used but shall have a maximum additional heating rates.
heat flow of less than 8 mW and shall be reported.
NOTE 8—A minimum of four determinations at heating rates typically
11.3.4 Cool the specimen container to ambient temperature
between1K/minand10K/minarerecommended.Additionalreplicatesor
heating rates (up to eight) will improve precision of the determinations.
and reweigh. Record and report any change in mass greater
Heating rates less than 1 K/min are thought to be more reliable for the
than 3% as from the initial mass from 11.3.1.
determination of kinetic values as self heating is avoided.
11.3.5 Prepare a display of the heat flow on the ordinate
11.4 Usingthefour(ormore)resultsfrom11.3.1to11.3.12,
(Y-axis) versus temperature on the abscissa (X-axis).
determine and report the Kissinger activation energy (E ), and
K
11.3.6 Construct a linear baseline under the reaction exo-
its standard deviation (σE ), the logarithm of the pre-
therm connecting a point on the baseline immediately before K
exponential factor (ln[Z]), its standard deviation (σ(ln[Z])), the
the reaction exotherm to a point on the baseline immediately
Farjas activation energy (E ), its standard deviation (σE ), the
afterthereactionexotherm.Determinethetemperature(T)and F F
i
reaction order (n or p) and its standard deviation (σ or σ )
n p
time (t) for the point on the baseline before the reaction and
i
using the calculations of Section 12.
temperature (T) and time (t) for the point on the baseline after
f f
the reaction. 11.5 Thevaluesof E and E shouldbethesamewithinthe
K F
11.3.7 From the baseline, determine the temperature (T) determined experimental error. If they are not, then the
value corresponding to the heat flow maximum of the reaction determination of E and/or reaction order n or p should be
F
exotherm. considered suspect.
E2890 − 21
FIG. 4 Determination of the Time at Full-Width-at-Half-Maximum
12. Calculations 12.4 Determination of Kissinger Activation Energy (E ):
K
12.4.1 From the heating rate (β) experimental result from
12.1 Determination of Thermal Resistance:
11.3, and the corrected temperature from 12.2, determine the
12.1.1 Prepare a display of the melting endotherm for the
values for ln[β/T ] and 1/T for each thermal (heating rate)
temperature calibration in 10.3 with heat flow on the ordinate m m
curve obtained.
(Y-axis) and temperature on the abscissa (X-axis) (see Fig. 1).
12.1.2 Determine the t
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2890 − 12 (Reapproved 2018) E2890 − 21
Standard Test Method for
Determination of Kinetic Parameters and Reaction Order for
Thermally Unstable Materials by Differential Scanning
Calorimetry Using the Kissinger Methodand Farjas
Methods
This standard is issued under the fixed designation E2890; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method describes the determination of the kinetic parameters of Arrhenius activation energy and pre-exponential
factor using the Kissinger variable heating rate iso-conversion method (1, 2) and activation energy and reaction order by the Farjas
method (3) for thermally unstable materials. The test method is applicable to the temperature range from 300 K to 900 K (27 °C
to 627°C).627 °C).
1.2 Both nth order and accelerating reactions are addressed by this method over the range of 0.5 < n < 4 and 1 < p < 4 where n
is the nth order reaction order and p is the Avrami reaction order (4). Reaction orders n and p are determined by the Farjas method
(3).
1.3 This test method uses the same experimental conditions as Test Method E698. The Flynn/Wall/Ozawa data treatment of Test
Method E698 may be simultaneously applied to these experimental results.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 There is no ISO equivalent to this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.01 on Calorimetry
and Mass Loss.
Current edition approved April 1, 2018Oct. 1, 2021. Published May 2018November 2021. Originally approved in 2012. Last previous approval in 20122018 as E2890
– 12. 12 (2018). DOI: 10.1520/E2890-12R18.10.1520/E2890-21.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2890 − 21
E473 Terminology Relating to Thermal Analysis and Rheology
E537 Test Method for Thermal Stability of Chemicals by Differential Scanning Calorimetry
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E698 Test Method for Kinetic Parameters for Thermally Unstable Materials Using Differential Scanning Calorimetry and the
Flynn/Wall/Ozawa Method
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E968 Practice for Heat Flow Calibration of Differential Scanning Calorimeters
E698 Test Method for Kinetic Parameters for Thermally Unstable Materials Using Differential Scanning Calorimetry and the
Flynn/Wall/Ozawa Method
E1142 Terminology Relating to Thermophysical Properties
E1231 Practice for Calculation of Hazard Potential Figures of Merit for Thermally Unstable Materials
E1860 Test Method for Elapsed Time Calibration of Thermal Analyzers
E1970 Practice for Statistical Treatment of Thermoanalytical Data
E2041 Test Method for Estimating Kinetic Parameters by Differential Scanning Calorimeter Using the Borchardt and Daniels
Method
E2161 Terminology Relating to Performance Validation in Thermal Analysis and Rheology
3. Terminology
3.1 Technical terms used in this test method are defined in Terminologies E473, E1142, and E2161. Referenced terms include
Arrhenius equation, baseline, calibration, Celsius, differential scanning calorimeter, endotherm, enthalpy, figure-of-merit,
first-deviation-from baseline, full-width-at-half-maximum, Kelvin, onset point, peak, peak value, relative standard deviation,
standard deviation, thermal analysis, and thermal curve.
4. Summary of Test Method
4.1 A series of thermally unstable test specimens are heated at a minimum of four different linear rates in a differential scanning
calorimeter through a region of exothermic reaction behavior. The rate of heat evolution, created by a chemical reaction, is
proportional to the rate of reaction and is measured as a function of temperature and time.
4.2 The temperature corresponding to the maximum rate of reaction (measured at the heat flow maximum of the exothermic
reaction peak) is recorded at each linear heating rate. This observed temperature is corrected for instrument thermal resistance.
Activation energy and pre-exponential factor are derived from the linear regression of the natural logarithm of the heating rate,
normalized to the square of the absolute temperature, versus the reciprocal absolute temperature of heat flow at the peak maximum.
The approach is known as the Kissinger method (1, 2).
4.3 A reaction type is determined for the specimen from the shape of the reaction exotherm under isothermal temperature
conditions.
4.4 Once a reaction type is determined kinetic parameters of order (either n or p) are determined using the shape of the reaction
exotherm measured by the time at full-width-at-half-maximum (t ). This approach is known at the Farjas method (3). The
FWHM
activation energy and reaction order are derived from the linear regression of the natural logarithm of the time at
full-width-at-half-maximum versus the reciprocal of absolute temperature at maximum reaction rate (heat flow).
5. Basis of Methodology
5.1 For reactions that are exothermic in nature, the rate of heat evolution is proportional to the rate of the reaction. Differential
scanning calorimetry measures the heat flow as the dependent experimental parameter versus temperature (or time) as the
independent parameter.
5.2 Reactions may be modeled with a number of suitable equations of the form:
da⁄dt 5 k T f α (1)
~ ! ~ !
where:
-1
da/dt = reaction rate (s ),
E2890 − 21
α = fraction reacted or conversion (dimensionless),
k(T) = specific rate constant at temperature T, and
f(α) = conversion function (dimensionless).
Commonly used functions include:
n
f ~α! 5 ~1 2 α! (2)
p 2 1 ⁄p
~ !
f α 5 p 1 2 α 2 ln 1 2 α (3)
~ ! ~ !@ ~ !#
where:
n = nth reaction order (dimensionless), and
p = Avrami reaction order (dimensionless).
NOTE 1—There are a large number of conversion function expressions for f(a)(5). Those described here are the more common ones but are not the only
functions suitable for this method. Eq 2 is known as the Law of Mass Action (6) while Eq 3 is the Avrami equation (4).
5.3 The Arrhenius equation (7) describes how the reaction rate changes as a function of temperature:
2E⁄RT
k T 5 Ze (4)
~ !
where:
-1
Z = pre-exponential factor (s ),
-1
E = activation energy (J mol ),
T = absolute temperature (K),
-1 -1
R = gas constant (8.314 J mol K ), and
e = natural logarithm base (2.7182818).
5.4 Eq 1 and Eq 4 may be combined to yield the general rate equation:
2E⁄RT
da⁄dt 5 f~α!Ze (5)
5.5 As the temperature increases, the rate of reaction will increase until a maximum is reached and then the rate declines back to
“zero” as the reactant is consumed. When the rate of reaction is displayed as a function of increasing temperature, the shape of
this response is called a “peak”. The mathematical derivative of the reaction rate at the peak maximum equals zero. Taking the
derivative of Eq 5 over time at the maximum point for the heating with constant rate β, then casting in logarithmic form and
assuming that ln@d f α ⁄ d t#50, leads to Eq 6.
~ ~ !!
ln β ⁄ T 5 ln Z R ⁄ E 2 E⁄RT (6)
@ # @ #
m m
where:
-1
β = heating rate (K s ), and
T = temperature a peak maximum (K).
m
NOTE 2—The assumption of ln@d ~f ~α!! ⁄ d t#50 holds strictly only for 1st order reaction but is considered a “reasonable” approximation for other nth
order or Avrami reactions.
5.6 Eq 6 is of the form Y5mX1b. If ln[β/T ] is set equal to Y and 1/T is set equal to X, then a display of Y versus X yields
m m
a slope (m ) equal to –E /R and an intercept (b ) equal to ln[ZR/E ] where Z and E are the pre-exponential factor and the
K K K K K
activation energy, respectively, determined by the Kissinger method.
5.7 The shape of the reaction exothermic peak may be characterized by the time at full-width-at-half-maximum (t )
fwhmFWHM
(3).
ln@t # 5 E ⁄RT 1ln@t ' ⁄ Z# (7)
FWHM F m
ln t 5 E ⁄RT 1ln t' ⁄ Z (7)
@ # @ #
FWHM F m
where:
t = the full-width-at-half-maximum time (s), and
fwhm
E2890 − 21
-1
t’ = an arbitrary function (s ).
t = the full-width-at-half-maximum time (s), and
FWHM
t’ = an arbitrary function.
5.8 Eq 7 is of the form Y5mX1b. If ln[t ] is set equal to Y and 1/T is set equal to X, then a display of Y versus X yields
FWHM m
a slope (m ) equal to E /R and an intercept (b ) equal to ln[t’/Z] where E is the activation energy determined by the Farjas method.
F F F F
5.9 The reaction order, n or p, is determined through an empirical relationship based on t’.
6. Significance and Use
6.1 This test method is useful for research and development, quality assurance, regulatory compliance and specification-based
acceptance.
6.2 The kinetic parameters determined by this method may be used to calculate thermal hazard figures-of-merit according to
Practice E1231.
7. Interferences
7.1 This test method assumes a single reaction mechanism constant over the reaction conversion temperature range of the material
under evaluation. Some overall reactions of interest are known to include a series of competing reaction mechanisms that lead to
changes in reaction order with conversion (8). This method addresses the reaction only at a single conversion value at the
maximum reaction rate—often rate — often about 0.7.
7.2 Method precision is enhanced with the selection of the appropriate conversion function [f(α)]. The shape of the thermal curve,
as described in 11.2, may confirm the selection of the nth order or accelerating reaction models.
7.2.1 Typically nth reactions include many (but not all) decomposition reactions or those where one of the participating species
is in excess.
7.2.2 Typical accelerating (Avrami) reactions include thermoset cure, crystallization, and some pyrotechnic reactions.
7.3 Since this method uses milligram quantities of material, it is essential for the test specimens to be homogeneous and
representative of the larger sample from which they are taken.
7.4 A critical literature evaluation of kinetic methods reports that the Kissinger method is the most accurate method for
determining activation energy in many cases (9).
8. Apparatus
8.1 Differential Scanning Calorimeter (DSC)—The essential instrumentation required to provide the minimum differential
scanning calorimetric capability for this method includes (a) a furnace(s) to provide uniform controlled heating or cooling of a
specimen and reference to a constant temperature or at a constant rate over the range of 300 K to 900 K, (b) a temperature sensor
to provide a measurement of the specimen temperature to 60.01 K, for the range of 100 K to 900 K and readable to 0.01 K to
measure the assumed temperature of the test specimen, (c)differential sensors to detect a heat flow difference between the specimen
and reference with a range of 100 mW readable to 61μW, 61 μW, (d) a means of sustaining a test chamber environment of inert
purge gas at a purge rate of 10 mL/min to 100 mL/min within 65 mL/min, (e) a temperature controller, capable of executing a
specific temperature program by operating the furnace(s) between selected temperature limits over the range of ambient to 900 K
(627°C) (627 °C) at a rate of 0.1 K/min to 20 K/min constant to 1 % or at an isothermal temperature constant to 0.1 K, (f) a data
collection device, to provide a means of acquiring, storing, and displaying measured or calculated signals or both. The minimum
output signals required are heat flow, temperature, and time.
8.2 Containers (pans, crucibles, vials, lids, closures, seals, etc.) that are inert to the specimen and reference materials (if any) and
that are of suitable structural shape and integrity to contain the specimen (even under internal pressure developed during the
reaction) and reference in accordance with the specific requirements of this test method.
E2890 − 21
NOTE 3—Many users find glass, gold or gold coated hermetically sealed containers of low headspace volume advantageous for testing with high energy
materials. The selected container shall meet the necessary internal pressure rating to withstand internal pressure buildup.
8.3 A means, tool or device to close, encapsulate or seal the container of choice.
8.4 Analytical Balance with a capacity of at least 100 mg to weigh specimens or containers, or both to within 610 μg.
8.5 Auxiliary instrumentation considered useful but not essential for conducting this method would include cooling capability to
hasten cooling to ambient temperature conditions at the end of the test.
9. Hazards
9.1 This test method is used to determine the properties of thermally reactive materials. The user of this test method shall use the
smallest quantity of material (typically a few milligrams) needed to obtain the desired analytical results.
9.2 Special precautions shall be taken to protect personnel and equipment when the apparatus in use requires the insertion of
specimens into a heated furnace. Typical special precautions include adequate shielding, ventilation of equipment and face and
hand protection for users. A safety analysis prior to testing is recommended.
10. Calibration and Standardization
10.1 Perform any calibration procedures recommended by the manufacturer as described in the operator’s manual to ensure that
the apparatus is calibrated at each heating rate used.
10.2 Calibrate the heat flow signal using 99.99+ % indium, Practice E968, and the same type of specimen container to be used
in the subsequent test for kinetic parameters.
10.3 Calibrate the temperature signal using 99.99+ % indium, Test Method E967, and the same type of specimen container and
heating rates to be used in the subsequent test for kinetic parameters.
10.4 Calibrate the elapsed time signal using Test Method E1860.
10.5 Determine the thermal resistance (φ) from the leading edge slope S 5 Δ q ⁄ Δ T in (mW/K) of the indium melting endotherm
~ !
as shown in Fig. 1 and 12.1.
11. Procedure
11.1 Scouting Experiment:
11.1.1 Using a 11mg to 5 mg test specimen, weighed to a precision of 60.1 mg, perform a scouting experiment using Test Method
E537 to determine the temperature of first-deviation-from baseline (T ) and the heat of reaction (ΔH).
o
11.2 Determination of Reaction Type:
11.2.1 Weigh into a specimen container 1 mg to 5 mg of the test specimen, with a precision of 60.1 mg, and hermetically seal
the container. DO NOT load the test specimen into the apparatus. Load an empty specimen container into the reference chamber.
Close the DSC chamber and prepare the apparatus for an experimental run.
11.2.2 Select an isothermal test temperature corresponding to 10 % peak area (ΔH) from the scouting experiment performed in
11.1. Equilibrate the apparatus for 1 min at this test temperature.
11.2.3 Initiate the experiment, recording heat flow as a function of time.
11.2.4 Open the DSC sample chamber and quickly load the test specimen container into the apparatus. Immediately close the
sample chamber. Record the thermal curve for 20 min or until the exothermic event is complete and the rate of heat flow
E2890 − 21
FIG. 1 Determination of Leading Edge Slope and Thermal Resistance
approaches zero. (Warning—Burn hazard. The sample chamber, heat shield and covers present a burn hazard to the operator.
Exercise great care in this operation. Protective safety equipment (such as heat resistant gloves and face shield) shall be used to
ensure the safety of the operator.)
11.2.5 Evaluate the shape of the resultant thermal curve and determine whether the reaction is nth order or autocatalytic.accel-
erating. An nth order reaction is likely when the heat flow curve reaches a maximum within seconds (nearly immediately) after
the specimen is loaded into the apparatus then slowly returns to baseline heat flow as shown in Fig. 2. An accelerating reaction
is likely when heat flow builds to a maximum (after tens of seconds) and then decays, as shown in Fig. 3.
11.3 Procedure for Measuring Kinetic Parameters:
11.3.1 Weigh 1 mg to 5 mg of the test specimen to a precision of 610 μg into a tarred sample container and hermetically seal the
container. Weigh the specimen and container to 610 μg. Load the test specimen into the apparatus. Prepare an equivalent empty,
sealed specimen container for use as the reference container. Close the DSC sample chamber and prepare the apparatus for an
experimental run.
11.3.2 Equilibrate the specimen for 1 min at a temperature (T ); that is, 10 min below the first-deviation-from-baseline temperature
s
(T ) observed in 11.1.
o
FIG. 2 Heat Flow Curve for an nth Order Reaction
E2890 − 21
FIG. 3 Heat Flow Curve for an Accelerating Reaction
NOTE 4—Starting temperature T 5T 2 10 min 3 β where β is the selected heating rate in K/min.
~ !
s o
-1
11.3.3 Heat the test specimen at a rate of 1 K minK/min to a temperature 10 min higher than the completion of the exothermic
reaction as indicated by the return to baseline. Record the heat flow, time, and sample temperature throughout this region.
NOTE 5—If the heat flow at peak maximum is greater than 8 mW, discard the result and re-examine the material using a smaller specimen size. Specimens
with heat flow greater than 8 mW may not be uniform in temperature and may produce erroneous results.
NOTE 6—Other heating rates may be used but shall have a maximum heat flow of less than 8 mW and shall be reported.
11.3.4 Cool the specimen container to ambient temperature and reweigh. Record and report any change in mass greater than 3 %
as from the initial mass from 11.3.1.
11.3.5 Prepare a display of the heat flow on the ordinate (Y-axis) versus temperature on the abscissa (X-axis).
11.3.6 Construct a linear baseline under the reaction exotherm connecting a point on the baseline immediately before the reaction
exotherm to a point on the baseline immediately after the reaction exotherm. Determine the temperature (T ) and time (t ) for the
i i
point on the baseline before the reaction and temperature (T ) and time (t ) for the point on the baseline after the reaction.
f f
11.3.7 From the baseline, determine the temperature (T) value corresponding to the heat flow maximum of the reaction exotherm.
11.3.8 Correct the observed temperature (T) using Eq 9 and the thermal resistance (φ) value determined in 10.410.5 to determine
T .
m
11.3.9 Determine the heating rate through the reaction using T ,T ,t , and t from 11.3.6 and Eq 10.
i f i f
11.3.10 Display the reaction exotherm and baseline of 11.3.6 versus time on the X-axis.
NOTE 7—Modern differential scanning calorimeters may easily transform the thermal curve between temperature and time.
11.3.11 From the baseline, determine the heat flow (q ) corresponding to the maximum of the reaction exotherm.
max
11.3.12 Determine the elapsed time between the leading and trailing edges of the reaction exothermic peak (t ) corresponding
FWHM
to the value of 0.5 of the maximum heat flow (q ) as shown in Fig. 4.
max
E2890 − 21
FIG. 4 Determination of the Time at Full-Width-at-Half-Maximum
11.3.13 Repeat steps 11.3.1 to 11.3.12 using at least three additional heating rates.
NOTE 8—A minimum of four determinations at heating rates typically between 1 K/min and 10 K/min are recommended. Additional replicates or heating
rates (up to eight) will improve precision of the determinations. Heating rates less thatthan 1 K/min are thought to be more reliable for the determination
of kinetic values.values as self heating is avoided.
11.4 Using the four (or more) results from 11.3.1 to 11.3.12, determine and report the Kissinger activation energy (E ), and its
K
standard deviation (σE ), the logarithm of the pre-exponential factor (ln[Z]), its standard deviation (σ(ln[Z])), the Farjas activation
K
energy (E ), its standard deviation (σE ), the reaction order (n or p) and its standard deviation (σ or σ ) using the calculations
F F n p
of Section 12.
11.5 The values of E and E should be the same within the determined experimental error. If they are not, then the determination
K F
of E and/or reaction order n or p should be considered suspect.
F
12. Calculations
12.1 Determination of Thermal Resistance:
12.1.1 Prepare a display of the melting endotherm for the temperature calibration in 10.3 with heat flow on the ordinate (Y-axis)
and temperature on the abscissa (X-axis) (see Fig. 1).
12.1.2 Determine the thermal resistance (φ) from the tangent to the point of greatest leading edge slope using Eq 8 (see Fig. 1).
φ 5 ΔT⁄Δq 5 1⁄S (8)
where:
ΔT = change in temperature (K),
Δq = change in heat flow (mW),
ϕ = thermal resistance (K/mW), and
S = the leading edge slope (mW/K).
NOTE 9—The values of Δq and φ are negative due to the endothermic transition.
12.2 Correct Observed Peak Temperatures for Thermal Resistance:
12.2.1 Using the thermal curve obtained in 11.3, construct a linear baseline by connecting the two points before and after the
reaction exotherm that first deviates from baseline. (See Fig. 4.)
12.2.2 Determine the maximum heat flow (q ) on the reaction exotherm above the baseline.
max
E2890 − 21
12.2.3 Determine the temperature point (T) on the reaction ex
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...