ASTM F2605-16
(Test Method)Standard Test Method for Determining the Molar Mass of Sodium Alginate by Size Exclusion Chromatography with Multi-angle Light Scattering Detection (SEC-MALS)
Standard Test Method for Determining the Molar Mass of Sodium Alginate by Size Exclusion Chromatography with Multi-angle Light Scattering Detection (SEC-MALS)
SIGNIFICANCE AND USE
4.1 The composition and sequential structure of alginate, as well as the molar mass and molar mass distribution, determines the functionality of alginate in an application. For instance, the gelling properties of an alginate are highly dependent upon the composition and molar mass of the polymer.
4.2 Light scattering is one of very few methods available for the determination of absolute molar mass and structure, and it is applicable over the broadest range of molar masses of any method. Combining light scattering detection with size exclusion chromatography (SEC), which sorts molecules according to size, gives the ability to analyze polydisperse samples, as well as to obtain information on branching and molecular conformation. This means that both the number-average and mass-average values for molar mass and size may be obtained for most samples. Furthermore, one has the ability to calculate the distributions of the molar masses and sizes.
4.3 Multi-angle laser light scattering (MALS) is a technique where measurements are made simultaneously over a range of different angles and used to determine the scattering at 0°, which directly relates to molecular weight. MALS detection can be used to obtain information on molecular size, since this parameter is determined by the angular variation of the scattered light. This can be related to branching, aggregation, and molecular conformation. Molar mass can also be determined by detecting scattered light at a single low angle (LALS) and assuming that this is not significantly different from the scattering at 0°.
4.4 Size exclusion chromatography uses columns, which are typically packed with polymer particles containing a network of uniform pores into which solute and solvent molecules can diffuse. While in the pores, molecules are effectively trapped and removed from the flow of the mobile phase. The average residence time in the pores depends upon the size of the solute molecules. Molecules that are larger than the ...
SCOPE
1.1 This test method covers the determination of the molar mass (typically expressed as grams/mole) of sodium alginate intended for use in biomedical and pharmaceutical applications as well as in tissue-engineered medical products (TEMPs) by size exclusion chromatography with multi-angle laser light scattering detection (SEC-MALS). A guide for the characterization of alginate has been published as Guide F2064.
1.2 Alginate used in TEMPs should be well characterized, including the molar mass and polydispersity (molar mass distribution) in order to ensure uniformity and correct functionality in the final product. This test method will assist end users in choosing the correct alginate for their particular application. Alginate may have utility as a scaffold or matrix material for TEMPs, in cell and tissue encapsulation applications, and in drug delivery formulations.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2016
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 01-May-2016
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Mar-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Sep-2003
- Effective Date
- 10-Nov-2000
- Effective Date
- 01-May-2016
Overview
ASTM F2605-16 is the Standard Test Method for Determining the Molar Mass of Sodium Alginate by Size Exclusion Chromatography with Multi-angle Light Scattering Detection (SEC-MALS), published by ASTM International. This standardized method is designed specifically for assessing the molar mass and molar mass distribution of sodium alginate, particularly for biomedical, pharmaceutical, and tissue-engineered medical product (TEMP) applications. Proper characterization of sodium alginate is essential, as its composition and molecular structure directly affect its performance in various industry applications, including gelling, encapsulation, and drug delivery.
Key Topics
- Molar Mass Determination: The standard outlines precise procedures for using SEC-MALS to obtain accurate molar mass and distribution data for sodium alginate. It supports the reliable calculation of both number-average (Mn) and mass-average (Mw) molar masses, ensuring comprehensive polymer characterization.
- Light Scattering Techniques: SEC-MALS combines size exclusion chromatography, which separates molecules by size, and multi-angle laser light scattering, which measures molecular weight and conformation. This powerful pairing provides absolute molar mass without relying on column calibration standards.
- Functional Property Insights: The method enables detailed understanding of branching, aggregation, and molecular conformation-all crucial for optimal material selection and end-use performance.
- Polydispersity Analysis: Assessment of molar mass distribution (polydispersity) helps confirm consistency, purity, and predictability of sodium alginate as an input for medical and pharmaceutical products.
Applications
Sodium alginate characterization using ASTM F2605-16 offers critical value across several high-impact sectors:
- Biomedical Devices and Scaffolds: In tissue engineering and regenerative medicine, alginate’s molar mass influences scaffold formation, mechanical stability, and biocompatibility. Uniform characterization ensures reliable scaffold properties for supporting cell growth and tissue formation.
- Pharmaceutical Formulations: The standard supports precise selection of alginate for use as excipients, matrix materials, or delivery agents in drug formulations. Knowing the molar mass helps optimize drug release rates and product stability.
- Cell and Tissue Encapsulation: Accurate measurement of molar mass and distribution is vital for applications where alginate is used to encapsulate living cells or tissues, such as in cell therapy, artificial organs, and immunoisolation devices.
- Quality Assurance and Regulatory Compliance: Routine use of this test method helps manufacturers document material consistency, meet regulatory expectations, and maintain product safety and efficacy.
Related Standards
For a comprehensive approach to sodium alginate evaluation, users may reference the following documents alongside ASTM F2605-16:
- ASTM F2064: Guide for Characterization and Testing of Alginates as Starting Materials Intended for Use in Biomedical and Tissue Engineered Medical Product Applications.
- ASTM F2315: Guide for Immobilization or Encapsulation of Living Cells or Tissue in Alginate Gels.
- USP <621>: Chromatography - A general chapter in the United States Pharmacopeia/National Formulary covering chromatography methods.
- NIST SP811: Special Publication providing guidelines for the use of the International System of Units (SI).
- ISO 31-8: International Standard for quantities and units in physical chemistry and molecular physics.
Conclusion
ASTM F2605-16 secures a consistent, validated approach to sodium alginate molar mass determination using SEC-MALS methods. This international standard empowers laboratories, manufacturers, and researchers to ensure reliable performance of alginate in high-stakes biomedical and pharmaceutical applications. By following this method, organizations can achieve greater quality control, compliance, and confidence in their alginate-based products.
Keywords: ASTM F2605-16, sodium alginate, molar mass, size exclusion chromatography, SEC-MALS, biomedical applications, pharmaceutical polymers, tissue engineering, polymer characterization, light scattering detection, polydispersity, alginate standards.
Buy Documents
ASTM F2605-16 - Standard Test Method for Determining the Molar Mass of Sodium Alginate by Size Exclusion Chromatography with Multi-angle Light Scattering Detection (SEC-MALS)
REDLINE ASTM F2605-16 - Standard Test Method for Determining the Molar Mass of Sodium Alginate by Size Exclusion Chromatography with Multi-angle Light Scattering Detection (SEC-MALS)
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2605-16 is a standard published by ASTM International. Its full title is "Standard Test Method for Determining the Molar Mass of Sodium Alginate by Size Exclusion Chromatography with Multi-angle Light Scattering Detection (SEC-MALS)". This standard covers: SIGNIFICANCE AND USE 4.1 The composition and sequential structure of alginate, as well as the molar mass and molar mass distribution, determines the functionality of alginate in an application. For instance, the gelling properties of an alginate are highly dependent upon the composition and molar mass of the polymer. 4.2 Light scattering is one of very few methods available for the determination of absolute molar mass and structure, and it is applicable over the broadest range of molar masses of any method. Combining light scattering detection with size exclusion chromatography (SEC), which sorts molecules according to size, gives the ability to analyze polydisperse samples, as well as to obtain information on branching and molecular conformation. This means that both the number-average and mass-average values for molar mass and size may be obtained for most samples. Furthermore, one has the ability to calculate the distributions of the molar masses and sizes. 4.3 Multi-angle laser light scattering (MALS) is a technique where measurements are made simultaneously over a range of different angles and used to determine the scattering at 0°, which directly relates to molecular weight. MALS detection can be used to obtain information on molecular size, since this parameter is determined by the angular variation of the scattered light. This can be related to branching, aggregation, and molecular conformation. Molar mass can also be determined by detecting scattered light at a single low angle (LALS) and assuming that this is not significantly different from the scattering at 0°. 4.4 Size exclusion chromatography uses columns, which are typically packed with polymer particles containing a network of uniform pores into which solute and solvent molecules can diffuse. While in the pores, molecules are effectively trapped and removed from the flow of the mobile phase. The average residence time in the pores depends upon the size of the solute molecules. Molecules that are larger than the ... SCOPE 1.1 This test method covers the determination of the molar mass (typically expressed as grams/mole) of sodium alginate intended for use in biomedical and pharmaceutical applications as well as in tissue-engineered medical products (TEMPs) by size exclusion chromatography with multi-angle laser light scattering detection (SEC-MALS). A guide for the characterization of alginate has been published as Guide F2064. 1.2 Alginate used in TEMPs should be well characterized, including the molar mass and polydispersity (molar mass distribution) in order to ensure uniformity and correct functionality in the final product. This test method will assist end users in choosing the correct alginate for their particular application. Alginate may have utility as a scaffold or matrix material for TEMPs, in cell and tissue encapsulation applications, and in drug delivery formulations. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 4.1 The composition and sequential structure of alginate, as well as the molar mass and molar mass distribution, determines the functionality of alginate in an application. For instance, the gelling properties of an alginate are highly dependent upon the composition and molar mass of the polymer. 4.2 Light scattering is one of very few methods available for the determination of absolute molar mass and structure, and it is applicable over the broadest range of molar masses of any method. Combining light scattering detection with size exclusion chromatography (SEC), which sorts molecules according to size, gives the ability to analyze polydisperse samples, as well as to obtain information on branching and molecular conformation. This means that both the number-average and mass-average values for molar mass and size may be obtained for most samples. Furthermore, one has the ability to calculate the distributions of the molar masses and sizes. 4.3 Multi-angle laser light scattering (MALS) is a technique where measurements are made simultaneously over a range of different angles and used to determine the scattering at 0°, which directly relates to molecular weight. MALS detection can be used to obtain information on molecular size, since this parameter is determined by the angular variation of the scattered light. This can be related to branching, aggregation, and molecular conformation. Molar mass can also be determined by detecting scattered light at a single low angle (LALS) and assuming that this is not significantly different from the scattering at 0°. 4.4 Size exclusion chromatography uses columns, which are typically packed with polymer particles containing a network of uniform pores into which solute and solvent molecules can diffuse. While in the pores, molecules are effectively trapped and removed from the flow of the mobile phase. The average residence time in the pores depends upon the size of the solute molecules. Molecules that are larger than the ... SCOPE 1.1 This test method covers the determination of the molar mass (typically expressed as grams/mole) of sodium alginate intended for use in biomedical and pharmaceutical applications as well as in tissue-engineered medical products (TEMPs) by size exclusion chromatography with multi-angle laser light scattering detection (SEC-MALS). A guide for the characterization of alginate has been published as Guide F2064. 1.2 Alginate used in TEMPs should be well characterized, including the molar mass and polydispersity (molar mass distribution) in order to ensure uniformity and correct functionality in the final product. This test method will assist end users in choosing the correct alginate for their particular application. Alginate may have utility as a scaffold or matrix material for TEMPs, in cell and tissue encapsulation applications, and in drug delivery formulations. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM F2605-16 is classified under the following ICS (International Classification for Standards) categories: 11.120.10 - Medicaments. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2605-16 has the following relationships with other standards: It is inter standard links to ASTM F2605-08e1, ASTM F2064-14, ASTM F2315-11, ASTM F2315-10, ASTM F2064-00(2006), ASTM F2064-00(2006)e1, ASTM F2315-03, ASTM F2064-00, ASTM F2064-17. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2605-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2605 − 16
Standard Test Method for
Determining the Molar Mass of Sodium Alginate by Size
Exclusion Chromatography with Multi-angle Light Scattering
Detection (SEC-MALS)
This standard is issued under the fixed designation F2605; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F2315 Guide for Immobilization or Encapsulation of Living
Cells or Tissue in Alginate Gels
1.1 This test method covers the determination of the molar
mass (typically expressed as grams/mole) of sodium alginate 2.2 United States Pharmacopeia/National Formulary:
intendedforuseinbiomedicalandpharmaceuticalapplications <621> Chromatography
as well as in tissue-engineered medical products (TEMPs) by 4
2.3 National Institute of Standards and Technology:
size exclusion chromatography with multi-angle laser light
NIST SP811 Special Publication: Guide for the Use of the
scattering detection (SEC-MALS). A guide for the character-
International System of Units
ization of alginate has been published as Guide F2064.
2.4 ISO Standards:
1.2 Alginate used in TEMPs should be well characterized,
ISO 31-8 Quantities and units- Part 8: Physical chemistry
including the molar mass and polydispersity (molar mass
and molecular physics
distribution) in order to ensure uniformity and correct func-
tionality in the final product. This test method will assist end
3. Terminology
users in choosing the correct alginate for their particular
3.1 Definitions:
application. Alginate may have utility as a scaffold or matrix
3.1.1 alginate, n—a polysaccharide substance extracted
material for TEMPs, in cell and tissue encapsulation
from brown algae, mainly occurring in the cell walls and
applications, and in drug delivery formulations.
intercellular spaces of brown seaweed and kelp. Its main
1.3 The values stated in SI units are to be regarded as
function is to contribute to the strength and flexibility of the
standard. No other units of measurement are included in this
seaweed plant. Sodium alginate, and in particular calcium
standard.
cross-linkedalginategelsareusedintissue-engineeredmedical
1.4 This standard does not purport to address all of the
products (TEMPs) as biomedical scaffolds and matrices, for
safety concerns, if any, associated with its use. It is the
immobilizing living cells (see Guide F2315), and in drug
responsibility of the user of this standard to establish appro-
delivery systems.
priate safety and health practices and determine the applica-
3.1.2 molar mass average, n—the given molar mass (Mw)
bility of regulatory limitations prior to use.
of an alginate will always represent an average of all of the
molecules in the population. The most common ways to
2. Referenced Documents
¯
expressthemolarmassareasthe number average(M )andthe
n
2.1 ASTM Standards: ¯
mass average (M ). The two averages are defined by the
w
F2064 Guide for Characterization and Testing of Alginates
following equations:
as Starting Materials Intended for Use in Biomedical and
N M w M N M
(i i i (i i i (i i i
Tissue Engineered Medical Product Applications
¯ ¯
M 5 and M 5 5 (1)
n w
N w N M
(i i (i i (i i i
This test method is under the jurisdiction ofASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.42 on Biomaterials and Biomolecules for TEMPs. Available from United States Pharmacopeia and National Formulary, U.S.
Current edition approved May 1, 2016. Published June 2016. Originally Pharmaceutical Convention, Inc. (USPC), Rockville, MD.
ϵ1
approved in 2008. Last previous edition approved in 2008 as F2605 – 08 . DOI: Available from National Institute of Standards and Technology (NIST), 100
10.1520/F2605-16. Bureau Dr., Stop 1070, Gaithersburg, MD 20899-1070, http://physics.nist.gov/cuu/
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Units/bibliography.html.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Available from International Organization for Standardization (ISO), ISO
Standards volume information, refer to the standard’s Document Summary page on Central Secretariat, BIBC II, Chemin de Blandonnet 8, CP 401, 1214 Vernier,
the ASTM website. Geneva, Switzerland, http://www.iso.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2605 − 16
where: volume available for diffusion; their retention will depend on
their molecular size, with the smaller molecules eluting last.
N = number of molecules having a specific molar mass M,
i i
and
4.5 For polyelectrolytes, dialysis against the elution buffer
w = mass of molecules having a specific molar mass M.
i i
has been suggested, in order to eliminate Donnan-type artifacts
in the molar mass determination by light scattering (1, 2).
3.1.2.1 Discussion—In a polydisperse molecular population
However, in the present method, the size exclusion chroma-
¯ ¯ ¯ ¯
the relation M > M is always valid.The coefficient M /M is
w n w n
tography step preceding the light scatter detection is an
referred to as the polydispersity index, and will typically be in
efficient substitute for a dialysis step. The sample is separated
the range 1.5 to 3.0 for commercial alginates.
onSECcolumnswithlargeexcessofelutionbufferfor30to40
NOTE 1—The term molecular weight (abbreviated MW) is obsolete and min, and it is therefore in full equilibrium with the elution
should be replaced by the SI (Système Internationale) equivalent of either
buffer when it reaches the MALS detector.
relative molecular mass (M ), which reflects the dimensionless ratio of the
r
mass of a single molecule to an atomic mass unit (see ISO 31-8), or molar
5. Materials
mass (M), which refers to the mass of a mole of a substance and is
5.1 Chemicals:
typically expressed as grams/mole. For polymers and other
macromolecules, use of the symbols M , M , and M continue, referring
w n z 5.1.1 Alginate sample.
to mass-average molar mass, number-average molar mass, and z-average
5.1.2 Deionized water (Milli-Q Plus or equivalent; conduc-
molar mass, respectively. For more information regarding proper utiliza-
tivity < 10 µS/cm).
tion of SI units, see NIST SP811.
5.1.3 Na SO (sodium sulfate).
2 4
5.1.4 EDTA (ethylene diamine tetraacetic acid).
4. Significance and Use
5.1.5 NaOH (1 mol/L).
4.1 The composition and sequential structure of alginate, as
5.1.6 Pullulan standards. See Note 2.
wellasthemolarmassandmolarmassdistribution,determines
NOTE 2—A series of linear homopolysaccharides with sufficiently
the functionality of alginate in an application. For instance, the
narrow dispersity to be suitable for utilization as molar mass calibration
gelling properties of an alginate are highly dependent upon the
standards in aqueous eluent.
composition and molar mass of the polymer.
5.2 The Mobile Phase:
4.2 Lightscatteringisoneofveryfewmethodsavailablefor 5.2.1 For SEC-MALS of alginate, a mobile phase of 0.05
the determination of absolute molar mass and structure, and it mol/L Na SO /0.01 mol/L EDTA in deionized water is used.
2 4
is applicable over the broadest range of molar masses of any Adjust pH to 6.0 using 1 mol/L NaOH.
method. Combining light scattering detection with size exclu- 5.2.2 Mobilephaseshouldbepreparedasastocksolutionof
sion chromatography (SEC), which sorts molecules according 0.10 mol/L Na SO /0.02 mol/L EDTA in deionized water,
2 4
to size, gives the ability to analyze polydisperse samples, as which can be stored cool (3 to 8°C) for 6 months. Before use
well as to obtain information on branching and molecular
as a mobile phase, the stock solution is diluted 1:1 (v/v) with
conformation. This means that both the number-average and deionized water and passed through a 0.22 µm filter.
mass-average values for molar mass and size may be obtained
5.3 Instruments:
for most samples. Furthermore, one has the ability to calculate
5.3.1 Analytical balance (0.1 mg).
the distributions of the molar masses and sizes.
5.3.2 Shaking device.
5.3.3 pH meter.
4.3 Multi-angle laser light scattering (MALS) is a technique
5.3.4 High Performance Liquid Chromotography (HPLC)
where measurements are made simultaneously over a range of
system with injector, pump, degassing unit.
different angles and used to determine the scattering at 0°,
5.3.5 Size exclusion columns: TSK-Gel PW columns
which directly relates to molecular weight. MALS detection
XL
from Tosoh Biosep., for example, PW -guard column +
can be used to obtain information on molecular size, since this XL
G6000 PW + G5000 PW + G3000 PW (last in the
parameter is determined by the angular variation of the
XL XL XL
series), or equivalent.
scattered light. This can be related to branching, aggregation,
5.3.6 Refractive Index (RI) detector, with a known calibra-
and molecular conformation. Molar mass can also be deter-
tion constant (dn/dV).
minedbydetectingscatteredlightatasinglelowangle(LALS)
5.3.7 Multiple Angle Laser Light Scattering (MALS)
and assuming that this is not significantly different from the
detector, with known calibration constant.
scattering at 0°.
5.3.8 Computer with suitable software.
4.4 Sizeexclusionchromatographyusescolumns,whichare
typically packed with polymer particles containing a network
6. Procedure
of uniform pores into which solute and solvent molecules can
6.1 Preparation of Standards and Alginate Samples for
diffuse. While in the pores, molecules are effectively trapped
SEC-MALS:
and removed from the flow of the mobile phase. The average
6.1.1 Samples are prepared at a concentration suitable for
residence time in the pores depends upon the size of the solute
injection of 200 µL of sample.
molecules. Molecules that are larger than the average pore size
of the packing are excluded and experience virtually no
retention; these are eluted first, in the void volume of the
The boldface numbers in parentheses refer to a list of references at the end of
column.Moleculeswhichpenetratetheporeswillhavealarger this standard.
F2605 − 16
6.1.2 Dissolve all samples in deionized water at twice the 6.3 Data Analysis:
required concentration for molar mass determination by shak-
6.3.1 Data analysis follows closely recommended proce-
-1
ingatabout100min overnightatcooltemperature(3to8°C).
dures for SEC-MALS data. Generally, the chromatograms are
6.1.3 Dilute samples 1+1 with stock solution of mobile
divided into a number of volume elements, defined by the peak
phase and shake gently for a few seconds.
width, the rate of data collection and the flow rate. The
6.1.4 Pass all samples through a 0.45 µm filter, and transfer
concentration of sample in each volume element (c) is deter-
i
to HPLC vials.
mined from the RI-detector response using known values of
6.1.5 Final concentration of pullulan standards of known
dn/dc and dn/dV (the RI-detector calibration constant).
¯
M values of approximately 11 800 to 47 300, 112 000,
w Furthermore, MALS-detector response is divided by c, the
i
212 000, and 404 000 g/mol should be approximately 4, 3, 2,
molar mass in each volume element (M) is considered
i
and 1.5 mg/mL, respectively.
monodisperse, and the mass is determined from a Zimm
6.1.6 Guidelines for the final concentration of alginates for
representation of a Debye plot by extrapolation to zero angle
molar mass determination are given in Table 1. If SEC-MALS
(which is essentially a solution to Eq X2.1 in X2.2). Once the
data display poor reproducibility with respect to replicates, this
values of c and M are known, calculation of the various
i i
might be an indication of column overload. In this case, less
average molar masses is straightforward.
sample should be injected.
6.3.2 In detail, the above procedure consists of the follow-
ing operations to be performed using suitable software:
6.2 Chromatography and Data Collection:
6.2.1 The complete experimental setup of the SEC-MALS
6.3.2.1 Define baselines for signals from both detectors.
system is shown in Fig. 1. The refractive index detector is
6.3.2.2 Calculate inter-detector delay volume using a
placed at the end of the solvent/sample line as it is highly
monodisperse low-molar mass pullulan standard.
sensitive to pressure changes.
6.3.2.3 Define the peak area of interest.
6.2.2 Pullulan standards should be injected and analyzed
6.3.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: F2605 − 08 F2605 − 16
Standard Test Method for
Determining the Molar Mass of Sodium Alginate by Size
Exclusion Chromatography with Multi-angle Light Scattering
Detection (SEC-MALS)
This standard is issued under the fixed designation F2605; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Subsection 6.1.5 was editorially corrected in September 2008.
1. Scope
1.1 This test method covers the determination of the molar mass (typically expressed as grams/mole) of sodium alginate
intended for use in biomedical and pharmaceutical applications as well as in tissue engineered tissue-engineered medical products
(TEMPs) by size exclusion chromatography with multi-angle laser light scattering detection (SEC-MALS). A guide for the
characterization of alginate has been published as Guide F2064.
1.2 Alginate used in TEMPs should be well characterized, including the molar mass and polydispersity (molar mass
distribution) in order to ensure uniformity and correct functionality in the final product. This test method will assist end users in
choosing the correct alginate for their particular application. Alginate may have utility as a scaffold or matrix material for TEMPs,
in cell and tissue encapsulation applications, and in drug delivery formulations.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
F2064 Guide for Characterization and Testing of Alginates as Starting Materials Intended for Use in Biomedical and Tissue
Engineered Medical Product Applications
F2315 Guide for Immobilization or Encapsulation of Living Cells or Tissue in Alginate Gels
2.2 United States Pharmacopeia/National Formulary:
<621> Chromatography
2.3 National Institute of Standards and Technology:
NIST SP811 Special Publication: Guide for the Use of the International System of Units
2.4 ISO Standards:
ISO 31-8 Quantities and units- Part 8: Physical chemistry and molecular physics
3. Terminology
3.1 Definitions:
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.42 on Biomaterials and Biomolecules for TEMPs.
ϵ1
Current edition approved Feb. 1, 2008May 1, 2016. Published May 2008June 2016. Originally approved in 2008. Last previous edition approved in 2008 as F2605 – 08 .
DOI: 10.1520/F2605-08E01.10.1520/F2605-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from United States Pharmacopeia and National Formulary, U.S. Pharmaceutical Convention, Inc. (USPC), Rockville, MD.
Available from National Institute of Standards and Technology (NIST), 100 Bureau Dr., Stop 1070, Gaithersburg, MD 20899-1070, http://physics.nist.gov/cuu/Units/
bibliography.html.
Available from International Organization for Standardization (ISO), ISO Central Secretariat, BIBC II, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
Switzerland, http://www.iso.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2605 − 16
3.1.1 alginate, n—a polysaccharide substance extracted from brown algae, mainly occurring in the cell walls and intercellular
spaces of brown seaweed and kelp. Its main function is to contribute to the strength and flexibility of the seaweed plant. Sodium
alginate, and in particular calcium cross-linked alginate gels are used in tissue engineered tissue-engineered medical products
(TEMPs) as biomedical scaffolds and matrices, for immobilizing living cells (see Guide F2315)), and in drug delivery systems.
3.1.2 molar mass average, n—the given molar mass (Mw) of an alginate will always represent an average of all of the molecules
in the population. The most common ways to express the molar mass are as the number average (M¯ ) and the mass average (M¯ ).
n w
The two averages are defined by the following equations:
N M w M N M
(i i i (i i i (i i i
¯ ¯
M 5 and M 5 5 (1)
n w
N w N M
(i i (i i (i i i
where:
N = number of molecules having a specific molar mass M , and
i i
w = mass of molecules having a specific molar mass M .
i i
3.1.2.1 Discussion—
In a polydisperse molecular population the relation M¯ > M¯ is always valid. The coefficient M¯ /M¯ is referred to as the
w n w n
polydispersity index, and will typically be in the range 1.5 to 3.0 for commercial alginates.
NOTE 1—The term molecular weight (abbreviated MW) is obsolete and should be replaced by the SI (Système Internationale) equivalent of either
relative molecular mass (M ), which reflects the dimensionless ratio of the mass of a single molecule to an atomic mass unit (see ISO 31-8), or molar
r
mass (M), which refers to the mass of a mole of a substance and is typically expressed as grams/mole. For polymers and other macromolecules, use of
the symbols M ,M , and M continue, referring to mass-average molar mass, number-average molar mass, and z-average molar mass, respectively. For
w n z
more information regarding proper utilization of SI units, see NIST SP811.
4. Significance and Use
4.1 The composition and sequential structure of alginate, as well as the molar mass and molar mass distribution, determines the
functionality of alginate in an application. For instance, the gelling properties of an alginate are highly dependent upon the
composition and molar mass of the polymer.
4.2 Light scattering is one of very few methods available for the determination of absolute molar mass and structure, and it is
applicable over the broadest range of molar masses of any method. Combining light scattering detection with size exclusion
chromatography (SEC), which sorts molecules according to size, gives the ability to analyze polydisperse samples, as well as
obtaining to obtain information on branching and molecular conformation. This means that both the number-average and
mass-average values for molar mass and size may be obtained for most samples. Furthermore, one has the ability to calculate the
distributions of the molar masses and sizes.
4.3 Multi-angle laser light scattering (MALS) is a technique where measurements are made simultaneously over a range of
different angles. angles and used to determine the scattering at 0°, which directly relates to molecular weight. MALS detection can
be used to obtain information on molecular size, since this parameter is determined by the angular variation of the scattered light.
Molar mass may in principle This can be related to branching, aggregation, and molecular conformation. Molar mass can also be
determined by detecting scattered light at a single low angle (LALLS). However, advantages with MALS as compared to LALLS
are: (LALS) (1) less noise at larger angles, (2) the precision of measurements are greatly improved by detecting at several angles,
and (3)assuming that the ability to detect angular variation allows determination of size, branching, aggregation, and molecular
conformation.this is not significantly different from the scattering at 0°.
4.4 Size exclusion chromatography uses columns, which are typically packed with polymer particles containing a network of
uniform pores into which solute and solvent molecules can diffuse. While in the pores, molecules are effectively trapped and
removed from the flow of the mobile phase. The average residence time in the pores depends upon the size of the solute molecules.
Molecules that are larger than the average pore size of the packing are excluded and experience virtually no retention; these are
eluted first, in the void volume of the column. Molecules,Molecules which may penetrate the pores will have a larger volume
available for diffusion, they will suffer retention depending diffusion; their retention will depend on their molecular size, with the
smaller molecules eluting last.
4.5 For polyelectrolytes, dialysis against the elution buffer has been suggested, in order to eliminate Donnan-type artifacts in
the molar mass determination by light scattering (1, 2). However, in the present method, the size exclusion chromatography step
preceding the light scatter detection is an efficient substitute for a dialysis step. The sample is separated on SEC columns with large
excess of elution buffer for 30 to 40 min, and it is therefore in full equilibrium with the elution buffer when it reaches the MALS
detector.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
F2605 − 16
5. Materials
5.1 Chemicals:
5.1.1 Alginate sample.
5.1.2 Deionized water (Milli-Q Plus or equivalent; conductivity < 10 μS/cm).
5.1.3 Na SO (sodium sulfate).
2 4
5.1.4 EDTA (ethylene diamine tetraacetic acid).
5.1.5 NaOH (1 mol/L).
5.1.6 Pullulan standards. See Note 2.
NOTE 2—A series of linear homopolysaccharides with sufficiently narrow dispersity to be suitable for utilization as molar mass calibration standards
in aqueous eluent.
5.2 The Mobile Phase:
5.2.1 For SEC-MALS of alginate, a mobile phase of 0.05 mol/L Na SO /0.01 mol/L EDTA in deionized water is used. Adjust
2 4
pH to 6.0 using 1 mol/L NaOH.
5.2.2 Mobile phase should be prepared as a stock solution of 0.10 mol/L Na SO /0.02 mol/L EDTA in deionized water, which
2 4
can be stored cool (3 to 8°C) for 6 months. Before use as a mobile phase, the stock solution is diluted 1:1 (v/v) with deionized
water and passed through a 0.22 μm filter.
5.3 Instruments:
5.3.1 Analytical balance (0.1 mg).
5.3.2 Shaking device.
5.3.3 pH meter.
5.3.4 HPLC High Performance Liquid Chromotography (HPLC) system with injector, pump, degassing unit.
5.3.5 Size exclusion columns: TSK-Gel PW columns from Tosoh Biosep., for example, PW -guard column + G6000
XL XL
PW + G5000 PW + G3000 PW (last in the series), or equivalent.
XL XL XL
5.3.6 Refractive Index (RI) detector, with a known calibration constant (dn/dV).
5.3.7 Multiple Angle Laser Light Scattering (MALS) detector, with known calibration constant.
5.3.8 Computer with suitable software.
6. Procedure
6.1 Preparation of Standards and Alginate Samples for SEC-MALS:
6.1.1 Samples are prepared at a concentration suitable for injection of 200 μL of sample.
6.1.2 Dissolve all samples in deionized water at twice the required concentration for molar mass determination by shaking at
-1
about 100 min overnight at cool temperature (3 to 8°C).
6.1.3 Dilute samples 1+1 with stock solution of mobile phase and shake gently for a few seconds.
6.1.4 Pass all samples through a 0.45 μm filter, and transfer to HPLC vials.
6.1.5 Final concentration of pullulan standards of known M¯ values of approximately 11 800 to 47 300, 112 000, 212 000, and
w
404 000 g/mol should be approximately 4, 3, 2, and 1.5 mg/mL, respectively.
6.1.6 Guidelines for the final concentration of alginates for molar mass determination are given in Table 1. If SEC-MALS data
display poor reproducibility with respect to replicates, this might be an indication of column overload. In this case, less sample
should be injected.
6.2 Chromatography and Data Collection:
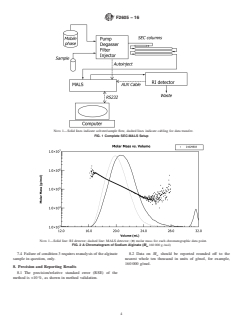
6.2.1 The complete experimental setup of the SEC-MALS system is shown in Fig. 1. The refractive index detector is placed
at the end of the solvent/sample line as it is highly sensitive to pressure changes.
6.2.2 Pullulan standards should be injected and analyzed with 2 replicates before and after all alginate samples (total of 4
replicates). 3 replicates should be injected for alginates.
6.2.3 A procedure for setting up the chromatography run and collecting the data is given below:
TABLE 1 Suggestions for Concentration and Injected Mass of
Alginate Samples for SEC-MALS
Apparent Concentration for Injected
M¯
w
A
Viscosity Injection
Mass
(g/mol)
(mg)
(mPas) (mg/mL)
<50 000 <10 4 0.8
50 000–75 000 10–20 3 0.6
75 000–100 000 20–40 2 0.4
100 000–150 000 40–100 1.5 0.3
150 000–250 000 100–300 1 0.2
>250 000 >300 0.5 0.1
A
Injected mass = Concentration*200 μL.
F2605 − 16
NOTE 1—Solid lines indicate solvent/sample flow, dashed lines indicate cabling for data transfer.
FIG. 1 Complete SEC-MALS Set-UpSetup
6.2.3.1 Use a flow rate of 0.5 mL/min.
6.2.3.2 Purge the injector with mobile phase before the sample set is run.
6.2.3.3 Purge the RI-detector refractive index (RI)-detector for at least 3
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...