ASTM D5953M-23
(Test Method)Standard Test Method for Determination of Non-methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic Preconcentration and Direct Flame Ionization Detection
Standard Test Method for Determination of Non-methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic Preconcentration and Direct Flame Ionization Detection
SIGNIFICANCE AND USE
5.1 Many regulators, industrial processes, and other stakeholders require determination of NMOC in atmospheres.
5.2 Accurate measurements of ambient NMOC concentrations are critical in devising air pollution control strategies and in assessing control effectiveness because NMOCs are primary precursors of atmospheric ozone and other oxidants (7, 8).
5.2.1 The NMOC concentrations typically found at urban sites may range up to 1 ppm C to 3 ppm C or higher. In order to determine transport of precursors into an area monitoring site, measurement of NMOC upwind of the site may be necessary. Rural NMOC concentrations originating from areas free from NMOC sources are likely to be less than a few tenths of 1 ppm C.
5.3 Conventional test methods based upon gas chromatography and qualitative and quantitative species evaluation are relatively time consuming, sometimes difficult and expensive in staff time and resources, and are not needed when only a measurement of NMOC is desired. The test method described requires only a simple, cryogenic pre-concentration procedure followed by direct detection with an FID. This test method provides a sensitive and accurate measurement of ambient total NMOC concentrations where speciated data are not required. Typical uses of this standard test method are as follows.
5.4 An application of the test method is the monitoring of the cleanliness of canisters.
5.5 Another use of the test method is the screening of canister samples prior to analysis.
5.6 Collection of ambient air samples in pressurized canisters provides the following advantages:
5.6.1 Convenient collection of integrated ambient samples over a specific time period,
5.6.2 Capability of remote sampling with subsequent central laboratory analysis,
5.6.3 Ability to ship and store samples, if necessary,
5.6.4 Unattended sample collection,
5.6.5 Analysis of samples from multiple sites with one analytical system,
5.6.6 Collection of replicate samples for ...
SCOPE
1.1 This test method2 presents a procedure for sampling and determination of non-methane organic compounds (NMOC) in ambient, indoor, or workplace atmospheres.
1.2 This test method describes the collection of integrated whole air samples in silanized or other passivated stainless steel canisters, and their subsequent laboratory analysis.
1.2.1 This test method describes a procedure for sampling in canisters at final pressures above atmospheric pressure (pressurized sampling).
1.3 This test method employs a cryogenic trapping procedure for concentration of the NMOC prior to analysis.
1.4 This test method describes the determination of the NMOC by the flame ionization detection (FID), without the use of gas chromatographic columns and other procedures necessary for species separation.
1.5 The range of this test method is from 20 ppb C to 10 000 ppb C (1, 2).3
1.6 This test method has a larger uncertainty for some halogenated or oxygenated hydrocarbons than for simple hydrocarbons or aromatic compounds. This is especially true if there are high concentrations of chlorocarbons or chlorofluorocarbons present.
1.7 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Techn...
General Information
- Status
- Published
- Publication Date
- 28-Feb-2023
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.03 - Ambient Atmospheres and Source Emissions
Relations
- Effective Date
- 01-Sep-2020
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Aug-2019
- Effective Date
- 15-Oct-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Dec-2014
- Effective Date
- 01-May-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Apr-2010
- Effective Date
- 01-Apr-2007
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-May-2005
- Effective Date
- 01-Mar-2005
- Effective Date
- 10-Dec-2001
Overview
ASTM D5953M-23 is the standard test method for the determination of non-methane organic compounds (NMOC) in ambient air using cryogenic preconcentration and direct flame ionization detection (FID). Established by ASTM International, this method outlines a procedure essential for regulators, environmental professionals, and industries needing reliable NMOC measurement for air quality assessment and pollution control strategy development. Accurate NMOC quantification is vital as these compounds are key precursors to ground-level ozone and other atmospheric oxidants in both urban and rural environments.
Key Topics
- Comprehensive NMOC Analysis: The standard provides a methodology for sampling and quantifying NMOC in ambient, indoor, or workplace atmospheres, emphasizing the importance of total NMOC measurements in air quality management.
- Cryogenic Preconcentration: Utilizes cryogenic trapping (typically at around -186 °C) with liquid argon to concentrate NMOC and separate methane, thus enhancing the sensitivity and selectivity of the analysis.
- Flame Ionization Detection (FID): Directs concentrated NMOC into an FID, enabling accurate quantification without the need for gas chromatographic separation, offering a faster and cost-effective solution compared to traditional GC-based methods.
- Sample Collection: Employs silanized or passivated stainless steel canisters for pressurized whole air sample collection, allowing for integrated sampling over defined time periods and simplifying sample transport and storage.
- Calibration and Precision: Emphasizes the use of dynamic calibration standards and conducting quality assurance checks to ensure accurate, repeatable results, even at low NMOC concentrations (from 20 ppb C up to 10,000 ppb C).
- Limitations: Notes higher uncertainty for certain halogenated and oxygenated hydrocarbons, particularly in samples with significant chlorocarbon or chlorofluorocarbon presence.
Applications
- Ambient and Workplace Air Monitoring: Widely used by regulatory agencies and environmental consultants to monitor NMOC levels in urban, rural, and industrial atmospheres.
- Air Pollution Control Strategy Development: Critical for assessing baseline NMOC concentrations and evaluating the effectiveness of emission reduction programs.
- Canister Cleanliness Verification: Enables laboratories to confirm the cleanliness of sampling canisters, ensuring sample integrity and reliable analytical outcomes.
- Screening Canister Samples: Facilitates rapid screening prior to more detailed analysis or speciation, saving both time and resources.
- Remote and Unattended Sampling: Supports convenient, unattended collection at multiple or remote sites with subsequent central laboratory analysis, meeting the demands of extensive monitoring networks.
- Quality Assurance and Data Comparability: Pressurized canister sampling allows for replicate sample collections and comparative analysis from multiple monitoring locations using a single analytical system.
Related Standards
- ASTM D1193: Specification for Reagent Water - outlines water quality requirements for laboratory protocols.
- ASTM D1356: Terminology Relating to Sampling and Analysis of Atmospheres - provides consistent definitions used in air quality measurements.
- ASTM D1357: Practice for Planning the Sampling of the Ambient Atmosphere - covers best practices for designing ambient air sampling strategies.
- ASTM D5466: Test Method for Determination of Volatile Organic Compounds in Atmospheres (Canister Sampling, Mass Spectrometry Analysis Methodology) - offers complementary methodologies for VOC sampling and analysis.
- EPA Method TO-12: Reference method for NMOC determination using cryogenic preconcentration and FID, forming the basis of this ASTM standard.
ASTM D5953M-23 is internationally recognized and aligns with WTO principles, ensuring its acceptance in global regulatory and industrial contexts. This standard delivers a reliable, efficient means for NMOC air monitoring, supporting air quality improvement, environmental compliance, and public health protection.
Buy Documents
ASTM D5953M-23 - Standard Test Method for Determination of Non-methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic Preconcentration and Direct Flame Ionization Detection
REDLINE ASTM D5953M-23 - Standard Test Method for Determination of Non-methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic Preconcentration and Direct Flame Ionization Detection
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.
Sponsored listings
Frequently Asked Questions
ASTM D5953M-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Non-methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic Preconcentration and Direct Flame Ionization Detection". This standard covers: SIGNIFICANCE AND USE 5.1 Many regulators, industrial processes, and other stakeholders require determination of NMOC in atmospheres. 5.2 Accurate measurements of ambient NMOC concentrations are critical in devising air pollution control strategies and in assessing control effectiveness because NMOCs are primary precursors of atmospheric ozone and other oxidants (7, 8). 5.2.1 The NMOC concentrations typically found at urban sites may range up to 1 ppm C to 3 ppm C or higher. In order to determine transport of precursors into an area monitoring site, measurement of NMOC upwind of the site may be necessary. Rural NMOC concentrations originating from areas free from NMOC sources are likely to be less than a few tenths of 1 ppm C. 5.3 Conventional test methods based upon gas chromatography and qualitative and quantitative species evaluation are relatively time consuming, sometimes difficult and expensive in staff time and resources, and are not needed when only a measurement of NMOC is desired. The test method described requires only a simple, cryogenic pre-concentration procedure followed by direct detection with an FID. This test method provides a sensitive and accurate measurement of ambient total NMOC concentrations where speciated data are not required. Typical uses of this standard test method are as follows. 5.4 An application of the test method is the monitoring of the cleanliness of canisters. 5.5 Another use of the test method is the screening of canister samples prior to analysis. 5.6 Collection of ambient air samples in pressurized canisters provides the following advantages: 5.6.1 Convenient collection of integrated ambient samples over a specific time period, 5.6.2 Capability of remote sampling with subsequent central laboratory analysis, 5.6.3 Ability to ship and store samples, if necessary, 5.6.4 Unattended sample collection, 5.6.5 Analysis of samples from multiple sites with one analytical system, 5.6.6 Collection of replicate samples for ... SCOPE 1.1 This test method2 presents a procedure for sampling and determination of non-methane organic compounds (NMOC) in ambient, indoor, or workplace atmospheres. 1.2 This test method describes the collection of integrated whole air samples in silanized or other passivated stainless steel canisters, and their subsequent laboratory analysis. 1.2.1 This test method describes a procedure for sampling in canisters at final pressures above atmospheric pressure (pressurized sampling). 1.3 This test method employs a cryogenic trapping procedure for concentration of the NMOC prior to analysis. 1.4 This test method describes the determination of the NMOC by the flame ionization detection (FID), without the use of gas chromatographic columns and other procedures necessary for species separation. 1.5 The range of this test method is from 20 ppb C to 10 000 ppb C (1, 2).3 1.6 This test method has a larger uncertainty for some halogenated or oxygenated hydrocarbons than for simple hydrocarbons or aromatic compounds. This is especially true if there are high concentrations of chlorocarbons or chlorofluorocarbons present. 1.7 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Techn...
SIGNIFICANCE AND USE 5.1 Many regulators, industrial processes, and other stakeholders require determination of NMOC in atmospheres. 5.2 Accurate measurements of ambient NMOC concentrations are critical in devising air pollution control strategies and in assessing control effectiveness because NMOCs are primary precursors of atmospheric ozone and other oxidants (7, 8). 5.2.1 The NMOC concentrations typically found at urban sites may range up to 1 ppm C to 3 ppm C or higher. In order to determine transport of precursors into an area monitoring site, measurement of NMOC upwind of the site may be necessary. Rural NMOC concentrations originating from areas free from NMOC sources are likely to be less than a few tenths of 1 ppm C. 5.3 Conventional test methods based upon gas chromatography and qualitative and quantitative species evaluation are relatively time consuming, sometimes difficult and expensive in staff time and resources, and are not needed when only a measurement of NMOC is desired. The test method described requires only a simple, cryogenic pre-concentration procedure followed by direct detection with an FID. This test method provides a sensitive and accurate measurement of ambient total NMOC concentrations where speciated data are not required. Typical uses of this standard test method are as follows. 5.4 An application of the test method is the monitoring of the cleanliness of canisters. 5.5 Another use of the test method is the screening of canister samples prior to analysis. 5.6 Collection of ambient air samples in pressurized canisters provides the following advantages: 5.6.1 Convenient collection of integrated ambient samples over a specific time period, 5.6.2 Capability of remote sampling with subsequent central laboratory analysis, 5.6.3 Ability to ship and store samples, if necessary, 5.6.4 Unattended sample collection, 5.6.5 Analysis of samples from multiple sites with one analytical system, 5.6.6 Collection of replicate samples for ... SCOPE 1.1 This test method2 presents a procedure for sampling and determination of non-methane organic compounds (NMOC) in ambient, indoor, or workplace atmospheres. 1.2 This test method describes the collection of integrated whole air samples in silanized or other passivated stainless steel canisters, and their subsequent laboratory analysis. 1.2.1 This test method describes a procedure for sampling in canisters at final pressures above atmospheric pressure (pressurized sampling). 1.3 This test method employs a cryogenic trapping procedure for concentration of the NMOC prior to analysis. 1.4 This test method describes the determination of the NMOC by the flame ionization detection (FID), without the use of gas chromatographic columns and other procedures necessary for species separation. 1.5 The range of this test method is from 20 ppb C to 10 000 ppb C (1, 2).3 1.6 This test method has a larger uncertainty for some halogenated or oxygenated hydrocarbons than for simple hydrocarbons or aromatic compounds. This is especially true if there are high concentrations of chlorocarbons or chlorofluorocarbons present. 1.7 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to inch-pound units that are provided for information only and are not considered standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Techn...
ASTM D5953M-23 is classified under the following ICS (International Classification for Standards) categories: 13.040.01 - Air quality in general; 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5953M-23 has the following relationships with other standards: It is inter standard links to ASTM D1356-20a, ASTM D1356-20, ASTM D1357-95(2019), ASTM D1356-15a, ASTM D1356-15, ASTM D1356-14b, ASTM D1356-14a, ASTM D1356-14, ASTM D1357-95(2011), ASTM D1356-05(2010), ASTM D5466-01(2007), ASTM D1193-06, ASTM D1356-05, ASTM D1357-95(2005), ASTM D5466-01. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5953M-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5953M − 23
Standard Test Method for
Determination of Non-methane Organic Compounds (NMOC)
in Ambient Air Using Cryogenic Preconcentration and Direct
Flame Ionization Detection
This standard is issued under the fixed designation D5953M; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope conversions to inch-pound units that are provided for informa-
tion only and are not considered standard.
1.1 This test method presents a procedure for sampling and
1.8 This standard does not purport to address all of the
determination of non-methane organic compounds (NMOC) in
safety concerns, if any, associated with its use. It is the
ambient, indoor, or workplace atmospheres.
responsibility of the user of this standard to establish appro-
1.2 This test method describes the collection of integrated
priate safety, health, and environmental practices and deter-
whole air samples in silanized or other passivated stainless
mine the applicability of regulatory limitations prior to use.
steel canisters, and their subsequent laboratory analysis.
1.9 This international standard was developed in accor-
1.2.1 This test method describes a procedure for sampling in
dance with internationally recognized principles on standard-
canisters at final pressures above atmospheric pressure (pres-
ization established in the Decision on Principles for the
surized sampling).
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.3 This test method employs a cryogenic trapping proce-
dure for concentration of the NMOC prior to analysis. Barriers to Trade (TBT) Committee.
1.4 This test method describes the determination of the
2. Referenced Documents
NMOC by the flame ionization detection (FID), without the
2.1 ASTM Standards:
use of gas chromatographic columns and other procedures
D1193 Specification for Reagent Water
necessary for species separation.
D1356 Terminology Relating to Sampling and Analysis of
1.5 The range of this test method is from 20 ppb C to
Atmospheres
10 000 ppb C (1, 2).
D1357 Practice for Planning the Sampling of the Ambient
Atmosphere
1.6 This test method has a larger uncertainty for some
halogenated or oxygenated hydrocarbons than for simple D5466 Test Method for Determination of Volatile Organic
Compounds in Atmospheres (Canister Sampling, Mass
hydrocarbons or aromatic compounds. This is especially true if
there are high concentrations of chlorocarbons or chlorofluo- Spectrometry Analysis Methodology)
rocarbons present.
2.2 Other References:
EPA Method TO-12 Determination of Non-Methane Organic
1.7 The values stated in SI units are to be regarded as
Compounds (NMOC) in Ambient Air Using Cryogenic
standard. The values given in parentheses are mathematical
Pre-Concentration and Direct Flame Ionization Detection
(PDFID)
This test method is under the jurisdiction of ASTM Committee D22 on Air
3. Terminology
Quality and is the direct responsibility of Subcommittee D22.03 on Ambient
Atmospheres and Source Emissions. 3.1 Definitions—For definitions of terms used in this test
Current edition approved March 1, 2023. Published March 2023. Originally
method, refer to Terminology D1356.
approved in 1996. Last previous edition approved in 2016 as D5953M – 16. DOI:
10.1520/D5953M-23.
This test method is based on EPA Compendium Method TO-12: “Determination
of Non-Methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Pre-Concentration and Direct Flame Ionization Detection (PDFID),” Compendium contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
of Methods for the Determination of Toxic Organic Compounds in Ambient Air, EPA Standards volume information, refer to the standard’s Document Summary page on
600 4-89-017, U.S. Environmental Protection Agency, Research Triangle Park, NC, the ASTM website.
March 1990. Available from United States Environmental Protection Agency (EPA), William
The boldface numbers in parentheses refer to the list of references at the end of Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460,
this standard. http://www.epa.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5953M − 23
3.2 Definitions of Terms Specific to This Standard: 4.5 The cryogenic trap simultaneously concentrates the
NMOC while separating and removing the methane from
3.2.1 cryogen—a refrigerant used to obtain very low tem-
samples. The technique thus directly measures NMOC with
peratures in analytical system cryogenic traps.
greater sensitivity than conventional continuous NMOC ana-
3.2.1.1 Discussion—Liquid argon (bp –185.7 °C at standard
lyzers due to the pre-concentration procedure.
pressure) is recommended and may be required for use in some
applications of this test method. Cryogens with lower boiling
4.6 The sample is injected into the hydrogen-rich flame of
points, such as liquid nitrogen, may be used if the cryogenic
an FID, where the organic vapors burn, producing ionized
trap temperature is actively maintained at –185°C to avoid the
molecular fragments. The resulting ion fragments are then
potential for trapping oxygen or methane from air samples.
collected and detected. Because this test method employs a
helium carrier gas, the detector response is nearly identical for
3.2.2 dynamic calibration—calibration of an analytical sys-
many hydrocarbon compounds commonly of interest. Thus, the
tem with pollutant concentrations that are generated in a
historical short-coming of varying FID response to aromatic,
dynamic, flowing system, such as by quantitative, flow-rate
olefinic, and paraffinic hydrocarbons is minimized. Users are
dilution of a high-concentration gas standard with zero gas.
cautioned that the FID is much less sensitive to most organic
3.2.3 NMOC—non-methane organic compounds.
compounds containing functional groups such as carbonyls,
3.2.3.1 Discussion—Total non-methane organic compounds
alcohols, halocarbons, etc. than simple hydrocarbons.
are compounds, excluding methane, measured using a flame
−2
ionization detector (FID), with vapor pressures above 10 kPa
5. Significance and Use
recovered from canisters.
5.1 Many regulators, industrial processes, and other stake-
3.2.4 ppm C and ppb C—concentration units of parts-per-
holders require determination of NMOC in atmospheres.
million and parts-per-billion of organic carbon as detected by
FID.
5.2 Accurate measurements of ambient NMOC concentra-
3.2.4.1 Discussion—For example, when calibrating with tions are critical in devising air pollution control strategies and
propane, concentrations of NMOC in samples are equivalent to in assessing control effectiveness because NMOCs are primary
parts-per-million by volume (ppm (v)) or parts-per-billion by precursors of atmospheric ozone and other oxidants (7, 8).
volume (ppb (v)) multiplied by the number of carbon atoms in
5.2.1 The NMOC concentrations typically found at urban
propane, which is three (3).
sites may range up to 1 ppm C to 3 ppm C or higher. In order
to determine transport of precursors into an area monitoring
4. Summary of Test Method (2-6)
site, measurement of NMOC upwind of the site may be
4.1 An air sample is collected directly from ambient air, necessary. Rural NMOC concentrations originating from areas
free from NMOC sources are likely to be less than a few tenths
using a pre-cleaned sample evacuated passivated canister,
which is then transported to a laboratory. of 1 ppm C.
5.3 Conventional test methods based upon gas chromatog-
4.2 A fixed-volume portion of the sample air is drawn from
the canister at a low flow rate through a silanized glass-bead raphy and qualitative and quantitative species evaluation are
relatively time consuming, sometimes difficult and expensive
filled trap that is cooled to approximately –186 °C with liquid
argon. The cryogenic trap simultaneously collects and concen- in staff time and resources, and are not needed when only a
trates the NMOC using condensation, while allowing the measurement of NMOC is desired. The test method described
nitrogen, oxygen, methane, and other compounds with boiling requires only a simple, cryogenic pre-concentration procedure
points below –186 °C to pass through the trap without reten- followed by direct detection with an FID. This test method
tion. The system is dynamically calibrated so that the volume provides a sensitive and accurate measurement of ambient total
of sample passing through the trap does not have to be NMOC concentrations where speciated data are not required.
quantitatively measured, but must be precisely repeatable Typical uses of this standard test method are as follows.
between the calibration and the analytical phases.
5.4 An application of the test method is the monitoring of
4.3 After the fixed-volume air sample has been drawn the cleanliness of canisters.
through the trap, a helium carrier gas flow is diverted to pass
5.5 Another use of the test method is the screening of
through the trap, in the opposite direction to the sample flow,
canister samples prior to analysis.
and into an FID. When the residual air and methane have been
5.6 Collection of ambient air samples in pressurized canis-
flushed from the trap and the FID baseline restabilizes, the
ters provides the following advantages:
cryogen is removed and the temperature of the trap is raised to
90 °C at 30 °C per minute. 5.6.1 Convenient collection of integrated ambient samples
over a specific time period,
4.4 The organic compounds previously collected on the trap
5.6.2 Capability of remote sampling with subsequent central
re-volatilize and are carried into the FID, resulting in a
laboratory analysis,
response peak or peaks from the FID. The area of the peak or
5.6.3 Ability to ship and store samples, if necessary,
peaks is integrated, and the integrated value is translated to
5.6.4 Unattended sample collection,
concentration units using a previously obtained calibration
curve relating integrated peak areas with known concentrations 5.6.5 Analysis of samples from multiple sites with one
of propane or other calibrant. analytical system,
D5953M − 23
5.6.6 Collection of replicate samples for assessment of 7.1.6 Particulate Matter Filter, inert in-line filter, 2 μm or
measurement precision, and less, or other suitable filter, used to filter the air sample.
5.6.7 Specific hydrocarbon analysis can be performed with
7.1.7 Auxiliary Vacuum Pump or Blower, draws sample air
the same sample system.
through the sample inlet line to reduce inlet residence time to
no greater than 10 s.
6. Interferences
7.1.7.1 Shock mount the pump to minimize vibration.
6.1 In laboratory evaluations, moisture in the sample has
7.1.8 Timer, programmable, and electrically connected to
been found to cause a positive shift in the FID baseline. The
the solenoid valve (7.1.4) and pumps (7.1.2 and 7.1.7), capable
effect of this shift is minimized by carefully selecting the
of controlling the pumps and the solenoid valve.
integration beginning and termination points and adjusting the
7.1.9 Sample Inlet Line, transports the sample air into the
baseline used for calculating the area of the NMOC peaks.
sample system, consisting of stainless steel tubing components.
6.2 With helium as a carrier gas, FID response is uniform
7.2 Sample Canister Cleaning System (Fig. 2).
for most hydrocarbon compounds, but the response can vary
7.2.1 Vacuum Pump, capable of evacuating sample canis-
considerably for other types of organic compounds such as
ter(s) to an absolute pressure of ≤1.69 kPa (29.5 in. Hg).
halogenated and oxygenated compounds.
7.2.2 Manifold, stainless steel manifold with connections
for simultaneously cleaning several canisters.
7. Apparatus
7.2.3 Shut-off Valve(s), nine required.
7.1 Sample Collection System, (Fig. 1).
7.2.4 Pressure Gauge, 0 kPa to 350 kPa (0 psig to
7.1.1 Sample Canister(s), stainless steel, stainless steel elec-
50 psig)—monitors zero-air pressure.
tropolished passivated Summa -polished or silanized vessel(s)
7.2.5 Cryogenic Trap (2 required), U-shaped open tubular
of 4 L to 6 L capacity, used for automatic collection of
trap cooled with liquid argon, used to prevent contamination
integrated air samples.
from back diffusion of oil from vacuum pump, and providing
7.1.1.1 Mark each canister with a unique identification
clean, zero-air to the sample canister(s).
number.
7.2.6 Vacuum Gauge, capable of measuring vacuum in the
7.1.2 Sample Pump, stainless steel, metal bellows type.
manifold to an absolute pressure of 1.69 kPa (29.5 in. Hg
7.1.2.1 Ensure that the pump is free of leaks, and uncon-
vacuum) or less, with scale divisions of 0.07 kPa (0.5 μm Hg).
taminated by oil or organic compounds.
7.2.7 Flow Control Valve, regulates flow of zero-air into the
7.1.2.2 Shock mount the pump to minimize vibration.
canister(s).
7.1.3 Pressure Gauge, 0 kPa to 210 kPa (0 psig to 30 psig).
7.1.4 Solenoid Valve, controls the sample flow to the canis- 7.2.8 Humidifier, water bubbler or other system capable of
ter with negligible temperature rise. providing moisture to the zero-air supply.
7.1.5 Flow Control Device, mass flow controller, micro-
7.2.9 Isothermal Oven, for heating canisters, not shown in
metering valve, or critical orifice, to maintain the sample flow
Fig. 2.
over the sampling period.
7.3 Analytical System (Fig. 3).
7.3.1 FID System, includes flow controls for the FID fuel
and combustion air, temperature control for the FID, and signal
The Summa process is a trademark of Molectrics, Inc., 4000 E. 89th St.,
processing electronics. Set the FID combustion air, hydrogen,
Cleveland, OH 44105.
and helium carrier flow rates according to the manufacturer’s
instructions.
7.3.2 Data Reduction Device, such as a computer, equipped
with data acquisition hardware and software and a laser printer,
or an electronic integrator, with chart recorder, capable of
integrating the area of one or more FID response peaks and
calculating peak area corrected for baseline drift.
7.3.2.1 If a discrete integrator and chart recorder are used,
exercise care to ensure that these components do not interfere
with each other electrically or electronically.
7.3.2.2 Range selector controls on both the integrator and
the FID analyzer may not provide accurate range ratios, so
prepare individual calibration curves for each range.
7.3.2.3 The integrator must be capable of marking the
beginning and ending of peaks, constructing the appropriate
baseline between the start and end of the integration period,
and calculating the peak area.
7.3.3 Cryogenic Trap, constructed from a single piece of
chromatographic-grade stainless steel tubing (3 mm outside
FIG. 1 Sample System for Automatic Collection of Integrated Air
Samples diameter, 2 mm inside diameter), as shown in Fig. 4.
D5953M − 23
FIG. 2 Canister Cleaning System
D5953M − 23
FIG. 3 NMOC Analytical System
D5953M − 23
7.3.10 Vacuum Reservoir, 1 L to 2 L capacity, typically 1 L.
7.3.11 Gas Purifiers (3 required), gas scrubbers containing
Drierite or silica gel and 5A molecular sieve to remove
moisture and organic impurities in the helium, air, and hydro-
gen gas flow. Check the purity of the gas purifiers prior to use
by passing zero-air through them and analyzing the gas in
accordance with 11.4. The gas purifiers are clean if the NMOC
concentration of the emitted gas is below the detection limit of
the test method.
7.3.12 Trap Heating System, chromatographic oven, direct
induction load heater, or other means to heat the trap to 90 °C
at a controlled rate of 30 °C per minute.
7.3.12.1 Repeatable types of heat sources are
recommended, including a temperature-programmed chro-
matograph oven, electrical heating of the trap itself, or any type
of heater that brings the temperature of the trap up to 90 °C in
1 min to 2 min. This is not shown in Fig. 3.
7.3.13 Toggle Shut-Off Valves (4 required), must be leak
free. Two are positioned on each side of the vacuum reservoir
(7.3.10), one at the absolute pressure gauge (7.3.9), and one at
the zero air cylinder (8.5) used for the analytical system leak
FIG. 4 Cryogenic Sample Trap
test (10.1).
7.3.14 Vacuum Pump, general purpose laboratory oil-less
7.3.3.1 Pack the central portion of the trap (70 mm to
diaphragm pump, must be capable of evacuating the vacuum
100 mm) with silanized 180 μm to 250 μm (60 ⁄80 mesh) glass
reservoir (7.3.10) to allow the desired sample volume to be
beads, with small silanized glass wool plugs, to retain the
drawn through the trap.
beads.
7.3.15 Vent, to keep the trap at atmospheric pressure during
7.3.3.2 The arms of the trap must be of such length to permit
trapping.
the beaded portion of the trap to be submerged below the level
7.3.16 Rotameter or Electronic Flow Measurement Device,
of cryogen in the Dewar flask.
verifies that there is vent flow at all times during trapping.
7.3.3.3 Connect the trap directly to the six-port valve (7.3.4)
7.3.17 Three-Way Valve.
to minimize the line length between the trap (7.3.3) and the
7.3.18 Chromatographic-Grade Stainless Steel Tubing and
FID (7.3.1).
Fittings, stainless steel tubing and fittings for interconnections.
7.3.3.4 Mount the trap to allow clearance so the Dewar flask
7.3.18.1 All such materials in contact with the sample,
may be applied and withdrawn to facilitate cooling and heating
analyte, or support gases prior to analysis must be of stainless
of the trap (see 7.3.12).
steel or other inert metal.
7.3.4 Six-Port Valve—Locate the six-port valve and as much
7.3.18.2 Do not use plastic or TFE-fluorocarbon tubing or
of the interconnecting tubing as practical inside an oven or
fittings.
otherwise heat it to 90 °C to minimize wall losses or
7.3.19 Pressure Gauge, capable of reading up to 500 kPa
adsorption/desorption in the connecting tubing. All lines must
(60 psig).
be as short as practical.
NOTE 1—A diaphragm type valve is recommended for use, as standard
8. Reagents and Materials
rotational valves do not typically perform well in many applications of this
test method.
8.1 Warning—Gas cylinders and compressed gas standards
should only be handled in well-ventilated locations, away from
7.3.5 Multistage Pressure Regulators (3 required), standard
sparks and flames. Improper handling of compressed gas
two-stage, stainless steel diaphragm regulators with pressure
cylinders containing air, nitrogen, hydrogen, or helium can
gauges, for helium, air, and hydrogen cylinders.
result in explosion. Rapid release of nitrogen or helium can
7.3.6 Auxilliary Flow or Pressure Regulators (2 required),
result in asphyxiation. Compressed air supports combustion.
to maintain constant flow rates, within 1 mL/min for the helium
Hydrogen is highly flammable and burns with a colorless,
carrier and the hydrogen.
transparent flame. Liquid argon is a freeze hazard as well as an
7.3.7 Fine Needle Valve (2 required)—One adjusts the
asphyxiate.
sample flow rate through the trap, and the other adjusts the
sample flow rate from the canister.
8.2 Gas Cylinders of Helium and Hydrogen, ultrahigh purity
7.3.8 Dewar Flask, holds cryogen used to cool the trap,
grade.
sized to contain the submerged portion of the trap.
8.3 Combustion Air, cylinder containing less than 0.02 ppm
7.3.9 Absolute Pressure Gauge, 0 kPa to 60 kPa (0 mm Hg
(v) hydrocarbons, or equivalent air source.
to 450 mm Hg), with scale divisions of 0.25 kPa (2 mm Hg),
monitors repeatable volumes of sample air through the cryo- 8.4 Propane Calibration Standard, cylinder containing
genic trap. 1 ppm to 100 ppm (v) (3 ppm to 300 ppm C) propane in air,
D5953M − 23
traceable to a National Institute of Standards and Technology 9.2.1 Record the final pressure and close the canister valve,
(NIST) Standard Reference Material (SRM) or to a NIST/EPA- then check the pressure after 24 h. If leak-tight, the pressure
approved Certified Reference Material (CRM). will not have not dropped by more than 15 kPa (2 psig) over
the 24-h period at constant temperature.
8.5 Zero-Air, cylinder containing less than or equal amounts
9.2.2 Record the leak check result on the Sampling Data
of total hydrocarbons as the detection limit of the test method.
Sheet, Fig. 5.
8.5.1 Zero-air may be obtained from a cylinder of zero-
grade compressed air scrubbed with anhydrous calcium sulfate
9.3 Clean the canister(s) as illustrated in Fig. 2.
or silica gel and 5A molecular sieve or activated charcoal, or by
9.3.1 Close all the valves.
catalytic cleanup of ambient air.
9.3.2 Add cryogen (8.6) to both the vacuum pump and
8.5.2 Pass the zero-air used for canister cleaning (9.3)
zero-air supply traps (7.2.5).
through a cryogenic cold trap (7.2.5) for final cleanup, then
9.3.3 Connect the canister(s) (7.1.1) to the manifold (7.2.2).
through a hydrocarbon-free water (8.7) humidifier (7.2.8) (or
Open the vent shut-off valve (E) and the canister valve(s) to
other device).
release any remaining pressure in the canister(s).
8.6 Cryogen (bp −185.7 °C), liquid argon recommended.
9.3.4 Now close the vent shut-off valve (E) and open the
8.6.1 If liquid argon cannot maintain the trap temperature at
vacuum shut-off valve (D).
−185.7 °C due to the location of the laboratory, such as at high
9.3.5 Energize the vacuum pump (7.2.1), open the vacuum
altitudes (where the normal atmospheric pressure is less than
shut-off valves F and H, and evacuate the canister(s) to
101.3 kPa), a mechanical refrigeration system can be used (see
≤1.69 kPa (29.5 in. Hg vacuum) for 30 min with optional
13.5.1).
heating to no more than 100 °C in an isothermal oven (7.2.9).
8.7 Purity of Water—Unless otherwise stated, water shall be
9.4 On a daily basis, or more often if necessary, blow out the
Type II reagent water conforming to Specification D1193.
cryogenic traps (7.2.5) with zero-air (8.5), using valves A and
9. Canister Cleanup and Preparation
I, to remove trapped water from previous canister cleaning
cycles.
9.1 Leak test and clean the canisters (7.1.1) of contaminants
before sample collection.
9.5 Close the vacuum and vacuum gauge shut-off valves (H
9.2 Leak test the canisters by pressurizing them to approxi- and D) and open the zero-air shut-off valves (B and C) to
mately 200 kPa above atmospheric pressure (30 psig) with pressurize the canister(s) with moist zero-air (8.5) to approxi-
zero-air (8.5), using the canister cleaning system (see Fig. 2). mately 200 kPa over atmospheric pressure (30 psig). Hold this
FIG. 5 Example Sampling Data Sheet
D5953M − 23
pressure for 30 min. If a zero gas generator system is used, P V n
F 5 (1)
limit the flow rate to maintain the zero-air quality. t
9.6 Close the zero-air shut-off valve (C) and allow the
where:
canister(s) to vent down to atmospheric pressure through the
F = flow rate, mL/min,
vent shut-off valve (E).
P = canister final absolute pressure ratio,
9.6.1 Close the vent shut-off valve (E).
= (P + P )/P ,
a g a
V = volume of the canister, mL,
9.7 As a blank check of the canister(s) and cleanup
n = number of canisters connected together (for simultane-
procedure, initially analyze the zero-air content of each canis-
ous sample collection),
ter until the cleanup system and canisters are proven to reliably
t = sample period, min,
result in blank tests of NMOC less than the MDL.
P = pressure in canister, kPa above atmospheric pressure,
g
9.7.1 Repeat the last three steps three times, or until the
(psig), and
blank is less than the detection limit of the procedure.
P = standard atmospheric pressure, 101.3 kPa (14.7 psig).
a
9.7.2 Do not use any canister that does not test at below the
MDL. 10.2.2.1 As an example, if one 6–L canister is to be filled to
approximately 100 kPa above atmospheric pressure in 3 h, the
9.8 Re-evacuate the canisters to ≤1.69 kPa (29.5 in. Hg
flow rate would calculated as follows:
vacuum), using the canister cleaning system.
9.8.1 Close the canister valve(s), remove the canister(s) 1001101.3
P 5 5 1.987 (2)
from the canister cleaning system, and cap the canister con- 101.3
nections with stainless steel or brass fittings.
1.987 × 6000 × 1
9.8.2 The canisters are now ready for the collection of air
F 5 5 66 mL/min
samples. Attach identification tags to the neck of each canister
for field notes and chain-of-custody purposes.
10.2.3 Adjust the flow control device (7.1.5) suitable for
9.8.3 Record the canister pressure as initial on the Sampling
maintaining a constant flow at the calculated flow rate into the
Data Sheet (see Fig. 5).
canister over the desired sampling period. This will maintain
9.9 Leak test the sample system and the outlet side of the
an approximately constant flow up to a canister pressure of
sample pump (7.1.2) prior to field use by attaching a vacuum
about 200 kPa (30 psig), after which the flow drops with
gauge (7.2.6) to the canister inlet using a connecting tubing
increasing pressure. At 101.3 kPa above atmospheric pressure
with a tee fitting, capping the pump inlet, and evacuating to
(14.7 psig), the flow will be about 10 % below the initial flow,
approximately 15 Pa (0.1 mm Hg). If the pressure remains at
depending upon pump performance.
60.4 Pa (3 μm Hg) for 15 min, with the pump energized, the
10.2.4 Place a particulate matter filter (7.1.6) in front of the
pump and connecting lines are leak free.
flow control device (7.1.5).
10.2.5 Check the sampling system for contamination by
10. Sampling
filling two evacuated, cleaned canisters (see 10.2) with humidi-
10.1 General:
fied zero-air (8.5) through the sampling system. Analyze the
10.1.1 See Practice D1357 for general sampling procedures.
canisters in accordance with 11.4. The sampling system is free
10.1.2 Choose a flow control device (7.1.5) to provide a
of contamination if the canisters contain less than the detection
constant flow rate such that the canister is pressurized to
limit of the system.
approximately 200 kPa (one atmosphere above ambient
10.2.6 Observe the flow rate into the sampling system
pressure), over the desired sampling period (see 10.2).
during the system contamination. Check to ensure that sample
10.1.3 Use a second canister when a duplicate sample is
flow rate remains relatively constant (610 %) up to about
desired for quality assurance (QA) purposes (see 12.3.4).
100 kPa above atmospheric pressure.
10.1.4 Exercise care in selecting, cleaning, and handling the
sample canisters and sampling apparatus to avoid losses or
NOTE 2—A drop in the flow rate may occur near the end of the sampling
contamination of the samples.
period as the canister pressure approaches its final pressure, depending
upon pump performance.
10.2 Sample Collection:
10.2.1 Assemble the sampling apparatus as shown in Fig. 1,
10.2.7 Reassemble the sampling system.
with the connecting lines between the sample pump (7.1.2) and
10.2.8 Verify that the timer (7.1.8), pumps (7.1.2 and 7.1.7)
the canisters (7.1.1) as short as possible to minimize their
and solenoid valve (7.1.4) are connected and operating prop-
volume.
erly.
10.2.1.1 Purge the sample inlet line (7.1.9) with a flow of
10.2.9 Verify that the timer (7.1.8) is correctly set for the
several L/min, using a small auxiliary vacuum pump (7.1.7), to
desired sample period, and that the solenoid valve (7.1.4) is
minimize the sample residence time.
closed.
10.2.2 Determine the flow rate required to pressurize the
10.2.10 Connect the cleaned, evacuated canister(s) (9.8) to
canisters to approximately 200 kPa (one atmosphere above
the non-contaminated sampling system, by way of the solenoid
ambient pressure or 2 atmospheres absolute pressure) during
the desired sample period, utilizing the following equation: valve (7.1.4), for sample collection.
D5953M − 23
10.2.11 Verify that the solenoid valve (7.1.4) is closed. pressure gauge toggle shut-off valve (C), and put the three-way
Open the canister valve(s). Temporarily connect a small valve in the sample position.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5953M − 16 D5953M − 23
Standard Test Method for
Determination of Non-methane Organic Compounds (NMOC)
in Ambient Air Using Cryogenic Preconcentration and Direct
Flame Ionization Detection
This standard is issued under the fixed designation D5953M; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method presents a procedure for sampling and determination of non-methane organic compounds (NMOC) in
ambient, indoor, or workplace atmospheres.
1.2 This test method describes the collection of integrated whole air samples in silanized or other passivated stainless steel
canisters, and their subsequent laboratory analysis.
1.2.1 This test method describes a procedure for sampling in canisters at final pressures above atmospheric pressure (pressurized
sampling).
1.3 This test method employs a cryogenic trapping procedure for concentration of the NMOC prior to analysis.
1.4 This test method describes the determination of the NMOC by the flame ionization detection (FID), without the use of gas
chromatographic columns and other procedures necessary for species separation.
1.5 The range of this test method is from 20 to 10 00020 ppb C to 10 000 ppb
ppb C (1, 2).
1.6 This test method has a larger uncertainty for some halogenated or oxygenated hydrocarbons than for simple hydrocarbons or
aromatic compounds. This is especially true if there are high concentrations of chlorocarbons or chlorofluorocarbons present.
1.7 The values stated in SI units are to be regarded as standard. The values given in parentheses are mathematical conversions to
inch-pound units that are provided for information only and are not considered standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
This test method is under the jurisdiction of ASTM Committee D22 on Air Quality and is the direct responsibility of Subcommittee D22.03 on Ambient Atmospheres
and Source Emissions.
Current edition approved Oct. 1, 2016March 1, 2023. Published October 2016March 2023. Originally approved in 1996. Last previous edition approved in 20092016 as
D5953M – 96 (2009).D5953M – 16. DOI: 10.1520/D5953M-16.10.1520/D5953M-23.
This test method is based on EPA Compendium Method TO-12: “Determination of Non-Methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic
Pre-Concentration and Direct Flame Ionization Detection (PDFID),” Compendium of Methods for the Determination of Toxic Organic Compounds in Ambient Air, EPA 600
4-89-017, U.S. Environmental Protection Agency, Research Triangle Park, NC, March 1990.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5953M − 23
1.9 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D1356 Terminology Relating to Sampling and Analysis of Atmospheres
D1357 Practice for Planning the Sampling of the Ambient Atmosphere
D5466 Test Method for Determination of Volatile Organic Compounds in Atmospheres (Canister Sampling, Mass Spectrometry
Analysis Methodology)
2.2 Other References:
EPA Method TO-12 Determination of Non-Methane Organic Compounds (NMOC) in Ambient Air Using Cryogenic
Pre-Concentration and Direct Flame Ionization Detection (PDFID)
3. Terminology
3.1 Definitions—For definitions of terms used in this test method, refer to Terminology D1356.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 cryogen—a refrigerant used to obtain very low temperatures in analytical system cryogenic traps.
3.2.1.1 Discussion—
Liquid argon (bp –185.7°C(bp –185.7 °C at standard pressure) is recommended and may be required for use in some applications
of this test method. Cryogens with lower boiling points, such as liquid nitrogen, may be used if the cryogenic trap temperature is
actively maintained at –185°C to avoid the potential for trapping oxygen or methane from air samples.
3.2.2 dynamic calibration—calibration of an analytical system with pollutant concentrations that are generated in a dynamic,
flowing system, such as by quantitative, flow-rate dilution of a high-concentration gas standard with zero gas.
3.2.3 NMOC—non-methane organic compounds.
3.2.3.1 Discussion—
Total non-methane organic compounds are compounds, excluding methane, measured using a flame ionization detector (FID), with
−2
vapor pressures above 10 kPa recovered from canisters.
3.2.4 ppm C and ppb C—concentration units of parts-per-million and parts-per-billion of organic carbon as detected by FID.
3.2.4.1 Discussion—
For example, when calibrating with propane, concentrations of NMOC in samples are equivalent to parts-per-million by volume
(ppm (v)) or parts-per-billion by volume (ppb (v)) multiplied by the number of carbon atoms in propane, which is three (3).
4. Summary of Test Method (2-6)
4.1 An air sample is collected directly from ambient air, using a pre-cleaned sample evacuated passivated canister, which is then
transported to a laboratory.
4.2 A fixed-volume portion of the sample air is drawn from the canister at a low flow rate through a silanized glass-bead filled
trap that is cooled to approximately –186°C–186 °C with liquid argon. The cryogenic trap simultaneously collects and concentrates
the NMOC using condensation, while allowing the nitrogen, oxygen, methane, and other compounds with boiling points below
–186°C–186 °C to pass through the trap without retention. The system is dynamically calibrated so that the volume of sample
passing through the trap does not have to be quantitatively measured, but must be precisely repeatable between the calibration and
the analytical phases.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from United States Environmental Protection Agency (EPA), William Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460,
http://www.epa.gov.
D5953M − 23
4.3 After the fixed-volume air sample has been drawn through the trap, a helium carrier gas flow is diverted to pass through the
trap, in the opposite direction to the sample flow, and into an FID. When the residual air and methane have been flushed from the
trap and the FID baseline restabilizes, the cryogen is removed and the temperature of the trap is raised to 90°C at 30°C90 °C at
30 °C per minute.
4.4 The organic compounds previously collected on the trap re-volatilize and are carried into the FID, resulting in a response peak
or peaks from the FID. The area of the peak or peaks is integrated, and the integrated value is translated to concentration units using
a previously obtained calibration curve relating integrated peak areas with known concentrations of propane or other calibrant.
4.5 The cryogenic trap simultaneously concentrates the NMOC while separating and removing the methane from samples. The
technique thus directly measures NMOC with greater sensitivity than conventional continuous NMOC analyzers due to the
pre-concentration procedure.
4.6 The sample is injected into the hydrogen-rich flame of an FID, where the organic vapors burn, producing ionized molecular
fragments. The resulting ion fragments are then collected and detected. Because this test method employs a helium carrier gas, the
detector response is nearly identical for many hydrocarbon compounds commonly of interest. Thus, the historical short-coming of
varying FID response to aromatic, olefinic, and paraffinic hydrocarbons is minimized. Users are cautioned that the FID is much
less sensitive to most organic compounds containing functional groups such as carbonyls, alcohols, halocarbons, etc. than simple
hydrocarbons.
5. Significance and Use
5.1 Many regulators, industrial processes, and other stakeholders require determination of NMOC in atmospheres.
5.2 Accurate measurements of ambient NMOC concentrations are critical in devising air pollution control strategies and in
assessing control effectiveness because NMOCs are primary precursors of atmospheric ozone and other oxidants (7, 8).
5.2.1 The NMOC concentrations typically found at urban sites may range up to 1 to 3 ppm 1 ppm C to 3 ppm C or higher. In order
to determine transport of precursors into an area monitoring site, measurement of NMOC upwind of the site may be necessary.
Rural NMOC concentrations originating from areas free from NMOC sources are likely to be less than a few tenths of 1 ppm 1 ppm
C.
5.3 Conventional test methods based upon gas chromatography and qualitative and quantitative species evaluation are relatively
time consuming, sometimes difficult and expensive in staff time and resources, and are not needed when only a measurement of
NMOC is desired. The test method described requires only a simple, cryogenic pre-concentration procedure followed by direct
detection with an FID. This test method provides a sensitive and accurate measurement of ambient total NMOC concentrations
where speciated data are not required. Typical uses of this standard test method are as follows.
5.4 An application of the test method is the monitoring of the cleanliness of canisters.
5.5 Another use of the test method is the screening of canister samples prior to analysis.
5.6 Collection of ambient air samples in pressurized canisters provides the following advantages:
5.6.1 Convenient collection of integrated ambient samples over a specific time period,
5.6.2 Capability of remote sampling with subsequent central laboratory analysis,
5.6.3 Ability to ship and store samples, if necessary,
5.6.4 Unattended sample collection,
5.6.5 Analysis of samples from multiple sites with one analytical system,
D5953M − 23
5.6.6 Collection of replicate samples for assessment of measurement precision, and
5.6.7 Specific hydrocarbon analysis can be performed with the same sample system.
6. Interferences
6.1 In laboratory evaluations, moisture in the sample has been found to cause a positive shift in the FID baseline. The effect of
this shift is minimized by carefully selecting the integration beginning and termination points and adjusting the baseline used for
calculating the area of the NMOC peaks.
6.2 With helium as a carrier gas, FID response is uniform for most hydrocarbon compounds, but the response can vary
considerably for other types of organic compounds such as halogenated and oxygenated compounds.
7. Apparatus
7.1 Sample Collection System, (Fig. 1).
7.1.1 Sample Canister(s), stainless steel, stainless steel electropolished passivated Summa -polished or silanized vessel(s) of 4 to
6 L 4 L to 6 L capacity, used for automatic collection of integrated air samples.
7.1.1.1 Mark each canister with a unique identification number.
7.1.2 Sample Pump, stainless steel, metal bellows type.
7.1.2.1 Ensure that the pump is free of leaks, and uncontaminated by oil or organic compounds.
7.1.2.2 Shock mount the pump to minimize vibration.
7.1.3 Pressure Gauge, 0 to 210 kPa (0 to 30 psig).0 kPa to 210 kPa (0 psig to 30 psig).
7.1.4 Solenoid Valve, controls the sample flow to the canister with negligible temperature rise.
7.1.5 Flow Control Device, mass flow controller, micro-metering valve, or critical orifice, to maintain the sample flow over the
sampling period.
FIG. 1 Sample System for Automatic Collection of Integrated Air Samples
The Summa process is a trademark of Molectrics, Inc., 4000 E. 89th St., Cleveland, OH 44105.
D5953M − 23
7.1.6 Particulate Matter Filter, inert in-line filter, 2 μm or less, or other suitable filter, used to filter the air sample.
7.1.7 Auxiliary Vacuum Pump or Blower, draws sample air through the sample inlet line to reduce inlet residence time to no greater
than 10 s.
7.1.7.1 Shock mount the pump to minimize vibration.
7.1.8 Timer, programmable, and electrically connected to the solenoid valve (7.1.4) and pumps (7.1.2 and 7.1.7), capable of
controlling the pumps and the solenoid valve.
7.1.9 Sample Inlet Line, transports the sample air into the sample system, consisting of stainless steel tubing components.
7.2 Sample Canister Cleaning System (Fig. 2).
7.2.1 Vacuum Pump, capable of evacuating sample canister(s) to an absolute pressure of ≤1.69 kPa (29.5 in. Hg).
7.2.2 Manifold, stainless steel manifold with connections for simultaneously cleaning several canisters.
7.2.3 Shut-off Valve(s), nine required.
7.2.4 Pressure Gauge, 0 to 350 kPa (0 to 50 psig)—monitors 0 kPa to 350 kPa (0 psig to 50 psig)—monitors zero-air pressure.
7.2.5 Cryogenic Trap (2 required), U-shaped open tubular trap cooled with liquid argon, used to prevent contamination from back
diffusion of oil from vacuum pump, and providing clean, zero-air to the sample canister(s).
7.2.6 Vacuum Gauge, capable of measuring vacuum in the manifold to an absolute pressure of 1.69 kPa (29.5 in. Hg vacuum) or
less, with scale divisions of 0.07 kPa (0.5 μm Hg).
7.2.7 Flow Control Valve, regulates flow of zero-air into the canister(s).
7.2.8 Humidifier, water bubbler or other system capable of providing moisture to the zero-air supply.
7.2.9 Isothermal Oven, for heating canisters, not shown in Fig. 2.
7.3 Analytical System (Fig. 3).
7.3.1 FID System, includes flow controls for the FID fuel and combustion air, temperature control for the FID, and signal
processing electronics. Set the FID combustion air, hydrogen, and helium carrier flow rates according to the manufacturer’s
instructions.
7.3.2 Data Reduction Device, such as a computer, equipped with data acquisition hardware and software and a laser printer, or
an electronic integrator, with chart recorder, capable of integrating the area of one or more FID response peaks and calculating peak
area corrected for baseline drift.
7.3.2.1 If a discrete integrator and chart recorder are used, exercise care to ensure that these components do not interfere with each
other electrically or electronically.
7.3.2.2 Range selector controls on both the integrator and the FID analyzer may not provide accurate range ratios, so prepare
individual calibration curves for each range.
7.3.2.3 The integrator must be capable of marking the beginning and ending of peaks, constructing the appropriate baseline
between the start and end of the integration period, and calculating the peak area.
7.3.3 Cryogenic Trap, constructed from a single piece of chromatographic-grade stainless steel tubing (3 mm outside diameter,
2 mm inside diameter), as shown in Fig. 4.
D5953M − 23
FIG. 2 Canister Cleaning System
D5953M − 23
FIG. 3 NMOC Analytical System
D5953M − 23
FIG. 4 Cryogenic Sample Trap
7.3.3.1 Pack the central portion of the trap (70 to 100 mm) with silanized 180 to 250 μm(70 mm to 100 mm) with silanized 180 μm
to 250 μm (60 (60/80 ⁄80 mesh) glass beads, with small silanized glass wool plugs, to retain the beads.
7.3.3.2 The arms of the trap must be of such length to permit the beaded portion of the trap to be submerged below the level of
cryogen in the Dewar flask.
7.3.3.3 Connect the trap directly to the six-port valve (7.3.4) to minimize the line length between the trap (7.3.3) and the FID
(7.3.1).
7.3.3.4 Mount the trap to allow clearance so the Dewar flask may be applied and withdrawn to facilitate cooling and heating of
the trap (see 7.3.12).
7.3.4 Six-Port Valve—Locate the six-port valve and as much of the interconnecting tubing as practical inside an oven or otherwise
heat it to 90°C90 °C to minimize wall losses or adsorption/desorption in the connecting tubing. All lines must be as short as
practical.
NOTE 1—A diaphragm type valve is recommended for use, as standard rotational valves do not typically perform well in many applications of this test
method.
7.3.5 Multistage Pressure Regulators (3 required), standard two-stage, stainless steel diaphragm regulators with pressure gauges,
for helium, air, and hydrogen cylinders.
7.3.6 Auxilliary Flow or Pressure Regulators (2 required), to maintain constant flow rates, within 1 mL/min for the helium carrier
and the hydrogen.
7.3.7 Fine Needle Valve (2 required)—One adjusts the sample flow rate through the trap, and the other adjusts the sample flow
rate from the canister.
7.3.8 Dewar Flask, holds cryogen used to cool the trap, sized to contain the submerged portion of the trap.
7.3.9 Absolute Pressure Gauge, 0 to 60 kPa (0 to 450 mm 0 kPa to 60 kPa (0 mm Hg to 450 mm Hg), with scale divisions of 0.25
kPa (2 mm Hg), monitors repeatable volumes of sample air through the cryogenic trap.
7.3.10 Vacuum Reservoir, 1 to 2 L capacity, typically 1 L.1 L to 2 L capacity, typically 1 L.
D5953M − 23
7.3.11 Gas Purifiers (3 required), gas scrubbers containing Drierite or silica gel and 5A molecular sieve to remove moisture and
organic impurities in the helium, air, and hydrogen gas flows.flow. Check the purity of the gas purifiers prior to use by passing
zero-air through them and analyzing the gas in accordance with 11.4. The gas purifiers are clean if the NMOC concentration of
the emitted gas is below the detection limit of the test method.
NOTE 2—Check the purity of the gas purifiers prior to use by passing zero-air through them and analyzing the gas in accordance with 11.4. The gas
purifiers are clean if the NMOC concentration of the emitted gas is below the detection limit of the test method.
7.3.12 Trap Heating System, chromatographic oven, direct induction load heater, or other means to heat the trap to 90°C90 °C at
a controlled rate of 30°C30 °C per minute.
7.3.12.1 Repeatable types of heat sources are recommended, including a temperature-programmed chromatograph oven, electrical
heating of the trap itself, or any type of heater that brings the temperature of the trap up to 90°C in 190 °C in 1 min to 2 min. This
is not shown in Fig. 3.
7.3.13 Toggle Shut-Off Valves (4 required), must be leak free. Two are positioned on each side of the vacuum reservoir (7.3.10),
one at the absolute pressure gauge (7.3.9), and one at the zero air cylinder (8.5) used for the analytical system leak test (10.1).
7.3.14 Vacuum Pump, general purpose laboratory oil-less diaphragm pump, must be capable of evacuating the vacuum reservoir
(7.3.10) to allow the desired sample volume to be drawn through the trap.
7.3.15 Vent, to keep the trap at atmospheric pressure during trapping.
7.3.16 Rotameter or Electronic Flow Measurement Device, verifies that there is vent flow at all times during trapping.
7.3.17 Three-Way Valve.
7.3.18 Chromatographic-Grade Stainless Steel Tubing and Fittings, stainless steel tubing and fittings for interconnections.
7.3.18.1 All such materials in contact with the sample, analyte, or support gases prior to analysis must be of stainless steel or other
inert metal.
7.3.18.2 Do not use plastic or TFE-fluorocarbon tubing or fittings.
7.3.19 Pressure Gauge, capable of reading up to 500 kPa (60 psig).
8. Reagents and Materials
8.1 Warning—Gas cylinders and compressed gas standards should only be handled in well-ventilated locations, away from sparks
and flames. Improper handling of compressed gas cylinders containing air, nitrogen, hydrogen, or helium can result in explosion.
Rapid release of nitrogen or helium can result in asphyxiation. Compressed air supports combustion. Hydrogen is highly flammable
and burns with a colorless, transparent flame. Liquid argon is a freeze hazard as well as an asphyxiate.
8.2 Gas Cylinders of Helium and Hydrogen, ultrahigh purity grade.
8.3 Combustion Air, cylinder containing less than 0.02 ppm (v) hydrocarbons, or equivalent air source.
8.4 Propane Calibration Standard, cylinder containing 1 to 100 ppm (v) (31 ppm to 100 ppm (v) (3 ppm to 300 ppm C) propane
in air, traceable to a National Institute of Standards and Technology (NIST) Standard Reference Material (SRM) or to a
NIST/EPA-approved Certified Reference Material (CRM).
8.5 Zero-Air, cylinder containing less than or equal amounts of total hydrocarbons as the detection limit of the test method.
8.5.1 Zero-air may be obtained from a cylinder of zero-grade compressed air scrubbed with anhydrous calcium sulfate or silica
gel and 5A molecular sieve or activated charcoal, or by catalytic cleanup of ambient air.
D5953M − 23
8.5.2 Pass the zero-air used for canister cleaning (9.3) through a cryogenic cold trap (7.2.5) for final cleanup, then through a
hydrocarbon-free water (8.7) humidifier (7.2.8) (or other device).
8.6 Cryogen (bp −185.7°C),−185.7 °C), liquid argon recommended.
8.6.1 If liquid argon cannot maintain the trap temperature at −185.7°C−185.7 °C due to the location of the laboratory, such as at
high altitudes (where the normal atmospheric pressure is less than 101.3 kPa), a mechanical refrigeration system can be used (see
13.5.1).
8.7 Purity of Water—Unless otherwise stated, water shall be Type II reagent water conforming to Specification D1193.
9. Canister Cleanup and Preparation
9.1 Leak test and clean the canisters (7.1.1) of contaminants before sample collection.
9.2 Leak test the canisters by pressurizing them to approximately 200 kPa above atmospheric pressure (30 psig) with zero-air
(8.5), using the canister cleaning system (see Fig. 2).
9.2.1 Record the final pressure and close the canister valve, then check the pressure after 24 h. If leak-tight, the pressure will not
have not dropped by more than 15 kPa (2 psig) over the 24-h period at constant temperature.
9.2.2 Record the leak check result on the Sampling Data Sheet, Fig. 5.
9.3 Clean the canister(s) as illustrated in Fig. 2.
9.3.1 Close all the valves.
FIG. 5 Example Sampling Data Sheet
D5953M − 23
9.3.2 Add cryogen (8.6) to both the vacuum pump and zero-air supply traps (7.2.5).
9.3.3 Connect the canister(s) (7.1.1) to the manifold (7.2.2). Open the vent shut-off valve (E) and the canister valve(s) to release
any remaining pressure in the canister(s).
9.3.4 Now close the vent shut-off valve (E) and open the vacuum shut-off valve (D).
9.3.5 Energize the vacuum pump (7.2.1), open the vacuum shut-off valves F and H, and evacuate the canister(s) to ≤1.69 kPa (29.5
in. ≤1.69 kPa (29.5 in. Hg vacuum) for 30 minutesmin with optional heating to no more than 100°C100 °C in an isothermal oven
(7.2.9).
9.4 On a daily basis, or more often if necessary, blow out the cryogenic traps (7.2.5) with zero-air (8.5), using valves A and I, to
remove trapped water from previous canister cleaning cycles.
9.5 Close the vacuum and vacuum gauge shut-off valves (H and D) and open the zero-air shut-off valves (B and C) to pressurize
the canister(s) with moist zero-air (8.5) to approximately 200 kPa over atmospheric pressure (30 psig). Hold this pressure for 30
minutes.min. If a zero gas generator system is used, limit the flow rate to maintain the zero-air quality.
9.6 Close the zero-air shut-off valve (C) and allow the canister(s) to vent down to atmospheric pressure through the vent shut-off
valve (E).
9.6.1 Close the vent shut-off valve (E).
9.7 As a blank check of the canister(s) and cleanup procedure, initially analyze the zero-air content of each canister until the
cleanup system and canisters are proven to reliably result in blank tests of NMOC less than the MDL.
9.7.1 Repeat the last three steps three times, or until the blank is less than the detection limit of the procedure.
9.7.2 Do not use any canister that does not test at below the MDL.
9.8 Re-evacuate the canisters to ≤1.69 kPa (29.5 in. Hg vacuum), using the canister cleaning system.
9.8.1 Close the canister valve(s), remove the canister(s) from the canister cleaning system, and cap the canister connections with
stainless steel or brass fittings.
9.8.2 The canisters are now ready for the collection of air samples. Attach identification tags to the neck of each canister for field
notes and chain-of-custody purposes.
9.8.3 Record the canister pressure as initial on the Sampling Data Sheet (see Fig. 5).
9.9 Leak test the sample system and the outlet side of the sample pump (7.1.2) prior to field use by attaching a vacuum gauge
(7.2.6) to the canister inlet using a connecting tubing with a tee fitting, capping the pump inlet, and evacuating to approximately
15 Pa (0.1 mm Hg). If the pressure remains at 60.4 Pa (3 μm Hg) for 15 min, with the pump energized, the pump and connecting
lines are leak free.
10. Sampling
10.1 General:
10.1.1 See Practice D1357 for general sampling procedures.
10.1.2 Choose a flow control device (7.1.5) to provide a constant flow rate such that the canister is pressurized to approximately
200 kPa (one atmosphere above ambient pressure), over the desired sampling period (see 10.2).
10.1.3 Use a second canister when a duplicate sample is desired for quality assurance (QA) purposes (see 12.3.4).
D5953M − 23
10.1.4 Exercise care in selecting, cleaning, and handling the sample canisters and sampling apparatus to avoid losses or
contamination of the samples.
10.2 Sample Collection:
10.2.1 Assemble the sampling apparatus as shown in Fig. 1, with the connecting lines between the sample pump (7.1.2) and the
canisters (7.1.1) as short as possible to minimize their volume.
10.2.1.1 Purge the sample inlet line (7.1.9) with a flow of several L/min, using a small auxiliary vacuum pump (7.1.7), to minimize
the sample residence time.
10.2.2 Determine the flow rate required to pressurize the canisters to approximately 200 kPa (one atmosphere above ambient
pressure or 2 atmospheres absolute pressure) during the desired sample period, utilizing the following equation:
P V n
F 5 (1)
t
where:
F = flow rate, mL/min,
P = canister final absolute pressure ratio,
= (P + P )/P ,
a g a
V = volume of the canister, mL,
n = number of canisters connected together (for simultaneous sample collection),
t = sample period, min,
P = pressure in canister, kPa above atmospheric pressure, (psig), and
g
P = standard atmospheric pressure, 101.3 kPa (14.7 psig).
a
10.2.2.1 As an example, if one 6–L canister is to be filled to approximately 100 kPa above atmospheric pressure in 3 h, the flow
rate would calculated as follows:
1001101.3
P 5 5 1.987 (2)
101.3
1.987 ×6000 ×1
F 5 5 66 mL/min
10.2.3 Adjust the flow control device (7.1.5) suitable for maintaining a constant flow at the calculated flow rate into the canister
over the desired sampling period. This will maintain an approximately constant flow up to a canister pressure of about 200 kPa
(30 psig), after which the flow drops with increasing pressure. At 101.3 kPa above atmospheric pressure (14.7 psig), the flow will
be about 10 % below the initial flow, depending upon pump performance.
10.2.4 Place a particulate matter filter (7.1.6) in front of the flow control device (7.1.5).
10.2.5 Check the sampling system for contamination by filling two evacuated, cleaned canisters (see 10.2) with humidified
zero-air (8.5) through the sampling system. Analyze the canisters in accordance with 11.4. The sampling system is free of
contamination if the canisters contain less than the detection limit of the system.
10.2.6 Observe the flow rate into the sampling system during the system contamination. Check to ensure that sample flow rate
remains relatively constant (610 %) up to about 100 kPa 100 kPa above atmospheric pressure.
NOTE 2—A drop in the flow rate may occur near the end of the sampling period as the canister pressure approaches its final pressure, depending upon
pump performance.
10.2.7 Reassemble the sampling system.
10.2.8 Verify that the timer (7.1.8), pumps (7.1.2 and 7.1.7) and solenoid valve (7.1.4) are connected and operating properly.
D5953M − 23
10.2.9 Verify that the timer (7.1.8) is correctly set for the desired sample period, and that the solenoid valve (7.1.4) is closed.
10.2.10 Connect the cleaned, evacuated canister(s) (9.8) to the non-contaminated sampling
...



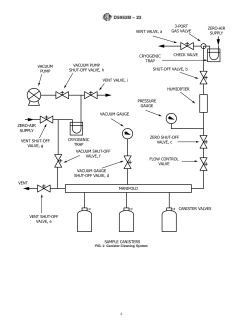




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...