ASTM D6667-21
(Test Method)Standard Test Method for Determination of Total Volatile Sulfur in Gaseous Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet Fluorescence
Standard Test Method for Determination of Total Volatile Sulfur in Gaseous Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet Fluorescence
SIGNIFICANCE AND USE
4.1 The sulfur content of LPG, used for fuel purposes, contributes to SOx emissions and can lead to corrosion in engine and exhaust systems. Some process catalysts used in petroleum and chemical refining can be poisoned by sulfur bearing materials in the feed stocks. This test method can be used to determine sulfur in process feeds, to measure sulfur in finished products, and can also be used for compliance determinations when acceptable to a regulatory authority.
SCOPE
1.1 This test method covers the determination of total volatile sulfur in gaseous hydrocarbons and liquefied petroleum (LP) gases. It is applicable to analysis of natural, processed, and final product materials. Precision has been determined for sulfur in gaseous hydrocarbons in the range of 1 mg/kg to 100 mg/kg and for sulfur in LP gases in the range of 1 mg/kg to 196 mg/kg (Note 1).
Note 1: An estimate of pooled limit of quantification (PLOQ), information regarding sample stability and other general information derived from the interlaboratory studies on precision can be referenced in the ASTM research reports.2,3
1.2 This test method may not detect sulfur compounds that do not vaporize under the conditions of the test.
1.3 This test method is applicable for total volatile sulfur determination in LP gases containing less than 0.35 % (mass/mass) halogen(s).
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See 3.1 and Sections 6 and 7 for specific warning statements.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2021
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.03 - Elemental Analysis
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Apr-2020
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Refers
ASTM D5287-08(2015) - Standard Practice for Automatic Sampling of Gaseous Fuels (Withdrawn 2024) - Effective Date
- 01-Jun-2015
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Jun-2013
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-May-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Nov-2009
Overview
ASTM D6667-21: Standard Test Method for Determination of Total Volatile Sulfur in Gaseous Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet Fluorescence is a key standard developed by ASTM International. This method outlines a reliable procedure for quantifying total volatile sulfur content in gaseous hydrocarbons-including natural gas-and liquefied petroleum (LP) gases such as propane and butane. Using ultraviolet (UV) fluorescence, this test method ensures consistent analysis of sulfur levels to help manage compliance, product quality, and environmental impact in the petroleum and gas sectors.
Key Topics
Scope and Applicability
- Measures total volatile sulfur in gaseous hydrocarbons and LP gases.
- Applicable to natural, processed, and finished product materials.
- Precision determined for sulfur concentrations ranging from 1 mg/kg to 100 mg/kg in gases and 1 mg/kg to 196 mg/kg in LP gases.
- Not suitable for sulfur compounds that do not vaporize under test conditions.
- Best for LP gases with halogen levels below 0.35% (mass/mass).
- SI units are standard-other units are not included.
Significance and Use
- Sulfur in LPG fuels contributes to SOx emissions and can cause corrosion in engines and exhaust systems.
- Key for protecting process catalysts in petroleum and chemical refining from sulfur poisoning.
- Used for process feed analysis, finished product measurement, and regulatory compliance verification.
Test Method
- Employs UV fluorescence detection after combustion of the sample to quantify sulfur as sulfur dioxide.
- Involves careful sample handling, calibration with certified standards, and stringent safety precautions due to flammable materials and high temperature operation.
- Results should be quality controlled daily with proper calibration factors and appropriate statistical process control.
Safety and Quality
- Addresses safety, health, and environmental considerations-users must establish best practices for safe operation.
- Requires verification of apparatus integrity and regular quality control monitoring.
Applications
ASTM D6667-21 is widely used in various applications, primarily within the oil, gas, and petrochemical industries:
Regulatory Compliance
- Supports monitoring and reporting sulfur levels to adhere to emission regulations and fuel standards.
Petroleum Refining
- Protects sensitive catalysts from sulfur contamination in process feeds, ensuring catalyst longevity and product yield.
Product Quality Control
- Verifies sulfur content in final LPG or hydrocarbon products to meet industry and customer specifications.
Environmental Management
- Aids in reducing SOx emissions linked to fuel combustion by enabling proper sulfur quantification.
Operational Safety
- Minimizes risk of equipment corrosion and prolongs the lifespan of engines and exhaust systems through accurate sulfur measurement.
Related Standards
Several other ASTM and industry standards complement or are referenced by ASTM D6667-21 for sampling, calibration, and hydrocarbon analysis:
- ASTM D1070 - Test Methods for Relative Density of Gaseous Fuels
- ASTM D1265 - Practice for Sampling Liquefied Petroleum (LP) Gases, Manual Method
- ASTM D2163 - Test Method for Determination of Hydrocarbons in LP Gases and Propane/Propene Mixtures by Gas Chromatography
- ASTM D2421 - Practice for Interconversion of Analysis of C and Lighter Hydrocarbons to Gas-Volume, Liquid-Volume, or Mass Basis
- ASTM D2598 - Practice for Calculation of Certain Physical Properties of LP Gases from Compositional Analysis
- ASTM D3700 - Practice for Obtaining LPG Samples Using a Floating Piston Cylinder
- ASTM D5287 - Practice for Automatic Sampling of Gaseous Fuels
- ASTM D6299 - Practice for Statistical Quality Assurance and Control Charting Techniques
- GPA 2166 - Obtaining Natural Gas Samples for Analysis by Gas Chromatography
Summary
Adopting ASTM D6667-21 ensures reliable and accurate measurement of total volatile sulfur in gaseous hydrocarbons and LPG. This test method supports industry efforts to manage sulfur-related risks, maintain product integrity, and comply with strict environmental and safety regulations. It is an essential standard for laboratories and facilities engaged in fuel quality testing, petrochemical processing, and environmental monitoring.
Keywords: ASTM D6667-21, volatile sulfur, LPG, gaseous hydrocarbons, ultraviolet fluorescence, sulfur analysis, petroleum standards, emissions, quality control.
Buy Documents
ASTM D6667-21 - Standard Test Method for Determination of Total Volatile Sulfur in Gaseous Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet Fluorescence
REDLINE ASTM D6667-21 - Standard Test Method for Determination of Total Volatile Sulfur in Gaseous Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet Fluorescence
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D6667-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Total Volatile Sulfur in Gaseous Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet Fluorescence". This standard covers: SIGNIFICANCE AND USE 4.1 The sulfur content of LPG, used for fuel purposes, contributes to SOx emissions and can lead to corrosion in engine and exhaust systems. Some process catalysts used in petroleum and chemical refining can be poisoned by sulfur bearing materials in the feed stocks. This test method can be used to determine sulfur in process feeds, to measure sulfur in finished products, and can also be used for compliance determinations when acceptable to a regulatory authority. SCOPE 1.1 This test method covers the determination of total volatile sulfur in gaseous hydrocarbons and liquefied petroleum (LP) gases. It is applicable to analysis of natural, processed, and final product materials. Precision has been determined for sulfur in gaseous hydrocarbons in the range of 1 mg/kg to 100 mg/kg and for sulfur in LP gases in the range of 1 mg/kg to 196 mg/kg (Note 1). Note 1: An estimate of pooled limit of quantification (PLOQ), information regarding sample stability and other general information derived from the interlaboratory studies on precision can be referenced in the ASTM research reports.2,3 1.2 This test method may not detect sulfur compounds that do not vaporize under the conditions of the test. 1.3 This test method is applicable for total volatile sulfur determination in LP gases containing less than 0.35 % (mass/mass) halogen(s). 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See 3.1 and Sections 6 and 7 for specific warning statements. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The sulfur content of LPG, used for fuel purposes, contributes to SOx emissions and can lead to corrosion in engine and exhaust systems. Some process catalysts used in petroleum and chemical refining can be poisoned by sulfur bearing materials in the feed stocks. This test method can be used to determine sulfur in process feeds, to measure sulfur in finished products, and can also be used for compliance determinations when acceptable to a regulatory authority. SCOPE 1.1 This test method covers the determination of total volatile sulfur in gaseous hydrocarbons and liquefied petroleum (LP) gases. It is applicable to analysis of natural, processed, and final product materials. Precision has been determined for sulfur in gaseous hydrocarbons in the range of 1 mg/kg to 100 mg/kg and for sulfur in LP gases in the range of 1 mg/kg to 196 mg/kg (Note 1). Note 1: An estimate of pooled limit of quantification (PLOQ), information regarding sample stability and other general information derived from the interlaboratory studies on precision can be referenced in the ASTM research reports.2,3 1.2 This test method may not detect sulfur compounds that do not vaporize under the conditions of the test. 1.3 This test method is applicable for total volatile sulfur determination in LP gases containing less than 0.35 % (mass/mass) halogen(s). 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See 3.1 and Sections 6 and 7 for specific warning statements. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6667-21 is classified under the following ICS (International Classification for Standards) categories: 19.100 - Non-destructive testing; 75.160.30 - Gaseous fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6667-21 has the following relationships with other standards: It is inter standard links to ASTM D6299-23a, ASTM D1265-23a, ASTM F307-13(2020), ASTM D6299-17b, ASTM D6299-17a, ASTM D6299-17, ASTM D5287-08(2015), ASTM D2163-14, ASTM D6299-13e1, ASTM F307-13, ASTM D3700-12, ASTM D1070-03(2010), ASTM D6299-10e2, ASTM D6299-10, ASTM D6299-09. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6667-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:D6667 −21
Standard Test Method for
Determination of Total Volatile Sulfur in Gaseous
Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet
Fluorescence
This standard is issued under the fixed designation D6667; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 This test method covers the determination of total
Barriers to Trade (TBT) Committee.
volatile sulfur in gaseous hydrocarbons and liquefied petro-
leum (LP) gases. It is applicable to analysis of natural,
2. Referenced Documents
processed, and final product materials. Precision has been
2.1 ASTM Standards:
determined for sulfur in gaseous hydrocarbons in the range of
D1070 Test Methods for Relative Density of Gaseous Fuels
1 mg⁄kg to 100 mg⁄kg and for sulfur in LP gases in the range
D1265 Practice for Sampling Liquefied Petroleum (LP)
of 1 mg⁄kg to 196 mg⁄kg (Note 1).
Gases, Manual Method
NOTE 1—An estimate of pooled limit of quantification (PLOQ),
D2163 Test Method for Determination of Hydrocarbons in
information regarding sample stability and other general information
Liquefied Petroleum (LP) Gases and Propane/Propene
derived from the interlaboratory studies on precision can be referenced in
2,3
Mixtures by Gas Chromatography
the ASTM research reports.
D2421 Practice for Interconversion of Analysis of C and
1.2 This test method may not detect sulfur compounds that
Lighter Hydrocarbons to Gas-Volume, Liquid-Volume, or
do not vaporize under the conditions of the test.
Mass Basis
1.3 This test method is applicable for total volatile sulfur
D2598 Practice for Calculation of Certain Physical Proper-
determination in LP gases containing less than 0.35 % (mass/
ties of Liquefied Petroleum (LP) Gases from Composi-
mass) halogen(s).
tional Analysis
D3700 Practice for Obtaining LPG Samples Using a Float-
1.4 The values stated in SI units are to be regarded as
ing Piston Cylinder
standard. No other units of measurement are included in this
D5287 Practice for Automatic Sampling of Gaseous Fuels
standard.
D6299 Practice for Applying Statistical Quality Assurance
1.5 This standard does not purport to address all of the
and Control Charting Techniques to Evaluate Analytical
safety concerns, if any, associated with its use. It is the
Measurement System Performance
responsibility of the user of this standard to establish appro-
F307 Practice for Sampling Pressurized Gas for Gas Analy-
priate safety, health, and environmental practices and deter-
sis
mine the applicability of regulatory limitations prior to use.
2.2 Gas Processor Association (GPA) Standards:
See 3.1 and Sections 6 and 7 for specific warning statements.
GPA 2166 Obtaining Natural Gas Samples for Analysis by
1.6 This international standard was developed in accor-
Gas Chromatography
dance with internationally recognized principles on standard-
GPA 2174 Obtaining Liquid Hydrocarbon Samples for
ization established in the Decision on Principles for the
Analysis by Gas Chromatography
3. Summary of Test Method
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
3.1 Aheatedsamplevalveisusedtoinjectgaseoussamples.
Subcommittee D02.03 on Elemental Analysis.
Liquefied petroleum gas (LPG) samples are injected by a
Current edition approved April 1, 2021. Published May 2021. Originally
approved in 2001. Last previous edition approved in 2019 as D6667 – 14 (2019).
DOI: 10.1520/D6667-21.
2 4
Supporting data have been filed at ASTM International Headquarters and may For referenced ASTM standards, visit the ASTM website, www.astm.org, or
beobtainedbyrequestingResearchReportRR:D02-1506.ContactASTMCustomer contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Service at service@astm.org. Standards volume information, refer to the standard’s Document Summary page on
Supporting data have been filed at ASTM International Headquarters and may the ASTM website.
beobtainedbyrequestingResearchReportRR:D02-1784.ContactASTMCustomer Available from Gas ProcessorsAssociation (GPA), 6526 E. 60th St.,Tulsa, OK
Service at service@astm.org. 74145.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6667−21
sample valve connected to a heated expansion chamber. The heated oxidation zone of the furnace. The combustion tube
gaseous sample then enters a high temperature combustion shall have side arms for the introduction of oxygen and carrier
tube where sulfur is oxidized to sulfur dioxide (SO)inan gas. The oxidation section shall be large enough (see Fig. 1)to
oxygen rich atmosphere. Water produced during the sample ensure complete combustion of the sample (see 11.3). Fig. 1
combustion is removed and the sample combustion gases are depicts a typical combustion tube. Other configurations are
next exposed to ultraviolet (UV) light. The SO absorbs the acceptable when precision is not degraded.
energy from the UV light and is converted to an excited sulfur
5.3 Flow Control—The apparatus shall be equipped with
*
dioxide (SO ). Fluorescence emitted from the excited SO as
2 2
flow controllers capable of maintaining a constant supply of
*
it returns to a stable state SO is detected by a photomultiplier
oxygen and carrier gas at the specified rates.
tube,theresultingsignalisameasureofthesulfurcontainedin
5.4 Drier Tube—The apparatus shall be equipped with a
the sample. (Warning—Exposure to excessive quantities of
mechanism for the removal of water vapor formed during
ultraviolet light is injurious to health. The operator shall avoid
sample combustion. This can be accomplished with a mem-
exposing their person, especially their eyes, not only to direct
brane drying tube, or a permeation dryer that utilizes a
UV light but also to secondary or scattered radiation that is
selective capillary action for water removal.
present.)
5.5 UV Fluorescence Detector—A quantitative detector ca-
4. Significance and Use
pable of measuring light emitted from the fluorescence of
sulfur dioxide by UV light.
4.1 The sulfur content of LPG, used for fuel purposes,
contributes to SOx emissions and can lead to corrosion in
5.6 Sample Inlet System—The system provides a heated
engine and exhaust systems. Some process catalysts used in
gas-sampling valve, or a LP gas-sampling valve, or both, with
petroleum and chemical refining can be poisoned by sulfur
a heated expansion chamber, connected to the inlet of the
bearing materials in the feed stocks. This test method can be
oxidation area, Fig. 2. The system is swept by an inert carrier
used to determine sulfur in process feeds, to measure sulfur in
gas and shall be capable of allowing the quantitative delivery
finished products, and can also be used for compliance deter-
of the material to be analyzed into the oxidation zone at a
minations when acceptable to a regulatory authority.
controlled and repeatable rate of approximately 30 mL⁄min.
Fig. 3 provides an example.
5. Apparatus
5.7 Strip Chart Recorder, equivalent electronic data logger,
5.1 Furnace—An electric furnace held at a temperature
integrator or, recorder (optional).
(1075 °C 6 25 °C) sufficient to pyrolyze the entire sample and
oxidize sulfur to SO . 6. Reagents
5.2 Combustion Tube—A quartz combustion tube con- 6.1 Purity of Reagents—Reagent grade chemicals shall be
structed to allow the direct injection of the sample into the used in tests. Unless otherwise indicated, it is intended that all
FIG. 1Example of a Typical Direct Inject Quartz Pyrolysis Tube
D6667−21
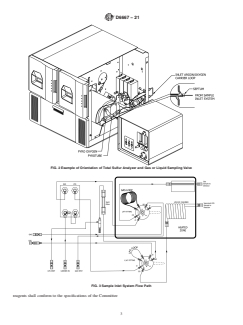
FIG. 2Example of Orientation of Total Sulfur Analyzer and Gas or Liquid Sampling Valve
FIG. 3Sample Inlet System Flow Path
reagents shall conform to the specifications of the Committee
D6667−21
on Analytical Reagents of the American Chemical Society, 8.2 If the sample is not used immediately, then thoroughly
where such specifications are available. Other grades may be mix it in its container prior to taking a test specimen. The use
used, provided it is first ascertained that the reagent is of of segregated or specially treated sample containers can help
sufficiently high purity to permit its use without lessening the reduce sample cross-contamination and improve sample sta-
accuracy of the determination. bility.
6.2 Inert Gas—Argon or helium only, high purity grade
9. Preparation of Apparatus
(that is, chromatography or zero grade), 99.998 % min purity,
9.1 Assembleandchecktheapparatusforleaksaccordingto
moisture 5 mg⁄kg max. (Warning—Argon or helium may be a
manufacturer’s instructions.
compressed gas under high pressure (7.1)).
9.2 Typical apparatus adjustments and conditions are listed
6.3 Oxygen—High purity (that is chromatography or zero
in Table 2.
grade), 99.75 % min purity, moisture 5 mg⁄kg max, dried over
molecular sieves. (Warning—Oxygen vigorously accelerates 9.3 Adjust instrument sensitivity and baseline stability and
combustion and may be compressed gas under high pressure
perform instrument-blanking procedures following manufac-
(7.1)).
turer’s guidelines.
6.4 Calibration Standards—Certified calibration standards
10. Calibration and Standardization
from commercial sources or calibration gases prepared using
10.1 Consult Table 3 and select a calibration range based on
certified permeation tube devices are required. Table 1 lists the
the anticipated sulfur concentrations present in samples to be
sulfur source material and diluent matrices used during the
analyzed, preferably using a sulfur compound and a diluent
inter-laboratory study (Notes 2 and 3).
type representative of the samples to be analyzed (Note 4).
NOTE 2—Other sulfur sources and diluent materials may be used if
Table 3 is representative of typical ranges, but narrower ranges
precision and accuracy are not degraded.
than those indicated may be used if desired. However, the
NOTE 3—Calibration standards are typically re-mixed and re-certified
method precision using narrower ranges than those indicated
on a regular basis depending upon frequency of use and age. These
calibration standards may have a useful life of about 6 months to
has not been determined. Ensure the standards used for
12 months.
calibration bracket the concentrations of the samples being
6.5 Quality Control (QC) Samples—preferably are portions analyzed.
of one or more gas or LP gas materials that are stable and
NOTE 4—The number of standards used per curve may vary.
representative of the samples of interest.
10.2 With the sample valve in the load position, connect the
pressurized sample container to the sample valve of the sample
7. Hazards
inlet system.
7.1 High temperature, flammable hydrocarbons, and gases
10.3 Obtain a quantitative measurement of the injected
under high pressures occur in the test method. Use materials
material by filling the sample loop of the sample valve system
that are rated for containing these pressurized hydrocarbons in
for the matrix being analyzed (see Table 2)(Notes 5 and 6).
all sample containers and sample transfer apparatus. Exercise
extra care when using flammable materials near the oxidative
NOTE 5—Injection of a constant or similar sample size for all materials
furnace.
analyzed in a selected operating range promotes consistent combustion
conditions and may simplify result calculations.
NOTE 6—An automatic sample transfer and injection device may be
8. Sampling
used.
8.1 Obtain a sample in accordance with Practices F307,
10.3.1 Flush the sample loop with sufficient calibrant to
D1265, D3700, D5287, or GPA 2174 or GPA 2166. Analyze
assure that the material to be injected is representative.
samples as soon as possible after taking from bulk supplies to
10.3.2 For LPG samples, if bubbles are present in the
prevent loss of sulfur or contamination due to exposure or
viewable portion of the liquid column, flush the sample loop to
contact with sample containers.
introduce a new liquid-full sample portion.
10.4 Start the analyzer and inject the calibration material
according to the manufacturer’s instructions.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington, 10.5 Calibrate the instrument using one of the following
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
techniques.
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. TABLE 2 Typical Operating Conditions
Sample inlet system temperature 85 °C ± 20 °C
Sample injection system carrier gas 25 mL ⁄min to 30 mL ⁄min
TABLE 1 Typical Standard Materials
Furnace temperature 1075 °C ± 25 °C
Sulfur Source Diluent
Furnace oxygen flow meter setting 375 mL ⁄min to 450 mL ⁄min
Dimethyl sulfide n, butane Inlet oxygen flow meter setting 10 mL ⁄min to 30 mL ⁄min
iso-butane Inlet carrier flow meter setting 130 mL ⁄min to 160 mL ⁄min
propylene Gas sample size 10 mL to 20 mL
propane LPG sample size 15 µL
D6667−21
TABLE 3 Typical Sulfur Calibration Ranges and Standard
11.3.1 Reduce the rate of injection or the sample size, or
Concentrations
both, of the specimen into the furnace when coke or sooting is
Curve I Curve II Curve III
observed.
Sulfur (mg/kg) Sulfur (mg/kg) Sulfur (mg/kg)
Blank Blank Blank
11.4 Cleaning and Re-calibration—Clean any coked or
510 50
sooted parts according to the manufacturer’s instructions.After
10 50 100
any cleaning or adjustment, assemble and check the apparatus
100 200
for leaks. Repeat instrument calibration prior to reanalysis of
the test specimen.
TABLE 4 Repeatability (r) and Reproducibility (R)
11.5 To obtain one result, measure each test specimen three
Concentration Repeatability Reproducibility
times and calculate the average detector response.
Gaseous Gaseous
S (mg/kg) LPG LPG
Hydrocarbons Hydrocarbons
11.6 Density values needed for calculations are to be
1 0.1 0.3 0.3 2.3
measured usingTest Methods D1070 or equivalent or calculate
5 0.6 0.8 1.6 7.2
25 2.9 2.6 7.8 22
accordingto12.2.3,atthetemperatureatwhichthesamplewas
50 5.8 4.2 16 36
tested (Note 8).
100 12 6.8 31 58
A
150 N/A 9N/A 77
NOTE 8—When sample matrix compositions are known, other tech-
196 N/A 11 N/A 93
niques may be used to derive sample density, provided accuracy and
A
N/A—Not applicable to the scope of Gaseous Hydrocarbons.
precision are not degraded.
12. Calculation
12.1 This section describes the calculation procedure for
10.5.1 Multi-point Calibration:
different calibration options and for correction of the sulfur
10.5.1.1 When the apparatus features an internal self-
result when the sample is different from the composition of the
calibration routine, analyze the calibration standards and blank
calibration material. When the sample is injected in the liquid
three times using the procedures described in 10.2 – 10.4.
phase, the density of the calibration mixture and the sample are
10.5.1.2 Calibrate the analyzer according to the manufac-
requiredtocalculatetheresults(12.2–12.4).Whenthesample
turer’s instructions to yield sulfur concentration (see Section
is injected in the gas phase, the molecular weight of the
14).Thiscurveistypicallylinearandsystemperformanceshall
calibration mixture and the samples are required to calculate
be checked at least once per day, each day of use. (Note 7).
the results (12.5 – 12.7).
NOTE 7—Other calibration curve techniques may be used when
accuracyandprecisionarenotdegraded.Thefrequencyofcalibrationmay 12.2 To calculate the density of the liquid calibration
be determined by the use of quality control charts or other quality
mixture and/or the sample:
assurance/quality control techniques.
12.2.1 Interconversion to Liquid Volume% from Mass%
10.5.2 One-point Calibration:
According to Practice D2421:
10.5.2.1 Utilize a calibration standard (6.4) with a sulfur
Mass% ⁄ Relative Density
~ !
x x
content close to that of the samples to be analyzed (625 %
Liquid Volume % 5 100 3
x #comp
max.).
~Mass% ⁄ Relative Density !
( n n
N51
10.5.2.2 Follow the instrument manufacturer’s instructions
to establish an instrument zero (instrument blank) by conduct- (1)
ing an analysis run without injection of the calibration stan-
where:
dard.
Liquid Volume% = liquid volume percent of all determined
10.5.2.3 Performmeasurementsofthecalibrationstandarda
components in the sample of which x is
minimum of three times.
one,
10.5.2.4 Calculate a calibration factor K, in counts per
#comp = the number of determined components
nanogram of sulfur (counts/ng S) as described in 12.2.
in the sample of which x is one,
Relative Density = the value (taken from Practice D2421,
x
11. Procedure
Table 2, Column 3) given for each
11.1 Obtain a test specimen using the procedure described
determined component x as a liquid,
in Section 8. Typically the sulfur concentration in the test
Mass% = the weight percent of each determined
x
specimen is less than the concentration of the highest standard
component taken from GC analysis
and greater than the concentration of the lowest standard used
such as D2163, and
#comp
in the calibration.
= the sum of the quotients Mass% divided
(
N51
by the relative density for all deter-
11.2 Measure the response for the test specimen using one
mined components.
of the procedures described in 10.2 – 10.4.
12.2.2 Interconversion to Liquid Volume% from Mole%
11.3 Inspect the combustion tube and other flow path
components to verify complete oxidation of the test specimen. According to Practice D2421:
D6667−21
Mole% 3 Volume Ratio A
~ !
x x c
Liquid Volume% 5 100 3 K 5 (6)
x #comp
V 3 S
c cv
~Mole% 3 Volume Ratio !
( n n
N51
where:
(2)
A = integrated detector response for calibration standard, in
c
where: counts, and
m = mass of calibration standard injected, in milligrams,
c
Liquid Volume % = liquid volume% of all determined com-
x
either measured directly or calculated from measured
ponents in the sample of whic
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6667 − 14 (Reapproved 2019) D6667 − 21
Standard Test Method for
Determination of Total Volatile Sulfur in Gaseous
Hydrocarbons and Liquefied Petroleum Gases by Ultraviolet
Fluorescence
This standard is issued under the fixed designation D6667; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method covers the determination of total volatile sulfur in gaseous hydrocarbons and liquefied petroleum (LP) gases.
It is applicable to analysis of natural, processed, and final product materials. Precision has been determined for sulfur in gaseous
hydrocarbons in the range of 1 mg ⁄kg to 100 mg ⁄kg and for sulfur in LP gases in the range of 1 mg ⁄kg to 196 mg ⁄kg (Note 1).
NOTE 1—An estimate of pooled limit of quantification (PLOQ), information regarding sample stability and other general information derived from the
2,3
interlaboratory studies on precision can be referenced in the ASTM research reports.
1.2 This test method may not detect sulfur compounds that do not vaporize under the conditions of the test.
1.3 This test method is applicable for total volatile sulfur determination in LP gases containing less than 0.35 % (mass/mass)
halogen(s).
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. See 3.1 and Sections 6 and 7 for specific warning statements.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1070 Test Methods for Relative Density of Gaseous Fuels
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.03 on Elemental Analysis.
Current edition approved May 1, 2019April 1, 2021. Published July 2019May 2021. Originally approved in 2001. Last previous edition approved in 20142019 as
D6667 – 14.D6667 – 14 (2019). DOI: 10.1520/D6667-14R19.10.1520/D6667-21.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D02-1506. Contact ASTM Customer
Service at service@astm.org.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D02-1784. Contact ASTM Customer
Service at service@astm.org.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6667 − 21
D1265 Practice for Sampling Liquefied Petroleum (LP) Gases, Manual Method
D2163 Test Method for Determination of Hydrocarbons in Liquefied Petroleum (LP) Gases and Propane/Propene Mixtures by
Gas Chromatography
D2421 Practice for Interconversion of Analysis of C and Lighter Hydrocarbons to Gas-Volume, Liquid-Volume, or Mass Basis
D2598 Practice for Calculation of Certain Physical Properties of Liquefied Petroleum (LP) Gases from Compositional Analysis
D3700 Practice for Obtaining LPG Samples Using a Floating Piston Cylinder
D5287 Practice for Automatic Sampling of Gaseous Fuels
D6299 Practice for Applying Statistical Quality Assurance and Control Charting Techniques to Evaluate Analytical Measure-
ment System Performance
F307 Practice for Sampling Pressurized Gas for Gas Analysis
2.2 Gas Processor Association (GPA) Standards:
GPA 2166 Obtaining Natural Gas Samples for Analysis by Gas Chromatography
GPA 2174 Obtaining Liquid Hydrocarbon Samples for Analysis by Gas Chromatography
3. Summary of Test Method
3.1 A heated sample valve is used to inject gaseous samples. Liquefied petroleum gas (LPG) samples are injected by a sample
valve connected to a heated expansion chamber. The gaseous sample then enters a high temperature combustion tube where sulfur
is oxidized to sulfur dioxide (SO ) in an oxygen rich atmosphere. Water produced during the sample combustion is removed and
the sample combustion gases are next exposed to ultraviolet (UV) light. The SO absorbs the energy from the UV light and is
* *
converted to an excited sulfur dioxide (SO ). Fluorescence emitted from the excited SO as it returns to a stable state SO is
2 2 2
detected by a photomultiplier tube, the resulting signal is a measure of the sulfur contained in the sample. (Warning—Exposure
to excessive quantities of ultraviolet light is injurious to health. The operator shall avoid exposing their person, especially their
eyes, not only to direct UV light but also to secondary or scattered radiation that is present.)
4. Significance and Use
4.1 The sulfur content of LPG, used for fuel purposes, contributes to SOx emissions and can lead to corrosion in engine and
exhaust systems. Some process catalysts used in petroleum and chemical refining can be poisoned by sulfur bearing materials in
the feed stocks. This test method can be used to determine sulfur in process feeds, to measure sulfur in finished products, and can
also be used for compliance determinations when acceptable to a regulatory authority.
FIG. 1 Example of a Typical Direct Inject Quartz Pyrolysis Tube
Available from Gas Processors Association (GPA), 6526 E. 60th St., Tulsa, OK 74145.
D6667 − 21
5. Apparatus
5.1 Furnace—An electric furnace held at a temperature (1075 °C 6 25 °C) sufficient to pyrolyze the entire sample and oxidize
sulfur to SO .
5.2 Combustion Tube—A quartz combustion tube constructed to allow the direct injection of the sample into the heated oxidation
zone of the furnace. The combustion tube shall have side arms for the introduction of oxygen and carrier gas. The oxidation section
shall be large enough (see Fig. 1) to ensure complete combustion of the sample (see 11.3). Fig. 1 depicts a typical combustion tube.
Other configurations are acceptable when precision is not degraded.
5.3 Flow Control—The apparatus shall be equipped with flow controllers capable of maintaining a constant supply of oxygen and
carrier gas at the specified rates.
5.4 Drier Tube—The apparatus shall be equipped with a mechanism for the removal of water vapor formed during sample
combustion. This can be accomplished with a membrane drying tube, or a permeation dryer that utilizes a selective capillary action
for water removal.
5.5 UV Fluorescence Detector—A quantitative detector capable of measuring light emitted from the fluorescence of sulfur dioxide
by UV light.
5.6 Sample Inlet System—The system provides a heated gas-sampling valve, or a LP gas-sampling valve, or both, with a heated
expansion chamber, connected to the inlet of the oxidation area, Fig. 2. The system is swept by an inert carrier gas and shall be
capable of allowing the quantitative delivery of the material to be analyzed into the oxidation zone at a controlled and repeatable
rate of approximately 30 mL ⁄min. Fig. 3 provides an example.
FIG. 2 Example of Orientation of Total Sulfur Analyzer and Gas or Liquid Sampling Valve
D6667 − 21
FIG. 3 Sample Inlet System Flow Path
5.7 Strip Chart Recorder, equivalent electronic data logger, integrator or, recorder (optional).
6. Reagents
6.1 Purity of Reagents—Reagent grade chemicals shall be used in tests. Unless otherwise indicated, it is intended that all reagents
shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such
specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without lessening the accuracy of the determination.
6.2 Inert Gas—Argon or helium only, high purity grade (that is, chromatography or zero grade), 99.998 % min purity, moisture
5 mg ⁄kg max. (Warning—Argon or helium may be a compressed gas under high pressure (7.1)).
6.3 Oxygen—High purity (that is chromatography or zero grade), 99.75 % min purity, moisture 5 mg ⁄kg max, dried over molecular
sieves. (Warning—Oxygen vigorously accelerates combustion and may be compressed gas under high pressure (7.1)).
6.4 Calibration Standards—Certified calibration standards from commercial sources or calibration gases prepared using certified
permeation tube devices are required. Table 1 lists the sulfur source material and diluent matrices used during the inter-laboratory
study (Notes 2 and 3).
TABLE 1 Typical Standard Materials
Sulfur Source Diluent
Dimethyl sulfide n, butane
iso-butane
propylene
propane
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
D6667 − 21
NOTE 2—Other sulfur sources and diluent materials may be used if precision and accuracy are not degraded.
NOTE 3—Calibration standards are typically re-mixed and re-certified on a regular basis depending upon frequency of use and age. These calibration
standards may have a useful life of about 6 months to 12 months.
6.5 Quality Control (QC) Samples—preferably are portions of one or more gas or LP gas materials that are stable and
representative of the samples of interest.
7. Hazards
7.1 High temperature, flammable hydrocarbons, and gases under high pressures occur in the test method. Use materials that are
rated for containing these pressurized hydrocarbons in all sample containers and sample transfer apparatus. Exercise extra care
when using flammable materials near the oxidative furnace.
8. Sampling
8.1 Obtain a sample in accordance with Practices F307, D1265, D3700, D5287, or GPA 2174 or GPA 2166. Analyze samples as
soon as possible after taking from bulk supplies to prevent loss of sulfur or contamination due to exposure or contact with sample
containers.
8.2 If the sample is not used immediately, then thoroughly mix it in its container prior to taking a test specimen. The use of
segregated or specially treated sample containers can help reduce sample cross-contamination and improve sample stability.
9. Preparation of Apparatus
9.1 Assemble and check the apparatus for leaks according to manufacturer’s instructions.
9.2 Typical apparatus adjustments and conditions are listed in Table 2.
9.3 Adjust instrument sensitivity and baseline stability and perform instrument-blanking procedures following manufacturer’s
guidelines.
10. Calibration and Standardization
10.1 Consult Table 3 and select a calibration range based on the anticipated sulfur concentrations present in samples to be
analyzed, preferably using a sulfur compound and a diluent type representative of the samples to be analyzed (Note 4). Table 3
is representative of typical ranges, but narrower ranges than those indicated may be used if desired. However, the method precision
using narrower ranges than those indicated has not been determined. Ensure the standards used for calibration bracket the
concentrations of the samples being analyzed.
NOTE 4—The number of standards used per curve may vary.
10.2 With the sample valve in the load position, connect the pressurized sample container to the sample valve of the sample inlet
system.
10.3 Obtain a quantitative measurement of the injected material by filling the sample loop of the sample valve system for the
matrix being analyzed (see Table 2) (Notes 5 and 6).
TABLE 2 Typical Operating Conditions
Sample inlet system temperature 85 °C ± 20 °C
Sample injection system carrier gas 25 mL ⁄min to 30 mL ⁄min
Furnace temperature 1075 °C ± 25 °C
Furnace oxygen flow meter setting 375 mL ⁄min to 450 mL ⁄min
Inlet oxygen flow meter setting 10 mL ⁄min to 30 mL ⁄min
Inlet carrier flow meter setting 130 mL ⁄min to 160 mL ⁄min
Gas sample size 10 mL to 20 mL
LPG sample size 15 μL
D6667 − 21
TABLE 3 Typical Sulfur Calibration Ranges and Standard
Concentrations
Curve I Curve II Curve III
Sulfur (mg/kg) Sulfur (mg/kg) Sulfur (mg/kg)
Blank Blank Blank
5 10 50
10 50 100
100 200
TABLE 4 Repeatability (r) and Reproducibility (R)
Concentration Repeatability Reproducibility
Gaseous Gaseous
S (mg/kg) LPG LPG
Hydrocarbons Hydrocarbons
1 0.1 0.3 0.3 2.3
5 0.6 0.8 1.6 7.2
25 2.9 2.6 7.8 22
50 5.8 4.2 16 36
100 12 6.8 31 58
A
150 N/A 9 N/A 77
196 N/A 11 N/A 93
A
N/A—Not applicable to the scope of Gaseous Hydrocarbons.
NOTE 5—Injection of a constant or similar sample size for all materials analyzed in a selected operating range promotes consistent combustion conditions
and may simplify result calculations.
NOTE 6—An automatic sample transfer and injection device may be used.
10.3.1 Flush the sample loop with sufficient calibrant to assure that the material to be injected is representative.
10.3.2 For LPG samples, if bubbles are present in the viewable portion of the liquid column, flush the sample loop to introduce
a new liquid-full sample portion.
10.4 Start the analyzer and inject the calibration material according to the manufacturer’s instructions.
10.5 Calibrate the instrument using one of the following techniques.
10.5.1 Multi-point Calibration:
10.5.1.1 When the apparatus features an internal self-calibration routine, analyze the calibration standards and blank three times
using the procedures described in 10.2 – 10.4.
10.5.1.2 Calibrate the analyzer according to the manufacturer’s instructions to yield sulfur concentration (see Section 14). This
curve is typically linear and system performance shall be checked at least once per day, each day of use. (Note 7).
NOTE 7—Other calibration curve techniques may be used when accuracy and precision are not degraded. The frequency of calibration may be determined
by the use of quality control charts or other quality assurance/quality control techniques.
10.5.2 One-point Calibration:
10.5.2.1 Utilize a calibration standard (6.4) with a sulfur content close to that of the samples to be analyzed (625 % max.).
10.5.2.2 Follow the instrument manufacturer’s instructions to establish an instrument zero (instrument blank) by conducting an
analysis run without injection of the calibration standard.
10.5.2.3 Perform measurements of the calibration standard a minimum of three times.
10.5.2.4 Calculate a calibration factor K, in counts per nanogram of sulfur (counts/ng S) as described in 12.2.
D6667 − 21
11. Procedure
11.1 Obtain a test specimen using the procedure described in Section 8. Typically the sulfur concentration in the test specimen is
less than the concentration of the highest standard and greater than the concentration of the lowest standard used in the calibration.
11.2 Measure the response for the test specimen using one of the procedures described in 10.2 – 10.4.
11.3 Inspect the combustion tube and other flow path components to verify complete oxidation of the test specimen.
11.3.1 Reduce the rate of injection or the sample size, or both, of the specimen into the furnace when coke or sooting is observed.
11.4 Cleaning and Re-calibration—Clean any coked or sooted parts according to the manufacturer’s instructions. After any
cleaning or adjustment, assemble and check the apparatus for leaks. Repeat instrument calibration prior to reanalysis of the test
specimen.
11.5 To obtain one result, measure each test specimen three times and calculate the average detector response.
11.6 Density values needed for calculations are to be measured using Test Methods D1070 or equivalent, equivalent or calculate
according to 12.2.3, at the temperature at which the sample was tested (Note 8).
NOTE 8—When sample matrix compositions are known, other techniques may be used to derive sample density, provided accuracy and precision are not
degraded.
12. Calculation
12.1 This section describes the calculation procedure for different calibration options and for correction of the sulfur result when
the sample is different from the composition of the calibration material. When the sample is injected in the liquid phase, the density
of the calibration mixture and the sample are required to calculate the results (12.2 – 12.4). When the sample is injected in the gas
phase, the molecular weight of the calibration mixture and the samples are required to calculate the results (12.5 – 12.7).
12.2 To calculate the density of the liquid calibration mixture and/or the sample:
12.2.1 Interconversion to Liquid Volume% from Mass% According to Practice D2421:
~Mass% ⁄ Relative Density !
x x
Liquid Volume % 5 100 3 (1)
#comp
x
~Mass% ⁄ Relative Density !
( n n
N51
where:
Liquid Volume% = liquid volume percent of all determined components in the sample of which x is one,
#comp = the number of determined components in the sample of which x is one,
Relative Density = the value (taken from Practice D2421, Table 2, Column 3) given for each determined component x as a
x
liquid,
Mass% = the weight percent of each determined component taken from GC analysis such as D2163, and
x
#comp
= the sum of the quotients Mass% divided by the relative density for all determined components.
(
N51
12.2.2 Interconversion to Liquid Volume% from Mole% According to Practice D2421:
Mole% 3 Volume Ratio
~ !
x x
Liquid Volume% 5 100 3 (2)
x #comp
Mole% 3 Volume Ratio
~ !
n n
(
N51
where:
Liquid Volume % = liquid volume% of all determined components in the sample of which x is one,
x
#comp = the number of determined components in the sample of which x is one,
D6667 − 21
Volume Ratio = the value (taken from Practice D2421, Table 2, Column 2) given for each determined component x,
x
Mole% = the mole percent of each determined component taken from GC analysis (equivalent to gas volume percent
x
assuming an ideal gas), and
#comp
= the sum of the quotients Mole%n multiplied by the volume ration for all determined components.
(
N51
12.2.3 Relative Density Calculation According to Practice D2598:
Relative Density 5
mix
#comp
Liquid Volume% 3 Relative Density
~ !
x x
(3)
(
x51
where:
Relative Density = relative density of the LPG mixture,
mix
#comp = the number of determined components in the sample of which x is one,
Relative Density = liquid relative density
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...