ASTM D4185-23
(Test Method)Standard Test Method for Measurement of Metals in Workplace Atmospheres by Flame Atomic Absorption Spectrophotometry
Standard Test Method for Measurement of Metals in Workplace Atmospheres by Flame Atomic Absorption Spectrophotometry
SIGNIFICANCE AND USE
5.1 The health of workers in many industries is at risk through exposure by inhalation to toxic metals. Industrial hygienists and other public health professionals need to determine the effectiveness of measures taken to control workers' exposures, and this is generally achieved by making workplace air measurements. Exposure to some metal-containing particles has been demonstrated to cause dermatitis, skin ulcers, eye problems, chemical pneumonitis, and other physical disorders (16).3
5.2 FAAS is capable of quantitatively determining many metals in air samples at the levels required by federal, state, and local occupational health and air pollution regulations. The analysis results can be used for the assessment of workplace exposures to metals in workplace air. The suitability of FAAS for elemental analysis for exposure assessment purposes must be investigated prior to carrying out workplace air sampling, in consideration of relevant occupational exposure limit values (OELVs) for metals of concern.
SCOPE
1.1 This test method covers the collection, dissolution, and determination of trace metals in workplace atmospheres, by flame atomic absorption spectrophotometry (FAAS).
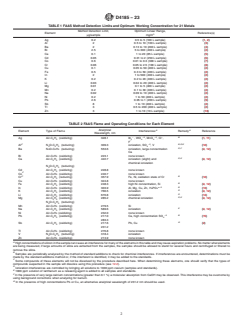
1.2 The estimated method detection limits and optimum working concentration ranges for 21 metals are given in Table 1.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (Specific safety precautionary statements are given in Section 9.)
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2023
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.04 - Workplace Air Quality
Relations
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
- Effective Date
- 01-Sep-2023
Overview
ASTM D4185-23 is an international standard developed by ASTM International that specifies the test method for the measurement of metals in workplace atmospheres using Flame Atomic Absorption Spectrophotometry (FAAS). This standard is designed to support occupational health and safety professionals, industrial hygienists, and environmental specialists in evaluating and controlling worker exposure to hazardous metals. By providing standardized guidance for sampling, dissolution, and analysis of trace metals in air, ASTM D4185-23 helps ensure reliable compliance with federal, state, and local workplace air quality regulations.
Key Topics
- Workplace Exposure Assessment: The standard outlines procedures for collecting air samples in workplace environments for the quantitative determination of a variety of metals, ensuring worker safety by monitoring exposure to toxic substances.
- Flame Atomic Absorption Spectrophotometry (FAAS): FAAS is highlighted as a sensitive analytical technique capable of detecting trace concentrations of up to 23 metals, including lead, cadmium, chromium, and nickel.
- Sampling and Sample Preparation: Guidance is provided on the collection of airborne particulate samples using membrane filters and personal sampling pumps, followed by dissolution of collected samples using acid digestion.
- Interference and Correction Procedures: The standard details identification and correction of possible analytical interferences, ensuring accuracy in FAAS measurements.
- Calibration and Quality Control: Recommendations for calibration of instruments, preparation of standards, and quality control protocols are included to support precise, repeatable results.
- Health and Safety Considerations: The document discusses safety precautions relevant to both laboratory procedures and the handling of hazardous metals and reagents.
Applications
ASTM D4185-23 is widely applicable across multiple settings where airborne metallic contaminants are a concern, including:
- Industrial Hygiene Programs: Used in industries such as metal fabrication, mining, foundries, welding operations, and battery manufacturing to assess workers’ exposure to hazardous metals.
- Regulatory Compliance: Supports compliance with Occupational Exposure Limit Values (OELVs) set by regulatory agencies such as OSHA, EPA, and other international bodies.
- Environmental Monitoring: Utilized in air quality monitoring programs to detect and control emissions of metals in occupational and ambient air.
- Workplace Risk Assessment: Provides data for risk assessments and the effectiveness evaluation of engineering controls, personal protective equipment, and workplace practices aimed at minimizing metal exposure.
- Research and Development: Serves laboratories and researchers investigating occupational exposure to metals or developing new control technologies.
Related Standards
ASTM D4185-23 is part of a suite of ASTM standards addressing air quality and metal analysis in workplace environments. Users often reference these standards concurrently to ensure comprehensive sampling and analytical practices:
- ASTM D1193: Specification for Reagent Water, relevant for sample preparation and analysis.
- ASTM D1356: Terminology relating to sampling and analysis of atmospheres.
- ASTM D1357: Practice for planning the sampling of ambient atmospheres.
- ASTM D3195: Practice for rotameter calibration.
- ASTM D5337: Practice for setting and verifying the flow rate of personal sampling pumps.
- ASTM D7035: Test method for determination of metals and metalloids in airborne particulate matter by Inductively Coupled Plasma Atomic Emission Spectrometry (ICP-AES).
- ASTM D8358: Guide for assessment and inclusion of wall deposits in the analysis of single-stage samplers for airborne particulate matter.
By following ASTM D4185-23 alongside these related standards, organizations can implement robust air monitoring protocols, safeguard worker health, and fulfill regulatory obligations for measuring metals in workplace atmospheres using FAAS.
Buy Documents
ASTM D4185-23 - Standard Test Method for Measurement of Metals in Workplace Atmospheres by Flame Atomic Absorption Spectrophotometry
REDLINE ASTM D4185-23 - Standard Test Method for Measurement of Metals in Workplace Atmospheres by Flame Atomic Absorption Spectrophotometry
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D4185-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Measurement of Metals in Workplace Atmospheres by Flame Atomic Absorption Spectrophotometry". This standard covers: SIGNIFICANCE AND USE 5.1 The health of workers in many industries is at risk through exposure by inhalation to toxic metals. Industrial hygienists and other public health professionals need to determine the effectiveness of measures taken to control workers' exposures, and this is generally achieved by making workplace air measurements. Exposure to some metal-containing particles has been demonstrated to cause dermatitis, skin ulcers, eye problems, chemical pneumonitis, and other physical disorders (16).3 5.2 FAAS is capable of quantitatively determining many metals in air samples at the levels required by federal, state, and local occupational health and air pollution regulations. The analysis results can be used for the assessment of workplace exposures to metals in workplace air. The suitability of FAAS for elemental analysis for exposure assessment purposes must be investigated prior to carrying out workplace air sampling, in consideration of relevant occupational exposure limit values (OELVs) for metals of concern. SCOPE 1.1 This test method covers the collection, dissolution, and determination of trace metals in workplace atmospheres, by flame atomic absorption spectrophotometry (FAAS). 1.2 The estimated method detection limits and optimum working concentration ranges for 21 metals are given in Table 1. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (Specific safety precautionary statements are given in Section 9.) 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The health of workers in many industries is at risk through exposure by inhalation to toxic metals. Industrial hygienists and other public health professionals need to determine the effectiveness of measures taken to control workers' exposures, and this is generally achieved by making workplace air measurements. Exposure to some metal-containing particles has been demonstrated to cause dermatitis, skin ulcers, eye problems, chemical pneumonitis, and other physical disorders (16).3 5.2 FAAS is capable of quantitatively determining many metals in air samples at the levels required by federal, state, and local occupational health and air pollution regulations. The analysis results can be used for the assessment of workplace exposures to metals in workplace air. The suitability of FAAS for elemental analysis for exposure assessment purposes must be investigated prior to carrying out workplace air sampling, in consideration of relevant occupational exposure limit values (OELVs) for metals of concern. SCOPE 1.1 This test method covers the collection, dissolution, and determination of trace metals in workplace atmospheres, by flame atomic absorption spectrophotometry (FAAS). 1.2 The estimated method detection limits and optimum working concentration ranges for 21 metals are given in Table 1. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. (Specific safety precautionary statements are given in Section 9.) 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4185-23 is classified under the following ICS (International Classification for Standards) categories: 13.040.30 - Workplace atmospheres. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4185-23 has the following relationships with other standards: It is inter standard links to ASTM D4185-17, ASTM D8344-20, ASTM D7035-21, ASTM E3193-21, ASTM D4844-16, ASTM D8358-21, ASTM E1583-21a, ASTM D7439-21, ASTM D4861-23. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4185-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4185 − 23
Standard Test Method for
Measurement of Metals in Workplace Atmospheres by
Flame Atomic Absorption Spectrophotometry
This standard is issued under the fixed designation D4185; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D1357 Practice for Planning the Sampling of the Ambient
Atmosphere
1.1 This test method covers the collection, dissolution, and
D3195 Practice for Rotameter Calibration
determination of trace metals in workplace atmospheres, by
D5337 Practice for Setting and Verifying the Flow Rate of
flame atomic absorption spectrophotometry (FAAS).
Personal Sampling Pumps
1.2 The estimated method detection limits and optimum
D7035 Test Method for Determination of Metals and Met-
working concentration ranges for 21 metals are given in Table
alloids in Airborne Particulate Matter by Inductively
1.
Coupled Plasma Atomic Emission Spectrometry (ICP-
1.3 The values stated in SI units are to be regarded as AES)
D8358 Guide for Assessment and Inclusion of Wall Deposits
standard. No other units of measurement are included in this
standard. in the Analysis of Single-Stage Samplers for Airborne
Particulate Matter
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
3. Terminology
responsibility of the user of this standard to establish appro-
3.1 Definitions—For definitions of terms used in this test
priate safety, health, and environmental practices and deter-
method, refer to Terminology D1356.
mine the applicability of regulatory limitations prior to use.
(Specific safety precautionary statements are given in Section 3.2 Definitions of Terms Specific to This Standard:
9.) 3.2.1 blank signal, n—that signal which results from all
1.5 This international standard was developed in accor- added reagents and clean sample media prepared and analyzed
dance with internationally recognized principles on standard-
exactly in the same way as the samples.
ization established in the Decision on Principles for the
3.2.2 working range for an analytical precision better than
Development of International Standards, Guides and Recom-
3 %, n—the range of sample concentrations that will absorb
mendations issued by the World Trade Organization Technical
10 % to 70 % of the incident radiation (0.05 to 0.52 absorbance
Barriers to Trade (TBT) Committee.
units).
2. Referenced Documents
4. Summary of Test Method
2.1 ASTM Standards:
4.1 Workplace air samples are collected in samplers con-
D1193 Specification for Reagent Water
taining filters or filter capsules and are then treated with acid
D1356 Terminology Relating to Sampling and Analysis of
mixtures to destroy the organic matrix and to dissolve the
Atmospheres
metals present. The analysis is subsequently made by flame
atomic absorption spectrophotometry (FAAS).
This test method is under the jurisdiction of ASTM Committee D22 on Air
4.2 Samples and standards are aspirated the flame of an
Quality and is the direct responsibility of Subcommittee D22.04 on Workplace Air
absorption spectrophotometer. A hollow cathode or electrode-
Quality.
less discharge lamp for the metal being determined provides a
Current edition approved Sept. 1, 2023. Published September 2023. Originally
source of characteristic radiation energy for that particular
approved in 1990. Last previous edition approved in 2017 as D4185 – 17. DOI:
10.1520/D4185-23.
metal. The absorption of this characteristic energy by the atoms
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
of interest in the flame is related to the concentration of the
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
metal in the aspirated sample. The flame and operating
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. conditions for each element are listed in Table 2.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4185 − 23
TABLE 1 FAAS Method Detection Limits and Optimum Working Concentration for 21 Metals
Method Detection Limit, Optimum Linear Range,
Element Reference(s)
μg/sample mg/m
Ag 0.2 0.5 to 5 (100 L sample) (1, 2)
Al 2 0.5 to 10 (100 L sample) (3)
Ba 2 0.13 to 10 (200 L sample) (4)
Bi 2.5 5 to 300 (400 L sample) (2)
Ca 0.1 1 to 20 (85 L sample) (5)
Cd 0.05 0.01 to 2 (250 L sample) (6)
Co 0.6 0.01 to 0.3 (300 L sample) (7)
Cr 0.06 0.05 to 2.5 (100 L sample) (8)
Cu 0.1 0.05 to 50 (400 L sample) (2)
Fe 0.5 0.3 to 50 (400 L sample) (2)
In 2 1 to 500 (400 L sample) (2)
K 0.2 0.2 to 20 (400 L sample) (2)
Li 0.03 0.04 to 20 (400 L sample) (2)
Mg 0.01 0.1 to 5 (400 L sample) (2)
Mn 0.2 0.1 to 30 (400 L sample) (2)
Na 0.02 0.09 to 10 (400 L sample) (2)
Ni 0.2 1 to 50 (400 L sample) (2)
Pb 2.6 0.05 to 1 (200 L sample) (9)
Sb 8 1 to 10 (400 L sample) (2)
Tl 3 0.5 to 200 (400 L sample) (2)
Zn 3 1 to 10 (10 L sample) (10)
TABLE 2 FAAS Flame and Operating Conditions for Each Element
Analytical
A A
Element Type of Flame Interferences Remedy Reference
Wavelength, nm
− −2 −2 − B
Ag Air-C H (oxidizing) 328.1 I0 , WO , MnO , Cl , (1, 11)
2 2 3 4 4
−
F
C −2 B,D,E
Al N O-C H (reducing) 309.3 ionization, SO , V (12)
2 2 2 4
D,F
Ba N O-C H (reducing) 553.6 ionization, large concentration (2, 12)
2 2 2
Ca
Bi Air-C H (oxidizing) 223.1 none known
2 2
D,E
Ca Air-C H (oxidizing) 422.7 ionization (slight) and (2, 12)
2 2
chemical ionization
N O-C H (reducing)
2 2 2
Cd Air-C H (oxidizing) 228.8 none known
2 2
C
Co Air-C H (oxidizing) 240.7 none known
2 2
C B
Cr Air-C H (reducing) 357.9 Fe, Ni, oxidation state of Cr (12)
2 2
Cu Air-C H (oxidizing) 324.8 none known
2 2
B
Fe Air-C H (oxidizing) 248.3 high Ni concentration, Si (2, 12)
2 2
x−3 B
In Air-C H (oxidizing) 303.9 Al, Mg, Cu, Zn, H PO (13)
2 2 x 4
D
K Air-C H (oxidizing) 766.5 ionization (2, 12)
2 2
D
Li Air-C H (oxidizing) 670.8 ionization (14)
2 2
D,E
Mg Air-C H (oxidizing) 285.2 chemical ionization (2, 12)
2 2
N O-C H (reducing)
2 2 2
Mn Air-C H (oxidizing) 279.5 Si
2 2
E
Na Air-C H (oxidizing) 589.6 ionization (2, 12)
2 2
Ni Air-C H (oxidizing) 232.0 none known
2 2
−2 B
Pb Air-C H (oxidizing) 217.0 Ca, high concentration SO (15)
2 2 4
283.3
G
Sb Air-C H (oxidizing) 217.6 Pb, Cu (2)
2 2
231.2
Tl Air-C H (oxidizing) 276.8 none known
2 2
V N O-C H (reducing) 318.4 ionization
2 2 2
Zn Air-C H (oxidizing) 213.9 none known
2 2
A
High concentrations of silicon in the sample can cause an interference for many of the elements in this table and may cause aspiration problems. No matter what elements
are being measured, if large amounts of silica are extracted from the samples, the samples should be allowed to stand for several hours and centrifuged or filtered to
remove the silica.
B
Samples are periodically analyzed by the method of standard additions to check for chemical interferences. If interferences are encountered, determinations must be
made by the standard additions method or, if the interferent is identified, it may be added to the standards.
C
Some compounds of these elements will not be dissolved by the procedure described here. When determining these elements, one should verify that the types of
compounds suspected in the sample will dissolve using this procedure (see 12.2).
D
Ionization interferences are controlled by bringing all solutions to 1000 ppm cesium (samples and standards).
E
1000 ppm solution of lanthanum as a releasing agent is added to all samples and standards.
F
In the presence of very large calcium concentrations (greater than 0.1 %) a molecular absorption from CaOH may be observed. This interference may be overcome by
using background corrections when analyzing for barium.
G
In the presence of high concentrations Pb or Cu, an alternative analytical wavelength of 231.2 nm should be used.
D4185 − 23
5. Significance and Use controlled by the addition of a high concentration of another
easily ionized element which will buffer the electron concen-
5.1 The health of workers in many industries is at risk
tration in the flame.
through exposure by inhalation to toxic metals. Industrial
6.5 Chemical interferences occur in FAAS when species
hygienists and other public health professionals need to deter-
present in the sample cause variations in the degree to which
mine the effectiveness of measures taken to control workers’
atoms are formed in the flame, or when different valence states
exposures, and this is generally achieved by making workplace
of a single element have different absorption characteristics.
air measurements. Exposure to some metal-containing particles
Such interferences may be controlled by adjusting the sample
has been demonstrated to cause dermatitis, skin ulcers, eye
matrix or by the method of standard additions (18). Also, the
problems, chemical pneumonitis, and other physical disorders
use of lanthanum as a releasing element minimizes the inter-
(16).
ference from the formation of nonvolatile compounds in the
5.2 FAAS is capable of quantitatively determining many
flame. Lanthanum forms nonvolatile compounds preferentially
metals in air samples at the levels required by federal, state,
with the interferent so that the analyte remains free.
and local occupational health and air pollution regulations. The
6.6 Physical interferences may result if the physical prop-
analysis results can be used for the assessment of workplace
erties of the samples vary significantly. Changes in viscosity
exposures to metals in workplace air. The suitability of FAAS
and surface tension can affect the sample aspiration rate and
for elemental analysis for exposure assessment purposes must
thus cause erroneous results. Sample dilution or the method of
be investigated prior to carrying out workplace air sampling, in
standard additions, or both, are used to correct such interfer-
consideration of relevant occupational exposure limit values
ences. High concentrations of silica in the sample can cause
(OELVs) for metals of concern.
aspiration problems. No matter what elements are being
determined, if large amounts of silica are extracted from the
6. Interferences
samples, they shall be allowed to stand for several hours and
6.1 In FAAS the occurrence of interferences is less common
centrifuged or filtered to remove the silica.
than in many other analytical techniques. Interferences can
6.7 This procedure describes a generalized method for
occur, however, and when encountered are corrected for as
sample preparation, which is applicable to the majority of
indicated in the following sections. The known interferences
samples. There are some relatively rare chemical forms of a
and correction methods for each metal are indicated in Table 2.
few of the elements listed in Table 1 that will not be dissolved
The methods of standard additions and background monitoring
by this procedure. If such chemical forms are suspected, results
and correction (11, 12, 17, 18) are used to identify the presence
obtained using this procedure shall be compared with those
of an interference. Insofar as possible, the matrix of sample and
obtained using an appropriately altered dissolution procedure.
standard are matched to minimize the possible interference.
Alternatively, the results may be compared with values ob-
6.2 Background or nonspecific absorption can occur from
tained using a technique that does not require dissolving the
particles produced in the flame which can scatter light and
sample (for example, X-ray fluorescence or neutron activation
produce an apparent absorption signal. Light scattering may be
analysis).
encountered when solutions of high salt content are being
7. Apparatus
analyzed. They are most severe when measurements are made
at shorter wavelengths (for example, below about 250 nm).
7.1 Sampling Apparatus:
Background absorption may also occur as the result of the
7.1.1 Samplers, containing mixed cellulose ester (MCE) or
formation of various molecular species which can absorb light.
cellulose nitrate membrane filters, with a pore size of 0.8 μm
The background absorption can be accounted for by the use of
mounted in a 25 mm or 37 mm diameter, two- or three-piece
background correction techniques (17).
filter holder. MCE filters attached to cellulose acetate capsules,
which are acid-soluble, are also suitable.
6.3 Spectral interferences are those interferences which
result from an atom different from the one being measured that
NOTE 1—Alternative sampling media, such as quartz fiber filters, may
absorbs a portion of the radiation. Such interferences are be suitable.
extremely rare in FAAS. In some cases multielement hollow
7.1.1.1 Appropriate workplace air samplers are described in
cathode lamps may cause a spectral interference by having
Test Method D7035. The background metal content of the
closely adjacent emission lines from two different elements. In
filters should be minimal (see Annex A1 of Test Method
general, the use of multielement hollow cathode lamps is
D7035).
discouraged.
7.1.2 Portable, Battery-Operated Personal Sampling
Pumps, equipped with a flow-monitoring device (rotameter,
6.4 Ionization interference occurs when easily ionized at-
critical orifice) or a constant-flow device, and capable of
oms are being measured. The degree to which such atoms are
drawing 1 L ⁄min to 5 L ⁄min of air through a 0.8 μm mixed
ionized is dependent upon the atomic concentration and the
cellulose ester membrane filter for a period of at least 8 h.
presence of other easily ionized atoms. This interference can be
7.2 Analytical Apparatus:
7.2.1 Flame Atomic Absorption Spectrophotometer,
equipped with air/acetylene and nitrous oxide/acetylene burner
Boldface numbers in parentheses refer to the list of references appended to
these methods. heads.
D4185 − 23
7.2.2 Hollow Cathode or Electrodeless Discharge Lamp, for 8.5.2 Stock Barium Solution—Dissolve 1.779 g of barium
each element to be determined. chloride (BaCl ·2H O) in water. Dilute to volume in a 1 L flask
2 2
with purified water.
7.2.3 Deuterium Continuum Lamp.
7.2.4 Compressed Air—Appropriate pressure reducing regu- 8.5.3 Stock Bismuth Solution—Dissolve 1.000 g of bismuth
lator with base connections (see instrument manufacturer’s
metal in a minimum volume of 6 N HNO . Dilute to volume in
instructions). a 1 L flask with 2 % (v/v) HNO .
7.2.5 Acetylene Gas and Regulator—A cylinder of acety-
8.5.4 Stock Cadmium Solution—Dissolve 1.000 g of cad-
lene equipped with a two-gauge, two-stage pressure-reducing
mium metal in a minimum volume of 6 N HCl. Dilute to
regulator with hose connections. (See instrument manufacturer
volume in a 1 L flask with 2 % (v/v) HNO .
instructions.)
8.5.5 Stock Calcium Solution—To 2.497 g of primary stan-
7.2.6 Nitrous Oxide Gas and Regulator—A cylinder of
dard calcium carbonate (CaCO ) add 50 mL of distilled water.
nitrous oxide equipped with a two-gauge, two-stage pressure-
Add dropwise a minimum volume of HCl (approximately
reducing regulator and hose connections. Heat tape with the
10 mL) to dissolve the CaCO . Dilute to volume in a 1 L flask
temperature controlled by a rheostat may be wound around the
with purified water.
second stage regulator and hose connection to prevent
8.5.6 Stock Chromium Solution—Dissolve 3.735 g of potas-
freeze-up of the line. (See instrument manufacturer instruc-
sium chromate (K CrO ) in distilled water. Dilute to volume in
2 4
tions.)
a 1 L flask with purified water.
7.2.7 Beakers, Phillips or Griffin, 125 mL, borosilicate
8.5.7 Stock Cobalt Solution—Dissolve 1.000 g of cobalt
glass.
metal in a minimum volume of 1 + 1 HCl. Dilute to volume in
7.2.8 Centrifuge Tubes, 15 mL, graduated, borosilicate
a 1 L flask with 2 % (v/v) HNO .
glass.
8.5.8 Stock Copper Solution—Dissolve 1.000 g of copper
7.2.9 Miscellaneous Borosilicate Glassware (Pipets and
metal in a minimum volume of 6 N HNO . Dilute to volume in
Volumetric Flasks)—All pipets and volumetric flasks shall be
a 1 L flask with 2 % (v/v) HNO .
calibrated Class A volumetric glassware.
8.5.9 Stock Indium Solution—Dissolve 1.000 g of indium
metal in a minimum volume of 1 + 1 HCl. Addition of a few
8. Reagents
drops of HNO and mild heating will aid in dissolving the
8.1 Purity of Reagents—Reagent grade chemicals shall be metal. Dilute to volume in a 1 L flask with 2 % (v/v) HNO .
used in all tests. Unless otherwise indicated, it is intended that 8.5.10 Stock Iron Solution—Dissolve 1.000 g of iron wire in
all reagents shall conform to the specifications of the Commit-
50 mL of 6 N HNO . Dilute to volume in a 1 L flask with 2 %
tee on Analytical Reagents of the American Chemical Society, (v/v) HNO .
where such specifications are available. Other grades may be
8.5.11 Stock Lead Solution—Dissolve 1.598 g of lead nitrate
used provided that it can be demonstrated that they are of
(Pb(NO ) ) in 2 % (v ⁄v) HNO . Dilute to volume in a 1 L flask
3 2 3
sufficiently high purity to permit their use without decreasing
with 2 % (v/v) HNO .
the accuracy of the determinations.
8.5.12 Stock Lithium Solution—Dissolve 5.324 g of lithium
carbonate (Li CO ) in a minimum volume of 6 N HCl. Dilute
8.2 Purity of Water—Unless otherwise indicated, reference
2 3
to volume in a 1 L flask with purified water.
to water shall be understood to mean Type II reagent water
conforming to Specification D1193. 8.5.13 Stock Magnesium Solution—Dissolve 1.000 g of
magnesium ribbon in a minimum volume of 6 N HCl. Dilute to
8.3 Hydrochloric Acid (HCl)—Concentrated hydrochloric
volume in a 1 L flask with 2 % (v/v) HNO .
acid, 12 N, specific gravity 1.19.
8.5.14 Stock Manganese Solution—Dissolve 1.000 g of
8.4 Nitric Acid (HNO )—Redistilled, concentrated nitric
manganese metal in a minimum volume of 6 N HNO . Dilute
acid, 16 N, specific gravity 1.42.
to volume in a 1 L flask with 2 % (v/v) HNO .
8.5 Standard Stock Solutions (1000 μg/mL) for each of the 8.5.15 Stock Nickel Solution—Dissolve 1.000 g of nickel
metals listed in Table 1. These solutions are stable for at least metal in a minimum volume of 6 N HNO . Dilute to volume in
one year when stored in polyethylene bottles, and can be a 1 L flask with 2 % (v/v) HNO .
obtained from commercial sources or prepared in the labora-
8.5.16 Stock Potassium Solution—Dissolve 1.907 g of po-
tory in the following manner:
tassium chloride (KCl) in purified water. Dilute to volume in a
8.5.1 Stock Aluminum Solution—Dissolve 1.000 g of alumi-
1 L flask with purified water.
num wire in a minimum volume of 1 + 1 HCl. Dilute to volume
8.5.17 Stock Rubidium Solution—Dissolve 1.415 g of ru-
in a 1 L flask with purified water.
bidium chloride (RbCl) in distilled water. Dilute to volume in
a 1 L flask with purified water.
8.5.18 Stock Silver Solution—Dissolve 1.575 g of silver
nitrate (AgNO ) in 100 mL of purified water. Dilute to volume
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
in a 1 L volumetric flask with 2 % (v ⁄v) HNO . The silver
DC. For suggestions on the testing of reagents not listed by the American Chemical
nitrate solution will deteriorate in light and must be stored in an
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
amber bottle away from direct light. New stock silver solution
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. shall be prepared every few months.
D4185 − 23
8.5.19 Stock Sodium Solution—Dissolve 2.542 g of sodium 9.4 The instrument exhaust gases contain the combustion
chloride (NaCl) in purified water. Dilute to volume in a 1 L products of the flame as well as metal vapor from the sample.
flask with purified water. Both the combustion products and the metal vapor (see 9.3) are
definite personnel hazards. The instrument combustion gases
8.5.20 Stock Strontium Solution—Dissolve 2.415 g of stron-
shall be mechanically exhausted from the laboratory (see
tium nitrate (Sr(NO ) ) in purified water. Dilute to volume in a
3 2
instrument manufacturer’s instructions).
1 L flask with purified water.
8.5.21 Stock Thallium Solution—Dissolve 1.303 g of thal-
10. Sampling
lium nitrate (TINO ) in 10 % (v ⁄v) HNO . Dilute to volume in
3 3
10.1 For general information on sampling, refer to Practice
a 1 L flask with 2 % (v/v) HNO .
D1357. Consider applicable r
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4185 − 17 D4185 − 23
Standard Test Method for
Measurement of Metals in Workplace Atmospheres by
Flame Atomic Absorption Spectrophotometry
This standard is issued under the fixed designation D4185; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the collection, dissolution, and determination of trace metals in workplace atmospheres, by flame
atomic absorption spectrophotometry (FAAS).
1.2 The sensitivity, detection limit,estimated method detection limits and optimum working concentration ranges for 2321 metals
are given in Table 1.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use. (Specific safety precautionary statements are given in Section 9.)
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D1356 Terminology Relating to Sampling and Analysis of Atmospheres
D1357 Practice for Planning the Sampling of the Ambient Atmosphere
D3195 Practice for Rotameter Calibration
D5337 Practice for Setting and Verifying the Flow Rate of Personal Sampling Pumps
D7035 Test Method for Determination of Metals and Metalloids in Airborne Particulate Matter by Inductively Coupled Plasma
Atomic Emission Spectrometry (ICP-AES)
D8358 Guide for Assessment and Inclusion of Wall Deposits in the Analysis of Single-Stage Samplers for Airborne Particulate
Matter
3. Terminology
3.1 Definitions—For definitions of terms used in this test method, refer to Terminology D1356.
This test method is under the jurisdiction of ASTM Committee D22 on Air Quality and is the direct responsibility of Subcommittee D22.04 on Workplace Air Quality.
Current edition approved March 1, 2017Sept. 1, 2023. Published March 2017September 2023. Originally approved in 1990. Last previous edition approved in 20112017
as D4185 – 06 (2011).D4185 – 17. DOI: 10.1520/D4185-17.10.1520/D4185-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4185 − 23
TABLE 1 FAAS Instrumental Detection Limits and Optimum Working Concentration for 23 Metals
Detection Limit, μg/mL
(approximately three Optimum Linear Range TLV, mg/m (elements,
Element times Upper Limit, compound classes, and
B
standard deviation of μg/mL oxides)
A
blank)
Ag 0.001 5 0.1 (metal) 0.01
(soluble compounds
as Ag)
Al 0.04 50 2.0 (soluble salts and
alkyls not otherwise
classified) 10 (metal
dust and oxide)
5 (pyro powder and
welding fume)
Ba 0.01 10 0.5 (soluble
compounds)
Bi 0.03 10 No Limit expressed for
this element
Ca 0.002 1 2 (oxide as CaO)
Cd 0.0008 1 0.01 (elemental and
compounds—total
dust)
0.002 (elemental
compounds—
respirable fraction)
Co 0.009 5 0.02 (elemental and
inorganic) 0.1
(carbonyl and
hydrocarbonyl)
Cr 0.003 5 0.5 (metal and Cr III
compounds) 0.05
(water soluble Cr VI
compounds)
0.01 (insoluble Cr VI
compounds)
Cu 0.002 5 0.2 (fume) 1 (dust and
mists as Cu)
Fe 0.005 5 5 (iron oxide fume) 5
(soluble salts as Fe)
In 0.03 50 0.1 (metal and
compounds)
K 0.003 1 No Limit expressed for
this element
Li 0.0008 1 No Limit expressed for
this element
Mg 0.0002 0.5 10 (as MgO fume)
Mn 0.002 5 0.2 (elemental and
inorganic
compounds)
Na 0.0003 0.5 No Limit expressed for
this element
Ni 0.006 5 0.05 (elemental,
soluble and insoluble
compounds)
Pb 0.02 10 0.15 (inorganic
compounds, fume,
dust)
Rb 0.003 5 No Limit expressed for
this element
Sr 0.003 5 No Limit expressed for
this element
Tl 0.02 50 0.1 (soluble
compounds)
V 0.06 100 0.05 (pentoxide,
respirable dust or
fume, as V O )
2 5
Zn 0.002 1 10 (oxide dust as ZnO)
5 (oxide fume as
ZnO)
TABLE 1 FAAS Method Detection Limits and Optimum Working Concentration for 21 Metals
Method Detection Limit, Optimum Linear Range,
Element Reference(s)
μg/sample mg/m
Ag 0.2 0.5 to 5 (100 L sample) (1, 2)
Al 2 0.5 to 10 (100 L sample) (3)
Ba 2 0.13 to 10 (200 L sample) (4)
D4185 − 23
Method Detection Limit, Optimum Linear Range,
Element Reference(s)
μg/sample mg/m
Bi 2.5 5 to 300 (400 L sample) (2)
Ca 0.1 1 to 20 (85 L sample) (5)
Cd 0.05 0.01 to 2 (250 L sample) (6)
Co 0.6 0.01 to 0.3 (300 L sample) (7)
Cr 0.06 0.05 to 2.5 (100 L sample) (8)
Cu 0.1 0.05 to 50 (400 L sample) (2)
Fe 0.5 0.3 to 50 (400 L sample) (2)
In 2 1 to 500 (400 L sample) (2)
K 0.2 0.2 to 20 (400 L sample) (2)
Li 0.03 0.04 to 20 (400 L sample) (2)
Mg 0.01 0.1 to 5 (400 L sample) (2)
Mn 0.2 0.1 to 30 (400 L sample) (2)
Na 0.02 0.09 to 10 (400 L sample) (2)
Ni 0.2 1 to 50 (400 L sample) (2)
Pb 2.6 0.05 to 1 (200 L sample) (9)
Sb 8 1 to 10 (400 L sample) (2)
Tl 3 0.5 to 200 (400 L sample) (2)
Zn 3 1 to 10 (10 L sample) (10)
A
These detection limits represent ideal laboratory conditions; variability due to sampling, digestion, reagents, and sample handling has not been taken into account.
B
Threshold Limit Values of Airborne Contaminants and Physical Agents adopted by ACGIH for 1994–1995. Values are elemental concentrations except as noted.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 blank signal—signal, n—that signal which results from all added reagents and a clean membrane filterclean sample media
prepared and analyzed exactly in the same way as the samples.
3.2.2 instrumental detection limit—that concentration of a given element which produces a signal three times the standard
deviation of the reagent blank signal.
3.2.2 working range for an analytical precision better than 3 %—3 %, n—the range of sample concentrations that will absorb
1010 % to 70 % of the incident radiation (0.05 to 0.52 absorbance units).
NOTE 1—Values for instrumental detection limit may vary from instrument to instrument.
4. Summary of Test Method
4.1 Workplace air samples are collected on membrane filters and treated with nitric acid in samplers containing filters or filter
capsules and are then treated with acid mixtures to destroy the organic matrix and to dissolve the metals present. The analysis is
subsequently made by flame atomic absorption spectrophotometry (FAAS).
4.2 Samples and standards are aspirated the flame of an absorption spectrophotometer. A hollow cathode or electrodeless discharge
lamp for the metal being determined provides a source of characteristic radiation energy for that particular metal. The absorption
of this characteristic energy by the atoms of interest in the flame is related to the concentration of the metal in the aspirated sample.
The flame and operating conditions for each element are listed in Table 2.
D4185 − 23
TABLE 2 FAAS Flame and Operating Conditions for Each Element
Analytical
Element Reference
A A
Type of Flame Wavelength, nm Interferences Remedy
Air-C H (oxidizing) 328.1 (2,
2 2
− −2 −2 − B
Ag I0 , WO , MnO , Cl ,
3 4 4
−
F
Air-C H (oxidizing) 328.1 (1,
2 2
− −2 −2 − B
Ag I0 , WO , MnO , Cl ,
3 4 4
−
F
N O-C H (reducing) 309.3 (4)
2 2 2
C −2 B,D,E
Al ionization, SO , V
N O-C H (reducing) 309.3 (12)
2 2 2
C −2 B,D,E
Al ionization, SO , V
N O-C H (reducing) 553.6 (1,
2 2 2
D,F
Ba ionization, large concentration
Ca
N O-C H (reducing) 553.6 (2,
2 2 2
D,F
Ba ionization, large concentration
Ca
Air-C H (oxidizing) 223.1
2 2
Bi none known
Air-C H (oxidizing) 422.7 (1,
2 2
D,E
Ca ionization (slight) and
chemical ionization
Air-C H (oxidizing) 422.7 (2,
2 2
D,E
Ca ionization (slight) and
chemical ionization
N O-C H (reducing)
2 2 2
Air-C H (oxidizing) 228.8
2 2
Cd none known
Air-C H (oxidizing) 240.7
2 2
C
Co none known
Air-C H (reducing) 357.9 (4)
2 2
C B
Cr Fe, Ni, oxidation state of Cr
Air-C H (reducing) 357.9 (12)
2 2
C B
Cr Fe, Ni, oxidation state of Cr
Air-C H (oxidizing) 324.8
2 2
Cu none known
Air-C H (oxidizing) 248.3 (1,
2 2
B
Fe high Ni concentration, Si
Air-C H (oxidizing) 248.3 (2,
2 2
B
Fe high Ni concentration, Si
Air-C H (oxidizing) 303.9 (5)
2 2
x−3 B
In Al, Mg, Cu, Zn, H PO
x 4
Air-C H (oxidizing) 303.9 (13)
2 2
x−3 B
In Al, Mg, Cu, Zn, H PO
x 4
Air-C H (oxidizing) 766.5 (1,
2 2
D
K ionization
Air-C H (oxidizing) 766.5 (2,
2 2
D
K ionization
Air-C H (oxidizing) 670.8 (6)
2 2
D
Li ionization
Air-C H (oxidizing) 670.8 (14)
2 2
D
Li ionization
Air-C H (oxidizing) 285.2 (1,
2 2
D,E
Mg chemical ionization
Air-C H (oxidizing) 285.2 (2,
2 2
D,E
Mg chemical ionization
N O-C H (reducing)
2 2 2
Air-C H (oxidizing) 279.5
2 2
Mn Si
Air-C H (oxidizing) 589.6 (1,
2 2
E
Na ionization
Air-C H (oxidizing) 589.6 (2,
2 2
E
Na ionization
Air-C H (oxidizing) 232.0
2 2
Ni none known
Air-C H (oxidizing) 217.0 (7)
2 2
−2 B
Pb 283.3 Ca, high concentration SO
Air-C H (oxidizing) 217.0 (15)
2 2
−2 B
Pb 283.3 Ca, high concentration SO
D
Air-C H (oxidizing) 780.0 ionization (1,
2 2
Rb
G
Sb Air-C H (oxidizing) 217.6 Pb, Cu
2 2
(2)
D4185 − 23
Analytical
Element Reference
A A
Type of Flame Wavelength, nm Interferences Remedy
Sr Air-CionizationH (oxidizing)and chemical 460.7 (1,
2 2
D,E
231.2
N O-C H (reducing)
2 2 2
ionization
Air-C H (oxidizing) 276.8
2 2
Tl none known
N O-C H (reducing) 318.4
2 2 2
Va ionization
V N O-C H (reducing) 318.4
2 2 2
ionization
Air-C H (oxidizing) 213.9
2 2
Zn none known
A
High concentrations of silicon in the sample can cause an interference for many of the elements in this table and may cause aspiration problems. No matter what elements
are being measured, if large amounts of silica are extracted from the samples, the samples should be allowed to stand for several hours and centrifuged or filtered to
remove the silica.
B
Samples are periodically analyzed by the method of standard additions to check for chemical interferences. If interferences are encountered, determinations must be
made by the standard additions method or, if the interferent is identified, it may be added to the standards.
C
Some compounds of these elements will not be dissolved by the procedure described here. When determining these elements, one should verify that the types of
compounds suspected in the sample will dissolve using this procedure (see 12.2).
D
Ionization interferences are controlled by bringing all solutions to 1000 ppm cesium (samples and standards).
E
1000-ppm 1000 ppm solution of lanthanum as a releasing agent is added to all samples and standards.
F
In the presence of very large calcium concentrations (greater than 0.1 %) a molecular absorption from CaOH may be observed. This interference may be overcome by
using background corrections when analyzing for barium.
G
In the presence of high concentrations Pb or Cu, an alternative analytical wavelength of 231.2 nm should be used.
5. Significance and Use
5.1 The health of workers in many industries is at risk through exposure by inhalation to toxic metals. Industrial hygienists and
other public health professionals need to determine the effectiveness of measures taken to control workers’ exposures, and this is
generally achieved by making workplace air measurements. Exposure to some metal-containing particles has been demonstrated
to cause dermatitis, skin ulcers, eye problems, chemical pneumonitis, and other physical disorders (116).
5.2 FAAS is capable of quantitatively determining mostmany metals in air samples at the levels required by federal, state, and
local occupational health and air pollution regulations. The analysis results can be used for the assessment of workplace exposures
to metals in workplace air. The suitability of FAAS for elemental analysis for exposure assessment purposes must be investigated
prior to carrying out workplace air sampling, in consideration of relevant occupational exposure limit values (OELVs) for metals
of concern.
6. Interferences
6.1 In FAAS the occurrence of interferences is less common than in many other analytical techniques. Interferences can occur,
however, and when encountered are corrected for as indicated in the following sections. The known interferences and correction
methods for each metal are indicated in Table 2. The methods of standard additions and background monitoring and correction
(211, 412, 817, 918) are used to identify the presence of an interference. Insofar as possible, the matrix of sample and standard
are matched to minimize the possible interference.
6.2 Background or nonspecific absorption can occur from particles produced in the flame which can scatter light and produce an
apparent absorption signal. Light scattering may be encountered when solutions of high salt content are being analyzed. They are
most severe when measurements are made at shorter wavelengths (for example, below about 250 nm). Background absorption may
also occur as the result of the formation of various molecular species which can absorb light. The background absorption can be
accounted for by the use of background correction techniques (817).
6.3 Spectral interferences are those interferences which result from an atom different from the one being measured that absorbs
a portion of the radiation. Such interferences are extremely rare in FAAS. In some cases multielement hollow cathode lamps may
cause a spectral interference by having closely adjacent emission lines from two different elements. In general, the use of
multielement hollow cathode lamps is discouraged.
Boldface numbers in parentheses refer to the list of references appended to these methods.
D4185 − 23
6.4 Ionization interference occurs when easily ionized atoms are being measured. The degree to which such atoms are ionized is
dependent upon the atomic concentration and the presence of other easily ionized atoms. This interference can be controlled by
the addition of a high concentration of another easily ionized element which will buffer the electron concentration in the flame.
6.5 Chemical interferences occur in FAAS when species present in the sample cause variations in the degree to which atoms are
formed in the flame, or when different valence states of a single element have different absorption characteristics. Such
interferences may be controlled by adjusting the sample matrix or by the method of standard additions (918). Also, the use of
lanthanum as a releasing element minimizes the interference from the formation of nonvolatile compounds in the flame.
Lanthanum forms nonvolatile compounds preferentially with the interferent so that the analyte remains free.
6.6 Physical interferences may result if the physical properties of the samples vary significantly. Changes in viscosity and surface
tension can affect the sample aspiration rate and thus cause erroneous results. Sample dilution or the method of standard additions,
or both, are used to correct such interferences. High concentrations of silica in the sample can cause aspiration problems. No matter
what elements are being determined, if large amounts of silica are extracted from the samples, they shall be allowed to stand for
several hours and centrifuged or filtered to remove the silica.
6.7 This procedure describes a generalized method for sample preparation, which is applicable to the majority of samples. There
are some relatively rare chemical forms of a few of the elements listed in Table 1 that will not be dissolved by this procedure. If
such chemical forms are suspected, results obtained using this procedure shall be compared with those obtained using an
appropriately altered dissolution procedure. Alternatively, the results may be compared with values obtained using a technique that
does not require dissolving the sample (for example, X-ray fluorescence or neutron activation analysis).
7. Apparatus
7.1 Sampling Apparatus:
7.1.1 Cellulose Ester or Cellulose Nitrate Membrane Filters, Samplers, containing mixed cellulose ester (MCE) or cellulose
nitrate membrane filters, with a pore size of 0.8 μm mounted in a 25-mm or 37-mm diameter25 mm or 37 mm diameter, two- or
three-piece filter holder. MCE filters attached to cellulose acetate capsules, which are acid-soluble, are also suitable.
NOTE 1—Appropriate workplace air samplers are described in Test MethodAlternative sampling media, such as quartz fiber filters, may D7035. The
background metal content of the filters should be minimal (see Annex A1 of Test Method be suitable. D7035).
7.1.1.1 Appropriate workplace air samplers are described in Test Method D7035. The background metal content of the filters
should be minimal (see Annex A1 of Test Method D7035).
7.1.2 Portable, Battery-Operated Personal Sampling Pumps, equipped with a flow-monitoring device (rotameter, critical orifice)
or a constant-flow device, and capable of drawing 1–51 L L/min ⁄min to 5 L ⁄min of air through the 0.8-μm a 0.8 μm mixed
cellulose ester membrane filter for a period of at least 8 h.
7.2 Analytical Apparatus:
7.2.1 Flame Atomic Absorption Spectrophotometer, equipped with air/acetylene and nitrous oxide/acetylene burner heads.
7.2.2 Hollow Cathode or Electrodeless Discharge Lamp, for each element to be determined.
7.2.3 Deuterium Continuum Lamp.
7.2.4 Compressed Air—Appropriate pressure reducing regulator with base connections (see instrument manufacturer’s instruc-
tions).
7.2.5 Acetylene Gas and Regulator—A cylinder of acetylene equipped with a two-gage,two-gauge, two-stage pressure-reducing
regulator with hose connections. (See instrument manufacturer instructions.)
7.2.6 Nitrous Oxide Gas and Regulator—A cylinder of nitrous oxide equipped with a two-gage,two-gauge, two-stage
D4185 − 23
pressure-reducing regulator and hose connections. Heat tape with the temperature controlled by a rheostat may be wound around
the second stage regulator and hose connection to prevent freeze-up of the line. (See instrument manufacturer instructions.)
7.2.7 Beakers, Phillips or Griffin, 125-mL,125 mL, borosilicate glass.
7.2.8 Centrifuge Tubes, 15-mL,15 mL, graduated, borosilicate glass.
7.2.9 Miscellaneous Borosilicate Glassware (Pipets and Volumetric Flasks)—All pipets and volumetric flasks shall be calibrated
Class A volumetric glassware.
8. Reagents
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used provided that it can be demonstrated that they are of sufficiently high
purity to permit their use without decreasing the accuracy of the determinations.
8.2 Purity of Water—Unless otherwise indicated, reference to water shall be understood to mean Type II reagent water conforming
to Specification D1193.
8.3 Hydrochloric Acid (HCl)—Concentrated hydrochloric acid, 12 N, specific gravity 1.19.
8.4 Nitric Acid (HNO )—Redistilled, concentrated nitric acid, 16 N, specific gravity 1.42.
8.5 Standard Stock Solutions (1000 μg/mL) for each of the metals listed in Table 1. These solutions are stable for at least one year
when stored in polyethylene bottles, and can be obtained from commercial sources or prepared in the laboratory in the following
manner:
8.5.1 Stock Aluminum Solution—Dissolve 1.000 g of aluminum wire in a minimum volume of 1 + 1 HCl. Dilute to volume in a
1-L1 L flask with purified water.
8.5.2 Stock Barium Solution—Dissolve 1.779 g of barium chloride (BaCl ·2H O) in water. Dilute to volume in a 1-L1 L flask with
2 2
purified water.
8.5.3 Stock Bismuth Solution—Dissolve 1.000 g of bismuth metal in a minimum volume of 6 N HNO . Dilute to volume in a
1-L1 L flask with 2 % (v/v) HNO .
8.5.4 Stock Cadmium Solution—Dissolve 1.000 g of cadmium metal in a minimum volume of 6 N HCl. Dilute to volume in a
1-L1 L flask with 2 % (v(v/v)⁄v) HNO .
8.5.5 Stock Calcium Solution—To 2.497 g of primary standard calcium carbonate (CaCO ) add 50 mL of distilled water. Add
dropwise a minimum volume of HCl (approximately 10 mL) 10 mL) to dissolve the CaCO . Dilute to volume in a 1-L1 L flask
with purified water.
8.5.6 Stock Chromium Solution—Dissolve 3.735 g of potassium chromate (K CrO ) in distilled water. Dilute to volume in a
2 4
1-L1 L flask with purified water.
8.5.7 Stock Cobalt Solution—Dissolve 1.000 g of cobalt metal in a minimum volume of 1 + 1 HCl. Dilute to volume in a 1-L1 L
flask with 2 % (v/v) HNO .
8.5.8 Stock Copper Solution—Dissolve 1.000 g of copper metal in a minimum volume of 6 N HNO . Dilute to volume in a 1-L1 L
flask with 2 % (v(v/v)⁄v) HNO .
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
D4185 − 23
8.5.9 Stock Indium Solution—Dissolve 1.000 g of indium metal in a minimum volume of 1 + 1 HCl. Addition of a few drops of
HNO and mild heating will aid in dissolving the metal. Dilute to volume in a 1-L1 L flask with 2 % (v/v) HNO .
3 3
8.5.10 Stock Iron Solution—Dissolve 1.000 g of iron wire in 50 mL of 6 N HNO . Dilute to volume in a 1-L1 L flask with 2 %
(v/v) HNO .
8.5.11 Stock Lead Solution—Dissolve 1.598 g of lead nitrate (Pb(NO ) ) in 2 % (v ⁄v) HNO . Dilute to volume in a 1-L1 L flask
3 2 3
with 2 % (v(v/v)⁄v) HNO .
8.5.12 Stock Lithium Solution—Dissolve 5.324 g of lithium carbonate (Li CO ) in a minimum volume of 6 N HCl. Dilute to
2 3
volume in a 1-L1 L flask with purified water.
8.5.13 Stock Magnesium Solution—Dissolve 1.000 g of magnesium ribbon in a minimum volume of 6 N HCl. Dilute to volume
in a 1-L1 L flask with 2 % (v(v/v)⁄v) HNO .
8.5.14 Stock Manganese Solution—Dissolve 1.000 g of manganese metal in a minimum volume of 6 N HNO . Dilute to volume
in a 1-L1 L flask with 2 % (v/v) HNO .
8.5.15 Stock Nickel Solution—Dissolve 1.000 g of nickel metal in a minimum volume of 6 N HNO . Dilute to volume in a 1-L1 L
flask with 2 % (v(v/v)⁄v) HNO .
8.5.16 Stock Potassium Solution—Dissolve 1.907 g of potassium chloride (KCl) in purified water. Dilute to volume in a 1-L1 L
flask with purified water.
8.5.17 Stock Rubidium Solution—Dissolve 1.415 g of rubidium chloride (RbCl) in distilled water. Dilute to volume in a 1-L1 L
flask with purified water.
8.5.18 Stock Silver Solution—Dissolve 1.575 g of silver nitrate
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...