ASTM E2524-22
(Test Method)Standard Test Method for Analysis of Hemolytic Properties of Nanoparticles
Standard Test Method for Analysis of Hemolytic Properties of Nanoparticles
SIGNIFICANCE AND USE
5.1 This test method is one of a series of tests listed in Practice F748 and ISO 10993-4 to assess the biocompatibility of materials contacting blood in medical applications.
5.2 This test method is similar to Practice F756 but modified to accommodate nanoparticulate materials.
SCOPE
1.1 This test method covers assessing the effect of nanoparticulate materials on the integrity of red blood cells.
1.2 This test method uses diluted whole blood incubated with nanoparticulate material and the hemoglobin released from damaged red blood cells is determined.
1.3 This test method is similar to Practice F756 with the volumes reduced to accommodate nanoparticulate material.
1.4 This test method is part of the in-vitro preclinical characterization and is important for nanoparticulate material that will contact the blood in medical applications.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2022
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.03 - Environment, Health, and Safety
Relations
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Mar-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jun-2010
- Refers
ASTM F1903-10 - Standard Practice for Testing For Biological Responses to Particles <i>In Vitro</i> - Effective Date
- 01-Jun-2010
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-May-2004
- Refers
ASTM F1903-98(2003) - Standard Practice for Testing for Biological Responses to Particles In Vitro - Effective Date
- 01-Nov-2003
- Effective Date
- 10-Apr-2003
Overview
ASTM E2524-22: Standard Test Method for Analysis of Hemolytic Properties of Nanoparticles sets forth procedures to evaluate the hemolytic activity of nanoparticulate materials in contact with blood. Nanoparticles are increasingly used in medical devices and drug delivery systems, making comprehensive assessment of their biocompatibility essential. This standard adapts and refines established test methods for hemolysis to accurately measure the integrity of red blood cells following exposure to nanoscale materials, ensuring their safety for blood-contacting applications.
Key Topics
- Hemolysis Assessment: This standard determines if nanoparticles cause the lysis (destruction) of red blood cells by quantifying hemoglobin release into plasma after incubation with the test material.
- In Vitro Characterization: Focus is on preclinical, laboratory-based testing using diluted human blood, assessing acute cytotoxicity before in vivo or clinical studies.
- Modified Protocols for Nanoparticles: ASTM E2524-22 modifies prior test methods (notably ASTM F756) to accommodate the unique characteristics of nanomaterials, such as altered volumes, dispersion, and possible assay interferences.
- Interference Considerations: Special attention is given to potential optical or physical interferences from nanoparticles, which might absorb light at assay wavelengths or adsorb hemoglobin, impacting results.
- Quality Control and Calibration: The method prescribes defined procedures for calibration and quality control, ensuring assay reliability and reproducibility.
- Application of International Protocols: Aligns with recommendations from ISO 10993-4 and is part of the broader biocompatibility assessment series in Practice ASTM F748.
Applications
ASTM E2524-22 is critically important for:
- Medical Device Evaluation: Ensuring the safety of devices such as intravenous catheters, implantable sensors, and stents, where nanomaterials come into direct blood contact.
- Nanomedicine Development: Supporting the preclinical validation of nanoparticle-based drug formulations, imaging agents, and targeted therapeutics intended for intravenous administration.
- Regulatory Submissions: Providing a recognized testing protocol cited in international guidance (e.g., ISO 10993-4) for regulatory submissions and safety assessments required by global health authorities.
- Research and Toxicology: Used in toxicological studies to screen for acute adverse effects related to nanoparticle-blood interactions, facilitating advancement in safe biomedical technology.
Related Standards
For a comprehensive biocompatibility assessment and method validation, the following related standards are often referenced with ASTM E2524-22:
- ASTM F748 - Practice for Selecting Generic Biological Test Methods for Materials and Devices: Outlines selection criteria for biological evaluation.
- ASTM F756 - Practice for Assessment of Hemolytic Properties of Materials: The foundational hemolysis method for non-nano materials.
- ISO 10993-4 - Biological Evaluation of Medical Devices – Part 4: Selection of Tests for Interactions with Blood: International guide for blood-contacting materials testing.
- ASTM F1877 - Practice for Characterization of Particles: Relevant for proper nanoparticle characterization prior to testing.
- ASTM F1903 - Practice for Testing for Cellular Responses to Particulates: Addresses broader cytotoxicity endpoints for particulate materials.
Practical Value
Adherence to ASTM E2524-22 ensures that manufacturers, researchers, and regulatory professionals can systematically evaluate the hemolytic properties of nanoparticles in blood-contacting medical products. By providing a standardized, internationally aligned approach to in vitro hemolysis testing, this standard supports the development of safe, effective, and regulatory-compliant nanomedical innovations.
Buy Documents
ASTM E2524-22 - Standard Test Method for Analysis of Hemolytic Properties of Nanoparticles
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E2524-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Analysis of Hemolytic Properties of Nanoparticles". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is one of a series of tests listed in Practice F748 and ISO 10993-4 to assess the biocompatibility of materials contacting blood in medical applications. 5.2 This test method is similar to Practice F756 but modified to accommodate nanoparticulate materials. SCOPE 1.1 This test method covers assessing the effect of nanoparticulate materials on the integrity of red blood cells. 1.2 This test method uses diluted whole blood incubated with nanoparticulate material and the hemoglobin released from damaged red blood cells is determined. 1.3 This test method is similar to Practice F756 with the volumes reduced to accommodate nanoparticulate material. 1.4 This test method is part of the in-vitro preclinical characterization and is important for nanoparticulate material that will contact the blood in medical applications. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is one of a series of tests listed in Practice F748 and ISO 10993-4 to assess the biocompatibility of materials contacting blood in medical applications. 5.2 This test method is similar to Practice F756 but modified to accommodate nanoparticulate materials. SCOPE 1.1 This test method covers assessing the effect of nanoparticulate materials on the integrity of red blood cells. 1.2 This test method uses diluted whole blood incubated with nanoparticulate material and the hemoglobin released from damaged red blood cells is determined. 1.3 This test method is similar to Practice F756 with the volumes reduced to accommodate nanoparticulate material. 1.4 This test method is part of the in-vitro preclinical characterization and is important for nanoparticulate material that will contact the blood in medical applications. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2524-22 is classified under the following ICS (International Classification for Standards) categories: 11.100.01 - Laboratory medicine in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2524-22 has the following relationships with other standards: It is inter standard links to ASTM F1903-18, ASTM F756-17, ASTM F1877-16, ASTM F748-16, ASTM F756-13, ASTM F748-06(2010), ASTM F1877-05(2010), ASTM F1903-10, ASTM F756-08, ASTM F748-06, ASTM F1877-05, ASTM F1877-05e1, ASTM F748-04, ASTM F1903-98(2003), ASTM F1877-98(2003)e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2524-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2524 − 22
Standard Test Method for
Analysis of Hemolytic Properties of Nanoparticles
This standard is issued under the fixed designation E2524; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F1877Practice for Characterization of Particles
F1903Practice for Testing for Cellular Responses to Par-
1.1 This test method covers assessing the effect of nanopar-
ticles in vitro
ticulate materials on the integrity of red blood cells.
2.2 ISO Standard:
1.2 This test method uses diluted whole blood incubated
ISO 10993-4Biological Evaluation of Medical Devices Part
with nanoparticulate material and the hemoglobin released
4: Selection of Tests for Interactions with Blood
from damaged red blood cells is determined.
3. Terminology
1.3 This test method is similar to Practice F756 with the
volumes reduced to accommodate nanoparticulate material.
3.1 Acronyms:
3.1.1 Cal—calibration standard
1.4 This test method is part of the in-vitro preclinical
characterization and is important for nanoparticulate material
3.1.2 CMH—cyanmethemoglobin
that will contact the blood in medical applications.
3.1.3 DPBS—Dulbecco’s phosphate-buffered saline
1.5 The values stated in SI units are to be regarded as
3.1.4 PFH—plasma-free hemoglobin
standard. No other units of measurement are included in this
3.1.5 QC—quality controls
standard.
3.1.6 TBH—total blood hemoglobin
1.6 This standard does not purport to address all of the
3.1.7 TBHd—blood sample diluted to 10 mg 6 1 mg/mL
safety concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
4. Summary of Test Method
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
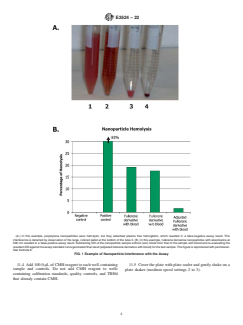
4.1 Thistestmethoddescribesaprotocolforassessingacute
1.7 This international standard was developed in accor-
in-vitro damage to red blood cells (that is, hemolysis) caused
dance with internationally recognized principles on standard-
by exposure to nanoparticles.
ization established in the Decision on Principles for the
4.2 This test method is based on the quantitative determi-
Development of International Standards, Guides and Recom-
nation of hemoglobin released into PFH as a percentage of the
mendations issued by the World Trade Organization Technical
TBH concentration when blood is exposed to nanoparticulate
Barriers to Trade (TBT) Committee.
materials.
2. Referenced Documents
4.3 Using an established colorimetric assay, hemoglobin
and its derivatives, such as sulfhemoglobin, are oxidized to
2.1 ASTM Standards:
methemoglobin by ferricyanide in the presence of alkali. A
F748PracticeforSelectingGenericBiologicalTestMethods
stable CMH concentration is measured using a plate reader
for Materials and Devices
spectrophotometer set at 540 nm.
F756Practice for Assessment of Hemolytic Properties of
Materials
4.4 Hemoglobin standards are used to create a standard
curvecoveringtherangefrom0.025mg⁄mLto0.8mg/mLand
prepare quality control samples at low (0.0625-mg/mL), mid
This test method is under the jurisdiction of ASTM Committee E56 on
(0.125-mg/mL), and high (0.625-mg/mL) concentrations to
Nanotechnology and is the direct responsibility of Subcommittee E56.03 on
Environment, Health, and Safety.
monitor assay performance. The required sample volume is
Current edition approved Sept. 1, 2022. Published October 2022. Originally
100 µL per test replicate.
approved in 2008. Last previous edition approved in 2013 as E2524 – 08(2013),
which was withdrawn in April 2022 and reinstated in September 2022. DOI:
10.1520/E2524-22.
2 3
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM 4th Floor, New York, NY 10036, http://www.ansi.org.
Standards volume information, refer to the standard’s Document Summary page on International Committee for Standardization in Haemotology, Journal of
the ASTM website. Clinical Pathology, Vol 31, 1978, pp. 139–143.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2524 − 22
TABLE 2 Quality Controls
4.5 The results are expressed as percent hemolysis to
evaluate the acute in-vitro hemolytic properties of nanopar- Nominal
Level Conc., Preparation Procedure
ticles.
mg/mL
QC 1 0.625 1.5 mL of stock solution + 0.42 mL of CMH reagent
5. Significance and Use
QC 2 0.125 200 µL of QC1 + 800 µL of CMH reagent
QC 3 0.0625 100 µL of QC1 + 900 µL of CMH reagent
5.1 This test method is one of a series of tests listed in
Practice F748 and ISO 10993-4 to assess the biocompatibility
of materials contacting blood in medical applications.
NOTE 1—The source of the reagents is shown for information purposes
5.2 ThistestmethodissimilartoPracticeF756butmodified
onlytoaidlaboratoriesinitiatingthisprocedure.Equivalentreagentsfrom
to accommodate nanoparticulate materials.
other suppliers can be used. Unless otherwise stated, reagents should be
used within their stated validity period (for example, before the manufac-
6. Apparatus
turer’s expiration date); processed reagents can have storage or use
periods shorter than the source chemical or material.
6.1 Pipettes covering range from 0.05mL to 10 mL.
6.2 Ninety-six well plates.
8. Preparation of Standards and Controls
NOTE 2—Aseptic precautions are not needed, but contamination of
6.3 Water bath set at 37°C 6 1°C.
reagents to be stored shall be avoided.
6.4 Plate reader capable of measuring at 540 nm.
8.1 Preparation of CMH Reagent—The CMH reagent is
6.5 Plate shaker.
supplied as a ready-to-use liquid.
6.6 Plastic beakers.
8.2 Preparation of Calibration Standards—Hemoglobin
standard is a commercially available reagent supplied as a
6.7 Microcentrifuge tubes, 1.5 mL, translucent, not colored.
solution with a fixed concentration.This stock is diluted in the
6.8 Centrifuge set at 700 g to 800 g.
CMH reagent to prepare calibration standards; mixing of
diluted samples is conducted by vortexing; volumes can be
7. Reagents
adjusted based on the need (see Table 1).
7.1 Purity of Reagents—Reagent-grade chemicals shall be
8.3 Preparation of Quality Controls—These controls are
used in all tests. Unless otherwise indicated, it is intended that
prepared from an independent aliquot of the hemoglobin stock
all reagents conform to the specifications of the Committee on
reagent; volumes can be adjusted based on the need (see Table
Analytical Reagents of theAmerican Chemical Society where
2).
such specifications are available. Other grades may be used,
provided it is first ascertained that the reagent is of sufficiently
8.4 Preparation of Positive Controls—Prepare 10% solu-
high purity to permit its use without lessening the accuracy of
tion (v/v) of Triton X-100 in water. Prepare aliquots for daily
the determination.
use and store at –20°C 6 3°C for up to 2 months.
2+ 2+
7.2 CMH reagent (also known as Drabkin’s reagent).
8.5 Preparation of Negative Control—Use Ca /Mg -free
DPBS as the negative control. Store the stock solution at 4°C
7.3 Hemoglobin standard.
6 3°C.
2+ 2+
7.4 Ca /Mg -free DPBS.
8.6 Preparation of Nanoparticulate Test Samples:
7.5 Pooled normal human whole blood anticoagulated with
8.6.1 For the initial dose, use the highest concentration of
Li-heparin.
nanoparticlesthatiswelldispersedinaphysiologicsolution.If
7.6 Triton X-100.
the concentration in the end-use application is known, that can
beusedasthestartingconcentration.Thematerialshallbewell
7.7 Distilled water.
characterized under physiological conditions according to
standard methods including those recommended in Practices
Reagent Chemicals, American Chemical Society Specifications, American F1877 and F1903. The nanoparticulate material for testing
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
shall be in physiologic solution which is isotonic with a pH of
listed by the American Chemical Society, see Analar Standards for Laboratory
7.2 62.Thissolutionshallbedefinedandtheparticlesshallbe
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
characterized in this solution. The number of particles/mLand
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
MD.
mg/mL shall be indicated. Prior characterization shall be
performedasappropriatetoallowadequatedatainterpretation.
TABLE 1 Calibration Standards
For example, lot-to-lot variations in particle size and surface
Nominal characteristics of the particles could result in different assay
Level Conc., Preparation Procedure
results. If the particles suspension is sterile, the method of
mg/mL
sterilization shall be indicated. The nanoparticulate material
Cal 1 0.8 2 mL of stock solution
and the buffer used for its storage/reconstitution shall be tested
Cal2 0.4 1mLofCal1+1mLofCMHreagent
Cal3 0.2 1mLofCal2+1mLofCMHreagent in the same assay. The assay requires at least 300 mL of test
Cal4 0.1 1mLofCal3+1mLofCMHreagent
material and enough for diluting.The starting suspension shall
Cal5 0.05 1mLofCal4+1mLofCMHreagent
be diluted in DPBS with serial one to five (1:5) dilutions at
Cal6 0.025 1mLofCal5+1mLofCMHreagent
least three times to give four test samples in the assay.
E2524 − 22
8.6.2 Sincesomenanoparticulatematerialscanabsorbatthe 10. Procedure with the Test Material
designated 540-nm wavelength, it is suggested that a trial of 2+ 2+
10.1 DilutepooledwholebloodwithCa /Mg -freeDPBS
the material in DPBS be tested at 540 nm. If absorption is
to adjust TBH concentration to 10mg⁄mL 6 1 mg/mL based
evident, it is advisable to determine if high-speed centrifuga-
on results from 9.5.
tion will pellet the particles and remove the interference. The
10.2 Add 100.0 µL of sample, reagent blank (PBS or other
maximum centrifugal force attainable using a microcentrifuge
bufferusedtodissolvenanoparticles),positivecontrolsolution,
is typically 16000 g to 22000 g, corresponding to speeds
andnegativecontrolsolutiontomicrocentrifugetubes.Prepare
upwardsof18000r⁄min.(SeeNote3.)Forthisprocedure,the
six tubes for each unknown sample, three tubes for the blank,
maximum force available should be applied for 30 min. If
two tubes for the positive control, and two tubes for the
successful, lower speeds can be tested if desired. When
negative control.
centrifugation is not applicable, an assay result obtained for a
2+ 2+
10.3 Add 700.0 µL of Ca /Mg -free DPBS to each tube.
particle incubated with blood is adjusted by subtracting result
obtained for the same particle in “minus blood” control (see
10.4 Add 100 µL of the whole blood prepared in 10.1 to
10.4).
each tube, except for three tubes of each test sample. In these
2+ 2+
tubes, add 100 µL of Ca /Mg -free DPBS. These samples
NOTE3—Therelationshipbetweencentrifugationspeedandtherelative
represent a “minus blood” control and are used to evaluate
centrifugal force (RCF, measured in multiples of gg, the force of gravity
potential interference of nanomaterial with the assay (for
at the Earth’s surface) depends on the radius (r) of the rotor, where RCF
= (r/min/1000) 2 × 1.12 × r (mm).
example, absorbance at or close to 540 nm, reactivity with
CMH reagent, and s
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...