ASTM D8126-21
(Test Method)Standard Test Method for Efficient Basicity Determination by Potentiometric Hydrochloric Acid Titration
Standard Test Method for Efficient Basicity Determination by Potentiometric Hydrochloric Acid Titration
SIGNIFICANCE AND USE
5.1 Lubricants can contain basic constituents that are present as additives. The relative amount of these materials can be determined by titration with acids. The base number is a measurement of the amount of basic substances in the oil under the conditions of the testing procedure.
5.2 A primary objective of a marine cylinder lubricant is the neutralization of sulfuric acid, produced during fuel combustion, to protect the engine from corrosion. The EBas method characterizes the more efficient basic species of the lubricant reacting with acids from the beginning of the neutralization (when the lubricant milieu is basic) until an equilibrium state where the lubricant becomes slightly acidic. The use of hydrochloric acid (HCl) allows differentiating basicities of various strengths during titration.
5.3 In marine lubricants, the constituents that can be considered to have basic properties are primarily organic and inorganic bases coming from the detergent. Basicity can also be brought to the lubricant by other components including dispersants, amino compounds, or any organic basic components. This test method uses the same titration system as that of Test Method D4739, however the evaluation and interpretation of the titration result is different. The hydrochloric acid is used as the titrant in this test method and Test Method D4739, whereas Test Method D2896 uses a stronger acid, perchloric acid. While all three methods can be considered as complementary to evaluate the basicity of lubricants, this test method is only used for marine cylinder lubricants.
5.4 This test method measures only the efficient basicity of the lubricant, which represents only the part of the total basicity, until the point of which the lubricant becomes slightly acidic. It will depend on the components of the formulation itself. In that respect, the EBas cannot be compared to base number given by Test Methods D4739 or D2896. The EBas is then complementary information to the base numbe...
SCOPE
1.1 This test method covers a procedure for the determination of the efficient basicity (EBas) in new marine cylinder lubricants that lubricate the upper part, that is, piston-ring-cylinder area, of two-stroke marine engines.
1.2 This test method has been developed for marine cylinder lubricants, having base number (BN) (measured by Test Method D2896) from 20 mg KOH/g to 100 mg KOH/g, and an EBas from 10 mg KOH/g to 36 mg KOH/g.
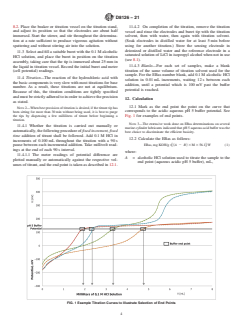
1.3 In this test method, only the efficient basicity is determined, corresponding to the components or part of components that have a major role in neutralization of the acidic species formed in the combustion of the fuel in the marine engine. It differentiates between the fastest reacting species and the last one to react like overbasing compounds in detergent (classically mineral calcium carbonate, CaCO3). The values obtained, however, are intended to be compared with the other values obtained by this test method only; base numbers obtained by this test method are not intended to be equal to values by other test methods.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Some specific hazards statements are given in Sections 6 and 7.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2021
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.06 - Analysis of Liquid Fuels and Lubricants
Relations
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2019
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Jun-2015
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 15-Jul-2013
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
- Effective Date
- 15-Dec-2008
- Effective Date
- 01-Feb-2008
- Effective Date
- 15-Jul-2007
- Effective Date
- 01-Jul-2007
Overview
ASTM D8126-21 is the Standard Test Method for Efficient Basicity Determination by Potentiometric Hydrochloric Acid Titration. Published by ASTM International, this standard outlines a precise procedure for measuring the efficient basicity (EBas) in new marine cylinder lubricants. Specifically designed for lubricants used in the piston-ring-cylinder area of two-stroke marine engines, D8126-21 is essential for quality assurance and formulation optimization within the marine lubricants industry. The efficient basicity determined by this method identifies the active basic species crucial for the neutralization of acidic byproducts generated during fuel combustion, helping protect engine components from corrosive damage.
Key Topics
Efficient Basicity (EBas):
- Represents the portion of basic components in marine lubricants that actively neutralize acidic species, especially sulfuric acid from fuel combustion.
- Measurement is specific to fast-reacting basic species, differentiating them from slow-reacting overbased compounds.
Potentiometric Titration with Hydrochloric Acid (HCl):
- Employs automatic or manual potentiometric titration methods using standardized alcoholic HCl as the titrant.
- Utilizes a pH 5 aqueous buffer to define the endpoint, ensuring consistent measurement of efficient basicity across samples.
Application Range:
- Developed for marine cylinder lubricants with base numbers (BN) from 20 mg KOH/g to 100 mg KOH/g and EBas values from 10 mg KOH/g to 36 mg KOH/g.
Significance in Marine Engines:
- EBas is crucial for evaluating a lubricant’s ability to neutralize strong acids and thereby prevent engine corrosion.
- Provides complementary data to other methods measuring total base number, offering insights into the efficiency of acid neutralization.
Precision and Safety:
- Details requirements for apparatus precision and sample preparation to ensure repeatability and reproducibility.
- Notes the importance of safety, health, and environmental practices, and references specific safety concerns.
Applications
- Marine Cylinder Lubricant Quality Control:
- Ensures that lubricants used in two-stroke marine diesel engines provide effective corrosion protection by quantifying the proportion of rapidly neutralizing base compounds.
- Lubricant Formulation Development:
- Assists formulators in optimizing detergent and dispersant content to maximize protective efficacy.
- Comparative Lubricant Assessment:
- Enables benchmarking of marine lubricants by comparing efficient basicity results, supporting evidence-based selection for specific engine conditions.
- Regulatory and Contractual Compliance:
- Facilitates compliance with marine engine manufacturer specifications and international shipping standards.
Related Standards
- ASTM D2896 – Test Method for Base Number of Petroleum Products by Potentiometric Perchloric Acid Titration: Used for total base number determination with a stronger acid.
- ASTM D4739 – Test Method for Base Number Determination by Potentiometric Hydrochloric Acid Titration: Measures a base number but with different interpretation criteria.
- ASTM D1193 – Specification for Reagent Water: Ensures water purity for reagent preparation.
- ASTM D4057 / ASTM D4177 – Practices for manual and automatic sampling of petroleum products, ensuring sample representativeness.
- ASTM D6300 – Practice for Determination of Precision and Bias Data for Use in Test Methods for Petroleum Products.
Conclusion
ASTM D8126-21 provides a robust test method for efficient basicity determination in marine cylinder lubricants via potentiometric hydrochloric acid titration. Its focus on efficient basicity offers practical insight into the functional additives actively protecting marine engines from corrosion. When used alongside complementary standards, D8126-21 enhances quality control and technical evaluation, supporting safe and effective marine lubricant performance.
Keywords: ASTM D8126-21, efficient basicity, EBas, potentiometric titration, hydrochloric acid titration, marine cylinder lubricants, corrosion protection, base number, marine engines, lubricant performance.
Buy Documents
ASTM D8126-21 - Standard Test Method for Efficient Basicity Determination by Potentiometric Hydrochloric Acid Titration
REDLINE ASTM D8126-21 - Standard Test Method for Efficient Basicity Determination by Potentiometric Hydrochloric Acid Titration
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D8126-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Efficient Basicity Determination by Potentiometric Hydrochloric Acid Titration". This standard covers: SIGNIFICANCE AND USE 5.1 Lubricants can contain basic constituents that are present as additives. The relative amount of these materials can be determined by titration with acids. The base number is a measurement of the amount of basic substances in the oil under the conditions of the testing procedure. 5.2 A primary objective of a marine cylinder lubricant is the neutralization of sulfuric acid, produced during fuel combustion, to protect the engine from corrosion. The EBas method characterizes the more efficient basic species of the lubricant reacting with acids from the beginning of the neutralization (when the lubricant milieu is basic) until an equilibrium state where the lubricant becomes slightly acidic. The use of hydrochloric acid (HCl) allows differentiating basicities of various strengths during titration. 5.3 In marine lubricants, the constituents that can be considered to have basic properties are primarily organic and inorganic bases coming from the detergent. Basicity can also be brought to the lubricant by other components including dispersants, amino compounds, or any organic basic components. This test method uses the same titration system as that of Test Method D4739, however the evaluation and interpretation of the titration result is different. The hydrochloric acid is used as the titrant in this test method and Test Method D4739, whereas Test Method D2896 uses a stronger acid, perchloric acid. While all three methods can be considered as complementary to evaluate the basicity of lubricants, this test method is only used for marine cylinder lubricants. 5.4 This test method measures only the efficient basicity of the lubricant, which represents only the part of the total basicity, until the point of which the lubricant becomes slightly acidic. It will depend on the components of the formulation itself. In that respect, the EBas cannot be compared to base number given by Test Methods D4739 or D2896. The EBas is then complementary information to the base numbe... SCOPE 1.1 This test method covers a procedure for the determination of the efficient basicity (EBas) in new marine cylinder lubricants that lubricate the upper part, that is, piston-ring-cylinder area, of two-stroke marine engines. 1.2 This test method has been developed for marine cylinder lubricants, having base number (BN) (measured by Test Method D2896) from 20 mg KOH/g to 100 mg KOH/g, and an EBas from 10 mg KOH/g to 36 mg KOH/g. 1.3 In this test method, only the efficient basicity is determined, corresponding to the components or part of components that have a major role in neutralization of the acidic species formed in the combustion of the fuel in the marine engine. It differentiates between the fastest reacting species and the last one to react like overbasing compounds in detergent (classically mineral calcium carbonate, CaCO3). The values obtained, however, are intended to be compared with the other values obtained by this test method only; base numbers obtained by this test method are not intended to be equal to values by other test methods. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Some specific hazards statements are given in Sections 6 and 7. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Lubricants can contain basic constituents that are present as additives. The relative amount of these materials can be determined by titration with acids. The base number is a measurement of the amount of basic substances in the oil under the conditions of the testing procedure. 5.2 A primary objective of a marine cylinder lubricant is the neutralization of sulfuric acid, produced during fuel combustion, to protect the engine from corrosion. The EBas method characterizes the more efficient basic species of the lubricant reacting with acids from the beginning of the neutralization (when the lubricant milieu is basic) until an equilibrium state where the lubricant becomes slightly acidic. The use of hydrochloric acid (HCl) allows differentiating basicities of various strengths during titration. 5.3 In marine lubricants, the constituents that can be considered to have basic properties are primarily organic and inorganic bases coming from the detergent. Basicity can also be brought to the lubricant by other components including dispersants, amino compounds, or any organic basic components. This test method uses the same titration system as that of Test Method D4739, however the evaluation and interpretation of the titration result is different. The hydrochloric acid is used as the titrant in this test method and Test Method D4739, whereas Test Method D2896 uses a stronger acid, perchloric acid. While all three methods can be considered as complementary to evaluate the basicity of lubricants, this test method is only used for marine cylinder lubricants. 5.4 This test method measures only the efficient basicity of the lubricant, which represents only the part of the total basicity, until the point of which the lubricant becomes slightly acidic. It will depend on the components of the formulation itself. In that respect, the EBas cannot be compared to base number given by Test Methods D4739 or D2896. The EBas is then complementary information to the base numbe... SCOPE 1.1 This test method covers a procedure for the determination of the efficient basicity (EBas) in new marine cylinder lubricants that lubricate the upper part, that is, piston-ring-cylinder area, of two-stroke marine engines. 1.2 This test method has been developed for marine cylinder lubricants, having base number (BN) (measured by Test Method D2896) from 20 mg KOH/g to 100 mg KOH/g, and an EBas from 10 mg KOH/g to 36 mg KOH/g. 1.3 In this test method, only the efficient basicity is determined, corresponding to the components or part of components that have a major role in neutralization of the acidic species formed in the combustion of the fuel in the marine engine. It differentiates between the fastest reacting species and the last one to react like overbasing compounds in detergent (classically mineral calcium carbonate, CaCO3). The values obtained, however, are intended to be compared with the other values obtained by this test method only; base numbers obtained by this test method are not intended to be equal to values by other test methods. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Some specific hazards statements are given in Sections 6 and 7. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D8126-21 is classified under the following ICS (International Classification for Standards) categories: 75.080 - Petroleum products in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8126-21 has the following relationships with other standards: It is inter standard links to ASTM D6300-24, ASTM D6300-23a, ASTM D6300-19a, ASTM D6300-16, ASTM D6300-15, ASTM D6300-14a, ASTM D6300-14ae1, ASTM D6300-14, ASTM D6300-13a, ASTM D6300-13, ASTM D4057-06(2011), ASTM D6300-08, ASTM D4739-08, ASTM D2896-07a, ASTM D6300-07. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8126-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8126 − 21
Standard Test Method for
Efficient Basicity Determination by Potentiometric
Hydrochloric Acid Titration
This standard is issued under the fixed designation D8126; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
1.1 This test method covers a procedure for the determina- 2.1 ASTM Standards:
tion of the efficient basicity (EBas) in new marine cylinder D1193Specification for Reagent Water
lubricants that lubricate the upper part, that is, piston-ring- D2896TestMethodforBaseNumberofPetroleumProducts
cylinder area, of two-stroke marine engines. by Potentiometric Perchloric Acid Titration
D4057Practice for Manual Sampling of Petroleum and
1.2 Thistestmethodhasbeendevelopedformarinecylinder
Petroleum Products
lubricants, having base number (BN) (measured by Test
D4177Practice for Automatic Sampling of Petroleum and
MethodD2896)from20mgKOH/gto100mgKOH⁄g,andan
Petroleum Products
EBas from 10mg KOH/g to 36mg KOH/g.
D4739Test Method for Base Number Determination by
1.3 In this test method, only the efficient basicity is
Potentiometric Hydrochloric Acid Titration
determined, corresponding to the components or part of com-
D6300Practice for Determination of Precision and Bias
ponents that have a major role in neutralization of the acidic
Data for Use in Test Methods for Petroleum Products,
species formed in the combustion of the fuel in the marine
Liquid Fuels, and Lubricants
engine.Itdifferentiatesbetweenthefastestreactingspeciesand
the last one to react like overbasing compounds in detergent
3. Terminology
(classically mineral calcium carbonate, CaCO ). The values
3.1 Definitions:
obtained, however, are intended to be compared with the other
3.1.1 base number, n—the quantity of a specified acid,
values obtained by this test method only; base numbers
expressed in terms of the equivalent number of milligrams of
obtained by this test method are not intended to be equal to
potassium hydroxide per gram of sample, required to titrate a
values by other test methods.
sample in a specified solvent to a specified endpoint using a
1.4 The values stated in SI units are to be regarded as
specified detection system.
standard. No other units of measurement are included in this
3.1.2 effıcient basicity, EBas, n—the quantity of hydrochlo-
standard.
ric acid, expressed in terms of the equivalent number of
1.5 This standard does not purport to address all of the
milligrams of potassium hydroxide per gram of sample, re-
safety concerns, if any, associated with its use. It is the
quired to neutralize the basic components, which are organic
responsibility of the user of this standard to establish appro-
species, of the tested lubricant in a specified solvent to a
priate safety, health, and environmental practices and deter-
specified buffer endpoint using a specified detection system.
mine the applicability of regulatory limitations prior to use.
Some specific hazards statements are given in Sections 6 and 7.
4. Summary of Test Method
1.6 This international standard was developed in accor-
4.1 The sample is dissolved in a mixture of toluene,
dance with internationally recognized principles on standard-
propan-2-ol (isopropyl alcohol), chloroform, and a small
ization established in the Decision on Principles for the
amount of water and titrated potentiometrically with alcoholic
Development of International Standards, Guides and Recom-
hydrochloric acid solution. The test results of this procedure
mendations issued by the World Trade Organization Technical
are obtained by titration mode of fixed increment and fixed
Barriers to Trade (TBT) Committee.
time additions of the titrant. An endpoint is selected from a
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.06 on Analysis of Liquid Fuels and Lubricants. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2021. Published November 2021. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2017. Last previous edition approved in 2017 as D8126–17. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D8126-21. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8126 − 21
titrationcurveaccordingtothecriteriagiveninSection12and 6.2 Sensing Electrode, standard pH with glass membrane,
used to calculate the EBas. suitable for non-aqueous titrations.
6.3 Reference Electrode, Silver/Silver Chloride (Ag/AgCl)
5. Significance and Use
referenceelectrodewithsleevejunction,filledwith1Mto3M
lithium chloride (LiCl) in ethanol.
5.1 Lubricants can contain basic constituents that are pres-
ent as additives. The relative amount of these materials can be 6.3.1 Combination Electrode—Sensingelectrodesmayhave
the silver/silver chloride (Ag/AgCl) reference electrode built
determined by titration with acids. The base number is a
measurementoftheamountofbasicsubstancesintheoilunder into the same electrode body, which offers the convenience of
working with and maintaining only one electrode. The combi-
the conditions of the testing procedure.
nation electrode shall have a sleeve junction type of reference
5.2 Aprimaryobjectiveofamarinecylinderlubricantisthe
and shall use an inert ethanol electrolyte, for example, 1M to
neutralization of sulfuric acid, produced during fuel
3M LiCl in ethanol.
combustion, to protect the engine from corrosion. The EBas
6.3.1.1 In the reference compartment, the sensing electrode
method characterizes the more efficient basic species of the
part shall use a glass membrane designed for nonaqueous
lubricant reacting with acids from the beginning of the neu-
titrations. These combination electrodes shall have the same
tralization (when the lubricant milieu is basic) until an equi-
response or better response than a dual electrode system.They
librium state where the lubricant becomes slightly acidic. The
shall have removable sleeves for easy rinsing and addition of
use of hydrochloric acid (HCl) allows differentiating basicities
electrolyte. When a movable sleeve is part of the electrode
of various strengths during titration.
system, ensure that the sleeve is unimpaired before every
5.3 In marine lubricants, the constituents that can be con-
titration.
sidered to have basic properties are primarily organic and
6.4 Stirrer, Buret, Stand, Titration Vessel, as specified in
inorganic bases coming from the detergent. Basicity can also
Annex A1, are required.
be brought to the lubricant by other components including
dispersants, amino compounds, or any organic basic compo-
7. Reagents
nents.Thistestmethodusesthesametitrationsystemasthatof
TestMethodD4739,howevertheevaluationandinterpretation
7.1 Purity of Reagents—Reagent-grade chemicals shall be
of the titration result is different.The hydrochloric acid is used used in all tests. Unless otherwise indicated, it is intended that
as the titrant in this test method and Test Method D4739,
all reagents shall conform to the specifications of the Commit-
whereas Test Method D2896 uses a stronger acid, perchloric tee onAnalytical Reagents of theAmerican Chemical Society,
acid. While all three methods can be considered as comple- where such specifications are available. Other grades may be
mentary to evaluate the basicity of lubricants, this test method used, provided it is first ascertained that the reagent is of
is only used for marine cylinder lubricants. sufficiently high purity to permit its use without lessening the
accuracy of the determination.
5.4 This test method measures only the efficient basicity of
the lubricant, which represents only the part of the total 7.2 Purity of Water—Unless otherwise indicated, references
basicity,untilthepointofwhichthelubricantbecomesslightly
to water shall be understood to mean reagent water that meets
acidic. It will depend on the components of the formulation the requirement of either Type I, II, or III of Specification
itself. In that respect, the EBas cannot be compared to base
D1193.
number given by Test Methods D4739 or D2896. The EBas is
7.3 Buffer, Aqueous Acid—CommercialpH5buffersolution
then complementary information to the base number, giving
withatoleranceof 60.02pHunitsat25°C.Thissolutionshall
performance in efficiency of neutralization of acidic species.
be replaced at regular intervals consistent with its stability or
when contamination is suspected. Information related to the
6. Apparatus
stability should be obtained from the manufacturer.
6.1 Potentiometric Titration, automatic or manual, with
7.4 Chloroform—Reagent grade. (Warning—Toxic and
capability of adding fixed increments of titrant at fixed time
suspected carcinogen.)
intervals (see Annex A1).
7.5 Hydrochloric Acid Solution, Standard Alcoholic (0.1
6.1.1 The titrimeter must automatically (or manually) con-
M)—Mix 9mLof reagent grade hydrochloric acid (HCl, sp gr
trol the rate of addition of titrant as follows: Delivery of titrant
1.19) (Warning—Toxic and corrosive), with 1Lof anhydrous
will be incremental; after delivery of precisely a 0.100mL
isopropyl alcohol. Standardize frequently enough to detect
increment (see 6.1.2), the delivery is stopped and a fixed time
normality changes of 0.0005 by potentiometric titration. of
period of 90s is allowed to pass before another 0.100mL
approximately 0.1g (weigh accurately at the nearest 0.0001g)
increment of titrant is delivered. This procedure is repeated
until the titration is completed.
6.1.2 The precision of addition of the 0.100mL increments
of titrant must be 60.001mL for automatic titrators. For
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
manual buret, it should be 60.005mL. A higher incremental
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
precision is required for an automatic buret, because the total
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
volume to the end point is summed from the individual
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
increments; it is read from a scale with a manual buret. copeial Convention, Inc. (USPC), Rockville, MD.
D8126 − 21
of primary standard Tris(hydroxymethyl)aminomethane dis- 8.1.2 When not in use, immerse the lower halves of the
solved in 60mL CO -free water. electrodes in either water (sensing) or the LiCl in isopropyl
alcohol electrolyte (reference). Do not allow them to remain
7.6 Ethanol—reagent grade. (Warning—Flammable and
immersedintitrationsolventforanyappreciableperiodoftime
toxic, especially when denatured.)
between titrations. While the electrodes are not extremely
7.7 Lithium Chloride Electrolyte—Prepareasolutionof1M
fragile, handle them carefully at all times.
to 3M LiCl in ethanol.
8.1.3 Electrode Life—Typically, electrode usage is limited
to 3 months to 6 months, depending upon usage. Electrodes
7.8 Propan-2-ol(IsopropylAlcohol)—Anhydrous,(lessthan
have a limited shelf life and shall be tested before use (8.3).
0.1% H O). (Warning—Flammable.)
8.2 Preparation of Electrodes:
7.9 Commercially available solutions may be used in place
8.2.1 When Ag/AgCl reference electrode is used for the
of laboratory preparations provided the solutions have been
titration and it contains an electrolyte that is not 1M to 3M
certified as equivalent.
LiCl in ethanol, replace the electrolyte. Drain the electrolyte
7.10 Alternative volumes of solutions may be prepared
from the electrode, wash away all the salt (if present) with
provided the final solution concentration is equivalent.
water and then rinse with ethanol. Rinse several times with the
LiClelectrolytesolution.Finally,replacethesleeveandfillthe
7.11 Toluene—Reagent grade. (Warning—Extremely flam-
electrode with the LiCl electrolyte to the filling hole. When
mable.)
refitting the sleeve, ensure that there will be a free flow of
7.12 Titration Solvent—In a brown reagent bottle, add
electrolyte into the system. A combination electrode shall be
30mL of water to 1L of isopropyl alcohol, and mix thor-
prepared in the same manner.The electrolyte in a combination
oughly. Add 1L each of toluene and chloroform, and mix
electrode can be removed with the aid of a vacuum suction.
thoroughly.
8.2.2 Prior to each titration, soak the prepared electrodes in
7.13 Tris(hydroxymethyl)aminomethane ((HOCH ) CNH ),
water,pH4.5–5.5acidifiedwithHCl,foratleast5min.Rinse
2 3 2
primary standard, dried—Crusha1gto3gportionofprimary
the electrode with propan-2-ol immediately before use, and
standard Tris(hydroxymethyl)aminomethane to a fine powder
then with the titration solvent.
using an agate or other nonreactive mortar and pestle for 60s
8.3 Testing of Electrodes—See Appendix X2 for the proce-
to 90s. Take special care to crush any larger crystals. Dry the
dure to check electrode performance.
crushedmaterialatroomtemperature(22°Cto23°C)for24h
inavacuumdesiccatoroveranhydrousmagnesiumperchlorate
9. Standardization of Apparatus
or equivalent. Drying of this material at elevated temperatures
9.1 Preparetheelectrodesasdescribedin8.2,immersethem
is not recommended, owing to the possibility of decomposi-
in the pH 5 aqueous buffer solution, and stir for at least 2min,
tion.
maintaining the temperature of the buffer solution at a tem-
perature within 2°C of that at which the titrations are to be
8. Preparation of Electrode System
made.Readthecellvoltage.ThereadingsoobtainedinthepH
8.1 Maintenance and Storage of Electrodes—Cleaning the
5 acidic buffer solution is taken as the end point for the EBas.
electrode thoroughly, keeping the ground-glass joint free of
foreign materials, and regular testing of the electrodes are very
10. Preparation of Lubricant Samples
importantinobtainingrepeatablepotentials,sincethecontami-
10.1 When applicable, refer to Practice D4057 (Manual
nation may introduce uncertain erratic and unnoticeable liquid
Sampling)orPracticeD4177(AutomaticSampling)forproper
contact potentials.While this is of secondary importance when
sampling techniques.
end points are chosen from inflection points in the titration
10.1.1 When sampling used lubricants, the specimen shall
curve, it may be very serious when end points are chosen at
be representative of the system sampled and shall be free of
experimentally determined cell potentials as outlined in the
contamination from external sources.
procedure.
10.1.2 Agitate used oil samples thoroughly to ensure that
8.1.1 CleanthepHindicatingelectrodeorthepHindicating
any sediment present is homogeneously suspen
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D8126 − 17 D8126 − 21
Standard Test Method for
Efficient Basicity Determination by Potentiometric
Hydrochloric Acid Titration
This standard is issued under the fixed designation D8126; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method covers a procedure for the determination of the efficient basicity (EBas) in new marine cylinder lubricants
that lubricate the upper part, that is, piston-ring-cylinder area, of two-stroke marine engines.
1.2 This test method has been developed for marine cylinder lubricants, having base number (BN) (measured by Test Method
D2896) from 20 mg KOH/g to 100 mg KOH ⁄g, and an EBas from 10 mg KOH/g to 36 mg KOH/g.
1.3 In this test method, only the efficient basicity is determined, corresponding to the components or part of components that have
a major role in neutralization of the acidic species formed in the combustion of the fuel in the marine engine. It differentiates
between the fastest reacting species and the last one to react like overbasing compounds in detergent (classically mineral calcium
carbonate, CaCO ). The values obtained, however, are intended to be compared with the other values obtained by this test method
only; base numbers obtained by this test method are not intended to be equal to values by other test methods.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. Some specific hazards statements are given in Sections 6 and 7.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D2896 Test Method for Base Number of Petroleum Products by Potentiometric Perchloric Acid Titration
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
D4739 Test Method for Base Number Determination by Potentiometric Hydrochloric Acid Titration
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.06 on Analysis of Liquid Fuels and Lubricants.
Current edition approved July 1, 2017Nov. 1, 2021. Published September 2017November 2021. Originally approved in 2017. Last previous edition approved in 2017 as
D8126 – 17. DOI: 10.1520/D8126-17.10.1520/D8126-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8126 − 21
D6300 Practice for Determination of Precision and Bias Data for Use in Test Methods for Petroleum Products, Liquid Fuels, and
Lubricants
3. Terminology
3.1 Definitions:
3.1.1 base number, n—the quantity of a specified acid, expressed in terms of the equivalent number of milligrams of potassium
hydroxide per gram of sample, required to titrate a sample in a specified solvent to a specified endpoint using a specified detection
system.
3.1.2 effıcient basicity, EBas, n—the quantity of hydrochloric acid, expressed in terms of the equivalent number of milligrams of
potassium hydroxide per gram of sample, required to neutralize the basic components, which are organic species, of the tested
lubricant in a specified solvent to a specified buffer endpoint using a specified detection system.
4. Summary of Test Method
4.1 The sample is dissolved in a mixture of toluene, propan-2-ol (isopropyl alcohol), chloroform, and a small amount of water and
titrated potentiometrically with alcoholic hydrochloric acid solution. The test results of this procedure are obtained by titration
mode of fixed increment and fixed time additions of the titrant. An endpoint is selected from a titration curve according to the
criteria given in Section 12 and used to calculate the EBas.
5. Significance and Use
5.1 Lubricants can contain basic constituents that are present as additives. The relative amount of these materials can be
determined by titration with acids. The base number is a measurement of the amount of basic substances in the oil under the
conditions of the testing procedure.
5.2 A primary objective of a marine cylinder lubricant is the neutralization of sulfuric acid, produced during fuel combustion, to
protect the engine from corrosion. The EBas method characterizes the more efficient basic species of the lubricant reacting with
acids from the beginning of the neutralization (when the lubricant milieu is basic) until an equilibrium state where the lubricant
becomes slightly acidic. The use of hydrochloric acid (HCl) allows differentiating basicities of various strengths during titration.
5.3 In marine lubricants, the constituents that can be considered to have basic properties are primarily organic and inorganic bases
coming from the detergent. Basicity can also be brought to the lubricant by other components including dispersants, amino
compounds, or any organic basic components. This test method uses the same titration system as that of Test Method D4739,
however the evaluation and interpretation of the titration result is different. The hydrochloric acid is used as the titrant in this test
method and Test Method D4739, whereas Test Method D2896 uses a stronger acid, perchloric acid. While all three methods can
be considered as complementary to evaluate the basicity of lubricants, this test method is only used for marine cylinder lubricants.
5.4 This test method measures only the efficient basicity of the lubricant, which represents only the part of the total basicity, until
the point of which the lubricant becomes slightly acidic. It will depend on the components of the formulation itself. In that respect,
the EBas cannot be compared to base number given by Test Methods D4739 or D2896. The EBas is then complementary
information to the base number, giving performance in efficiency of neutralization of acidic species.
6. Apparatus
6.1 Potentiometric Titration, automatic or manual, with capability of adding fixed increments of titrant at fixed time intervals (see
Annex A1).
6.1.1 The titrimeter must automatically (or manually) control the rate of addition of titrant as follows: Delivery of titrant will be
incremental; after delivery of precisely a 0.100 mL increment (see 6.1.2), the delivery is stopped and a fixed time period of 90 s
is allowed to pass before another 0.100 mL increment of titrant is delivered. This procedure is repeated until the titration is
completed.
6.1.2 The precision of addition of the 0.100 mL increments of titrant must be 60.001 mL for automatic titrators. For manual buret,
it should be 60.005 mL. A higher incremental precision is required for an automatic buret, because the total volume to the end
point is summed from the individual increments; it is read from a scale with a manual buret.
D8126 − 21
6.2 Sensing Electrode, standard pH with glass membrane, suitable for non-aqueous titrations.
6.3 Reference Electrode, Silver/Silver Chloride (Ag/AgCl) reference electrode with sleeve junction, filled with 1 M to 3 M lithium
chloride (LiCl) in ethanol.
6.3.1 Combination Electrode—Sensing electrodes may have the silver/silver chloride (Ag/AgCl) reference electrode built into the
same electrode body, which offers the convenience of working with and maintaining only one electrode. The combination electrode
shall have a sleeve junction type of reference and shall use an inert ethanol electrolyte, for example, 1 M to 3 M LiCl in ethanol.
6.3.1.1 In the reference compartment, the sensing electrode part shall use a glass membrane designed for nonaqueous titrations.
These combination electrodes shall have the same response or better response than a dual electrode system. They shall have
removable sleeves for easy rinsing and addition of electrolyte. When a movable sleeve is part of the electrode system, ensure that
the sleeve is unimpaired before every titration.
6.4 Stirrer, Buret, Stand, Titration Vessel, as specified in Annex A1, are required.
7. Reagents
7.1 Purity of Reagents—Reagent-grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
7.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water that meets the
requirement of either Type I, II, or III of Specification D1193.
7.3 Buffer, Aqueous Acid—Commercial pH 5 buffer solution with a tolerance of 60.02 pH units at 25 °C. This solution shall be
replaced at regular intervals consistent with its stability or when contamination is suspected. Information related to the stability
should be obtained from the manufacturer.
7.4 Chloroform—Reagent grade. (Warning—Toxic and suspected carcinogen.)
7.5 Hydrochloric Acid Solution, Standard Alcoholic (0.1 M)—Mix 9 mL of reagent grade hydrochloric acid (HCl, sp gr 1.19)
(Warning—Toxic and corrosive), with 1 L of anhydrous isopropyl alcohol. Standardize frequently enough to detect normality
changes of 0.0005 by potentiometric titration. of approximately 0.1 g (weigh accurately at the nearest 0.0001 g) of primary
standard Tris(hydroxymethyl)aminomethane dissolved in 60 mL CO -free water.
7.6 Ethanol—reagent grade. (Warning—Flammable and toxic, especially when denatured.)
7.7 Lithium Chloride Electrolyte—Prepare a solution of 1 M to 3 M LiCl in ethanol.
7.8 Propan-2-ol (Isopropyl Alcohol)—Anhydrous, (less than 0.1 % H O). (Warning—Flammable.)
7.9 Commercially available solutions may be used in place of laboratory preparations provided the solutions have been certified
as equivalent.
7.10 Alternative volumes of solutions may be prepared provided the final solution concentration is equivalent.
7.11 Toluene—Reagent grade. (Warning—Extremely flammable.)
ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference Materials, American Chemical Society, Washington, DC. For
suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and
the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D8126 − 21
7.12 Titration Solvent—In a brown reagent bottle, add 30 mL of water to 1 L of isopropyl alcohol, and mix thoroughly. Add 1 L
each of toluene and chloroform, and mix thoroughly.
7.13 Tris(hydroxymethyl)aminomethane ((HOCH ) CNH ), primary standard, dried—Crush a 1 g to 3 g portion of primary
2 3 2
standard Tris(hydroxymethyl)aminomethane to a fine powder using an agate or other nonreactive mortar and pestle for 60 s to 90 s.
Take special care to crush any larger crystals. Dry the crushed material at room temperature (22 °C to 23 °C) for 24 h in a vacuum
desiccator over anhydrous magnesium perchlorate or equivalent. Drying of this material at elevated temperatures is not
recommended, owing to the possibility of decomposition.
8. Preparation of Electrode System
8.1 Maintenance and Storage of Electrodes—Cleaning the electrode thoroughly, keeping the ground-glass joint free of foreign
materials, and regular testing of the electrodes are very important in obtaining repeatable potentials, since the contamination may
introduce uncertain erratic and unnoticeable liquid contact potentials. While this is of secondary importance when end points are
chosen from inflection points in the titration curve, it may be very serious when end points are chosen at experimentally determined
cell potentials as outlined in the procedure.
8.1.1 Clean the pH indicating electrode or the pH indicating part of the combination electrode at frequent intervals based on use
and type of samples being analyzed by immersing in non-chromium containing, strongly oxidizing cleaning solution.
8.1.1.1 The electrode shall be cleaned periodically when in use or when a new electrode is installed. Drain the LiCl electrolyte
from the electrode at least once each week, and refill with fresh LiCl electrolyte as far as the filling hole. Ensure that there are no
air bubbles in the electrode liquid. If air bubbles are observed, hold the electrode in a vertical position and gently tap it to release
the bubbles. Maintain the electrolyte level in the electrode above that of the liquid in the titration beaker at all times.
8.1.2 When not in use, immerse the lower halves of the electrodes in either water (sensing) or the LiCl in isopropyl alcohol
electrolyte (reference). Do not allow them to remain immersed in titration solvent for any appreciable period of time between
titrations. While the electrodes are not extremely fragile, handle them carefully at all times.
8.1.3 Electrode Life—Typically, electrode usage is limited to 3 months to 6 months, depending upon usage. Electrodes have a
limited shelf life and shall be tested before use (8.3).
8.2 Preparation of Electrodes:
8.2.1 When Ag/AgCl reference electrode is used for the titration and it contains an electrolyte that is not 1 M to 3 M LiCl in
ethanol, replace the electrolyte. Drain the electrolyte from the electrode, wash away all the salt (if present) with water and then
rinse with ethanol. Rinse several times with the LiCl electrolyte solution. Finally, replace the sleeve and fill the electrode with the
LiCl electrolyte to the filling hole. When refitting the sleeve, ensure that there will be a free flow of electrolyte into the system.
A combination electrode shall be prepared in the same manner. The electrolyte in a combination electrode can be removed with
the aid of a vacuum suction.
8.2.2 Prior to each titration, soak the prepared electrodes in water, pH 4.5 – 5.5 acidified with HCl, for at least 5 min. Rinse the
electrode with propan-2-ol immediately before use, and then with the titration solvent.
8.3 Testing of Electrodes—See Appendix X2 for the procedure to check electrode performance.
9. Standardization of Apparatus
9.1 Prepare the electrodes as described in 8.2, immerse them in the pH 5 aqueous buffer solution, and stir for at least 2 min,
maintaining the temperature of the
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...