ASTM A262-15(2021)
(Practice)Standard Practices for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels
Standard Practices for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels
ABSTRACT
This specification covers the standard practices for detecting susceptibility to intergranular attack in austenitic stainless steels. These practices include five intergranular corrosion tests, namely: (1) oxalic acid etch test for classification of etch structures of austenitic stainless steels; (2) ferric sulfate-sulfuric acid test, (3) nitric acid test and (4) copper-copper sulfate-sulfuric acid test for detecting susceptibility to intergranular attack in austenitic stainless steels; and (5) copper-copper sulfate-50% sulfuric acid test for detecting susceptibility to intergranular attack in molybdenum-bearing cast austenitic stainless steels. Methods for preparing the test specimens, rapid screening tests, apparatus setup and testing procedures, and calculations and report contents are described for each testing practice. The etch structure types used to classify the specimens are: step structure, dual structure, ditch structure, isolated ferrite, interdendritic ditches, end-grain pitting I, and end-grain pitting II.
SIGNIFICANCE AND USE
6.1 Use of the etch test allows rapid acceptance of specific lots of material without the need to perform time-consuming and costly hot acid immersion tests on those lots.
SCOPE
1.1 These practices cover the following five tests:
1.1.1 Practice A—Oxalic Acid Etch Test for Classification of Etch Structures of Austenitic Stainless Steels (Sections 4 to 13, inclusive),
1.1.2 Practice B—Ferric Sulfate-Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 14 to 25, inclusive),
1.1.3 Practice C—Nitric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 26 to 36, inclusive),
1.1.4 Practice E—Copper–Copper Sulfate–Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 37 to 46, inclusive), and
1.1.5 Practice F—Copper–Copper Sulfate–50 % Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Molybdenum-Bearing Austenitic Stainless Steels (Sections 47 to 58, inclusive).
1.2 The Oxalic Acid Etch Test is a rapid method of identifying, by simple etching, those specimens of certain stainless steel grades that are essentially free of susceptibility to intergranular attack associated with chromium carbide precipitates. These specimens will have low corrosion rates in certain corrosion tests and therefore can be eliminated (screened) from testing as “acceptable.” The etch test is applicable only to those grades listed in the individual hot acid tests and classifies the specimens either as “acceptable” or as “suspect.”
1.3 The ferric sulfate-sulfuric acid test, the copper–copper sulfate–50 % sulfuric acid test, and the nitric acid test are based on weight loss determinations and, thus, provide a quantitative measure of the relative performance of specimens evaluated. In contrast, the copper–copper sulfate–16 % sulfuric acid test is based on visual examination of bend specimens and, therefore, classifies the specimens only as acceptable or nonacceptable.
1.4 The presence or absence of intergranular attack in these tests is not necessarily a measure of the performance of the material in other corrosive environments. These tests do not provide a basis for predicting resistance to forms of corrosion other than intergranular, such as general corrosion, pitting, or stress-corrosion cracking.
Note 1: See Appendix X1 for information regarding test selection.
1.5 The values stated in SI units are to be regarded as standard. The inch-pound equivalents are in parentheses and may be approximate.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitatio...
General Information
- Status
- Published
- Publication Date
- 31-Aug-2021
- Technical Committee
- A01 - Steel, Stainless Steel and Related Alloys
- Drafting Committee
- A01.14 - Methods of Corrosion Testing
Relations
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Jul-2019
- Refers
ASTM A370-17a - Standard Test Methods and Definitions for Mechanical Testing of Steel Products - Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Nov-2015
- Effective Date
- 15-May-2014
- Effective Date
- 15-Nov-2013
- Effective Date
- 15-Feb-2013
- Refers
ASTM A370-12a - Standard Test Methods and Definitions for Mechanical Testing of Steel Products - Effective Date
- 15-Oct-2012
- Effective Date
- 15-Mar-2012
- Refers
ASTM A370-11a - Standard Test Methods and Definitions for Mechanical Testing of Steel Products - Effective Date
- 15-Nov-2011
- Effective Date
- 15-Jun-2010
- Refers
ASTM A370-09a - Standard Test Methods and Definitions for Mechanical Testing of Steel Products - Effective Date
- 01-Jun-2009
- Refers
ASTM A370-09ae1 - Standard Test Methods and Definitions for Mechanical Testing of Steel Products - Effective Date
- 01-Jun-2009
- Effective Date
- 01-Jan-2009
Overview
ASTM A262-15(2021) is a comprehensive standard developed by ASTM International for detecting susceptibility to intergranular attack (IGA) in austenitic stainless steels. This standard provides detailed testing practices that help determine if austenitic stainless steels are at risk of intergranular corrosion-a form of localized corrosion that attacks the grain boundaries of a material, often due to the precipitation of chromium carbides or presence of certain phases. The document outlines five recognized intergranular corrosion test methods, ensuring that users can effectively assess the resilience of stainless steels in critical applications.
Key Topics
ASTM A262-15(2021) addresses several crucial topics:
- Five Corrosion Test Methods:
- Oxalic Acid Etch Test (A): Rapid classification of etch structures to screen out non-susceptible samples.
- Ferric Sulfate-Sulfuric Acid Test (B): Measures weight loss after boiling exposure, determining attack susceptibility.
- Nitric Acid Test (C): Quantifies weight loss from nitric acid boiling, used for specific stainless grades.
- Copper-Copper Sulfate-Sulfuric Acid Test (E): Relies on a visual pass/fail inspection of bend specimens.
- Copper-Copper Sulfate-50% Sulfuric Acid Test (F): Specifically for molybdenum-bearing cast austenitic stainless steels.
- Specimen Preparation and Sampling: Guidelines for preparing representative and process-affected metal samples.
- Rapid Screening: Procedures for quickly identifying "acceptable" vs "suspect" materials, reducing reliance on lengthy immersion tests.
- Reporting and Evaluation: Instructions on data calculation, acceptance criteria, and thorough documentation.
- Safety Practices: Notes on proper handling of hazardous reagents and use of laboratory safety precautions.
Applications
Practical Value of ASTM A262-15(2021):
- Quality Control: Enables manufacturers and suppliers to certify the resistance of stainless steel products to intergranular attack.
- Material Certification: Ensures compliance with procurement specifications in industries such as chemical processing, food handling, and piping systems.
- Weld Testing: Assesses weld-affected zones for localized corrosion susceptibility, critical for pressure vessels and piping systems.
- Failure Analysis: Provides standardized methods for diagnosing intergranular corrosion-related failures in service.
- Screening for Process-Affected Metal: Identifies metals altered by welding, carburizing, or heat treatment so that proper testing and classification can be applied.
These standardized practices are relied upon by quality assurance teams, material engineers, and testing laboratories to maintain high performance and safety standards in stainless steel components.
Related Standards
Key Standards Referenced in ASTM A262-15(2021):
- ASTM A370: Test Methods and Definitions for Mechanical Testing of Steel Products
- ASTM A380/A380M: Practices for Cleaning, Descaling, and Passivation of Stainless Steel Parts
- ASTM D1193: Specification for Reagent Water
- ASTM E3: Guide for Preparation of Metallographic Specimens
- ISO 3651-2: Determination of resistance to intergranular corrosion of stainless steels
- ASME Boiler and Pressure Vessel Code, Section IX: Welding and brazing qualifications
These documents complement ASTM A262 by providing additional requirements for specimen preparation, cleaning, and testing procedures in various industrial contexts.
By following ASTM A262-15(2021), industries ensure that austenitic stainless steels used in critical environments are thoroughly evaluated for intergranular corrosion, thus enhancing material reliability and safety. This standard remains a key resource for quality control, regulatory compliance, and material selection in corrosive service conditions.
Buy Documents
ASTM A262-15(2021) - Standard Practices for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM A262-15(2021) is a standard published by ASTM International. Its full title is "Standard Practices for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels". This standard covers: ABSTRACT This specification covers the standard practices for detecting susceptibility to intergranular attack in austenitic stainless steels. These practices include five intergranular corrosion tests, namely: (1) oxalic acid etch test for classification of etch structures of austenitic stainless steels; (2) ferric sulfate-sulfuric acid test, (3) nitric acid test and (4) copper-copper sulfate-sulfuric acid test for detecting susceptibility to intergranular attack in austenitic stainless steels; and (5) copper-copper sulfate-50% sulfuric acid test for detecting susceptibility to intergranular attack in molybdenum-bearing cast austenitic stainless steels. Methods for preparing the test specimens, rapid screening tests, apparatus setup and testing procedures, and calculations and report contents are described for each testing practice. The etch structure types used to classify the specimens are: step structure, dual structure, ditch structure, isolated ferrite, interdendritic ditches, end-grain pitting I, and end-grain pitting II. SIGNIFICANCE AND USE 6.1 Use of the etch test allows rapid acceptance of specific lots of material without the need to perform time-consuming and costly hot acid immersion tests on those lots. SCOPE 1.1 These practices cover the following five tests: 1.1.1 Practice A—Oxalic Acid Etch Test for Classification of Etch Structures of Austenitic Stainless Steels (Sections 4 to 13, inclusive), 1.1.2 Practice B—Ferric Sulfate-Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 14 to 25, inclusive), 1.1.3 Practice C—Nitric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 26 to 36, inclusive), 1.1.4 Practice E—Copper–Copper Sulfate–Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 37 to 46, inclusive), and 1.1.5 Practice F—Copper–Copper Sulfate–50 % Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Molybdenum-Bearing Austenitic Stainless Steels (Sections 47 to 58, inclusive). 1.2 The Oxalic Acid Etch Test is a rapid method of identifying, by simple etching, those specimens of certain stainless steel grades that are essentially free of susceptibility to intergranular attack associated with chromium carbide precipitates. These specimens will have low corrosion rates in certain corrosion tests and therefore can be eliminated (screened) from testing as “acceptable.” The etch test is applicable only to those grades listed in the individual hot acid tests and classifies the specimens either as “acceptable” or as “suspect.” 1.3 The ferric sulfate-sulfuric acid test, the copper–copper sulfate–50 % sulfuric acid test, and the nitric acid test are based on weight loss determinations and, thus, provide a quantitative measure of the relative performance of specimens evaluated. In contrast, the copper–copper sulfate–16 % sulfuric acid test is based on visual examination of bend specimens and, therefore, classifies the specimens only as acceptable or nonacceptable. 1.4 The presence or absence of intergranular attack in these tests is not necessarily a measure of the performance of the material in other corrosive environments. These tests do not provide a basis for predicting resistance to forms of corrosion other than intergranular, such as general corrosion, pitting, or stress-corrosion cracking. Note 1: See Appendix X1 for information regarding test selection. 1.5 The values stated in SI units are to be regarded as standard. The inch-pound equivalents are in parentheses and may be approximate. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitatio...
ABSTRACT This specification covers the standard practices for detecting susceptibility to intergranular attack in austenitic stainless steels. These practices include five intergranular corrosion tests, namely: (1) oxalic acid etch test for classification of etch structures of austenitic stainless steels; (2) ferric sulfate-sulfuric acid test, (3) nitric acid test and (4) copper-copper sulfate-sulfuric acid test for detecting susceptibility to intergranular attack in austenitic stainless steels; and (5) copper-copper sulfate-50% sulfuric acid test for detecting susceptibility to intergranular attack in molybdenum-bearing cast austenitic stainless steels. Methods for preparing the test specimens, rapid screening tests, apparatus setup and testing procedures, and calculations and report contents are described for each testing practice. The etch structure types used to classify the specimens are: step structure, dual structure, ditch structure, isolated ferrite, interdendritic ditches, end-grain pitting I, and end-grain pitting II. SIGNIFICANCE AND USE 6.1 Use of the etch test allows rapid acceptance of specific lots of material without the need to perform time-consuming and costly hot acid immersion tests on those lots. SCOPE 1.1 These practices cover the following five tests: 1.1.1 Practice A—Oxalic Acid Etch Test for Classification of Etch Structures of Austenitic Stainless Steels (Sections 4 to 13, inclusive), 1.1.2 Practice B—Ferric Sulfate-Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 14 to 25, inclusive), 1.1.3 Practice C—Nitric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 26 to 36, inclusive), 1.1.4 Practice E—Copper–Copper Sulfate–Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels (Sections 37 to 46, inclusive), and 1.1.5 Practice F—Copper–Copper Sulfate–50 % Sulfuric Acid Test for Detecting Susceptibility to Intergranular Attack in Molybdenum-Bearing Austenitic Stainless Steels (Sections 47 to 58, inclusive). 1.2 The Oxalic Acid Etch Test is a rapid method of identifying, by simple etching, those specimens of certain stainless steel grades that are essentially free of susceptibility to intergranular attack associated with chromium carbide precipitates. These specimens will have low corrosion rates in certain corrosion tests and therefore can be eliminated (screened) from testing as “acceptable.” The etch test is applicable only to those grades listed in the individual hot acid tests and classifies the specimens either as “acceptable” or as “suspect.” 1.3 The ferric sulfate-sulfuric acid test, the copper–copper sulfate–50 % sulfuric acid test, and the nitric acid test are based on weight loss determinations and, thus, provide a quantitative measure of the relative performance of specimens evaluated. In contrast, the copper–copper sulfate–16 % sulfuric acid test is based on visual examination of bend specimens and, therefore, classifies the specimens only as acceptable or nonacceptable. 1.4 The presence or absence of intergranular attack in these tests is not necessarily a measure of the performance of the material in other corrosive environments. These tests do not provide a basis for predicting resistance to forms of corrosion other than intergranular, such as general corrosion, pitting, or stress-corrosion cracking. Note 1: See Appendix X1 for information regarding test selection. 1.5 The values stated in SI units are to be regarded as standard. The inch-pound equivalents are in parentheses and may be approximate. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitatio...
ASTM A262-15(2021) is classified under the following ICS (International Classification for Standards) categories: 77.140.20 - Stainless steels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM A262-15(2021) has the following relationships with other standards: It is inter standard links to ASTM A370-24, ASTM A370-19, ASTM A370-17a, ASTM A370-17, ASTM A370-15, ASTM A370-14, ASTM A370-13, ASTM A380/A380M-13, ASTM A370-12a, ASTM A370-12, ASTM A370-11a, ASTM A370-10, ASTM A370-09a, ASTM A370-09ae1, ASTM A370-09. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM A262-15(2021) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: A262 − 15 (Reapproved 2021)
Standard Practices for
Detecting Susceptibility to Intergranular Attack in Austenitic
Stainless Steels
This standard is issued under the fixed designation A262; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope based on visual examination of bend specimens and, therefore,
classifies the specimens only as acceptable or nonacceptable.
1.1 These practices cover the following five tests:
1.4 The presence or absence of intergranular attack in these
1.1.1 Practice A—Oxalic Acid Etch Test for Classification
of Etch Structures ofAustenitic Stainless Steels (Sections 4 to tests is not necessarily a measure of the performance of the
material in other corrosive environments. These tests do not
13, inclusive),
provide a basis for predicting resistance to forms of corrosion
1.1.2 Practice B—Ferric Sulfate-Sulfuric Acid Test for De-
other than intergranular, such as general corrosion, pitting, or
tecting Susceptibility to Intergranular Attack in Austenitic
Stainless Steels (Sections 14 to 25, inclusive), stress-corrosion cracking.
1.1.3 Practice C—Nitric Acid Test for Detecting Suscepti-
NOTE 1—See Appendix X1 for information regarding test selection.
bility to Intergranular Attack in Austenitic Stainless Steels
1.5 The values stated in SI units are to be regarded as
(Sections 26 to 36, inclusive),
standard. The inch-pound equivalents are in parentheses and
1.1.4 Practice E—Copper–Copper Sulfate–Sulfuric Acid
may be approximate.
Test for Detecting Susceptibility to Intergranular Attack in
1.6 This standard does not purport to address all of the
Austenitic Stainless Steels (Sections 37 to 46, inclusive), and
safety concerns, if any, associated with its use. It is the
1.1.5 Practice F—Copper–Copper Sulfate–50% Sulfuric
responsibility of the user of this standard to establish appro-
Acid Test for Detecting Susceptibility to Intergranular Attack
priate safety, health, and environmental practices and deter-
in Molybdenum-Bearing Austenitic Stainless Steels (Sections
mine the applicability of regulatory limitations prior to use.
47 to 58, inclusive).
Some specific hazards statements are given in 10.1, 20.1.1,
1.2 The Oxalic Acid Etch Test is a rapid method of
20.1.9, 31.3, 34.4, 53.1.1, and 53.1.10.
identifying, by simple etching, those specimens of certain
1.7 This international standard was developed in accor-
stainless steel grades that are essentially free of susceptibility
dance with internationally recognized principles on standard-
to intergranular attack associated with chromium carbide
ization established in the Decision on Principles for the
precipitates. These specimens will have low corrosion rates in
Development of International Standards, Guides and Recom-
certain corrosion tests and therefore can be eliminated
mendations issued by the World Trade Organization Technical
(screened) from testing as “acceptable.” The etch test is
Barriers to Trade (TBT) Committee.
applicable only to those grades listed in the individual hot acid
tests and classifies the specimens either as “acceptable” or as
2. Referenced Documents
“suspect.”
2.1 ASTM Standards:
1.3 The ferric sulfate-sulfuric acid test, the copper–copper
A370Test Methods and Definitions for Mechanical Testing
sulfate–50%sulfuricacidtest,andthenitricacidtestarebased
of Steel Products
on weight loss determinations and, thus, provide a quantitative
A380/A380MPractice for Cleaning, Descaling, and Passi-
measureoftherelativeperformanceofspecimensevaluated.In
vation of Stainless Steel Parts, Equipment, and Systems
contrast, the copper–copper sulfate–16% sulfuric acid test is
D1193Specification for Reagent Water
E3Guide for Preparation of Metallographic Specimens
These practices are under the jurisdiction of ASTM Committee A01 on Steel,
StainlessSteelandRelatedAlloysandarethedirectresponsibilityofSubcommittee
A01.14 on Methods of Corrosion Testing. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Sept. 1, 2021. Published September 2021. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1943. Last previous edition approved in 2015 as A262–15. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/A0262-15R21. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
A262 − 15 (2021)
2.2 ASME Code: E—Copper-CopperSulfate–16%SulfuricAcidTest,andPrac-
BPVCASME Boiler and Pressure Vessel Code, Section IX tice F—Copper-Copper Sulfate–50% Sulfuric Acid Test.
2.3 ACS Specifications:
4.4 Each of these other practices contains a table showing
ACS Reagent ChemicalsSpecifications and Procedures
which classifications of etch structures on a given stainless
2.4 ISO Standard:
steelgradeareequivalenttoacceptableorsuspectperformance
ISO 3651-2Determination of resistance to intergranular
in that particular test. Specimens having acceptable etch
corrosion of stainless steels — Part 2: Ferritic, austenitic
structuresneednotbesubjectedtothehotacidtest.Specimens
and ferritic-austenitic (duplex) stainless steels — Corro-
having suspect etch structures must be tested in the specified
sion test in media containing sulfuric acid
hot acid solution.
4.5 There are two classes of specimens to be considered:
3. Purity of Reagents
base metal, and process-affected metal.
3.1 Purity of Reagents—Reagent grade chemicals shall be
4.5.1 Process-affected metal contains any condition that
used in all tests. Unless otherwise indicated, it is intended that
affectsthecorrosionpropertiesofthematerialinanon-uniform
all reagents conform to the specifications of the Committee on
way, such as (but not limited to) welds; carburized. nitrided, or
AnalyticalReagentsoftheAmericanChemicalSociety where
oxidized surfaces; mechanical deformation; and areas affected
such specifications are available. Other grades may be used,
by heat. Base metal has none of these conditions.
provided it is first ascertained that the reagent is of sufficiently
4.5.2 Because Practices B, C, and F involve immersing the
high purity to permit its use without lessening the accuracy of
entire specimen and averaging the mass loss over the total
the test result.
specimenarea,andbecausewelding,carburization,mechanical
3.2 Purity of Water—Unless otherwise indicated, references
deformation, and the like affect only part of a specimen, the
to water shall be understood to mean reagent water as defined
presence of process-affected metal in a specimen can affect the
by Type IV of Specification D1193.
test result in an unpredictable way depending on the propor-
tions of the area affected.
PRACTICE A—OXALIC ACID ETCH TEST FOR
4.5.3 Ifthepresenceoftheseorotherlocalizedconditionsis
CLASSIFICATION OF ETCH STRUCTURES OF
a concern to the purchaser, then tests that do not average the
AUSTENITIC STAINLESS STEELS (1)
masslossoverthetotalspecimensurfacearea,suchasPractice
A,theOxalicAcidEtchTest,orPracticeE,theCopper–Copper
4. Scope
Sulfate–Sulfuric Acid Test for Detecting Susceptibility to
4.1 The Oxalic Acid Etch Test is used for acceptance of
Intergranular Attack in Austenitic Stainless Steels, should be
wrought or cast austenitic stainless steel material but not for
considered.
rejection of material. Use of A262 PracticeAas a stand-alone
test may reject material that the applicable hot acid test would 5. Summary of Practice
find acceptable; such use is outside the scope of this practice.
5.1 A specimen representative of the material to be evalu-
ated is polished to a specified finish and over-etched using
4.2 This test is intended to be used in connection with other
evaluation tests described in these practices to provide a rapid oxalic acid electrolytically. The etched specimen is then
examined using a metallurgical microscope. The etched struc-
method for identifying qualitatively those specimens that are
certain to be free of susceptibility to rapid intergranular attack ture is compared with reference photographs to determine
whether the material is acceptable or suspect. Suspect material
intheseothertests.Suchspecimenshavelowcorrosionratesin
the various hot acid tests which require from 15 to 240 h of is then subjected to the specified hot acid immersion test.
exposure. These specimens are identified by means of their
6. Significance and Use
etch structures, which are classified according to the criteria
given in Section 11.
6.1 Use of the etch test allows rapid acceptance of specific
lots of material without the need to perform time-consuming
4.3 The Oxalic Acid Etch Test may be used to screen
and costly hot acid immersion tests on those lots.
specimens intended for testing in Practice B—Ferric Sulfate-
Sulfuric Acid Test, Practice C—Nitric Acid Test, Practice
7. Apparatus
7.1 Etching Cell:
Available from American Society of Mechanical Engineers (ASME), ASME
7.1.1 An etching cell may be assembled using components
International Headquarters, Two Park Ave., New York, NY 10016-5990, http://
as described in this section. Alternatively, a commercial
www.asme.org.
4 electropolisher/etcher (as used for metallographic sample
Available from American Chemical Society (ACS), 1155 Sixteenth St., NW,
Washington, DC 20036, http://www.acs.org.
preparation) may be used for small specimens provided the
Available from International Organization for Standardization (ISO), ISO
current density requirement of 10.2 is met.
Central Secretariat, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
7.1.2 Source of Direct Current—Battery, generator, or rec-
Switzerland, https://www.iso.org.
tifier capable of supplying about 15 V and 20 A.
For suggestions on the testing of reagents not listed by theAmerican Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
7.1.3 Ammeter—For direct current; used to measure the
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
current on the specimen to be etched.
copeial Convention, Inc. (USPC), Rockville, MD.
7.1.4 Variable Resistance—Used to control the current on
The boldface numbers in parentheses refer to a list of references at the end of
this standard. the specimen to be etched.
A262 − 15 (2021)
7.1.5 Cathode—A stainless steel container, for example, a 10. Procedure
1L (1-qt) stainless steel beaker.
10.1 (Warning—Etching should be carried out under a
7.1.5.1 Alternate Cathode—Apiece of flat stainless steel at
ventilatedhood.Gas,whichisrapidlyevolvedattheelectrodes
least as large as the specimen surface.
with some entrainment of oxalic acid, is poisonous and
7.1.6 Electrical Clamp—To hold the specimen to be etched
irritating to mucous membranes.)
andtocompletetheelectricalcircuitbetweenthespecimenand
10.2 Etch the polished specimen at 1 A/cm for 1.5 min.
the power source such that the specimen is the anode of the
10.2.1 To obtain the correct specified current density:
cell.
10.2.1.1 Measurethetotalimmersedareaofthespecimento
7.1.7 Thepowersource,resistor,andammetermustbesized
be etched in square centimetres.
appropriately for providing and controlling the current as
10.2.1.2 Adjust the variable resistance until the ammeter
specified in 10.2 of this practice.
reading in amperes is equal to the total immersed area of the
7.1.8 As described, the electrolyte container is the cathode;
specimen in square centimetres.
it may be a stainless steel beaker or fabricated from stainless
steel such as by welding a section of tube or pipe to a flat plate 10.3 A yellow-green film is gradually formed on the cath-
ode.Thisincreasestheresistanceoftheetchingcell.Whenthis
or sheet. Alternatively, the electrolyte container may be glass
(or other non-conducting, corrosion resisting material) in lieu occurs, remove the film by rinsing the inside of the stainless
steelbeaker(orthesteelusedasthecathode)withanacidsuch
ofastainlesssteelcontainer,andthecathodemaybeaflatplate
orsheetofacorrosionresistingalloy.Inthislattercase,theflat as 30% HNO .
surface of the cathode must be at least as large as, facing, and
10.4 The temperature of the etching solution gradually
approximately centered on, the prepared surface of the speci-
increases during etching. Keep the temperature below 50°C.
men. Other configurations of the electrodes might not provide
This may be done by alternating two containers. One may be
uniform etching over the specimen surface. In any case, the
cooled in tap water while the other is used for etching.
sizeandshapeofthespecimendictatethesizeandconstruction
10.4.1 The rate of heating depends on the total current
of the etching cell and of the power source and controls. The
(ammeter reading) passing through the cell. Therefore, keep
overriding principle is that the etch needs to be uniform over
theareatobeetchedassmallaspossiblewhileatthesametime
the surface to be examined.
meeting the requirements of desirable minimum area to be
7.2 Metallurgical Microscope—For examination of etched etched.
microstructures at 250 to 500 diameters.
10.5 Avoid immersing the clamp holding the specimen in
the etching solution.
8. Reagents and Materials
10.6 Rinsing—Following etching, rinse the specimen thor-
8.1 Etching Solution (10 %)—Dissolve 100 g of reagent
oughly in hot water and then in acetone or alcohol to avoid
grade oxalic acid crystals (H C O ·2H O) in 900 mL of
2 2 4 2
crystallization of oxalic acid on the etched surface during
reagent water. Stir until all crystals are dissolved.
drying.
8.1.1 Alternate Etching Solution(See10.7)—Dissolve100g
10.7 It may be difficult to reveal the presence of step
of reagent grade ammonium persulfate ((NH ) S O)in
4 2 2 8
structures on some specimens containing molybdenum (AISI
900mL of reagent water. Stir until dissolved.
316, 316L, 317, 317L), which are free of chromium carbide
sensitization, by electrolytic etching with oxalic acid. In such
9. Sampling and Test Specimens
cases, an alternate electrolyte of ammonium persulfate may be
9.1 The specified hot acid test provides instructions for
used in place of oxalic acid. (See 8.1.1.) An etch for 5 or 10
sampling and for specimen preparation such as a sensitization min at 1 A/cm in a solution at room temperature readily
heat treatment. Additional instructions specific to Practice A
develops step structures on such specimens.
follow:
11. Classification of Etch Structures
9.2 The preferred specimen is a cross-section including the
productsurfacetobeexposedinservice.Onlysuchfinishingof
11.1 Examine the etched surface on a metallurgical micro-
the product surface should be performed as is required to scopeat250×to500×forwroughtsteelsandatabout250×for
remove foreign material.
cast steels.
11.2 Examine the etched cross-sectional areas thoroughly
9.3 Whenever practical, use a cross-sectional area of 1 cm
or more. If any cross-sectional dimension is less than 1 cm, by complete traverse from inside to outside diameters of rods
then the other dimension of the cross-section should be a and tubes, from face to face on plates.
minimum of 1 cm. When both dimensions of the product are
11.2.1 Microscopical examination of a specimen shall be
less than 1 cm, use a full cross section.
made on metal unaffected by cold-working, carburization,
welding,andthelike.Ifanyoftheseconditionsarefound,note
9.4 Polishing—On all types of materials, polish cross sec-
their presence in the report.
tionalsurfacesthroughCAMI/ANSI600[FEPA/ISOP1200]in
accordance with Guide E3 prior to etching and examination. 11.3 Classify the etch structures into the following types
Not all scratches need to be removed. (Note 2):
A262 − 15 (2021)
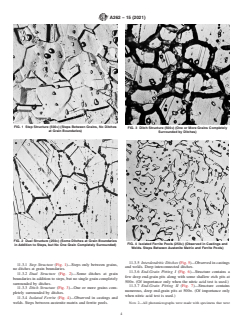
FIG. 1 Step Structure (500×) (Steps Between Grains, No Ditches
FIG. 3 Ditch Structure (500×) (One or More Grains Completely
at Grain Boundaries)
Surrounded by Ditches)
FIG. 2 Dual Structure (250×) (Some Ditches at Grain Boundaries
FIG. 4 Isolated Ferrite Pools (250×) (Observed in Castings and
in Addition to Steps, but No One Grain Completely Surrounded)
Welds. Steps Between Austenite Matrix and Ferrite Pools)
11.3.5 Interdendritic Ditches(Fig.5)—Observedincastings
11.3.1 Step Structure (Fig. 1)—Steps only between grains,
and welds. Deep interconnected ditches.
no ditches at grain boundaries.
11.3.6 End-Grain Pitting I (Fig. 6)—Structure contains a
11.3.2 Dual Structure (Fig. 2)—Some ditches at grain
few deep end-grain pits along with some shallow etch pits at
boundaries in addition to steps, but no single grain completely
500×. (Of importance only when the nitric acid test is used.)
surrounded by ditches.
11.3.7 End-Grain Pitting II (Fig. 7)—Structure contains
11.3.3 Ditch Structure (Fig. 3)—One or more grains com-
numerous, deep end-grain pits at 500×. (Of importance only
pletely surrounded by ditches.
when nitric acid test is used.)
11.3.4 Isolated Ferrite (Fig. 4)—Observed in castings and
welds. Steps between austenite matrix and ferrite pools. NOTE 2—All photomicrographs were made with specimens that were
A262 − 15 (2021)
FIG. 5 Interdendritic Ditches (250×) (Observed in Castings and
NOTE1—Thisoragreaterconcentrationofendgrainpitsat500×(using
Welds. Deep Interconnected Ditches)
standard etching conditions) indicates that the specimen must be tested
when screening is for nitric acid test.
FIG. 7 End Grain Pitting II (500×)
required to determine if there are any grains completely
encircled. If an encircled grain is found, classify the steel as a
ditch structure.
11.4.1 On stainless steel castings (also on weld metal), the
stepsbetweengrainsformedbyelectrolyticoxalicacidetching
tend to be less prominent than those on wrought materials or
areentirelyabsent.However,anysusceptibilitytointergranular
attack is readily detected by pronounced ditches.
11.4.2 Some wrought specimens, especially from bar stock,
maycontainarandompatternofpits.Ifthesepitsaresharpand
so deep that they appear black (Fig. 7) it is possible that the
specimen may be susceptible to end grain attack in nitric acid
only.Therefore,eventhoughthegrainboundariesallhavestep
structures,specimenshavingasmuchormoreendgrainpitting
thanthatshowninFig.7cannotbesafelyassumedtohavelow
nitric acid rates and should be subjected to the nitric acid test
whenever it is specified. Such sharp, deep pits should not be
confused with the shallow pits shown in Figs. 1 and 6.
NOTE 1—To differentiate between the types of pits, use a magnification
of 500× and focus in the plane of etched surface. The pits which now
appear completely black are end grain pits. 12. Use of Etch Structure Classifications
FIG. 6 End Grain Pitting I (500×) (A Few Deep End Grain Pits
12.1 Theuseoftheseclassificationsdependsonthehotacid
(See 1 in Figure) and Shallow Etch Pits (2))
corrosion test for which stainless steel specimens are being
screened by etching in oxalic acid and is described in each of
the practices.
etched under standard conditions: 10% oxalic acid, room temperature,
13. Precision and Bias
1.5min at 1 A/cm .
11.4 The evaluation of etch structures containing only steps 13.1 Precision and Bias—Noinformationispresentedabout
and of those showing grains completely surrounded by ditches either the precision or bias of Practice A—Oxalic Acid Etch
ineveryfieldcanbecarriedoutrelativelyrapidly.Incasesthat TestforclassificationofEtchStructuresofAusteniticStainless
appear to be dual structures, more extensive examination is Steels since the test result is nonquantitative.
A262 − 15 (2021)
TABLE 1 Use of Etch Structure Classifications from the Oxalic
PRACTICE B—FERRIC SULFATE–SULFURIC
A
Acid Etch Test with Ferric Sulfate-Sulfuric Acid Test
ACID TEST FOR DETECTING SUSCEPTIBILITY
Grade Acceptable Etch Suspect
TO INTERGRANULAR ATTACK IN
B
Structures Etch Structures
AUSTENITIC STAINLESS STEELS (2)
304 Step, dual, end grain, I & II Ditch
304L Step, dual, end grain, I & II Ditch
14. Scope
316 Step, dual, end grain, I & II Ditch
316L Step, dual, end grain, I & II Ditch
14.1 This practice describes the procedure for conducting
317 Step, dual, end grain, I & II Ditch
the boiling 120-h ferric sulfate–50% sulfuric acid test which 317L Step, dual, end grain, I & II Ditch
CF-3 Step, dual, isolated ferrite pools Ditch, interdendritic ditches
measures the susceptibility of austenitic stainless steels to
CF-8 Step, dual, isolated ferrite pools Ditch, interdendritic ditches
intergranular attack.
CF-3M Step, dual, isolated ferrite pools Ditch, interdendritic ditches
CF-8M Step, dual, isolated ferrite pools Ditch, interdendritic ditches
14.2 The presence or absence of intergranular attack in this
A
Grades not listed in this table either have not been evaluated for use of Practice
test is not necessarily a measure of the performance of the
A with Practice B or have been found to give acceptable results in the etch test
material in other corrosive environments. The test does not
while giving unacceptable results in Practice B. In the latter case Practices A would
pass material that should have been subjected to the ferric sulfate-sulfuric acid
provide a basis for predicting resistance to forms of corrosion
test.
other than intergranular, such as general corrosion, pitting, or
B
Specimens having these structures shall be tested in the ferric sulfate-sulfuric
stress-corrosion cracking.
acid test.
15. Summary of the Ferric Sulfate-Sulfuric Acid Practice
B
intergranular attack in the ferric sulfate-sulfuric acid test. Such
15.1 Aspecimen representative of the material to be evalu-
specimens are acceptable without testing in the ferric sulfate-
ated is immersed in a boiling solution of ferric sulfate and
sulfuric acid test.All specimens having suspect etch structures
sulfuric acid for a specified time. The resulting mass loss is
shall be tested in the ferric sulfate-sulfuric acid test.
converted to a corrosion rate, which is compared to a specified
maximum value to determine whether the material has the 18. Apparatus
resistance to attack expected of the grade of material being
18.1 The apparatus is illustrated in Fig. 8.
tested.
NOTE 4—Other ground glass joints, such as the 45/40 joint may also be
used.
16. Significance and Use
16.1 The ferric sulfate-sulfuric acid test detects susceptibil-
ity to intergranular attack associated primarily with chromium
carbide precipitate in unstabilized austenitic stainless steels,
and to intergranular attack associated with sigma phase.
16.2 The corrosion potential of the ferric sulfate-sulfuric
acid test has been reported as 0.6 V versus a standard calomel
electrode(SCE),ascomparedwith0.75to1.0VforPracticeC,
and 0.1 V for Practices E and F (3).
NOTE3—Ahighercorrosionpotentialindicatesmoreseverelyoxidizing
conditions.
17. Rapid Screening Test
17.1 Before testing in the ferric sulfate-sulfuric acid test,
specimens of certain grades of stainless steels (see Table 1)
may be given a rapid screening test in accordance with
procedures given in Practice A, Oxalic Acid Etch Test for
Classification of Etch Structures ofAustenitic Stainless Steels.
Preparation, etching, and the classification of etch structures
are described therein. The use of etch structure evaluations in
connection with the ferric sulfate-sulfuric acid test is specified
in Table 1.
17.2 Heat treat the material in accordance with22.1 prior to
performing the etch test.
17.3 Ignore “process-affected” areas (see Section 21); ap-
plication of the ferric sulfate-sulfuric acid test to process-
affected areas is currently outside the scope of Practice B.
17.4 Corrosion test specimens having acceptable etch struc-
tures in the Oxalic Acid Etch Test will be essentially free of FIG. 8 Apparatus for Ferric Sulfate-Sulfuric Acid Test
A262 − 15 (2021)
18.1.1 An Allihn condenser with a minimum of four bulbs 20.1.7 Cover flask with condenser and circulate cooling
and with a ground glass joint to match that of the flask. water.
18.1.1.1 Substitutions for this condenser or flask are not 20.1.8 Boil the solution until all ferric sulfate is dissolved
allowed. Specifically, the cold-finger type of condenser with (see Note 7).
standard Erlenmeyer flasks shall not be used. Corrosion rates 20.1.9 (Warning—It has been reported that violent boiling
obtainedusingthecold-fingertypeofcondenserarelowerthan resulting in acid spills can occur. It is important to ensure that
those obtained using theAllihn type of condenser whether due theconcentrationofaciddoesnotincreaseandthatanadequate
to loss of vapor or to higher oxygen content in the solution or number of boiling chips (which are resistant to attack by the
both.Suchlowercorrosionratesleadtoacceptanceofmaterial test solution) are present.)
that should be rejected.
18.1.2 A 1 L Erlenmeyer flask with a ground glass joint to 21. Sampling
match that of the condenser. The flask opening limits the size
21.1 Obtain and prepare only base metal samples.
of the specimen; a larger opening is desirable.
21.1.1 There are two classes of specimens to be considered:
18.1.3 Glass Cradle (Note 5)—Can be supplied by a glass-
base metal, and process-affected metal. Process-affected metal
blowing shop. It must be sized so as to fit, with the specimen,
contains any condition that affects the corrosion properties of
through the flask opening. It must be designed to allow free
the material in a non-uniform way, such as (but not limited to)
flow of the testing solution around the specimen.
welds; carburized. nitrided, or oxidized surfaces; mechanical
deformation; and areas affected by heat. Base metal has none
NOTE 5—Other equivalent means of specimen support, such as glass
hooks or stirrups, may also be used. of these conditions.
21.1.2 The Practice B test involves immersing the entire
18.1.4 Boiling Chips—Used to prevent bumping.
specimenandaveragingthemasslossovertheentiresurfaceof
18.1.5 High Vacuum Silicone Grease—Forthegroundglass
thespecimen.Welding,carburization,mechanicaldeformation,
joint.
and the like, affect only part of a specimen.
18.1.6 Hot plate, capable of providing heat for continuous
21.1.3 The mass loss rate from process-affected metal is
boiling of the solution.
expected to differ from that from base metal; the presence of
18.1.7 An analytical balance capable of weighing to the
process-affected metal in a specimen will affect the calculated
nearest 0.001 g.
test result in an unpredictable way.
NOTE 6—During testing, there is some deposition of iron oxides on the
21.1.4 If the presence of these or other localized conditions
upper part of the Erlenmeyer flask.This can be readily removed, after test
is a concern to the purchaser, then tests that do not average the
completion, by boiling a solution of 10 % hydrochloric acid in the flask.
masslossoverthetotalspecimensurfacearea,suchasPractice
18.1.8 Desiccator—Forstorageofpreparedspecimensprior
A,theOxalicAcidEtchTest,orPracticeE,theCopper–Copper
to testing.
Sulfate–16% SulfuricAcid Test for Detecting Susceptibility to
Intergranular Attack in Austenitic Stainless Steels, should be
19. Reagents and Materials
considered. Details of the test and acceptance criteria shall be
19.1 Ferric Sulfate Hydrate (Fe (SO ) ·xH O), about 75% as agreed by the purchaser and producer.
2 4 3 2
(Fe (SO ) ) by mass.
2 4 3
21.2 Unless otherwise specified by the purchaser, the pro-
19.1.1 Ferric sulfate is a specific additive that establishes
cedures for obtaining representative base metal samples, for
and controls the corrosion potential. Substitutions are not
removing the specimens from the samples, and the number of
permitted.
specimens shall be at the discretion of the producer.
19.2 Sulfuric Acid (H (SO) ), 95.0 to 98.0 % by mass.
2 4
22. Preparation of Test Specimens
20. Ferric Sulfate-Sulfuric Acid Test Solution
22.1 Heat treat extra-low carbon and stabilized grades at
650 to 675°C (1200 to 1250°F), which is the range of
20.1 Prepare 600 mL of 50% (49.4 to 50.9%) solution as
maximum carbide precipitation, prior to testing. The length of
follows:
timeofheating,andthemethodofsubsequentcoolingusedfor
20.1.1 (Warning—Protect the eyes and use rubber gloves
this sensitizing treatment together with the corresponding
for handling acid. Place the test flask under a hood.)
maximum permissible corrosion rate shall be as agreed be-
20.1.2 First, measure 400.0 mL of Type IV reagent water
tween the material producer and purchaser.
and pour into the Erlenmeyer flask.
20.1.3 Then measure 236.0 mL of reagent-grade sulfuric
NOTE 8—The most commonly used sensitizing treatment is1hat
acid. Add the acid slowly and with constant stirring to the
675°C (1250°F).
water in the Erlenmeyer flask to avoid boiling by the heat
22.2 Preparethespecimens,eachhavingatotalsurfacearea
evolved. 2
of5to20cm .
NOTE 7—Loss of vapor results in concentration of the acid.
22.3 Where feasible for the product form, grind all the
20.1.4 Weigh 25 g of reagent-grade ferric sulfate to the
specimen surfaces using CAMI/ANSI 120 [FEPA/ISO P120]
nearest 0.1 g and add to the sulfuric acid solution. paper-backed, wet or dry, closed coated abrasive paper, with
20.1.5 Drop boiling chips into the flask. water as a coolant. If abrasive paper is used dry, polish slowly
20.1.6 Lubricate ground glass joint with silicone grease. to avoid overheating. Do not use abrasives with grinding aids;
A262 − 15 (2021)
some grinding aids contain fluorides that can affect the 23.13 Several specimens may be tested simultaneously.The
measured corrosion rate. number (3 or 4) is limited only by the number of glass cradles
that can be fitted into the flask.
22.4 Remove all traces of oxide scale and heat tint formed
during heat treatments. Any scale that cannot be removed by
24. Calculation and Report
grinding (for example, in stamped numbers) may be removed
24.1 The effect of the acid solution on the material is
by using one of the pickling solutions described in Practice
measured by determining the loss of weight of the specimen.
A380/A380M, Table A1.1. (Residual oxide scale causes gal-
The corrosion rates should be reported as millimetres of
vanic action and consequent activation in the test solution.)
penetration per month (Note 9), calculated as follows:
22.5 Measurethespecimens,includingtheinnersurfacesof
Millimetrepermonth 5 ~7305 3 W!/~A 3 t 3 d! (1)
any holes, to the nearest 0.05 mm (0.001 in.) and calculate the
where:
total exposed area.
t = time of exposure, h,
22.6 Degrease the specimens using suitable nonchlorinated
A = area, cm ,
agents, such as soap and lukewarm water, or acetone. Dry the
W = weight loss, g, and
specimensandweigheachonetothenearest0.001g.Storethe 3
d = density, g/cm
specimens in a desiccator until the test is to be performed.
for chromium-nickel steels, d = 7.9 g/cm
for chromium-nickel-molybdenum steels, d = 8.00 g/cm
23. Procedure
NOTE 9—Conversion factors to other commonly used units for corro-
23.1 If the test solution is not already boiling, bring it to
sion rates are as follows:
boiling.
Millimetres per month × 0.04 = inches per month
Millimetres per month × 0.47 = inches per year
23.1.1 Keep the flask covered with the condenser (with
Millimetres per month × 12 = millimetres per year
cooling water flowing) except when inserting or removing
Millimetres per month × 472 = mils per year
specimens. (See Note 7.)
Millimetres per month × 1000 × density/3 = milligrams per square
decimetre per day
23.2 Turn off the heat source and allow the boiling to
Millimetrespermonth×1.39×density=gramspersquaremetreperhour
subside.
25. Precision and Bias
23.3 Place specimens in glass cradles.
25.1 Precision—The precision of Practice B is being deter-
23.4 Uncover the flask.
mined.
23.5 Insert the specimens.
25.2 Bias—This practice has no bias because the resistance
to intergranular corrosion is defined only in terms of this
23.6 Replace the condenser immediately, restore cooling
practice.
water flow, and turn on the heat source.
23.7 Marktheliquidlevelontheflasktoprovideacheckon PRACTICE C—NITRIC ACID TEST FOR
vapor loss, which would result in concentration of the acid. If DETECTING SUSCEPTIBILITY TO
there is an appreciable change in the level, repeat the test with INTERGRANULAR ATTACK IN
fresh solution and reground and reweighed specimens. AUSTENITIC STAINLESS STEELS
23.8 Continue the immersion of the specimens for a total of
26. Scope
120h(fivedays),thenremovethespecimens,rinseinwateror
26.1 This practice describes the procedure for conducting
acetone, and dry.
the boiling nitric acid test (2) as employed to measure the
23.9 Weigh the specimens and subtract the new weights
relative susceptibility of austenitic stainless steels to inter-
from original weights.
granular attack.
26.2 The presence or absence of intergranular attack in this
23.10 Intermediateweighingsareusuallynotnecessary.The
test is not necessarily a measure of the performance of the
test can be run without interruption for 120 h. However, if
material in other corrosive environments; in particular, it does
preliminary results are desired, the specimens can be removed
not provide a basis for predicting resistance to forms of
at any time for weighing.
corrosion other than intergranular, such as general corrosion,
23.11 Changes to the solution during the 120-h test periods
pitting, or stress-corrosion cracking.
are not necessary.
27. Summary of Test Method C, the Nitric Acid Test
23.12 If the corrosion rate is extraordinarily high, as evi-
denced by a change in the color (from yellow to green) of the 27.1 Aspecimen representative of the material to be evalu-
solution, additional ferric sulfate inhibitor may need to be ated is immersed in a boiling solution of nitric acid for a
added during the test. If the total weight loss of all the specified time. The resulting mass loss is converted to a
specimens in a flask exceeds 2 g, more ferric sulfate must be corrosion rate, which is compared to a specified maximum
added. (During the test, ferric sulfate is consumed at a rate of value to determine whether the material has the resistance to
10gforeach1gof dissolved stainless steel.) attack expected of the grade of material being tested.
A262 − 15 (2021)
28. Significance and Use
28.1 The nitric acid test detects susceptibility to rapid
intergranular attack associated with chromium carbide precipi-
tate
28.2 The corrosion potential of the nitric acid test (Practice
C)hasbeenreportedas0.75to1.0Vversusastandardcalomel
electrode as compared with 0.6Vfor Practice B, and 0.1Vfor
Practices E and F. (3).
NOTE10—Highercorrosionpotentialindicatesmoreseverelyoxidizing
conditions.Thehighcorrosionpotentialofthenitricacidtestsuggeststhat
it should be invoked only when the material is destined for nitric acid
service.
29. Rapid Screening Test
29.1 Before testing in the nitric acid test, specimens of
FIG. 9 Flask and Condenser for Nitric Acid Test
certain grades of stainless steel, as given in Table 2, may be
given a rapid screening test in accordance with procedures
given in PracticeA, OxalicAcid EtchTest for Classification of
30.4 Balance—An analytical balance capable of weighing
Etch Structures of Austenitic Stainless Steels. The use of the
to at least the nearest 0.001 g.
etchstructureevaluationsinconnectionwiththenitricacidtest
is specified in Table 2.
30.5 Desiccator—For storage of prepared specimens prior
to testing.
29.2 Heat treat the material in accordance with33.1 prior to
performing the etch test.
31. Nitric Acid Test Solution
29.3 Ignore “process-affected” areas, if any (see Section
31.1 Thetestsolutionshallbe65.0 60.2weight%asnitric
32); application of the nitric acid test to process-affected areas
acid determined by analysis.
is currently outside the scope of Practice C.
31.2 Prepare this solution by adding reagent grade nitric
29.4 Corrosion test specimens having acceptable etch struc-
acid (HNO Table 3) to reagent water at the rate of 108 mLof
tures in the Oxalic Acid Etch Test will be essentially free of
reagent water per litre of reagent nitric acid.
intergranular attack in the nitric acid test; such specimens are
31.3 (Warning—Protect the eyes and use rubber gloves for
acceptable without testing in the nitric acid test.All specimens
handling acid. Place the test flask under a hood.)
having suspect etch structures shall be tested in the nitric acid
test.
31.4 The nitric acid used shall conform to the American
Chemical Society Specifications for Reagent Chemicals and
30. Apparatus
the additional requirements of this test method as shown in
30.1 Container—A 1 L Erlenmeyer flask equipped with a
Table 3.
cold finger-type condenser, as illustrated in Fig. 9.
32. Sampling
30.2 Specimen Supports—Glass hooks, stirrups, or cradles
32.1 Obtain and prepare only base metal samples.
for supporting the specimens in the flask fully immersed at all
32.1.1 There are two classes of specimens to be considered:
times during the test and so designed that specimens tested in
base metal, and process-affected metal. Process-affected metal
the same container do not come in contact with each other.
contains any condition that affects the corrosion properties of
30.3 Heater—A means for heating the test solutions and of
the material in a non-uniform way, such as (but not limited to)
keepingthemboilingthroughoutthetestperiod.Anelectrically
welds; carburized. nitrided, or oxidized surfaces; mechanical
heated hot plate is satisfactory for this purpose.
deformation; and areas affected by heat. Base metal has none
of these conditions.
TABLE 2 Use of Etch Structure Classification from Oxalic Acid
A
Etch Test with Nitric Acid Test
TABLE 3 Nitric Acid Composition Limits
Acceptable Etch Suspect Etch
Grade
B
Structures Structures
Minimum Maximum
AISI 304 Step, dual, end grain I Ditch, end grain II
Nitric Acid (HNO ), mass percent 69.0 71.0
AISI 304L Step, dual, end grain I Ditch, end grain II
Ash, ppm { 5
ACI CF-8 Step, dual, isolated ferrite pools Ditch, interdendritic ditches
Chloride as Cl, ppm { 0.5
ACI CF-3 Step, dual, isolated ferrite pools Ditch, interdendritic ditches
Sulfate, as (SO ), ppm { 1
A
Grades not listed in this table either have not been evaluated for use of Practice Arsenic (As), ppm { 0.01
Heavy metals, as Pb, ppm { 0.2
A with Practice B or have been found to give acceptable results in the etch test
while giving unacceptable results in Practice B. In the latter case Practice A would Iron, (Fe), ppm { 0.2
Additional limits per Practices A262
pass material that should have been subjected to the ferric sulfate-sulfuric acid
test. Fluorine (F), ppm { 1
B
Phosphate (PO ), ppm { 0.2
Specimens having these structures shall be tested in the nitric acid test.
A262 − 15 (2021)
32.1.2 The Practice C test involves immersing the entire 33.5 Degrease the specimen using suitable nonchlorinated
specimenandaveragingthemasslossovertheentiresurfaceof agents,suchassoapandlukewarmwater,oracetone(Note13).
thespecimen.Welding,carburization,mechanicaldeformation, Dry the specimens and weigh each one to the nearest 0.001 g.
and the like, affect only part of a specimen. Store the specimens in a desiccator until the test is to be
performed.
32.1.3 The mass loss rate from process-affected metal is
expected to differ from that from base metal; the presence of
NOTE 13—The cleaning treatment described may be supplemented by
process-affected metal in a specimen will affect the calculated
immersing the specimen in nitric acid (for example, 20 weight% at 49 to
test result in an unpredictable way.
60°C (120 to 140°F)) for 20 min, followed by rinsing, drying, and
weighing. In the case of small-diameter tubular specimens which cannot
32.1.4 If the presence of these or other localized conditions
be conveniently resurfaced on the inside, it is desirable to include in the
is a concern to the purchaser, then tests that do not average the
preparation an immersion in boiling nitric acid (65%) for 2 to 4 h using
masslossoverthetotalspecimensurfacearea,suchasPractice
the same apparatus as for the actual test. The purpose of these treatments
A,theOxalicAcidEtchTest,orPracticeE,theCopper–Copper
is to remove any surface contamination that may not be accomplished by
Sulfate–Sulfuric Acid Test for Detecting Susceptibility to the regular cleaning method and which may increase the apparent weight
loss of the specimen during the early part of the test.
Intergranular Attack in Austenitic Stainless Steels, should be
considered. Details of the test and acceptance criteria shall be 33.6 The standard test is to test only one specimen of each
as agreed by the purchaser and producer.
materialorlotofmaterial.However,incaseofdispute,theuse
of at least two specimens for check purposes is recommended.
32.2 Unless otherwise specified by the purchaser, the pro-
cedures for obtaining representative base metal samples, for
34. Procedure
removing the specimens from the samples, and the number of
specimens shall be at the discretion of the producer.
34.1 Use a sufficient quantity of the nitric acid test solution
to cover the specimens and to provide a volume of at least 20
32.3 Whenspecimensarecutbyshearing,theshearededges
2 2
mL/cm (125 mL/in. ) of specimen surface. Normally, a
shall be refinished by machining or grinding prior to testing.
volume of about 600 mL is used.
33. Preparation of Test Specimens 34.2 Use a separate container for each test specimen.
34.2.1 As many as three specimens may be tested in the
33.1 Heat treat extra-low carbon and stabilized grades at
samecontainerprovidedthattheyallareofthesamegradeand
650 to 675°C (1200 to 1250°F), which is the range of
all show satisfactory resistance to corrosion.
maximum carbide precipitation, prior to testing. The length of
34.2.2 If more than one of the specimens tested in the same
timeofheating,andthemethodofsubsequentcoolingusedfor
container fail to pass the test, retest all the specimens in
this sensitizing treatment together with the corresponding
separate containers.
maximum permissible corrosion rate shall be as agreed be-
tween the material producer and purchaser. NOTE 14—Excessive corrosion of one specimen may result in acceler-
ated corrosion of the other specimens tested with it. Excessive corrosion
NOTE 11—The most commonly used sensitizing treatment is1hat
may often be detected by changes in the color of the test solution, and it
675°C (1250°F).
may be appropriate to provide separate containers for such specimens
NOTE 12—The size and shape of the specimen must be considered with
without waiting until the end of the test period.Arecord should be made
respect to available facilities for accurate weighing and the volume of test
showing which specimens were tested together.
solution to be used. Normally, the maximum convenient weight of a
34.3 Afterthespecimenshavebeenplacedintheacidinthe
specimenisabout100g.Inthecaseofbar,wire,andtubularproducts,the
proportion of the total area represented by the exposed cross section may
container, pass cooling water through the condenser, bring the
influence the results. Cross-sectional areas in these products may be
acid to a boil on the hot plate, and keep boiling throughout the
subjecttoendgrainattackinnitricacid.Theproportionofendgraininthe
test period (Note 15). After each test period, rinse the speci-
specimen should therefore be kept low unless such surface is actually to
mens with water and treat by scrubbing with rubber or a nylon
be exposed in service involving nitric acid. In this latter case, the
brush under running water to remove any adhering corrosion
proportion of end grain in the specimen should be kept high.
products, then dry and weigh them. Drying may be facilitated,
33.2 Where feasible for the product form, grind all the
if desired, by dipping the specimens in acetone after they are
specimen surfaces using CAMI/ANSI 120 [FEPA/ISO P120]
scrubbed.
paper-backed, wet or dry, closed coated abrasive paper, with
34.4 (Warning—It has been reported that violent boiling
water as a coolant. If abrasive paper is used dry, polish slowly
resulting in acid spil
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...