ASTM C560-20
(Test Method)Standard Test Methods for Chemical Analysis of Graphite
Standard Test Methods for Chemical Analysis of Graphite
SIGNIFICANCE AND USE
4.1 These test methods provide a practical way to measure the concentration of certain trace elements in graphite. Many end uses of graphite require that it be free of elements which may be incompatible with certain nuclear applications. Other elemental contamination can affect the rate of oxidative degradation.
4.2 These test methods allow measurement of trace amounts of contaminants with a minimal amount of costly equipment. The colorimetric procedures used are accessible to most laboratories.
4.3 Other instrumental analysis techniques are available, capable of simultaneous quantitative analysis of 76 stable elements in a single run, with detectability limits in the parts per million range. Standards are currently being developed for elemental analysis of impurities in graphite using glow discharge mass spectrometry (GDMS), inductively coupled plasma optical emission spectroscopy (ICP-OES), combustion ion chromatography (CIC).
SCOPE
1.1 These test methods cover the chemical analysis of graphite.
1.2 The analytical procedures appear in the following order:
Sections
Silicon by the Molybdenum Blue (Colorimetric) Test Method
9 to 15
Iron by the o-Phenanthroline (Colorimetric) Test Method
16 to 22
Calcium by the Permanganate (Colorimetric) Test Method
23 to 29
Aluminum by the 2-Quinizarin Sulfonic Acid Test Method
30 to 36
Titanium by the Peroxide (Colorimetric) Test Method
37 to 44
Vanadium by the 3,3′-Dimethylnaphthidine (Colorimetric)
Test Method
45 to 52
Boron by the Curcumin-Oxalic Acid (Colorimetric) Test Method
53 to 60
1.3 The preferred concentration of sought element in the final solution, the limits of sensitivity, and the precision of the results are given in Table 1.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See 56.1 for specific caution statement.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2020
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.F0 - Manufactured Carbon and Graphite Products
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Nov-2006
- Effective Date
- 15-Sep-2006
- Effective Date
- 01-May-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Jun-2005
- Effective Date
- 01-Dec-2004
- Effective Date
- 10-May-2002
- Effective Date
- 10-Apr-2000
- Effective Date
- 10-May-1999
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM C560-20 - Standard Test Methods for Chemical Analysis of Graphite provides a comprehensive set of colorimetric and photometric test methods for determining trace elemental impurities in graphite materials. These standardized methods allow manufacturers, laboratories, and quality assurance specialists to reliably measure the concentrations of elements that could impact the performance, safety, or regulatory compliance of graphite, particularly for critical applications such as nuclear energy and high-temperature environments.
Key elements analyzed include silicon, iron, calcium, aluminum, titanium, vanadium, and boron. Accurate elemental analysis ensures graphite purity, supporting its specialized uses in demanding industries.
Key Topics
- Trace Element Analysis: Procedures are included for the detection and quantification of low concentrations of elements that, even at the parts per million (ppm) level, may affect graphite’s properties.
- Colorimetric and Photometric Methods: The standard outlines practical, accessible laboratory techniques such as the molybdenum blue test for silicon, o-phenanthroline test for iron, and others based on color change and absorbance.
- Sensitivity and Precision: Detection limits are provided for each element, ensuring that laboratories can meet sensitivity requirements without advanced or expensive instrumentation.
- Silicon: Detection down to 1 μg/100 mL solution
- Iron: 40 μg/100 mL
- Calcium: 50 μg/100 mL
- Aluminum: 2 μg/100 mL
- Titanium: 200 μg/100 mL
- Vanadium: 5 μg/50 mL
- Boron: 0.1 μg/50 mL
- Sample Preparation and Calibration: Procedures for proper sample preparation, reagent specifications, and the use of calibration curves are detailed to ensure accurate results.
- Interference and Reproducibility: The methods are designed to minimize interference from other elements typically present in commercial graphite, with reproducibility data provided for most measurements.
Applications
The standardized test methods outlined in ASTM C560-20 are crucial in several key areas:
- Nuclear Graphite Production: Ensuring graphite contains only allowable trace elements, as certain impurities can be incompatible with nucleonic functions or contribute to undesirable radiation effects.
- High-Temperature/High-Purity Applications: In metallurgy, semiconductor manufacturing, and aerospace, even small amounts of elemental contaminants can affect electrical conductivity, mechanical strength, or oxidation resistance.
- Quality Control in Graphite Manufacturing: Routine monitoring of elemental impurities during production prevents batch failures and supports compliance with customer and regulatory requirements.
- Research and Development: Laboratories engaged in developing new graphite materials or characterizing novel products rely on these methods for accurate impurity assessment.
- Accessible Laboratory Settings: The prescribed colorimetric techniques require minimal specialized equipment, making them suitable for most commercial and industrial laboratories.
Related Standards
When performing chemical analysis of graphite or ensuring full compliance and comparability of results, reference can be made to these closely associated ASTM standards:
- ASTM C561 - Test Method for Ash in a Graphite Sample
- ASTM D1193 - Specification for Reagent Water
- ASTM E29 - Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
Additionally, emerging standards for advanced instrumental techniques such as Glow Discharge Mass Spectrometry (GDMS), Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES), and Combustion Ion Chromatography (CIC) provide supplemental methods for more comprehensive graphite impurity analysis.
Keywords: ASTM C560, chemical analysis of graphite, trace elements in graphite, colorimetric methods, graphite impurity analysis, graphite quality control, nuclear graphite, purity testing, elemental analysis in graphite.
Buy Documents
ASTM C560-20 - Standard Test Methods for Chemical Analysis of Graphite
REDLINE ASTM C560-20 - Standard Test Methods for Chemical Analysis of Graphite
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM C560-20 is a standard published by ASTM International. Its full title is "Standard Test Methods for Chemical Analysis of Graphite". This standard covers: SIGNIFICANCE AND USE 4.1 These test methods provide a practical way to measure the concentration of certain trace elements in graphite. Many end uses of graphite require that it be free of elements which may be incompatible with certain nuclear applications. Other elemental contamination can affect the rate of oxidative degradation. 4.2 These test methods allow measurement of trace amounts of contaminants with a minimal amount of costly equipment. The colorimetric procedures used are accessible to most laboratories. 4.3 Other instrumental analysis techniques are available, capable of simultaneous quantitative analysis of 76 stable elements in a single run, with detectability limits in the parts per million range. Standards are currently being developed for elemental analysis of impurities in graphite using glow discharge mass spectrometry (GDMS), inductively coupled plasma optical emission spectroscopy (ICP-OES), combustion ion chromatography (CIC). SCOPE 1.1 These test methods cover the chemical analysis of graphite. 1.2 The analytical procedures appear in the following order: Sections Silicon by the Molybdenum Blue (Colorimetric) Test Method 9 to 15 Iron by the o-Phenanthroline (Colorimetric) Test Method 16 to 22 Calcium by the Permanganate (Colorimetric) Test Method 23 to 29 Aluminum by the 2-Quinizarin Sulfonic Acid Test Method 30 to 36 Titanium by the Peroxide (Colorimetric) Test Method 37 to 44 Vanadium by the 3,3′-Dimethylnaphthidine (Colorimetric) Test Method 45 to 52 Boron by the Curcumin-Oxalic Acid (Colorimetric) Test Method 53 to 60 1.3 The preferred concentration of sought element in the final solution, the limits of sensitivity, and the precision of the results are given in Table 1. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See 56.1 for specific caution statement. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 These test methods provide a practical way to measure the concentration of certain trace elements in graphite. Many end uses of graphite require that it be free of elements which may be incompatible with certain nuclear applications. Other elemental contamination can affect the rate of oxidative degradation. 4.2 These test methods allow measurement of trace amounts of contaminants with a minimal amount of costly equipment. The colorimetric procedures used are accessible to most laboratories. 4.3 Other instrumental analysis techniques are available, capable of simultaneous quantitative analysis of 76 stable elements in a single run, with detectability limits in the parts per million range. Standards are currently being developed for elemental analysis of impurities in graphite using glow discharge mass spectrometry (GDMS), inductively coupled plasma optical emission spectroscopy (ICP-OES), combustion ion chromatography (CIC). SCOPE 1.1 These test methods cover the chemical analysis of graphite. 1.2 The analytical procedures appear in the following order: Sections Silicon by the Molybdenum Blue (Colorimetric) Test Method 9 to 15 Iron by the o-Phenanthroline (Colorimetric) Test Method 16 to 22 Calcium by the Permanganate (Colorimetric) Test Method 23 to 29 Aluminum by the 2-Quinizarin Sulfonic Acid Test Method 30 to 36 Titanium by the Peroxide (Colorimetric) Test Method 37 to 44 Vanadium by the 3,3′-Dimethylnaphthidine (Colorimetric) Test Method 45 to 52 Boron by the Curcumin-Oxalic Acid (Colorimetric) Test Method 53 to 60 1.3 The preferred concentration of sought element in the final solution, the limits of sensitivity, and the precision of the results are given in Table 1. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See 56.1 for specific caution statement. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM C560-20 is classified under the following ICS (International Classification for Standards) categories: 71.060.10 - Chemical elements. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C560-20 has the following relationships with other standards: It is inter standard links to ASTM C560-15e1, ASTM C561-91(2010)e1, ASTM E29-08, ASTM E29-06b, ASTM E29-06a, ASTM E29-06, ASTM D1193-06, ASTM C561-91(2005), ASTM E29-04, ASTM E29-02e1, ASTM C561-91(2000), ASTM E29-93a(1999), ASTM D1193-99e1, ASTM D1193-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C560-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C560 − 20
Standard Test Methods for
Chemical Analysis of Graphite
This standard is issued under the fixed designation C560; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* D1193 Specification for Reagent Water
E29 Practice for Using Significant Digits in Test Data to
1.1 These test methods cover the chemical analysis of
Determine Conformance with Specifications
graphite.
1.2 The analytical procedures appear in the following order:
3. Terminology
Sections
3.1 Definitions:
Silicon by the Molybdenum Blue (Colorimetric) Test Method 9 to 15
Ironbythe o-Phenanthroline (Colorimetric) Test Method 16 to 22
3.1.1 calibration solutions, n—solutions of accurately
Calcium by the Permanganate (Colorimetric) Test Method 23 to 29
known concentrations of the chemical element to be deter-
Aluminum by the 2-Quinizarin Sulfonic Acid Test Method 30 to 36
mined using the calibration curve method.
Titanium by the Peroxide (Colorimetric) Test Method 37 to 44
Vanadium by the 3,3'-Dimethylnaphthidine (Colorimetric) 45 to 52
3.1.2 colorimetric analysis, n—photometric analysis
Test Method
Boron by the Curcumin-Oxalic Acid (Colorimetric) Test Method 53 to 60
method of using absorption of monochromatic light in the
visible spectrum.
1.3 The preferred concentration of sought element in the
final solution, the limits of sensitivity, and the precision of the
3.1.3 photometric analysis, n—analytical chemistry method
results are given in Table 1.
for quantitative chemical analysis based on the relationship
1.4 The values stated in SI units are to be regarded as between solution concentrations and the absorption of mono-
chromatic light, as expressed by the Beer law.
standard. No other units of measurement are included in this
standard.
3.2 Definitions of Terms Specific to This Standard:
1.5 This standard does not purport to address all of the
3.2.1 calibration curve, n—graphical or mathematical rep-
safety concerns, if any, associated with its use. It is the
resentation of the relationship between known concentrations
responsibility of the user of this standard to establish appro-
of an element in a series of standard calibration solutions and
priate safety, health, and environmental practices and deter-
the measured response from the measurement system.
mine the applicability of regulatory limitations prior to use.
See 56.1 for specific caution statement.
4. Significance and Use
1.6 This international standard was developed in accor-
4.1 These test methods provide a practical way to measure
dance with internationally recognized principles on standard-
the concentration of certain trace elements in graphite. Many
ization established in the Decision on Principles for the
end uses of graphite require that it be free of elements which
Development of International Standards, Guides and Recom-
may be incompatible with certain nuclear applications. Other
mendations issued by the World Trade Organization Technical
elemental contamination can affect the rate of oxidative deg-
Barriers to Trade (TBT) Committee.
radation.
2. Referenced Documents
4.2 Thesetestmethodsallowmeasurementoftraceamounts
2.1 ASTM Standards:
of contaminants with a minimal amount of costly equipment.
C561 Test Method for Ash in a Graphite Sample
The colorimetric procedures used are accessible to most
laboratories.
These test methods are under the jurisdiction of ASTM Committee D02 on
4.3 Other instrumental analysis techniques are available,
Petroleum Products and Lubricants and are the direct responsibility of Subcommit-
capable of simultaneous quantitative analysis of 76 stable
tee D02.F0 on Petroleum Products, Liquid Fuels, and Lubricants
elements in a single run, with detectability limits in the parts
Current edition approved May 1, 2020. Published May 2020. Originally
ε1
per million range. Standards are currently being developed for
approved in 1965. Last previous edition approved in 2015 as C560 – 15 . DOI:
10.1520/C0560-20.
elemental analysis of impurities in graphite using glow dis-
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
charge mass spectrometry (GDMS), inductively coupled
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
plasma optical emission spectroscopy (ICP-OES), combustion
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. ion chromatography (CIC).
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C560 − 20
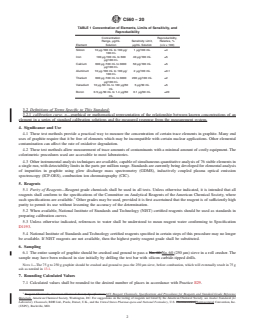
TABLE 1 Concentration of Elements, Limits of Sensitivity, and
8. Precision and Bias
Reproducibility
8.1 No statement is being made about either the precision or
Concentration Reproducibility,
bias of these test methods. At this time Committee C05 is
Range, µg/mL Sensitivity Limit, Relative, %
Element Solution µg/mL Solution (σ/x × 100)
investigating new standard methods of chemical analysis of
Silicon 10 µg ⁄100 mL to 100 µg/ 1 µg/100 mL ±4 graphite that will eventually replace these test methods. For
100 mL
this reason, no statistical study of these test methods has been
Iron 100 µg ⁄100 mL to 600 40 µg/100 mL ±5
planned.
µg/100 mL
Calcium 600 µg ⁄100 mL to 3000 50 µg/100 mL ±5
8.2 The relative reproducibility data in Table 1 has no
µg/100 mL
supportive research report on file and does not conform to
Aluminum 10 µg ⁄100 mL to 100 µg/ 2 µg/100 mL ±0.1
100 mL
ASTM precision and bias standards.
Titanium 600 µg ⁄100 mL to 3000 200 µg/100 mL ±2
µg/100 mL
SILICON BY THE MOLYBDENUM BLUE TEST
Vanadium 10 µg ⁄50 mL to 130 µg/50 5 µg/50 mL ±5
mL METHOD
Boron 0.5 µg ⁄50 mL to 1.4 µg/50 0.1 µg/50 mL ±20
mL
9. Summary of Test Method
9.1 Silicomolybdic acid is formed by adding ammonium
molybdate to soluble silicates in acid solution. The heteropoly
acid is reduced with stannous chloride to form a deep blue
5. Reagents
colloidal solution. Photometric measurement is made at
5.1 Purity of Reagents—Reagent grade chemicals shall be
765 nm. Regular classical gravimetric methods for silica using
used in all tests. Unless otherwise indicated, it is intended that
sodium carbonate fusion followed by hydrofluoric acid vola-
all reagents shall conform to the specifications of the Commit-
tilization may be suitable for use.
tee onAnalytical Reagents of theAmerican Chemical Society,
where such specifications are available. Other grades may be
10. Stability of Color
used, provided it is first ascertained that the reagent is of
10.1 The blue colored solution should be disposed of and
sufficiently high purity to permit its use without lessening the
the determination repeated if a period of 12 h has elapsed
accuracy of the determination.
between color development and measurements.
5.2 When available, National Institute of Standards and
Technology (NIST) certified reagents should be used as stan-
11. Interferences
dards in preparing calibration curves.
11.1 There is no interference from the ions usually present
5.3 Unless otherwise indicated, references to water shall be
in graphite.
understood to mean reagent water conforming to Specification
D1193.
12. Reagents
5.4 National Institute of Standards and Technology certified
12.1 Ammonium Molybdate (50 g⁄L)—Dissolve 50 g of am-
reagents specified in certain steps of this procedure may no
monium molybdate ((NH ) -Mo O ·4H O) in water and di-
4 6 7 24 2
longerbeavailable.IfNISTreagentsarenotavailable,thenthe
lute to 1 L.
highest purity reagent grade shall be substituted.
12.2 Hydrochloric Acid (HCl) (1+1)—Mix equal volumes
of concentrated HCl, sp gr 1.19 and water.
6. Sampling
12.3 Silicon, Standard Solution (1 mL = 1 mg Si)—Dissolve
6.1 The entire sample of graphite should be crushed and
10.1 g of sodium silicate (Na SiO ·9H O) in water and dilute
2 3 2
ground to pass a No. 60 (250 µm) sieve in a roll crusher. The
to 1 L in a volumetric flask. Store in a polyethylene bottle.
sample may have been reduced in size initially by drilling the
Determine exact concentration by the standard gravimetric
test bar with silicon carbide-tipped drills.
procedure.
NOTE 1—The 75 g to 250 g graphite should be crushed and ground to
12.4 Silicon, Working Solution (1 mL = 0.01 mg Si)—Dilute
pass the 250 µm sieve, before combustion, which will eventually result in
10 mL of standard silicon solution (1 mL = mg Si) to 1 L in a
75 g ash as needed in 13.1.
volumetric flask. Transfer to a polyethylene bottle.
7. Rounding Calculated Values
12.5 SodiumCarbonateSolution(100 g⁄L)—Dissolve100 g
7.1 Calculated values shall be rounded to the desired num-
ofsodiumcarbonate(Na CO )inwateranddiluteto1 L.Store
2 3
ber of places in accordance with Practice E29.
in a polyethylene bottle.
12.6 Stannous Chloride Solution—Dissolve 2.5 g of stan-
nous chloride (SnCl ·2H O) in 5 mL of hot concentrated HCl
3 2 2
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
(sp gr 1.19) and dilute to 250 mL with water. Prepare a fresh
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical solution every 2 weeks.
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
12.7 Sulfuric Acid (H SO ) (1+3)—Carefully mix 1 volume
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma- 2 4
copeial Convention, Inc. (USPC), Rockville, MD. of concentrated H SO , sp gr 1.84 with 3 volumes of water.
2 4
C560 − 20
13. Preparation of Calibration Curve where:
A = silicon per 100 mL of solution found in the aliquot
13.1 Calibration Solutions—Transfer 0 mL, 1.0 mL,
used, µg,
3.0 mL, 5.0 mL, 7.0 mL, and 10 mL of silicon working solu-
B = aliquot factor = original volume divided by aliquot
tion (1 mL = 0.01 mg Si) to 100 mL volumetric flasks. Add 5
taken for analysis, and
drops of H SO (1+3) and dilute to approximately 10 mL.
2 4
W = original sample weight, g.
13.2 Color Development—Add 2.5 mL of (NH ) Mo O
4 6 7 24
solution to each flask and let stand 5 min. Then add 5.0 mL of
IRON BY THE ORTHO-PHENANTHROLINE
H SO (1+3), mix well, and add 5 drops of SnCl solution. (PHOTOMETRIC) TEST METHOD
2 4 2
Dilute to volume and let stand 5 min.
16. Summary of Test Method
13.3 Photometry—Transfer a suitable portion of the reagent
16.1 After suitable dilution of an aliquot from the carbonate
blank solution to a 1 cm absorption cell and adjust the
fusion is adjusted to a pH of 3.0, the iron is reduced with
photometertotheinitialsetting,usingawavelengthof765 nm.
hydroxylamine hydrochloride. The ferrous ortho-
While maintaining this photometer adjustment, take the pho-
tometric readings of the calibration solutions. phenanthroline complex is formed, and its absorption is mea-
sured at a wavelength of 490 nm.
13.4 Calibration Curve—Plot the photometric readings (ab-
sorbance) of the calibration solution against micrograms of
17. Stability of Color
silicon per 100 mL of solution.
17.1 The color becomes stable within 15 min and does not
change for at least 48 h.
14. Procedure for Carbonate Fusion
14.1 Sample Solution—Rinse the ash (from a 50 g to 75 g
18. Interferences
ash sample) from the platinum dish into a mullite mortar with
18.1 No interfering elements are normally present in graph-
three 0.5 g portions of Na CO passing a No. 100 (150 µm)
2 3
ite.
sieve (see Test Method C561). Grind the resulting mixture to
pass a No. 200 (75 µm) sieve to ensure intimate contact of the
19. Reagents
ash with the flux. Then transfer the mixture to a platinum
19.1 Ammonium Hydroxide (NH OH) (1+1)—Mix equal
crucible(containing0.5 gofNa CO )withthree0.5 grinsesof 4
2 3
volumes of concentrated NH OH, sp gr 0.90 and water.
Na CO . Add sufficient Na CO to bring the total Na CO 4
2 3 2 3 2 3
content to 6 g. Cover the crucible, and fuse gently over a
19.2 Bromine Water—Add 10 mL of bromine to 1 L of
bunsen burner.
water. Allow to stand for 24 h.
NOTE 2—In order to get 75 g ash, one needs to combust 250 kg high
19.3 Hydrochloric Acid (HCl) (1+1)—Mix equal volumes
puruty graphite (300 ppm ash) or 75 kg low purity graphite (1000 ppm
of concentrated HCl, sp gr 1.19 and water.
ash).
19.4 Hydroxylamine Hydrochloride Solution—Dissolve
14.1.1 When fusion is complete (usually 30 min to 1 h),
10 g of hydroxylamine hydrochloride (NH OH·HCl) in water
removethecruciblefromtheburner,swirltodistributethemelt
and dilute to 100 mL. Discard the solution if color develops on
on the sides of the crucible, and allow to cool. Then place the
standing for long periods of time.
crucible and contents in a 200 mL high-form beaker and add
19.5 Iron, Standard Solution (1 mL = 0.1 mg Fe)—Into a
25 mL of water. Cover the beaker with a watch glass, and
100 mLbeaker, weigh 0.1000 g of iron wire. Dissolve the wire
cautiously add HCl (1+1) to decompose the melt. When
in 50 mLof HCl (1+1).Add 1 mLof bromine water to oxidize
solution of the melt is complete, boil for several minutes on a
the iron to the ferric state. Boil the solution to expel the excess
hot plate and cool.
bromine and dilute to 1 L in a volumetric flask.
14.1.2 Transfer to a 100 mL volumetric flask, dilute to
volume, and mix. Transfer a suitable aliquot of this solution to
19.6 Iron Wire, primary standard, over 99.9 % pure.
a 100 mL volumetric flask.
19.7 o-Phenanthroline—Dissolve 2 g of 1,10-
14.2 Color Development—Adjust the pH of the aliquot to 6
phenanthrolineinethylalcoholanddiluteto250 mLwithethyl
to 8 with Na CO solution, then proceed in accordance with
2 3 alcohol in a volumetric flask. Discard this solution if color
14.2.
develops upon long standing.
14.3 Photometry—Proceed in accordance with 13.3.
20. Preparation of Calibration Curve
14.4 Calibration—Convert the photometric reading of the
20.1 Calibration Solutions—Transfer 0.0 mL, 1.0 mL,
sample solution to micrograms of silicon by means of the
2.0 mL, 3.0 mL, 4.0 mL, 5.0 mL, and 6.0 mL of iron solution
calibration curve.
(1 mL= 0.1 mg Fe) to 100 mLvolumetric flasks.Add NH OH
(1+1) until the brown hydrous precipitate of ferric hydroxide
15. Calculation
(Fe(OH) ) is just visible.Then add HCl (1+1) drop-wise, while
15.1 Calculate the parts per million (ppm) of silicon in the
stirring, until the precipitate just dissolves. Bring the pH of the
original sample as follows:
solutionto3.0byadding2additionaldropsofHCl(1+1).Then
Silicon, ppm A 3B /W add 2 mL of NH OH·HCl solution.
~ !
C560 − 20
20.2 Color Development—Heat the solutions in the flasks 26.3 Bromocresol Green Indicator Solution—Use the water
almost to boiling. Add 1 mL of o-phenanthroline solution and soluble sodium salt. Dissolve 0.040 g in water and dilute to
allow the solutions to cool. Then dilute to the mark with water. 100 mL. Store in a glass-stoppered brown bottle.
26.4 Formate Buffer Solution (pH 3.7)—Dissolve 31.5 g of
20.3 Photometry—Transfer a suitable portion of the reagent
blank solution to a 1 cm absorption cell, and adjust the ammonium formate in about 200 mLof water and transfer to a
1 L volumetric flask. Add 20.8 mL of formic acid, dilute to
spectrophotometer to the initial setting using a wavelength of
volume, and mix well.
490 nm. While maintaining this photometer adjustment, take
the photometric readings of the calibration solutions.
26.5 Hydrochloric Acid (HCl) (1+1)—Mix equal volumes
of concentrated HCl, sp gr 1.19 and water.
20.4 Calibration Curve—Plot the absorbance of the calibra-
tion solution against micrograms of iron per 100 mL of
26.6 Oxalate, Standard Solution (1 mL = 0.125 mg Ca)—
solution.
Dry approximately 2 g of sodium oxalate (Na C O ) at 105 °C
2 2 4
for 1 h, and cool in a desiccator. Weigh accurately 0.2090 g
21. Procedure
into a 250 mL beaker, dissolve in boiled water, and dilute to
500 mL in a volumetric flask.
21.1 Sample Solution—Proceed in accordance with 14.1.
26.7 Potassium Permanganate, Standard Solution—
21.2 Color Development—Proceed in accordance with 20.2.
Dissolve 3.25 g of NIST potassium permanganate (KMnO )in
21.3 Photometry—Proceed in accordance with 20.2.
1 L of hot water. Let stand in the dark for 12 h. Filter through
21.4 Calibration—Convert the photometric reading of the inert filter medium into a dark colored bottle.
sample solution to micrograms of iron by means of the 26.7.1 Standardize as follows: dissolve 3.0 g of dried NIST
calibration curve. sodium oxalate (Na C O ) in boiled water and dilute to
2 2 4
500 mL in a volumetric flask. Pipet 25 mL aliquots of the
22. Calculation oxalate solution into 600 mL beakers. Add 250 mL of
H SO (1+33),heatto55 °Cto60 °C,andtitratetoafaintpink
2 4
22.1 Calculate the ppm of iron in the original sample as
end point that persists for 30 s. For a blank, add permanganate
follows:
solution, dropwise, to 250 mL of H SO (1+33). Note the
2 4
Fe, ppm A 3B /W
~ !
volume required to impart a pink color. Calculate the normality
of the permanganate solution.
where:
26.7.2 Prepare 0.0200 N KMnO solution by appropriate
A = iron per 100 mL of solution in the aliquot used, µg,
dilution of the standardized solution.
B = aliquot factor = original volume divided by aliquot
taken for analysis, and 26.8 Sulfuric Acid (H SO ) (1+3)—Carefully mix 1 volume
2 4
W = original sample weight, g.
of concentrated H SO , sp gr 1.84 with 3 volumes of water.
2 4
27. Preparation of Calibration Curve
CALCIUM BY THE PERMANGANATE
(COLORIMETRIC) TEST METHOD
27.1 Calibration Solutions—Transfer 0.0 mL, 5.0 mL,
10.0 mL, 15.0 mL, and 25.0 mL of standard oxalate solution
23. Summary of Test Method
into 100 mLvolumetric flasks.Add 40 mLof H SO (1+3) and
2 4
10 mL of boiled and cooled water. Place the flasks in a water
23.1 Calcium is precipitated as the oxalate, filtered off, and
bath at 55 °C to 60 °C for 5 min.
dissolved in sulfuric acid.The acid solution is added to a dilute
potassium permanganate solution, and the decrease in absorp-
27.2 Color Development—Pipet into each flask 10.0 mL of
tion is measured at a wavelength of 528 nm.
the 0.0200 N KMnO solution. Remove from the bath and
allow to stand at room temperature for 5 min for the color
24. Stability of Color
change to be completed. Place in a cold-water bath, and cool to
room temperature. Dilute to volume with CO -free water and
24.1 Potassium permanganate solution is decomposed rap- 2
mix.
idly by exposure to air or light. Photometric readings should be
made at once.
27.3 Photometry—Transfer a portion of the reagent blank
solution to a 1 cm absorption cell.Transfer a portion of the first
standard into a second absorption cell. Adjust the spectropho-
25. Interferences
tometer to zero, with the standard in the light path. Then
25.1 Ashedgraphitesamplesarenormallyfreeofsignificant
measure the absorbance of the reference solution. Repeat the
concentrations of possible interfering ions.
procedure using the other standard solutions.
27.4 Calibration Curve—Plot the absorption of the calibra-
26. Reagents
tion solutions against micrograms of calcium per 100 mL of
26.1 Ammonium Hydroxide (NH OH ) (1+6)—Mix 1 vol-
4 2
solution.
ume of concentrated NH OH , sp gr 0.90 with 6 volumes of
4 2
water.
28. Procedure
26.2 Ammonium Oxalate Solution—Prepare a saturated so- 28.1 Sample Solution—Proceed in accordance with 14.1.
lution of ammonium oxalate ((NH ) C O ·2H O). However, after the sample solution has been diluted to volume
4 2 2 4 2
C560 − 20
and mixed, proceed as follows: pipet a suitable aliquot (usually transfer to a 500 mL volumetric flask. Cover the salt with
25 mL) into a 50 mL beaker. Add 1 or 2 drops of bromocresol 200 mL of absolute methanol. Add 10 mL of concentrated
green indicator, 1 mL of formate buffer, and 1 mL of saturated hydrochloric acid (HCl, sp gr 1.19) to dissolve the salt, and
(NH ) C O solution. Add, dropwise, NH OH
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: C560 − 15 C560 − 20 An American National Standard
Standard Test Methods for
Chemical Analysis of Graphite
This standard is issued under the fixed designation C560; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Subsection 1.2 was corrected editorially in February 2017.
1. Scope*
1.1 These test methods cover the chemical analysis of graphite.
1.2 The analytical procedures appear in the following order:
Sections
Silicon by the Molybdenum Blue (Colorimetric) Test Method 9 to 15
Iron by the o-Phenanthroline (Colorimetric) Test Method 16 to 22
Calcium by the Permanganate (Colorimetric) Test Method 23 to 29
Aluminum by the 2-Quinizarin Sulfonic Acid Test Method 30 to 36
Titanium by the Peroxide (Colorimetric) Test Method 37 to 44
Vanadium by the 3,3'-Dimethylnaphthidine (Colorimetric) 45 to 52
Test Method
Boron by the Curcumin-Oxalic Acid (Colorimetric) Test Method 53 to 60
1.3 The preferred concentration of sought element in the final solution, the limits of sensitivity, and the precision of the results
are given in Table 1.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. See 56.1 for specific caution statement.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C561 Test Method for Ash in a Graphite Sample
D1193 Specification for Reagent Water
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
3. Terminology
3.1 Definitions:
3.1.1 calibration curve, n—graphical or mathematical representation of the relationship between known concentrations of an
element in a series of standard calibration solutions and the measured response from the measurement system.
3.1.1 calibration solutions, n—solutions of accurately known concentrations of the chemical element to be determined using the
calibration curve method.
3.1.2 colorimetric analysis, n—photometric analysis method of using absorption of monochromatic light in the visible spectrum.
3.1.3 photometric analysis, n—analytical chemistry method for quantitative chemical analysis based on the relationship between
solution concentrations and the absorption of monochromatic light, as expressed by the Beer law.
These test methods are under the jurisdiction of ASTM Committee D02 on Petroleum Products and Lubricants and are the direct responsibility of Subcommittee D02.F0
on Petroleum Products, Liquid Fuels, and Lubricants
Current edition approved Oct. 1, 2015May 1, 2020. Published November 2015May 2020. Originally approved in 1965. Last previous edition approved in 20102015 as
ε1
C560 – 88 (2010)C560 – 15 . DOI: 10.1520/C0560-15E01.10.1520/C0560-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C560 − 20
TABLE 1 Concentration of Elements, Limits of Sensitivity, and
Reproducibility
Concentration Reproducibility,
Range, μg/mL Sensitivity Limit, Relative, %
Element Solution μg/mL Solution (σ/x × 100)
Silicon 10 μg ⁄100 mL to 100 μg/ 1 μg/100 mL ±4
100 mL
Iron 100 μg ⁄100 mL to 600 40 μg/100 mL ±5
μg/100 mL
Calcium 600 μg ⁄100 mL to 3000 50 μg/100 mL ±5
μg/100 mL
Aluminum 10 μg ⁄100 mL to 100 μg/ 2 μg/100 mL ±0.1
100 mL
Titanium 600 μg ⁄100 mL to 3000 200 μg/100 mL ±2
μg/100 mL
Vanadium 10 μg ⁄50 mL to 130 μg/50 5 μg/50 mL ±5
mL
Boron 0.5 μg ⁄50 mL to 1.4 μg/50 0.1 μg/50 mL ±20
mL
3.2 Definitions of Terms Specific to This Standard:
3.2.1 calibration curve, n—graphical or mathematical representation of the relationship between known concentrations of an
element in a series of standard calibration solutions and the measured response from the measurement system.
4. Significance and Use
4.1 These test methods provide a practical way to measure the concentration of certain trace elements in graphite. Many end
uses of graphite require that it be free of elements which may be incompatible with certain nuclear applications. Other elemental
contamination can affect the rate of oxidative degradation.
4.2 These test methods allow measurement of trace amounts of contaminants with a minimal amount of costly equipment. The
colorimetric procedures used are accessible to most laboratories.
4.3 Other instrumental analysis techniques are available, capable of simultaneous quantitative analysis of 76 stable elements in
a single run, with detectability limits in the parts per million range. Standards are currently being developed for elemental analysis
of impurities in graphite using glow discharge mass spectrometry (GDMS), inductively coupled plasma optical emission
spectroscopy (ICP-OES), combustion ion chromatography (CIC).
5. Reagents
5.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
5.2 When available, National Institute of Standards and Technology (NIST) certified reagents should be used as standards in
preparing calibration curves.
5.3 Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to Specification
D1193.
5.4 National Institute of Standards and Technology certified reagents specified in certain steps of this procedure may no longer
be available. If NIST reagents are not available, then the highest purity reagent grade shall be substituted.
6. Sampling
6.1 The entire sample of graphite should be crushed and ground to pass a No. 60 No. 60 (250 μm) sieve in a roll crusher. The
sample may have been reduced in size initially by drilling the test bar with silicon carbide-tipped drills.
NOTE 1—The 75 g to 250 g graphite should be crushed and ground to pass the 250 μm sieve, before combustion, which will eventually result in 75 g
ash as needed in 13.1.
7. Rounding Calculated Values
7.1 Calculated values shall be rounded to the desired number of places in accordance with Practice E29.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. PharmaceuticalPharmacopeial Convention, Inc.
(USPC), Rockville, MD.
C560 − 20
8. Precision and Bias
8.1 No statement is being made about either the precision or bias of these test methods. At this time Committee C05 is
investigating new standard methods of chemical analysis of graphite that will eventually replace these test methods. For this
reason, no statistical study of these test methods has been planned.
8.2 The relative reproducibility data in Table 1 has no supportive research report on file and does not conform to ASTM
precision and bias standards.
SILICON BY THE MOLYBDENUM BLUE TEST METHOD
9. Summary of Test Method
9.1 Silicomolybdic acid is formed by adding ammonium molybdate to soluble silicates in acid solution. The heteropoly acid is
reduced with stannous chloride to form a deep blue colloidal solution. Photometric measurement is made at 765 nm. Regular
classical gravimetric methods for silica using sodium carbonate fusion followed by hydrofluoric acid volatilization may be suitable
for use.
10. Stability of Color
10.1 The blue colored solution should be disposed of and the determination repeated if a period of 12 h has elapsed between
color development and measurements.
11. Interferences
11.1 There is no interference from the ions usually present in graphite.
12. Reagents
12.1 Ammonium Molybdate (50 (50 g g/L)—⁄L)—Dissolve 50 g of ammonium molybdate ((NH ) -Mo O ·4H O) in water and
4 6 7 24 2
dilute to 1 L.
12.2 Hydrochloric Acid (HCl) (1+1)—Mix equal volumes of concentrated HCl, sp gr 1.19 and water.
12.3 Silicon, Standard Solution (1 mL = 1 mg Si)—Dissolve 10.1 g of sodium silicate (Na SiO ·9H O) in water and dilute to
2 3 2
1 L in a volumetric flask. Store in a polyethylene bottle. Determine exact concentration by the standard gravimetric procedure.
12.4 Silicon, Working Solution (1 mL = 0.01 mg Si)—Dilute 10 mL of standard silicon solution (1 mL = mg Si) to 1 L in a
volumetric flask. Transfer to a polyethylene bottle.
12.5 Sodium Carbonate Solution (100 g ⁄L)—Dissolve 100 g of sodium carbonate (Na CO ) in water and dilute to 1 L. Store in
2 3
a polyethylene bottle.
12.6 Stannous Chloride Solution—Dissolve 2.5 g of stannous chloride (SnCl ·2H O) in 5 mL of hot concentrated HCl (sp gr
2 2
1.19) and dilute to 250 mL with water. Prepare a fresh solution every 2 weeks.
12.7 Sulfuric Acid (H SO ) (1+3)—Carefully mix 1 volume of concentrated H SO , sp gr 1.84 with 3 volumes of water.
2 4 2 4
13. Preparation of Calibration Curve
13.1 Calibration Solutions—Transfer 0 mL, 1.0 mL, 3.0 mL, 5.0 mL, 7.0 mL, and 10 mL of silicon working solution (1 mL =
0.01 mg Si) to 100 mL volumetric flasks. Add 5 drops of H SO (1+3) and dilute to approximately 10 mL.
2 4
13.2 Color Development—Add 2.5 mL of (NH ) Mo O solution to each flask and let stand 5 min. Then add 5.0 mL of
4 6 7 24
H SO (1+3), mix well, and add 5 drops of SnCl solution. Dilute to volume and let stand 5 min.
2 4 2
13.3 Photometry—Transfer a suitable portion of the reagent blank solution to a 1 cm absorption cell and adjust the photometer
to the initial setting, using a wavelength of 765 nm. While maintaining this photometer adjustment, take the photometric readings
of the calibration solutions.
13.4 Calibration Curve—Plot the photometric readings (absorbance) of the calibration solution against micrograms of silicon
per 100 mL of solution.
14. Procedure for Carbonate Fusion
14.1 Sample Solution—Rinse the ash (from a 50 g to 75 g ash sample) from the platinum dish into a mullite mortar with three
0.5 g portions of Na CO passing a No. 100 (150 μm) sieve (see Test Method C561). Grind the resulting mixture to pass a No.
2 3
200 (75 μm) sieve to ensure intimate contact of the ash with the flux. Then transfer the mixture to a platinum crucible (containing
0.5 g of Na CO ) with three 0.5 g rinses of Na CO . Add sufficient Na CO to bring the total Na CO content to 6 g. Cover the
2 3 2 3 2 3 2 3
crucible, and fuse gently over a bunsen burner.
NOTE 2—In order to get 75 g ash, one needs to combust 250 kg high puruty graphite (300 ppm ash) or 75 kg low purity graphite (1000 ppm ash).
C560 − 20
14.1.1 When fusion is complete (usually 30 min to 1 h), remove the crucible from the burner, swirl to distribute the melt on the
sides of the crucible, and allow to cool. Then place the crucible and contents in a 200 mL high-form beaker and add 25 mL of water.
Cover the beaker with a watch glass, and cautiously add HCl (1+1) to decompose the melt. When solution of the melt is complete,
boil for several minutes on a hot plate and cool.
14.1.2 Transfer to a 100 mL volumetric flask, dilute to volume, and mix. Transfer a suitable aliquot of this solution to a 100 mL
volumetric flask.
14.2 Color Development—Adjust the pH of the aliquot to 6 to 8 with Na CO solution, then proceed in accordance with 14.2.
2 3
14.3 Photometry—Proceed in accordance with 13.3.
14.4 Calibration—Convert the photometric reading of the sample solution to micrograms of silicon by means of the calibration
curve.
15. Calculation
15.1 Calculate the parts per million (ppm) of silicon in the original sample as follows:
Silicon, ppm ~A 3B!/W
where:
A = silicon per 100 mL of solution found in the aliquot used, μg,
B = aliquot factor = original volume divided by aliquot taken for analysis, and
W = original sample weight, g.
IRON BY THE ORTHO-PHENANTHROLINE
(PHOTOMETRIC) TEST METHOD
16. Summary of Test Method
16.1 After suitable dilution of an aliquot from the carbonate fusion is adjusted to a pH of 3.0, the iron is reduced with
hydroxylamine hydrochloride. The ferrous ortho-phenanthroline complex is formed, and its absorption is measured at a wavelength
of 490 nm.
17. Stability of Color
17.1 The color becomes stable within 15 min and does not change for at least 48 h.
18. Interferences
18.1 No interfering elements are normally present in graphite.
19. Reagents
19.1 Ammonium Hydroxide (NH OH) (1+1)—Mix equal volumes of concentrated NH OH, sp gr 0.90 and water.
4 4
19.2 Bromine Water—Add 10 mL of bromine to 1 L of water. Allow to stand for 24 h.
19.3 Hydrochloric Acid (HCl) (1+1)—Mix equal volumes of concentrated HCl, sp gr 1.19 and water.
19.4 Hydroxylamine Hydrochloride Solution—Dissolve 10 g of hydroxylamine hydrochloride (NH OH·HCl) in water and dilute
to 100 mL. Discard the solution if color develops on standing for long periods of time.
19.5 Iron, Standard Solution (1 mL = 0.1 mg Fe)—Into a 100 mL beaker, weigh 0.1000 g of iron wire. Dissolve the wire in
50 mL of HCl (1+1). Add 1 mL of bromine water to oxidize the iron to the ferric state. Boil the solution to expel the excess bromine
and dilute to 1 L in a volumetric flask.
19.6 Iron Wire, primary standard, over 99.9 % pure.
19.7 o-Phenanthroline—Dissolve 2 g of 1,10-phenanthroline in ethyl alcohol and dilute to 250 mL with ethyl alcohol in a
volumetric flask. Discard this solution if color develops upon long standing.
20. Preparation of Calibration Curve
20.1 Calibration Solutions—Transfer 0.0, mL0.0 mL, 1.0 mL, 2.0 mL, 3.0 mL, 4.0 mL, 5.0 mL, and 6.0 mL of iron solution
(1 mL = 0.1 mg Fe) to 100 mL volumetric flasks. Add NH OH (1+1) until the brown hydrous precipitate of ferric hydroxide
(Fe(OH) ) is just visible. Then add HCl (1+1) drop-wise, while stirring, until the precipitate just dissolves. Bring the pH of the
solution to 3.0 by adding 2 additional drops of HCl (1+1). Then add 2 mL of NH OH·HCl solution.
20.2 Color Development—Heat the solutions in the flasks almost to boiling. Add 1 mL of o-phenanthroline solution and allow
the solutions to cool. Then dilute to the mark with water.
C560 − 20
20.3 Photometry—Transfer a suitable portion of the reagent blank solution to a 1 cm absorption cell, and adjust the
spectrophotometer to the initial setting using a wavelength of 490 nm. While maintaining this photometer adjustment, take the
photometric readings of the calibration solutions.
20.4 Calibration Curve—Plot the absorbance of the calibration solution against micrograms of iron per 100 mL of solution.
21. Procedure
21.1 Sample Solution—Proceed in accordance with 14.1.
21.2 Color Development—Proceed in accordance with 20.2.
21.3 Photometry—Proceed in accordance with 20.2.
21.4 Calibration—Convert the photometric reading of the sample solution to micrograms of iron by means of the calibration
curve.
22. Calculation
22.1 Calculate the ppm of iron in the original sample as follows:
Fe, ppm A 3B /W
~ !
where:
A = iron per 100 mL of solution in the aliquot used, μg,
B = aliquot factor = original volume divided by aliquot taken for analysis, and
W = original sample weight, g.
CALCIUM BY THE PERMANGANATE
(COLORIMETRIC) TEST METHOD
23. Summary of Test Method
23.1 Calcium is precipitated as the oxalate, filtered off, and dissolved in sulfuric acid. The acid solution is added to a dilute
potassium permanganate solution, and the decrease in absorption is measured at a wavelength of 528 nm.
24. Stability of Color
24.1 Potassium permanganate solution is decomposed rapidly by exposure to air or light. Photometric readings should be made
at once.
25. Interferences
25.1 Ashed graphite samples are normally free of significant concentrations of possible interfering ions.
26. Reagents
26.1 Ammonium Hydroxide (NH OH ) (1+6)—Mix 1 volume of concentrated NH OH , sp gr 0.90 with 6 volumes of water.
4 2 4 2
26.2 Ammonium Oxalate Solution—Prepare a saturated solution of ammonium oxalate ((NH ) C O ·2H O).
4 2 2 4 2
26.3 Bromocresol Green Indicator Solution—Use the water soluble sodium salt. Dissolve 0.040 g in water and dilute to 100 mL.
Store in a glass-stoppered brown bottle.
26.4 Formate Buffer Solution (pH 3.7)—Dissolve 31.5 g of ammonium formate in about 200 mL of water and transfer to a 1 L
volumetric flask. Add 20.8 mL of formic acid, dilute to volume, and mix well.
26.5 Hydrochloric Acid (HCl) (1+1)—Mix equal volumes of concentrated HCl, sp gr 1.19 and water.
26.6 Oxalate, Standard Solution (1 mL = 0.125 mg Ca)—Dry approximately 2 g of sodium oxalate (Na C O ) at 105 °C for 1 h,
2 2 4
and cool in a desiccator. Weigh accurately 0.2090 g into a 250 mL beaker, dissolve in boiled water, and dilute to 500 mL in a
volumetric flask.
26.7 Potassium Permanganate, Standard Solution—Dissolve 3.25 g of NIST potassium permanganate (KMnO ) in 1 L of hot
water. Let stand in the dark for 12 h. Filter through inert filter medium into a dark colored bottle.
26.7.1 Standardize as follows: dissolve 3.0 g of dried NIST sodium oxalate (Na C O ) in boiled water and dilute to 500 mL in
2 2 4
a volumetric flask. Pipet 25 mL aliquots of the oxalate solution into 600 mL beakers. Add 250 mL of H SO (1+33), heat to 55 °C
2 4
to 60 °C, and titrate to a faint pink end point that persists for 30 s. For a blank, add permanganate solution, dropwise, to 250 mL
of H SO (1+33). Note the volume required to impart a pink color. Calculate the normality of the permanganate solution.
2 4
26.7.2 Prepare 0.0200 N KMnO solution by appropriate dilution of the standardized solution.
26.8 Sulfuric Acid (H SO ) (1+3)—Carefully mix 1 volume of concentrated H SO , sp gr 1.84 with 3 volumes of water.
2 4 2 4
C560 − 20
27. Preparation of Calibration Curve
27.1 Calibration Solutions—Transfer 0.0 mL, 5.0 mL, 10.0 mL, 15.0 mL, and 25.0 mL of standard oxalate solution into 100 mL
volumetric flasks. Add 40 mL of H SO (1+3) and 10 mL of boiled and cooled water. Place the flasks in a water bath at 55 °C to
2 4
60 °C for 5 min.
27.2 Color Development—Pipet into each flask 10.0 mL of the 0.0200 N KMnO solution. Remove from the bath and allow to
stand at room temperature for 5 min for the color change to be completed. Place in a cold-water bath, and cool to room temperature.
Dilute to volume with CO -free water and mix.
27.3 Photometry—Transfer a portion of the reagent blank solution to a 1 cm absorption cell. Transfer a portion of the first
standard into a second absorption cell. Adjust the spectrophotometer to zero, with the standard in the light path. Then measure the
absorbance of the reference solution. Repeat the procedure using the other standard solutions.
27.4 Calibration Curve—Plot the absorption of the calibration solutions against micrograms of calcium per 100 mL of solution.
28. Procedure
28.1 Sample Solution—Proceed in accordance with 14.1. However, after the sample solution has been diluted to volume and
mixed, proceed as follows: pipet a suitable aliquot (usually 25 mL) into a 50 mL beaker. Add 1 or 2 drops of bromocresol green
indicator, 1 mL of formate buffer, and 1 mL of saturated (NH ) C O solution. Add,
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...