ASTM D7542-21
(Test Method)Standard Test Method for Air Oxidation of Carbon and Graphite in the Kinetic Regime
Standard Test Method for Air Oxidation of Carbon and Graphite in the Kinetic Regime
SIGNIFICANCE AND USE
5.1 This test method can be used to measure the rate of oxidation for various grades of manufactured carbon and graphite in standard conditions, and can be used for quality control purposes.
5.2 The following conditions are standardized in this test method: size and shape of the graphite specimens; their placement in the vertical furnace with upwards air flow; the method for continuous weight variation measurement using an analytical scale with under-the-scale port; the air flow rate, which must be high enough to ensure that oxidation is not oxygen-starved at the highest temperature used; the initial and final points on the weight loss curve used for calculation of oxidation rate.
5.3 This test method also provides kinetic parameters (apparent activation energy and logarithm of pre-exponential factor) for the oxidation reaction, and a standard oxidation temperature. The results characterize the effect of temperature on oxidation rates in air, and the oxidation resistance of machined carbon or graphite specimens with standard size and shape, in the kinetic, or chemically controlled, oxidation regime. This information is useful for discrimination between material grades with different impurity levels, grain size, pore structure, degree of graphitization, or antioxidation treatments, or a combination thereof.
5.4 Accurately determined kinetic parameters, like activation energy and logarithm of pre-exponential factor, can be used for prediction of oxidation rates in air as a function of temperature in conditions similar to those of this test method. However, extrapolation of such predictions outside the temperature range where Arrhenius plots are linear (outside the kinetic or chemically controlled regime of oxidation) should be made with extreme caution. In conditions where (1) oxidation rates become controlled by a mechanism other than chemical reactions (such as in-pore diffusion or boundary transport of the oxidant gas), or (2) the oxidant supply rate is no...
SCOPE
1.1 This test method recommends a standard procedure for measuring oxidation rates in air of various grades of nuclear graphite and/or manufactured carbon. Following the standard procedure recommended here, one can obtain kinetic parameters that characterize the oxidation resistance in standard conditions of tested materials and that can be used to for materials selection and qualification, and for quality control purposes in the fabrication process.
1.2 This test method covers the rate of oxidative weight loss per exposed nominal geometric surface area, or per initial weight of machined test specimens of standard size and shape, or both. The test is valid in the temperature range where the rate of air oxidation of graphite and manufactured carbon is limited by reaction kinetics.
1.3 This test method also provides a standard oxidation temperature (as defined in 3.1.7), and the kinetic parameters of the oxidation reaction, namely the apparent activation energy and the logarithm of pre-exponential factor in Arrhenius equation. The kinetic parameters of Arrhenius equation are calculated from the temperature dependence of oxidation rates measured over the temperature range where Arrhenius plots (as defined in 3.1.8) are linear, which is defined as the “kinetic” or “chemical control” oxidation regime. For typical nuclear grade graphite materials it was found that the practical range of testing temperatures is from about 500 °C to 550 °C up to about 700 °C to 750 °C.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international s...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2021
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.F0 - Manufactured Carbon and Graphite Products
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jan-2016
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Aug-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Apr-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Oct-2005
- Effective Date
- 01-Jun-2005
- Effective Date
- 01-May-2004
Overview
ASTM D7542-21: Standard Test Method for Air Oxidation of Carbon and Graphite in the Kinetic Regime details a standardized procedure for measuring oxidation rates of different grades of nuclear graphite and manufactured carbon. This method focuses on the kinetic, or chemically controlled, regime of oxidation-where reaction kinetics, not mass transport, control oxidation rates. The standard enables precise evaluation of oxidation resistance and the collection of kinetic parameters under specified laboratory conditions. These results aid in material selection, quality control, and qualification in areas where carbon and graphite are exposed to oxidizing air atmospheres at elevated temperatures.
Key Topics
- Oxidation Rate Measurement: The test determines either area-normalized or weight-normalized oxidation rates using cylindrical specimens of standard dimensions in a controlled air flow and temperature environment.
- Kinetic Parameters: This method calculates the apparent activation energy and the logarithm of the pre-exponential factor from Arrhenius plots, providing insight into how temperature impacts the oxidation rate.
- Standardization of Conditions:
- Specimen size and shape
- Placement in a vertical tube furnace with upwards air flow
- Continuous weight measurement via an analytical scale
- Air flow regulation to maintain non-oxygen-starved conditions at all test temperatures
- Use of both area-normalized and weight-normalized results for versatility
- Temperature Range: Valid for the kinetic regime, typically from about 500°C–750°C, depending on the density and grade of the graphite or carbon material.
- Quality Control and Material Qualification: The procedure is designed for repeatable, reliable use in manufacturing and research environments.
Applications
The ASTM D7542-21 test method is essential for:
- Quality Control: Ensuring consistency in graphite and carbon products by monitoring oxidation resistance across different lots or production runs.
- Materials Selection: Comparing grades with varying impurity levels, grain sizes, porosities, and antioxidants to select optimal materials for high-temperature and oxidative environments.
- Product Qualification: Documenting oxidation performance for critical applications-such as nuclear reactors, metallurgy, and electronics-where predictable material lifetimes are crucial.
- Predictive Analysis: Using measured kinetic parameters to estimate service life or performance at temperatures similar to those tested, within the kinetic regime.
- Research and Development: Allowing researchers to analyze the impact of microstructural or compositional changes on oxidation resistance.
Related Standards
Practitioners using ASTM D7542-21 should also be aware of the following related ASTM standards, which support accurate measurement and analysis:
- ASTM C559: Test Method for Bulk Density by Physical Measurements of Manufactured Carbon and Graphite Articles
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM E898: Practice for Calibration of Non-Automatic Weighing Instruments
- ASTM E1582: Test Method for Temperature Calibration of Thermogravimetric Analyzers
- ASTM E1970: Practice for Statistical Treatment of Thermoanalytical Data
Practical Value
Implementing ASTM D7542-21 ensures:
- Reliable and Repeatable Testing: Critical for quality assurance programs in carbon and graphite manufacturing.
- Transferable, Comparable Results: Standardized definitions and procedures enable meaningful comparison between materials and laboratories.
- Compliance and Certification: Meets international expectations for standardization in high-reliability industries.
Keywords: air oxidation, graphite, manufactured carbon, oxidation resistance, kinetic regime, activation energy, Arrhenius equation, quality control, ASTM D7542-21, standard test method, high temperature oxidation, material testing, pre-exponential factor.
Buy Documents
ASTM D7542-21 - Standard Test Method for Air Oxidation of Carbon and Graphite in the Kinetic Regime
REDLINE ASTM D7542-21 - Standard Test Method for Air Oxidation of Carbon and Graphite in the Kinetic Regime
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D7542-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Air Oxidation of Carbon and Graphite in the Kinetic Regime". This standard covers: SIGNIFICANCE AND USE 5.1 This test method can be used to measure the rate of oxidation for various grades of manufactured carbon and graphite in standard conditions, and can be used for quality control purposes. 5.2 The following conditions are standardized in this test method: size and shape of the graphite specimens; their placement in the vertical furnace with upwards air flow; the method for continuous weight variation measurement using an analytical scale with under-the-scale port; the air flow rate, which must be high enough to ensure that oxidation is not oxygen-starved at the highest temperature used; the initial and final points on the weight loss curve used for calculation of oxidation rate. 5.3 This test method also provides kinetic parameters (apparent activation energy and logarithm of pre-exponential factor) for the oxidation reaction, and a standard oxidation temperature. The results characterize the effect of temperature on oxidation rates in air, and the oxidation resistance of machined carbon or graphite specimens with standard size and shape, in the kinetic, or chemically controlled, oxidation regime. This information is useful for discrimination between material grades with different impurity levels, grain size, pore structure, degree of graphitization, or antioxidation treatments, or a combination thereof. 5.4 Accurately determined kinetic parameters, like activation energy and logarithm of pre-exponential factor, can be used for prediction of oxidation rates in air as a function of temperature in conditions similar to those of this test method. However, extrapolation of such predictions outside the temperature range where Arrhenius plots are linear (outside the kinetic or chemically controlled regime of oxidation) should be made with extreme caution. In conditions where (1) oxidation rates become controlled by a mechanism other than chemical reactions (such as in-pore diffusion or boundary transport of the oxidant gas), or (2) the oxidant supply rate is no... SCOPE 1.1 This test method recommends a standard procedure for measuring oxidation rates in air of various grades of nuclear graphite and/or manufactured carbon. Following the standard procedure recommended here, one can obtain kinetic parameters that characterize the oxidation resistance in standard conditions of tested materials and that can be used to for materials selection and qualification, and for quality control purposes in the fabrication process. 1.2 This test method covers the rate of oxidative weight loss per exposed nominal geometric surface area, or per initial weight of machined test specimens of standard size and shape, or both. The test is valid in the temperature range where the rate of air oxidation of graphite and manufactured carbon is limited by reaction kinetics. 1.3 This test method also provides a standard oxidation temperature (as defined in 3.1.7), and the kinetic parameters of the oxidation reaction, namely the apparent activation energy and the logarithm of pre-exponential factor in Arrhenius equation. The kinetic parameters of Arrhenius equation are calculated from the temperature dependence of oxidation rates measured over the temperature range where Arrhenius plots (as defined in 3.1.8) are linear, which is defined as the “kinetic” or “chemical control” oxidation regime. For typical nuclear grade graphite materials it was found that the practical range of testing temperatures is from about 500 °C to 550 °C up to about 700 °C to 750 °C. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international s...
SIGNIFICANCE AND USE 5.1 This test method can be used to measure the rate of oxidation for various grades of manufactured carbon and graphite in standard conditions, and can be used for quality control purposes. 5.2 The following conditions are standardized in this test method: size and shape of the graphite specimens; their placement in the vertical furnace with upwards air flow; the method for continuous weight variation measurement using an analytical scale with under-the-scale port; the air flow rate, which must be high enough to ensure that oxidation is not oxygen-starved at the highest temperature used; the initial and final points on the weight loss curve used for calculation of oxidation rate. 5.3 This test method also provides kinetic parameters (apparent activation energy and logarithm of pre-exponential factor) for the oxidation reaction, and a standard oxidation temperature. The results characterize the effect of temperature on oxidation rates in air, and the oxidation resistance of machined carbon or graphite specimens with standard size and shape, in the kinetic, or chemically controlled, oxidation regime. This information is useful for discrimination between material grades with different impurity levels, grain size, pore structure, degree of graphitization, or antioxidation treatments, or a combination thereof. 5.4 Accurately determined kinetic parameters, like activation energy and logarithm of pre-exponential factor, can be used for prediction of oxidation rates in air as a function of temperature in conditions similar to those of this test method. However, extrapolation of such predictions outside the temperature range where Arrhenius plots are linear (outside the kinetic or chemically controlled regime of oxidation) should be made with extreme caution. In conditions where (1) oxidation rates become controlled by a mechanism other than chemical reactions (such as in-pore diffusion or boundary transport of the oxidant gas), or (2) the oxidant supply rate is no... SCOPE 1.1 This test method recommends a standard procedure for measuring oxidation rates in air of various grades of nuclear graphite and/or manufactured carbon. Following the standard procedure recommended here, one can obtain kinetic parameters that characterize the oxidation resistance in standard conditions of tested materials and that can be used to for materials selection and qualification, and for quality control purposes in the fabrication process. 1.2 This test method covers the rate of oxidative weight loss per exposed nominal geometric surface area, or per initial weight of machined test specimens of standard size and shape, or both. The test is valid in the temperature range where the rate of air oxidation of graphite and manufactured carbon is limited by reaction kinetics. 1.3 This test method also provides a standard oxidation temperature (as defined in 3.1.7), and the kinetic parameters of the oxidation reaction, namely the apparent activation energy and the logarithm of pre-exponential factor in Arrhenius equation. The kinetic parameters of Arrhenius equation are calculated from the temperature dependence of oxidation rates measured over the temperature range where Arrhenius plots (as defined in 3.1.8) are linear, which is defined as the “kinetic” or “chemical control” oxidation regime. For typical nuclear grade graphite materials it was found that the practical range of testing temperatures is from about 500 °C to 550 °C up to about 700 °C to 750 °C. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international s...
ASTM D7542-21 is classified under the following ICS (International Classification for Standards) categories: 71.060.10 - Chemical elements. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7542-21 has the following relationships with other standards: It is inter standard links to ASTM E898-20, ASTM C559-16(2020), ASTM C559-16, ASTM E898-88(2013), ASTM E691-13, ASTM E691-11, ASTM E1970-11, ASTM C559-90(2010), ASTM E1582-10, ASTM E691-08, ASTM E1970-06, ASTM E691-05, ASTM E898-88(2005), ASTM C559-90(2005), ASTM E1582-04. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7542-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7542 − 21
Standard Test Method for
Air Oxidation of Carbon and Graphite in the Kinetic Regime
This standard is issued under the fixed designation D7542; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 1.6 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.1 This test method recommends a standard procedure for
ization established in the Decision on Principles for the
measuring oxidation rates in air of various grades of nuclear
Development of International Standards, Guides and Recom-
graphite and/or manufactured carbon. Following the standard
mendations issued by the World Trade Organization Technical
procedure recommended here, one can obtain kinetic param-
Barriers to Trade (TBT) Committee.
eters that characterize the oxidation resistance in standard
conditions of tested materials and that can be used to for
2. Referenced Documents
materials selection and qualification, and for quality control
2.1 ASTM Standards:
purposes in the fabrication process.
C559Test Method for Bulk Density by Physical Measure-
1.2 Thistestmethodcoverstherateofoxidativeweightloss
ments of Manufactured Carbon and Graphite Articles
per exposed nominal geometric surface area, or per initial
E691Practice for Conducting an Interlaboratory Study to
weight of machined test specimens of standard size and shape,
Determine the Precision of a Test Method
or both. The test is valid in the temperature range where the
E898Practice for Calibration of Non-Automatic Weighing
rate of air oxidation of graphite and manufactured carbon is
Instruments
limited by reaction kinetics.
E1582Test Method forTemperature Calibration ofThermo-
gravimetric Analyzers
1.3 This test method also provides a standard oxidation
E1970PracticeforStatisticalTreatmentofThermoanalytical
temperature (as defined in 3.1.7), and the kinetic parameters of
Data
the oxidation reaction, namely the apparent activation energy
and the logarithm of pre-exponential factor in Arrhenius
3. Terminology
equation. The kinetic parameters of Arrhenius equation are
3.1 Definitions:
calculated from the temperature dependence of oxidation rates
measuredoverthetemperaturerangewhereArrheniusplots(as 3.1.1 Definitions are ordered by oxidation rates first, fol-
lowed by activation energy as calculated from oxidation rates.
defined in 3.1.8) are linear, which is defined as the “kinetic” or
“chemical control” oxidation regime. For typical nuclear grade 3.1.2 area-normalized oxidation rate (OR ), n—rate of
a
weight loss due to oxidation of a machined test specimen at a
graphite materials it was found that the practical range of
testing temperatures is from about 500°C to 550°C up to given temperature, divided by the nominal geometric surface
area of the specimen.
about 700°C to 750°C.
3.1.2.1 Discussion—The rate of weight loss is determined
1.4 The values stated in SI units are to be regarded as
by a linear fit of the weight loss plotted against time in the
standard. No other units of measurement are included in this
range from 5% to 10% loss of original specimen weight. The
standard.
-1 -2
units of area-normalized oxidation rate, OR,aregh m .
a
1.5 This standard does not purport to address all of the
3.1.3 weight-normalized oxidation rate (OR ), n—rate of
w
safety concerns, if any, associated with its use. It is the
weightlossduetooxidationofamachinedspecimenatagiven
responsibility of the user of this standard to establish appro-
temperature, divided by the initial weight of the specimen.
priate safety, health, and environmental practices and deter-
3.1.3.1 Discussion—The rate of weight loss is determined
mine the applicability of regulatory limitations prior to use.
by a linear fit of the weight loss plotted against time in the
range from 5% to 10% loss of original specimen weight. The
units of weight-normalized oxidation rate, OR are:
w
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.F0 on Manufactured Carbon and Graphite Products. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2021. Published November 2021. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2009. Last previous edition approved in 2015 as D7542–15. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D7542-21. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7542 − 21
21 21 21
g g h or, equivalent, h (1) testing.Thelinearrateofweightlossbetween5%and10%of
@ #@ # ~ !
oxidized specimen
~ ! ~ !
the specimen’s initial weight is determined during exposure.
3.1.4 nominal geometric surface area, n—exposed area (A)
Experience has shown that this is the most linear part of the
of the test specimen determined by measuring its diameter (D)
curve because weight loss below 5% of the specimen starting
and height (H) before testing and using the formula:
weight includes an induction period where reactive surface is
A 5 2πD /41πDH (2)
created.Forweightlossesabove10%ofthespecimenstarting
weight, the sample dimensions may become significantly
The units of nominal geometric surface area are m .
distorted. The area-normalized oxidation rate (OR ) is calcu-
a
3.1.5 weight-normalized standard oxidation rate (SOR ),
w
latedbydividingtherateofweightlossbytheoriginalnominal
n—valueofweight-normalizedoxidationratecorrespondingto
-4 geometric surface area of the specimen. The result is reported
1% weight loss in 24h (equivalent to SOR = 4.17 × 10 g
w
-1 -2
-1 -1 ingh m . The weight-normalized oxidation rate (OR )is
w
g h ).
calculated by dividing the rate of weight loss by the original
3.1.6 area-normalized standard oxidation rate (SOR ), -1 -1
a
weight of the specimen. The result is reported in g g h . The
n—value of area normalized oxidation rate corresponding to
results can be used to compare the oxidation resistance of
1% weight loss in 24h. Area-normalized standard oxidation
different graphite materials and to estimate their service life at
rate, SOR ,dependsontheinitialspecimendensity.Forcarbon
a
equivalent oxidation conditions.
-3
and graphite samples (density 1.2 to 2.2 g cm ) SOR varies
a
-1 -2
4.2 In order to calculate the kinetic parameters of the
between 2 and4gh m .
oxidation reaction and the standard oxidation temperature, the
3.1.7 standard oxidation temperature (SOT),
procedure is repeated with fresh specimens for a total of four
n—temperature in degrees Celsius at which a sample would
temperatures. An Arrhenius plot is obtained as explained in
reach the standard oxidation rate, that is, it would lose by
3.1.8. Only those data points in the linear range of the
oxidation 1% of its initial weight in 24h.
Arrhenius plot should be used for calculation of slope and
3.1.7.1 Discussion—In this procedure, SOT is estimated by
intercept. If deviation from linearity of Arrhenius plots is
plotting the decimal logarithm of oxidation rate data deter-
observed at high temperatures for certain materials, the data
mined at several temperatures against the reciprocal of the
outside the linear segment should not be used, and more
absolute temperature (in Kelvin) of the measurement. The plot
oxidation rate measurements should be performed at lower
should yield a straight line. The temperature at which the line
temperatures. For typical nuclear graphite materials it was
predicts a rate corresponding to 1% weight loss in 24h
found that the practical range of testing temperatures is from
-4 -1 -1
(equivalent to SOR =4.17×10 gg h ) is the standard
w
about 500°C to 550°C up to about 700°C to 750°C.
oxidation temperature (SOT).
3.1.8 activation energy (E ), n—measure of temperature
a
5. Significance and Use
effects on the rate of oxidation in the kinetic, or chemical
5.1 This test method can be used to measure the rate of
control, regime. Activation energy is calculated from the
oxidation for various grades of manufactured carbon and
Arrhenius equation:
graphite in standard conditions, and can be used for quality
OR 5 Zexp~2E /RT! (3)
a
control purposes.
where:
5.2 The following conditions are standardized in this test
OR = oxidation rate, method: size and shape of the graphite specimens; their
-1 -1
R = 8.314 J mole K is the universal gas constant,
placement in the vertical furnace with upwards air flow; the
T = absolute temperature (in Kelvin), and
method for continuous weight variation measurement using an
Z = pre-exponential factor.
analytical scale with under-the-scale port; the air flow rate,
which must be high enough to ensure that oxidation is not
The activation energy and pre-exponential factor are calcu-
oxygen-starved at the highest temperature used; the initial and
lated from linearized form ofArrhenius equation, that is, from
final points on the weight loss curve used for calculation of
the slope and intercept of the linear plot of the logarithm of
oxidation rate.
oxidationrateversustheinverseofabsolutetemperature(1/T):
5.3 This test method also provides kinetic parameters (ap-
log ~OR! 5 log Z 2 E /~2.303RT! (4)
10 10 a
Activation energy is expressed in units of kJ/mol. Pre-
parent activation energy and logarithm of pre-exponential
exponential factor is expressed in the same units as the oxi-
factor) for the oxidation reaction, and a standard oxidation
-1 -2
dation rates, namely g h m (for Z calculated from area-
a
temperature. The results characterize the effect of temperature
-1 -1
normalized oxidation rates, OR)orgg h (for Z
a w
on oxidation rates in air, and the oxidation resistance of
calculated from weight-normalized oxidation rates, OR ).
w
machined carbon or graphite specimens with standard size and
4. Summary of Test Method shape, in the kinetic, or chemically controlled, oxidation
regime. This information is useful for discrimination between
4.1 This test method provides the rate of oxidation in air of
material grades with different impurity levels, grain size, pore
cylindrical test specimens with standard size, machined of
structure, degree of graphitization, or antioxidation treatments,
carbon and graphite. During tests, the specimens hang freely
or a combination thereof.
from a continuously recording balance in a stream of dry air
preheated at a preselected test temperature. The nominal 5.4 Accurately determined kinetic parameters, like activa-
geometrical surface area of the specimen is determined before tion energy and logarithm of pre-exponential factor, can be
D7542 − 21
FIG. 1 Oxidation Apparatus
used for prediction of oxidation rates in air as a function of 6.2 The specimen and the air supply to the furnace shall be
temperature in conditions similar to those of this test method.
free of moisture. A desiccant column shall be used on the air
However, extrapolation of such predictions outside the tem-
supply line.
perature range where Arrhenius plots are linear (outside the
kineticorchemicallycontrolledregimeofoxidation)shouldbe
7. Apparatus
made with extreme caution. In conditions where (1) oxidation
7.1 Oxidation Apparatus, Shown schematically in Fig. 1
rates become controlled by a mechanism other than chemical
and consisting of the following:
reactions (such as in-pore diffusion or boundary transport of
the oxidant gas), or (2) the oxidant supply rate is not large 7.1.1 Vertical Tube Furnace—Capable of obtaining 900°C.
enough to prevent oxidant starving conditions at high A three-zone furnace with proportional–integral–derivative
temperature, prediction of oxidation rates using kinetic param- (PID) controllers is recommended. Temperature control accu-
eters determined with this test method will produce overesti-
racyshouldbe 62°C.Thetemperatureofeachzoneshouldbe
mated results.
independently controlled by its thermocouple. A separate
sample temperature thermocouple should also be used; it is
6. Interferences
recommended that the sample temperature thermocouple is
located in the gas stream below the sample within maximum
6.1 Specimens shall not be contaminated during handling.
5mmofit.Itshouldindicatethetemperatureofthegasstream
They should be machined without oil, using diamond or
carbide tools, and handled with cotton gloves. just before the sample (sample temperature). Safety interlocks
D7542 − 21
FIG. 2 Tube Furnace
D7542 − 21
FIG. 3 Top Cover
with thermocouples placed on the outside of the pipe are
recommended for each zone.
7.1.2 Oxidation Resistant Furnace Tube—Such as Inconel
2 ⁄2in. schedule 40 pipe (7.30cm outer diameter; 6.27cm
inner diameter) should be used. Tubes of alumina or quartz
with equivalent inner diameter may also be used. It is recom-
mended that the ends extending from the furnace, especially
thetopendofthetube,arecooledbywatercirculatingthrough
copper tubing wrapped around the furnace tube (see Fig. 2).
7.1.3 Top Cover Block—Manufactured from a refractory
material, such as boron nitride, and should be used as thermal
shieldprotectionfortheanalyticalbalance(Fig.3).Alternately,
a grooved copper plate can be used, having a copper tube
threaded through the grooves for water circulation.The role of
thermalshieldistoensurethattheanalyticalbalanceplacedon
top of the vertical furnace is maintained as constant
temperature, as close as possible to room temperature, as
required for proper operation. The same effect can be obtained
by allowing sufficient air gap between the top end of the
furnace tube and the analytical scale, and by removing the hot
FIG. 4 Platinum Wire Basket
gases coming from the furnace tube through a snorkel con-
nected to the local ventilation system.
7.1.4 Platinum Wire and Platinum Basket—For holding
7.4 Nitrogen Flow Meter—0L⁄min to 10L⁄min, 65%
suspended specimen (Fig. 4).
full-scale accuracy.
7.2 Analytical Balance—With weigh-below port feature, at
7.5 Desiccator—Charged with indicating desiccant for stor-
least 200g capacity, 60.001g resolution.
age of conditioned specimens before use.
7.3 Air Flow Meter—0L⁄min to 10L⁄min, 65% full-scale
7.6 Cotton Gloves—For handling specimens.
accuracy.
8. Reagents and Materials
8.1 Alumina or Silica Beads or Spheres, –12 / +32 mesh.
InconelisatrademarkofSpecialMetalsCorporation.Thesolesourceofsupply
of the apparatus known to the committee at this time is Special Metals Corporation,
8.2 Air Supply, desiccated.
4317 Middle Settlement Rd., New Hartford, NY 13413-5392. If you are aware of
alternative suppliers, please provide this information to ASTM International
8.3 Nitrogen Supply (99.99 %), desiccated.
Headquarters.Your comments will receive careful consideration at a meeting of the
responsible technical committee, which you may attend. 8.4 Desiccant Column.
D7542 − 21
9. Hazards temperature used. If variations larger than 5 % are found, the
testtemperatureistoohighandalowertesttemperatureshould
9.1 Burns—The test involves high temperatures. Appropri-
be used.
ate steps should be taken to avoid contact with hot surfaces.
12.6 Record the weight, sample temperature, and elapsed
Guarding is recommended.
timeoftenenoughsothatchangesbetweenreadingsarealways
9.2 Fire—Hot surfaces could be a source of ignition.
less than 0.1% of the initial weight, W . An automated data
o
collection system is recommended. The frequency of data
10. Sampling and Test Specimens
collection should be adjusted depending on the anticipated
10.1 At least four test specimens with standard size and duration of test.
shapearerequired.Itisrecommendedtoprepareatotalof8or
12.7 Continue the test until the specimen has lost about
10 specimens for duplicate measurements at a minimum of
15% of its initial weight, that is, its weight becomes 0.85 W .
o
four temperatures.
Stoptheautomateddatacollectionprogram(ifitisbeingused)
and save the data.
10.2 The standard size of test specimens for the oxidation
testisacylinderwitha25.4mmdiameterand25.4mmlength.
12.8 After cooling, remove the specimen by disconnecting
Machining should be done with carbide or diamond tools. The
the wire at the balance.
machining tolerances should be 60.15mm. Surface finish is
12.9 Using fresh specimens each time, repeat steps 12.1 –
not critical.
12.8 above at least at three different test temperatures. A
10.3 Wipethespecimenswithlint-freepapertoremovedust
minimum of four different test temperatures should be used,
from machining.
typically between 500°C and 750°C, spaced at 50°C to
100°Conefromanother.Selectionoftesttemperaturesshould
10.4 Condition the specimens at 110°C to 150°C for a
be based on the oxidation resistance of the particular graphite
minimum of 3h. Remove and cool in a desiccat
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7542 − 15 D7542 − 21
Standard Test Method for
Air Oxidation of Carbon and Graphite in the Kinetic Regime
This standard is issued under the fixed designation D7542; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method recommends a standard procedure for measuring oxidation rates in air of various grades of nuclear graphite
and/or manufactured carbon. Following the standard procedure recommended here, one can obtain kinetic parameters that
characterize the oxidation resistance in standard conditions of tested materials and that can be used to for materials selection and
qualification, and for quality control purposes in the fabrication process.
1.2 This test method covers the rate of oxidative weight loss per exposed nominal geometric surface area, or per initial weight of
machined test specimens of standard size and shape, or both. The test is valid in the temperature range where the rate of air
oxidation of graphite and manufactured carbon is limited by reaction kinetics.
1.3 This test method also provides a standard oxidation temperature (as defined in 3.1.7), and the kinetic parameters of the
oxidation reaction, namely the apparent activation energy and the logarithm of pre-exponential factor in Arrhenius equation. The
kinetic parameters of Arrhenius equation are calculated from the temperature dependence of oxidation rates measured over the
temperature range where Arrhenius plots (as defined in 3.1.8) are linear, which is defined as the “kinetic” or “chemical control”
oxidation regime. For typical nuclear grade graphite materials it was found that the practical range of testing temperatures is from
about 500 °C to 550 °C up to about 700 °C to 750 °C.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C559 Test Method for Bulk Density by Physical Measurements of Manufactured Carbon and Graphite Articles
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E898 Practice for Calibration of Non-Automatic Weighing Instruments
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.F0 on Manufactured Carbon and Graphite Products.
Current edition approved Oct. 1, 2015Nov. 1, 2021. Published November 2015November 2021. Originally approved in 2009. Last previous edition approved in 20092015
as D7542 – 09.D7542 – 15. DOI: 10.1520/D7542-15.10.1520/D7542-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7542 − 21
E1582 Test Method for Temperature Calibration of Thermogravimetric Analyzers
E1970 Practice for Statistical Treatment of Thermoanalytical Data
3. Terminology
3.1 Definitions:
3.1.1 Definitions are ordered by oxidation rates first, followed by activation energy as calculated from oxidation rates.
3.1.2 area-normalized oxidation rate (OR )—), n—rate of weight loss due to oxidation of a machined test specimen at a given
a
temperature, divided by the nominal geometric surface area of the specimen.
3.1.2.1 Discussion—
The rate of weight loss is determined by a linear fit of the weight loss plotted against time in the range from 5 % to 10 % loss of
-1 -2
original specimen weight. The units of area-normalized oxidation rate, OR , are g h m .
a
3.1.3 weight-normalized oxidation rate (OR )—), n—rate of weight loss due to oxidation of a machined specimen at a given
w
temperature, divided by the initial weight of the specimen.
3.1.3.1 Discussion—
The rate of weight loss is determined by a linear fit of the weight loss plotted against time in the range from 5 % to 10 % loss of
original specimen weight. The units of weight-normalized oxidation rate, OR are:
w
21 21 21
g g h ~or, equivalent, h ! (1)
@ # @ #
~oxidized! ~specimen!
3.1.4 nominal geometric surface area—area, n—exposed area (A) of the test specimen determined by measuring its diameter (D)
and height (H) before testing and using the formula:
A 5 2πD /41πDH (2)
The units of nominal geometric surface area are m .
3.1.5 weight-normalized standard oxidation rate (SOR )—), n—value of weight-normalized oxidation rate corresponding to 1 %
w
-4 -1 -1
weight loss in 24 h (equivalent to SOR = 4.17 × 10 g g h ).
w
3.1.6 area-normalized standard oxidation rate (SOR )—), n—value of area normalized oxidation rate corresponding to 1 % weight
a
loss in 24 h. Area-normalized standard oxidation rate, SOR , depends on the initial specimen density. For carbon and graphite
a
-3 -1 -2
samples (density 1.2 to 2.2 g cm ) SOR varies between 2 and 4 g h m .
a
3.1.7 standard oxidation temperature (SOT)—(SOT), n—temperature in degrees Celsius at which a sample would reach the
standard oxidation rate, that is, it would lose by oxidation 1 % of its initial weight in 24 h.
3.1.7.1 Discussion—
In this procedure, SOT is estimated by plotting the decimal logarithm of oxidation rate data determined at several temperatures
against the reciprocal of the absolute temperature (in Kelvin) of the measurement. The plot should yield a straight line. The
-4 -1 -1
temperature at which the line predicts a rate corresponding to 1 % weight loss in 24 h (equivalent to SOR = 4.17 × 10 g g h )
w
is the standard oxidation temperature (SOT).
3.1.8 activation energy (E )—), n—measure of temperature effects on the rate of oxidation in the kinetic, or chemical control,
a
regime. Activation energy is calculated from the Arrhenius equation:
OR 5 Zexp~2E /RT! (3)
a
where:
OR = oxidation rate,
-1 -1
R = 8.314 J mole K is the universal gas constant,
T = absolute temperature (in Kelvin), and
Z = pre-exponential factor.
The activation energy and pre-exponential factor are calculated from linearized form of Arrhenius equation, that is, from the
slope and intercept of the linear plot of the logarithm of oxidation rate versus the inverse of absolute temperature (1/T):
log OR 5 log Z 2 E / 2.303 RT (4)
~ ! ~ !
10 10 a
Activation energy is expressed in units of kJ/mol. Pre-exponential factor is expressed in the same units as the oxidation rates,
D7542 − 21
-1 -2 -1 -1
namely g h m (for Z calculated from area-normalized oxidation rates, OR ) or g g h (for Z calculated from weight-
a a w
normalized oxidation rates, OR ).
w
4. Summary of Test Method
4.1 This test method provides the rate of oxidation in air of cylindrical test specimens with standard size, machined of carbon and
graphite. During tests, the specimens hang freely from a continuously recording balance in a stream of dry air preheated at a
preselected test temperature. The nominal geometrical surface area of the specimen is determined before testing. The linear rate
of weight loss between 5 % and 10 % of the specimen’s initial weight is determined during exposure. Experience has shown that
this is the most linear part of the curve because weight loss below 5 % of the specimen starting weight includes an induction period
where reactive surface is created. For weight losses above 10 % of the specimen starting weight, the sample dimensions may
become significantly distorted. The area-normalized oxidation rate (OR ) is calculated by dividing the rate of weight loss by the
a
-1 -2
original nominal geometric surface area of the specimen. The result is reported in g h m . The weight-normalized oxidation rate
-1 -1
(OR ) is calculated by dividing the rate of weight loss by the original weight of the specimen. The result is reported in g g h .
w
The results can be used to determine relative service life of samples in a series, at a preselected temperature.compare the oxidation
resistance of different graphite materials and to estimate their service life at equivalent oxidation conditions.
4.2 In order to calculate the kinetic parameters of the oxidation reaction and the standard oxidation temperature, the procedure is
repeated with fresh specimens for a total of four temperatures. An Arrhenius plot is obtained as explained in 3.1.8. Only those data
points in the linear range of the Arrhenius plot should be used for calculation of slope and intercept. If deviation from linearity
of Arrhenius plots is observed at high temperatures for certain materials, the data outside the linear segment should not be used,
and more oxidation rate measurements should be performed at lower temperatures. For typical nuclear graphite materials it was
found that the practical range of testing temperatures is from about 500 °C to 550 °C up to about 700 °C to 750 °C.
5. Significance and Use
5.1 This test method can be used to measure the rate of oxidation for various grades of manufactured carbon and graphite in
standard conditions, and can be used for quality control purposes.
5.2 The following conditions are standardized in this test method: size and shape of the graphite specimens; their placement in
the vertical furnace with upwards air flow; the method for continuous weight variation measurement using an analytical scale with
under-the-scale port; the air flow rate, which is must be high enough to ensure that oxidation is not oxygen-starved at the highest
temperature used; the initial and final points on the weight loss curve used for calculation of oxidation rate.
5.3 This test method also provides kinetic parameters (activation (apparent activation energy and logarithm of pre-exponential
factor) for the oxidation reaction, and a standard oxidation temperature. The results uniquely characterize the effect of temperature
on oxidation rates in air, and the oxidation resistance of machined carbon or graphite specimens with standard size and shape, in
the kinetic, or chemically controlled, oxidation regime. This information is useful for discrimination between material grades with
different impurity levels, grain size, pore structure, degree of graphitization, or antioxidation treatments, or a combination thereof.
5.4 Accurately determined kinetic parameters, like activation energy and logarithm of pre-exponential factor, can be used for
prediction of oxidation rates in air as a function of temperature in conditions similar to those of this test method. However,
extrapolation of such predictions outside the temperature range where Arrhenius plots are linear (outside the kinetic or chemically
controlled regime of oxidation) should be made with extreme caution. In conditions where (1) oxidation rates become controlled
by a mechanism other than chemical reactions, suchreactions (such as in-pore diffusion or boundary transport of the oxidant gas,
gas), or (2) the oxidant supply rate is not large enough to prevent oxidant starving conditions at high temperature, prediction of
oxidation rates using kinetic parameters determined with this test method will produce overestimated results.
6. Interferences
6.1 Specimens shall not be contaminated during handling. They should be machined without oil, using diamond or carbide tools,
and handled with cotton gloves.
6.2 The specimen and the air supply to the furnace shall be free of moisture. A desiccant column shall be used on the air supply
line.
D7542 − 21
FIG. 1 Oxidation Apparatus
7. Apparatus
7.1 Oxidation Apparatus, Shown schematically in Fig. 1 and consisting of the following:
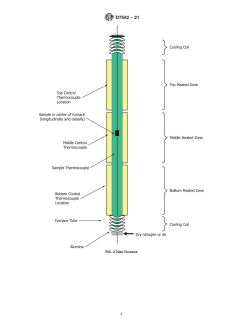
7.1.1 Vertical Tube Furnace—Capable of obtaining 900 °C. A three-zone furnace with proportional–integral–derivative (PID)
controllers is recommended. Temperature control accuracy should be 62 °C. The temperature of each zone should be
independently controlled by its thermocouple. A separate sample temperature thermocouple should also be used; it is recommended
that the sample temperature thermocouple is located in the gas stream below the sample within maximum 5 mm of it. It should
indicate the temperature of the gas stream just before the sample (sample temperature). Safety interlocks with thermocouples
placed on the outside of the pipe are recommended for each zone.
7.1.2 Oxidation Resistant Furnace Tube—Such as Inconel 2 ⁄2 in. schedule 40 pipe (7.30 cm outer diameter; 6.27 cm inner
diameter) should be used. Tubes of alumina or quartz with equivalent inner diameter may also be used. It is recommended that
the ends extending from the furnace, especially the top end of the tube, are cooled by water circulating through copper tubing
wrapped around the furnace tube (see Fig. 2).
Inconel is a trademark of Special Metals Corporation. The sole source of supply of the apparatus known to the committee at this time is Special Metals Corporation,
4317 Middle Settlement Rd., New Hartford, NY 13413-5392. If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters.
Your comments will receive careful consideration at a meeting of the responsible technical committee, which you may attend.
D7542 − 21
FIG. 2 Tube Furnace
7.1.3 Top Cover Block—Manufactured from a refractory material, such as boron nitride, and should be used as thermal shield
protection for the analytical balance (Fig. 3). Alternately, a grooved copper plate can be used, having a copper tube threaded
through the grooves for water circulation. The role of thermal shield is to ensure that the analytical balance placed on top of the
vertical furnace is maintained as constant temperature, as close as possible to room temperature, as required for proper operation.
The same effect can be obtained by allowing sufficient air gap between the top end of the furnace tube and the analytical scale,
and by removing the hot gases coming from the furnace tube through a snorkel connected to the local ventilation system.
7.1.4 Platinum Wire and Platinum Basket—For holding suspended specimen (Fig. 4).
D7542 − 21
FIG. 3 Top Cover
FIG. 4 Platinum Wire Basket
7.2 Analytical Balance—With weigh-below port feature, at least 200 g capacity, 60.001 g resolution.
7.3 Air Flow Meter—0 L ⁄min to 10 L ⁄min, 65 % full-scale accuracy.
7.4 Nitrogen Flow Meter—0 L ⁄min to 10 L ⁄min, 65 % full-scale accuracy.
7.5 Desiccator—Charged with indicating desiccant for storage of conditioned specimens before use.
7.6 Cotton Gloves—For handling specimens.
D7542 − 21
8. Reagents and Materials
8.1 Alumina or Silica Beads or Spheres, –12 / +32 mesh.
8.2 Air Supply, desiccated.
8.3 Nitrogen Supply (99.99 %), desiccated.
8.4 Desiccant Column.
9. Hazards
9.1 Burns—The test involves high temperatures. Appropriate steps should be taken to avoid contact with hot surfaces. Guarding
is recommended.
9.2 Fire—Hot surfaces could be a source of ignition.
10. Sampling and Test Specimens
10.1 At least four test specimens with standard size and shape are required. It is recommended to prepare a total of 8 or 10
specimens for duplicate measurements at a minimum of four temperatures.
10.2 The standard size of test specimens for the oxidation test is a cylinder with a 25.4 mm diameter and 25.4 mm length.
Machining should be done with carbide or diamond tools. The machining tolerances should be 60.15 mm. Surface finish is not
critical.
10.3 Wipe the specimens with lint-free paper to remove dust from machining.
10.4 Condition the specimens at 110 °C to 150 °C for a minimum of 3 h. Remove and cool in a desiccator for a minimum of
30 min. Keep specimens in desiccator until ready to perform test.
11. Calibration and Standardization
11.1 The recommended practice for calibration of temperature scale for thermogravimmetry is Practice E1582.
11.2 The recommended test method for testing top-loading, direct-reading laboratory scales and balances is Test Method E898.
12. Procedure
12.1 Measure the diameter (D) and height (H) of the specimen to the nearest 60.03 mm.
12.2 Assemble the furnace as shown in Fig. 1. Charge the bottom of the furnace with about 5 cm layer of alumina or silica beads
to act as a gas distributor.
12.3 Hang the wire basket on the weigh-below hook of the analytical balance. Ensure that the wire is in the middle of the furnace
chamber and does not touch the walls of the furnace tube. Tare the balance. Remove the wire basket and insert the specimen.
Re-hang the basket and specimen in the furnace. Weigh the specimen to the nearest 60.01 g and record weight as W.
12.4 Start flowing dry nitrogen through the tube at a flow rate of
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...