ASTM D8442-22
(Test Method)Standard Test Method for Determination of Cannabinoids in Cannabis Raw Materials and Resin Cannabis Products by Gas Chromatography and Flame Ionization Detection
Standard Test Method for Determination of Cannabinoids in Cannabis Raw Materials and Resin Cannabis Products by Gas Chromatography and Flame Ionization Detection
SIGNIFICANCE AND USE
5.1 Gas chromatography and flame ionization detection provides a rapid means to identify and quantify cannabinoids in a variety of samples of interest. This test method allows producers of cannabis products to improve and optimize the quality of their products. For example, hemp extractors can use it to determine the efficiency of extraction processes and to verify that products meet regulatory requirements, ensuring safety and quality of products.
5.2 Cannabinoids, such as CBD and THC can be monitored throughout the production process. The determination of Δ9-THC is often required for regulatory purposes and the determination of other THC isomers is often of interest. The United Nations Office on Drugs and Crime provides experimental details and guidance for use of GC to analyze cannabis related samples, including conditions suitable for decarboxylation of cannabinoid acids.3
5.3 Post-decarboxylated methodology is used. In decarboxylation, heat is used to liberate carbon dioxide from carboxylic acid cannabinoids, forming their corresponding neutral cannabinoids, for example, THC from THCA. It should be recognized that the hot temperature of the GC injection port itself is capable of effecting at least some decarboxylation (250 °C – Table 2), and many sample types, such as distillates, require no decarboxylation because it would have occurred during material processing. Therefore, some knowledge of sample properties and material processing is useful. Resulting determinations are for the total cannabinoid content of specific isomers, for example, total Δ9-THC. For those samples requiring decarboxylation, the method is validated per Practice D8282 through the use of reference materials, spike and recovery of knowns, or through comparison with LC results. For example, carrying out the decarboxylation procedure of a standard containing known amounts of CBDA and CBN should yield the correct amounts of CBD and CBN, where CBN is not significantly changed and the mass...
SCOPE
1.1 This test method covers the analysis of cannabinoids in cannabis products by gas chromatography (GC) and flame ionization detection (FID).
1.2 This test method is applicable to cannabis raw materials and resin cannabis products as defined in Guide D8245, including those from hemp. Such material includes: biomass; plant material; flowers; resins; extracts; distillates; recovered solvents; and other intermediate processing material. The applicable concentration range of analysis will vary to some extent depending on the nature of the sample, for instance measurement of delta-9-tetrahydrocannabinol (Δ9-THC) for regulatory purposes in hemp would require calibration to lower concentration levels compared to measurement of CBD in its isolate; however, in most cases, the test method is applicable to the determination of major and minor cannabinoids above about 0.1 mass% in concentration. Dilution of sample solutions is used to adjust concentrations to fall within appropriate calibration curves. Particular emphasis is placed on the determination of Δ9-THC for regulatory compliance purposes and control. This test method can measure any cannabinoid that is eluted and detected from a GC column with sufficient resolution from any interfering compounds. Typical cannabinoids of interest that can be determined by this test method are shown in Table 1. Use of an HPLC technique is recommended if individual measurement of acids, such as THCA, is required.
1.3 The test method does not purport to identify all individual cannabinoids; however, individual users can adapt this test method for specific custom analyses to meet their needs.
1.4 Units—Values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard t...
General Information
- Status
- Published
- Publication Date
- 14-Jul-2022
- Technical Committee
- D37 - Cannabis
- Drafting Committee
- D37.03 - Laboratory
Relations
- Effective Date
- 01-Oct-2023
- Refers
ASTM D4626-95(2019) - Standard Practice for Calculation of Gas Chromatographic Response Factors - Effective Date
- 01-Dec-2019
- Refers
ASTM D8282-19 - Standard Practice for Laboratory Test Method Validation and Method Development - Effective Date
- 01-Sep-2019
- Effective Date
- 01-Sep-2019
- Effective Date
- 15-Jan-2019
- Effective Date
- 01-Oct-2015
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Mar-2007
- Effective Date
- 01-Mar-2006
- Refers
ASTM D4626-95(2005) - Standard Practice for Calculation of Gas Chromatographic Response Factors - Effective Date
- 01-May-2005
- Effective Date
- 01-Feb-2005
- Effective Date
- 01-May-2004
- Effective Date
- 01-Jan-2001
- Effective Date
- 01-Jan-2001
Overview
ASTM D8442-22 is a standard test method developed by ASTM International for the determination of cannabinoids in cannabis raw materials and resin cannabis products using Gas Chromatography (GC) coupled with Flame Ionization Detection (FID). This standard provides a validated approach for fast and accurate identification and quantification of cannabinoids such as CBD, THC, and their isomers in a wide range of cannabis-derived materials, including hemp. The method is designed to support quality control, regulatory compliance, and product optimization throughout the cannabis production chain.
Key Topics
Test Method Scope:
- Applicable to a variety of cannabis materials: biomass, plant material, flowers, resins, extracts, distillates, and recovered solvents.
- Provides accurate results for major and minor cannabinoids at concentrations above ~0.1 mass%.
- Specific calibration for low levels of Δ9-tetrahydrocannabinol (Δ9-THC), essential for compliance testing, especially in hemp products.
Gas Chromatography and FID:
- Utilizes GC with FID for separation, identification, and quantification of selected cannabinoids.
- Capable of measuring cannabinoids that elute with sufficient resolution from interfering compounds.
- Decarboxylation (conversion of cannabinoid acids to their neutral forms) may occur during analysis, particularly in the GC injection port.
Sample Preparation:
- Biomass and plant materials require drying and often decarboxylation.
- Extracts and distillates may not need extra decarboxylation due to prior processing.
- Calibration is achieved with known standards; validation can use reference materials, spike/recovery, or comparisons with LC (liquid chromatography) methods.
Regulatory Relevance:
- Emphasizes determination of Δ9-THC to support compliance with legal limits on psychoactive content.
- Supports product labeling and safety requirements by verifying cannabinoid profiles.
Applications
Quality Control in Production:
- Enables producers to monitor cannabinoid content at each stage, improving product consistency and safety.
- Assists hemp extractors in evaluating extraction efficiency and consistency across batches.
Regulatory Compliance:
- Provides essential data for regulatory submissions regarding THC and other cannabinoid concentrations.
- Facilitates routine testing for legal compliance, critical in jurisdictions with stringent THC limits.
Research and Development:
- Supports method adaptation for custom analyses, accommodating emerging needs in cannabinoid profiling and product innovation.
Product Labeling and Verification:
- Supplies accurate cannabinoid concentration data for product labels and consumer safety documentation.
- Aids in verifying that products meet customer and regulatory expectations.
Related Standards
- ASTM D8245: Guide for Disposal of Resin-Containing Cannabis Raw Materials and Downstream Products
- ASTM D8270: Terminology Relating to Cannabis
- ASTM D8282: Practice for Laboratory Test Method Validation and Method Development
- ASTM D8334/D8334M: Practice for Sampling of Cannabis/Hemp Post-Harvest Batches for Laboratory Analyses
- ASTM D8375: Test Method for Determination of Cannabinoid Concentration in Dried Cannabis and Hemp Raw Materials Using Liquid Chromatography Tandem Mass Spectrometry
- ASTM D4626: Practice for Calculation of Gas Chromatographic Response Factors
Practical Value
By following ASTM D8442-22, laboratories and producers can ensure reliable, reproducible cannabinoid quantitation in a wide variety of cannabis and hemp products. This supports both product integrity and legal compliance, helping stakeholders maintain high standards of quality and safety in a rapidly evolving market.
Relevant keywords: cannabinoids analysis, cannabis testing, gas chromatography, flame ionization detection, Δ9-THC, CBD, hemp compliance, cannabis quality control, ASTM D8442-22.
Buy Documents
ASTM D8442-22 - Standard Test Method for Determination of Cannabinoids in Cannabis Raw Materials and Resin Cannabis Products by Gas Chromatography and Flame Ionization Detection
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM D8442-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Cannabinoids in Cannabis Raw Materials and Resin Cannabis Products by Gas Chromatography and Flame Ionization Detection". This standard covers: SIGNIFICANCE AND USE 5.1 Gas chromatography and flame ionization detection provides a rapid means to identify and quantify cannabinoids in a variety of samples of interest. This test method allows producers of cannabis products to improve and optimize the quality of their products. For example, hemp extractors can use it to determine the efficiency of extraction processes and to verify that products meet regulatory requirements, ensuring safety and quality of products. 5.2 Cannabinoids, such as CBD and THC can be monitored throughout the production process. The determination of Δ9-THC is often required for regulatory purposes and the determination of other THC isomers is often of interest. The United Nations Office on Drugs and Crime provides experimental details and guidance for use of GC to analyze cannabis related samples, including conditions suitable for decarboxylation of cannabinoid acids.3 5.3 Post-decarboxylated methodology is used. In decarboxylation, heat is used to liberate carbon dioxide from carboxylic acid cannabinoids, forming their corresponding neutral cannabinoids, for example, THC from THCA. It should be recognized that the hot temperature of the GC injection port itself is capable of effecting at least some decarboxylation (250 °C – Table 2), and many sample types, such as distillates, require no decarboxylation because it would have occurred during material processing. Therefore, some knowledge of sample properties and material processing is useful. Resulting determinations are for the total cannabinoid content of specific isomers, for example, total Δ9-THC. For those samples requiring decarboxylation, the method is validated per Practice D8282 through the use of reference materials, spike and recovery of knowns, or through comparison with LC results. For example, carrying out the decarboxylation procedure of a standard containing known amounts of CBDA and CBN should yield the correct amounts of CBD and CBN, where CBN is not significantly changed and the mass... SCOPE 1.1 This test method covers the analysis of cannabinoids in cannabis products by gas chromatography (GC) and flame ionization detection (FID). 1.2 This test method is applicable to cannabis raw materials and resin cannabis products as defined in Guide D8245, including those from hemp. Such material includes: biomass; plant material; flowers; resins; extracts; distillates; recovered solvents; and other intermediate processing material. The applicable concentration range of analysis will vary to some extent depending on the nature of the sample, for instance measurement of delta-9-tetrahydrocannabinol (Δ9-THC) for regulatory purposes in hemp would require calibration to lower concentration levels compared to measurement of CBD in its isolate; however, in most cases, the test method is applicable to the determination of major and minor cannabinoids above about 0.1 mass% in concentration. Dilution of sample solutions is used to adjust concentrations to fall within appropriate calibration curves. Particular emphasis is placed on the determination of Δ9-THC for regulatory compliance purposes and control. This test method can measure any cannabinoid that is eluted and detected from a GC column with sufficient resolution from any interfering compounds. Typical cannabinoids of interest that can be determined by this test method are shown in Table 1. Use of an HPLC technique is recommended if individual measurement of acids, such as THCA, is required. 1.3 The test method does not purport to identify all individual cannabinoids; however, individual users can adapt this test method for specific custom analyses to meet their needs. 1.4 Units—Values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard t...
SIGNIFICANCE AND USE 5.1 Gas chromatography and flame ionization detection provides a rapid means to identify and quantify cannabinoids in a variety of samples of interest. This test method allows producers of cannabis products to improve and optimize the quality of their products. For example, hemp extractors can use it to determine the efficiency of extraction processes and to verify that products meet regulatory requirements, ensuring safety and quality of products. 5.2 Cannabinoids, such as CBD and THC can be monitored throughout the production process. The determination of Δ9-THC is often required for regulatory purposes and the determination of other THC isomers is often of interest. The United Nations Office on Drugs and Crime provides experimental details and guidance for use of GC to analyze cannabis related samples, including conditions suitable for decarboxylation of cannabinoid acids.3 5.3 Post-decarboxylated methodology is used. In decarboxylation, heat is used to liberate carbon dioxide from carboxylic acid cannabinoids, forming their corresponding neutral cannabinoids, for example, THC from THCA. It should be recognized that the hot temperature of the GC injection port itself is capable of effecting at least some decarboxylation (250 °C – Table 2), and many sample types, such as distillates, require no decarboxylation because it would have occurred during material processing. Therefore, some knowledge of sample properties and material processing is useful. Resulting determinations are for the total cannabinoid content of specific isomers, for example, total Δ9-THC. For those samples requiring decarboxylation, the method is validated per Practice D8282 through the use of reference materials, spike and recovery of knowns, or through comparison with LC results. For example, carrying out the decarboxylation procedure of a standard containing known amounts of CBDA and CBN should yield the correct amounts of CBD and CBN, where CBN is not significantly changed and the mass... SCOPE 1.1 This test method covers the analysis of cannabinoids in cannabis products by gas chromatography (GC) and flame ionization detection (FID). 1.2 This test method is applicable to cannabis raw materials and resin cannabis products as defined in Guide D8245, including those from hemp. Such material includes: biomass; plant material; flowers; resins; extracts; distillates; recovered solvents; and other intermediate processing material. The applicable concentration range of analysis will vary to some extent depending on the nature of the sample, for instance measurement of delta-9-tetrahydrocannabinol (Δ9-THC) for regulatory purposes in hemp would require calibration to lower concentration levels compared to measurement of CBD in its isolate; however, in most cases, the test method is applicable to the determination of major and minor cannabinoids above about 0.1 mass% in concentration. Dilution of sample solutions is used to adjust concentrations to fall within appropriate calibration curves. Particular emphasis is placed on the determination of Δ9-THC for regulatory compliance purposes and control. This test method can measure any cannabinoid that is eluted and detected from a GC column with sufficient resolution from any interfering compounds. Typical cannabinoids of interest that can be determined by this test method are shown in Table 1. Use of an HPLC technique is recommended if individual measurement of acids, such as THCA, is required. 1.3 The test method does not purport to identify all individual cannabinoids; however, individual users can adapt this test method for specific custom analyses to meet their needs. 1.4 Units—Values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard t...
ASTM D8442-22 is classified under the following ICS (International Classification for Standards) categories: 11.120.99 - Other standards related to pharmaceutics; 65.020.20 - Plant growing. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8442-22 has the following relationships with other standards: It is inter standard links to ASTM D4626-23, ASTM D4626-95(2019), ASTM D8282-19, ASTM E594-96(2019), ASTM D8245-19, ASTM D4307-99(2015), ASTM E594-96(2011), ASTM D4307-99(2010), ASTM E355-96(2007), ASTM E594-96(2006), ASTM D4626-95(2005), ASTM E1510-95(2005), ASTM D4307-99(2004), ASTM E355-96(2001), ASTM E355-96. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8442-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8442 − 22
Standard Test Method for
Determination of Cannabinoids in Cannabis Raw Materials
and Resin Cannabis Products by Gas Chromatography and
Flame Ionization Detection
This standard is issued under the fixed designation D8442; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
1.1 This test method covers the analysis of cannabinoids in
1.6 This international standard was developed in accor-
cannabis products by gas chromatography (GC) and flame
dance with internationally recognized principles on standard-
ionization detection (FID).
ization established in the Decision on Principles for the
1.2 This test method is applicable to cannabis raw materials
Development of International Standards, Guides and Recom-
and resin cannabis products as defined in Guide D8245,
mendations issued by the World Trade Organization Technical
including those from hemp. Such material includes: biomass;
Barriers to Trade (TBT) Committee.
plant material; flowers; resins; extracts; distillates; recovered
2. Referenced Documents
solvents; and other intermediate processing material. The
applicable concentration range of analysis will vary to some
2.1 ASTM Standards:
extent depending on the nature of the sample, for instance
D4307Practice for Preparation of Liquid Blends for Use as
measurement of delta-9-tetrahydrocannabinol (∆ -THC) for
Analytical Standards
regulatorypurposesinhempwouldrequirecalibrationtolower
D4626Practice for Calculation of Gas Chromatographic
concentration levels compared to measurement of CBD in its
Response Factors
isolate;however,inmostcases,thetestmethodisapplicableto
D8245Guide for Disposal of Resin-Containing Cannabis
the determination of major and minor cannabinoids above
Raw Materials and Downstream Products
about0.1mass%inconcentration.Dilutionofsamplesolutions
D8270Terminology Relating to Cannabis
is used to adjust concentrations to fall within appropriate
D8282Practice for Laboratory Test Method Validation and
calibration curves. Particular emphasis is placed on the deter-
Method Development
mination of ∆ -THC for regulatory compliance purposes and
D8334/D8334MPractice for Sampling of Cannabis/Hemp
control. This test method can measure any cannabinoid that is
Post-Harvest Batches for Laboratory Analyses
eluted and detected from a GC column with sufficient resolu-
D8375Test Method for Determination of Cannabinoid Con-
tion from any interfering compounds. Typical cannabinoids of
centration in Dried Cannabis and Hemp Raw Materials
interest that can be determined by this test method are shown
using Liquid Chromatography Tandem Mass Spectrom-
in Table1. Use of an HPLC technique is recommended if
etry (LC-MS/MS)
individual measurement of acids, such as THCA, is required.
E355PracticeforGasChromatographyTermsandRelation-
1.3 The test method does not purport to identify all indi- ships
E594Practice for Testing Flame Ionization Detectors Used
vidual cannabinoids; however, individual users can adapt this
test method for specific custom analyses to meet their needs. in Gas or Supercritical Fluid Chromatography
E1510Practice for Installing Fused Silica Open Tubular
1.4 Units—ValuesstatedinSIunitsaretoberegardedasthe
Capillary Columns in Gas Chromatographs
standard. No other units of measurement are included in this
standard.
3. Terminology
1.5 This standard does not purport to address all of the
3.1 Definitions:
safety concerns, if any, associated with its use. It is the
3.1.1 Refer to Terminology D8270 for guidance on termi-
responsibility of the user of this standard to establish appro-
nology relating to cannabis.
1 2
This test method is under the jurisdiction of ASTM Committee D37 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Cannabis and is the direct responsibility of Subcommittee D37.03 on Laboratory. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
CurrenteditionapprovedJuly15,2022.PublishedOctober2022.DOI:10.1520/ Standards volume information, refer to the standard’s Document Summary page on
D8442-22. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8442 − 22
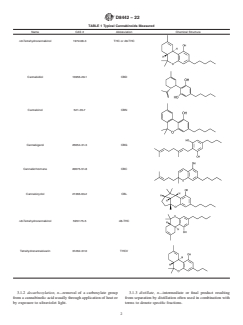
TABLE 1 Typical Cannabinoids Measured
Name CAS # Abbreviation Chemical Structure
∆9-Tetrahydrocannabinol 1972-08-3 THC or d9-THC
Cannabidiol 13956-29-1 CBD
Cannabinol 521-35-7 CBN
Cannabigerol 25654-31-3 CBG
Cannabichromene 20675-51-8 CBC
Cannabicyclol 21366-63-2 CBL
∆8-Tetrahydrocannabinol 5957-75-5 d8-THC
Tetrahydrocannabivarin 31262-37-0 THCV
3.1.2 decarboxylation, n—removal of a carboxylate group 3.1.3 distillate, n—intermediate or final product resulting
fromacannabinolicacidusuallythroughapplicationofheator from separation by distillation often used in combination with
by exposure to ultraviolet light. terms to denote specific fractions.
D8442 − 22
TABLE 1 Continued
Name CAS # Abbreviation Chemical Structure
Cannabidivarin 24274-48-4 CBDV
Cannabicitran 31508-71-1 CBT
3.2 Definitions: with authentic standards. The FID produces a highly sensitive
3.2.1 CBC—cannabichromene and linear response that is validated by the guidelines of
Practice D8282.
3.2.2 CBD—cannabidiol
3.2.3 CBDV—cannabidivarin
5. Significance and Use
3.2.4 CBG—cannabigerol
5.1 Gas chromatography and flame ionization detection
3.2.5 CBL—cannabicyclol providesarapidmeanstoidentifyandquantifycannabinoidsin
a variety of samples of interest. This test method allows
3.2.6 CBN—cannabinol
producers of cannabis products to improve and optimize the
3.2.7 CBT—cannabicitran
qualityoftheirproducts.Forexample,hempextractorscanuse
3.2.8 CRM—certified reference material
it to determine the efficiency of extraction processes and to
verify that products meet regulatory requirements, ensuring
3.2.9 FID—flame ionization detector
safety and quality of products.
3.2.10 GC—gas chromatography
5.2 Cannabinoids, such as CBD andTHC can be monitored
3.2.11 HPLC—high performance liquid chromatography
throughout the production process. The determination of
3.2.12 ILS—interlaboratory study
∆ -THC is often required for regulatory purposes and the
3.2.13 LC—liquid chromatography
determination of other THC isomers is often of interest. The
3.2.14 PPE—personal protective equipment United Nations Office on Drugs and Crime provides experi-
mental details and guidance for use of GC to analyze cannabis
3.2.15 PTFE—polytetrafluoroethylene
related samples, including conditions suitable for decarboxy-
3.2.16 PTV—programmed temperature vaporization
lation of cannabinoid acids.
3.2.17 THC—tetrahydrocannabinol
5.3 Post-decarboxylated methodology is used. In
3.2.18 THCV—tetrahydrocannabivarin
decarboxylation, heat is used to liberate carbon dioxide from
3.2.19 ∆ -THC—delta-9-tetrahydrocannabinol
carboxylic acid cannabinoids, forming their corresponding
neutralcannabinoids,forexample,THCfromTHCA.Itshould
3.2.20 ∆ -THC—delta-8-tetrahydrocannabinol
berecognizedthatthehottemperatureoftheGCinjectionport
3.2.21 WCOT, n—wall-coated open tubular
itself is capable of effecting at least some decarboxylation
(250°C – Table 2), and many sample types, such as distillates,
4. Summary of Test Method
require no decarboxylation because it would have occurred
4.1 The sample is analyzed by GC and FID. Standards are
during material processing. Therefore, some knowledge of
used to cover the desired concentration range of the analysis.
sample properties and material processing is useful. Resulting
Cannabinoid peaks are identified by means of their retention
determinations are for the total cannabinoid content of specific
times. Generally, a known amount of sample is weighed and 9
isomers, for example, total∆ -THC. For those samples requir-
diluted with a solvent. Many samples of interest, for example,
ing decarboxylation, the method is validated per Practice
distillates, require little sample preparation. Biomass samples
D8282 through the use of reference materials, spike and
shall be dried and decarboxylated separately before their
recovery of knowns, or through comparison with LC results.
extraction, dilution, and analysis.
4.2 FID—Unknown peaks may be quantified using the
Recommended Methods for the Identification and Analysis of Cannabis and
average FID response factor for calibrated cannabinoids,
Cannabis Products, MANUAL FOR USE BY NATIONAL DRUG ANALYSIS
however, it is desirable to identify and calibrate components LABORATORIES, United Nations, New York, 2009.
D8442 − 22
TABLE 2 Typical GC Conditions
7.1.4 Column—The test method uses a WCOT (capillary)
Instrument GC equipped with a split/splitless injection port column. Columns and conditions used must provide adequate
Column Capillary column: 15 m, 0.25 mm inside diameter,
separation of the cannabinoids of interest. Fused silica, glass,
0.25 µm, Rxi-35Sil MS
and metal capillaries of 10m to 60 m lengths with 0.25mm,
Flow conditions 1 mL/min hydrogen carrier (constant flow)
Inlet 260 °C
0.32mm,or0.53mminternaldiametersandbondedstationary
Detector FID
phases more polar than 100% dimethyl-polysiloxane and with
FID Temperature: 350 °C
film thicknesses of 0.1µm to 0.5µm have been successfully
40 mL/min H2, 400 mL/min Air
Oven 80 °C (hold 1 min) to 320 °C at 20 °C ⁄min (hold 3
used. An example chromatogram showing elution order and
min)
baseline separation of several typical cannabinoids of interest
Sample injection 1 µL, split 10:1
using the conditions of Table 1 is illustrated in Fig. 1.Itisup
totheusertovalidatethesuitabilityofalternativecolumnsand
conditions.
7.1.5 Microsyringe—A microsyringe with a 23 gauge or
For example, carrying out the decarboxylation procedure of a
smaller stainless-steel needle is used for sample introduction.
standardcontainingknownamountsofCBDAandCBNshould
Syringes of 0.1µL to 10µL capacity are commercially avail-
yieldthecorrectamountsofCBDandCBN,whereCBNisnot
able. Automatic syringe injection is recommended. Other
significantly changed and the mass of CBD formed from
sample introduction devices are allowed provided that repeat-
CBDA should be 0.877 × CBDA. The same ratio applies to
ableinjectionareasoflessthan10%relativestandarddeviation
THC and THCA. For other cannabinoids of interest, the
are achieved.
conversion factor is derived from the ratio of formula masses
of the neutral to that of its acid. If the observed conversion
7.2 Data Acquisition System—Use of an electronic integrat-
deviates significantly from expected results, then corrective
ingdeviceorcomputerismandatory.Thedeviceshallhavethe
action is necessary.
following capabilities: (1) graphic presentation of the chro-
5.4 As an aid, users are referred to other methods for
matogram; (2) digital display of chromatographic peak areas;
determination of individual cannabinoids and their acids, for
(3) measurement of area and retention times; and (4) calcula-
example, Test Method D8375.
tion and use of response factors in accordance with Practice
D4626, for example, external standardization.
NOTE 1—Other ASTM standards are in development.
7.3 Analytical balance capable of meeting the requirements
6. Interferences
of Practice D4307, for example, able to measure mass to a
6.1 Coelution of cannabinoids with other species, if
precision of 0.0001g.
occurring, will result in inaccurate determinations. Cannabi-
7.4 Grinder/homogenizer for biomass samples; cryogenic
noids in cannabis and minimally processed products are
grinder optional.
generally amenable to GC analysis, however, if validation
indicates inaccurate recovery of any cannabinoids of interest,
7.5 Laboratory oven, able to minimally reach and maintain
thencorrectiveactionmustbetaken,forexample,switchingto
a temperature of 110°C.
a higher resolution column. More sophisticated methods, such
7.6 Hot plate optional.
as Test Method D8375, shall be used if greater selectivity,
specificity, and the need for determination of neutrals and
7.7 Vortex mixer optional.
cannabinoid acids individually is needed.
7.8 Centrifuge optional.
7. Apparatus
7.9 Solvent dispenser optional.
7.1 Chromatograph—Use a GC that has the following
7.10 Dispensing pipettes optional.
performance characteristics (see Practice E355):
7.1.1 Column Temperature Programmer—The gas chro-
7.11 Cartridge or syringe filters, 0.45µm or smaller, PTFE
matograph shall be capable of linear programmed temperature
membrane.
operationoverarangesufficienttoelutethelastcompoundsof
7.12 Centrifuge tubes optional.
interest within a suitable analysis time. The programming rate
shall be sufficiently reproducible to obtain a retention time
7.13 Glass vials.
repeatability of 0.01 min.
7.14 Class A volumetric flasks.
7.1.2 Inlet—A split/splitless inlet with liner optimized for
cannabis analysis is recommended. PTV and programmable
7.15 Forceps optio
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...