ASTM E928-19
(Test Method)Standard Test Method for Purity by Differential Scanning Calorimetry
Standard Test Method for Purity by Differential Scanning Calorimetry
SIGNIFICANCE AND USE
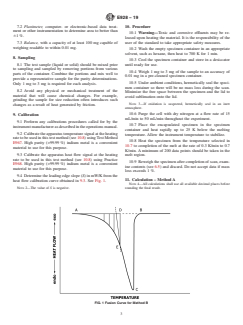
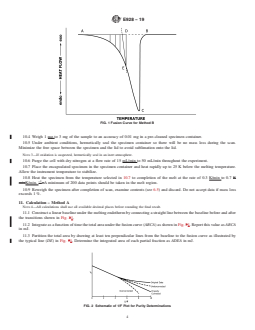
5.1 The melting temperature range of a compound broadens as the impurity level rises. This phenomenon is described approximately by the van’t Hoff equation for melting point depressions. Measuring and recording the instantaneous heat flow into the specimen as a function of temperature during such a melting process is a practical way for the generation of data suitable for analysis by the van’t Hoff equation.
5.2 The results obtained include: sample purity (expressed as mole percent); enthalpy of fusion (expressed as J/mol); and the melting temperature (expressed in Kelvin) of the pure form of the major component.
5.3 Generally, the repeatability of this test method decreases as the purity level decreases. This test method is ordinarily considered unreliable when the purity level of the major component of the mixture is less than 98.5 mol % or when the incremental enthalpy correction (c) exceeds 20 % of the original detected enthalpy of fusion.
5.4 This test method is used for quality control, specification acceptance, and research.
SCOPE
1.1 This test method describes the determination of purity of materials greater than 98.5 mole percent purity using differential scanning calorimetry and the van’t Hoff equation.
1.2 This test method is applicable to thermally stable compounds with well-defined melting temperatures.
1.3 Determination of purity by this test method is only applicable when the impurity dissolves in the melt and is insoluble in the crystal.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2019
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.01 - Calorimetry and Mass Loss
Relations
- Effective Date
- 01-May-2019
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Sep-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Mar-2013
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Aug-2011
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-Apr-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Aug-2009
- Effective Date
- 01-Sep-2008
- Effective Date
- 01-Sep-2008
- Effective Date
- 01-Sep-2008
Overview
ASTM E928-19 is the internationally recognized standard test method for determining the purity of high-purity compounds (greater than 98.5 mol %) using Differential Scanning Calorimetry (DSC). Developed by ASTM International, this method leverages the van’t Hoff equation to relate melting point depression to impurity concentration. By capturing the heat flow as a function of temperature during the compound’s melting process, the method provides reliable quantitative data for key properties including purity (mole percent), enthalpy of fusion (J/mol), and the melting temperature (K) of the sample's main component.
This standard is widely utilized for quality control, specification acceptance, and research purposes where precise purity assessment is critical, such as in pharmaceutical, chemical, and materials industries.
Key Topics
- Differential Scanning Calorimetry (DSC): Measures the heat flow into a sample during a controlled temperature program, identifying transitions like melting.
- van’t Hoff Equation: Relates the depression and broadening of melting temperature to the quantity of impurity, allowing calculation of sample purity.
- Applicable Materials: Only suitable for thermally stable compounds with well-defined melting points; impurities must dissolve in the melted phase and not in the solid.
- Result Parameters:
- Purity (mole percent)
- Enthalpy of fusion (J/mol)
- Melting temperature of the pure main component (K)
- Limitations:
- Not reliable below 98.5 mol % purity or if the enthalpy correction exceeds 20%
- Not specific to the type of impurity, only quantifies total impurity content
Applications
The ASTM E928-19 test method is a vital tool in industries and laboratories that require accurate purity determination for high-purity materials. Key applications include:
- Quality Control: Ensures that batches of chemicals or pharmaceuticals meet specified purity thresholds before distribution.
- Product Specification and Acceptance: Validates that supplied or received materials conform to required standards for purity and melting behavior.
- Research and Development: Assesses the effectiveness of purification processes or verifies the behavior of new materials under controlled thermal conditions.
- Reference Material Characterization: Establishes benchmark data for the purity and thermal properties of standard reference materials.
By using DSC and the van’t Hoff equation, manufacturers and researchers can rapidly assess and document the quality of their high-purity compounds, supporting regulatory compliance and market requirements.
Related Standards
Several ASTM standards complement ASTM E928-19 in the field of thermal analysis and calorimetry:
- ASTM E473: Terminology Relating to Thermal Analysis and Rheology
- ASTM E793: Test Method for Enthalpies of Fusion and Crystallization by Differential Scanning Calorimetry
- ASTM E794: Test Method for Melting and Crystallization Temperatures by Thermal Analysis
- ASTM E967: Test Method for Temperature Calibration of Differential Scanning Calorimeters
- ASTM E968: Practice for Heat Flow Calibration of Differential Scanning Calorimeters
- ASTM E1970: Practice for Statistical Treatment of Thermoanalytical Data
- ASTM E2161: Terminology Relating to Performance Validation in Thermal Analysis and Rheology
These related standards help ensure consistency, accuracy, and reproducibility across all thermal analysis procedures and interpretations.
Keywords: ASTM E928-19, differential scanning calorimetry, DSC, purity determination, van’t Hoff equation, melting point depression, enthalpy of fusion, quality control, thermal analysis, high-purity materials, reference materials
Buy Documents
ASTM E928-19 - Standard Test Method for Purity by Differential Scanning Calorimetry
REDLINE ASTM E928-19 - Standard Test Method for Purity by Differential Scanning Calorimetry
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E928-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Purity by Differential Scanning Calorimetry". This standard covers: SIGNIFICANCE AND USE 5.1 The melting temperature range of a compound broadens as the impurity level rises. This phenomenon is described approximately by the van’t Hoff equation for melting point depressions. Measuring and recording the instantaneous heat flow into the specimen as a function of temperature during such a melting process is a practical way for the generation of data suitable for analysis by the van’t Hoff equation. 5.2 The results obtained include: sample purity (expressed as mole percent); enthalpy of fusion (expressed as J/mol); and the melting temperature (expressed in Kelvin) of the pure form of the major component. 5.3 Generally, the repeatability of this test method decreases as the purity level decreases. This test method is ordinarily considered unreliable when the purity level of the major component of the mixture is less than 98.5 mol % or when the incremental enthalpy correction (c) exceeds 20 % of the original detected enthalpy of fusion. 5.4 This test method is used for quality control, specification acceptance, and research. SCOPE 1.1 This test method describes the determination of purity of materials greater than 98.5 mole percent purity using differential scanning calorimetry and the van’t Hoff equation. 1.2 This test method is applicable to thermally stable compounds with well-defined melting temperatures. 1.3 Determination of purity by this test method is only applicable when the impurity dissolves in the melt and is insoluble in the crystal. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The melting temperature range of a compound broadens as the impurity level rises. This phenomenon is described approximately by the van’t Hoff equation for melting point depressions. Measuring and recording the instantaneous heat flow into the specimen as a function of temperature during such a melting process is a practical way for the generation of data suitable for analysis by the van’t Hoff equation. 5.2 The results obtained include: sample purity (expressed as mole percent); enthalpy of fusion (expressed as J/mol); and the melting temperature (expressed in Kelvin) of the pure form of the major component. 5.3 Generally, the repeatability of this test method decreases as the purity level decreases. This test method is ordinarily considered unreliable when the purity level of the major component of the mixture is less than 98.5 mol % or when the incremental enthalpy correction (c) exceeds 20 % of the original detected enthalpy of fusion. 5.4 This test method is used for quality control, specification acceptance, and research. SCOPE 1.1 This test method describes the determination of purity of materials greater than 98.5 mole percent purity using differential scanning calorimetry and the van’t Hoff equation. 1.2 This test method is applicable to thermally stable compounds with well-defined melting temperatures. 1.3 Determination of purity by this test method is only applicable when the impurity dissolves in the melt and is insoluble in the crystal. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E928-19 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E928-19 has the following relationships with other standards: It is inter standard links to ASTM E928-08(2014), ASTM E473-23b, ASTM E2161-15, ASTM E473-14, ASTM E2161-13, ASTM E793-06(2012), ASTM E794-06(2012), ASTM E1970-11, ASTM E473-11a, ASTM E473-11, ASTM E473-10, ASTM E473-09, ASTM E968-02(2008), ASTM E473-08, ASTM E967-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E928-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E928 − 19
Standard Test Method for
Purity by Differential Scanning Calorimetry
This standard is issued under the fixed designation E928; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* E967Test Method for Temperature Calibration of Differen-
tial Scanning Calorimeters and Differential ThermalAna-
1.1 Thistestmethoddescribesthedeterminationofpurityof
lyzers
materials greater than 98.5 mole percent purity using differen-
E968Practice for Heat Flow Calibration of Differential
tial scanning calorimetry and the van’t Hoff equation.
Scanning Calorimeters
1.2 This test method is applicable to thermally stable
E1970PracticeforStatisticalTreatmentofThermoanalytical
compounds with well-defined melting temperatures.
Data
E2161Terminology Relating to Performance Validation in
1.3 Determination of purity by this test method is only
applicable when the impurity dissolves in the melt and is Thermal Analysis and Rheology
insoluble in the crystal.
3. Terminology
1.4 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
3.1 Definitions—The definitions relating to this standard
standard.
appear in Terminologies E473 and E2161, including baseline,
calibration, detection limit, differential scanning calorimeter,
1.5 This standard does not purport to address all of the
endotherm, extrapolated onset, first-deviation-from-baseline,
safety concerns, if any, associated with its use. It is the
peak quantitation limit, repeatability, reproducibility, and val-
responsibility of the user of this standard to establish appro-
ley.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
4. Summary of Test Method
1.6 This international standard was developed in accor-
dance with internationally recognized principles on standard-
4.1 This test method is based upon the van’t Hoff equation
ization established in the Decision on Principles for the 3
(1):
Development of International Standards, Guides and Recom-
T 5 T 2 RT χ / HF (1)
~ ! ~ !
s o o
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
where:
T = specimen temperature, K,
s
2. Referenced Documents
T = melting temperature of 100% pure material, K,
o
−1 −1
R = gas constant (= 8.314 J mol K ),
2.1 ASTM Standards:
χ = mole fraction of impurity,
E473Terminology Relating to Thermal Analysis and Rhe-
−1
H = heat of fusion, J mol , and
ology
F = fraction melted.
E793Test Method for Enthalpies of Fusion and Crystalliza-
tion by Differential Scanning Calorimetry
4.2 This test method consists of melting the test specimen
E794TestMethodforMeltingAndCrystallizationTempera-
that is subjected to a temperature-controlled program while
tures By Thermal Analysis
recording the heat flow into the specimen as a function of
temperature.Theresultingmeltingendothermareaismeasured
to yield the enthalpy of fusion, H.The melting endotherm area
is then partitioned into a series of fractional areas (about ten,
ThistestmethodisunderthejurisdictionofASTMCommitteeE37onThermal
Measurements and is the direct responsibility of Subcommittee E37.01 on Calo-
comprisingthefirst10to50%ofthetotalarea).Thefractional
rimetry and Mass Loss.
area, divided by the total area, yields the fraction melted, F.
Current edition approved May 1, 2019. Published May 2019. Originally
Each fractional area is assigned a temperature, T .
approved in 1983. Last previous edition approved in 2014 as E928–08 (2014). s
DOI: 10.1520/E0928-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on The boldface numbers in parentheses refer to a list of references at the end of
the ASTM website. this standard.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E928 − 19
4.3 Eq1hastheformofY=mX+bwhereY= T,X=1/F, 6. Interferences
s
m=−(RT χ)/ H, and b = T . A plot of Y versus X should
o o
6.1 This test method is nonspecific. Many impurities may
produce a straight line with slope m and intercept b.
causethemeltingtemperaturebroadening.Thus,itisnotuseful
in identifying the nature of the impurity or impurities but only
4.4 In practice, however, the resultant plot of T versus 1 /F
s
the total mol percent of impurity present.
is seldom a straight line. To linearize the plot, an incremental
amount of area is added to the total area and to each fractional
6.2 The van’t Hoff theory assumes the following:
area to produce a revised value for F. The process of 6.2.1 The impurities dissolve in the melt of the major
incremental addition of area is continued until a straight line is constituent forming a solution approximately described by
obtained. ideal solution theory;
6.2.2 Thesolubilityoftheimpurityinthesolidofthemajor
F 5 A 1c / A 1c (2)
~ ! ~ !
part total
constituent is negligible; and
where:
6.2.3 The major constituent displays a single well-defined
meltingendotherminthetemperaturerangeofinterest.Micro-
A = area of fraction melted, mJ,
part
A = total area, mJ, and scopic investigations of the melt and the solid may help to
total
c = incremental area, mJ.
establish whether or not solid or liquid solutions have been
formed.
NOTE 1—The best fit straight line may be determined by the least
squares method. (See Practice E1970.)
6.2.4 The solute and solvent are close in molecular size.
6.3 In some cases the sample may react with air during the
4.5 The values of mole fraction impurity χ and melting
temperature cycle, causing an incorrect transition to be mea-
temperatureofthe100%purematerialT aredeterminedfrom
o
sured. Where it has been shown that this effect is present,
the slope m and intercept b of the resultant straight line. This
provision shall be made for sealing the specimen and running
is Method A.
the test under an inert gas blanket. Since some materials
4.6 An alternative form of the van’t Hoff equation is given
degrade near the melting region, carefully distinguish between
by (2):
degradation and transition. See Appendix X1.
A 52c1 T c 2RT χ m/M /T 1T A /T (3)
@ #
part o o s o part s
6.4 Since milligram quantities of sample are used, ensure
that samples are homogeneous and representative.
where:
m = mass of the sample, mg, and 6.5 Sublimation or decomposition will lead to a different
−1
M = molecular weight, g mol . heatconsumptionand,perhaps,achangeincompositionofthe
specimen. The specimen holder should be examined after the
4.7 Eq 3 has the form of Y = αW+ βX+ γ Z where Y =
measurement for crystals not part of the resolidified melt.
A , α=−c,W=1, β=[T c−RT χm/M],X=1/ T ,
part o o s
γ = T ,andZ=A / T . Eq 3 may be evaluated by multiple
o part s 7. Apparatus
linear regression and χ and T determined form the resultant
o
7.1 The essential equipment required to provide the mini-
values of α, β and γ. This is Method B.
mum instrument capability for this test method includes:
7.1.1 Differential Scanning Calorimeter (DSC), consisting
5. Significance and Use
of:
7.1.1.1 DSC Test Chamber, composed of a furnace(s) to
5.1 Themeltingtemperaturerangeofacompoundbroadens
provide uniform controlled heating of a specimen and refer-
as the impurity level rises. This phenomenon is described
ence to a constant temperature or at a constant rate within the
approximately by the van’t Hoff equation for melting point
applicabletemperaturerangeofthistestmethod;atemperature
depressions. Measuring and recording the instantaneous heat
sensor to provide an indication of the specimen temperature
flowintothespecimenasafunctionoftemperatureduringsuch
readable to 60.1 K; a differential sensor to detect a heat flow
a melting process is a practical way for the generation of data
differencebetweenthespecimenandreferenceequivalentto10
suitable for analysis by the van’t Hoff equation.
µW; and a means of sustaining a test chamber environment of
5.2 The results obtained include: sample purity (expressed
N at a purge rate of 15 mL/min to 50 mL/min 6 5 mL/min.
as mole percent); enthalpy of fusion (expressed as J/mol); and
7.1.1.2 Temperature Controller, capable of executing a spe-
themeltingtemperature(expressedinKelvin)ofthepureform
cific temperature program by operating the furnace(s) between
of the major component.
selected temperature limits at a rate of temperature change of
0.3 K/min to 0.7 K/min constant to 60.01 K/min.
5.3 Generally,therepeatabilityofthistestmethoddecreases
7.1.1.3 Data Collection Device, to provide a means of
as the purity level decreases. This test method is ordinarily
acquiring, storing, and displaying measured or calculated
considered unreliable when the purity level of the major
signals,orboth.TheminimumoutputsignalsrequiredforDSC
component of the mixture is less than 98.5 mol% or when the
are heat flow, temperature, and time.
incremental enthalpy correction (c) exceeds 20% of the
7.1.2 Containers,thatareinerttothespecimen,andthatare
original detected enthalpy of fusion.
ofsuitablestructuralshapeandintegrityforuseintheDSCtest
5.4 This test method is used for quality control, specifica-
chamber, made of materials of high thermal conductivity, such
tion acceptance, and research. as aluminum.
E928 − 19
7.2 Planimeter, computer- or electronic-based data treat- 10. Procedure
ment or other instrumentation to determine area to better than
10.1 Warning—Toxic and corrosive effluents may be re-
61%.
leased upon heating the material. It is the responsibility of the
user of the standard to take appropriate safety measures.
7.3 Balance, with a capacity of at least 100 mg capable of
weighing readable to within 0.01 mg.
10.2 Wash the empty specimen container in an appropriate
solvent, such as hexane, then heat to 700 K for 1 min.
8. Sampling
10.3 Cool the specimen container and store in a desiccator
8.1 The test sample (liquid or solid) should be mixed prior
until ready for use.
to sampling and sampled by removing portions from various
10.4 Weigh 1 mg to 3 mg of the sample to an accuracy of
parts of the container. Combine the portions and mix well to
0.01 mg in a pre-cleaned specimen container.
provide a representative sample for the purity determinations.
10.5 Under ambient conditions, hermetically seal the speci-
Only 1 mg to 3 mg is required for each analysis.
men container so there will be no mass loss during the scan.
8.2 Avoid any physical or mechanical treatment of the
Minimize the free space between the specimen and the lid to
material that will cause chemical changes. For example,
avoid sublimation onto the lid.
grinding the sample for size reduction often introduces such
NOTE 3—If oxidation is suspected, hermetically seal in an inert
changes as a result of heat generated by friction.
atmosphere.
10.6 Purge the cell with dry nitrogen at a flow rate of 15
9. Calibration
mL/min to 50 mL/min throughout the experiment.
9.1 Perform any calibrations procedures called for by the
10.7 Place the encapsulated specimen in the specimen
instrumentmanufacturerasdescribedintheoperati
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E928 − 08 (Reapproved 2014) E928 − 19
Standard Test Method for
Purity by Differential Scanning Calorimetry
This standard is issued under the fixed designation E928; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method describes the determination of purity of materials greater than 98.5 mole percent purity using differential
scanning calorimetry and the van’t Hoff equation.
1.2 This test method is applicable to thermally stable compounds with well-defined melting temperatures.
1.3 Determination of purity by this test method is only applicable when the impurity dissolves in the melt and is insoluble in
the crystal.
1.4 There is no ISO method equivalent to this test method.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E473 Terminology Relating to Thermal Analysis and Rheology
E793 Test Method for Enthalpies of Fusion and Crystallization by Differential Scanning Calorimetry
E794 Test Method for Melting And Crystallization Temperatures By Thermal Analysis
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E968 Practice for Heat Flow Calibration of Differential Scanning Calorimeters
E1970 Practice for Statistical Treatment of Thermoanalytical Data
E2161 Terminology Relating to Performance Validation in Thermal Analysis and Rheology
3. Terminology
3.1 Definitions—The definitions relating to thermal analysis appearing in Terminologythis standard appear in Terminologies
E473 shalland E2161be considered, including applicable to this test method. baseline, calibration, detection limit, differential
scanning calorimeter, endotherm, extrapolated onset, first-deviation-from-baseline, peak quantitation limit, repeatability,
reproducibility, and valley.
4. Summary of Test Method
4.1 This test method is based upon the van’tvan’t Hoff equation:equation (1):
T 5 T 2 RT χ / H F (1)
~ ! ~ !
s o o
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.01 on Calorimetry
and Mass Loss.
Current edition approved Aug. 15, 2014May 1, 2019. Published September 2014May 2019. Originally approved in 1983. Last previous edition approved in 20082014 as
E928 – 08.E928 – 08 (2014). DOI: 10.1520/E0928-08R14.10.1520/E0928-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
Brennan, W. P., DiVito, M. P., Fynas, R. L., Gray, A. P., “An Overview of the Calorimetric Purity Measurement”, in Purity Determinations by Thermal Methods, R. L.
Blaine and C. K. Schoff (Eds.), Special Technical Publication 838, American Society for Testing and Materials, West Conshohocken, PA, 1984, pp. 5–15.The boldface numbers
in parentheses refer to a list of references at the end of this standard.
Widman, G., Scherrer, O., “A New Program for DSC Purity Analysis”, Journal of Thermal Analysis, 371987, pp. 1957–1964.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E928 − 19
where:
T = specimen temperature, K,
s
T = melting temperature of 100 % pure material, K,
o
−1 − 1
R = gas constant (= 8.314 J mol K ),
−1 −1
R = gas constant (= 8.314 J mol K ),
χ = mole fraction of impurity,
− 1
H = heat of fusion, J mol , and
−1
H = heat of fusion, J mol , and
F = fraction melted.
4.2 This test method consists of melting the test specimen that is subjected to a temperature-controlled program while recording
the heat flow into the specimen as a function of temperature. The resulting melting endotherm area is measured to yield the
enthalpy of fusion, H. The melting endotherm area is then partitioned into a series of fractional areas (about ten, comprising the
first 10 to 50 % of the total area). The fractional area, divided by the total area, yields the fraction melted, F. Each fractional area
is assigned a temperature, T .
s
4.3 Eq 1 has the form of Y = mX +b where Y = T , X = 1/F, m = −(R T χ) / H, and b = T . A plot of Y versus X should produce
s o o
a straight line with slope m and intercept b.
4.4 In practice, however, the resultant plot of T versus 1 /F is seldom a straight line. To linearize the plot, an incremental amount
s
of area is added to the total area and to each fractional area to produce a revised value for F. The process of incremental addition
of area is continued until a straight line is obtained.
F 5 A 1c / A 1c (2)
~ ! ~ !
part total
where:
A = area of fraction melted, mJ,
part
A = total area, mJ, and
total
c = incremental area, mJ.
NOTE 1—The best fit straight line may be determined by the least squares method. See(See Practice E1970.)
4.5 The values of mole fraction impurity χ and melting temperature of the 100 % pure material T are determined from the slope
o
m and intercept b of the resultant straight line. This is Method A.
4.6 An alternative form of the van’tvan’t Hoff equation is given by:by (2):
A 52c1 T c 2 R T χ m/M /T 1T A /T (3)
@ #
part o o s o part s
where:
m = mass of the sample, mg, and
−1
M = molecular weight, g mol .
4.7 Eq 3 has the form of Y = α W + β X + γ Z where Y = A , α = −c, W = 1, β = [T c − R T χ m / M], X = 1 / T , γ =
part o o s
T ,and Z = A / T .Eq 3 may be evaluated by multiple linear regression and χ and T determined form the resultant values of
o part s o
α, β and γ. This is Method B.
5. Significance and Use
5.1 The melting temperature range of a compound broadens as the impurity level rises. This phenomenon is described
approximately by the van’t Hoff equation for melting point depressions. Measuring and recording the instantaneous heat flow into
the specimen as a function of temperature during such a melting process is a practical way for the generation of data suitable for
analysis by the van’t Hoff equation.
5.2 The results obtained include: sample purity (expressed as mole percent); enthalpy of fusion (expressed as joules per mole);
J/mol); and the melting temperature (expressed in Kelvin) of the pure form of the major component.
5.3 Generally, the repeatability of this test method decreases as the purity level decreases. This test method is ordinarily
considered unreliable when the purity level of the major component of the mixture is less than 98.5 mol % or when the incremental
enthalpy correction (c) exceeds 20 % of the original detected enthalpy of fusion.
5.4 This test method is used for quality control, specification acceptance, and research.
6. Interferences
6.1 This test method is nonspecific. Many impurities may cause the melting temperature broadening. Thus, it is not useful in
identifying the nature of the impurity or impurities but only the total mol percent of impurity present.
Journal of Research of the National Bureau of Standards, Vol 92, p. 85.
E928 − 19
6.2 The van’t Hoff theory assumes the following:
6.2.1 The impurities dissolve in the melt of the major constituent forming a solution approximately described by ideal solution
theory;
6.2.2 The solubility of the impurity in the solid of the major constituent is negligible; and
6.2.3 The major constituent displays a single well-defined melting endotherm in the temperature range of interest. Microscopic
investigations of the melt and the solid may help to establish whether or not solid or liquid solutions have been formed.
6.2.4 The solute and solvent are close in molecular size.
6.3 In some cases the sample may react with air during the temperature cycle, causing an incorrect transition to be measured.
Where it has been shown that this effect is present, provision shall be made for sealing the specimen and running the test under
an inert gas blanket. Since some materials degrade near the melting region, carefully distinguish between degradation and
transition. See Appendix X1.
6.4 Since milligram quantities of sample are used, ensure that samples are homogeneous and representative.
6.5 Sublimation or decomposition will lead to a different heat consumption and, perhaps, a change in composition of the
specimen. The specimen holder should be examined after the measurement for crystals not part of the resolidified melt.
7. Apparatus
7.1 The essential equipment required to provide the minimum instrument capability for this test method includes:
7.1.1 Differential Scanning Calorimeter (DSC), consisting of:
7.1.1.1 DSC Test Chamber, composed of a furnace(s) to provide uniform controlled heating of a specimen and reference to a
constant temperature or at a constant rate within the applicable temperature range of this test method; a temperature sensor to
provide an indication of the specimen temperature readable to 60.1 K; a differential sensor to detect a heat flow difference between
the specimen and reference equivalent to 10 μW; and a means of sustaining a test chamber environment of N at a purge rate of
15 mL/min to 50 mL/min 6 5 mL/min.
7.1.1.2 Temperature Controller, capable of executing a specific temperature program by operating the furnace(s) between
selected temperature limits at a rate of temperature change of 0.3 K/min to 0.7 K/min constant to 60.01 K/min.
7.1.1.3 Data Collection Device, to provide a means of acquiring, storing, and displaying measured or calculated signals, or both.
The minimum output signals required for DSC are heat flow, temperature, and time.
7.1.2 Containers, that are inert to the specimen, and that are of suitable structural shape and integrity for use in the DSC test
chamber, made of materials of high thermal conductivity, such as aluminum.
7.2 Planimeter, computer- or electronic-based data treatment or other instrumentation to determine area to within 61 %
precision.better than 61 %.
7.3 Balance, with a capacity of at least 100 mg capable of weighing to an accuracy of readable to within 0.01 mg.
8. Sampling
8.1 The test sample (liquid or solid) should be mixed prior to sampling and sampled by removing portions from various parts
of the container. Combine the portions and mix well to provide a representative sample for the purity determinations. Only 1 mg
to 3 mg is required for each analysis.
8.2 Avoid any physical or mechanical treatment of the material that will cause chemical changes. For example, grinding the
sample for size reduction often introduces such changes as a result of heat generated by friction.
9. Calibration
9.1 Perform any calibrations procedures called for by the instrument manufacturer as described in the operations manual.
9.2 Calibrate the apparatus temperature signal at the heating rate to be used in this test method (see 10.8) using Test Method
E967. High purity (>99.99 %) indium metal is a convenient material to use for this purpose.
9.3 Calibrate the apparatus heat flow signal at the heating rate to be used in this test method (see 10.8) using Practice E968.
High purity (>99.99 %) indium metal is a convenient material to use for this purpose.
9.4 Determine the leading edge slope (S) in mW/K f
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...