ASTM E2253-21
(Test Method)Standard Test Method for Temperature and Enthalpy Measurement Validation of Differential Scanning Calorimeters
Standard Test Method for Temperature and Enthalpy Measurement Validation of Differential Scanning Calorimeters
SIGNIFICANCE AND USE
5.1 This test method may be used to determine and validate the performance of a particular DSC apparatus.
5.2 This test method may be used to determine and validate the performance of a particular method based upon a DSC temperature and enthalpic measurement, or both.
5.3 This test method may be used to determine the repeatability of a particular apparatus, operator, or laboratory.
5.4 This test method may be used for specification and regulatory compliance purposes.
SCOPE
1.1 This test method provides procedures for validating temperature and enthalpy measurements of differential scanning calorimeters (DSC) and analytical methods based upon the measurement of temperature or enthalpy (or heat), or both, by DSC. Performance parameters determined include temperature and calorimetric repeatability (precision), detection limit, quantitation limit, linearity, and bias. This test method is applicable to exothermic events.
1.2 Validation of apparatus performance and analytical methods is requested or required for quality initiatives or where results may be used for legal purposes.
1.3 The values stated in SI units are to regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2021
- Technical Committee
- E37 - Thermal Measurements
- Drafting Committee
- E37.10 - Fundamental, Statistical and Mechanical Properties
Relations
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Sep-2015
- Effective Date
- 01-May-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-May-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 15-Sep-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Mar-2013
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Aug-2011
- Effective Date
- 01-Aug-2011
Overview
ASTM E2253-21: Standard Test Method for Temperature and Enthalpy Measurement Validation of Differential Scanning Calorimeters (DSC) provides essential procedures for validating the temperature and enthalpy measurements of DSC instruments. Widely used in analytical laboratories, this standard establishes robust methodologies for ensuring the reliability, precision, and consistency of results involving differential scanning calorimetry, which is critical in fields ranging from pharmaceuticals to materials science.
This standard outlines requirements for assessing the performance of both DSC equipment and related analytical methods. By systematically validating temperature and enthalpic data, organizations can comply with quality assurance initiatives and regulatory requirements, and achieve reliable results for specification or legal purposes.
Key Topics
- DSC Performance Validation: Procedures specify how to determine and validate temperature and enthalpy measurements, confirming that a DSC apparatus performs accurately under defined operational conditions.
- Analytical Method Validation: Provides guidance for validating methods dependent on DSC measurements, which is vital for method development and regulatory compliance.
- Precision and Repeatability: Describes how to determine repeatability of measurements across apparatus, operators, and laboratories, supporting inter-laboratory consistency.
- Detection and Quantitation Limits: Offers methodology for establishing the minimum levels at which analytes can be reliably detected or quantified by DSC.
- Linearity and Bias: Ensures that results are proportional and accurate across the range of interest, using linear regression techniques and comparisons to reference standards.
- Use of Certified Reference Materials: Specifies the use of high-purity indium, bismuth, and zinc metals with known thermal properties for calibration and validation.
- Regulatory and Specification Compliance: Supports compliance with internal, industry, and regulatory requirements for temperature and enthalpy measurement accuracy.
Applications
ASTM E2253-21 delivers practical value in various sectors where thermal analysis is a cornerstone technique:
- Quality Control: Assures performance of DSC equipment and analytical procedures, allowing manufacturers and laboratories to meet internal QC or external regulatory requirements.
- Method Development: Critical in developing or validating new DSC methods for material identification, purity analysis, and thermal stability studies.
- Interlaboratory Studies: Enables reliable comparisons and benchmarking of results from different operators or facilities through standardized precision and bias assessments.
- Specification Testing: Used in acceptance testing of materials based on their thermal properties, aligning with customer or regulatory specifications.
- Regulatory Submissions: Helps meet documentation requirements for regulatory submissions (e.g., pharmaceutical filings), where validated measurement methods are mandatory.
- Legal and Forensic Uses: Ensures that DSC-derived data uphold necessary standards for evidentiary or compliance purposes.
Related Standards
ASTM E2253-21 references several other key standards and regulatory guidelines to support comprehensive DSC validation:
- ASTM E177 - Use of Terms Precision and Bias
- ASTM E473 - Terminology Relating to Thermal Analysis and Rheology
- ASTM E967 - Temperature Calibration of DSC and Differential Thermal Analyzers
- ASTM E968 - Heat Flow Calibration of DSC
- ASTM E1142 - Terminology Relating to Thermophysical Properties
- ASTM E1860 - Elapsed Time Calibration of Thermal Analyzers
- ASTM E1970 - Statistical Treatment of Thermoanalytical Data
- ASTM E2161 - Terminology Relating to Performance Validation in Thermal Analysis and Rheology
- FDA Q2B - Validation of Analytical Procedures: Methodology
For laboratories and industries dependent on differential scanning calorimetry, ASTM E2253-21 provides a recognized framework for ensuring the highest standards in measurement validation, supporting data credibility and regulatory compliance in thermal analysis.
Buy Documents
ASTM E2253-21 - Standard Test Method for Temperature and Enthalpy Measurement Validation of Differential Scanning Calorimeters
REDLINE ASTM E2253-21 - Standard Test Method for Temperature and Enthalpy Measurement Validation of Differential Scanning Calorimeters
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM E2253-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Temperature and Enthalpy Measurement Validation of Differential Scanning Calorimeters". This standard covers: SIGNIFICANCE AND USE 5.1 This test method may be used to determine and validate the performance of a particular DSC apparatus. 5.2 This test method may be used to determine and validate the performance of a particular method based upon a DSC temperature and enthalpic measurement, or both. 5.3 This test method may be used to determine the repeatability of a particular apparatus, operator, or laboratory. 5.4 This test method may be used for specification and regulatory compliance purposes. SCOPE 1.1 This test method provides procedures for validating temperature and enthalpy measurements of differential scanning calorimeters (DSC) and analytical methods based upon the measurement of temperature or enthalpy (or heat), or both, by DSC. Performance parameters determined include temperature and calorimetric repeatability (precision), detection limit, quantitation limit, linearity, and bias. This test method is applicable to exothermic events. 1.2 Validation of apparatus performance and analytical methods is requested or required for quality initiatives or where results may be used for legal purposes. 1.3 The values stated in SI units are to regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method may be used to determine and validate the performance of a particular DSC apparatus. 5.2 This test method may be used to determine and validate the performance of a particular method based upon a DSC temperature and enthalpic measurement, or both. 5.3 This test method may be used to determine the repeatability of a particular apparatus, operator, or laboratory. 5.4 This test method may be used for specification and regulatory compliance purposes. SCOPE 1.1 This test method provides procedures for validating temperature and enthalpy measurements of differential scanning calorimeters (DSC) and analytical methods based upon the measurement of temperature or enthalpy (or heat), or both, by DSC. Performance parameters determined include temperature and calorimetric repeatability (precision), detection limit, quantitation limit, linearity, and bias. This test method is applicable to exothermic events. 1.2 Validation of apparatus performance and analytical methods is requested or required for quality initiatives or where results may be used for legal purposes. 1.3 The values stated in SI units are to regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2253-21 is classified under the following ICS (International Classification for Standards) categories: 17.200.10 - Heat. Calorimetry. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2253-21 has the following relationships with other standards: It is inter standard links to ASTM E473-23b, ASTM E1142-23b, ASTM E2161-15, ASTM E1142-15, ASTM E1142-14b, ASTM E473-14, ASTM E177-14, ASTM E1142-14a, ASTM E1142-14, ASTM E1860-13, ASTM E177-13, ASTM E2161-13, ASTM E1142-12, ASTM E1142-11b, ASTM E1970-11. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2253-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2253 − 21
Standard Test Method for
Temperature and Enthalpy Measurement Validation of
Differential Scanning Calorimeters
This standard is issued under the fixed designation E2253; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E967 Test Method for Temperature Calibration of Differen-
tial Scanning Calorimeters and Differential Thermal Ana-
1.1 This test method provides procedures for validating
lyzers
temperature and enthalpy measurements of differential scan-
E968 Practice for Heat Flow Calibration of Differential
ning calorimeters (DSC) and analytical methods based upon
Scanning Calorimeters
the measurement of temperature or enthalpy (or heat), or both,
E1142 Terminology Relating to Thermophysical Properties
by DSC. Performance parameters determined include tempera-
E1860 Test Method for Elapsed Time Calibration of Ther-
ture and calorimetric repeatability (precision), detection limit,
mal Analyzers
quantitation limit, linearity, and bias. This test method is
E1970 PracticeforStatisticalTreatmentofThermoanalytical
applicable to exothermic events.
Data
1.2 Validation of apparatus performance and analytical
E2161 Terminology Relating to Performance Validation in
methodsisrequestedorrequiredforqualityinitiativesorwhere
Thermal Analysis and Rheology
results may be used for legal purposes.
2.2 FDA Publications:
1.3 The values stated in SI units are to regarded as standard. Q2B Validation of Analytical Procedures Methodology, 62
No other units of measurement are included in this standard.
FR 27464, May 19, 1997
1.4 This standard does not purport to address all of the
3. Terminology
safety concerns, if any, associated with its use. It is the
3.1 Technical terms used in this standard are defined in
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter- Practice E177 and in Terminologies E473, E1142, and E2161
including analyte, detection limit, differential scanning
mine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accor- calorimetry, enthalpy, extrapolated onset value (temperature),
dance with internationally recognized principles on standard- linearity, mean, precision, quantitation limit, relative standard
ization established in the Decision on Principles for the deviation,repeatability,standarddeviation,thermalcurve, and
Development of International Standards, Guides and Recom- validation.
mendations issued by the World Trade Organization Technical
4. Summary of Test Method
Barriers to Trade (TBT) Committee.
4.1 Temperature and time are the primary independent
2. Referenced Documents
parameters and heat flow is the primary dependent experimen-
2.1 ASTM Standards: tal parameter provided by DSC. Integration of heat flow, as a
E177 Practice for Use of the Terms Precision and Bias in
function of time, yields enthalpy (heat).
ASTM Test Methods 4.1.1 Time, measured by the DSC apparatus, shall conform
E473 Terminology Relating to Thermal Analysis and Rhe-
to better than 0.1 % verified by Test Method E1860 and
ology reported.
4.1.2 Temperature is directly measured by a temperature
sensor that is an integral part of the differential scanning
This test method is under the jurisdiction ofASTM Committee E37 on Thermal
calorimetry apparatus.
Measurements and is the direct responsibility of Subcommittee E37.10 on
Fundamental, Statistical and Mechanical Properties. 4.1.3 Heat flow, a measured value, is validated by its
Current edition approved July 1, 2021. Published July 2021. Originally approved
integration over time to obtain the desired calorimetric (en-
in 2003. Last previous edition approved in 2016 as E2253 – 16. DOI: 10.1520/
thalpic) information of interest.
E2253-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available from U.S. Food and Drug Administration (FDA), 10903 New
the ASTM website. Hampshire Ave., Silver Spring, MD 20993, http://www.fda.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2253 − 21
4.2 Calorimetric validation of a differential scanning calo- 6.1.1.1 Furnace(s), to provide uniform controlled heating or
rimetric apparatus at a single temperature is performed using cooling of a specimen and reference to a constant temperature
the indium metal melt as an analyte. or at a constant rate within the applicable temperature range of
the test method.
4.3 Validation of a DSC method based upon enthalpic
6.1.1.2 Temperature Sensor, to provide an indication of the
measurement may be performed using the test specimen as the
specimen temperature to readability required.
analyte.
6.1.1.3 Differential Sensor, to detect a heat flow difference
4.4 The enthalpy of three (or more) specimens (nominally
between the specimen and reference.
representing the maximum, midpoint and minimum of the
6.1.1.4 A means of sustaining a test chamber environment
range of the test method) are measured in triplicate (or more).
of an inert purge gas at a rate of 10 mL/min to 50 mL/min 6
A fourth blank specimen, containing no analyte, is also
10 %.
measured in triplicate.
NOTE 2—Typically, 99+ % pure nitrogen (or other inert gas, such as
argon or helium) is employed when oxidation in air is a concern. Unless
NOTE1—Repeatabilityisdeterminedbyperformingasufficientnumber
of determinations to calculate statistically valid estimates of the standard the effects of moisture are to be studied, the use of a dry purge gas is
recommended, especially for operation at subambient temperatures.
deviation or relative standard deviation of the measurement.
6.1.2 Temperature Controller, capable of executing a spe-
4.4.1 Calorimetric linearity and bias are determined from
cific temperature program by operating the furnace(s) between
the best-fit (linear regression) straight-line correlation of the
selected temperature limits at a rate of temperature change
results from measurements of the three (or more) specimens.
constant to 61 % or at an isothermal temperature constant to
4.4.2 Calorimetric detection limit and quantitation limit are
60.5 °C.
determined from the standard deviation of the blank determi-
6.1.3 Data Collection Device, to provide a means of
nation.
acquiring, storing, and displaying measured or calculated
4.4.3 Calorimetric repeatability is determined from the re-
signals, or both. The minimum output signals required for
peatability measurement of the three (or more) specimens.
differential scanning calorimetry are heat flow, temperature,
4.5 The temperature validation of a differential scanning
and time.
calorimetric apparatus is performed at three temperatures using
6.1.4 Containers, (pans, crucibles, vials, lids, closures,
indium, bismuth, and zinc metal melts as an analyte (see
seals, etc.) that are inert to the specimen and reference
Section 7).
materials and that are of suitable structural shape and integrity
4.5.1 The melting temperature of three (or more) materials
to contain the specimen and reference in accordance with the
representing the maximum, midpoint, and minimum of the
specific requirements of the test method.
temperature range of the test method are measured in triplicate
6.2 Balance, of 100 mg or greater capacity to weigh
(or more) (see Note 1).
specimens and containers to 61 µg.
4.5.2 Temperature linearity and bias are determined from
NOTE 3—A balance of this high precision is required so that weighing
the best-fit (linear regression) straight-line correlation of the
imprecision is not part of the overall method imprecision.
results from the temperature measurements at the three (or
more) temperatures.
7. Reagents and Materials
4.5.3 Temperature repeatability, detection limit and quanti-
7.1 Indium (In) Metal, 99.99+ % purity, preferably a certi-
tation limit are determined from the standard deviation of the
fied reference material for which the melting temperature and
replicate temperature measurements.
enthalpy of fusion are known.
7.2 Bismuth (Bi) Metal, 99.99+ % purity, preferably a cer-
5. Significance and Use
tified reference material for which the melting temperature is
5.1 This test method may be used to determine and validate
known.
the performance of a particular DSC apparatus.
7.3 Zinc (Zn) Metal, 99.99+ % purity, preferably a certified
5.2 This test method may be used to determine and validate
reference material for which the melting temperature is known.
the performance of a particular method based upon a DSC
temperature and enthalpic measurement, or both.
8. Calibration and Standardization
5.3 This test method may be used to determine the repeat-
8.1 After turning the power on, allow the instrument to
ability of a particular apparatus, operator, or laboratory.
equilibrate for at least one hour prior to any measurements.
5.4 This test method may be used for specification and
8.2 Perform any cleaning and calibration procedures de-
regulatory compliance purposes.
scribed by the manufacturer in the apparatus Operator’s
Manual.
6. Apparatus
8.3 If not previously established, perform temperature and
6.1 DifferentialScanningCalorimeter(DSC)—Theessential
heat flow calibrations according to Test Method E967 and
instrumentation required to provide the minimum differential
Practice E968 respectively, using the same purge gas, purge
scanning calorimetric capability for this test method includes:
gas flow rate and heating rate (here 10 °C/min) to be used for
6.1.1 DSC Test Chamber, composed of: validation experiments.
E2253 − 21
8.4 If not previously established, obtain the instrument’s 9.6 Heat the test specimen at 10 °C/min through the indium
elapsed time conformance using Test Method E1860. melting transition to 180 °C and record the thermal curve.
NOTE 6—Other heating rates may be used but shall be reported.
9. Procedure for Determining Calorimetric Repeatability,
9.7 Cool the test specimen to 120 °C at 5 °C/min, then cool
Detection Limit, Quantitation Limit, Linearity, and Bias
to ambient temperature at any convenient rate. The thermal
9.1 This process involves characterizing, in triplicate, a
curve need not be recorded.
blank and three (or more) test specimens taken to represent the
9.8 Construct a baseline for the melting endotherm by
low, medium and high extremes of the range over which
selecting a point on the curve immediately before and another
performance is to be validated.
immediately after the endotherm. Record the temperatures of
NOTE 4—The details of this procedure are written using indium as an
these two points as T and T . Construct a linear baseline
analyte. For validation of an enthalpic method, test specimens represent-
1 2
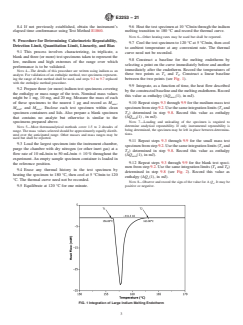
ing the range of that method shall be used, and steps 9.2 to 9.7 replaced between the two points (see Fig. 1).
with the enthalpic method procedure.
9.9 Integrate, as a function of time, the heat flow described
9.2 Prepare three (or more) indium test specimens covering
by the constructed baseline and the melting endotherm. Record
the enthalpy or mass range of the tests. Nominal mass values
this value as enthalpy (∆Q (1), in mJ).
max
might be 1 mg, 10 mg, and 20 mg. Measure the mass of each
of these specimens to the nearest 1 µg and record as M , 9.10 Repeat steps 9.3 through 9.9 for the medium mass test
min
specimenfromstep9.2.Usethesameintegrationlimits(T and
M , and M . Enclose each test specimen within clean
mid max 1
specimen containers and lids. Also prepare a blank specimen T ) determined in step 9.8. Record this value as enthalpy
(∆Q (1) , in mJ).
that contains no analyte but otherwise is similar to the
mid
specimens prepared above.
NOTE 7—Loading and unloading of the specimen is required to
determine analytical repeatability. If only instrumental repeatability is
NOTE 5—Most thermoanalytical methods cover 1.5 to 2 decades of
range. The mass values selected should be approximately equally distrib- being determined, the specimen may be left in place between determina-
tions.
uted over the anticipated range. Other masses and mass ranges may be
used but shall be reported.
9.11 Repeat steps 9.3 through 9.9 for the small mass test
9.3 Load the largest specimen into the instrument chamber,
specimenfromstep9.2.Usethesameintegrationlimits(T and
purge the chamber with dry nitrogen (or other inert gas) at a
T ) determined in step 9.8. Record this value as enthalpy
flow rate of 10 mL/min to 50 mL/min 6 10 % throughout the
(∆Q (1), in mJ).
min
experiment. An empty sample specimen container is loaded in
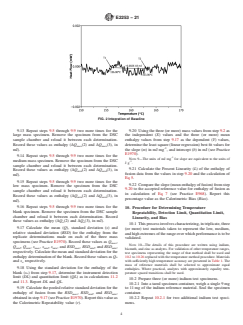
9.12 Repeat steps 9.3 through 9.9 for the blank test speci-
the reference position.
men from step 9.2. Use the same integration limits (T and T )
1 2
9.4 Erase any thermal history in the test specimen by
determined in step 9.8 (see Fig. 2). Record this value as
heating the specimen to 180 °C, then cool at 5 °C/min to 120
enthalpy (∆Q (1), in mJ).
o
°C. The thermal curve need not be recorded.
NOTE 8—Observe and record the sign of the value for∆ Q . It may be
o
9.5 Equilibrate at 120 °C for one minute. positive or negative.
FIG. 1 Integration of Large Indium Melting Endotherm
E2253 − 21
FIG. 2 Integration of Baseline
9.13 Repeat steps 9.5 through 9.9 two more times for the 9.20 Using the three (or more) mass values from step 9.2 as
large mass specimen. Remove the specimen from the DSC the independent (X) values and the three (or more) mean
sample chamber and reload it between each determination. enthalpy values from step 9.17 as the dependent (Y) values,
Record these values as enthalpy (∆Q (2) and ∆Q (3), in determine the least square (linear regression) best fit values for
max max
-1
mJ). the slope (m)inmJmg , and intercept (b) in mJ (see Practice
E1970).
9.14 Repeat steps 9.5 through 9.9 two more times for the
-1
NOTE 9—The units of mJ mg for slope are equivalent to the units of
medium mass specimen. Remove the specimen from the DSC
-1
Jg .
sample chamber and reload it between each determination.
9.21 Calculate the Percent Linearity (L) of the enthalpy of
Record these values as enthalpy (∆Q (2) and ∆Q (3), in
mid mid
fusion data
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2253 − 16 E2253 − 21
Standard Test Method for
Temperature and Enthalpy Measurement Validation of
Differential Scanning Calorimeters
This standard is issued under the fixed designation E2253; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*Scope
1.1 This test method provides procedures for validating temperature and enthalpy measurements of differential scanning
calorimeters (DSC) and analytical methods based upon the measurement of temperature or enthalpy (or heat), or both, by DSC.
Performance parameters determined include temperature and calorimetric repeatability (precision), detection limit, quantitation
limit, linearity, and bias. This test method is applicable to exothermic events.
1.2 Validation of apparatus performance and analytical methods is requested or required for quality initiatives or where results may
be used for legal purposes.
1.3 The values stated in SI units are to regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E473 Terminology Relating to Thermal Analysis and Rheology
E967 Test Method for Temperature Calibration of Differential Scanning Calorimeters and Differential Thermal Analyzers
E968 Practice for Heat Flow Calibration of Differential Scanning Calorimeters
E1142 Terminology Relating to Thermophysical Properties
E1860 Test Method for Elapsed Time Calibration of Thermal Analyzers
E1970 Practice for Statistical Treatment of Thermoanalytical Data
E2161 Terminology Relating to Performance Validation in Thermal Analysis and Rheology
2.2 FDA Publications:
Q2B Validation of Analytical Procedures Methodology, 62 FR 27464, May 19, 1997
This test method is under the jurisdiction of ASTM Committee E37 on Thermal Measurements and is the direct responsibility of Subcommittee E37.10 on Fundamental,
Statistical and Mechanical Properties.
Current edition approved April 1, 2016July 1, 2021. Published April 2016July 2021. Originally approved in 2003. Last previous edition approved in 20112016 as
E2253 – 11.E2253 – 16. DOI: 10.1520/E2253-16.10.1520/E2253-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from U.S. Food and Drug Administration (FDA), 10903 New Hampshire Ave., Silver Spring, MD 20993-0002,20993, http://www.fda.gov.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2253 − 21
3. Terminology
3.1 Technical terms used in this standard are defined in Practice E177 and in Terminologies E473, E1142, and E2161 including
analyte, detection limit, differential scanning calorimetry, enthalpy, extrapolated onset value (temperature), linearity, mean,
precision, quantitation limit, relative standard deviation, repeatability, standard deviation, thermal curve, and validation.
4. Summary of Test Method
4.1 Temperature and time are the primary independent parameters and heat flow is the primary dependent experimental parameter
provided by DSC. Integration of heat flow, as a function of time, yields enthalpy (heat).
4.1.1 Time, measured by the DSC apparatus, shall conform to better than 0.1 % verified by Test Method E1860 and reported.
4.1.2 Temperature is directly measured by a temperature sensor that is an integral part of the differential scanning calorimetry
apparatus.
4.1.3 Heat flow, a measured value, is validated by its integration over time to obtain the desired calorimetric (enthalpic)
information of interest.
4.2 Calorimetric validation of a differential scanning calorimetric apparatus at a single temperature is performed using the indium
metal melt as an analyte.
4.3 Validation of a DSC method based upon enthalpic measurement may be performed using the test specimen as the analyte.
4.4 The enthalpy of three (or more) specimens (nominally representing the maximum, midpoint and minimum of the range of the
test method) are measured in triplicate (or more). A fourth blank specimen, containing no analyte, is also measured in triplicate.
NOTE 1—Repeatability is determined by performing a sufficient number of determinations to calculate statistically valid estimates of the standard deviation
or relative standard deviation of the measurement.
4.4.1 Calorimetric linearity and bias are determined from the best-fit (linear regression) straight-line correlation of the results from
measurements of the three (or more) specimens.
4.4.2 Calorimetric detection limit and quantitation limit are determined from the standard deviation of the blank determination.
4.4.3 Calorimetric repeatability is determined from the repeatability measurement of the three (or more) specimens.
4.5 The temperature validation of a differential scanning calorimetric apparatus is performed at three temperatures using indium,
bismuth, and zinc metal melts as an analyte (see Section 7).
4.5.1 The melting temperature of three (or more) materials representing the maximum, midpoint, and minimum of the temperature
range of the test method are measured in triplicate (or more) (see Note 1).
4.5.2 Temperature linearity and bias are determined from the best-fit (linear regression) straight-line correlation of the results from
the temperature measurements at the three (or more) temperatures.
4.5.3 Temperature repeatability, detection limit and quantitation limit are determined from the standard deviation of the replicate
temperature measurements.
5. Significance and Use
5.1 This test method may be used to determine and validate the performance of a particular DSC apparatus.
5.2 This test method may be used to determine and validate the performance of a particular method based upon a DSC temperature
and enthalpic measurement, or both.
E2253 − 21
5.3 This test method may be used to determine the repeatability of a particular apparatus, operator, or laboratory.
5.4 This test method may be used for specification and regulatory compliance purposes.
6. Apparatus
6.1 Differential Scanning Calorimeter (DSC)—The essential instrumentation required to provide the minimum differential
scanning calorimetric capability for this test method includes:
6.1.1 DSC Test Chamber, composed of:
6.1.1.1 Furnace(s), to provide uniform controlled heating or cooling of a specimen and reference to a constant temperature or at
a constant rate within the applicable temperature range of the test method.
6.1.1.2 Temperature Sensor, to provide an indication of the specimen temperature to readability required.
6.1.1.3 Differential Sensor, to detect a heat flow difference between the specimen and reference.
6.1.1.4 A means of sustaining a test chamber environment of an inert purge gas at a rate of 10 mL/min to 50 mL/min 6 10 %.
NOTE 2—Typically, 99+ % pure nitrogen (or other inert gas, such as argon or helium) is employed when oxidation in air is a concern. Unless the effects
of moisture are to be studied, the use of a dry purge gas is recommended, especially for operation at subambient temperatures.
6.1.2 Temperature Controller, capable of executing a specific temperature program by operating the furnace(s) between selected
temperature limits at a rate of temperature change constant to 61 % or at an isothermal temperature constant to 60.5°C.60.5 °C.
6.1.3 Data Collection Device, to provide a means of acquiring, storing, and displaying measured or calculated signals, or both.
The minimum output signals required for differential scanning calorimetry are heat flow, temperature, and time.
6.1.4 Containers, (pans, crucibles, vials, lids, closures, seals, etc.) that are inert to the specimen and reference materials and that
are of suitable structural shape and integrity to contain the specimen and reference in accordance with the specific requirements
of the test method.
6.2 Balance, of 100 mg or greater capacity to weigh specimens and containers to 61 μg.
NOTE 3—A balance of this high precision is required so that weighing imprecision is not part of the overall method imprecision.
7. Reagents and Materials
7.1 Indium (In) Metal, 99.99+ % purity, preferably a certified reference material for which the melting temperature and enthalpy
of fusion are known.
7.2 Bismuth (Bi) Metal, 99.99+ % purity, preferably a certified reference material for which the melting temperature is known.
7.3 Zinc (Zn) Metal, 99.99+ % purity, preferably a certified reference material for which the melting temperature is known.
8. Calibration and Standardization
8.1 After turning the power on, allow the instrument to equilibrate for at least one hour prior to any measurements.
8.2 Perform any cleaning and calibration procedures described by the manufacturer in the apparatus Operator’s Manual.
8.3 If not previously established, perform temperature and heat flow calibrations according to Test Method E967 and Practice
E968 respectively, using the same purge gas, purge gas flow rate and heating rate (here 10°C/min) 10 °C/min) to be used for
validation experiments.
E2253 − 21
8.4 If not previously established, obtain the instrument’s elapsed time conformance using Test Method E1860.
9. Procedure for Determining Calorimetric Repeatability, Detection Limit, Quantitation Limit, Linearity, and Bias
9.1 This process involves characterizing, in triplicate, a blank and three (or more) test specimens taken to represent the low,
medium and high extremes of the range over which performance is to be validated.
NOTE 4—The details of this procedure are written using indium as an analyte. For validation of an enthalpic method, test specimens representing the range
of that method shall be used, and steps 9.2 to 9.7 replaced with the enthalpic method procedure.
9.2 Prepare three (or more) indium test specimens covering the enthalpy or mass range of the tests. Nominal mass values might
be 1, 10, 1 mg, 10 mg, and 20 mg. Measure the mass of each of these specimens to the nearest 1 μg and record as M ,M , and
min mid
M . Enclose each test specimen within clean specimen containers and lids. Also prepare a blank specimen that contains no
max
analyte but otherwise is similar to the specimens prepared above.
NOTE 5—Most thermoanalytical methods cover 1.5 to 2 decades of range. The mass values selected should be approximately equally distributed over the
anticipated range. Other masses and mass ranges may be used but shall be reported.
9.3 Load the largest specimen into the instrument chamber, purge the chamber with dry nitrogen (or other inert gas) at a flow rate
of 10 mL/min to 50 mL/min 6 10 % throughout the experiment. An empty sample specimen container is loaded in the reference
position.
9.4 Erase any thermal history in the test specimen by heating the specimen to 180°C, 180 °C, then cool at 5°C/min to 120°C. 5
°C/min to 120 °C. The thermal curve need not be recorded.
9.5 Equilibrate at 120°C 120 °C for one minute.
9.6 Heat the test specimen at 10°C/min 10 °C/min through the indium melting transition to 180°C 180 °C and record the thermal
curve.
NOTE 6—Other heating rates may be used but shall be reported.
9.7 Cool the test specimen to 120°C at 5°C/min, 120 °C at 5 °C/min, then cool to ambient temperature at any convenient rate.
The thermal curve need not be recorded.
9.8 Construct a baseline for the melting endotherm by selecting a point on the curve immediately before and another immediately
after the endotherm. Record the temperatures of these two points as T and T . Construct a linear baseline between the two points
1 2
(see Fig. 1).>
9.9 Integrate, as a function of time, the heat flow described by the constructed baseline and the melting endotherm. Record this
value as enthalpy (ΔQ (1), in mJ).
max
9.10 Repeat steps 9.3 through 9.9 for the medium mass test specimen from step 9.2. Use the same integration limits (T and T )
1 2
determined in step 9.8. Record this value as enthalpy (ΔQ (1) , in mJ).
mid
NOTE 7—Loading and unloading of the specimen is required to determine analytical repeatability. If only instrumental repeatability is being determined,
the specimen may be left in place between determinations.
9.11 Repeat steps 9.3 through 9.9 for the small mass test specimen from step 9.2. Use the same integration limits (T and T )
1 2
determined in step 9.8. Record this value as enthalpy (ΔQ (1), in mJ).
min
9.12 Repeat steps 9.3 through 9.9 for the blank test specimen from step 9.2. Use the same integration limits (T and T ) determined
1 2
in step 9.8 (see Fig. 2). Record this value as enthalpy (ΔQ (1), in mJ).>
o
NOTE 8—Observe and record the sign of the value for Δ Q . It may be positive or negative.
o
E2253 − 21
FIG. 1 Integration of Large Indium Melting Endotherm
FIG. 2 Integration of Baseline
9.13 Repeat steps 9.5 through 9.9 two more times for the large mass specimen. Remove the specimen from the DSC sample
chamber and reload it between each determination. Record these values as enthalpy (ΔQ (2) and ΔQ (3), in mJ).
max max
9.14 Repeat steps 9.5 through 9.9 two more times for the medium mass specimen. Remove the specimen from the DSC sample
chamber and reload it between each determination. Record these values as enthalpy (ΔQ (2) and ΔQ (3), in mJ).
mid mid
9.15 Repeat steps 9.5 through 9.9 two more times for the low mass specimen. Remove the specimen from the DSC sample
chamber and reload it between each determination. Record these values as enthalpy (ΔQ (2) and ΔQ (3), in mJ).
min min
9.16 Repeat steps 9.5 through 9.9 two more times for the blank specimen. Remove the specimen from the DSC sample chamber
and reload it between each determination. Record these values as enthalpy (ΔQ (2) and ΔQ (3), in mJ).
o o
E2253 − 21
9.17 Calculate the mean (Q), standard deviation (s) and relative standard deviation (RSD) for th
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...