ASTM D3588-98(2017)e1
(Practice)Standard Practice for Calculating Heat Value, Compressibility Factor, and Relative Density of Gaseous Fuels
Standard Practice for Calculating Heat Value, Compressibility Factor, and Relative Density of Gaseous Fuels
SIGNIFICANCE AND USE
5.1 The heating value is a measure of the suitability of a pure gas or a gas mixture for use as a fuel; it indicates the amount of energy that can be obtained as heat by burning a unit of gas. For use as heating agents, the relative merits of gases from different sources and having different compositions can be compared readily on the basis of their heating values. Therefore, the heating value is used as a parameter for determining the price of gas in custody transfer. It is also an essential factor in calculating the efficiencies of energy conversion devices such as gas-fired turbines. The heating values of a gas depend not only upon the temperature and pressure, but also upon the degree of saturation with water vapor. However, some calorimetric methods for measuring heating values are based upon the gas being saturated with water at the specified conditions.
5.2 The relative density (specific gravity) of a gas quantifies the density of the gas as compared with that of air under the same conditions.
SCOPE
1.1 This practice covers procedures for calculating heating value, relative density, and compressibility factor at base conditions (14.696 psia and 60°F (15.6°C)) for natural gas mixtures from compositional analysis.2 It applies to all common types of utility gaseous fuels, for example, dry natural gas, reformed gas, oil gas (both high and low Btu), propane-air, carbureted water gas, coke oven gas, and retort coal gas, for which suitable methods of analysis as described in Section 6 are available. Calculation procedures for other base conditions are given.
1.2 The values stated in inch-pound units are to be regarded as the standard. The SI units given in parentheses are for information only.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2017
- Technical Committee
- D03 - Gaseous Fuels
Relations
- Effective Date
- 01-Nov-2015
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Jan-2010
- Effective Date
- 01-Jun-2006
- Effective Date
- 01-Nov-2004
- Effective Date
- 10-May-2003
- Effective Date
- 01-Jan-2001
- Effective Date
- 01-Jan-2000
- Effective Date
- 10-Apr-1999
- Effective Date
- 01-Jan-1996
Overview
ASTM D3588-98(2017)e1, developed by ASTM International, specifies standard procedures for calculating the heating value (heat content), compressibility factor, and relative density of natural gas and other common utility gaseous fuels. These calculations are essential for the energy sector, particularly when determining the energy content and suitability of a gas for use as a fuel. The standard applies to natural gas, reformed gas, oil gas, propane-air, and various manufactured gases, covering both dry and water-saturated conditions at defined base temperature and pressure.
Key Topics
Heating Value Calculation: Provides guidance for determining the gross and net heating value based on the compositional analysis of gaseous fuels. The heating value signifies the available energy from combustion and is commonly used as the basis for pricing and energy billing.
Relative Density (Specific Gravity): Offers procedures for quantifying the density of a gas in comparison to dry air under identical conditions. This property is crucial for process calculations, custody transfer, and system design.
Compressibility Factor (Z-factor): Details the process for calculating the compressibility factor, which accounts for deviations from ideal gas behavior. Accurate Z-factor determination is important for volumetric flow rate calculations and custody transfer accounting.
Base Conditions: Establishes calculations at standard base conditions of 14.696 psia and 60°F (15.6°C), with procedures for conversion to alternative conditions as required for international and local regulatory purposes.
Compositional Analysis: Lists acceptable analytical methods (such as gas chromatography and mass spectrometry) for determining gas composition, which is the foundational data for all subsequent calculations.
Applications
The procedures in ASTM D3588 are widely used across the energy and utilities sectors where natural gas quality, custody transfer, and system performance are critical. Major applications include:
Energy Billing and Contractual Settlement: Calculated heating values serve as the basis for metering and billing, ensuring fair value in the commodity exchange of natural gas and related fuels.
Efficiency Assessment: Boiler, gas turbine, and combustion system efficiencies rely on accurate calorific values and gas properties for performance optimization.
Pipeline and Distribution Operations: Determination of compressibility and relative density supports accurate flow measurement, pressure regulation, and system balancing.
Comparative Analysis: Facilitates benchmarking of fuel quality between different sources and types of gaseous fuels.
Regulatory Compliance: Supports adherence to local, national, and international rules for gas quality, transfer, and measurement.
Related Standards
Organizations and professionals using ASTM D3588 often reference the following related standards:
- ASTM D1945 – Standard Test Method for Analysis of Natural Gas by Gas Chromatography
- ASTM D2163 – Determination of Hydrocarbons in Liquefied Petroleum Gases
- ASTM D2650 – Chemical Composition of Gases by Mass Spectrometry
- GPA 2145 – Physical Constants for Paraffin Hydrocarbons and Other Components of Natural Gas
- GPA 2172 – Calculation of Gross Heating Value, Relative Density, and Compressibility Factor for Natural Gas Mixtures from Compositional Analysis
- ANSI Z132.1 – Base Conditions of Pressure and Temperature for the Volumetric Measurement of Natural Gas
Practical Value
Adoption of ASTM D3588-98(2017)e1 ensures consistency, accuracy, and traceability in the calculation of key gas properties necessary for energy management, system safety, and financial operations. By providing a standard approach to analyzing and expressing gas quality and energy content, this practice underpins reliable gas trade, operational decision-making, and regulatory conformance in the global natural gas industry.
Buy Documents
ASTM D3588-98(2017)e1 - Standard Practice for Calculating Heat Value, Compressibility Factor, and Relative Density of Gaseous Fuels
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.
Sponsored listings
Frequently Asked Questions
ASTM D3588-98(2017)e1 is a standard published by ASTM International. Its full title is "Standard Practice for Calculating Heat Value, Compressibility Factor, and Relative Density of Gaseous Fuels". This standard covers: SIGNIFICANCE AND USE 5.1 The heating value is a measure of the suitability of a pure gas or a gas mixture for use as a fuel; it indicates the amount of energy that can be obtained as heat by burning a unit of gas. For use as heating agents, the relative merits of gases from different sources and having different compositions can be compared readily on the basis of their heating values. Therefore, the heating value is used as a parameter for determining the price of gas in custody transfer. It is also an essential factor in calculating the efficiencies of energy conversion devices such as gas-fired turbines. The heating values of a gas depend not only upon the temperature and pressure, but also upon the degree of saturation with water vapor. However, some calorimetric methods for measuring heating values are based upon the gas being saturated with water at the specified conditions. 5.2 The relative density (specific gravity) of a gas quantifies the density of the gas as compared with that of air under the same conditions. SCOPE 1.1 This practice covers procedures for calculating heating value, relative density, and compressibility factor at base conditions (14.696 psia and 60°F (15.6°C)) for natural gas mixtures from compositional analysis.2 It applies to all common types of utility gaseous fuels, for example, dry natural gas, reformed gas, oil gas (both high and low Btu), propane-air, carbureted water gas, coke oven gas, and retort coal gas, for which suitable methods of analysis as described in Section 6 are available. Calculation procedures for other base conditions are given. 1.2 The values stated in inch-pound units are to be regarded as the standard. The SI units given in parentheses are for information only. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The heating value is a measure of the suitability of a pure gas or a gas mixture for use as a fuel; it indicates the amount of energy that can be obtained as heat by burning a unit of gas. For use as heating agents, the relative merits of gases from different sources and having different compositions can be compared readily on the basis of their heating values. Therefore, the heating value is used as a parameter for determining the price of gas in custody transfer. It is also an essential factor in calculating the efficiencies of energy conversion devices such as gas-fired turbines. The heating values of a gas depend not only upon the temperature and pressure, but also upon the degree of saturation with water vapor. However, some calorimetric methods for measuring heating values are based upon the gas being saturated with water at the specified conditions. 5.2 The relative density (specific gravity) of a gas quantifies the density of the gas as compared with that of air under the same conditions. SCOPE 1.1 This practice covers procedures for calculating heating value, relative density, and compressibility factor at base conditions (14.696 psia and 60°F (15.6°C)) for natural gas mixtures from compositional analysis.2 It applies to all common types of utility gaseous fuels, for example, dry natural gas, reformed gas, oil gas (both high and low Btu), propane-air, carbureted water gas, coke oven gas, and retort coal gas, for which suitable methods of analysis as described in Section 6 are available. Calculation procedures for other base conditions are given. 1.2 The values stated in inch-pound units are to be regarded as the standard. The SI units given in parentheses are for information only. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D3588-98(2017)e1 is classified under the following ICS (International Classification for Standards) categories: 75.160.30 - Gaseous fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3588-98(2017)e1 has the following relationships with other standards: It is inter standard links to ASTM D1946-90(2015)e1, ASTM D2163-14, ASTM D1946-90(2011), ASTM D2650-10, ASTM D1945-03(2010), ASTM D1946-90(2006), ASTM D2650-04, ASTM D1945-03, ASTM D1945-96(2001), ASTM D1946-90(2000), ASTM D2650-99, ASTM D2163-91(1996). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3588-98(2017)e1 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
´1
Designation: D3588 − 98 (Reapproved 2017)
Standard Practice for
Calculating Heat Value, Compressibility Factor, and Relative
Density of Gaseous Fuels
This standard is issued under the fixed designation D3588; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Note 2 was editorially corrected in April 2020.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This practice covers procedures for calculating heating
D1717Test Method for Test for Analysis of Commerical
value, relative density, and compressibility factor at base
conditions (14.696 psia and 60°F (15.6°C)) for natural gas Butane-Butene Mixtures and Isolutylene by Gas Chroma-
tography (Withdrawn 1984)
mixtures from compositional analysis. It applies to all com-
D1945Test Method for Analysis of Natural Gas by Gas
montypesofutilitygaseousfuels,forexample,drynaturalgas,
Chromatography
reformed gas, oil gas (both high and low Btu), propane-air,
D1946Practice for Analysis of Reformed Gas by Gas
carbureted water gas, coke oven gas, and retort coal gas, for
Chromatography
which suitable methods of analysis as described in Section 6
D2163Test Method for Determination of Hydrocarbons in
are available. Calculation procedures for other base conditions
Liquefied Petroleum (LP) Gases and Propane/Propene
are given.
Mixtures by Gas Chromatography
1.2 The values stated in inch-pound units are to be regarded
D2650Test Method for Chemical Composition of Gases by
as the standard. The SI units given in parentheses are for
Mass Spectrometry
information only.
2.2 GPA Standards:
1.3 This standard does not purport to address all of the
GPA2145Physical Constants for the Paraffin Hydrocarbons
safety concerns, if any, associated with its use. It is the and Other Components in Natural Gas
responsibility of the user of this standard to establish appro-
GPA Standard 2166Methods of Obtaining Natural Gas
priate safety, health, and environmental practices and deter- Samples for Analysis by Gas Chromatography
mine the applicability of regulatory limitations prior to use. GPA 2172Calculation of Gross Heating Value, Relative
Density, and Compressibility Factor for Natural Gas
1.4 This international standard was developed in accor-
5,6
Mixtures from Compositional Analysis
dance with internationally recognized principles on standard-
GPAStandard2261MethodofAnalysisforNaturalGasand
ization established in the Decision on Principles for the
Similar Gaseous Mixtures by Gas Chromatography
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
This practice is under the jurisdiction of ASTM Committee D03 on Gaseous the ASTM website.
Fuels and is the direct responsibility of Subcommittee D03.03 on Determination of The last approved version of this historical standard is referenced on
Heating Value and Relative Density of Gaseous Fuels. www.astm.org.
Current edition approved April 1, 2017. Published April 2017. Originally AvailablefromGasProcessorsAssociation(GPA),6526E.60thSt.,Tulsa,OK
approved in 1998. Last previous edition approved in 2011 as D3588–98(2011). 74145, http://www.gasprocessors.com.
DOI: 10.1520/D3588-98R17E01. The sole source of supply of the program in either BASIC or FORTRAN
A more rigorous calculation of Z(T,P) at both base conditions and higher suitable for running on computers known to the committee at this time is the Gas
pressures can be made using the calculation procedures in “Compressibility and ProcessorsAssociation.Ifyouareawareofalternativesuppliers,pleaseprovidethis
Super Compressibility for Natural Gas and Other Hydrocarbon Gases,” American information to ASTM International Headquarters. Your comments will receive
Gas Association Transmission Measurement Committee Report 8, AGA Cat. No. careful consideration at a meeting of the responsible technical committee , which
XQ1285, 1985, AGA, 1515 Wilson Blvd., Arlington, VA 22209. you may attend.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
´1
D3588 − 98 (2017)
GPATechnical Publication TP-17Table of Physical Proper- Conversionstootherbaseconditionsshouldbemadeattheend
ties of Hydrocarbons for Extended Analysis of Natural of the calculation to reduce roundoff errors.
Gases
3.1.7 standard temperature (USA)—60°F (15.6°C).
GPSA Data Book,Fig. 23-2, Physical Constants
3.2 Symbols:
2.3 TRC Document:
7 3.2.1 Nomenclature:
TRC Thermodynamic Tables—Hydrocarbons
3.2.1.1 B—second virial coefficient for gas mixture
2.4 ANSI Standard:
3.2.1.2 =β —summation factor for calculating real gas
ANSI Z 132.1-1969:Base Conditions of Pressure and Tem- ij
correction (alternate method)
perature for the Volumetric Measurement of Natural
8,9
Gas
3.2.1.3 (cor)—corrected for water content
3.2.1.4 (dry)—value on water-free basis
3. Terminology
3.2.1.5 d—density for gas relative to the density of air.
id
3.1 Definitions:
3.2.1.6 d —ideal relative density or relative molar mass,
3.1.1 British thermal unit—the defined International Tables
that is, molar mass of gas relative to molar mass of air
id
British thermal unit (Btu).
3.2.1.7 G —molar mass ratio
id
3.1.1.1 Discussion—The defining relationships are:
3.2.1.8 H —gross heating value per unit mass
m
–1 –1
1 Btu•lb = 2.326 J•g (exact)
id
3.2.1.9 H —gross heating value per unit volume
v
1 lb = 453.592 37 g (exact)
id
3.2.1.10 H —gross heating value per unit mole
n
Bytheserelationships,1Btu=1055.05585262J(exact).For
id
most purposes, the value (rounded) 1 Btu = 1055.056 J is
3.2.1.11 h —net heating value per unit mass
m
adequate.
id
3.2.1.12 h —net heating value per unit volume
v
3.1.2 compressibility factor (z)—the ratio of the actual
id
3.2.1.13 h —net heating value per unit mole
n
volume of a given mass of gas at a specified temperature and
3.2.1.14 a, b, c—in Eq 1, integers required to balance the
pressure to its volume calculated from the ideal gas law under
the same conditions. equation: C, carbon; H, hydrogen; S, sulfur; O, oxygen
3.2.1.15 (id)—ideal gas state
3.1.3 gross heating value—theamountofenergytransferred
3.2.1.16 (l)—liquid phase
asheatfromthecomplete,idealcombustionofthegaswithair,
3.2.1.17 M—molar mass
at standard temperature, in which all the water formed by the
3.2.1.18 m—mass flow rate
reaction condenses to liquid. The values for the pure gases
3.2.1.19 n—number of components
appear in GPAStandard 2145, which is revised annually. If the
3.2.1.20 P—pressure in absolute units (psia)
gross heating value has a volumetric rather than a mass or
id
3.2.1.21 Q —ideal energy per unit time released as heat
molar basis, a base pressure must also be specified.
upon combustion
3.1.4 netheatingvalue—theamountofenergytransferredas
3.2.1.22 R—gasconstant,10.7316psia.ft /(lbmol•R)inthis
heat from the total, ideal combustion of the gas at standard
practice (based upon R = 8.31448 J/(mol•K))
temperature in which all the water formed by the reaction
3.2.1.23 (sat)—denotes saturation value
remains in the vapor state. Condensation of any “spectator”
3.2.1.24 T—absolute temperature, °R = °F + 459.67 or K =
water does not contribute to the net heating value. If the net
°C + 273.15
heating value has a volumetric rather than a mass or molar
3.2.1.25 (T, P)—value dependent upon temperature and
basis, a base pressure must also be specified.
pressure
3.1.5 relativedensity—theratioofthedensityofthegaseous
3.2.1.26 V—gas volumetric flow rate
fuel,underobservedconditionsoftemperatureandpressure,to
3.2.1.27 x—mole fraction
the density of dry air (of normal carbon dioxide content) at the
3.2.1.28 Z—gascompressibilityfactorrepeatabilityofprop-
same temperature and pressure.
erty
3.1.6 standard cubic foot of gas—the amount of gas that
3.2.1.29 δ—repeatability of property
3 3
occupies 1 ft (0.028 m ) at a temperature of 60°F (15.6°C)
3.2.1.30 ρ—density in mass per unit volume
n
under a given base pressure and either saturated with water
3.2.1.31 —property summed for Components 1 through
(
vapor(wet)orfreeofwatervapor(dry)asspecified(seeANSI
j51
n, where n represents the total number of components in the
Z 132.1). In this practice, calculations have been made at
mixture
14.696 psia and 60°F (15.6°C), because the yearly update of
GPA2145 by theThermodynamics Research Center, on which
3.2.2 Superscripts:
these calculations are based, are given for this base pressure.
3.2.2.1 id—ideal gas value
3.2.2.2 l—liquid
3.2.2.3 σ—value at saturation (vapor pressure)
AvailablefromThermodynamicsResearchCenter,TheTexasA&MUniversity,
College Station, TX 77843-3111.
3.2.2.4 '—reproducibility
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
3.2.3 Subscripts:
4th Floor, New York, NY 10036, http://www.ansi.org.
9 3.2.3.1 a—value for air
Supporting data have been filed atASTM International Headquarters and may
be obtained by requesting Research Report RR:D03-1007. 3.2.3.2 a—relative number of atoms of carbon in Eq 1
´1
D3588 − 98 (2017)
3.2.3.3 b—relative number of atoms of hydrogen in Eq 1 where id denotes the ideal gas state and l denotes liquid
3.2.3.4 c—relative number of atoms of sulfur in Eq 1 phase. The ideal net heating value results when all the water
3.2.3.5 j—property for component j remains in the ideal gas state. The ideal gross heating value
3.2.3.6 ii—non-ideal gas property for component i results when all the water formed by the reaction condenses to
id
3.2.3.7 ij—non-ideal gas property for mixture of i and j liquid. For water, the reduction from H O(id)toH O(l)is H
2 2 w
l
3.2.3.8 jj—non-ideal gas property for component j – H , the ideal enthalpy of vaporization, which is somewhat
w
y l
3.2.3.9 w—value for water larger than the enthalpy of vaporization H – H ' .
w w
3.2.3.10 1—property for Component 1
7.1.1 Because the gross heating value results from an ideal
3.2.3.11 2—property for Component 2
combustion reaction, ideal gas relationships apply. The ideal
id
gross heating value per unit mass for a mixture, H , is:
m
4. Summary of Practice
n n
4.1 The ideal gas heating value and ideal gas relative
id id
H 5 x M H / x M (2)
m ( j j m,j ( j j
density at base conditions (14.696 psia and 60°F (5.6°C)) are j51 j51
calculatedfromthemolarcompositionandtherespectiveideal
where:x isthemolefractionofComponentj,M isthemolar
j j
gas values for the components; these values are then adjusted
mass of Component j from Table 1, and n is the total number
by means of a calculated compressibility factor.
of components.
id
7.1.2 H is the pure component, ideal gross heating value
m,j
5. Significance and Use
per unit mass for Component j (at 60°F (15.6°C) in Table 1).
5.1 The heating value is a measure of the suitability of a
id
Values of H are independent of pressure, but they vary with
m
pure gas or a gas mixture for use as a fuel; it indicates the
temperature.
amountofenergythatcanbeobtainedasheatbyburningaunit
7.2 Ideal Gas Density
of gas. For use as heating agents, the relative merits of gases
id
7.2.1 The ideal gas density, ρ , is:
from different sources and having different compositions can
n
be compared readily on the basis of their heating values.
id
ρ 5 ~P/RT! x M 5 MP/RT (3)
( j j
Therefore, the heating value is used as a parameter for
j51
determining the price of gas in custody transfer. It is also an
where: M is the molar mass of the mixture,
essential factor in calculating the efficiencies of energy con-
n
version devices such as gas-fired turbines. The heating values
M 5 x M (4)
( j j
of a gas depend not only upon the temperature and pressure,
j51
but also upon the degree of saturation with water vapor.
P is the base pressure in absolute units (psia), R is the gas
However, some calorimetric methods for measuring heating
constant, 10.7316 psia.ft /(lb mol•°R) in this practice, based
valuesarebaseduponthegasbeingsaturatedwithwateratthe
upon R = 8.31448 J/(mol•K), T is the base temperature in
specified conditions.
absolute units (°R = °F + 459.67). Values of the ideal gas
5.2 Therelativedensity(specificgravity)ofagasquantifies
density at 60°F (15.6°C) and 14.696 psia are in GPAStandard
the density of the gas as compared with that of air under the
2145.
same conditions.
7.3 Ideal Relative Density:
id
6. Methods of Analysis 7.3.1 The ideal relative density d is:
n
6.1 Determine the molar composition of the gas in accor-
id
d 5 x d 5 x M /M 5 M/M (5)
( j j ( j j a a
dancewithanyASTMorGPAmethodthatyieldsthecomplete
j51
composition, exclusive of water, but including all other com-
where: M isthemolarmassofair.Theidealrelativedensity
a
ponents present in amounts of 0.1% or more, in terms of
is the molar mass ratio.
componentsorgroupsofcomponentslistedinTable1.Atleast
98%ofthesamplemustbereportedasindividualcomponents
7.4 Gross Heating Value per Unit Volume:
(that is, not more than a total of 2% reported as groups of
7.4.1 Multiplicationofthegrossheatingvalueperunitmass
components such as butanes, pentanes, hexanes, butenes, and
by the ideal gas density provides the gross heating value per
id
so forth).Any group used must be one of those listed in Table
unit volume, H :
v
1 for which average values appear.The following test methods
n
id id id id
are applicable to this practice when appropriate for the sample
H 5 ρ H 5 x H (6)
v m ( j v,j
j51
under test: Test Methods D1717, D1945, D2163, and D2650.
id
H is the pure component gross heating value per unit
v,j
7. Calculation—Ideal Gas Values; Ideal Heating Value
volume for Component j at specified temperature and pressure
7.1 An ideal combustion reaction in general terms for fuel
(60°F (15.6°C) and 14.696 psia in Table 1, ideal gas values).
and air in the ideal gas state is:
7.4.2 Conversion of values in Table 1 to different pressure
C H S id 1 a1b/41c O id 5aCO id 1 h/2 H O idor l
~ ! ~ ! ~ ! ~ ! ~ ! ~ !
bases results from multiplying by the pressure ratio:
a b c 2 2 2
(1)
id id
H P 5 H P 5 14.696 3P/14.696 (7)
~ ! ~ !
v v
1cSO id 7.5 Real Gas Values—Compressibility Factor:
~ !
´1
D3588 − 98 (2017)
A
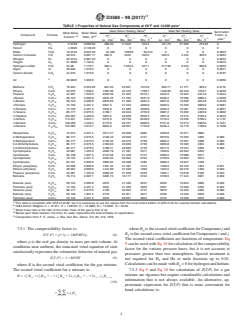
TABLE 1 Properties of Natural Gas Components at 60°F and 14.696 psia
D
Ideal Gross Heating Value Ideal Net Heating Value
Summation
Molar Mass, Molar Mass,
id id id id id id
Compound Formula Factor, b ,
–1B idC i
H , H , H , h , h , h ,
lb·lbmol Ratio, G n m v n m v
−1
–1 –1 –3 –1 –1 –3 psia
kJ · mol Btu · lbm Btu · ft kJ · mol Btu · lbm Btu · ft
Hydrogen H 2.0159 0.069 60 286.20 6 1022 324.2 241.79 51 566 273.93 0
Helium He 4.0026 0.138 20 0 0 0 0 0 0 0
Water H O 18.0153 0.622 02 44.409 1059.8 50.312 0 0 0 0.0623
Carbon monoxide CO 28.010 0.967 11 282.9 4342 320.5 282.9 4 342 320.5 0.0053
Nitrogen N 28.0134 0.967 23 0 0 0 0 0 0 0.0044
Oxygen O 31.9988 1.104 8 0 0 0 0 0 0 0.0073
Hydrogen sulfide H S 34.08 1.176 7 562.4 7 094.2 637.1 517.99 6 534 586.8 0.0253
Argon Ar 39.948 1.379 3 0 0 0 0 0 0 0.0071
Carbon dioxide CO 44.010 1.519 6 0 0 0 0 0 0 0.0197
E
Air 28.9625 1.000 0 0 0 0 0 0 0 0.0050
Methane CH 16.043 0.553 92 891.63 23 891 1010.0 802.71 21 511 909.4 0.0116
Ethane C H 30.070 1.038 2 1562.06 22 333 1769.7 1428.83 20 429 1618.7 0.0239
2 6
Propane C H 44.097 1.522 6 2220.99 21 653 2516.1 2043.3 19 922 2314.9 0.0344
3 8
i-Butane C H 58.123 2.006 8 2870.45 21 232 3251.9 2648.4 19 590 3000.4 0.0458
4 10
n-Butane C H 58.123 2.006 8 2879.63 21 300 3262.3 2657.6 19 658 3010.8 0.0478
4 10
i-Pentane C H 72.150 2.491 2 3531.5 21 043 4000.9 3265.0 19 456 3699.0 0.0581
5 12
n-Pentane C H 72.150 2.491 2 3535.8 21 085 4008.9 3269.3 19 481 3703.9 0.0631
5 12
n-Hexane C H 86.177 2.975 5 4198.1 20 943 4755.9 3887.2 19 393 4403.9 0.0802
6 14
n-Heptane C H 100.204 3.459 8 4857.2 20 839 5502.5 4501.9 19 315 5100.3 0.0944
7 16
n-Octane C H 114.231 3.944 1 5515.9 20 759 6248.9 5116.2 19 256 5796.2 0.1137
8 18
n-Nonane C H 128.258 4.428 4 6175.9 20 701 6996.5 5731.8 19 213 6493.6 0.1331
9 20
n-Decane C H 142.285 4.912 7 6834.9 20 651 7742.9 6346.4 19 176 7189.9 0.1538
10 22
Neopentane C H 72.015 2.491 2 3517.27 20 958 3985 3250.8 19 371 3683
5 12
2-Methylpentane C H 86.177 2.975 5 4190.43 20 905 4747 3879.6 19 355 4395 0.080
6 14
3-Methylpentane C H 86.177 2.975 5 4193.03 20 918 4750 3882.2 19 367 4398 0.080
6 14
2,2-Dimethylbutane C H 86.177 2.975 5 4180.63 20 856 4736 3869.8 19 306 4384 0.080
6 14
2,3-Dimethylbutane C H 86.177 2.975 5 4188.41 20 895 4745 3877.5 19 344 4393 0.080
6 14
Cyclopropane C H 42.081 1.452 9 2092.78 21 381 2371 1959.6 20 020 2220 . . .
3 6
Cyclobutane C H 56.108 1.937 3 2747.08 21 049 2747 2569.4 19 688 2911 . . .
4 8
Cyclopentane C H 70.134 2.421 5 3322.04 20 364 3764 3100.0 19 003 3512 . . .
5 10
Cyclohexane C H 84.161 2.905 9 3955.84 20 208 4482 3689.4 18 847 4180 . . .
6 12
Ethyne (acetylene) C H 26.038 0.899 0 1301.32 21 487 1474 1256.9 20 753 1424 0.021
2 2
Ethene (ethylene) C H 28.054 0.968 6 1412.06 21 640 1600 1323.2 20 278 1499 0.020
2 4
Propene (propylene) C H 42.081 1.452 9 2059.35 21 039 2333 1926.1 19 678 2182 0.033
3 6
Benzene C H 78.114 2.697 1 3202.74 18 177 3742 3169.5 17 444 3591 0.069
6 6
Butanes (ave) C H 58.123 2.006 8 2875 21 266 3257 2653 19 623 3006 0.046
4 10
Pentanes (ave) C H 72.150 2.491 2 3534 21 056 4003 3267 19 469 3702 0.062
5 12
Hexanes (ave) C H 86.177 2.975 5 4190 20 904 4747 3879 19 353 4395 0.080
6 14
Butenes (ave) C H 56.108 1.937 2 2716 20 811 3077 2538 19 450 2876 0.046
4 8
Pentenes (ave) C H 70.134 2.421 5 3375 20 691 3824 3153 19 328 3572 0.060
5 10
A
This table is consistent with GPA 2145-89, but it is necessary to use the values from the most recent edition of GPA 2145 for custody transfer calculations.
B
1984 Atomic Weights: C = 12.011, H = 1.00794, O = 15.9994, N = 14.0067, S = 32.06.
C
Molar mass ratio is the ratio of the molar mass of the gas to that of air.
D
Based upon ideal reaction; the entry for water represents the total enthalpy of vaporization.
E
Composition from: F. E. Jones, J. Res. Nat. Bur. Stand., Vol. 83, 419, 1978.
7.5.1 The compressibility factor is: where B isthesecondvirialcoefficientforComponent jand
jj
B isthesecondcrossvirialcoefficientforComponents iand j.
id
ij
Z~T,P! 5 ρ /ρ 5 ~MP/RT!/ρ (8)
The second virial coefficients are functions of temperature. Eq
where ρ is the real gas density in mass per unit volume. At
9 can be used with Eq 10 for calculation of the compressibility
conditions near ambient, the truncated virial equation of state
factor for the various pressure bases, but it is not accurate at
satisfactorilyrepresentsthevolumetricbehaviorofnaturalgas:
pressures greater than two atmospheres. Special treatment is
Z~T,P! 5 11BP/RT (9)
not required for H and He at mole fractions up to 0.01.
CalculationscanbemadewithB =0forhydrogenandhelium.
jj
where B is the second virial coefficient for the gas mixture.
7.5.2 Eq 9 and Eq 10 for calculation of Z(T,P) for a gas
The second virial coefficient for a mixture is:
mixture are rigorous but require considerable calculations and
2 2 2
B 5 x B 1x B 1···1x B 12x x B 1···12x x B
1 11 2 22 n nn 1 2 12 n21 n n2i,n
information that is not always available. An alternative, ap-
(10)
proximate expression for Z(T,P) that is more convenient for
n n
hand calculations is:
5 x x B
( ( i j ij
i51j51
´1
D3588 − 98 (2017)
n 2
The mole fraction of water can range from zero up to the
Z T,P 5 1 2 P x =β (11)
~ ! F G
( j jj
saturated value. The saturated value for x is, assuming
j51 w
Raoult’s Law:
where β =B /RT and =β is the summation factor for
jj jj jj
σ
x sat 5 P /P (15)
~ !
w w
Component j.Values of =β at 60°F (15.6°C) appear in Table
jj
σ
2. The method based upon Eq 11 has been adopted for this where: P is the vapor pressure of water (0.25636 psia at
w
practice. 60°F (15.6°C)).
7.6 Real Gas Density:
7.9.2 Technically, water has a gross heating value, the ideal
7.6.1 The real gas density ρ at a specific temperature and
enthalpy of condensation. If only the water that is formed
pressure is:
during the combustion condenses, then the heat released upon
id combustion of a wet gas with dry air becomes:
ρ 5 ρ /Z (12)
id id
id H wetgas 5 1 2 x H drygas (16)
~ ! ~ ! ~ !
v w v
where: ρ and Z are evaluated at the same temperature and
pressure.
Forwater-saturatedgas, x at60°F(15.6°C)is0.25636⁄P
w b
where P is the base pressure. Eq 16 is adequate for custody
7.7 Real Relative Density: b
transfer applications as a matter of definition. H
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...